Supplement Use and Dietary Sources of Folate, Vitamin D, and n-3 Fatty Acids during Preconception: The GLIMP2 Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Dietary Assessment
2.3. Biochemical Analyses
2.4. Covariates
2.5. Data Analyses
3. Results
3.1. Participant Characteristics
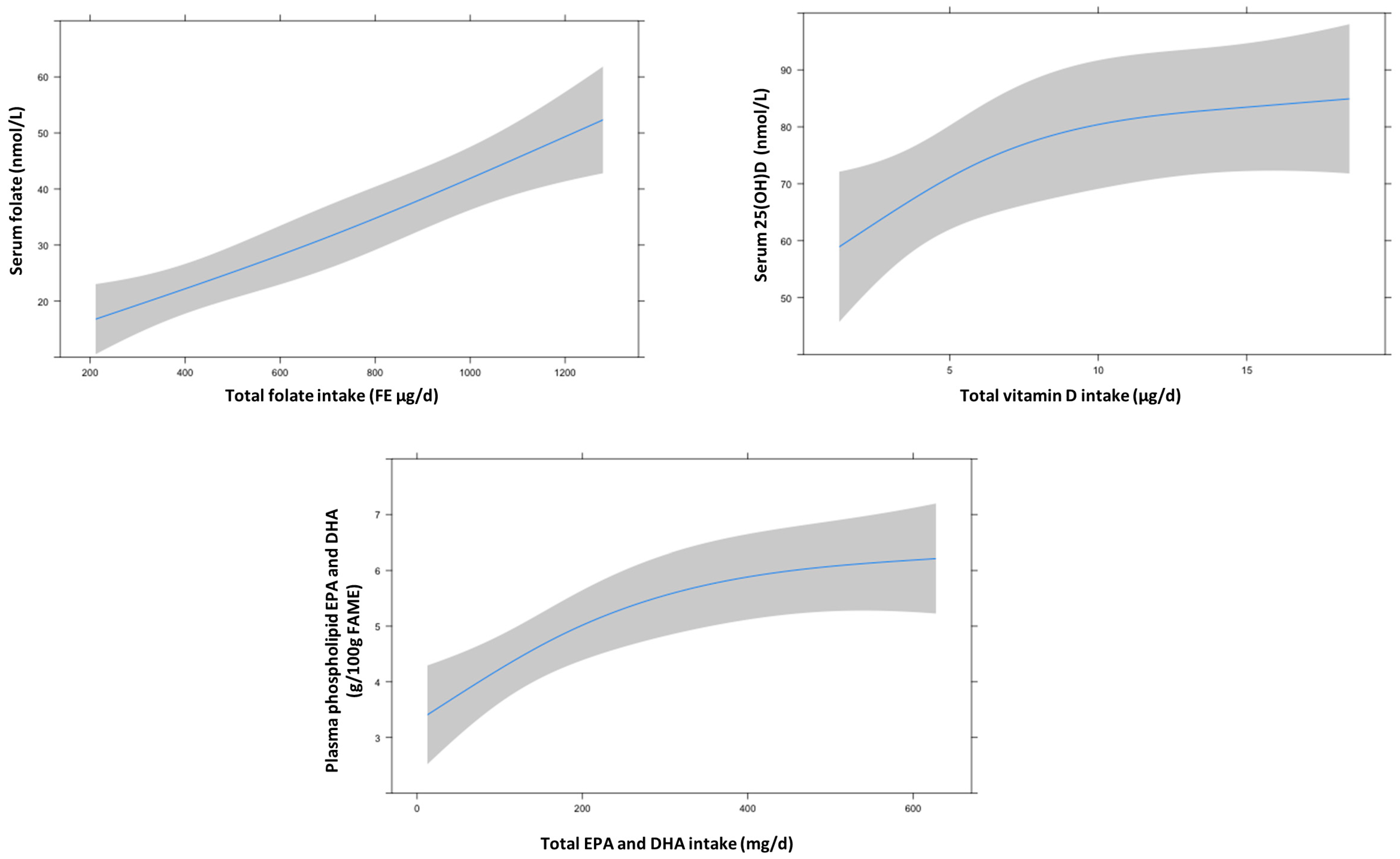
3.2. Folate Intake and Status
3.3. Vitamin D Intake and Status
3.4. EPA and DHA Intake and Status
4. Discussion
4.1. Folate Intake
4.2. Vitamin D Intake
4.3. EPA and DHA Intake
4.4. Supplement Use
4.5. Study Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abu-Saad, K.; Fraser, D. Maternal nutrition and birth outcomes. Epidemiol. Rev. 2010, 32, 5–25. [Google Scholar] [CrossRef] [PubMed]
- Emmett, P.M.; Jones, L.R.; Golding, J. Pregnancy diet and associated outcomes in the Avon longitudinal study of parents and children. Nutr. Rev. 2015, 73 (Suppl. 3), 154–174. [Google Scholar] [CrossRef] [PubMed]
- Langley-Evans, S.C. Nutrition in early life and the programming of adult disease: A review. J. Hum. Nutr. Diet. 2015, 28 (Suppl. 1), 1–14. [Google Scholar] [CrossRef] [PubMed]
- Parisi, F.; Laoreti, A.; Cetin, I. Multiple micronutrient needs in pregnancy in industrialized countries. Ann. Nutr. Metab. 2014, 65, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Huijgen, N.A.; van de Kamp, M.E.; Twigt, J.M.; de Vries, J.H.; Eilers, P.H.; Steegers, E.A.; Laven, J.S.; Steegers-Theunissen, R.P. The preconception dietary risk score; a simple tool to assess an inadequate habitual diet for clinical practice. e-SPEN J. 2014, 9, e13–e19. [Google Scholar] [CrossRef]
- Health Council of the Netherlands. Preconception Care: A Good Beginning; Health Council of The Netherlands: The Hague, The Netherlands, 2007. [Google Scholar]
- Van der Meer, I.M.; Karamali, N.S.; Boeke, A.J.P.; Lips, P.; Middelkoop, B.J.; Verhoeven, I.; Wuister, J.D. High prevalence of vitamin d deficiency in pregnant non-western women in the Hague, Netherlands. Am. J. Clin. Nutr. 2006, 84, 350–353. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Fernández-Gaxiola, A.C.; Dowswell, T.; Peña-Rosas, J.P. Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2010, 10. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Catov, J.M.; Simhan, H.N.; Holick, M.F.; Powers, R.W.; Roberts, J.M. Maternal vitamin D deficiency increases the risk of preeclampsia. J. Clin. Endocrinol. Metab. 2007, 92, 3517–3522. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Qiu, C.; Hu, F.B.; David, R.M.; Van Dam, R.M.; Bralley, A.; Williams, M.A. Maternal plasma 25-hydroxyvitamin d concentrations and the risk for gestational diabetes mellitus. PLoS ONE 2008, 3, e3753. [Google Scholar] [CrossRef] [PubMed]
- Morley, R.; Carlin, J.; Pasco, J.; Wark, J.; Ponsonby, A. Maternal 25-hydroxyvitamin D concentration and offspring birth size: Effect modification by infant vdr genotype. Eur. J. Clin. Nutr. 2009, 63, 802–804. [Google Scholar] [CrossRef] [PubMed]
