New Insights about How to Make an Intervention in Children and Adolescents with Metabolic Syndrome: Diet, Exercise vs. Changes in Body Composition. A Systematic Review of RCT
Abstract
1. Introduction
1.1. Definitions
1.2. Noncommunicable Diseases (NCDs)
1.3. Metabolic Syndrome (MS): Concept and Prevalence (Table 1 and Table 2)
1.4. Strategies for Intervention in Overweight, Obesity and T2DM
1.5. Changes in Body Composition Andmetabolic Abnormalities
1.6. Use of Pharmacology in the Interventions of Changes Body Composition
1.7. Theoretical Framework and Purpose of the Review
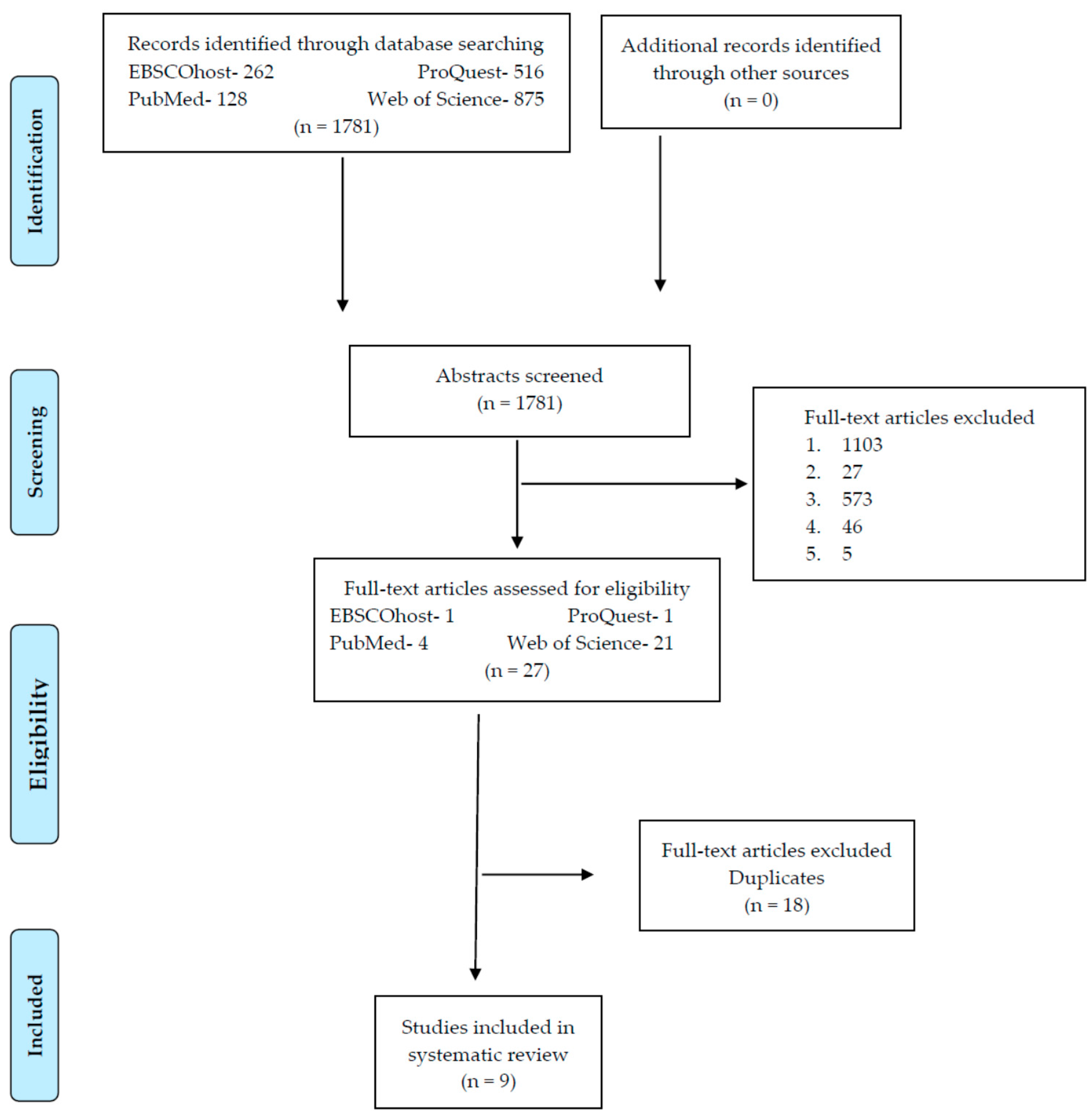
2. Method
2.1. Selection Criteria of the Clinical Trials and Search Strategy
2.2. Data Extraction, Synthesis of Results and Risk of Information Loss
3. Results
3.1. Search Characteristics and Types of Identified Interventions
- Include a part or a sample without the objective pathology of the systematic revision
- Dietary and physical exercise interventions or education to changes body composition not defined
- No comparison or analysis of the anthropometric parameter of interest, before or after intervention
- Include adult sample (older than 18 years old)
- Sample not human
3.2. Variations in Body Composition; Exclusive vs. Multidisciplinary Intervention (Table 6)
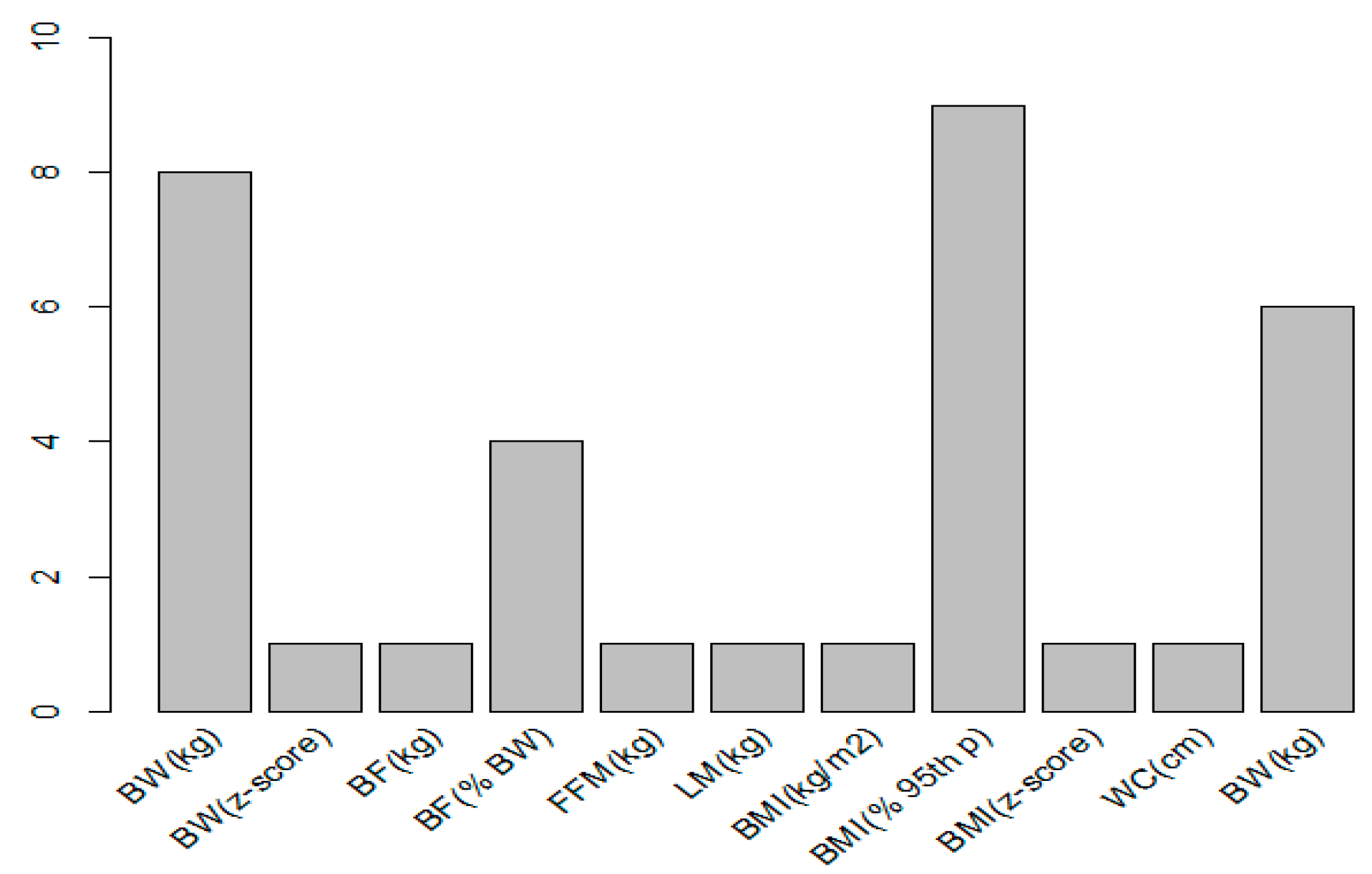
3.3. Anthropometric Parameters and Units of Measurement to Express the Changes in Body Composition
4. Discussion
4.1. Changes in Body Composition. Comparison between This Work and Other Reviews and/or Meta-Analyses
4.2. Practical Recommendations for the Design of Future Clinical Trials of Patients with Overweight, Obesity, T2DM and MS (Table 3 and Table 4)
- Based on the lifestyle changes in weight loss programs, the following results were obtained [32]: (6a) One contact with the patient in ≥26 h, demonstrating small reductions in weight excess in overweight and obese patients, without evidence of harm. (6b) One contact with the patient in ≥52 h, demonstrating an improved blood pressure and other cardio-metabolic improvements.
- In the dietary intervention, at the stage of changes in body composition, the type of energy restriction must be selected depending on the degree of excess weight (Table 3 and Table 4). Besides, the following recommendations must also be considered: (7a) an adaptation in the daily planning of the energy density and macronutrients if the patient completes the physical exercises regularly, especially the intake of complex carbohydrates [79]. (7b) be especially cautious regarding the planning of the carbohydrate intake guidelines for the days where physical exercise is completed [80].
- Table 3 and Table 4 summarize the strategies that must be shown by an intervention of physical exercise in patients looking for changes of body composition (BW, BF, BMI and WC). In addition, according to the American College of Sports Medicine (ACSM) in clinical trials including physical exercise, the following aspects should be recorded [82]: (9a) Cardiorespiratory fitness exercise of resistance: the frequency (days per week), intensity (mild, moderate or vigorous), time (duration), type, volume (the distance travelled or the expenditure of energy that causes), pattern (one or more than one session for day) and progression (in volume of exercise adjusted to the duration, the frequency and/or intensity). (9b) Resistance exercise: frequency (each muscle group should be trained 2–3 times per week), intensity (mild, moderate or vigorous in function of the maximum repetitions and on the weight lifted), time (to be determined), the type (depending on the muscle group involved and the weight lifted); repetitions, session or patterns (rest intervals of 2–3 min between each set of repetitions; it is recommended ≥48 h between sessions for any muscle group) and progression (gradual increase of greater resistance, and/or more replicates per set).
- In the article by Pieles GE and colleagues (Table 1 and Figure 1), it is shown that progress has been made in the recommendations for children and adolescents. However, a more accurate vision should be taken as indicated by ACSM both theoretically and practically to be able to prove effectiveness [82,83]. For example, in the maintenance and/or reduction of body composition in patients with overweight, obesity, T2DM and MS, the amount of time they can be seated in front of the television, must be limited and relative to their ages (Table 4). However, occasionally this recommendation is not met [84].
