Low Serum Carotenoids Are Associated with Self-Reported Cognitive Dysfunction and Inflammatory Markers in Breast Cancer Survivors
Abstract
1. Introduction
2. Materials and Methods
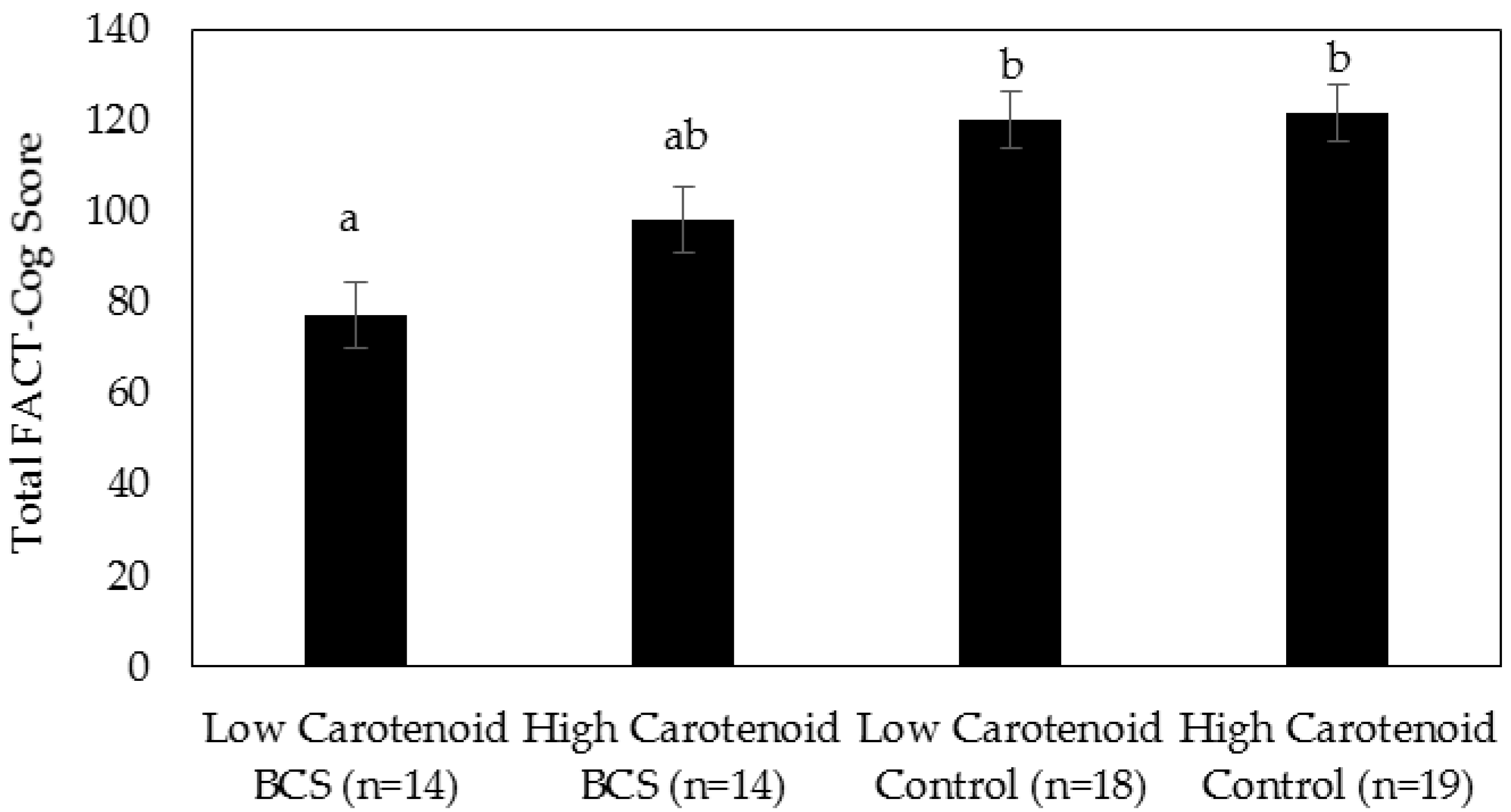
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Howlader, N.; Noone, A.M.; Krapcho, M.; Miller, D.; Bishop, K.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; et al. SEER Cancer Statistics Review (CSR) 1975–2014. Available online: https://seer.cancer.gov/csr/1975_2015/ (accessed on 16 April 2018).