- Javaid, M.; Crozier, S.; Harvey, N.; Gale, C.; Dennison, E.; Boucher, B.; Arden, N.; Godfrey, K.; Cooper, C. Maternal vitamin D status during pregnancy and childhood bone mass at age 9 years: A longitudinal study. Lancet 2006, 367, 36–43. [Google Scholar] [CrossRef]
- Camargo, C.A.; Rifas-Shiman, S.L.; Litonjua, A.A.; Rich-Edwards, J.W.; Weiss, S.T.; Gold, D.R.; Kleinman, K.; Gillman, M.W. Maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age. Am. J. Clin. Nutr. 2007, 85, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Devereux, G.; Litonjua, A.A.; Turner, S.W.; Craig, L.C.; McNeill, G.; Martindale, S.; Helms, P.J.; Seaton, A.; Weiss, S.T. Maternal vitamin D intake during pregnancy and early childhood wheezing. Am. J. Clin. Nutr. 2007, 85, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Brouwer-Brolsma, E.M.; Vaes, A.M.; van der Zwaluw, N.L.; van Wijngaarden, J.P.; Swart, K.M.; Ham, A.C.; van Dijk, S.C.; Enneman, A.W.; Sohl, E.; van Schoor, N.M. Relative importance of summer sun exposure, vitamin D intake, and genes to vitamin D status in dutch older adults: The b-proof study. J. Steroid Biochem. Mol. Biol. 2016, 164, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Health Council of the Netherlands. Evaluation of the Dietary Reference Values for Vitamin D; Health Council of the Netherlands: The Hague, The Netherlands, 2012. [Google Scholar]
- Uauy, R.; Dangour, A.D. Nutrition in brain development and aging: Role of essential fatty acids. Nutr. Rev. 2006, 64, S24–S33. [Google Scholar] [CrossRef] [PubMed]
- Health Council of the Netherlands. Richtlijnen Goede Voeding 2015; Health Council of the Netherlands: The Hague, The Netherlands, 2015. [Google Scholar]
- Netherlands Nutrition Centre. Richtlijnen Schijf van vijf Netherlands; Nutrition Centre: The Hague, The Netherlands, 2016. (In Dutch) [Google Scholar]
- Verkleij-Hagoort, A.C.; de Vries, J.H.; Stegers, M.P.; Lindemans, J.; Ursem, N.T.; Steegers-Theunissen, R.P. Validation of the assessment of folate and vitamin B12 intake in women of reproductive age: The method of triads. Eur. J. Clin. Nutr. 2007, 61, 610–615. [Google Scholar] [CrossRef] [PubMed]
- Siebelink, E.; Geelen, A.; de Vries, J.H. Self-reported energy intake by FFQ compared with actual energy intake to maintain body weight in 516 adults. Br. J. Nutr. 2011, 106, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Streppel, M.T.; de Vries, J.H.; Meijboom, S.; Beekman, M.; de Craen, A.J.; Slagboom, P.E.; Feskens, E.J. Relative validity of the food frequency questionnaire used to assess dietary intake in the Leiden Longevity Study. Nutr. J. 2013, 12, 75. [Google Scholar] [CrossRef] [PubMed]
- Stichting Nederlands Voedingsstoffenbestand. NEVO-Tabel: Nederlands Voedingsstoffenbestand 2006; Nutrition Centre: The Hague, The Netherlands, 2006. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; Institute of Medicine: Washington, DC, USA, 1998. [Google Scholar]
- Health Council of the Netherlands. Towards an Optimal Use of Folic Acid; Health Council of the Netherlands: The Hague, The Netherlands, 2008. [Google Scholar]