- Mark the objective of the changes in body composition, around a 5, 10 or 15% changes of body weight or body fat, without necessity to set an ideal BMI [85,86]. One should be cautious when it comes to not regaining the weight and/or fat [87] and be more tolerant of variations in body composition [85].
4.3. Limitations and Strength of the Systematic Review
- The range of search dates of this systematic review, having found 1781 clinical trials from 2005–2017.
- The following contributions to the review of this subject: (2a) The MS definitions and diagnostic criteria (Table 1 and Table 2). (2b) A synthesis of guides about the treatment of overweight, obesity, and T2DM (Table 3 and Table 4) together with the development of practical recommendations for the design of future clinical trials related to MS in children and adolescents. This is due to the lack of guidelines and consensus on MS in children and adolescents.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflict of Interest
References
- Diem, G.; Brownson, R.C.; Grabauskas, V.; Shatchkute, A.; Stachenko, S.; Diem, G.; Brownson, R.C.; Grabauskas, V.; Shatchkute, A.; Stachenko, S. Prevention and control of noncommunicable diseases through evidence-based public health: Implementing the NCD 2020 Action Plan. Glob. Health Promot. 2016, 23, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Riley, L.; Guthold, R.; Cowan, M.; Savin, S.; Bhatti, L.; Timothy Armstrong, T.; Bonita, R. The World Health Organization STEPwise Approach to Noncommunicable Disease Risk-Factor Surveillance: Methods, Challenges, and Opportunities. Am. J. Public Health 2016, 106, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Monforte, M.; Sánchez, E.; Barrio, F.; Costa, B.; Flores-Mateo, G. Metabolic syndrome and dietary patterns: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2016, 56, 925–947. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Tango, T. Effects of lifestyle modification on metabolic syndrome: A systematic review and meta-analysis. BMC Med. 2012, 10, 138. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Metabolic syndrome update. Trends Cardiovasc. Med. 2016, 26, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Han, T.S.; Lean, M.E. A clinical perspective of obesity, metabolic syndrome and cardiovascular disease. JRSM Cardiovasc. Dis. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Dulloo, A.G.; Montani, J.P. Pathways from dieting to weight regain, to obesity and to the metabolic syndrome: An overview. Obes. Rev. 2015, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hong, A.R.; Lim, S. Clinical characteristics of metabolic syndrome in Korea, and its comparison with other Asian countries. J. Diabetes Investig. 2015, 6, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Calton, E.K.; James, A.P.; Pannu, P.K.; Soares, M.J. Certain dietary patterns are beneficial for the metabolic syndrome: Reviewing the evidence. Nutr. Res. 2014, 34, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J. A Comprehensive Review on Metabolic Syndrome. Cardiol. Res. Pract. 2014, 2014, 943162. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.; Bremer, A.A.; Lustig, R.H. What is metabolic syndrome, and why are children getting it? Ann. N. Y. Acad. Sci. 2013, 1281, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.K.; Badjatya, V.; Nema, R.K. A review on the metabolic syndrome: Plethora of disease. Adv. Pharmacol. Toxicol. 2013, 14, 29–42. [Google Scholar]
- Nelson, R.A.; Bremer, A.A. Insulin Resistance and Metabolic Syndrome in the Pediatric Population. Metab. Syndr. Relat. Disord. 2010, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Halpern, A.; Mancini, M.C.; Magalhães, M.E.C.; Fisberg, M.; Radominski, R.; Bertolami, M.C.; Adriana Bertolami, A.; de Melo, M.E.; Zanella, M.T.; Queiroz, M.S. Metabolic syndrome, dyslipidemia, hypertension and type 2 diabetes in youth: From diagnosis to treatment. Diabetol. Metab. Syndr. 2010, 2, 55–75. [Google Scholar] [CrossRef] [PubMed]
- Fleischman, A.; Rhodes, E.T. Management of obesity, insulin resistance and type 2 diabetes in children: Consensus and controversy. Diabetes Metab. Syndr. Obes. 2009, 2, 185–202. [Google Scholar] [PubMed]
