Potential Micronutrients and Phytochemicals against the Pathogenesis of Chronic Obstructive Pulmonary Disease and Lung Cancer
Abstract
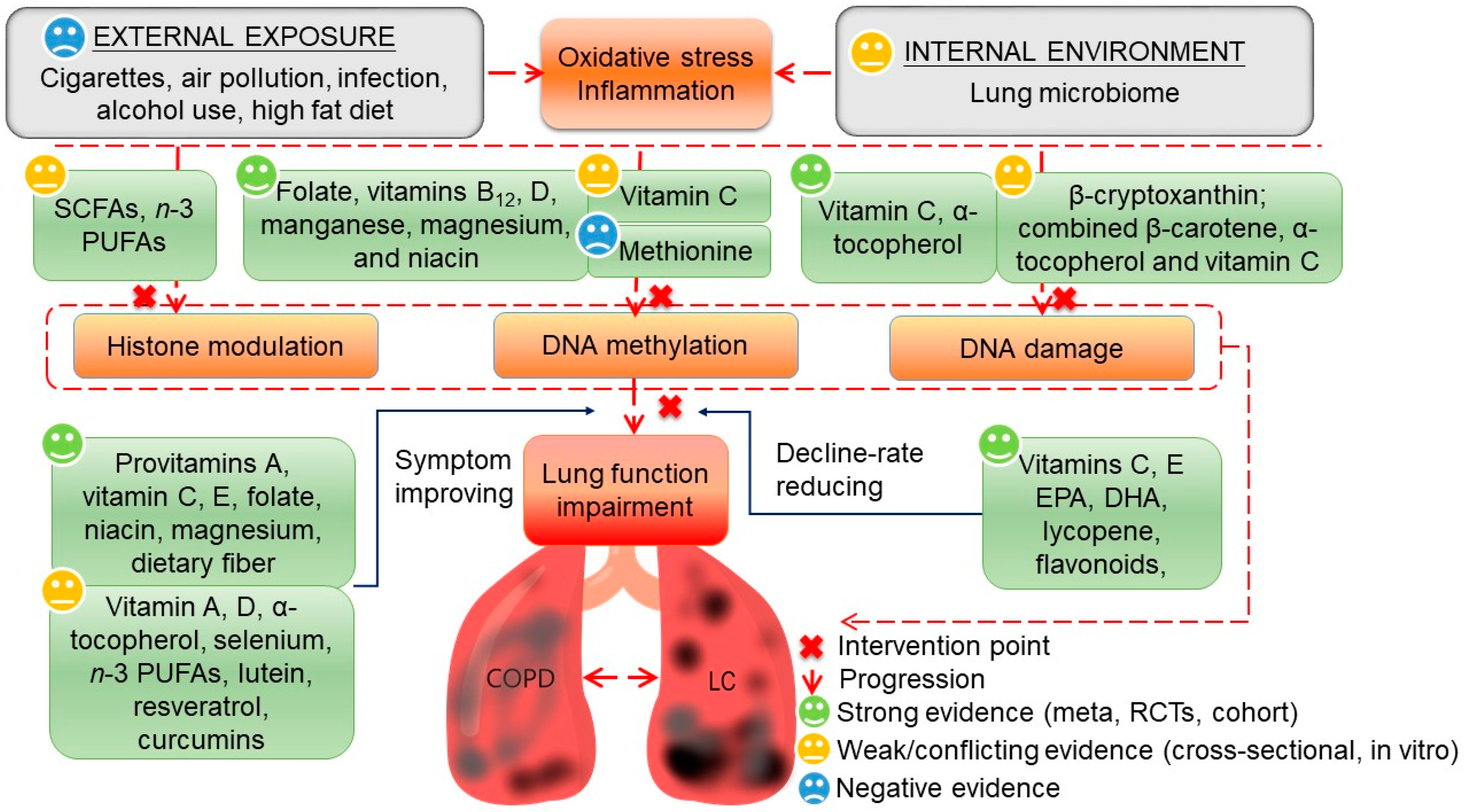
1. Introduction
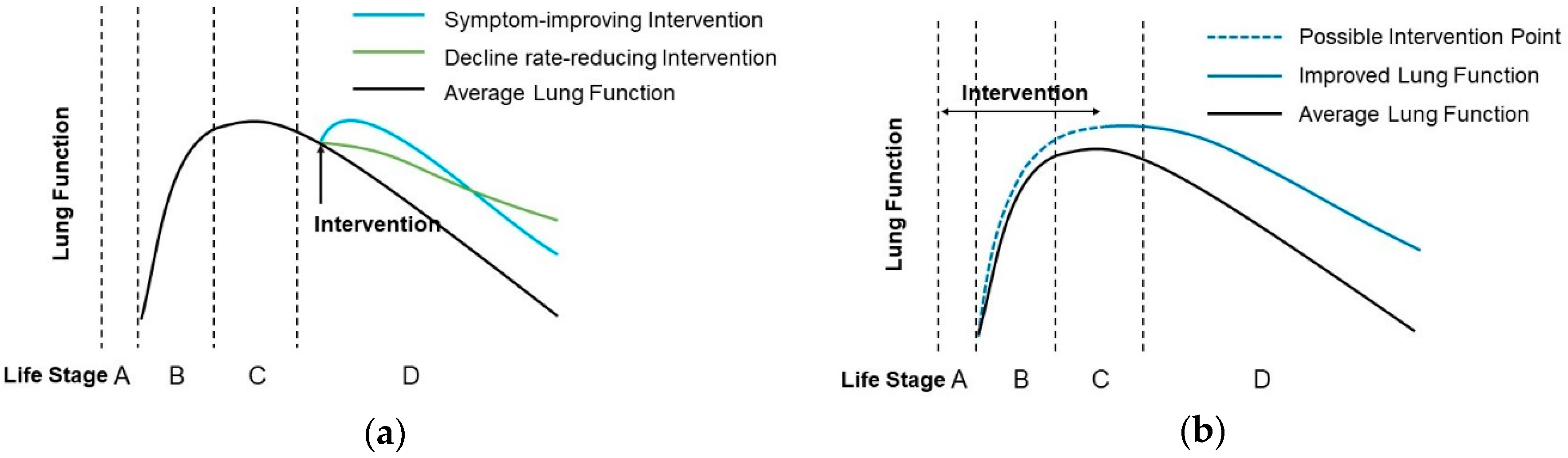
2. Nutrients and Phytochemicals against Lung Function Attenuation during Adulthood and Old Age
2.1. Carotenoids and Vitamin A
2.2. Vitamin C
2.3. Vitamin D
2.4. Vitamin E
2.5. Minerals
2.6. Fatty Acids
2.7. Phytochemicals
3. Dietary Intervention in Early Life
4. Nutrients Intervention in Lung Cancer Chemoprevention
4.1. Nutrients Targeting Epigenetics
4.2. Nutrients Targeting DNA Damage Repair
4.3. Lessons from Supplementation Trials
5. Nutrients and Microbiomes
6. Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Kasala, E.R.; Bodduluru, L.N.; Barua, C.C.; Sriram, C.S.; Gogoi, R. Benzo(a)pyrene induced lung cancer: Role of dietary phytochemicals in chemoprevention. Pharmacol. Rep. 2015, 67, 996–1009. [Google Scholar] [CrossRef] [PubMed]
- WHO. The Top 10 Causes of Death. Available online: http://www.who.int/mediacentre/factsheets/fs310/en/index1.html (accessed on 13 March 2018).