- Health Council of the Netherlands. Towards an Adequate Intake of Vitamin D; Publication; Health Council of the Netherlands: The Hague, The Netherlands, 2008. [Google Scholar]
- Glatz, J.; Soffers, A.; Katan, M.B. Fatty acid composition of serum cholesteryl esters and erythrocyte membranes as indicators of linoleic acid intake in man. Am. J. Clin. Nutr. 1989, 49, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Scholtens, S.; Smidt, N.; Swertz, M.A.; Bakker, S.J.; Dotinga, A.; Vonk, J.M.; van Dijk, F.; van Zon, S.K.; Wijmenga, C.; Wolffenbuttel, B.H.; et al. Cohort profile: Lifelines, a three-generation cohort study and biobank. Int. J. Epidemiol. 2015, 44, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- De Walle, H.E.; de Jong-van den Berg, L.T. Ten years after the dutch public health campaign on folic acid: The continuing challenge. Eur. J. Clin. Pharmacol. 2008, 64, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Livock, M.; Anderson, P.J.; Lewis, S.; Bowden, S.; Muggli, E.; Halliday, J. Maternal micronutrient consumption periconceptionally and during pregnancy: A prospective cohort study. Public Health Nutr. 2017, 20, 294–304. [Google Scholar] [CrossRef] [PubMed]
- McKenna, E.; Hure, A.; Perkins, A.; Gresham, E. Dietary supplement use during preconception: The Australian longitudinal study on women’s health. Nutrients 2017, 9, 1119. [Google Scholar] [CrossRef] [PubMed]
- Dubois, L.; Diasparra, M.; Bedard, B.; Colapinto, C.K.; Fontaine-Bisson, B.; Morisset, A.S.; Tremblay, R.E.; Fraser, W.D. Adequacy of nutritional intake from food and supplements in a cohort of pregnant women in Quebec, Canada: The 3D cohort study (Design, Develop, Discover). Am. J. Clin. Nutr. 2017, 106, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Arkkola, T.; Uusitalo, U.; Pietikainen, M.; Metsala, J.; Kronberg-Kippila, C.; Erkkola, M.; Veijola, R.; Knip, M.; Virtanen, S.M.; Ovaskainen, M.L. Dietary intake and use of dietary supplements in relation to demographic variables among pregnant Finnish women. Br. J. Nutr. 2006, 96, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Gomez, M.F.; Field, C.J.; Olstad, D.L.; Loehr, S.; Ramage, S.; McCargar, L.J.; Team, A.P.S. Use of micronutrient supplements among pregnant women in Alberta: Results from the Alberta Pregnancy Outcomes and nutrition (APrON) cohort. Matern. Child Nutr. 2015, 11, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Berti, C.; Fekete, K.; Dullemeijer, C.; Trovato, M.; Souverein, O.W.; Cavelaars, A.; Dhonukshe-Rutten, R.; Massari, M.; Decsi, T.; Van’T Veer, P. Folate intake and markers of folate status in women of reproductive age, pregnant and lactating women: A meta-analysis. J. Nutr. Metab. 2012, 2012, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Van Rossum, C.T.M.; Fransen, H.P.; Verkaik-Kloosterman, J.; Buurma-Rethans, E.J.M.; Ocké, M. Dutch National Food Consumption Survey 2007–2010: Diet of Children and Adults Aged 7 to 69 Years; RIVM Rapport 350050006 2011; National Institue for Public Health and the Environment: Bilthoven, The Netherlands, 2011.