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA. Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef] [PubMed]
- Palesh, O.; Scheiber, C.; Kesler, S.; Mustian, K.; Koopman, C.; Schapira, L. Management of side effects during and post-treatment in breast cancer survivors. Breast J. 2018, 24, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Janelsins, M.C.; Kesler, S.R.; Ahles, T.A.; Morrow, G.R. Prevalence, mechanisms, and management of cancer-related cognitive impairment. Int. Rev. Psychiatry 2014, 26, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Koppelmans, V.; Breteler, M.M.B.; Boogerd, W.; Seynaeve, C.; Gundy, C.; Schagen, S.B. Neuropsychological performance in survivors of breast cancer more than 20 years after adjuvant chemotherapy. J. Clin. Oncol. 2012, 30, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Janelsins, M.C.; Mustian, K.M.; Palesh, O.G.; Mohile, S.G.; Peppone, L.J.; Sprod, L.K.; Heckler, C.E.; Roscoe, J.A.; Katz, A.W.; Williams, J.P.; et al. Differential expression of cytokines in breast cancer patients receiving different chemotherapies: Implications for cognitive impairment research. Support Care Cancer 2012, 20, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.M.; Shah, R.; Shayne, M.; Huston, A.J.; Krebs, M.; Murray, N.; Thompson, B.D.; Doyle, K.; Korotkin, J.; van Wijngaarden, E.; et al. Associations between inflammatory markers and cognitive function in breast cancer patients receiving chemotherapy. J. Neuroimmunol. 2018, 314, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Lyon, D.E.; Cohen, R.; Chen, H.; Kelly, D.L.; McCain, N.L.; Starkweather, A.; Ahn, H.; Sturgill, J.; Jackson-Cook, C.K. Relationship of systemic cytokine concentrations to cognitive function over two years in women with early stage breast cancer. J. Neuroimmunol. 2016, 301, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Cheung, Y.T.; Ng, T.; Shwe, M.; Ho, H.K.; Foo, K.M.; Cham, M.T.; Lee, J.A.; Fan, G.; Tan, Y.P.; Yong, W.S.; et al. Association of proinflammatory cytokines and chemotherapy-associated cognitive impairment in breast cancer patients: A multi-centered, prospective, cohort study. Ann. Oncol. 2015, 26, 1446–1451. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Walitt, B.; Saligan, L.; Tiwari, A.F.; Cheung, C.W.; Zhang, Z.J. Chemobrain: A critical review and causal hypothesis of link between cytokines and epigenetic reprogramming associated with chemotherapy. Cytokine 2015, 72, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Wahl, D.; Cogger, V.C.; Solon-Biet, S.M.; Waern, R.V.; Gokarn, R.; Pulpitel, T.; Cabo, R.; Mattson, M.P.; Raubenheimer, D.; Simpson, S.J.; et al. Nutritional strategies to optimise cognitive function in the aging brain. Ageing Res. Rev. 2016, 31, 80–92. [Google Scholar] [CrossRef] [PubMed]
- Loef, M.; Walach, H. Fruit, vegetables and prevention of cognitive decline or dementia: A systematic review of cohort studies. J. Nutr. Heal. Aging 2012, 16, 626–630. [Google Scholar] [CrossRef]
- Nurk, E.; Refsum, H.; Drevon, C.A.; Tell, G.S.; Nygaard, H.A.; Engedal, K.; Smith, A.D. Cognitive performance among the elderly in relation to the intake of plant foods. The Hordaland Health Study. Br. J. Nutr. 2010, 104, 1190–1201. [Google Scholar] [CrossRef] [PubMed]