- Papi, A.; Casoni, G.; Caramori, G.; Guzzinati, I.; Boschetto, P.; Ravenna, F.; Calia, N.; Petruzzelli, S.; Corbetta, L.; Cavallesco, G.; et al. COPD increases the risk of squamous histological subtype in smokers who develop non-small cell lung carcinoma. Thorax 2004, 59, 679–681. [Google Scholar] [CrossRef] [PubMed]
- Houghton, A.M. Mechanistic links between COPD and lung cancer. Nat. Rev. Cancer 2013, 13, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Durham, A.L.; Adcock, I.M. The relationship between COPD and lung cancer. Lung Cancer 2015, 90, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Hole, D.J.; Watt, G.C.; Davey-Smith, G.; Hart, C.L.; Gillis, C.R.; Hawthorne, V.M. Impaired lung function and mortality risk in men and women: Findings from the Renfrew and Paisley prospective population study. Br. Med. J. 1996, 313, 711–716. [Google Scholar] [CrossRef]
- Sin, D.D.; Wu, L.; Man, S.F.P. The relationship between reduced lung function and cardiovascular mortality: A population-based study and a systematic review of the literature. Chest 2005, 127, 1952–1959. [Google Scholar] [CrossRef] [PubMed]
- Fry, J.S.; Hamling, J.S.; Lee, P.N. Systematic review with meta-analysis of the epidemiological evidence relating FEV1decline to lung cancer risk. BMC Cancer 2012, 12, 498. [Google Scholar] [CrossRef] [PubMed]
- Wasswa-Kintu, S.; Gan, W.Q.; Man, S.F.P.; Pare, P.D.; Sin, D.D. Relationship between reduced forced expiratory volume in one second and the risk of lung cancer: A systematic review and meta-analysis. Thorax 2005, 60, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Carr, L.L.; Jacobson, S.; Lynch, D.A.; Foreman, M.G.; Flenaugh, E.L.; Hersh, C.P.; Sciurba, F.C.; Wilson, D.O.; Sieren, J.C.; Mulhall, P.; et al. Features of Chronic Obstructive Pulmonary Disease as Predictors of Lung Cancer. Chest 2018, 153, 1326–1335. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.; Diergaarde, B.; Picchi, M.A.; Wilson, D.O.; Gilliland, F.D.; Yuan, J.-M.; Siegfried, J.M.; Belinsky, S.A. Gene Promoter Hypermethylation Detected in Sputum Predicts FEV1 Decline and All-cause Mortality in Smokers. Am. J. Respir. Crit. Care Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- James, A.L.; Palmer, L.J.; Kicic, E.; Maxwell, P.S.; Lagan, S.E.; Ryan, G.F.; Musk, A.W. Decline in lung function in the Busselton Health Study: The effects of asthma and cigarette smoking. Am. J. Respir. Crit. Care Med. 2005, 171, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Salvi, S.S.; Barnes, P.J. Chronic obstructive pulmonary disease in non-smokers. Lancet 2009, 374, 733–743. [Google Scholar] [CrossRef]
- Pelkonen, M.; Notkola, I.L.; Tukiainen, H.; Tervahauta, M.; Tuomilehto, J.; Nissinen, A. Smoking cessation, decline in pulmonary function and total mortality: A 30 year follow up study among the Finnish cohorts of the Seven Countries Study. Thorax 2001, 56, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Lepeule, J.; Litonjua, A.A.; Coull, B.; Koutrakis, P.; Sparrow, D.; Vokonas, P.S.; Schwartz, J. Long-term effects of traffic particles on lung function decline in the elderly. Am. J. Respir. Crit. Care Med. 2014, 190, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Leas, E.C.; Pierce, J.P.; Benmarhnia, T.; White, M.M.; Noble, M.L.; Trinidad, D.R.; Strong, D.R. Effectiveness of Pharmaceutical Smoking Cessation Aids in a Nationally Representative Cohort of American Smokers. J. Natl. Cancer Inst. 2017. [Google Scholar] [CrossRef] [PubMed]
- Reyfman, P.A.; Washko, G.R.; Dransfield, M.T.; Spira, A.; Han, M.K.; Kalhan, R. Defining Impaired Respiratory Health: A Paradigm Shift for Pulmonary Medicine. Am. J. Respir. Crit. Care Med. 2018, 1996–2000. [Google Scholar] [CrossRef] [PubMed]
- Kelly, Y.; Sacker, A.; Marmot, M. Nutrition and respiratory health in adults: Findings from the health survey for Scotland. Eur. Respir. J. 2003, 21, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Potts, J.F.; Omenaas, E.; Heinrich, J.; Svanes, C.; Garcia-Aymerich, J.; Burney, P.G.; Jarvis, D.L. Dietary antioxidants and 10-year lung function decline in adults from the ECRHS survey. Eur. Respir. J. 2017, 50, 1602286. [Google Scholar] [CrossRef] [PubMed]
- Siedlinski, M.; Boer, J.M.A.; Smit, H.A.; Postma, D.S.; Boezen, H.M. Dietary factors and lung function in the general population: Wine and resveratrol intake. Eur. Respir. J. 2012, 39, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Carey, I.M.; Strachan, D.P.; Cook, D.G. Effects of Changes in Fresh Fruit Consumption on Ventilatory Function in Healthy British Adults. Am. J. Respir. Crit. Care Med. 1998, 158, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Baines, K.J.; Backer, V.; Gibson, P.G.; Powell, H.; Porsbjerg, C.M. Investigating the effects of arctic dietary intake on lung health. Eur. J. Clin. Nutr. 2015, 69, 1262–1266. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.; Paik, D.C.; Hankinson, J.L.; Barr, R.G. Cured meat consumption, lung function, and chronic obstructive pulmonary disease among United States adults. Am. J. Respir. Crit. Care Med. 2007, 175, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Kamholz, S. Wine, Spirits and the Lung: Good, Bad or Indifferent? Trans. Am. Clin. Climatol. Assoc. 2006, 117, 129–145. [Google Scholar] [PubMed]
