Effects of Long-Term Walnut Supplementation on Body Weight in Free-Living Elderly: Results of a Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Intervention
2.3. Assessment of Diet
2.4. Anthropometry, Body Composition, and Physical Activity
2.5. Biomarker Analyses
2.6. Statistical Analyses
3. Results
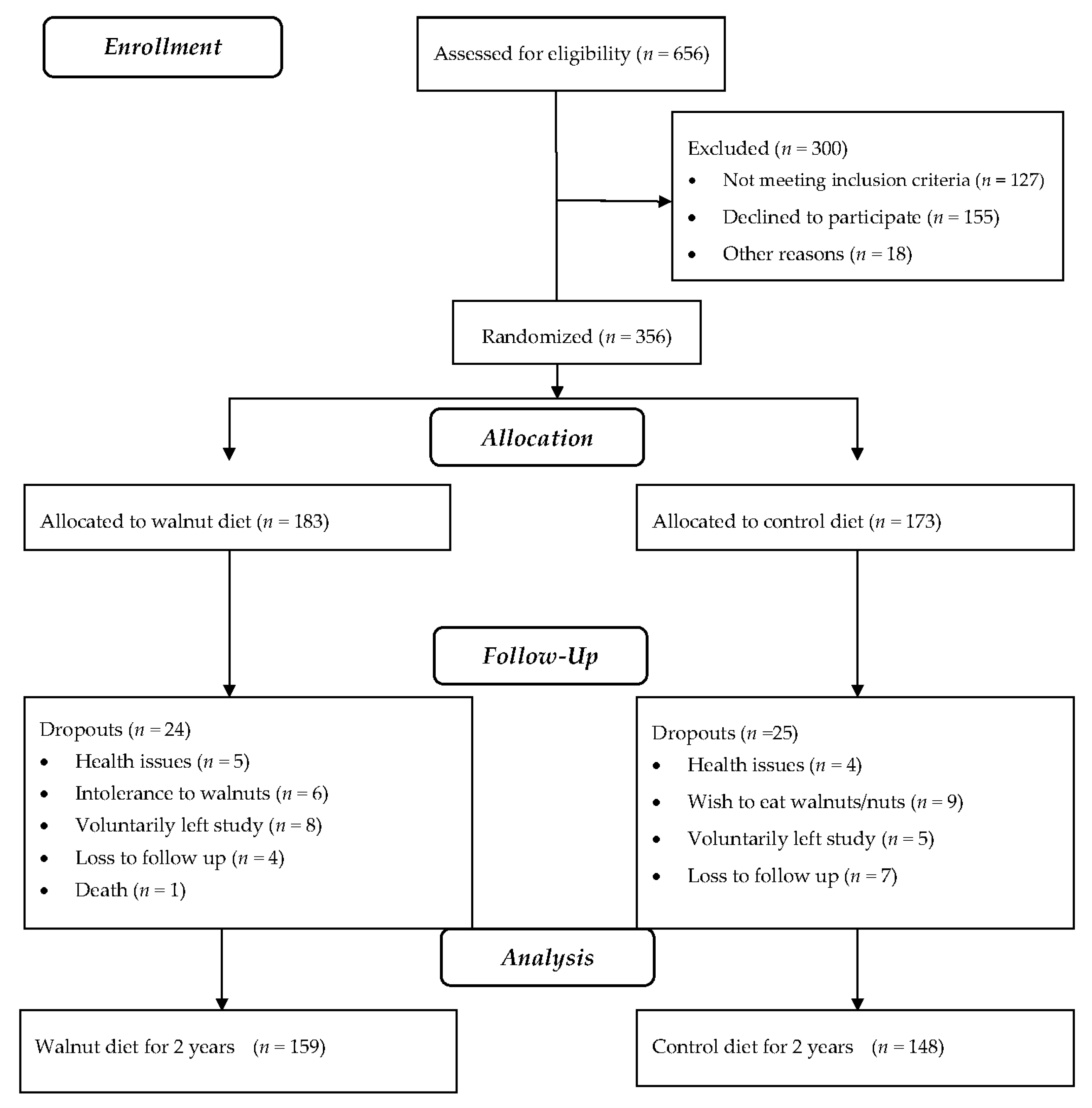
3.1. Participants
3.2. Compliance with Treatment
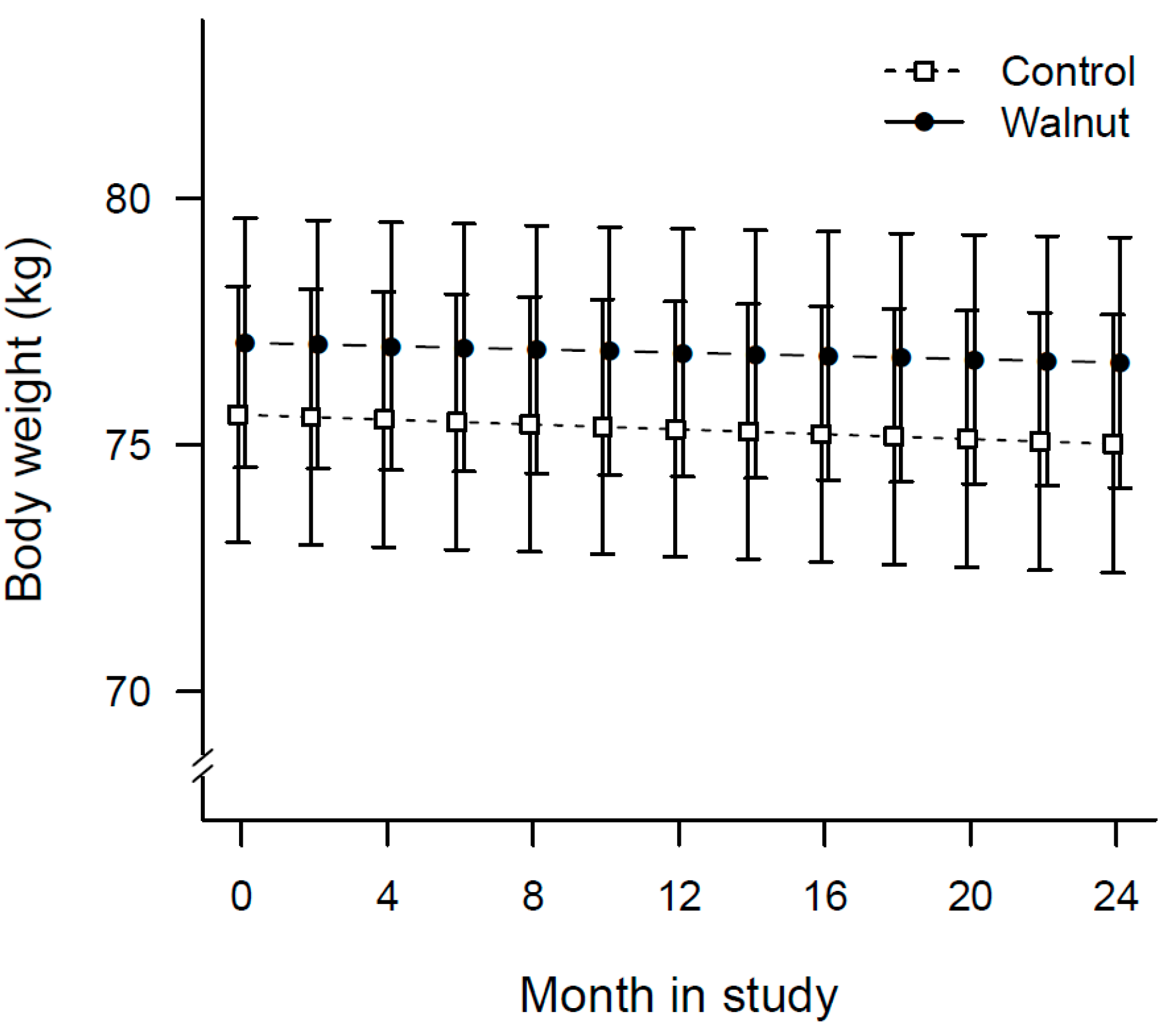
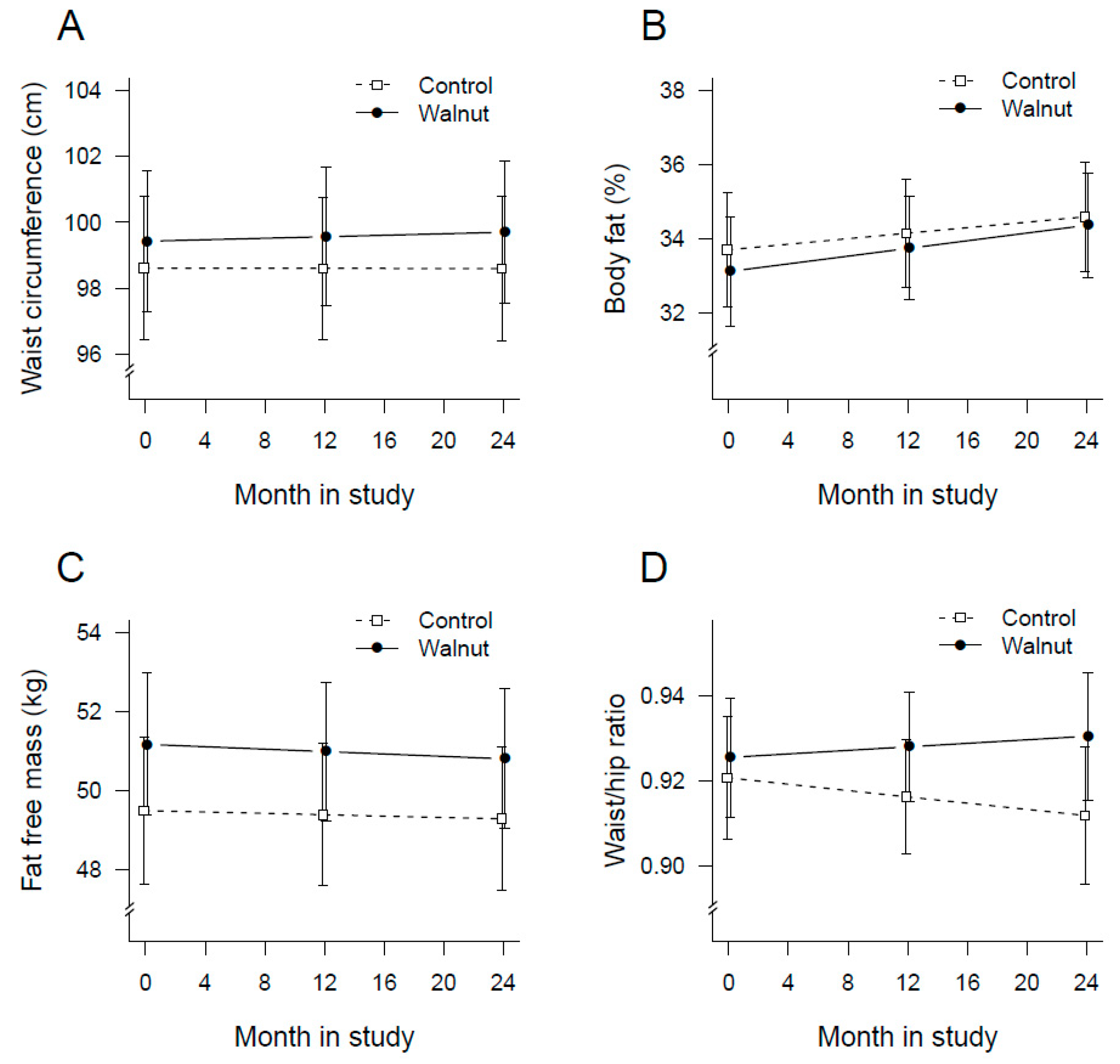
3.3. Changes in Physical Activity and Anthropometric Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ogden, C.L.; Carroll, M.D.; Fryar, C.D.; Flegal, K.M. Prevalence of Obesity among Adults and Youth: United States, 2011–2014; US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics: Washington, DC, USA, 2015.