- Hanefeld, M.; Pistrosch, F.; Bornstein, S.R.; Birkenfeld, A.L. The metabolic vascular syndrome—Guide to an individualized treatment. Rev. Endocr. Metab. Disord. 2016, 17, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Pucci, G.; Alcidi, R.; Tap, L.; Battista, F.; Mattace-Raso, F.; Schillaci, G. Sex-and gender-related prevalence, cardiovascular risk and therapeutic approach in metabolic syndrome: A review of the literature. Pharmacol. Res. 2017, 120, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Graf, C.; Ferrari, N. Metabolic Syndrome in Children and Adolescents. Visc. Med. 2016, 32, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Kelishadi, R.; Hovsepian, S.; Djalalinia, S.; Qorbani, M. A systematic review on the prevalence of metabolic syndrome in Iranian children and adolescents. J. Res. Med. Sci. 2016, 21, 90. [Google Scholar] [PubMed]
- Oliveros, E.; Somers, V.K.; Sochor, O.; Goel, K.; Lopez-Jimenez, F. The concept of normal weight obesity. Prog. Cardiovasc. Dis. 2014, 56, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, L.; Anania, C.; Martino, F.; Poggiogalle, E.; Chiarelli, F.; Arca, M.; Chiesa, C. Management of metabolic syndrome in children and adolescents. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.T.K.; Ball, G.D.C.; Franks, P.W. Metabolic syndrome in youth: Current issues and Challenges. Appl. Physiol. Nutr. Metab. 2007, 32, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Titmuss, A.T.; Srinivasan, S. Metabolic syndrome in children and adolescents: Old concepts in a young population. J. Paediatr. Child Health 2016, 52, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Tavares Giannini, D.; Caetano Kuschnir, M.C.; Szklo, M. Metabolic Syndrome in Overweight and Obese Adolescents: A Comparison of Two Different Diagnostic Criteria. Ann. Nutr. Metab. 2014, 64, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Haemer, M.A.; Grow, H.M.; Fernandez, C.; Lukasiewicz, G.J.; Rhodes, E.T.; Shaffer, L.A.; Sweeney, B.; Woolford, S.J.; Estrada, E. Addressing prediabetes in childhood obesity treatment programs: Support from research and current practice. Child. Obes. 2014, 10, 292–303. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association (ADA). Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 2014, 37, S81–S90. [Google Scholar]
- Hoelscher, D.M.; Kirk, S.; Ritchie, L.; Cunningham-Sabo, L. Academy Positions Committee. Position of the Academy of Nutrition and Dietetics: Interventions for the Prevention and Treatment of Pediatric Overweight and Obesity. J. Acad. Nutr. Dietet. 2013, 113, 1375–1394. [Google Scholar] [CrossRef] [PubMed]
- Seagle, H.M.; Strain, G.W.; Makris, A.; Reeves, R.S. American Dietetic Association. Position of the American Dietetic Association: Weight management. J. Am. Dietet. Assoc. 2009, 109, 330–346. [Google Scholar]
- Fitch, A.; Fox, C.; Bauerly, K.; Gross, A.; Heim, C.; Judge-Dietz, J.; Kaufman, T.; Krych, E.; Kumar, S.; Landin, D.; et al. Prevention and Management of Obesity for Children and Adolescents. Institute for Clinical Systems Improvement. Available online: https://www.ohcoop.org/wp-content/uploads/Clinical-Guidelines-Prevention-and-Management-Obesity-in-Children-and-Adolscent.pdf (accessed on 1 February 2018).
- Copeland, K.C.; Silverstein, J.; Moore, K.R.; Prazar, G.E.; Raymer, T.; Shiffman, R.N.; Springer, S.C.; Thaker, V.V.; Anderson, M.; Spann, S.J.; et al. Management of newly diagnosed type 2 Diabetes Mellitus (T2DM) in children and adolescents. Pediatrics 2013, 131, 364–382. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association (ADA). 4. Lifestyle Management. Diabetes Care 2017, 40, 33–43. [Google Scholar]
- O’Connor, E.A.; Evans, C.V.; Burda, B.U.; Walsh, E.S.; Eder, M.; Lozano, P. Screening for obesity and intervention for weight management in children and adolescents: Evidence report and systematic review for the US Preventive Services Task Force. JAMA 2017, 317, 2427–2444. [Google Scholar] [CrossRef] [PubMed]
- Consideration of the Evidence on Childhood Obesity for the Commission on Ending Childhood Obesity: Report of the ad Hoc Working Group on Science and Evidence for Ending Childhood Obesity, Geneva, Switzerland. 2016. Available online: http://apps.who.int/iris/bitstream/10665/206549/1/9789241565332_eng.pdf?ua=1 (accessed on 1 February 2017).
- Onge, E.S.; Miller, S.A.; Motycka, C.; DeBerry, A. A review of the treatment of type 2 diabetes in children. J. Pediatr. Pharmacol. Ther. 2015, 20, 4–16. [Google Scholar] [PubMed]
- National Health and Medical Research Council (NHMRC). Clinical Practice Guidelines for the Management of Overweight and Obesity in Adults, Adolescents and Children in Australia. Available online: https://www.nhmrc.gov.au/guidelines-publications/n57 (accessed on 3 July 2018).
