Inulin Supplementation Does Not Reduce Plasma Trimethylamine N-Oxide Concentrations in Individuals at Risk for Type 2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
2.3. Controlled Feeding
2.4. Procedures
2.5. Fasting and Postprandial Blood Collection
2.6. Mass Spectrometry
2.7. Statistical Analyses
3. Results
3.1. Participant Characteristics
3.2. Dietary Intake
3.3. Fasting and Postprandial Plasma TMAO and TMA Moiety Concentrations
3.4. Correlations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tabák, A.G.; Herder, C.; Rathmann, W.; Brunner, E.J.; Kivimäki, M. Prediabetes: A high-risk state for diabetes development. Lancet 2012, 379, 2279–2290. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Abdul-Ghani, M. Assessment and treatment of cardiovascular risk in prediabetes: Impaired glucose tolerance and impaired fasting glucose. Am. J. Cardiol. 2011, 108, 3B–24B. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Cai, X.; Mai, W.; Li, M.; Hu, Y. Association between prediabetes and risk of cardiovascular disease and all cause mortality: Systematic review and meta-analysis. Br. Med. J. 2016, 355, i5953. [Google Scholar]
- Tang, W.H.; Wang, Z.; Li, X.S.; Fan, Y.; Li, D.S.; Wu, Y.; Hazen, S.L. Increased trimethylamine n-oxide portends high mortality risk independent of glycemic control in patients with type 2 diabetes mellitus. Clin. Chem. 2017, 63, 297–306. [Google Scholar] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [PubMed]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Li, X.S.; Obeid, S.; Klingenberg, R.; Gencer, B.; Mach, F.; Raber, L.; Windecker, S.; Rodondi, N.; Nanchen, D.; Muller, O.; et al. Gut microbiota-dependent trimethylamine n-oxide in acute coronary syndromes: A prognostic marker for incident cardiovascular events beyond traditional risk factors. Eur. Heart J. 2017, 38, 814–824. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Warrier, M. Trimethylamine n-oxide, the microbiome, and heart and kidney disease. Annu. Rev. Nutr. 2017, 37, 157–181. [Google Scholar] [PubMed]
- Tang, W.H.; Kitai, T.; Hazen, S.L. Gut microbiota in cardiovascular health and disease. Circ. Res. 2017, 120, 1183–1196. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Hazen, S.L. The contributory role of gut microbiota in cardiovascular disease. J. Clin. Investig. 2014, 124, 4204–4211. [Google Scholar] [CrossRef] [PubMed]
- Boutagy, N.E.; Neilson, A.P.; Osterberg, K.L.; Smithson, A.T.; Englund, T.R.; Davy, B.M.; Hulver, M.W.; Davy, K.P. Probiotic supplementation and trimethylamine-n-oxide production following a high-fat diet. Obesity 2015, 23, 2357–2363. [Google Scholar] [CrossRef] [PubMed]
- Borges, N.A.; Stenvinkel, P.; Bergman, P.; Qureshi, A.R.; Lindholm, B.; Moraes, C.; Stockler-Pinto, M.B.; Mafra, D. Effects of probiotic supplementation on trimethylamine-n-oxide plasma levels in hemodialysis patients: A pilot study. Probiot. Antimicrob. Proteins 2018. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.M.; Davy, B.M.; Halliday, T.M.; Hulver, M.W.; Neilson, A.P.; Ponder, M.A.; Davy, K.P. The effect of prebiotic supplementation with inulin on cardiometabolic health: Rationale, design, and methods of a controlled feeding efficacy trial in adults at risk of type 2 diabetes. Contemp. Clin. Trials 2015, 45, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Mifflin, M.D.; St Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Conway, J.M.; Ingwersen, L.A.; Vinyard, B.T.; Moshfegh, A.J. Effectiveness of the us department of agriculture 5-step multiple-pass method in assessing food intake in obese and nonobese women. Am. J. Clin. Nutr. 2003, 77, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Demarquoy, J.; Georges, B.; Rigault, C.; Royer, M.; Clairet, A.; Soty, M.; Lekounoungou, S.; Le Borgne, F. Radioisotopic determination of l-carnitine content in foods commonly eaten in western countries. Food Chem. 2004, 86, 137–142. [Google Scholar] [CrossRef]
- Carnitine: Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/Carnitine-HealthProfessional/ (accessed on 6 March 2018).