- Hales, C.M.; Carroll, M.D.; Fryar, C.D.; Ogden, C.L. Prevalence of Obesity among Adults and Youth: United States, 2015–2016; US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics: Washington, DC, USA, 2017.
- King, D.E.; Matheson, E.; Chirina, S.; Shankar, A.; Broman-Fulks, J. The status of baby boomers’ health in the United States: The healthiest generation? JAMA Intern. Med. 2013, 173, 385–386. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 207. [Google Scholar] [CrossRef] [PubMed]
- Villareal, D.T.; Chode, S.; Parimi, N.; Sinacore, D.R.; Hilton, T.; Armamento-Villareal, R.; Napoli, N.; Qualls, C.; Shah, K. Weight loss, exercise, or both and physical function in obese older adults. N. Engl. J. Med. 2011, 364, 1218–1229. [Google Scholar] [CrossRef] [PubMed]
- Ros, E. Nuts and CVD. Br. J. Nutr. 2015, 113, S111–S120. [Google Scholar] [CrossRef] [PubMed]
- Ros, E.; Mataix, J. Fatty acid composition of nuts–implications for cardiovascular health. Br. J. Nutr. 2006, 96, S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Ros, E. Health benefits of nut consumption. Nutrients 2010, 2, 652–682. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.E. Qualified Health Claims: Letter of Enforcement Discretion-Walnuts and Coronary Heart Disease (Docket No 02P-0292); US Food and Drug Administration: Silver Spring, MD, USA, 2004.
- Krauss, R.M.; Eckel, R.H.; Howard, B.; Appel, L.J.; Daniels, S.R.; Deckelbaum, R.J.; Erdman, J.W., Jr.; Kris-Etherton, P.; Goldberg, I.J.; Kotchen, T.A.; et al. AHA dietary guidelines revision 2000: A statement for healthcare professionals from the Nutrition Committee of the American Heart Association. Circulation 2000, 102, 2284–2299. [Google Scholar] [CrossRef] [PubMed]
- Vannice, G.; Rasmussen, H. Position of the academy of nutrition and dietetics: Dietary fatty acids for healthy adults. J. Acad. Nutr. Diet. 2014, 114, 136–153. [Google Scholar] [CrossRef] [PubMed]
- Flores-Mateo, G.; Rojas-Rueda, D.; Basora, J.; Ros, E.; Salas-Salvadó, J. Nut intake and adiposity: Meta-analysis of clinical trials. Am. J. Clin. Nutr. 2013, 97, 1346–1355. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D.; Kris-Etherton, P.M.; Foster, G.D. Impact of peanuts and tree nuts on body weight and healthy weight loss in adults. J. Nutr. 2008, 138, 1741S–1745S. [Google Scholar] [CrossRef] [PubMed]
- Rajaram, S.; Valls-Pedret, C.; Cofán, M.; Sabaté, J.; Serra-Mir, M.; Pérez-Heras, A.M.; Arechiga, A.; Casaroli-Marano, R.P.; Alforja, S.; Sala-Vila, A.; Doménech, M. The Walnuts and Healthy Aging Study (WAHA): Protocol for a Nutritional Intervention Trial with Walnuts on Brain Aging. Front. Aging Neurosci. 2017, 8, 333. [Google Scholar] [CrossRef] [PubMed]
- Volp, A.C.P.; Oliveira, F.C.E.; Alves, R.D.M.; Esteves, E.; Bressan, J. Energy expenditure: Components and evaluation methods. Nutr. Hosp. 2011, 26, 430–440. [Google Scholar]
- Nutrition Coordinating Center. Nutrition Data System for Research; University of Minnesota: Minneapolis, MN, USA, 2011. [Google Scholar]
- Fraser, G.E.; Bennett, H.W.; Jaceldo, K.B.; Sabaté, J. Effect on body weight of a free 76 kilojoule (320 calorie) daily supplement of almonds for six months. J. Am. Coll. Nutr. 2002, 21, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Sabate, J.; Fraser, G.E.; Burke, K.; Knutsen, S.F.; Bennett, H.; Lindsted, K.D. Effects of walnuts on serum lipid levels and blood pressure in normal men. N. Engl. J. Med. 1993, 328, 603–607. [Google Scholar] [CrossRef] [PubMed]
- National Center for Health Statistics. Anthropometry Procedures Manual—National Health and Nutrition Examination Survey (NHANES); NCHS: Hyattsville, MD, USA, 2013.