- Cheung, B.H.; Ho, I.C.; Chan, R.S.; Sea, M.M.; Woo, J. Current evidence on dietary pattern and cognitive function. Adv. Food Nutr. Res. 2014, 71, 137–163. [Google Scholar] [PubMed]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement. 2015, 11, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Allès, B.; Samieri, C.; Féart, C.; Jutand, M.A.; Laurin, D.; Barberger-Gateau, P. Dietary patterns: A novel approach to examine the link between nutrition and cognitive function in older individuals. Nutr. Res. Rev. 2012, 25, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Shi, Y.; Bao, P.; Cai, H.; Hong, Z.; Ding, D.; Jackson, J.; Shu, X.-O.; Dai, Q. Associations of dietary intake and supplement use with post-therapy cognitive recovery in breast cancer survivors. Breast Cancer Res. Treat. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zuniga, K.E.; Mackenzie, M.J.; Roberts, S.A.; Raine, L.B.; Hillman, C.H.; Kramer, A.F.; McAuley, E. Relationship between fruit and vegetable intake and interference control in breast cancer survivors. Eur. J. Nutr. 2016, 55, 1555–1562. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R., Jr.; Renzi, L.M. Nutrient Information: Carotenoids. Adv. Nutr. 2013, 4, 474–476. [Google Scholar] [CrossRef] [PubMed]
- Akbaraly, N.T.; Faure, H.; Gourlet, V.; Favier, A.; Berr, C. Plasma carotenoid levels and cognitive performance in an elderly population: Results of the EVA study. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Andreeva, V.A.; Ducros, V.; Jeandel, C.; Julia, C.; Hercberg, S.; Galan, P. Carotenoid-rich dietary patterns during midlife and subsequent cognitive function. Br. J. Nutr. 2014, 111, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J. Role of lutein and zeaxanthin in visual and cognitive function throughout the lifespan. Nutr. Rev. 2014, 72, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Mohn, E.S.; Erdman, J.W.; Kuchan, M.J.; Neuringer, M.; Johnson, E.J. Lutein accumulates in subcellular membranes of brain regions in adult rhesus macaques: Relationship to DHA oxidation products. PLoS ONE 2017, 12, e0186767. [Google Scholar] [CrossRef] [PubMed]
- Ruxton, C.H.S.; Derbyshire, E.; Toribio-Mateas, M. Role of fatty acids and micronutrients in healthy ageing: A systematic review of randomised controlled trials set in the context of European dietary surveys of older adults. J. Hum. Nutr. Diet. 2016, 29, 308–324. [Google Scholar] [CrossRef] [PubMed]
- Lindbergh, C.A.; Renzi-Hammond, L.M.; Hammond, B.R.; Terry, D.P.; Mewborn, C.M.; Puente, A.N.; Miller, L.S. Lutein and zeaxanthin influence brain function in older adults: A randomized controlled trial. J. Int. Neuropsychol. Soc. 2018, 24, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R.; Miller, L.S.; Bello, M.O.; Lindbergh, C.A.; Mewborn, C.; Renzi-Hammond, L.M.; Renzi-Hammond, L.M. Effects of lutein/zeaxanthin supplementation on the cognitive function of community dwelling older adults: A randomized, double-masked, placebo-controlled trial. Front. Aging Neurosci. 2017, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Power, R.; Coen, R.F.; Beatty, S.; Mulcahy, R.; Moran, R.; Stack, J.; Howard, A.N.; Nolan, J.M. Supplemental retinal carotenoids enhance memory in healthy individuals with Low levels of macular pigment in a randomized, double-blind, placebo-ontrolled clinical trial. J. Alzheimers Dis. 2018, 61, 947–961. [Google Scholar] [CrossRef] [PubMed]
- Grodstein, F.; Kang, J.H.; Glynn, R.J.; Cook, N.R.; Gaziano, J.M. A randomized trial of beta carotene supplementation and cognitive function in men: The Physicians’ Health Study II. Arch. Intern. Med. 2007, 167, 2184. [Google Scholar] [CrossRef] [PubMed]
- Block, G.; Wakimoto, P.; Jensen, C.; Mandel, S.; Green, R.R. Validation of a food frequency questionnaire for Hispanics. Prev. Chronic Dis. 2006, 3, A77. [Google Scholar] [PubMed]