- Logue, J. Management of Obesity. A National Clinical Guideline; NHS Quality Improvement Scotland: Edinburgh, UK, 2010. [Google Scholar]
- August, G.P.; Caprio, S.; Fennoy, I.; Freemark, M.; Kaufman, F.R.; Lustig, R.H.; Silverstein, J.H.; Speiser, P.W.; Styne, D.M.; Montori, V.M. Prevention and treatment of pediatric obesity: An endocrine society clinical practice guideline based on expert opinion. J. Clin. Endocrinol. Metab. 2008, 93, 4576–4599. [Google Scholar] [CrossRef] [PubMed]
- Reig, M.; Rizo, M.M.; Cortés, E. Predictors of weight loss and fat in the dietary management: Sex, age, BMI and consultin assistance. Nutr. Hosp. 2015, 32, 1028–1035. [Google Scholar]
- Gutiérrez, A.; Reig, M.; Rizo, M.; Cortés, E.; Mur, N.; Aguilar, M.I. Measurement units used in treatments to reduce weight and obesity. Systematic review. Nutr. Hosp. 2014, 30, 478–485. [Google Scholar]
- McCarthy, H.D.; Samani-Radia, D.; Jebb, S.A.; Prentice, A.M. Skeletal muscle mass reference curves for children and adolescents. Ped. Obes. 2014, 9, 249–259. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, H.D. Measuring growth and obesity across childhood and adolescence. Proc. Nutr. Soc. 2014, 73, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Oguoma, V.M.; Nwose, E.U.; Richards, R.S. Prevalence of cardio-metabolic syndrome in Nigeria: A systematic review. Public Health 2015, 129, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Duprez, D.; Toleuova, A. Prehypertension and the cardiometabolic syndrome: Pathological and clinical consequences. Expert Rev. Cardiovasc. Ther. 2013, 11, 1725–1733. [Google Scholar] [CrossRef] [PubMed]
- Stoner, L.; Rowlands, D.; Morrison, A.; Credeur, D.; Hamlin, M.; Gaffney, K.; Lambrick, D.; Matheson, A. Efficacy of Exercise Intervention for Weight Loss in Overweight and Obese Adolescents: Meta-Analysis and Implications. Sports Med. 2016, 46, 1737–1751. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Garnett, S.P.; Baur, L.; Burrows, T.; Stewart, L.; Neve, M.; Collins, C. Effectiveness of lifestyle interventions in child obesity: Systematic review with meta-analysis. Pediatrics 2012, 130, e1647–e1671. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.Á.; Martín-Calvo, N. The major European dietary patterns and metabolic syndrome. Rev. Endocr. Metab. Disord. 2013, 14, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Quinn, S.M.; Baur, L.A.; Garnett, S.P.; Cowell, C.T. Treatment of clinical insulin resistance in children: A systematic review. Obes. Rev. 2010, 11, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Khokhar, A.; Umpaichitra, V.; Chin, V.L.; Perez-Colon, S. Metformin Use in Children and Adolescents with Prediabetes. Pediatr. Clin. N. Am. 2017, 64, 1341–1353. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Garnett, S.P.; Baur, L.A. Childhood Obesity and Insulin Resistance: How Should It Be Managed? Curr. Treat. Opt. Cardiovasc. Med. 2014, 16, 351. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association (ADA). 12. Children and Adolescents. Diabetes Care 2017, 40, S105–S113. [Google Scholar]
- Rodd, C.; Metzger, D.L.; Sharma, A. Canadian Pediatric Endocrine Group (CPEG) Working Committee for National Growth Charts. Extending World Health Organization weight-for-age reference curves to older children. BMC Pediatr. 2014, 14, 32. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Inter. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.A.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-analyses of Health Care Interventions: Checklist and Explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Wiesman, F.; Hasman, A.; van den Herik, H.J. Information retrieval: An overview of system characteristics. Int. J. Med. Inform. 1997, 47, 5–26. [Google Scholar] [CrossRef]
- de Mello, M.T.; de Piano, A.; Carnier, J.; Sanches, P.D.L.; Corrêa, F.A.; Tock, L.; Ernandes, R.M.Y.; Tufik, S.; Dâmaso, A.R. Long-term effects of aerobic plus resistance training on the metabolic syndrome and adiponectinemia in obese adolescents. J. Clin. Hypertens. 2011, 13, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Armeno, M.L.; Krochik, A.G.; Mazza, C.S. Evaluation of two dietary treatments in obese hyperinsulinemic adolescents. J. Pediatr. Endocrinol. Metab. 2011, 24, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Van der Aa, M.P.; Elst, M.A.J.; Van De Garde, E.M.W.; Van Mil, E.G.A.H.; Knibbe, C.A.J.; Van der Vorst, M.M.J. Long-term treatment with metformin in obese, insulin-resistant adolescents: Results of a randomized double-blinded placebo controlled trial. Nutr. Diabetes 2016, 6, e228. [Google Scholar] [CrossRef] [PubMed]
- Garnett, S.P.; Gow, M.; Ho, M.; Baur, L.A.; Noakes, M.; Woodhead, H.J.; Broderick, C.R.; Burrell, S.; Chisholm, K.; Halim, J.; et al. Optimal macronutrient content of the diet for adolescents with prediabetes; RESIST a randomised control trial. J. Clin. Endocrinol. Metab. 2013, 98, 2116–2125. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Díaz, R.A.; Talavera, J.O.; Pool, E.C.; Ortiz-Navarrete, F.V.; Solórzano-Santos, F.; Mondragón-González, R.; Valladares-Salgado, A.; Cruz, M.; Aguilar-Salinas, C.A.; Wacher, N.H. Metformin decreases plasma resistin concentrations in pediatric patients with impaired glucose tolerance: A placebo-controlled randomized clinical trial. Metabolism 2012, 61, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
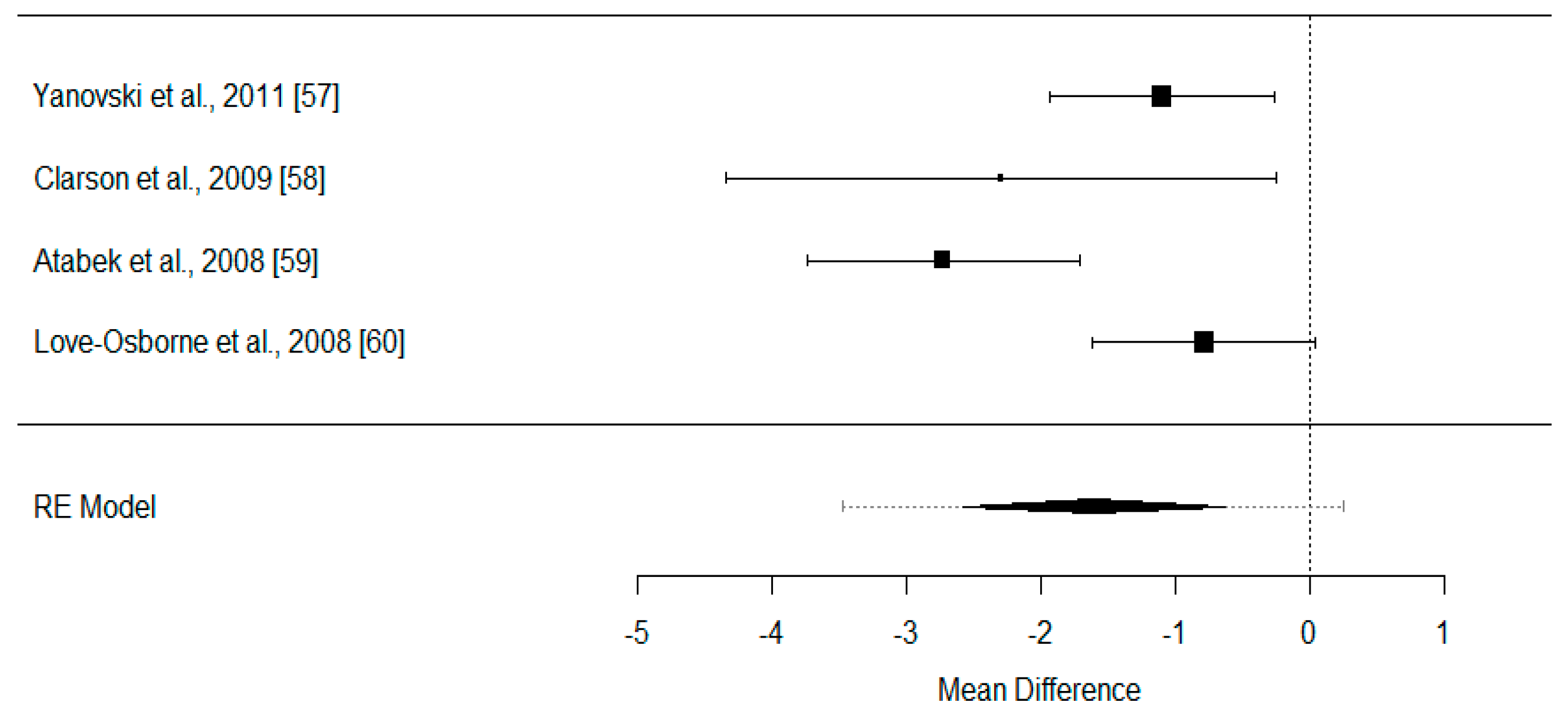
