Longitudinal Study of the Role of Epidermal Growth Factor on the Fractional Excretion of Magnesium in Children: Effect of Calcineurin Inhibitors
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Magnesium Intake Questionnaire and Nubel®
2.3. Determination of Creatinine, Magnesium, and CsA Levels
2.4. Determination of Urinary EGF
2.5. Statistical Analysis
3. Results
3.1. Population Demographics
Group Descriptions
3.2. Kidney Function, Magnesium, EGF, and CNI Levels
Patients with Hypomagnesemia versus Patients with Normomagnesemia (presented in Table 3)
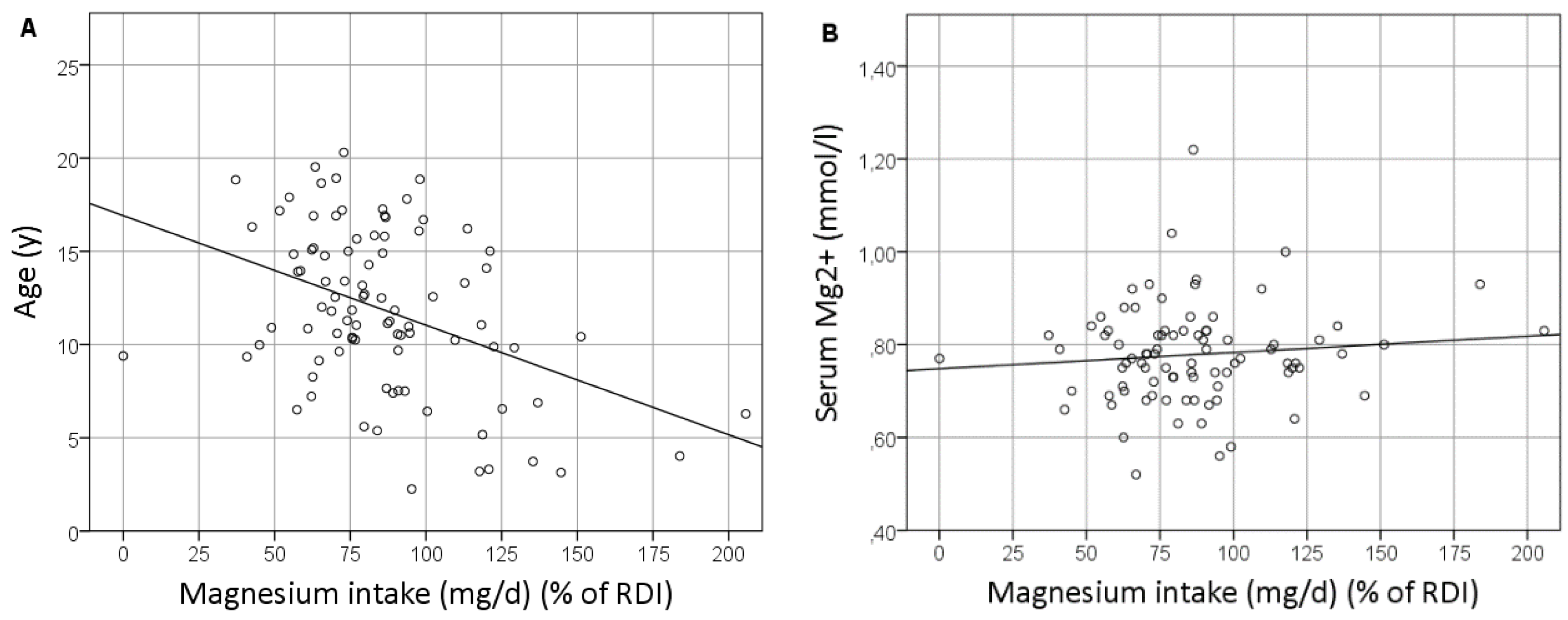
3.3. Magnesium Intake
3.4. The Healthy Control Group
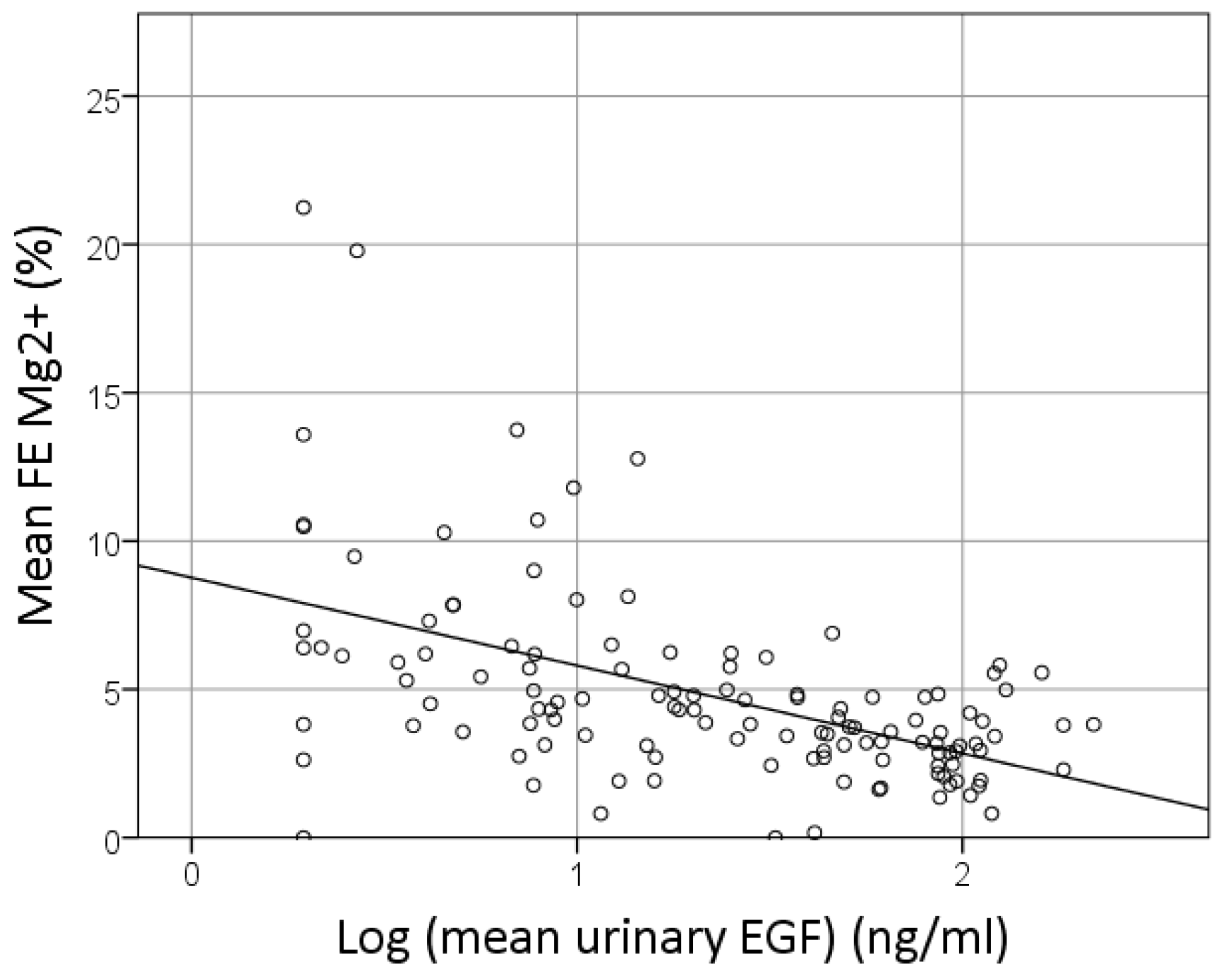
3.5. Predictors of FE Mg2+
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Narayanan, S.; Scalici, P. Serum magnesium levels in pediatric inpatients: A study in laboratory overuse. Hosp. Pediatr. 2015, 5, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.A.; Chen, Z.; Hawthorne, K.M. Magnesium metabolism in 4-year-old to 8-year-old children. J. Bone Miner. Res. 2014, 29, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kidney J. 2012, 5, i3–i14. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M. Calcium and magnesium requirements of children and adolescents and peak bone mass. Nutrition 2000, 16, 514–516. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Hu, F.B.; Ros, E.; Sabate, J. The role of tree nuts and peanuts in the prevention of coronary heart disease: Multiple potential mechanisms. J. Nutr. 2008, 138, 1746S–1751S. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Dairy and bone health. J. Am. Coll. Nutr. 2009, 28 (Suppl. 1), 82S–90S. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.F. Influence of vegetable protein sources on trace element and mineral bioavailability. J. Nutr. 2003, 133, 2973S–2977S. [Google Scholar] [CrossRef] [PubMed]
- Tornaritis, M.J.; Philippou, E.; Hadjigeorgiou, C.; Kourides, Y.A.; Panayi, A.; Savva, S.C. A study of the dietary intake of cypriot children and adolescents aged 6–18 years and the association of mother’s educational status and children’s weight status on adherence to nutritional recommendations. BMC Public Health 2014, 14, 13. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Aranda, P.; Perez de la Cruz, A.; Llopis, J. Magnesium and zinc status in patients with chronic renal failure: Influence of a nutritional intervention. Magnes. Res. 2009, 22, 72–80. [Google Scholar] [PubMed]