- Brevik, A.; Vollset, S.E.; Tell, G.S.; Refsum, H.; Ueland, P.M.; Loeken, E.B.; Drevon, C.A.; Andersen, L.F. Plasma concentration of folate as a biomarker for the intake of fruit and vegetables: The hordaland homocysteine study. Am. J. Clin. Nutr. 2005, 81, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Blumfield, M.L.; Hure, A.J.; Macdonald-Wicks, L.; Smith, R.; Collins, C.E. A systematic review and meta-analysis of micronutrient intakes during pregnancy in developed countries. Nutr. Rev. 2013, 71, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Vaes, A.; Brouwer-Brolsma, E.; van der Zwaluw, N.; van Wijngaarden, J.; Berendsen, A.; van Schoor, N.; van der Velde, N.; Uitterlinden, A.; Lips, P.; Dhonukshe-Rutten, R. Food sources of vitamin D and their association with 25-hydroxyvitamin D status in dutch older adults. J. Steroid Biochem. Mol. Biol. 2016, 173, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, U.; Gjessing, H.R.; Hirche, F.; Mueller-Belecke, A.; Gudbrandsen, O.A.; Ueland, P.M.; Mellgren, G.; Lauritzen, L.; Lindqvist, H.; Hansen, A.L. Efficacy of fish intake on vitamin D status: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2015, 102, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Lemming, E.W.; Nälsén, C.; Becker, W.; Ridefelt, P.; Mattisson, I.; Lindroos, A.K. Relative validation of the dietary intake of fatty acids among adults in the Swedish national dietary survey using plasma phospholipid fatty acid composition. J. Nutr. Sci. 2015, 4, e25. [Google Scholar] [CrossRef] [PubMed]
- Aronsson, C.A.; Vehik, K.; Yang, J.; Uusitalo, U.; Hay, K.; Joslowski, G.; Riikonen, A.; Ballard, L.; Virtanen, S.M.; Norris, J.M.; et al. Use of dietary supplements in pregnant women in relation to sociodemographic factors—A report from the environmental determinants of diabetes in the young (teddy) study. Public Health Nutr. 2013, 16, 1390–1402. [Google Scholar] [CrossRef] [PubMed]
- Pouchieu, C.; Levy, R.; Faure, C.; Andreeva, V.A.; Galan, P.; Hercberg, S.; Touvier, M. Socioeconomic, lifestyle and dietary factors associated with dietary supplement use during pregnancy. PLoS ONE 2013, 8, e70733. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Who Recommendations on Antenatal Care for a Positive Pregnancy Experience; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Tolppanen, A.; Fraser, A.; Fraser, W.; Lawlor, D. Risk factors for variation in 25-Hydroxyvitamin D3 and D2 concentrations and vitamin D deficiency in children. J. Clin. Endocrinol. Metab. 2012, 4, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Saadatian-Elahi, M.; Slimani, N.; Chajès, V.; Jenab, M.; Goudable, J.; Biessy, C.; Ferrari, P.; Byrnes, G.; Autier, P.; Peeters, P.H. Plasma phospholipid fatty acid profiles and their association with food intakes: Results from a cross-sectional study within the European prospective investigation into cancer and nutrition. Am. J. Clin. Nutr. 2009, 89, 331–346. [Google Scholar] [CrossRef] [PubMed]