- Yanovski, J.A.; Krakoff, J.; Salaita, C.G.; McDuffie, J.R.; Kozlosky, M.; Sebring, N.G.; Reynolds, J.C.; Brady, S.M.; Calis, K.A. Effects of metformin on body weight and body composition in obese insulin-resistant children: A randomized clinical trial. Diabetes 2011, 60, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Clarson, C.L.; Mahmud, F.H.; Baker, J.E.; Clark, H.E.; Mckay, W.M.; Schauteet, V.D.; Hill, D.J. Metformin in combination with structured lifestyle intervention improved body mass index in obese adolescents, but did not improve insulin resistance. Endocrine 2009, 36, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Atabek, M.E.; Pirgon, O. Use of metformin in obese adolescents with hyperinsulinemia: A 6-month, randomized, double-blind, placebo-controlled clinical trial. J. Pediatr. Endocrinol. Metab. 2008, 21, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Love-Osborne, K.; Sheeder, J.; Zeitler, P. Addition of metformin to a lifestyle modification program in adolescents with insulin resistance. J. Pediatr. 2008, 152, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Mead, E.; Brown, T.; Rees, K.; Azevedo, L.B.; Whittaker, V.; Jones, D.; Olajide, J.; Mainardi, G.M.; Corpeleijn, E.; O’Malley, C.; et al. Diet, physical activity and behavioural interventions for the treatment of overweight or obese children from the age of 6 to 11 years. Cochrane Database Syst. Rev. 2017, 6, CD012651. [Google Scholar] [CrossRef] [PubMed]
- García-Hermoso, A.; Sánchez-López, M.; Martínez-Vizcaíno, V. Effects of Aerobic Plus Resistance Exercise on Body Composition Related Variables in Pediatric Obesity: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Pediatr. Exerc. Sci. 2015, 27, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Garnett, S.P.; Baur, L.A.; Burrows, T.; Stewart, L.; Neve, M.; Collins, C. Impact of dietary and exercise interventions on weight change and metabolic outcomes in obese children and adolescents: A systematic review and meta-analysis of randomized trials. JAMA Pediatr. 2013, 167, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.M.; Dalskov, S.; Van Baak, M.; Jebb, S.; Kafatos, A.; Pfeiffer, A.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunešová, M.; Holst, C.; et al. The Diet, Obesity and Genes (Diogenes) Dietary Study in eight European countries – a comprehensive design for long-term intervention. Obes. Rev. 2010, 11, 76–91. [Google Scholar] [CrossRef] [PubMed]
- Whitlock, E.P.; O’Connor, E.A.; Williams, S.B.; Beil, T.L.; Lutz, K.W. Effectiveness of Weight Management Interventions in Children: A Targeted Systematic Review for the USPSTF. Pediatrics 2010, 125, e396–e418. [Google Scholar] [CrossRef] [PubMed]
- Gow, M.L.; Garnett, S.P.; Baur, L.A.; Lister, N.B. The Effectiveness of Different Diet Strategies to Reduce Type 2 Diabetes Risk in Youth. Nutrients 2016, 8, 486. [Google Scholar] [CrossRef] [PubMed]
- Kroeger, C.M.; Hoddy, K.K.; Varady, K.A. Impact of weight regain on metabolic disease risk: A review of human trials. J. Obes. 2014. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gøtzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. Int. J. Surg. 2012, 10, 28–55. [Google Scholar] [CrossRef] [PubMed]
- Argimon, J.M.; Jimenéz, J. Métodos de investigación clínica y epidemiológica. Elsevier 2013, 27, 109–112. (In Spanish) [Google Scholar]
- Reig, M.; Rizo, M.M.; Cortés, E. Indicators of success in the dietary management of overweight and obesity: Weight, body fat loss and quality. Nutr. Hosp. 2015, 32, 1009–1016. [Google Scholar]
- Millstein, R.A. Measuring Outcomes in Adult Weight Loss Studies That Include Diet and Physical Activity: A Systematic Review. J. Nutr. Metab. 2014, 2014, 421423. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, H.D.; Cole, T.J.; Fry, T.; Jebb, S.A.; Prentice, A.M. Body fat reference curves for children. Int. J. Obes. (Lond.) 2006, 30, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Reig, M.R.; Castell, E.C.; Baeza, M.R.; Hervás, A.G. The variability in adherence to dietary treatment and quality of weight loss: Overweight and obesity. Nutr. Hosp. 2015, 31, 2017–2024. [Google Scholar]
- Stubbs, J.; Whybrow, S.; Teixeira, P.; Blundell, J.; Lawton, C.; Westenhoefer, J.; Engel, D.; Shepherd, R.; Mcconnon, Á.; Gilbert, P.; et al. Problems in identifying predictors and correlates of weight loss and maintenance: Implications for weight control therapies based on behaviour change. Obes. Rev. 2011, 12, 688–708. [Google Scholar] [CrossRef] [PubMed]
- Desbrow, B.; McCormack, J.; Burke, L.M.; Cox, G.R.; Fallon, K.; Hislop, M.; Logan, R.; Marino, N.; Sawyer, S.M.; Shaw, G.; et al. Sports Dietitians Australia Position Statement: Sports Nutrition for the Adolescent Athlete. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 570–584. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E. Nutrition for endurance sports: Marathon, triathlon, and road cycling. J. Sports Sci. 2011, 29, S91–S99. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.D.; Ryan, D.H. 2013 AHA/ACC/TOS. Guideline for management of overweight and obesity in adults. J. Am. Coll. Cardiol. 2014, 63, 2994–3000. [Google Scholar] [CrossRef] [PubMed]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand (ACSM). Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef] [PubMed]
- Pieles, G.E.; Horn, R.; Williams, C.A.; Stuart, A.G. Paediatric exercise training in prevention and treatment. Arch. Dis. Child. 2014, 99, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Mielgo-Ayuso, J.; Aparicio-Ugarriza, R.; Castillo, A.; Ruiz, E.; Avila, J.M.; Aranceta-Bartrina, J.; Angel Gil, A.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; et al. Sedentary behavior among Spanish children and adolescents: Findings from the ANIBES study. BMC Public Health 2017, 17, 94. [Google Scholar] [CrossRef] [PubMed]