- Heaton, R.K.; Akshoomoff, N.; Tulsky, D.; Mungas, D.; Weintraub, S.; Dikmen, S.; Beaumont, J.; Casaletto, K.B.; Conway, K.; Slotkin, J.; et al. Reliability and validity of composite scores from the NIH toolbox cognition battery in adults. J. Int. Neuropsychol. Soc. 2014, 20, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Cella, D.F.; Tulsky, D.S.; Gray, G.; Sarafian, B.; Linn, E.; Bonomi, A.; Silberman, M.; Yellen, S.B.; Winicour, P.; Brannon, J.; et al. The Functional Assessment of Cancer Therapy scale: Development and validation of the general measure. J. Clin. Oncol. 1993, 11, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.K.; Engelmann, N.J.; Lila, M.A.; Erdman, J.W. Phytoene, phytofluene, and lycopene from tomato powder differentially accumulate in tissues of male Fisher 344 rats. Nutr. Res. 2007, 27, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Choi, J.H.; Engelmann Moran, N.; Jin, Y.S.; Erdman, J.W. Laboratory-scale production of 13C-labeled lycopene and phytoene by bioengineered Escherichia coli. J. Agric. Food Chem. 2011, 59, 9996–10005. [Google Scholar] [CrossRef] [PubMed]
- Jim, H.S.; Phillips, K.M.; Chait, S.; Faul, L.A.; Popa, M.A.; Lee, Y.H.; Hussin, M.G.; Jacobsen, P.B.; Small, B.J. Meta-analysis of cognitive functioning in breast cancer survivors previously treated with standard-dose chemotherapy. J. Clin. Oncol. 2012, 30, 3578–3587. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Ogilvie, J.M.; Wilson, J.S.; Green, H.J.; Chambers, S.K.; Ownsworth, T.; Shum, D.H.K. A meta-analysis of cognitive impairment and decline associated with adjuvant chemotherapy in women with breast cancer. Front. Oncol. 2015, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, L.J.; McCreath, G.A.; Komeylian, Z.; Rich, J.B. Cognitive impairment in breast cancer survivors treated with chemotherapy depends on control group type and cognitive domains assessed: A multilevel meta-analysis. Neurosci. Biobehav. Rev. 2017, 83, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, A.D.; Hosking, J.R.; Kichenadasse, G.; Mattiske, J.K.; Wilson, C. Objective and subjective cognitive impairment following chemotherapy for cancer: A systematic review. Cancer Treat. Rev. 2012, 38, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Pullens, M.J.; De Vries, J.; Roukema, J.A. Subjective cognitive dysfunction in breast cancer patients: A systematic review. Psychooncology 2010, 19, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Clarnette, R.M.; Almeida, O.P.; Forstl, H.; Paton, A.; Martins, R.N. Clinical characteristics of individuals with subjective memory loss in Western Australia: Results from a cross-sectional survey. Int. J. Geriatr. Psychiatry 2001, 16, 168–174. [Google Scholar] [CrossRef]
- Montejo, P.; Montenegro, M.; Fernandez, M.A.; Maestu, F. Subjective memory complaints in the elderly: Prevalence and influence of temporal orientation, depression and quality of life in a population-based study in the city of Madrid. Aging Ment. Health 2011, 15, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Reid, L.; MacLullich, A.M. Subjective memory complaints and cognitive impairment in older people. Dement. Geriatr. Cogn. Disord. 