- McKeever, T.M.; Lewis, S.A.; Cassano, P.A.; Ocke, M.; Burney, P.; Britton, J.; Smit, H.A. Patterns of dietary intake and relation to respiratory disease, forced expiratory volume in 1 s, and decline in 5-y forced expiratory volume. Am. J. Clin. Nutr. 2010, 92, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Sorli-Aguilar, M.; Martin-Lujan, F.; Flores-Mateo, G.; Arija-Val, V.; Basora-Gallisa, J.; Sola-Alberich, R. Dietary patterns are associated with lung function among Spanish smokers without respiratory disease. BMC Pulm. Med. 2016, 16, 162. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.O.; Jameson, K.A.; Syddall, H.E.; Aihie Sayer, A.; Dennison, E.M.; Cooper, C.; Robinson, S.M. The relationship of dietary patterns with adult lung function and COPD. Eur. Respir. J. 2010, 36, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Smit, H.A. Chronic obstructive pulmonary disease, asthma and protective effects of food intake: From hypothesis to evidence? Respir. Res. 2001, 2, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Hanson, C.; Rutten, E.P.A.; Wouters, E.F.M.; Rennard, S. Diet and vitamin D as risk factors for lung impairment and COPD. Transl. Res. 2013, 162, 219–236. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.; Picchi, M.A.; Tesfaigzi, Y.; Wu, G.; James Gauderman, W.; Xu, F.; Gilliland, F.D.; Belinsky, S.A. Dietary nutrients associated with preservation of lung function in hispanic and Non-Hispanic white smokers from New Mexico. Int. J. Chronic Obstr. Pulm. Dis. 2017, 12, 3171–3181. [Google Scholar] [CrossRef] [PubMed]
- McKeever, T.M.; Lewis, S.A.; Smit, H.A.; Burney, P.; Cassano, P.A.; Britton, J. A multivariate analysis of serum nutrient levels and lung function. Respir. Res. 2008, 9, 67. [Google Scholar] [CrossRef] [PubMed]
- Chuwers, P.; Barnhart, S.; Blanc, P.; Brodkin, C.A.; Cullen, M.; Kelly, T.; Keogh, J.; Omenn, G.; Williams, J.; Balmes, J.R. The protective effect of beta-carotene and retinol on ventilatory function in an asbestos-exposed cohort. Am. J. Respir. Crit. Care Med. 1997, 155, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- Thyagarajan, B.; Meyer, K.A.; Smith, L.J.; Beckett, W.S.; Williams, O.D.; Gross, M.D.; Jacobs, D.R., Jr. Serum carotenoid concentrations predict lung function evolution in young adults: The Coronary Artery Risk Development in Young Adults (CARDIA) study. Am. J. Clin. Nutr. 2011, 94, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Guénégou, A.; Leynaert, B.; Pin, I.; Le Moël, G.; Zureik, M.; Neukirch, F. Serum carotenoids, vitamins A and E, and 8 year lung function decline in a general population. Thorax 2006, 61, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Van Lent, D.M.; Leermakers, E.T.M.; Darweesh, S.K.L.; Moreira, E.M.; Tielemans, M.J.; Muka, T.; Vitezova, A.; Chowdhury, R.; Bramer, W.M.; Brusselle, G.G.; et al. The effects of lutein on respiratory health across the life course: A systematic review. Clin. Nutr. ESPEN 2016, 13, e1–e7. [Google Scholar] [CrossRef] [PubMed]
- Van Lent, D.M.; Leermakers, E.T.M.; Hofman, A.; Stricker, B.H.; Brusselle, G.G.; Franco, O.H.; Lahousse, L.; Kiefte-de Jong, J.C. Association between lutein intake and lung function in adults: The Rotterdam Study. Br. J. Nutr. 2017, 117, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Cassano, P.A. Antioxidant nutrients and pulmonary function: The Third National Health and Nutrition Examination Survey (NHANES III). Am. J. Epidemiol. 2000, 151, 975–981. [Google Scholar] [CrossRef] [PubMed]
- Palozza, P.; Serini, S.; Trombino, S.; Lauriola, L.; Ranelletti, F.O.; Calviello, G. Dual role of beta-carotene in combination with cigarette smoke aqueous extract on the formation of mutagenic lipid peroxidation products in lung membranes: Dependence on pO2. Carcinogenesis 2006, 27, 2383–2391. [Google Scholar] [CrossRef] [PubMed]
- Omenn, G.S. Chemoprevention of lung cancers: Lessons from CARET, the beta-carotene and retinol efficacy trial, and prospects for the future. Eur. J. Cancer Prev. 2007, 16, 184–191. [Google Scholar] [CrossRef] [PubMed]
- McKeever, T.M.; Scrivener, S.; Broadfield, E.; Jones, Z.; Britton, J.; Lewis, S.A. Prospective study of diet and decline in lung function in a general population. Am. J. Respir. Crit. Care Med. 2002, 165, 1299–1303. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.J.; Grant, B.J.; Freudenheim, J.L.; Muti, P.; Browne, R.W.; Drake, J.A.; Klocke, R.A.; Trevisan, M. The relation of serum levels of antioxidant vitamins C and E, retinol and carotenoids with pulmonary function in the general population. Am. J. Respir. Crit. Care Med. 2001, 163, 1246–1255. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Tunstall-Pedoe, H.; Bolton-Smith, C.; Hannah, M.K.; Morrison, C. Association of dietary antioxidants and waist circumference with pulmonary function and airway obstruction. Am. J. Epidemiol. 2001, 153, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.S. Vitamin a: History, current uses, and controversies. Semin. Cutan. Med. Surg. 2012, 31, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Redlich, C.A.; Grauer, J.N.; Van Bennekum, A.M.; Clever, S.L.; Ponn, R.B.; Blaner, W.S. Characterization of carotenoid, vitamin A, and alpha-tocopheral levels in human lung tissue and pulmonary macrophages. Am. J. Respir. Crit. Care Med. 1996, 154, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Tsiligianni, I.G.; van der Molen, T. A systematic review of the role of vitamin insufficiencies and supplementation in COPD. Respir. Res. 2010, 11, 171. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Kim, W.J.; Lee, S.A. The effect of dietary antioxidant on the COPD risk: The community-based KoGES (Ansan–Anseong) cohort. Int. J. COPD 2015, 10, 2159–2168. [Google Scholar] [CrossRef]