- Ravussin, E.; Ryan, D. Energy Expenditure and Weight Control: Is the Biggest Loser the Best Loser? Obesity 2016, 24, 1607–1608. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.; Heaner, M. Guidelines (2013) for managing overweight and obesity in adults: Preface to the full report. Obesity (Silver Spring) 2014, 22, S1–S3. [Google Scholar] [CrossRef] [PubMed]
- Cefalu, W.T.; Bray, G.A.; Home, P.D.; Garvey, W.T.; Klein, S.; Pi-Sunyer, F.X.; Hu, F.B.; Raz, I.; Gaal, V.L.; Wolfe, B.M.; et al. Advances in the Science, Treatment, and Prevention of the Disease of Obesity: Ref lections From a Diabetes Care Editors’ Expert Forum. Diabetes Care 2015, 38, 1567–1582. [Google Scholar] [CrossRef] [PubMed]
- Delahanty, L.M. An expanded role for dietitians in maximising retention in nutrition and lifestyle intervention trials: Implications for clinical practice. J. Hum. Nutr. Diet. 2010, 23, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Spear, B.A.; Barlow, S.E.; Ervin, C.; Ludwig, D.S.; Saelens, B.E.; Schetzina, K.E.; Taveras, E.M. Recommendations for Treatment of Child and Adolescent Overweight and Obesity. Pediatrics 2007, 120, 254–288. [Google Scholar] [CrossRef] [PubMed]
- Reig, M. The management of qualitative and quantitative dietary treatment for overweight and obesity: Methodology and a new perspective on individualised assessment. Nutr. Hosp. 2015, 32, 2019–2029. [Google Scholar]
- Apovian, C.M.; Garvey, W.T.; Ryan, D.H. Challenging Obesity: Patient, Provider, and Expert Perspectives on the Roles of Available and Emerging Nonsurgical Therapies. Obesity 2015, 23, S1–S26. [Google Scholar] [CrossRef] [PubMed]
- Quiles, P.; Reig, M. Glycemic control through physical exercise in type 2 diabetes systematic review. Nutr. Hosp. 2015, 31, 1465–1472. [Google Scholar]
- Albert Pérez, E.J.; Reig García-Galbis, M. Effects of green tea on the nutritional status of the exercise. Nutr. Hosp. 2015, 32, 1417–1431. [Google Scholar] [PubMed]
| AHA Criteria [21] | IDF Criteria | WHO Criteria | NCEP ATP III Criteria | ||||
|---|---|---|---|---|---|---|---|
| Necessary components for the MS diagnosis | 3 of the 5 must be present | Central obesity and 2 of 4 other components must be present [21] | At least 3 or hyperinsulinemia and at least 2 must be present [22] | At least 3 must be present [23] | |||
| Age (years) | 12–19 | 6–9 [21] | 10–15 [21] 10–16 [23] | >15 [21] ≥16 [24] | ND | ND | |
| Essential criteria | ND | ND | ND | ND | Insulin resistance [23] | None [23] | |
| Waist circumference | WC ≥ 90th percentile for age, sex and race/ethnicity | WC ≥ 90th percentile for age (MS as entity is not diagnosed) [21] | WC ≥ 90th percentile [23] or adult cut-off if lower [21] | WC ≥ 90 cm in boys and ≥80 cm in girls [24] WC ≥ 94 cm in boys and ≥80 cm in girls [21] | Waist-to-hip ratio > 0.9 in boys and >0.85 in girls [23] BMI ≥ 75/85/95th percentile by age, sex [22] | WC ≥102 cm in boys and ≥88 cm in girls [23] WC > 90th percentile or BMI ≥ 97th percentile [22] WC > 75th percentile for age and sex [24] | |
| BMI | ND | ND | ND | ND | >30 kg/m2 [23] | ND | |
| Blood pressure | ≥90th percentile for age, sex, and height | ND | SBP ≥ 130 mmHg [23] SBP ≥ 130 mmHg or DBP ≥ 85 mmHg [24] | SBP ≥ 130 mmHg or DBP ≥85 mmHg [24] or treatment of previously diagnosed hypertension [21] | SBP ≥ 140 mmHg [23] | SBP ≥ 130 mmHg [23] SBP > 90th percentile for age and sex [24] | |
| Dyslipidemia | Triglyceride | ≥1.23 mmol/L (≥110 mg/dL) | ND | ≥1.7 mmol/L (≥150 mg/dL) [23] | ≥1.7 mmol/L (≥150 mg/dL) [24] or specific treatment for high triglycerides [21] | ≥1.7 mmol/L (≥150 mg/dL) [23] | ≥1.7 mmol/L (≥150 mg/dL) [23] ≥100 mg/dL [24] |
| HDL-C | ≤10th percentile for race and sex [21] | ND | <1.03 mmol/L (<40 mg/dL) [23] | <1.03 mmol/L (<40 mg/dL) in boys and <1.29 mmol/L (<50 mg/dL) in girls [24] or specific treatment for low HDL-C [21] | <0.91 mmol/L in boys <1.0 mmol/L in girls [23] | <1.0 mmol/L [23] 500 mg/dL, except boys from 15 to 18 years, whose cutoff point was <45 mg/dL [24] | |
| Glucose | Fasting glucose ≥5.6 mmol/L (≥100 mg/dL) [21] | ND | Fasting glucose ≥5.6 mmol/L (≥100 mg/dL) [23] | Fasting glucose ≥5.6 mmol/L (≥100 mg/dL) [24] or known T2DM [21] | Insulin resistance or diabetes [23] Fasting glucose ≥ 6.1 mmol/L (≥110 mg/dL) or ≥80/90th percentile by age, sex or diabetes [22] | Fasting glucose ≥6.1 mmol/L (≥110 mg/dL) [23] Fasting glucose ≥5.6 or 6.1 mmol/L (≥100 or 110 mg/dL) or 2 h glucose ≥140 mg/dL [22] | |
| Insulin | ND | ND | ND | ND | Insulin resistance [23] | ND | |
| WHO Criteria | ADA Criteria | |||
|---|---|---|---|---|
| Prediabetes [25] | Glucose | Fasting plasma glucose | 110–125 mg/dL (6.1–6.9 mmol/L) | 100–125 mg/dL (5.6–6.9 mmol/L) |
| Random Plasma Glucose | ND | Not applicable | ||
| 2-h plasma glucose (OGTT) | 140–200 mg/dL (7.8–11.0 mmol/L) | 140–200 mg/dL (7.8–11.0 mmol/L) | ||
| Hemoglobin A1c | ND | 5.7–6.4% | ||
| Impaired glucose tolerance [26] | Glucose | 2-h plasma glucose (OGTT) | ND | 140–199 mg/dL (7.8–11.0 mmol/L) |
| Type 2 Diabetes Mellitus [25,26] | Glucose | Fasting plasma glucose | ND | ≥126 mg/dL (7.0 mmol/L) |
| Random Plasma Glucose | ND | ≥200 mg/dL (11.1 mmol/L) | ||
| 2-h plasma glucose (OGTT) | ND | ≥200 mg/dL (11.1 mmol/L) | ||
| Hemoglobin A1c | ND | ≥6.5% | ||
| Author | Recommendations in Dietary Intervention and Exercise | ||
|---|---|---|---|
| Overweight and obesity | AND | [27] | Intervention: divided into three levels: primary, secondary and tertiary prevention Evidence: 2009 Academy of Nutrition and Dietetics (Figure 1) [28] |
| ICSI | [29] | Intervention: during the day, diet and physical activity. It identifies 4 levels of intervention in patients with BMI ≥ 85th percentile: prevention, structures weight management, integral multidisciplinary intervention, tertiary intervention Dietary intervention: the consumption of a diet with very low energy density BW: age, 2–11 years = 1 lb or 0.45 kg mo−1; age, 12–18 years = 2 lb or 0.91 kg wk−1 Evidence: [29] | |