- Boutagy, N.E.; Neilson, A.P.; Osterberg, K.L.; Smithson, A.T.; Englund, T.R.; Davy, B.M.; Hulver, M.W.; Davy, K.P. Short-term high-fat diet increases postprandial trimethylamine-n-oxide in humans. Nutr. Res. 2015, 35, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wang, Z.; Tang, W.H.W.; Hazen, S.L. Gut microbe-generated trimethylamine n-oxide from dietary choline is prothrombotic in subjects. Circulation 2017, 135, 1671–1673. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2015, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Smits, L.P.; Kootte, R.S.; Levin, E.; Prodan, A.; Fuentes, S.; Zoetendal, E.G.; Wang, Z.; Levison, B.S.; Cleophas, M.C.P.; Kemper, E.M.; et al. Effect of vegan fecal microbiota transplantation on carnitine- and choline-derived trimethylamine-n-oxide production and vascular inflammation in patients with metabolic syndrome. J. Am. Heart Assoc. 2018, 7, e008342. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Klatt, K.C.; Caudill, M.A. Choline. Adv. Nutr. 2018, 9, 58–60. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Roberts, A.B.; Buffa, J.A.; Levison, B.S.; Zhu, W.; Org, E.; Gu, X.; Huang, Y.; Zamanian-Daryoush, M.; Culley, M.K.; et al. Non-lethal inhibition of gut microbial trimethylamine production for the treatment of atherosclerosis. Cell 2015, 163, 1585–1595. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, N.; Williams, P.T.; Lamendella, R.; Faghihnia, N.; Grube, A.; Li, X.; Wang, Z.; Knight, R.; Jansson, J.K.; Hazen, S.L.; et al. Diets high in resistant starch increase plasma levels of trimethylamine-n-oxide, a gut microbiome metabolite associated with CVD risk. Br. J. Nutr. 2016, 116, 2020–2029. [Google Scholar] [CrossRef] [PubMed]
- Nutrient Intakes from Food and Beverages: Mean Amounts Consumed per Individual, by Gender and Age, What We Eat in America, NHANES 2013–2014. Available online: www.ars.usda.gov/nea/bhnrc/fsrg (accessed on 6 March 2018).
- Rebouche, C.J. Kinetics, pharmacokinetics, and regulation of l-carnitine and acetyl-l-carnitine metabolism. Ann. N. Y. Acad. Sci. 2004, 1033, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Kolida, S.; Tuohy, K.; Gibson, G.R. Prebiotic effects of inulin and oligofructose. Br. J. Nutr. 2002, 87 (Suppl. 2), S193–S197. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.; Stasse-Wolthuis, M. The bifidogenic effect of inulin and oligofructose and its consequences for gut health. Eur. J. Clin. Nutr. 2009, 63, 1277–1289. [Google Scholar] [CrossRef] [PubMed]
- Bouhnik, Y.; Raskine, L.; Champion, K.; Andrieux, C.; Penven, S.; Jacobs, H.; Simoneau, G. Prolonged administration of low-dose inulin stimulates the growth of bifidobacteria in humans. Nutr. Res. 2007, 27, 187–193. [Google Scholar] [CrossRef]
 placebo at baseline,
placebo at baseline,  inulin at baseline,
inulin at baseline,  placebo post-intervention,
placebo post-intervention,  inulin post-intervention.
inulin post-intervention.
 placebo at baseline,
placebo at baseline,  inulin at baseline,
inulin at baseline,  placebo post-intervention,
placebo post-intervention,  inulin post-intervention.