- Bentley, A.R.; Kritchevsky, S.B.; Harris, T.B.; Holvoet, P.; Jensen, R.L.; Newman, A.B.; Lee, J.S.; Yende, S.; Bauer, D.; Cassano, P.A. Dietary antioxidants and forced expiratory volume in 1 s decline: The Health, Aging and Body Composition study. Eur. Respir. J. 2012, 39, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.O.; Jameson, K.A.; Robinson, S.M.; Boucher, B.J.; Syddall, H.E.; Sayer, A.A.; Cooper, C.; Holloway, J.W.; Dennison, E.M. Relationship of vitamin D status to adult lung function and COPD. Thorax 2011, 66, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Lange, N.E.; Sparrow, D.; Vokonas, P.; Litonjua, A.A. Vitamin D deficiency, smoking, and lung function in the normative aging study. Am. J. Respir. Crit. Care Med. 2012, 186, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; Chang, S.S.; Sun, K.; Cappola, A.R.; Ferrucci, L.; Fried, L.P. Serum 25-hydroxyvitamin D and pulmonary function in older disabled community-dwelling women. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2012, 67, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Millen, A.E.; Bodnar, L.M. Vitamin D assessment in population-based studies: A review of the issues. Am. J. Clin. Nutr. 2008, 87, 1102S–1105S. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.W.; Joyce, A.; Ingold, K.U. First proof that vitamin E is major lipid-soluble, chain-breaking antioxidant in human blood plasma. Lancet 1982, 2, 327. [Google Scholar] [CrossRef]
- Meydani, M.; Cohn, J.S.; Macauley, J.B.; McNamara, J.R.; Blumberg, J.B.; Schaefer, E.J. Postprandial changes in the plasma concentration of alpha- and gamma-tocopherol in human subjects fed a fat-rich meal supplemented with fat-soluble vitamins. J. Nutr. 1989, 119, 1252–1258. [Google Scholar] [CrossRef] [PubMed]
- Hanson, C.; Lyden, E.; Furtado, J.; Campos, H.; Sparrow, D.; Vokonas, P.; Litonjua, A.A. Serum tocopherol levels and vitamin E intake are associated with lung function in the normative aging study. Clin. Nutr. 2016, 35, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.J.; McCann, S.; Grant, B.J.B.; Trevisan, M.; Muti, P.; Freudenheim, J.L. Lung function in relation to intake of carotenoids and other antioxidant vitamins in a population-based study. Am. J. Epidemiol. 2002, 155, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Cassano, P.A.; Guertin, K.A.; Kristal, A.R.; Ritchie, K.E.; Bertoia, M.L.; Arnold, K.B.; Crowley, J.J.; Hartline, J.; Goodman, P.J.; Tangen, C.M.; et al. A randomized controlled trial of vitamin E and selenium on rate of decline in lung function. Respir. Res. 2015, 16, 35. [Google Scholar] [CrossRef] [PubMed]
- Berdnikovs, S.; Abdala-Valencia, H.; McCary, C.; Somand, M.; Cole, R.; Garcia, A.; Bryce, P.; Cook-Mills, J.M. Isoforms of vitamin E have opposing immunoregulatory functions during inflammation by regulating leukocyte recruitment. J. Immunol. 2009, 182, 4395–4405. [Google Scholar] [CrossRef] [PubMed]
- Grievink, L.; Smit, H.A.; Veer, P.; Brunekreef, B.; Kromhout, D. Plasma concentrations of the antioxidants beta-carotene and alpha-tocopherol in relation to lung function. Eur. J. Clin. Nutr. 1999, 53, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Grievink, L.; de Waart, F.G.; Schouten, E.G.; Kok, F.J. Serum carotenoids, alpha-tocopherol, and lung function among Dutch elderly. Am. J. Respir. Crit. Care Med. 2000, 161, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Bivona, J.J., 3rd; Patel, S.; Vajdy, M. Induction of cellular and molecular Immunomodulatory pathways by vitamin E and vitamin C. Expert Opin. Biol. Ther. 2017, 17, 1539–1551. [Google Scholar] [CrossRef] [PubMed]
- Britton, J.R.; Pavord, I.D.; Richards, K.A.; Knox, A.J.; Wisniewski, A.F.; Lewis, S.A.; Tattersfield, A.E.; Weiss, S.T. Dietary antioxidant vitamin intake and lung function in the general population. Am. J. Respir. Crit. Care Med. 1995, 151, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- Pearson, P.; Britton, J.; McKeever, T.; Lewis, S.A.; Weiss, S.; Pavord, I.; Fogarty, A. Lung function and blood levels of copper, selenium, vitamin C and vitamin E in the general population. Eur. J. Clin. Nutr. 2005, 59, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G.; Attia, J.; McElduff, P.; McEvoy, M.; Gibson, P.G. Assessment of dietary fat intake and innate immune activation as risk factors for impaired lung function. Eur. J. Clin. Nutr. 2010, 64, 818–825. [Google Scholar] [CrossRef] [PubMed]