| T2DM | [30] | Dietary intervention:
|
| Dietary Intervention | ||
| Energy restriction | Overweight and obesity | 1000 a 2000 Kcal day−1 [27] |
| T2DM | ≥1200 Kcal day−1 in ages between 6 and 12 years old [30] | |
| VLCD | Overweight and obesity | ≤1.000 Kcal day−1 ó 600 a 800 Kcal day−1 (PSMF) [27] |
| T2DM | ≥900 Kcal day−1 in ages between 6 and 12 years old [30] | |
| Macronutrients and diets | Different quantities of macronutrients (carbohydrates, proteins and fats) and different types of diets; PSMF (10–20 weeks), proteins (1.5 to 2.0 g kg−1 to reach the optimum body weight), carbohydrates (20–25 g day−1), water and other liquids without calories (2 L day−1), daily multivitamin supplements, balanced diet (for 10 weeks) [27] | |
| Physical exercise | ||
| Overweight and obesity | ≤2 years old should not watch television, supervised free play is encouraged; 4 to 6 years old, up to 120 min of moderate to rigorous physical activity (MVPA) each day, 60 min in structured activity and 60 min of free play; ≥10 years old, at least ≥60 min day−1 of physical activity which should consist primarily of MVPA. In adolescents, promote and incorporate more complex and personalized activities [29] | |
| T2DM | Children and adolescents with T2DM should practice moderate to vigorous physical activity for at least 60 min day−1 a day [27,31] Limited television time, to less than 2 h per day [27] Evidence grade D: expert opinions and evidence from metabolic syndrome and obesity studies. Prevalence of benefits over the harms. | |
| Search strategy | EBSCOhost Identified/Included | ProQuest Identified/Included | PubMed Identified/Included | Web of Science Identified/Included |
|---|---|---|---|---|
| “metabolic syndrome” AND “children” OR “teens” OR “pediatrics” AND “diet” OR “dietary treatment” OR “feeding” AND “nutrition” OR “nutritional counseling” OR “lifestyle” | 12/0 | 25/0 | 3/0 | 32/1 |
| “metabolic syndrome” AND “children” OR “teens” OR “pediatrics” AND “exercise” OR “physical activity” OR “sport” OR “weightlifting” | 54/0 | 90/0 | 13/0 | 44/2 |
| “metabolic syndrome” AND “children” OR “teens” OR “pediatrics” AND “weight loss” OR “weight reduction” OR “fat loss” OR “fat reduction” | 12/0 | 27/0 | 8/0 | 277/3 |
| “type II diabetes” OR “insulin resistance” OR “hyperinsulinism” OR “hyperinsulinaemia” OR “hyperglycemia” OR “dyslipidemia” OR “prediabetes” AND “children” OR “teens” OR “pediatrics” AND “weight loss” OR “weight reduction” OR “fat loss” OR “fat reduction” | 29/1 | 75/1 | 25/2 | 88/7 |
| “type II diabetes” OR “insulin resistance” OR “hyperinsulinism” OR “hyperinsulinaemia” OR “hyperglycemia” OR “dyslipidemia” OR “prediabetes” AND “children” OR “teens” OR “pediatrics” AND “diet” OR “dietary treatment” OR “feeding” AND “nutrition” OR “nutritional counseling” OR “lifestyle” | 23/0 | 55/0 | 13/1 | 78/2 |
| “type II diabetes” OR “insulin resistance” OR “hyperinsulinism” OR “hyperinsulinaemia” OR “hyperglycemia” OR “dyslipidemia” OR “prediabetes” AND “children” OR “teens” OR “pediatrics” AND “exercise” OR “physical activity” OR “sport” OR “weightlifting” | 114/0 | 200/0 | 65/1 | 135/6 |
| “metabolic syndrome” AND “hypertension” OR “high blood pressure” AND “children” OR “teens” OR “pediatrics” AND “weight loss” OR “weight reduction” OR “fat loss” OR “fat reduction” | 3/0 | 6/0 | 0/0 | 45/0 |
| “metabolic syndrome” AND “hypertension” OR “high blood pressure” AND “children” OR “teens” OR “pediatrics” AND “diet” OR “dietary treatment” OR “feeding” AND “nutrition” OR “nutritional counseling” OR “lifestyle” | 4/0 | 13/0 | 0/0 | 40/0 |
| “metabolic syndrome” AND “hypertension” OR “high blood pressure” AND “children” OR “teens” OR “pediatrics” AND “exercise” OR “physical activity” OR “sport” OR “weightlifting” | 11/0 | 25/0 | 1/0 | 136/0 |
| Author [56,57,58,59,60,61,62,63,64] | Sample/Diagnostic Criteria | Duration (Months) | Intervention and Comparative Statistical Analysis of the Body Composition | BW (kg or z-Score/%) | BF (kg or % of BW) | FFM (kg/) | LM (kg) | BMI (kg/m2 or % of 95th Percentile or z-Score) | WC (cm) | Changes in Body Composition Mean ± sd or Mean ± (SE) or Mean (CI, 95%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Armeno et al., 2011 [57] | n = 86 IG1: 47 IG2: 39 Girls: 58% Age: 11–19 years old Population: South America (Argentina) Obesity and Insulin Resistance, source of diagnostic criteria: 95th Percentile /NE | 4 | Dietary intervention Within groups: IG1 (low insulin response diet) IG2 (conventional diet) Between groups | YES (kg and z-score) NE NE NS | YES (kg) NE NE NE | NO | NO | YES (kg/m2 andz-score) NE NE NS | YES NE NE p < 0.05 | BW: IG1: 8.9 kg IG2: −6.4 kg IG1: −0.53 ± 0.5 (z-score) IG2: −0.54 ± 0.4 (z-score) BF: IG1: −5.12 kg IG2: NE BMI: IG1: −3.9 kg/m2 IG2: −2.9 kg/m2 IG1: −0.35 ± 0.2 (z-score) IG2: −0.36 ± 0.2 (z-score) WC: IG1: −9.1 ± 4.8 cm IG2: −6.6 ± 4.6 cm |
| Van der Aa et al., 2016 [58] | n = 42 IG1: 23 IG2: 19 Girls: 66% Age: 10–16 years old Population: Europe (The Netherlands) Obesity and Insulin Resistance, source of diagnostic criteria: NE/NE | 18 | Physical exercise intervention, pharmacology Within groups: IG1 (metformin) IG2 (placebo) Between groups | YES (kg) NE NE NE | YES (kg and % of BW) NE NE p < 0.05/NS | YES NE NE p < 0.05 | NO | YES (kg/m2) NE NE p < 0.05 | YES NE NE NE | BW (3): IG1: 1.6 kg (−4.2, 5.9) IG2: 12 kg (2.7, 17) BF (3): IG1: −0.2 kg (−5.2, 2.1) IG2: 2 kg (1.2, 6.4) IG1: −3.1% (−4.8, 0.3) IG2: −0.8% (−3.2, 1.6) FFM (3): IG1: 2.0 kg (−0.1, 4) IG2: 4.5 kg (1.3, 11.6) BMI (3): IG1: 0.2 kg/m2 (−2.9, +1.3) IG2: 1.2 kg/m2 (−0.3, 2.4) WC (3): IG1 and IG2 NE (cm) |