inulin post-intervention.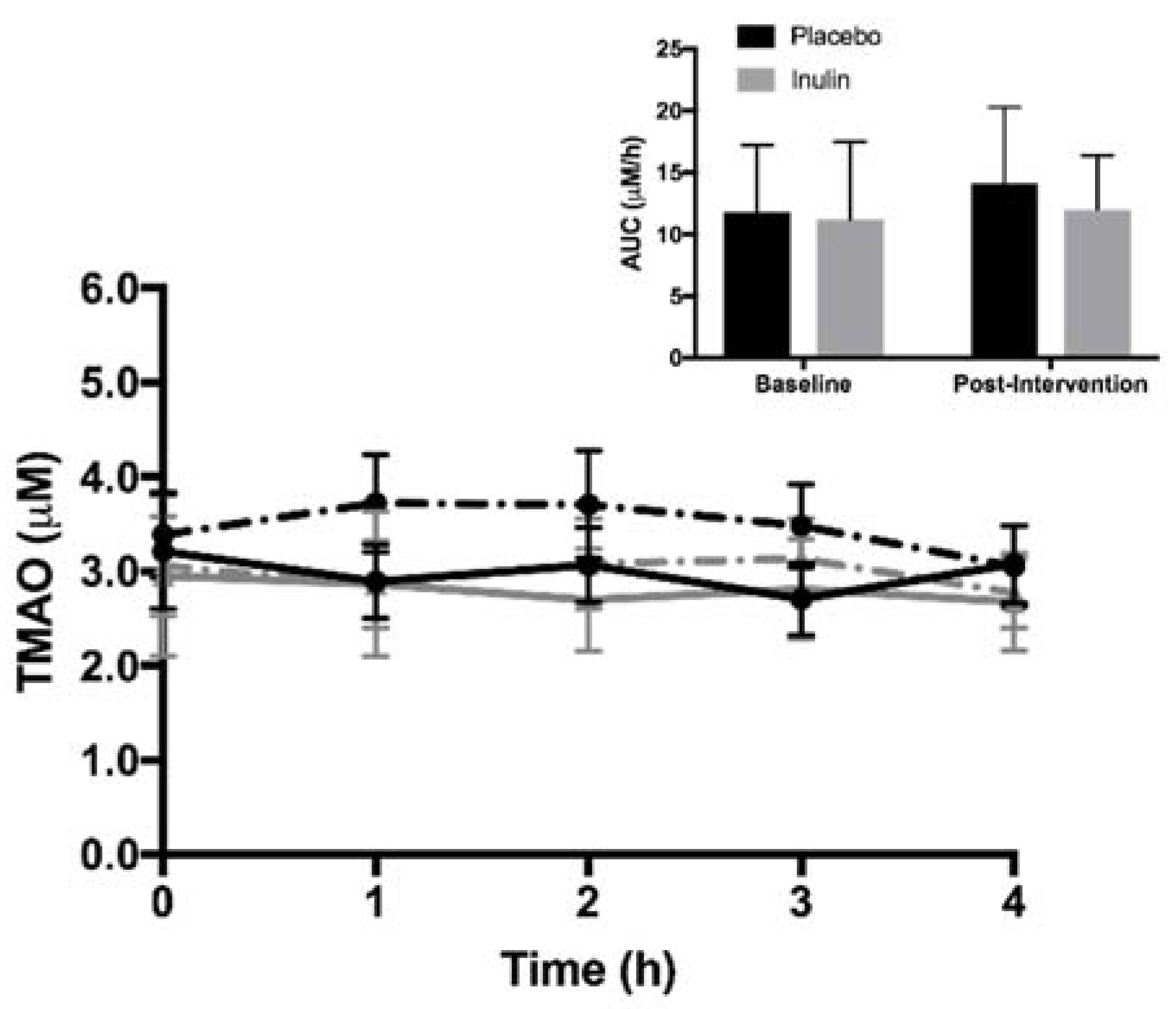
 placebo at baseline,
placebo at baseline,  inulin at baseline,
inulin at baseline,  placebo post-intervention,
placebo post-intervention,  inulin post-intervention.
inulin post-intervention.
 placebo at baseline,
placebo at baseline,  inulin at baseline,
inulin at baseline,  placebo post-intervention,
placebo post-intervention,  inulin post-intervention.