- Gluba-Brzozka, A.; Franczyk, B.; Rysz, J. Vegetarian diet in chronic kidney disease-a friend or foe. Nutrients 2017, 9, 374. [Google Scholar] [CrossRef] [PubMed]
- Lameris, A.L.; Monnens, L.A.; Bindels, R.J.; Hoenderop, J.G.J. Drug-induced alterations in mg2+ homoeostasis. Clin. Sci. 2012, 123, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Milbouw, S.; Verhaegen, J.; Verrijken, A.; Schepens, T.; De Winter, B.Y.; Van Gaal, L.F.; Ledeganck, K.J.; De Block, C.E.M. Predictors of insulin resistance in obesity and type 2 diabetes mellitus—The role of magnesium. J. Metab. Syndr. 2017, 6, 235. [Google Scholar] [CrossRef]
- Liu, F.; Mao, J.H. Calcineurin inhibitors and nephrotoxicity in children. World J. Pediatr. 2018, 14, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Burdmann, E.A.; Andoh, T.F.; Lindsley, J.; Houghton, D.C.; Bennett, W.M. Effects of oral magnesium supplementation on acute experimental cyclosporin nephrotoxicity. Nephrol. Dial. Transplant. 1994, 9, 16–21. [Google Scholar] [PubMed]
- Higgins, R.; Ramaiyan, K.; Dasgupta, T.; Kanji, H.; Fletcher, S.; Lam, F.; Kashi, H. Hyponatraemia and hyperkalaemia are more frequent in renal transplant recipients treated with tacrolimus than with cyclosporin. Further evidence for differences between cyclosporin and tacrolimus nephrotoxicities. Nephrol. Dial. Transplant. 2004, 19, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Hoenderop, J.G.J.; Bindels, R.J.M. Epithelial ca2+ and mg2+ channels in health and disease. J. Am. Soc. Nephrol. 2005, 16, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, K.P.; Waldegger, S.; Konrad, M.; Chubanov, V.; Gudermann, T. Trpm6 and trpm7—Gatekeepers of human magnesium metabolism. Biochim. Biophys. Acta 2007, 1772, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Thebault, S.P.; Alexander, R.T.; Tiel Groenestege, W.M.; Hoenderop, J.G.; Bindels, R.J. Egf increases trpm6 activity and surface expression. J. Am. Soc. Nephrol. 2009, 20, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Groenestege, W.M.; Thebault, S.; van der Wijst, J.; van den Berg, D.; Janssen, R.; Tejpar, S.; van den Heuvel, L.P.; van Cutsem, E.; Hoenderop, J.G.; Knoers, N.V.; et al. Impaired basolateral sorting of pro-egf causes isolated recessive renal hypomagnesemia. J. Clin. Invest. 2007, 117, 2260–2267. [Google Scholar] [CrossRef] [PubMed]