2006, 22, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; van Belle, G.; Crane, P.K.; Kukull, W.A.; Bowen, J.D.; McCormick, W.C.; Larson, E.B. Subjective memory deterioration and future dementia in people aged 65 and older. J. Am. Geriatr. Soc. 2004, 52, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Saykin, A.J.; Wishart, H.A.; Rabin, L.A.; Santulli, R.B.; Flashman, L.A.; West, J.D.; McHugh, T.L.; Mamourian, A.C. Older adults with cognitive complaints show brain atrophy similar to that of amnestic MCI. Neurology 2006, 67, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Cheung, Y.T.; Foo, Y.L.; Shwe, M.; Tan, Y.P.; Fan, G.; Yong, W.S.; Madhukumar, P.; Ooi, W.S.; Chay, W.Y.; Dent, R.A.; et al. Minimal clinically important difference (MCID) for the functional assessment of cancer therapy: Cognitive function (FACT-Cog) in breast cancer patients. J. Clin. Epidemiol. 2014, 67, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Bray, V.J.; Dhillon, H.M.; Bell, M.L.; Kabourakis, M.; Fiero, M.H.; Yip, D.; Boyle, F.; Price, M.A.; Vardy, J.L. Evaluation of a web-based cognitive rehabilitation program in cancer survivors reporting cognitive symptoms after chemotherapy. J. Clin. Oncol. 2017, 35, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Hartman, S.J.; Nelson, S.H.; Myers, E.; Natarajan, L.; Sears, D.D.; Palmer, B.W.; Weiner, L.S.; Parker, B.A.; Patterson, R.E. Randomized controlled trial of increasing physical activity on objectively measured and self-reported cognitive functioning among breast cancer survivors: The memory and motion study. Cancer 2018, 124, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Derry, H.M.; Jaremka, L.M.; Bennett, J.M.; Peng, J.; Andridge, R.; Shapiro, C.; Malarkey, W.B.; Emery, C.F.; Layman, R.; Mrozek, E.; et al. Yoga and self-reported cognitive problems in breast cancer survivors: A randomized controlled trial. Psychooncology 2015, 24, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Milbury, K.; Chaoul, A.; Biegler, K.; Wangyal, T.; Spelman, A.; Meyers, C.A.; Arun, B.; Palmer, J.L.; Taylor, J.; Cohen, L. Tibetan sound meditation for cognitive dysfunction: Results of a randomized controlled pilot trial. Psychooncology 2013, 22, 2354–2363. [Google Scholar] [CrossRef] [PubMed]
- Lacourt, T.E.; Heijnen, J.C. Mechanisms of neurotoxic symptoms as a result of breast cancer and its treatment: Considerations on the contribution of stress, inflammation, and cellular bioenergetics. Curr. Breast Cancer Rep. 2017, 9, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Ganz, P.A.; Bower, J.E.; Kwan, L.; Castellon, S.A.; Silverman, D.H.; Geist, C.; Breen, E.C.; Irwin, M.R.; Cole, S.W. Does tumor necrosis factor-alpha (TNF-a) play a role in post-chemotherapy cerebral dysfunction? Brain Behav. Immun. 2013, 30. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Green, R.C.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between serum and brain carotenoids, α -tocopherol, and retinol concentrations and cognitive performance in the oldest old from the georgia centenarian study. J. Aging Res. 2013, 2013, 951786. [Google Scholar] [CrossRef] [PubMed]
- Snodderly, D.M. Evidence for protection against age-related macular degeneration by carotenoids and antioxidant vitamins. Am. J. Clin. Nutr. 1995, 62, 1448S–1461S. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jungsuwadee, P.; Vore, M.; Butterfield, D.A.; St Clair, D.K. Collateral damage in cancer chemotherapy: Oxidative stress in nontargeted tissues. Mol. Interv. 2007, 7, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Vera-Ramirez, L.; Ramirez-Tortosa, Mc.; Perez-Lopez, P.; Granados-Principal, S.; Battino, M.; Quiles, J.L. Long-term effects of systemic cancer treatment on DNA oxidative damage: The potential for targeted therapies. Cancer Lett. 2012, 327, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.F.; Gu, S.; Shan, C.; Marchado, S.; Arias-Carrión, O. Oxidative Stress and Adult Neurogenesis. Stem Cell Rev. Rep. 2015, 11, 706–709. [Google Scholar] [CrossRef] [PubMed]