- Haskell, W.L.; Lee, I.M.; Pate, R.R.; Powell, K.E.; Blair, S.N.; Franklin, B.A.; Macera, C.A.; Heath, G.W.; Thompson, P.D.; Bauman, A. Physical activity and public health: Updated recommendation for adults from the American College of Sports Medicine and the American Heart Association. Circulation 2007, 116, 1081. [Google Scholar] [CrossRef] [PubMed]
- Sala-Vila, A.; Harris, W.S.; Cofán, M.; Pérez-Heras, A.M.; Pintó, X.; Lamuela-Raventós, R.M.; Covas, M.I.; Estruch, R.; Ros, E. Determinants of the omega-3 index in a Mediterranean population at increased risk for CHD. Br. J. Nutr. 2011, 106, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Bitok, E.; Jaceldo-Siegl, K.; Rajaram, S.; Serra-Mir, M.; Roth, I.; Feitas-Simoes, T.; Ros, E.; Sabaté, J. Favourable nutrient intake and displacement with long-term walnut supplementation among elderly: Results of a randomised trial. Br. J. Nutr. 2017, 118, 201. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.; Ruts, E.; Visser, M.; Heshka, S.; Baumgartner, R.N.; Wang, J.; Pierson, R.N.; Pi-Sunyer, F.X.; Heymsfield, S.B. Weight stability masks sarcopenia in elderly men and women. Am. J. Physiol.-Endocrinol. Metab. 2000, 279, E366–E375. [Google Scholar] [CrossRef] [PubMed]
- Forbes, G.B. Longitudinal changes in adult fat-free mass: Influence of body weight. Am. J. Clin. Nutr. 1999, 70, 1025–1031. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, M.; Zoico, E.; Scartezzini, T.; Mazzali, G.; Tosoni, P.; Zivelonghi, A.; Gallagher, D.; De Pergola, G.; Di Francesco, V.; Bosello, O. Body composition changes in stable-weight elderly subjects: The effect of sex. Aging Clin. Exp. Res. 2003, 15, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Cordero-MacIntyre, Z.; Siapco, G.; Torabian, S.; Haddad, E. Does regular walnut consumption lead to weight gain? Br. J. Nutr. 2005, 94, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Kirkmeyer, S.; Mattes, R.D. Effects of food attributes on hunger and food intake. International journal of obesity and related metabolic disorders. J. Int. Assoc. Study Obes. 2000, 24, 1167–1175. [Google Scholar] [CrossRef]
- Mattes, R.D.; Dreher, M.L. Nuts and healthy body weight maintenance mechanisms. Asia Pac. J. Clin. Nutr. 2010, 19, 137–141. [Google Scholar] [PubMed]
- Alper, C.; Mattes, R.D. Effects of chronic peanut consumption on energy balance and hedonics. International journal of obesity and related metabolic disorders. J. Int. Assoc. Study Obes. 2002, 26, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Hollis, J.; Mattes, R.D. Effect of chronic consumption of almonds on body weight in healthy humans. Br. J. Nutr. 2007, 98, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.S.; Silvis, S.E. Absorption of whole peanuts, peanut oil, and peanut butter. N. Engl. J. Med. 1980, 303, 917–918. [Google Scholar] [CrossRef] [PubMed]