- Fekete, K.; Marosvölgyi, T.; Jakobik, V.; Decsi, T. Methods of assessment of n–3 long-chain polyunsaturated fatty acid status in humans: A systematic review. Am. J. Clin. Nutr. 2009, 89, 2070–2084. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Total Population (n = 66) | Folate Intake < 680 FE µg/day (n = 33) | Folate Intake ≥ 680 FE µg/day (n = 33) | Vitamin D Intake < 10 µg/day (n = 44) | Vitamin D Intake ≥ 10 µg/day (n = 22) | EPA+DHA Intake < 200 mg/day (n = 37) | EPA+DHA Intake ≥ 200 mg/day (n = 29) |
|---|---|---|---|---|---|---|---|
| Maternal age (years) | 31.7 ± 4.1 | 31.2 ± 4.7 | 32.1 ± 3.4 | 31.0 ± 4.0 | 33.0 ± 4.0 | 31.7 ± 4.5 | 31.7 ± 3.5 |
| BMI (kg/m2) | 25.2 ± 4.0 | 26.1 ± 4.4 | 24.3 ± 4.2 | 25.3 ± 4.5 | 25.0 ± 4.4 | 24.9 ± 4.6 | 25.6 ± 4.2 |
| Western ethnicity (%) | 63 (95.5%) | 30 (90.9%) | 33 (100%) | 42 (95.5%) | 21 (95.5%) | 34 (91.9%) | 29 (100.0%) |
| Marital status married (%) | 55 (83.3%) | 27 (81.8%) | 28 (84.9%) | 37 (84.1%) | 18 (81.8%) | 32 (86.5%) | 23 (79.3%) |
| Parity, ≥1 child (%) | 60 (90.9%) | 29 (87.9%) | 31 (93.9%) | 39 (88.6%) | 21 (95.5%) | 34 (91.9%) | 26 (89.7%) |
| Educational level (%) 1 | |||||||
| Low | 3 (4.6%) | 2 (6.1%) | 1 (2.6%) | 3 (6.8%) | 0 (0.0%) | 2 (5.4%) | 1 (3.5%) |
| Intermediate | 22 (33.3%) | 12 (36.4%) | 10 (30.3%) | 14 (31.8%) | 8 (36.4%) | 12 (32.4%) | 10 (34.5%) |
| High | 41 (62.1%) | 19 (57.6%) | 22 (66.7%) | 27 (61.4%) | 14 (63.6%) | 23 (62.2%) | 18 (62.7%) |
| Smokers (%) | 8 (12.1%) | 5 (15.2%) | 3 (9.1%) | 6 (13.6%) | 2 (9.1%) | 4 (10.8%) | 4 (13.8%) |
| Alcohol (g/day) | 0.9 (4.1) | 0.5 (4.1) | 0.9 (3.6) | 0.8 (3.8) | 1.0 (4.3) | 0.5 (1.5) | 1.8 (3.9) * |
| Blood sampling between December and April (%) | 10 (15.2%) | 5 (15.2%) | 5 (15.2%) | 7 (15.9%) | 3 (13.6%) | 4 (10.8%) | 6 (20.7%) |
| Energy (kJ) | 8424 (2701) | 8152 (2888) | 8697 (2058) | 8338 (2752) | 8495 (2218) | 8152 (2376) | 8913 (2172) |
| Total carbohydrate (E%) 2 | 45.4 (5.8) | 44.9 (6.6) | 46.0 (6.3) | 45.4 (7.5) | 45.2 (4.6) | 45.7 (6.4) | 44.4 (7.7) |
| Total protein (E%) 2 | 15.7 (2.5) | 15.9 (1.6) | 14.9 (3.2) | 15.4 (2.6) | 15.9 (2.4) | 15.8 (2.9) | 15.6 ± 2.1 |
| Total fat (E%) 2 | 36.2 (5.9) | 36.2 (5.0) | 36.3 (5.4) | 36.2 (6.6) | 36.4 (5.0) | 35.1 (5.3) | 37.1 (6.3) |
| Total folate (FE µg) | 713 (672) | 272 (102) | 943 (124) ** | 323 (657) | 898 (145) ** | 338 (700) | 892 (551) |