- Ledeganck, K.J.; Boulet, G.A.; Horvath, C.A.; Vinckx, M.; Bogers, J.J.; Van den Bossche, R.; Verpooten, G.A.; De Winter, B.Y. Expression of renal distal tubule transporters trpm6 and ncc in a rat model of cyclosporine nephrotoxicity and effect of egf treatment. Am. J. Physiol. Renal Physiol. 2011, 301, F486–F493. [Google Scholar] [CrossRef] [PubMed]
- Ledeganck, K.J.; De Winter, B.Y.; Van den Driessche, A.; Jurgens, A.; Bosmans, J.L.; Couttenye, M.M.; Verpooten, G.A. Magnesium loss in cyclosporine-treated patients is related to renal epidermal growth factor downregulation. Nephrol. Dial. Transplant. 2013, 29, 1097–1102. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Harris, R.C. Epidermal growth factor, from gene organization to bedside. Semin. Cell Dev. Biol. 2014, 28, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Tomaszewska, R.; Dembinski, A.; Warzecha, Z.; Ceranowicz, P.; Konturek, S.J.; Stachura, J. The influence of epidermal growth factor on the course of ischemia-reperfusion induced pancreatitis in rats. J. Physiol. Pharmacol. 2002, 53, 183–198. [Google Scholar] [PubMed]
- Warzecha, Z.; Dembinski, A.; Konturek, P.C.; Ceranowicz, P.; Konturek, S.J. Epidermal growth factor protects against pancreatic damage in cerulein-induced pancreatitis. Digestion 1999, 60, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Konturek, P.C.; Dembinski, A.; Warzecha, Z.; Ceranowicz, P.; Konturek, S.J.; Stachura, J.; Hahn, E.G. Expression of transforming growth factor-beta 1 and epidermal growth factor in caerulein-induced pancreatitis in rat. J. Physiol. Pharmacol. 1997, 48, 59–72. [Google Scholar] [PubMed]
- Bennett, S.P.; Griffiths, G.D.; Schor, A.M.; Leese, G.P.; Schor, S.L. Growth factors in the treatment of diabetic foot ulcers. Br. J. Surg. 2003, 90, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Huysentruyt, K.; Laire, D.; Van Avondt, T.; De Schepper, J.; Vandenplas, Y. Energy and macronutrient intakes and adherence to dietary guidelines of infants and toddlers in belgium. Eur. J. Nutr. 2016, 55, 1595–1604. [Google Scholar] [CrossRef] [PubMed]
- Cochat, P. Espn Handbook; European Society for Paediatric Nephrology; Medcom: Lyon, France, 2002. [Google Scholar]
- Schwartz, G.J.; Munoz, A.; Schneider, M.F.; Mak, R.H.; Kaskel, F.; Warady, B.A.; Furth, S.L. New equations to estimate gfr in children with ckd. J. Am. Soc. Nephrol. 2009, 20, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; van der Wijst, J.; van der Kemp, A.; van Zeeland, F.; Bindels, R.J.; Hoenderop, J.G. Regulation of the epithelial mg2+ channel trpm6 by estrogen and the associated repressor protein of estrogen receptor activity (rea). J. Biol. Chem. 2009, 284, 14788–14795. [Google Scholar] [CrossRef] [PubMed]