- Best, K.P.; Gold, M.; Kennedy, D.; Martin, J.; Makrides, M. Omega-3 long-chain PUFA intake during pregnancy and allergic disease outcomes in the offspring: A systematic review and meta-analysis of observational studies and randomized controlled trials. Am. J. Clin. Nutr. 2016, 103, 128–143. [Google Scholar] [CrossRef] [PubMed]
- Willemsen, L.E.M. Dietary n-3 long chain polyunsaturated fatty acids in allergy prevention and asthma treatment. Eur. J. Pharmacol. 2016, 785, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Pontes-Arruda, A.; DeMichele, S.; Seth, A.; Singer, P. The use of an inflammation-modulating diet in patients with acute lung injury or acute respiratory distress syndrome: A meta-analysis of outcome data. J. Parenter. Enter. Nutr. 2008, 32, 596–605. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Zhang, Y.; Li, S.; Gan, L.; Feng, H.; Nie, W. Enteral omega-3 fatty acid supplementation in adult patients with acute respiratory distress syndrome: A systematic review of randomized controlled trials with meta-analysis and trial sequential analysis. Intensive Care Med. 2014, 40, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Amigo, H.; Bustos, P.; Bakolis, I.; Rona, R.J. Ventilatory function in young adults and dietary antioxidant intake. Nutrients 2015, 7, 2879–2896. [Google Scholar] [CrossRef] [PubMed]
- Lago, J.H.G.; Toledo-Arruda, A.C.; Mernak, M.; Barrosa, K.H.; Martins, M.A.; Tiberio, I.F.L.C.; Prado, C.M. Structure-activity association of flavonoids in lung diseases. Molecules 2014, 19, 3570–3595. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.J.; Cassidy, A.; Litonjua, A.A.; Sparrow, D.; Vokonas, P.; Schwartz, J. Dietary anthocyanin intake and age-related decline in lung function: Longitudinal findings from the VA normative aging study. Am. J. Clin. Nutr. 2016, 103, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Thawer, N.; Charles, D.; Cassidy, A.; Van Zele, T.; Thilsing, T.; Ahlström, M.; Haahtela, T.; Keil, T.; Matricardi, P.M.; et al. Dietary intake of flavonoids and ventilatory function in European adults: A GA2LEN study. Nutrients 2018, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.P.; Niti, M.; Yap, K.B.; Tan, W.C. Curcumins-rich curry diet and pulmonary function in Asian older adults. PLoS ONE 2012, 7, e51753. [Google Scholar] [CrossRef] [PubMed]
- Tabak, C.; Smit, H.A.; Räsänen, L.; Fidanza, F.; Menotti, A.; Nissinen, A.; Feskens, E.J.M.; Heederik, D.; Kromhout, D. Alcohol consumption in relation to 20-year COPD mortality and pulmonary function in middle-aged men from three European countries. Epidemiology 2001, 12, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Preedy, V.R. Free radicals as mediators of alcohol toxicity. Adverse Drug React. Toxicol. Rev. 1999, 18, 235–252. [Google Scholar] [PubMed]
- Maxwell, S.; Cruickshank, A.; Thorpe, G. Red wine and antioxidant activity in serum. Lancet 1994, 344, 193–194. [Google Scholar] [CrossRef]
- Fauconneau, B.; Waffo-Teguo, P.; Huguet, F.; Barrier, L.; Decendit, A.; Merillon, J.M. Comparative study of radical scavenger and antioxidant properties of phenolic compounds from Vitis vinifera cell cultures using in vitro tests. Life Sci. 1997, 61, 2103–2110. [Google Scholar] [CrossRef]
- Schünemann, H.J.; Grant, B.J.B.; Freudenheim, J.L.; Muti, P.; McCann, S.E.; Kudalkar, D.; Ram, M.; Nochajski, T.; Russell, M.; Trevisan, M. Beverage specific alcohol intake in a population-based study: Evidence for a positive association between pulmonary function and wine intake. BMC Pulm. Med. 2002, 2, 3. [Google Scholar] [CrossRef]
- Viegi, G.; Scognamiglio, A.; Baldacci, S.; Pistelli, F.; Carrozzi, L. Epidemiology of chronic obstructive pulmonary disease (COPD). Respiration 2001, 68, 4–19. [Google Scholar] [CrossRef] [PubMed]
- Lange, P.; Celli, B.; Agustí, A.; Boje Jensen, G.; Divo, M.; Faner, R.; Guerra, S.; Marott, J.L.; Martinez, F.D.; Martinez-Camblor, P.; et al. Lung-Function Trajectories Leading to Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2015, 373, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Svanes, C.; Sunyer, J.; Plana, E.; Dharmage, S.; Heinrich, J.; Jarvis, D.; De Marco, R.; Norbäck, D.; Raherison, C.; Villani, S.; et al. Early life origins of chronic obstructive pulmonary disease. Thorax 2010, 65, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Gauderman, W.J.; Urman, R.; Avol, E.; Berhane, K.; McConnell, R.; Rappaport, E.; Chang, R.; Lurmann, F.; Gilliland, F. Association of improved air quality with lung development in children. N. Engl. J. Med. 2015, 372, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Stern, D.A.; Morgan, W.J.; Wright, A.L.; Guerra, S.; Martinez, F.D. Poor airway function in early infancy and lung function by age 22 years: A non-selective longitudinal cohort study. Lancet 2007, 370, 758–764. [Google Scholar] [CrossRef]