- Haddad, E.; Sabate, J. (Eds.) Effect of pecan consumption on stool fat. FASEB J. 2000, 14, A294. [Google Scholar]
- Kris-Etherton, P.M.; Hu, F.B.; Ros, E.; Sabaté, J. The role of tree nuts and peanuts in the prevention of coronary heart disease: Multiple potential mechanisms. J. Nutr. 2008, 138, 1746S–1751S. [Google Scholar] [CrossRef] [PubMed]
- Zemaitis, J.; Sabaté, J. (Eds.) Effect of almond consumption on stool weight and stool fat. FASEB J. 2001, 15, A602. [Google Scholar]
- Ellis, P.R.; Kendall, C.W.; Ren, Y.; Parker, C.; Pacy, J.F.; Waldron, K.W.; Jenkins, D.J. Role of cell walls in the bioaccessibility of lipids in almond seeds. Am. J. Clin. Nutr. 2004, 80, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.; Gebauer, S.; Novotny, J. Atwater factors overestimate the calorie content of walnuts (371.1). FASEB J. 2014, 28 (Suppl. 1), 371. [Google Scholar]
| Variable | Walnut | Control | p-Value | |
|---|---|---|---|---|
| n (%) | 183 (51.4) | 173 (48.6) | -- | |
| Age—year. (mean ± SD) | 69.7 (4.1) | 69.1 (3.7) | 0.137 a | |
| Sex—no. (%) | Women | 119 (65.0) | 118 (68.2) | 0.525 b |
| Men | 64 (35.0) | 55 (31.8) | ||
| Ethnicity—no. (%) | White | 144 (78.7) | 131 (75.7) | 0.221 a |
| Non-white | 39 (21.3) | 42 (24.3) | ||
| Height—cm | 167.2 (9.8) | 165.9 (8.8) | 0.176 a | |
| Weight—kg | 77.1 (17.2) | 75.6 (16.1) | 0.348 a | |
| Body mass index (BMI)—kg/m2 | 27.5 (4.8) | 27.4 (4.8) | 0.833 a | |
| Waist circumference—cm | 99.2 (14.1) | 98.4 (13.4) | 0.615 a | |
| Smoking—no. (%) | Never | 174 (95.1) | 169 (97.7) | 0.503 c |
| Former | 7 (3.8) | 3 (1.7) | ||
| Current | 2 (1.1) | 1 (0.6) | ||
| Physical activity—METs-h/week | 3.54 (3.5) | 3.70 (3.4) | 0.840 a | |
| Variable | Walnut (n = 159) | Control (n = 148) | p-Value d |
|---|---|---|---|
| Mean * (SD) | Mean * (SD) | ||
| Energy (kcal) | 1821 (503) | 1593 (423) | <0.0001 |
| Total carbohydrate (g) | 204 (76) | 192 (64) | 0.199 |
| Total protein (g) | 70 (18) | 65 (19) | 0.011 |
| Vegetable protein (g) | 30 (11) | 24 (11) | <0.0001 |
| Total fat (g) | 84 (24) | 63 (20) | <0.0001 |
| Saturated fat (g) | 22 (9) | 21 (9) | 0.185 |
| Monounsaturated fat (g) | 25 (8) | 22 (7) | 0.001 |
| Polyunsaturated fat (g) | 31 (8) | 14 (5) | <0.0001 |
| Dietary cholesterol (mg) | 202 (102) | 218 (114) | 0.308 |
| Total dietary fiber (g) | 24 (10) | 20 (8) | <0.0001 |
| Total carbohydrate (% E) | 42.8 (10.2) | 47.3 (11.4) | <0.0001 |
| Total protein (% E) | 15.5 (5) | 16.6 (5.6) | 0.01 |
| Total fat (% E) | 40.2 (8.7) | 33.6 (9.7) | <0.0001 |
| Saturated fat (% E) | 10 (3.9) | 11 (4.9) | 0.01 |
| Monounsaturated fat (% E) | 11.8 (3.7) | 11.9 (4.4) | 0.662 |