- Nozue, T.; Kobayashi, A.; Kodama, T.; Uemasu, F.; Endoh, H.; Sako, A.; Takagi, Y. Pathogenesis of cyclosporine-induced hypomagnesemia. J. Pediatr. 1992, 120, 638–640. [Google Scholar] [CrossRef]
- Riva, N.; Schaiquevich, P.; Caceres Guido, P.; Halac, E.; Dip, M.; Imventarza, O. Pharmacoepidemiology of tacrolimus in pediatric liver transplantation. Pediatr. Transplant. 2017, 21. [Google Scholar] [CrossRef] [PubMed]
- Hayes, W.; Boyle, S.; Carroll, A.; Bockenhauer, D.; Marks, S.D. Hypomagnesemia and increased risk of new-onset diabetes mellitus after transplantation in pediatric renal transplant recipients. Pediatr. Nephrol. 2017, 32, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Yanik, G.; Levine, J.E.; Ratanatharathorn, V.; Dunn, R.; Ferrara, J.; Hutchinson, R.J. Tacrolimus (fk506) and methotrexate as prophylaxis for acute graft-versus-host disease in pediatric allogeneic stem cell transplantation. Bone Marrow Transplant. 2000, 26, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Nijenhuis, T.; Hoenderop, J.G.; Bindels, R.J. Downregulation of ca(2+) and mg(2+) transport proteins in the kidney explains tacrolimus (fk506)-induced hypercalciuria and hypomagnesemia. J. Am. Soc. Nephrol. 2004, 15, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Freundlich, M. Bone mineral content and mineral metabolism during cyclosporine treatment of nephrotic syndrome. J. Pediatr. 2006, 149, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Ariceta, G.; Rodriguez-Soriano, J.; Vallo, A. Renal magnesium handling in infants and children. Acta Paediatr. 1996, 85, 1019–1023. [Google Scholar] [CrossRef] [PubMed]
- Viering, D.; de Baaij, J.H.F.; Walsh, S.B.; Kleta, R.; Bockenhauer, D. Genetic causes of hypomagnesemia, a clinical overview. Pediatr. Nephrol. 2017, 32, 1123–1135. [Google Scholar] [CrossRef] [PubMed]
- Whang, R. Magnesium deficiency: Pathogenesis, prevalence, and clinical implications. Am. J. Med. 1987, 82, 24–29. [Google Scholar] [CrossRef]
- Agus, Z.S. Mechanisms and causes of hypomagnesemia. Curr. Opin. Nephrol. Hypertens. 2016, 25, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Gheissari, A.; Andalib, A.; Labibzadeh, N.; Modarresi, M.; Azhir, A.; Merrikhi, A. Fractional excretion of magnesium (femg), a marker for tubular dysfunction in children with clinically recovered ischemic acute tubular necrosis. Saudi J. Kidney Dis. Transpl. 2011, 22, 476–481. [Google Scholar] [PubMed]