- Lam, V.; Hackett, M.; Takechi, R. Antioxidants and Dementia Risk: Consideration through a Cerebrovascular Perspective. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Aderka, D. The potential biological and clinical significance of the soluble tumor necrosis factor receptors. Cytokine Growth Factor Rev. 1996, 7, 231–240. [Google Scholar] [CrossRef]
- Patel, S.K.; Wong, A.L.; Wong, F.L.; Breen, E.C.; Hurria, A.; Smith, M.; Kinjo, C.; Paz, I.B.; Kruper, L.; Somlo, G.; et al. Inflammatory biomarkers, comorbidity, and neurocognition in women With newly diagnosed breast cancer. J. Natl. Cancer Inst. 2015, 107, djv131. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, V.; Thwaites, R.; Cercignani, M.; Sacre, S.; Harrison, N.; Whiteley-Jones, H.; Mullen, L.; Chamberlain, G.; Davies, K.; Zammit, C.; et al. A feasibility study exploring the role of pre-operative assessment when examining the mechanism of “chemo-brain” in breast cancer patients. Springerplus 2016, 5, 390. [Google Scholar] [CrossRef] [PubMed]
- Kesler, S.; Janelsins, M.; Koovakkattu, D.; Palesh, O.; Mustian, K.; Morrow, G.; Dhabhar, F.S. Reduced hippocampal volume and verbal memory performance associated with interleukin-6 and tumor necrosis factor-alpha levels in chemotherapy-treated breast cancer survivors. Brain Behav. Immun. 2013, 30, S109–S116. [Google Scholar] [CrossRef] [PubMed]
- Shibayama, O.; Yoshiuchi, K.; Inagaki, M.; Matsuoka, Y.; Yoshikawa, E.; Sugawara, Y.; Akechi, T.; Wada, N.; Imoto, S.; Murakami, K.; et al. Association between adjuvant regional radiotherapy and cognitive function in breast cancer patients treated with conservation therapy. Cancer Med. 2014, 3, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress—implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.A.; Stendell-Hollis, N.R.; Rock, C.L.; Cussler, E.C.; Flatt, S.W.; Pierce, J.P. Plasma and dietary carotenoids are associated with reduced oxidative stress in women previously treated for breast cancer. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 2008–2015. [Google Scholar] [CrossRef] [PubMed]
- Butalla, A.C.; Crane, T.E.; Patil, B.; Wertheim, B.C.; Thompson, P.; Thomson, C.A. Effects of a carrot juice intervention on plasma carotenoids, oxidative stress, and inflammation in overweight breast cancer survivors. Nutr. Cancer 2012, 64, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine, Panel on Micrnutrients. DRI: Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2002. [Google Scholar]
- Polidori, M.C.; Praticó, D.; Mangialasche, F.; Mariani, E.; Aust, O.; Anlasik, T.; Mang, N.; Pientka, L.; Stahl, W.; Sies, H.; et al. High fruit and vegetable intake is positively correlated with antioxidant status and cognitive performance in healthy subjects. J. Alzheimers Dis. 2009, 17, 921–927. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, 384S–392S. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.J.; Saunders, C.; Butler, L.T.; Spencer, J.P. Fruits, vegetables, 100% juices, and cognitive function. Nutr. Rev. 2014, 72, 774–789. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.G.; Thangthaeng, N.; Poulose, S.M.; Shukitt-Hale, B. Role of fruits, nuts, and vegetables in maintaining cognitive health. Exp. Gerontol. 2017, 94, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Pomykala, K.L.; Ganz, P.A.; Bower, J.E.; Kwan, L.; Castellon, S.A.; Mallam, S.; Cheng, I.; Ahn, R.; Breen, E.C.; Irwin, M.R.; et al. The association between pro-inflammatory cytokines, regional cerebral metabolism, and cognitive complaints following adjuvant chemotherapy for breast cancer. Brain Imaging Behav. 2013, 7, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Simen, A.A.; Bordner, K.A.; Martin, M.P.; Moy, L.A.; Barry, L.C. Cognitive dysfunction with aging and the role of inflammation. Ther. Adv. Chronic Dis. 2011, 2, 175–195. [Google Scholar] [CrossRef] [PubMed]