| Folate supplement (%) | 37 (56.1%) | 4 (12.1%) | 33 (100%) ** | 18 (40.9%) | 19 (86.4%) ** | 16 (43.2%) | 21 (72.4%) * |
| Total vitamin D (µg) | 5.9 (8.5) | 3.7 (3.0) | 10.3 (7.4) ** | 3.7 (3.2) | 13.0 (2.1) ** | 4.8 (8.9) | 6.4 (7.0) |
| Vitamin D supplement (%) | 30 (45.5%) | 7 (21.2%) | 23 (69.7%) ** | 8 (18.2%) | 22 (100.0%) ** | 15 (40.5%) | 15 (51.7%) |
| Total EPA+DHA (mg) | 170 (200) | 130 (150) | 220 (250) | 180 (160) | 160 (270) | 100 (110) | 310 (240) ** |
| EPA+DHA supplement (%) | 3 (4.6%) | 1 (3.0%) | 2 (6.1%) | 3 (6.8%) | 0 (0.0%) | 0 (0.0%) | 3 (10.3%) * |
| Total vitamin B6 (mg) | 1.9 (1.4) | 1.8 (0.8) | 2.1 (3.4) * | 1.8 (0.7) | 2.9 (4.5) ** | 1.8 (1.6) | 1.9 (1.2) |
| Total vitamin B12 (µg) | 4.6 (3.3) | 4.3 (2.1) | 5.4 (8.0) * | 4.4 (2.3) | 5.6 (9.8) | 4.1 (2.2) | 5.8 (4.7) ** |
| Plasma folate (nmol/L) | 29.4 ± 18.7 | 17.9 ± 9.7 | 40.9 ± 18.6 ** | 26.9 ± 18.4 | 34.4 ± 18.7 | 26.9 ± 15.5 | 32.6 ± 22.0 |
| Insufficient plasma folate (<10 nmol/L) | 9 (13.6%) | 8 (24.2%) | 1 (3.0%) * | 8 (18.2%) | 1 (4.5%) | 4 (10.8%) | 5 (17.2%) |
| Serum 25(OH)D (nmol/L) | 70.6 ± 23.8 | 65.7 ± 26.2 | 75.5 ± 20.5 | 67.6 ± 25.7 | 76.5 ± 18.5 | 68.6 ± 22.9 | 73.1 ± 25.5 |
| Insufficient serum 25(OH)D (<50 nmol/L) | 15 (22.7%) | 10 (30.3%) | 5 (15.2%) | 13 (29.6%) | 2 (9.1%) | 9 (24.3%) | 6 (20.7%) |
| Plasma phospholipid EPA+DHA (g/100 g FAME) | 5.2 ± 1.7 | 5.0 ± 1.6 | 5.3 ± 1.8 | 5.2 ± 1.8 | 5.0 ± 1.5 | 4.6 ± 1.6 | 5.9 ± 1.5 ** |
| Contribution | Correlation | Adjusted Means 4 with 95%CIs | p for Trend | |||||
|---|---|---|---|---|---|---|---|---|
| µg/day 1 | % | R 2 | R 3 | Tertile 1 | Tertile 2 | Tertile 3 | ||
| Total folate intake | 713 (672) | 0.58 ** | 0.55 ** | ≤294 | 295–894 | ≥895 | <0.001 | |
| Plasma folate | 17.6 | 28.6 | 42.0 | |||||
| (10.6–24.7) | (21.7–35.4) | (34.8–49.2) | ||||||
| Folate from supplements | 340 (680) | 0.68 ** | 0.67 ** | ≤0 | 0.1–679 | ≥680 | <0.001 | |
| Plasma folate | 17.6 | 29.7 | 40.7 | |||||
| (12.2–23.0) | (19.1–40.3) | (35.5–46.0) | ||||||
| Total dietary folate | 262 (102) | 100 | −0.08 | −0.20 | ≤223 | 224–293 | ≥294 | 0.257 |
| Plasma folate | 31.4 | 30.2 | 26.5 | |||||
| (25.5–37.3) | (24.5–35.9) | (20.6–32.5) | ||||||
| Folate from vegetables | 63.2 (50.2) | 25 | 0.06 | 0.04 | ≤47.9 | 48.0–83.3 | ≥83.4 | 0.324 |
| Plasma folate | 30.1 | 25.2 | 33.1 | |||||
| (24.6–35.6) | (19.8–30.7) | (27.6–38.6) | ||||||
| Folate from bread and cereal products 5 | 57.5 (39.3) | 22 | −0.10 | −0.19 | ≤46.0 | 46.1–69.5 | ≥69.6 | 0.763 |
| Plasma folate | 29.9 | 29.7 | 28.6 | |||||
| (24.3–35.5) | (24.0–35.4) | (22.8–34.4) | ||||||
| Folate from dairy products 6 | 26.8 (24.0) | 10 | 0.01 | −0.15 | ≤18.7 | 18.8–34.0 | ≥34.1 | 0.355 |