- Zylka, A.; Gala-Bladzinska, A.; Dumnicka, P.; Ceranowicz, P.; Kuzniewski, M.; Gil, K.; Olszanecki, R.; Kusnierz-Cabala, B. Is urinary ngal determination useful for monitoring kidney function and assessment of cardiovascular disease? A 12-month observation of patients with type 2 diabetes. Dis. Markers 2016, 2016, 8489543. [Google Scholar] [CrossRef] [PubMed]
- Shlipak, M.G.; Mattes, M.D.; Peralta, C.A. Update on cystatin c: Incorporation into clinical practice. Am. J. Kidney Dis. 2013, 62, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Geist, B.K.; Diemling, M.; Staudenherz, A. Glomerular filtration rate and error calculation based on the slope-intercept method with chromium-51 ethylenediaminetetraacetic acid via a new clinical software: Gfrcalc. Med. Princ. Pract. 2016, 25, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Uslu Gokceoglu, A.; Comak, E.; Dogan, C.S.; Koyun, M.; Akbas, H.; Akman, S. Magnesium excretion and hypomagnesemia in pediatric renal transplant recipients. Ren. Fail. 2014, 36, 1056–1059. [Google Scholar] [CrossRef] [PubMed]
- Osorio, J.M.; Bravo, J.; Perez, A.; Ferreyra, C.; Osuna, A. Magnesemia in renal transplant recipients: Relation with immunosuppression and posttransplant diabetes. Transplant. Proc. 2010, 42, 2910–2913. [Google Scholar] [CrossRef] [PubMed]
- Slev, P.R.; Bunker, A.M.; Owen, W.E.; Roberts, W.L. Pediatric reference intervals for random urine calcium, phosphorus and total protein. Pediatr. Nephrol. 2010, 25, 1707–1710. [Google Scholar] [CrossRef] [PubMed]
- Ariceta, G. Clinical practice: Proteinuria. Eur. J. Pediatr. 2011, 170, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Sobaler, A.M.; Aparicio, A.; Gonzalez-Rodriguez, L.G.; Cuadrado-Soto, E.; Rubio, J.; Marcos, V.; Sanchidrian, R.; Santos, S.; Perez-Farinos, N.; Dal Re, M.A.; et al. Adequacy of usual vitamin and mineral intake in spanish children and adolescents: Enalia study. Nutrients 2017, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Mavrogianni, C.; Bos, R.; Singh-Povel, C. Micronutrient intakes among children and adults in greece: The role of age, sex and socio-economic status. Nutrients 2014, 6, 4073–4092. [Google Scholar] [CrossRef] [PubMed]
- Affenito, S.G.; Thompson, D.R.; Franko, D.L.; Striegel-Moore, R.H.; Daniels, S.R.; Barton, B.A.; Schreiber, G.B.; Schmidt, M.; Crawford, P.B. Longitudinal assessment of micronutrient intake among african-american and white girls: The national heart, lung, and blood institute growth and health study. J. Am. Diet. Assoc. 2007, 107, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