| Polyunsaturated fat (% E) | 15.1 (4.7) | 7.8 (3.7) | <0.0001 |
| Variable | Timepoint | Walnut (n = 159) | Control (n = 148) | p-Value e | |
|---|---|---|---|---|---|
| Mean (95% CI) | Mean (95% CI) | Time Effect | Group × Time Interaction Effect | ||
| Weight—kg | Baseline | 77.1 (74.5, 79.6) | 75.6 (73.0, 78.2) | 0.031 | 0.671 |
| Year 1 | 76.9 (74.4, 79.4) | 75.3 (72.7, 77.9) | |||
| Year 2 | 76.7 (74.1, 79.2) | 75.0 (72.4, 77.6) | |||
| Body fat—kg | Baseline | 25.5 (24.4, 26.7) | 25.5 (24.3, 26.2) | 0.0001 | 0.528 |
| Year 1 | 25.9 (24.9, 27.0) | 25.7 (24.6, 26.8) | |||
| Year 2 | 26.4 (25.3, 27.4) | 26.0 (24.8, 27.1) | |||
| Lean body mass—kg | Baseline | 51.2 (49.4, 53.0) | 49.5 (47.6, 51.3) | 0.220 | 0.740 |
| Year 1 | 51.0 (49.2, 52.7) | 49.4 (47.6, 51.2) | |||
| Year 2 | 50.8 (49.0, 52.6) | 49.3 (47.4, 51.1) | |||
| Waist circumference—cm | Baseline | 99.4 (97.3, 101.6) | 98.6 (96.4, 100.8) | 0.680 | 0.651 |
| Year 1 | 99.6 (97.5, 101.7) | 98.6 (96.5, 100.8) | |||
| Year 2 | 99.7 (97.6, 101.8) | 98.6 (96.4, 100.8) | |||
| Waist-to-hip ratio | Baseline | 0.93 (0.91, 0.94) | 0.92 (0.91, 0.94) | 0.697 | 0.160 |
| Year 1 | 0.93 (0.92, 0.94) | 0.92 (0.90, 0.93) | |||
| Year 2 | 0.93 (0.92, 0.95) | 0.91 (0.90, 0.93) | |||
| PA-METS—h/week | Baseline | 3.54 (3.06, 4.02) | 3.70 (3.21, 4.19) | <0.001 | 0.841 |
| Year 1 | 3.83 (3.40, 4.25) | 4.02 (3.58, 4.46) | |||
| Year 2 | 4.11 (3.62, 4.61) | 4.34 (3.83, 4.85) | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bitok, E.; Rajaram, S.; Jaceldo-Siegl, K.; Oda, K.; Sala-Vila, A.; Serra-Mir, M.; Ros, E.; Sabaté, J. Effects of Long-Term Walnut Supplementation on Body Weight in Free-Living Elderly: Results of a Randomized Controlled Trial. Nutrients 2018, 10, 1317. https://doi.org/10.3390/nu10091317
Bitok E, Rajaram S, Jaceldo-Siegl K, Oda K, Sala-Vila A, Serra-Mir M, Ros E, Sabaté J. Effects of Long-Term Walnut Supplementation on Body Weight in Free-Living Elderly: Results of a Randomized Controlled Trial. Nutrients. 2018; 10(9):1317. https://doi.org/10.3390/nu10091317
Chicago/Turabian StyleBitok, Edward, Sujatha Rajaram, Karen Jaceldo-Siegl, Keiji Oda, Aleix Sala-Vila, Mercè Serra-Mir, Emilio Ros, and Joan Sabaté. 2018. "Effects of Long-Term Walnut Supplementation on Body Weight in Free-Living Elderly: Results of a Randomized Controlled Trial" Nutrients 10, no. 9: 1317. https://doi.org/10.3390/nu10091317
APA StyleBitok, E., Rajaram, S., Jaceldo-Siegl, K., Oda, K., Sala-Vila, A., Serra-Mir, M., Ros, E., & Sabaté, J. (2018). Effects of Long-Term Walnut Supplementation on Body Weight in Free-Living Elderly: Results of a Randomized Controlled Trial. Nutrients, 10(9), 1317. https://doi.org/10.3390/nu10091317