inulin post-intervention.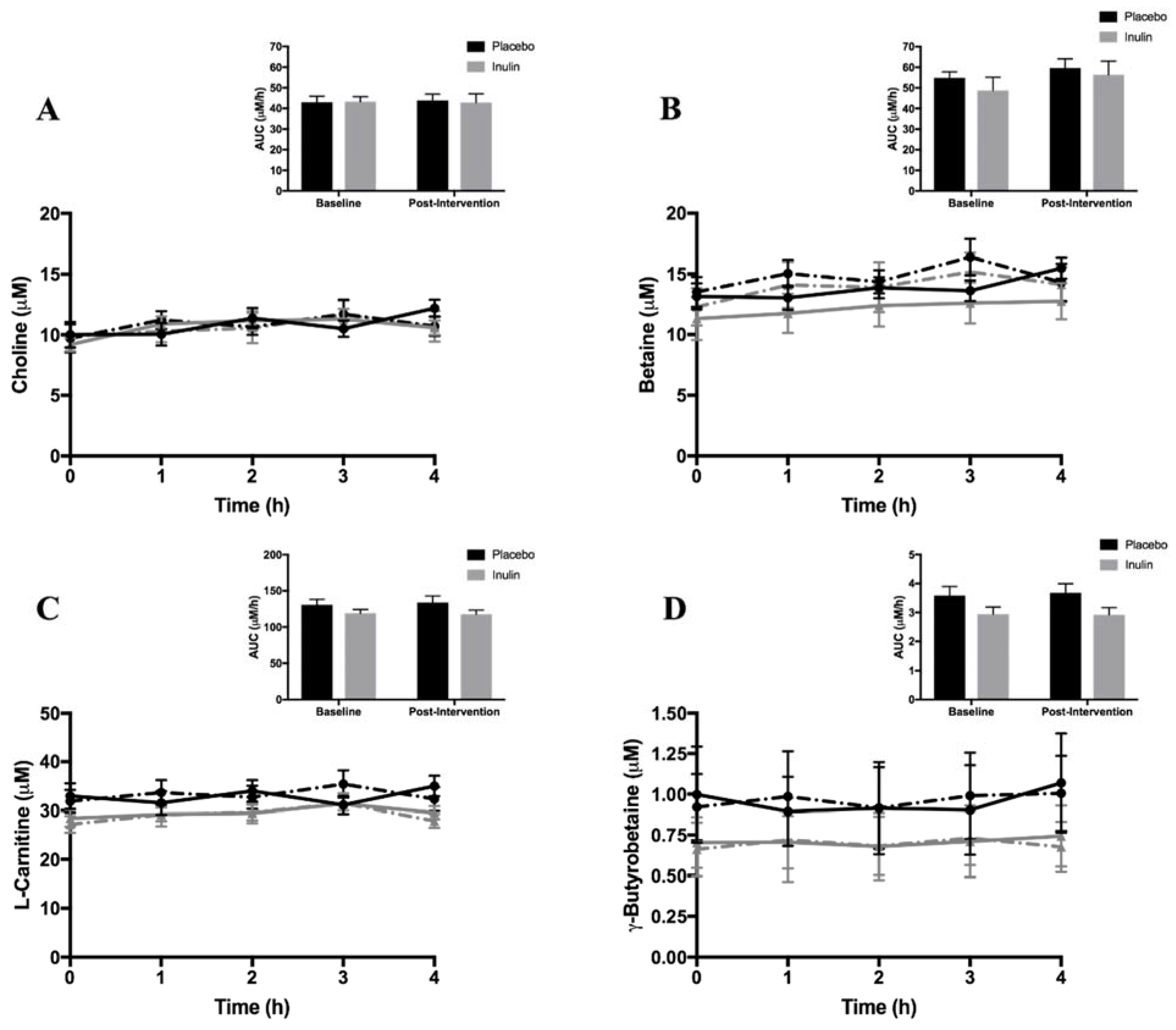
| Compound | Retention Time (min) | Parent [M + H]+ (m/z) | Daughter (m/z) | Cone Voltage (V) | Collision Energy (eV) |
|---|---|---|---|---|---|
| Betaine | 1.25 | 117.15 | 118.24 | 59 | 44 |
| γ-Butyrobetaine | 0.98 | 146.26 | 87.00 | 26 | 16 |
| Betaine-d9 | 1.25 | 126.14 | 127.3 | 68 | 46 |
| Choline | 1.13 | 103.16 | 104.2 | 60 | 38 |
| Choline-d9 | 1.11 | 112.16 | 113.32 | 69 | 40 |
| TMAO | 2.01 | 75.11 | 76.16 | 59 | 40 |
| TMAO-d9 | 1.98 | 84.12 | 85.22 | 68 | 40 |
| Characteristic | Placebo n = 11 (6 Females) | Inulin n = 7 (5 Females) |
|---|---|---|
| Age, years | 54 ± 2 | 58 ± 3 |
| BMI, kg/m2 | 30.8 ± 0.8 | 31.0 ± 0.92 |
| Body Fat, % | 39.2 ± 1.8 | 41.7 ± 4.0 |
| FPG, mg/dL | 95 ± 4 | 88 ± 4 |
| 2-h OGT, mg/dL § | 121 ± 12 | 125 ± 19 |
| ADA T2DM Risk Assessment Score | 5 | 5 |
| Plasma Total Cholesterol, mg/dL | 208 ± 8 | 212 ± 13 |
| Plasma LDL Cholesterol, mg/dL | 131 ± 9 | 124 ± 17 |
| Plasma HDL Cholesterol, mg/dL | 49 ± 3 | 56 ± 7 |
| Plasma Triglycerides, mg/dL | 141 ± 16 | 160 ± 41 |
| Placebo n = 10 (5 Females) | Inulin n = 7 (5 Females) | |
|---|---|---|
| Energy (kcal/day) | 2172 ± 184 | 2009 ± 218 |
| Protein (g/day) | 91 ± 7 | 74 ± 8 * |
| Protein (% kcal/day) | 17 ± 1 | 15 ± 1 |
| Carbohydrate (g/day) | 236 ± 19 | 242 ± 27 |
| Carbohydrate (% kcal/day) | 44 ± 2 | 48 ± 3 |
| Total Fat (g/day) | 94 ± 11 | 82 ± 13 |
| Total Fat (% kcal/day) | 38 ± 2 | 36 ± 2 |
| Saturated Fat (g/day) | 32 ± 3 | 27 ± 5 |
| Saturated Fat (% kcal/day) | 13 ± 1 | 12 ± 1 |
| Fiber (g/day) | 17 ± 2 | 20 ± 2 |