| Plasma folate | 32.6 | 27.3 | 28.5 | |||||
| (26.6–38.6) | (21.8–32.9) | (22.2–34.8) | ||||||
| Folate from fruit | 25.6 (22.4) | 10 | 0.13 | −0.14 | ≤17.6 | 17.7–30.7 | ≥30.8 | 0.502 |
| Plasma folate | 29.5 | 32.0 | 26.5 | |||||
| (23.5–35.5) | (26.5–37.5) | (20.4–32.6) | ||||||
| Folate from oils and fats 7 | 0.27 (3.32) | 5 | −0.23 | −0.20 | ≤0.07 | 0.08–0.63 | ≥0.64 | 0.102 |
| Plasma folate | 32.0 | 30.7 | 25.4 | |||||
| (26.4–37.7) | (25.0–36.4) | (19.7–31.2) | ||||||
| Contribution | Correlation | Adjusted Means 4 with 95%CIs | p for Trend | |||||
|---|---|---|---|---|---|---|---|---|
| µg/day 1 | % | R 2 | R 3 | Tertile 1 | Tertile 2 | Tertile 3 | ||
| Total vitamin D | 5.9 (8.5) | 0.32 ** | 0.42 ** | ≤3.6 | 3.7–10.2 | ≥10.3 | 0.04 | |
| Serum 25(OH)D | 62.5 | 71.2 | 78.0 | |||||
| (52.6–72.5) | (61.5–80.9) | (68.4–87.5) | ||||||
| Vitamin D from supplements | 0 (7.5) | 0.30 * | 0.40 ** | ≤0 | 0.1–4.9 | ≥5.0 | 0.006 | |
| Serum 25(OH)D | 64.5 | 55.6 | 79.4 | |||||
| (57.6–71.5) | (24.8–86.3) | (71.5–87.3) | ||||||
| Total dietary vitamin D | 3.3 (2.0) | 100 | 0.12 | 0.30 * | ≤2.8 | 2.9–3.8 | ≥3.9 | 0.001 |
| Serum 25(OH)D | 57.4 | 71.5 | 82.8 | |||||
| (48.2–66.6) | (62.8–80.2) | (72.9–92.6) | ||||||
| Vitamin D from oils and fats 5 | 1.1 (2.0) | 39 | 0.20 | 0.38 ** | ≤0.6 | 0.7–2.0 | ≥2.1 | 0.02 |
| Serum 25(OH)D | 60.6 | 69.6 | 80.7 | |||||
| (50.2–71.1) | (60.8–78.3) | (70.1–91.3) | ||||||
| Vitamin D from fish | 0.53 (0.76) | 20 | −0.04 | 0.18 | ≤0.2 | 0.3–0.7 | ≥0.8 | 0.18 |
| Serum 25(OH)D | 63.3 | 73.8 | 74.1 | |||||
| (53.5–73.2) | (65–82.6) | (64.8–83.3) | ||||||
| Vitamin D from meat | 0.43 (0.37) | 14 | 0.09 | 0.03 | ≤0.3 | 0.4–0.6 | ≥0.7 | 0.43 |
| Serum 25(OH)D | 69.2 | 69.5 | 73.0 | |||||
| (59.6–78.8) | (59.9–79) | (63.1–83.0) | ||||||
| Vitamin D from egg | 0.23 (0.35) | 10 | −0.14 | −0.07 | ≤0.1 | 0.2–0.3 | ≥0.4 | 0.96 |
| Serum 25(OH)D | 72.0 | 67.5 | 71.8 | |||||
| (62.1–81.9) | (57.4–77.6) | (61.7–81.9) | ||||||
| Vitamin D from dairy products 6 | 0.12 (0.11) | 5 | −0.06 | 0.13 | ≤0.01 | 0.02–0.10 | ≥0.11 | 0.11 |
| Serum 25(OH)D | 74.0 | 63.7 | 73.5 | |||||
| (65.0–83.0) | (54.0–73.4) | (63.8–83.2) | ||||||
| Contribution | Correlation | Adjusted Means4 with 95%CIs | p for Trend | |||||
|---|---|---|---|---|---|---|---|---|
| µg/day 1 | % | R 2 | R 3 | Tertile 1 | Tertile 2 | Tertile 3 | ||
| Total EPA+DHA intake | 170 (200) | 0.63 ** | 0.67 ** | <100 | 100–240 | >240 | 0.002 | |
| PPL g/100 g FAME | 4.1 | 5.3 | 6.0 | |||||
| (3.4–4.8) | (4.6–6.0) | (5.4–6.7) | ||||||
| EPA+DHA from supplements | 0 (0) | 0.24 | 0.38 * | 0 | 1–500 | - a | - a | |