| Garnett et al., 2013 [59] | n = 111 IG1: 55 IG2: 56 Girls: 61% Age: 10–17 years old Population: Oceania (Australia) Overweight and Obesity/Prediabetes and/or Insulin Resistance, source of diagnostic criteria: International Obesity Task Force/ADA/NE | 6 | Dietary and physical exercise intervention, pharmacology Within groups: IG1 (high CH diet) IG2 (low CH diet) Between groups | YES (kg) NE NE NE | NO | NO | NO | YES (% of 95th percentile) p < 0.05 p < 0.05 NS | NO | BW: Total: −3.7 kg (median) BMI: Total NE % 95th percentile |
| Gómez-Díaz et al., 2012 [60] | n = 52 IG1: 28 IG2: 24 Girls: 56% Age: 4–17 years old Population: North America (Mexico) Glucose Intolerance, source of diagnostic criteria: ADA | 3 | Dietary intervention, physical exercise education, pharmacology Within groups: IG1 (metformin) IG2 (placebo) Between groups | YES (kg) /YES p < 0.05 p < 0.05 NS/ p < 0.05 | NO | NO | NO | YES (kg/m2) p < 0.05 p < 0.05 NS | YES p < 0.05 p < 0.05 NS | BW: IG1: −2.7 kg IG1: −5.86% IG2: −1.6 kg IG2: −2.75% BMI: IG1: −4.3 kg/m2 IG2: −1.0 kg/m2 WC: IG1: −9.3 cm IG2: −5.7 cm |
| de Mello et al., 2011 [56] | n = 30 IG1: 15 IG2: 15 Boys: 67% Age: 15–19 years old Population: South America (Brazil) Metabolic Syndrome/ Obesity, source of diagnostic criteria: IDF/>95th Percentile | 12 | Physical exercise, dietary and psychological intervention, clinical therapy Within groups: IG1 (aerobic training) IG2 (aerobic plus resistance training) Between groups | YES (kg) p < 0.05 p < 0.05 p < 0.05 | YES (kg and % of BW) p < 0.05 p < 0.05 p < 0.05 | NO | YES NS NS p < 0.05 | YES (kg/m2)/NO p < 0.05 p < 0.05 p < 0.05 | YES p < 0.05 p < 0.05 p < 0.05 | BW: IG1: −7.91 ± 7.48 kg IG2: −15.45 ± 6.95 kg BF: IG1: −5.67 ± 8.05 kg IG1: −2.87 ± 6.01%IG2: −17.34 ± 6.5 kg IG2: −11.42 ± 6.10% LM: IG1: −2.29 ± 4.01 kg IG2: 2.31 ± 5.22 kg BMI: IG1: −2.62 ± 2.35 kg/m2 IG2: −5.54 ± 2.41 kg/m2 WC: IG1: −5.7 ± 6.37 cm IG2: −17.06 ± 11.38 cm |
| Yanovski et al., 2011 [61] | n = 100 IG1: 53 IG2: 47 Girls: 60% Age: 6–12 years old Population: North America (USA) Obesity/ Insulin resistance, source of diagnostic criteria: ≥ 95th Percentile /NE | 6 | Dietary and physical exercise intervention, pharmacology Within groups: IG1 (metformin) IG2 (placebo) Between groups | YES (kg) NS p < 0.05 p < 0.001 | YES (kg) NS p < 0.05 (1) NS (2) p < 0.05 | NO | NO | YES (kg/m2 and z-score)/NO p < 0.05 NS p < 0.05 | YES NS p < 0.05 p < 0.05 | BW: IG1: 1.47 kg (−0.31, 3.24) IG2: 4.85 kg (2.84, 6.85) BF: IG1: −0.48 kg (−0.8, 1.76) (1) IG2: −1.88 kg (0.44, 3.31) (1) IG1: −1.51 kg (−4.56, 1.54) (2) IG2: 1.81 kg (−1.64, 5.25) (2) BMI: IG1: −0.78 kg/m2 (−1.54, −0.01) IG2: 0.32 kg/m2 (−0.54, 1.18) IG1: −0.11 (−0.16, −0.05) (z-score) IG2: −0.04 (−0.1, 0.02) (z-score) WC: IG1: 1.84 cm (−1, 4.69) IG2: 4.38 cm (1.23, 7.53) |
| Clarson et al., 2009 [62] | n = 25 IG1: 11 IG2: 14 Boys: 56% Age: 10–16 years old Population: North America (Canada) Obesity/Insulin Resistance, source of diagnostic criteria: >95th Percentile /NE | 6 | Physical exercise intervention, dietary education, pharmacology Within groups: IG1 (metformin) IG2 (lifestyle alone) Between groups | NO | NO | NO | NO | YES (kg/m2) p < 0.05 NS p < 0.05 | YES NS NS NS | BMI: IG1: −1.8 ± (0.8) kg/m2 IG2: 0.5 ± (0.3) kg/m2 WC: IG1 and IG2 NE (cm) |
| Atabek et al., 2008 [63] | n = 120 IG1: 90 IG2: 30 Girls: 50% Age: 9–17 years old Population: Europe (Turkey) Obesity/Hyperinsulinemia, source of diagnostic criteria: >95th Percentile /NE | 6 | Dietary and physical exercise intervention pharmacology Within groups: IG1 (metformin) IG2 (placebo) Between groups | YES (kg) p < 0.001 NS NE | NO | NO | NO | YES (kg/m2) p < 0.001 NS p < 0.01 | NO | BW: IG1: −3.4 kg IG2: 3.6 kg BMI: IG1: −2.08 ± 2.32 kg/m2IG2: 0.65 ± 2.5 kg/m2 |
| Love-Osborne et al., 2008 [64] | n = 64 IG1: 48 IG2: 16 Girls: 71% Age: 12–19 years old Population: North America (USA) Obesity/Insulin Resistance, source of diagnostic criteria: >95th Percentile /NE | 6 | Dietary and physical exercise intervention, pharmacology Within groups: IG1 (metformin) IG2 (placebo) Between groups | YES (kg) NE NE NS | NO | NO | NO | YES (kg/m2) NE NE NS | NO | BW: IG1 and IG2: NE (kg) BMI: IG1: −0.16 ± 1.89 kg/m2 IG2: 0.63 ± 1.29 kg/m2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albert Pérez, E.; Mateu Olivares, V.; Martínez-Espinosa, R.M.; Molina Vila, M.D.; Reig García-Galbis, M. New Insights about How to Make an Intervention in Children and Adolescents with Metabolic Syndrome: Diet, Exercise vs. Changes in Body Composition. A Systematic Review of RCT. Nutrients 2018, 10, 878. https://doi.org/10.3390/nu10070878
Albert Pérez E, Mateu Olivares V, Martínez-Espinosa RM, Molina Vila MD, Reig García-Galbis M. New Insights about How to Make an Intervention in Children and Adolescents with Metabolic Syndrome: Diet, Exercise vs. Changes in Body Composition. A Systematic Review of RCT. Nutrients. 2018; 10(7):878. https://doi.org/10.3390/nu10070878
Chicago/Turabian StyleAlbert Pérez, Enrique, Victoria Mateu Olivares, Rosa María Martínez-Espinosa, Mariola D Molina Vila, and Manuel Reig García-Galbis. 2018. "New Insights about How to Make an Intervention in Children and Adolescents with Metabolic Syndrome: Diet, Exercise vs. Changes in Body Composition. A Systematic Review of RCT" Nutrients 10, no. 7: 878. https://doi.org/10.3390/nu10070878
APA StyleAlbert Pérez, E., Mateu Olivares, V., Martínez-Espinosa, R. M., Molina Vila, M. D., & Reig García-Galbis, M. (2018). New Insights about How to Make an Intervention in Children and Adolescents with Metabolic Syndrome: Diet, Exercise vs. Changes in Body Composition. A Systematic Review of RCT. Nutrients, 10(7), 878. https://doi.org/10.3390/nu10070878