| Insoluble Fiber (g/day) | 11 ± 1 | 14 ± 2 |
| Soluble Fiber (g/day) | 6 ± 1 | 6 ± 1 |
| Sodium (mg/day) | 3797 ± 315 | 2871 ± 248 * |
| Betaine (mg/day) | 153 ± 21 | 187 ± 33 |
| Choline (mg/day) | 408 ± 32 | 262 ± 28 ** |
| l-Carnitine (mg/day) | 51 ± 6 | 36 ± 3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baugh, M.E.; Steele, C.N.; Angiletta, C.J.; Mitchell, C.M.; Neilson, A.P.; Davy, B.M.; Hulver, M.W.; Davy, K.P. Inulin Supplementation Does Not Reduce Plasma Trimethylamine N-Oxide Concentrations in Individuals at Risk for Type 2 Diabetes. Nutrients 2018, 10, 793. https://doi.org/10.3390/nu10060793
Baugh ME, Steele CN, Angiletta CJ, Mitchell CM, Neilson AP, Davy BM, Hulver MW, Davy KP. Inulin Supplementation Does Not Reduce Plasma Trimethylamine N-Oxide Concentrations in Individuals at Risk for Type 2 Diabetes. Nutrients. 2018; 10(6):793. https://doi.org/10.3390/nu10060793
Chicago/Turabian StyleBaugh, Mary Elizabeth, Cortney N. Steele, Christopher J. Angiletta, Cassie M. Mitchell, Andrew P. Neilson, Brenda M. Davy, Matthew W. Hulver, and Kevin P. Davy. 2018. "Inulin Supplementation Does Not Reduce Plasma Trimethylamine N-Oxide Concentrations in Individuals at Risk for Type 2 Diabetes" Nutrients 10, no. 6: 793. https://doi.org/10.3390/nu10060793
APA StyleBaugh, M. E., Steele, C. N., Angiletta, C. J., Mitchell, C. M., Neilson, A. P., Davy, B. M., Hulver, M. W., & Davy, K. P. (2018). Inulin Supplementation Does Not Reduce Plasma Trimethylamine N-Oxide Concentrations in Individuals at Risk for Type 2 Diabetes. Nutrients, 10(6), 793. https://doi.org/10.3390/nu10060793