| PPL g/100 g FAME | 5.0 | 7.9 | ||||||
| (4.6–5.4) | (6.1–9.7) | |||||||
| Total dietary EPA+DHA | 165 (190) | 100 | 0.59 ** | 0.63 ** | <100 | 100–230 | >230 | 0.001 |
| PPL g/100 g FAME | 4.2 | 5.5 | 5.9 | |||||
| (3.5–4.8) | (4.8–6.2) | (5.2–6.5) | ||||||
| EPA+DHA from fish | 135 (190) | 69 | 0.60 ** | 0.67 ** | <70 | 70–90 | >190 | <0.001 |
| PPL g/100 g FAME | 3.8 | 5.9 | 6.0 | |||||
| (3.2–4.4) | (5.2–6.5) | (5.4–6.5) | ||||||
| EPA+DHA from fatty fish | 105 (170) | 46 | 0.60 ** | 0.51 ** | <20 | 20–160 | >160 | 0.009 |
| PPL g/100 g FAME | 4.4 | 5.4 | 5.7 | |||||
| (3.7–5) | (4.7–6) | (5–6.4) | ||||||
| EPA+DHA from lean fish | 25 (40) | 18 | 0.22 | 0.10 | <10 | 10–30 | >30 | 0.408 |
| PPL g/100 g FAME | 4.9 | 5.3 | 5.3 | |||||
| (4.2–5.5) | (4.7–6.0) | (4.6–6.0) | ||||||
| EPA+DHA from shell fish | 0 (10) | 5 | 0.32 * | 0.24 | 0 | 1–140 | - a | - a |
| PPL g/100 g FAME | 4.9 | 6.0 | ||||||
| (4.4–5.3) | (5.2–6.8) | |||||||
| EPA+DHA from meat | 10 (10) | 6 | −0.06 | −0.30 * | 0 | 1–30 | - a | - a |
| PPL g/100 g FAME | 5.6 | 4.8 | ||||||
| (5–6.1) | (4.3–5.3) | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Looman, M.; Van den Berg, C.; Geelen, A.; Samlal, R.A.K.; Heijligenberg, R.; Klein Gunnewiek, J.M.T.; Balvers, M.G.J.; Leendertz-Eggen, C.L.; Wijnberger, L.D.E.; Feskens, E.J.M.; et al. Supplement Use and Dietary Sources of Folate, Vitamin D, and n-3 Fatty Acids during Preconception: The GLIMP2 Study. Nutrients 2018, 10, 962. https://doi.org/10.3390/nu10080962
Looman M, Van den Berg C, Geelen A, Samlal RAK, Heijligenberg R, Klein Gunnewiek JMT, Balvers MGJ, Leendertz-Eggen CL, Wijnberger LDE, Feskens EJM, et al. Supplement Use and Dietary Sources of Folate, Vitamin D, and n-3 Fatty Acids during Preconception: The GLIMP2 Study. Nutrients. 2018; 10(8):962. https://doi.org/10.3390/nu10080962
Chicago/Turabian StyleLooman, Moniek, Claudia Van den Berg, Anouk Geelen, Rahul A. K. Samlal, Rik Heijligenberg, Jacqueline M. T. Klein Gunnewiek, Michiel G. J. Balvers, Caroline L. Leendertz-Eggen, Lia D. E. Wijnberger, Edith J. M. Feskens, and et al. 2018. "Supplement Use and Dietary Sources of Folate, Vitamin D, and n-3 Fatty Acids during Preconception: The GLIMP2 Study" Nutrients 10, no. 8: 962. https://doi.org/10.3390/nu10080962
APA StyleLooman, M., Van den Berg, C., Geelen, A., Samlal, R. A. K., Heijligenberg, R., Klein Gunnewiek, J. M. T., Balvers, M. G. J., Leendertz-Eggen, C. L., Wijnberger, L. D. E., Feskens, E. J. M., & Brouwer-Brolsma, E. M. (2018). Supplement Use and Dietary Sources of Folate, Vitamin D, and n-3 Fatty Acids during Preconception: The GLIMP2 Study. Nutrients, 10(8), 962. https://doi.org/10.3390/nu10080962