- McEvoy, C.T.; Spindel, E.R. Pulmonary Effects of Maternal Smoking on the Fetus and Child: Effects on Lung Development, Respiratory Morbidities, and Life Long Lung Health. Paediatr. Respir. Rev. 2017, 21, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Sarginson, J.E.; Killen, J.D.; Lazzeroni, L.C.; Fortmann, S.P.; Ryan, H.S.; Schatzberg, A.F.; Murphy, G.M.J. Markers in the 15q24 nicotinic receptor subunit gene cluster (CHRNA5-A3-B4) predict severity of nicotine addiction and response to smoking cessation therapy. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2011, 156, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.; Huy, C.; Schutz, J.; Diehl, K. Smoking cessation during pregnancy: A systematic literature review. Drug Alcohol Rev. 2010, 29, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.G.; Carey, I.M.; Whincup, P.H.; Papacosta, O.; Chirico, S.; Bruckdorfer, K.R.; Walker, M. Effect of fresh fruit consumption on lung function and wheeze in children. Thorax 1997, 52, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-Y.; Forno, E.; Alvarez, M.; Colon-Semidey, A.; Acosta-Perez, E.; Canino, G.; Celedon, J.C. Diet, Lung Function, and Asthma Exacerbations in Puerto Rican Children. Pediatr. Allergy Immunol. Pulmonol. 2017, 30, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Gilliland, F.D.; Berhane, K.T.; Li, Y.-F.; Kim, D.H.; Margolis, H.G. Dietary magnesium, potassium, sodium, and children’s lung function. Am. J. Epidemiol. 2002, 155, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Checkley, W.; Keith, P.; West, J.; Wise, R.A.; Baldwin, M.R.; Wu, L.; LeClerq, S.C.; Christian, P.; Katz, J.; Tielsch, J.M.; et al. Maternal Vitamin A Supplementation and Lung Function in Offspring. N. Engl. J. Med. 2010, 362, 1784–1794. [Google Scholar] [CrossRef] [PubMed]
- McEvoy, C.T.; Schilling, D.; Clay, N.; Jackson, K.; Go, M.D.; Spitale, P.; Bunten, C.; Leiva, M.; Gonzales, D.; Hollister-Smith, J.; et al. Vitamin C supplementation for pregnant smoking women and pulmonary function in their newborn infants: A randomized clinical trial. JAMA 2014, 311, 2074–2082. [Google Scholar] [CrossRef] [PubMed]
- Shorey-Kendrick, L.E.; McEvoy, C.T.; Ferguson, B.; Burchard, J.; Park, B.S.; Gao, L.; Vuylsteke, B.H.; Milner, K.F.; Morris, C.D.; Spindel, E.R. Vitamin C Prevents Offspring DNA Methylation Changes Associated with Maternal Smoking in Pregnancy. Am. J. Respir. Crit. Care Med. 2017, 196, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Waidyatillake, N.T.; Stoney, R.; Thien, F.; Lodge, C.J.; Simpson, J.A.; Allen, K.J.; Abramson, M.J.; Erbas, B.; Svanes, C.; Dharmage, S.C.; et al. Breast milk polyunsaturated fatty acids: Associations with adolescent allergic disease and lung function. Allergy 2017, 72, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Bédard, A.; Northstone, K.; Henderson, A.J.; Shaheen, S.O. Maternal intake of sugar during pregnancy and childhood respiratory and atopic outcomes. Eur. Respir. J. 2017, 50, 1700073. [Google Scholar] [CrossRef] [PubMed]
- Hulbert, A.; Jusue-Torres, I.; Stark, A.; Chen, C.; Rodgers, K.; Lee, B.; Griffin, C.; Yang, A.; Huang, P.; Wrangle, J.; et al. Early Detection of Lung Cancer Using DNA Promoter Hypermethylation in Plasma and Sputum. Clin. Cancer Res. 2017, 23, 1998–2005. [Google Scholar] [CrossRef] [PubMed]
- Duruisseaux, M.; Esteller, M. Lung cancer epigenetics: From knowledge to applications. Semin. Cancer Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Mari-Alexandre, J.; Diaz-Lagares, A.; Villalba, M.; Juan, O.; Crujeiras, A.B.; Calvo, A.; Sandoval, J. Translating cancer epigenomics into the clinic: Focus on lung cancer. Transl. Res. 2017, 189, 76–92. [Google Scholar] [CrossRef] [PubMed]
- Camarena, V.; Wang, G. The epigenetic role of vitamin C in health and disease. Cell. Mol. Life Sci. 2016, 73, 1645–1658. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Karlsson, O.; Wang, G.; Li, J.; Guo, Y.; Lin, X.; Zemplenyi, M.; Sanchez-Guerra, M.; Trevisi, L.; Urch, B.; et al. B vitamins attenuate the epigenetic effects of ambient fine particles in a pilot human intervention trial. Proc. Natl. Acad. Sci. USA 2017, 114, 3503–3508. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Perkins, J.T.; Hennig, B. EGCG prevents PCB 126-induced endothelial cell inflammation via epigenetic modifications of NF-κB target genes in human endothelial cells. J. Nutr. Biochem. 2016, 28, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Shankar, E.; Kanwal, R.; Candamo, M.; Gupta, S. Dietary phytochemicals as epigenetic modifiers in cancer: Promise and challenges. Semin. Cancer Biol. 2016, 40–41, 82–99. [Google Scholar] [CrossRef] [PubMed]
- Crider, K.S.; Yang, T.P.; Berry, R.J.; Bailey, L.B. Folate and DNA methylation: A review of molecular mechanisms and the evidence for folate’s role. Adv. Nutr. 2012, 3, 21–38. [Google Scholar] [CrossRef] [PubMed]