- Sartori, A.; Vance, D. The impact of inflammation on cognitive function in older adults: Implications for health care practice and research. J. Neurosci. Nurs. 2012, 44, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Fardell, J.E.; Vardy, J.; Johnston, I.N.; Winocur, G. Chemotherapy and cognitive impairment: Treatment options. Clin. Pharmacol. Ther. 2011, 90, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Wefel, J.S.; Kesler, S.R.; Noll, K.R.; Schagen, S.B. Clinical characteristics, pathophysiology, and management of noncentral nervous system cancer-related cognitive impairment in adults. CA Cancer J. Clin. 2015, 65, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Oh, P.J.; Kim, J. The effects of nonpharmacologic interventions on cognitive function in patients with cancer: A meta-analysis. Oncol. Nurs. Forum 2016, 43, E205–E217. [Google Scholar] [CrossRef] [PubMed]
- Treanor, C.J.; McMenamin, U.C.; O’Neill, R.F.; Cardwell, C.R.; Clarke, M.J.; Cantwell, M.; Donnelly, M. Non-pharmacological interventions for cognitive impairment due to systemic cancer treatment. Cochrane Database Syst. Rev. 2016, CD011325. [Google Scholar] [CrossRef] [PubMed]
- Demark-Wahnefried, W.; Aziz, N.M.; Rowland, J.H.; Pinto, B.M. Riding the crest of the teachable moment: Promoting long-term health after the diagnosis of cancer. J. Clin. Oncol. 2005, 23, 5814–5830. [Google Scholar] [CrossRef] [PubMed]
| Breast Cancer Survivors | Controls | p | |
|---|---|---|---|
| Age, y (Mean ± SD) | 50.1 ± 10.1 | 50.8 ± 10.0 | 0.783 |
| Income, n (%) | |||
| ≥$60,000 | 16 (61.5) | 29 (78.4) | 0.145 |
| Race, n (%) | |||
| White | 20 (69.0) | 33 (86.8) | 0.075 |
| Black | 3 (10.3) | 0 (0.0) | |
| Asian | 2 (6.9) | 1 (2.6) | |
| More than One Race | 4 (13.8) | 4 (10.5) | |
| Ethnicity, n (%) | |||
| Hispanic or Latino | 6 (20.7) | 9 (23.7) | 0.771 |
| Education, n (%) | |||
| ≥ 4 year College Degree | 20 (69.0) | 32 (84.2) | 0.138 |
| Body Mass Index (BMI), kg/m2 (Mean ± SD) | 29.7 ± 6.3 | 27.3 ± 7.6 | 0.168 |
| Stage at Diagnosis, n (%) | |||
| Ductal Carcinoma in Situ (DCIS) | 3 (10.3) | - | |
| Stage I | 8 (27.6) | - | |
| Stage II | 11 (37.9) | - | |
| Stage III | 6 (20.7) | - | |
| Unknown | 1 (3.4) | - | |
| Treatment, n (%) | |||
| Chemotherapy Only | 10 (34.5) | - | |
| Radiation Only | 8 (27.6) | - | |
| Chemotherapy + Radiation | 11 (37.9) | - | |
| Current Hormone Therapy | 19 (65.5) | - | |
| Surgery | 29 (100) | - | |
| Time since Treatment-Months (Mean ± SD) | 18.6 ± 16.3 | - | |
| Total Cholesterol (mg/dL) 1 | 167 (130–189) | 166 (146–189) | 0.463 |
| Serum Carotenoid Concentrations (nmol/L) 1 | |||
| Alpha-carotene | 41.1 (19.7–64.5) | 53.3 (35.8–122.7) | 0.121 |
| Beta-carotene | 163.0 (79.5–298.8) | 217.5 (122.6–367.2) | 0.260 |
| Lycopene | 298.1 (225.8–369.8) | 312.4 (213.6–411.3) | 0.968 |
| Lutein & Zeaxanthin | 163.2 (92.9–272.9) | 214.7 (145.7–308.3) | 0.199 |
| Beta-cryptoxanthin | 68.7 (45.3–115.8) | 78.2 (49.6–154.9) | 0.354 |
| Phytofluene | 65.2 (50.7–92.4) | 62.1 (48.7–100.2) | 0.958 |
| Phytoene | 68.6 (50.3–82.4) | 53.6 (43.1–75.6) | 0.083 |
| Total Carotenoids | 933.3 (663.2–1120.5) | 1052.3 (782.2–1356.1) | 0.314 |
| Average Daily Intakes (Mean ± SD) | |||