| Group | Renal Tx + CNI (n = 23) | CKD − CNI (n = 24) | NS + CNI (n = 27) | NS − CNI (n = 22) |
|---|---|---|---|---|
| Age (year) | 13.4 (2.2–20.3) | 11.1 (3.2–18.9) | 12.5 (3.1–19.5) | 12.3 (3.7–18.7) |
| Gender (M/F; %) | 87/13 | 62/38 | 71/29 | 64/36 |
| Length (cm) | 151 (88–183) | 145 (95–176) | 149 (92–190) | 149 (101–183) |
| Weight (kg) | 43 ± 17 | 42 ± 16 | 46 ± 23 | 42 ± 15 |
| BMI z-score | 0.40 (−2.10–1.13) | 0.24 (−2.24–2.05) | 0.17 (−1.57–2.12) | −0.09 (−1.86–2.82) |
| Mg2+ intake (% of RDI) | 89 (37–684) | 86 (63–436) | 87 (45–299) | 86 (41–383) |
| Patients who exceeded the RDI for Mg2+ intake (%) | 27.3 | 30.4 | 30.8 | 42.1 |
| Mg2+ supplements (%) | 13.0 | 8.0 | 13.8 | 25.0 |
| Group | Renal Tx + CNI (n = 23) | CKD − CNI (n = 24) | NS + CNI (n = 28) | NS − CNI (n = 22) |
|---|---|---|---|---|
| Serum Creatinine (mg/dL) | 1.11 (0.08) §# | 1.27 (0.14) #§ | 0.72 (0.07) #$* | 0.54 (0.03) $*§ |
| Creatinine clearance (mL/min/1.73 m2) | 59 (3) §# | 62 (6) #§ | 98 (5) #$* | 117 (4) $*§ |
| Urinary protein/creatinine (mg/g) | 347.4 (71.8) $ | 613.2 (115.0) * | 848.0 (333.9) | 544.9 (215.8) |
| Serum Mg2+ (mg/dL) | 0.76 (0.02) #$ | 0.82 (0.02) *§ | 0.78 (0.02) #$ | 0.84 (0.01) *§ |
| HypoMg (%) | 39.1 $# | 16.0 *§ | 44.8 $# | 10.0 *§ |
| FE Mg2+ (%) | 7.82 (0.84 ) §# | 7.76 (0.84) | 3.95 (0.32) *$ | 3.57 (0.28) *$ |
| CsA levels (ng/mL) | 666 (45) | - | 579 (38) | - |
| Tacrolimus levels (ng/mL) | 8.62 (0.91) | - | 7.71 (0.79) | - |
| Serum EGF (pg/mL) | 776.6 (39.9) | 742.1 (47.8) § | 865.5 (40.7) $ | 817.2 (55.0) |
| Urine EGF (ng/mL) | 7.0 (1.1) #§ | 11.5 (2.4) #§ | 35.4 (6.0) $*$ | 47.7 (6.6) *$ |
| Urine EGF/creatinine (ng/mg) | 0.11 (0.01) $#§ | 0.19 (0.03) #*§ | 0.33 (0.05) #$* | 0.51 (0.07) $*§ |
| Group | Normomagnesemic Patients (n = 69) | Hypomagnesemic Patients (n = 28) | p-Value |
|---|---|---|---|
| Age (year) | 11.79 (3.20–18.66) | 13.91 (3.14–19.53) | 0.492 |
| Length (cm) | 147 (94.5–190) | 155.4 (92–77.5) | 0.582 |
| Weight (kg) | 42.95 ± 18.39 | 43.61 ± 19.31 | 0.683 |
| BMI z-score | 0.14 (−2.24–2.82) | 0.13 (−1.66–2.12) | 0.418 |
| Mg2+ intake (% of RDI) | 87 (41–436) | 88 (37–684) | 0.692 |
| Patients who exceeded the RDI for Mg2+ intake (%) | 31.3 | 34.6 | 0.419 |
| Serum Creatinine (mg/dL) | 0.88 (0.06) | 1.01 (0.11) | 0.287 |