- Stidley, C.A.; Picchi, M.A.; Leng, S.; Willink, R.; Crowell, R.E.; Flores, K.G.; Kang, H.; Byers, T.; Gilliland, F.D.; Belinsky, S.A. Multivitamins, folate, and green vegetables protect against gene promoter methylation in the aerodigestive tract of smokers. Cancer Res. 2010, 70, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.; Picchi, M.A.; Kang, H.; Wu, G.; Filipczak, P.T.; Juri, D.E.; Zhang, X.; Gauderman, W.J.; Gilliland, F.D.; Belinsky, S.A. Dietary nutrient intake, ethnicity, and epigenetic silencing of lung cancer genes detected in sputum in New Mexican smokers. Cancer Prev. Res. 2018, 11, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Vineis, P.; Chuang, S.-C.; Vaissière, T.; Cuenin, C.; Ricceri, F.; Collaborators, G.; Johansson, M.; Ueland, P.; Brennan, P.; Herceg, Z. DNA methylation changes associated with cancer risk factors and blood levels of vitamin metabolites in a prospective study. Epigenetics 2011, 6, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Piyathilake, C.J.; Bell, W.C.; Johanning, G.L.; Cornwell, P.E.; Heimburger, D.C.; Grizzle, W.E. The accumulation of ascorbic acid by squamous cell carcinomas of the lung and larynx is associated with global methylation of DNA. Cancer 2000, 89, 171–176. [Google Scholar] [CrossRef]
- Piyathilake, C.J.; Johanning, G.L.; Macaluso, M.; Whiteside, M.; Oelschlager, D.K.; Heimburger, D.C.; Grizzle, W.E. Localized folate and vitamin B-12 deficiency in squamous cell lung cancer is associated with global DNA hypomethylation. Nutr. Cancer 2000, 37, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Eckschlager, T.; Plch, J.; Stiborova, M.; Hrabeta, J. Histone Deacetylase Inhibitors as Anticancer Drugs. Int. J. Mol. Sci. 2017, 18, 1414. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Wang, C.; Xia, L.; Fan, C.; Dong, H.; Deckelbaum, R.J.; Qi, K. Epigenetic modification of the leptin promoter in diet-induced obese mice and the effects of N-3 polyunsaturated fatty acids. Sci. Rep. 2014, 4, 5282. [Google Scholar] [CrossRef] [PubMed]
- Pai, J.-T.; Hsu, C.-Y.; Hua, K.-T.; Yu, S.-Y.; Huang, C.-Y.; Chen, C.-N.; Liao, C.-H.; Weng, M.-S. NBM-T-BBX-OS01, Semisynthesized from Osthole, Induced G1 Growth Arrest through HDAC6 Inhibition in Lung Cancer Cells. Molecules 2015, 20, 8000–8019. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.; Trac, C.; Du, J.; Natarajan, R.; Schones, D.E. Persistent Chromatin Modifications Induced by High Fat Diet. J. Biol. Chem. 2016, 291, 10446–10455. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xu, G.B.; Zhou, D.; Pan, Y.-X. High-fat diet modifies expression of hepatic cellular senescence gene p16(INK4a) through chromatin modifications in adult male rats. Genes Nutr. 2018, 13, 6. [Google Scholar] [CrossRef] [PubMed]
- Licciardi, P.V.; Ververis, K.; Karagiannis, T.C. Histone deacetylase inhibition and dietary short-chain Fatty acids. ISRN Allergy 2011, 2011, 869647. [Google Scholar] [CrossRef] [PubMed]
- Tesei, A.; Brigliadori, G.; Carloni, S.; Fabbri, F.; Ulivi, P.; Arienti, C.; Sparatore, A.; Del Soldato, P.; Pasini, A.; Amadori, D.; et al. Organosulfur derivatives of the HDAC inhibitor valproic acid sensitize human lung cancer cell lines to apoptosis and to cisplatin cytotoxicity. J. Cell. Physiol. 2012, 227, 3389–3396. [Google Scholar] [CrossRef] [PubMed]
- Mooney, L.A.; Madsen, A.M.; Tang, D.; Orjuela, M.A.; Tsai, W.-Y.; Garduno, E.R.; Perera, F.P. Antioxidant vitamin supplementation reduces benzo(a)pyrene-DNA adducts and potential cancer risk in female smokers. Cancer Epidemiol. Biomark. Prev. 2005, 14, 237–242. [Google Scholar]
- Liu, C.; Bronson, R.T.; Russell, R.M.; Wang, X.-D. beta-Cryptoxanthin supplementation prevents cigarette smoke-induced lung inflammation, oxidative damage, and squamous metaplasia in ferrets. Cancer Prev. Res. 2011, 4, 1255–1266. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lian, F.; Yeum, K.-J.; Chongviriyaphan, N.; Choi, S.-W.; Russell, R.M.; Wang, X.-D. The effects of combined antioxidant (beta-carotene, alpha-tocopherol and ascorbic acid) supplementation on antioxidant capacity, DNA single-strand breaks and levels of insulin-like growth factor-1/IGF-binding protein 3 in the ferret model of lung cancer. Int. J. Cancer 2007, 120, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Buchner, F.L.; Bueno-de-Mesquita, H.B.; Ros, M.M.; Overvad, K.; Dahm, C.C.; Hansen, L.; Tjonneland, A.; Clavel-Chapelon, F.; Boutron-Ruault, M.-C.; Touillaud, M.; et al. Variety in fruit and vegetable consumption and the risk of lung cancer in the European prospective investigation into cancer and nutrition. Cancer Epidemiol. Biomark. Prev. 2010, 19, 2278–2286. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.T.; Lai, G.Y.; Liao, L.M.; Subar, A.F.; Bertazzi, P.A.; Pesatori, A.C.; Freedman, N.D.; Landi, M.T.; Lam, T.K. Nut Consumption and Lung Cancer Risk: Results from Two Large Observational Studies. Cancer Epidemiol. Biomark. Prev. 2017, 26, 826–836. [Google Scholar] [CrossRef] [PubMed]
- Seow, A.; Koh, W.-P.; Wang, R.; Lee, H.-P.; Yu, M.C. Reproductive variables, soy intake, and lung cancer risk among nonsmoking women in the Singapore Chinese Health Study. Cancer Epidemiol. Biomark. Prev. 2009, 18, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Balder, H.F.; Goldbohm, R.A.; van den Brandt, P.A. Dietary patterns associated with male lung cancer risk in The Netherlands Cohort Study. Cancer Epidemiol. Biomark. Prev. 2005, 14, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Guo, Y.; Di, Q.; Zheng, Y.; Xian, H.; Li, X.; Liu, T.; Xiao, J.; Zeng, W.; Howard, S.W.; et al. Consumption of fruit and vegetables might mitigate the adverse effects of ambient PM2.5 on lung function among adults. Environ. Res. 2018, 160, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Fanidi, A.; Muller, D.C.; Yuan, J.-M.; Stevens, V.L.; Weinstein, S.J.; Albanes, D.; Prentice, R.; Thomsen, C.A.; Pettinger, M.; Cai, Q.; et al. Circulating Folate, Vitamin B6, and Methionine in Relation to Lung Cancer Risk in the Lung Cancer Cohort Consortium (LC3). J. Natl. Cancer Inst. 2018, 110. [Google Scholar] [CrossRef]