| Fruit (cups) | 1.0 ± 0.8 | 1.1 ± 0.9 | 0.568 |
| Vegetables (cups) | 2.8 ± 1.7 | 2.6 ± 1.6 | 0.761 |
| Total Carotenoid Intake (mg) | 20.8 ± 13.5 | 18.7 ± 10.3 | 0.514 |
| Breast Cancer Survivors | Controls | p | |
|---|---|---|---|
| List-Sorting Working Memory | 108.8 (17.5) | 105.3 (11.2) | 0.353 |
| Picture Vocabulary | 108.2 (16.2) | 115.2 (14.5) | 0.072 |
| Picture Sequence Memory | 100.5 (18.9) | 103.1 (14.7) | 0.536 |
| Oral Reading Recognition | 117.4 (14.3) | 116.7 (16.4) | 0.864 |
| Breast Cancer Survivors | Controls | p | |
|---|---|---|---|
| Total FACT-Cog Score 1 | 88.70 (5.27) | 119.17 (4.60) | <0.001 |
| Perceived Cognitive Impairments | 45.43 (3.01) | 61.66 (2.63) | <0.001 |
| Impact of Perceived Cognitive Impairments on Quality of Life | 10.06 (0.83) | 13.60 (0.73) | 0.002 |
| Comments from Others | 12.94 (0.53) | 15.57 (0.46) | <0.001 |
| Perceived Cognitive Abilities | 20.28 (1.44) | 28.34 (1.26) | <0.001 |
| Serum Carotenoid Concentrations (nmol/L). | Low Carotenoid BCS | High Carotenoid BCS | Low Carotenoid Control | High Carotenoid Control |
|---|---|---|---|---|
| Alpha-carotene | 27.3 (13.0) | 153.8 (269.9) | 43.3 (33.6) | 90.3 (150.8) |
| Beta-carotene | 103.9 (58.7) a | 461.1 (427.4) b | 140.7 (78.5) a | 425.4 (215.7) b |
| Lycopene | 275.0 (74.4) a | 344.1 (99.4) ab | 241.4 (80.8) a | 417.3 (198.4) b |
| Lutein & Zeaxanthin | 117.4 (44.5) a | 366.3 (263.6) b | 174.0 (79.5) ac | 299.8 (121.7) bc |
| Beta-cryptoxanthin | 54.4 (34.5) a | 135.9 (133.5) ab | 69.1 (58.1) ab | 149.4 (105.3) b |
| Phytofluene | 55.5 (34.6) ab | 113.8 (91.8) a | 53.9 (22.5) b | 108.4 (63.4) a |
| Phytoene | 56.5 (23.1) ab | 87.5 (40.1) a | 47.9 (9.9) b | 79.7 (39.0) a |
| Total Carotenoids | 689.9 (185.0) a | 1662.4 (1065.7) b | 770.3 (179.9) a | 1619.6 (604.2) b |
| Breast Cancer Survivors | Controls | p | |
|---|---|---|---|
| IL-6 (pg/mL) | 2.0 (1.1) | 1.8 (1.1) | 0.431 |
| IL-1ra (pg/mL) | 476.8 (91.6) | 513.1 (78.4) | 0.766 |
| sTNFRII (pg/mL) | 3037.41 (128.0) | 2717.3 (115.8) | 0.073 |
| CRP (ng/mL) | 2156.8 (358.2) | 3012.3 (296.3) | 0.072 |
| IL-6 | sTNFR-II | CRP | IL-1ra | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β | p | Model Adjusted R2 | β | p | Model Adjusted R2 | β | p | Model Adjusted R2 | β | p | Model Adjusted R2 | |
| 0.483 ** | 0.175 * | 0.142 * | 0.032 | |||||||||
| Age | 0.225 | 0.020 | 0.284 | 0.022 | −0.064 | 0.622 | 0.264 | 0.046 | ||||
| BMI | 0.433 | <0.001 | 0.036 | 0.790 | 0.337 | 0.024 | −0.018 | 0.900 | ||||
| Serum Carotenoids | −0.353 | 0.001 | −0.404 | 0.005 | −0.158 | 0.277 | −0.162 | 0.267 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuniga, K.E.; Moran, N.E. Low Serum Carotenoids Are Associated with Self-Reported Cognitive Dysfunction and Inflammatory Markers in Breast Cancer Survivors. Nutrients 2018, 10, 1111. https://doi.org/10.3390/nu10081111
Zuniga KE, Moran NE. Low Serum Carotenoids Are Associated with Self-Reported Cognitive Dysfunction and Inflammatory Markers in Breast Cancer Survivors. Nutrients. 2018; 10(8):1111. https://doi.org/10.3390/nu10081111
Chicago/Turabian StyleZuniga, Krystle E., and Nancy E. Moran. 2018. "Low Serum Carotenoids Are Associated with Self-Reported Cognitive Dysfunction and Inflammatory Markers in Breast Cancer Survivors" Nutrients 10, no. 8: 1111. https://doi.org/10.3390/nu10081111
APA StyleZuniga, K. E., & Moran, N. E. (2018). Low Serum Carotenoids Are Associated with Self-Reported Cognitive Dysfunction and Inflammatory Markers in Breast Cancer Survivors. Nutrients, 10(8), 1111. https://doi.org/10.3390/nu10081111