| Creatinine clearance (mL/min/1.73 m2) | 86 (4) | 75 (6) | 0.122 |
| Urinary protein/creatinine (mg/g) | 625.2 (155.8) | 561.7 (120.5) | 0.747 |
| Serum estradiol (pg/mL) | 60.28 (15.83) | 67.24 (37.75) | 0.865 |
| Serum Mg2+ (mg/dL) | 0.83 (0.01) | 0.70 (0.01) | <0.001 |
| FE Mg2+ (%) | 5.41 (0.43) | 6.55 (0.72) | 0.178 |
| CsA levels (ng/mL) | 566.13 (30.70) | 685.75 (53.56) | 0.053 |
| Tacrolimus levels (ng/mL) | 6.90 (0.64) | 8.55 (0.99) | 0.163 |
| Serum EGF (pg/mL) | 802.77 (27.93) | 795.60 (40.08) | 0.883 |
| Urine EGF (ng/mL) | 27.46 (3.50) | 18.58 (4.52) | 0.120 |
| Urine EGF/creatinine (ng/mg) | 0.31 (0.03) | 0.22 (0.05) | 0.120 |
| β | p-Value | 95% CI | ||
|---|---|---|---|---|
| Lower | Upper | |||
| Log Urinary EGF (ng/mL) | −2.084 | <0.001 | −3.153 | −1.015 |
| eGFR (mL/min/1.73 m2) | −0.049 | <0.001 | −0.067 | −0.032 |
| Serum Mg2+ (mg/dL) | −6.239 | 0.034 | −12.014 | −0.463 |
| Age (year) | −0.239 | 0.001 | −0.384 | −0.094 |
| Constant | 20.078 | <0.001 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ledeganck, K.J.; Anné, C.; De Monie, A.; Meybosch, S.; Verpooten, G.A.; Vinckx, M.; Van Hoeck, K.; Van Eyck, A.; De Winter, B.Y.; Trouet, D. Longitudinal Study of the Role of Epidermal Growth Factor on the Fractional Excretion of Magnesium in Children: Effect of Calcineurin Inhibitors. Nutrients 2018, 10, 677. https://doi.org/10.3390/nu10060677
Ledeganck KJ, Anné C, De Monie A, Meybosch S, Verpooten GA, Vinckx M, Van Hoeck K, Van Eyck A, De Winter BY, Trouet D. Longitudinal Study of the Role of Epidermal Growth Factor on the Fractional Excretion of Magnesium in Children: Effect of Calcineurin Inhibitors. Nutrients. 2018; 10(6):677. https://doi.org/10.3390/nu10060677
Chicago/Turabian StyleLedeganck, Kristien J., Charlotte Anné, Amandine De Monie, Sarang Meybosch, Gert A. Verpooten, Marleen Vinckx, Koen Van Hoeck, Annelies Van Eyck, Benedicte Y. De Winter, and Dominique Trouet. 2018. "Longitudinal Study of the Role of Epidermal Growth Factor on the Fractional Excretion of Magnesium in Children: Effect of Calcineurin Inhibitors" Nutrients 10, no. 6: 677. https://doi.org/10.3390/nu10060677
APA StyleLedeganck, K. J., Anné, C., De Monie, A., Meybosch, S., Verpooten, G. A., Vinckx, M., Van Hoeck, K., Van Eyck, A., De Winter, B. Y., & Trouet, D. (2018). Longitudinal Study of the Role of Epidermal Growth Factor on the Fractional Excretion of Magnesium in Children: Effect of Calcineurin Inhibitors. Nutrients, 10(6), 677. https://doi.org/10.3390/nu10060677