- Shen, H.; Wei, Q.; Pillow, P.C.; Amos, C.I.; Hong, W.K.; Spitz, M.R. Dietary folate intake and lung cancer risk in former smokers: A case-control analysis. Cancer Epidemiol. Biomark. Prev. 2003, 12, 980–986. [Google Scholar]
- Männistö, S.; Smith-Warner, S.A.; Spiegelman, D.; Albanes, D.; Anderson, K.; Van Den Brandt, P.A.; Cerhan, J.R.; Colditz, G.; Feskanich, D.; Freudenheim, J.L.; et al. Dietary Carotenoids and Risk of Lung Cancer in a Pooled Analysis of Seven Cohort Studies. Cancer Epidemiol. Biomark. Prev. 2004, 13, 40–48. [Google Scholar] [CrossRef]
- Cheng, T.-Y.D.; Neuhouser, M.L. Serum 25-hydroxyvitamin D, vitamin A, and lung cancer mortality in the US population: A potential nutrient-nutrient interaction. Cancer Causes Control 2012, 23, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Martínez, M.E.; Jacobs, E.T.; Baron, J.A.; Marshall, J.R.; Byers, T. Dietary supplements and cancer prevention: Balancing potential benefits against proven harms. J. Natl. Cancer Inst. 2012, 104, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Gahche, J.; Bailey, R.; Burt, V.; Hughes, J.; Yetley, E.; Dwyer, J.; Picciano, M.F.; McDowell, M.; Sempos, C. Dietary supplement use among U.S. adults has increased since NHANES III (1988–1994). NCHS Data Brief 2011, 61, 1–8. [Google Scholar]
- Hilty, M.; Burke, C.; Pedro, H.; Cardenas, P.; Bush, A.; Bossley, C.; Davies, J.; Ervine, A.; Poulter, L.; Pachter, L.; et al. Disordered microbial communities in asthmatic airways. PLoS ONE 2010, 5, e8578. [Google Scholar] [CrossRef] [PubMed]
- Sze, M.A.; Hogg, J.C.; Sin, D.D. Bacterial microbiome of lungs in COPD. Int. J. Chronic Obstr. Pulm. Dis. 2014, 9, 229–238. [Google Scholar] [CrossRef]
- Winter, S.E.; Baumler, A.J. Why related bacterial species bloom simultaneously in the gut: Principles underlying the “Like will to like” concept. Cell. Microbiol. 2014, 16, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Bernasconi, E.; Pattaroni, C.; Koutsokera, A.; Pison, C.; Kessler, R.; Benden, C.; Soccal, P.M.; Magnan, A.; Aubert, J.-D.; Marsland, B.J.; et al. Airway Microbiota Determines Innate Cell Inflammatory or Tissue Remodeling Profiles in Lung Transplantation. Am. J. Respir. Crit. Care Med. 2016, 194, 1252–1263. [Google Scholar] [CrossRef] [PubMed]
- Huffnagle, G.B.; Dickson, R.P.; Lukacs, N.W. The respiratory tract microbiome and lung inflammation: A two-way street. Mucosal Immunol. 2017, 10, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Kanner, R.E.; Anthonisen, N.R.; Connett, J.E. Lower respiratory illnesses promote FEV(1) decline in current smokers but not ex-smokers with mild chronic obstructive pulmonary disease: Results from the lung health study. Am. J. Respir. Crit. Care Med. 2001, 164, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, G.C.; Seemungal, T.A.R.; Bhowmik, A.; Wedzicha, J.A. Relationship between exacerbation frequency and lung function decline in chronic obstructive pulmonary disease. Thorax 2002, 57, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Roggenbuck, M.; Anderson, D.; Barfod, K.K.; Feelisch, M.; Geldenhuys, S.; Sørensen, S.J.; Weeden, C.E.; Hart, P.H.; Gorman, S. Vitamin D and allergic airway disease shape the murine lung microbiome in a sex-specific manner. Respir. Res. 2016, 17, 116. [Google Scholar] [CrossRef] [PubMed]
- Vargas, A.J.; Harris, C.C. Biomarker development in the precision medicine era: Lung cancer as a case study. Nat. Rev. Cancer 2016, 16, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Wang, C. Surviving With Smog and Smoke: Precision Interventions? Chest 2017, 152, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Murgia, C.; Adamski, M.M. Translation of nutritional genomics into nutrition practice: The next step. Nutrients 2017, 9, 366. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhai, T.; Li, S.; Hu, W.; Li, D.; Leng, S. Potential Micronutrients and Phytochemicals against the Pathogenesis of Chronic Obstructive Pulmonary Disease and Lung Cancer. Nutrients 2018, 10, 813. https://doi.org/10.3390/nu10070813
Zhai T, Li S, Hu W, Li D, Leng S. Potential Micronutrients and Phytochemicals against the Pathogenesis of Chronic Obstructive Pulmonary Disease and Lung Cancer. Nutrients. 2018; 10(7):813. https://doi.org/10.3390/nu10070813
Chicago/Turabian StyleZhai, Ting, Shizhen Li, Wei Hu, Duo Li, and Shuguang Leng. 2018. "Potential Micronutrients and Phytochemicals against the Pathogenesis of Chronic Obstructive Pulmonary Disease and Lung Cancer" Nutrients 10, no. 7: 813. https://doi.org/10.3390/nu10070813
APA StyleZhai, T., Li, S., Hu, W., Li, D., & Leng, S. (2018). Potential Micronutrients and Phytochemicals against the Pathogenesis of Chronic Obstructive Pulmonary Disease and Lung Cancer. Nutrients, 10(7), 813. https://doi.org/10.3390/nu10070813




