Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function
Abstract
:1. Introduction
2. Materials and Methods
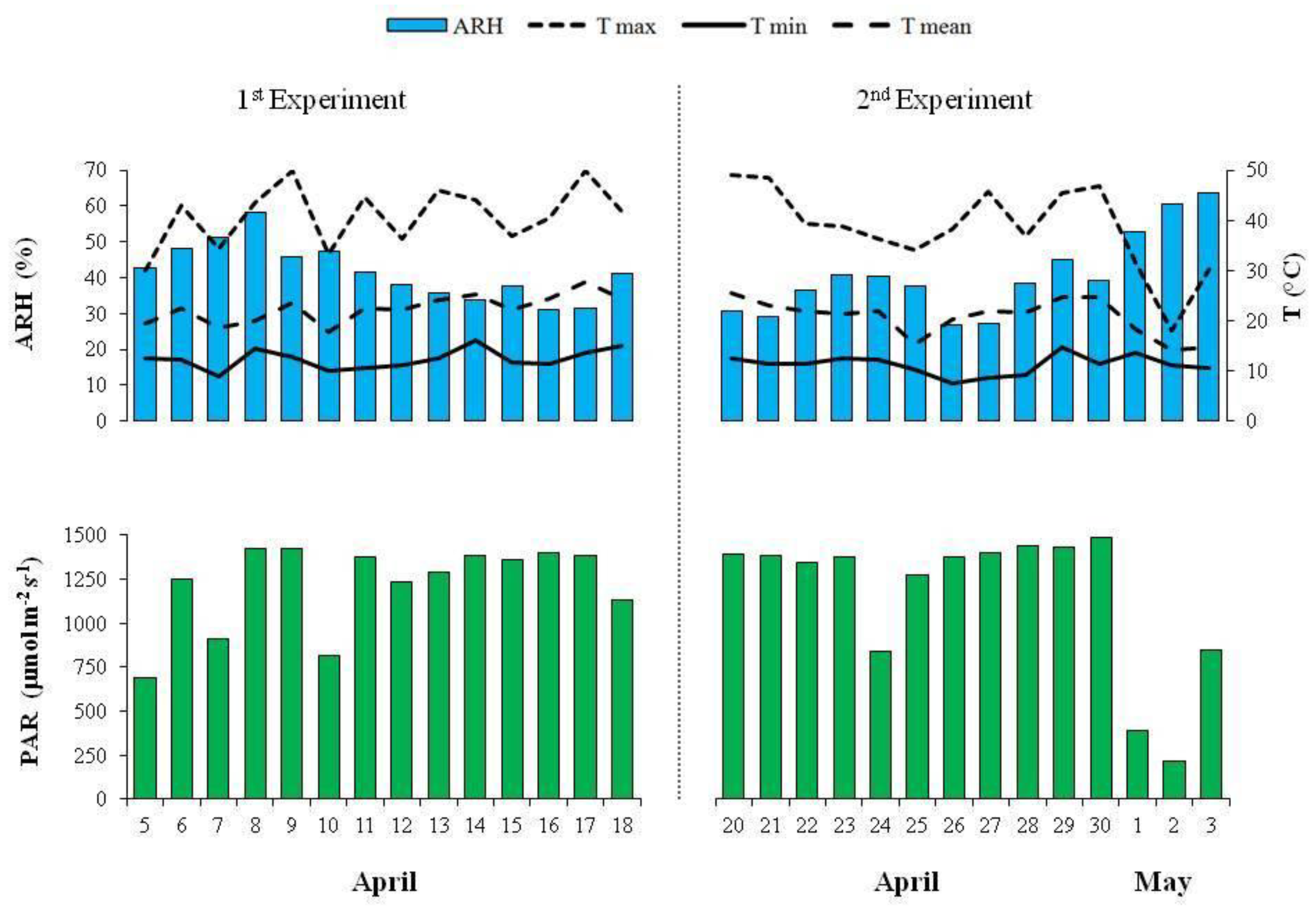
2.1. Experimental Conditions
2.2. Harvesting, Yield Assessment, and Samples Preparation
2.3. Chemical Analysis
2.4. Statistical Analysis
3. Results
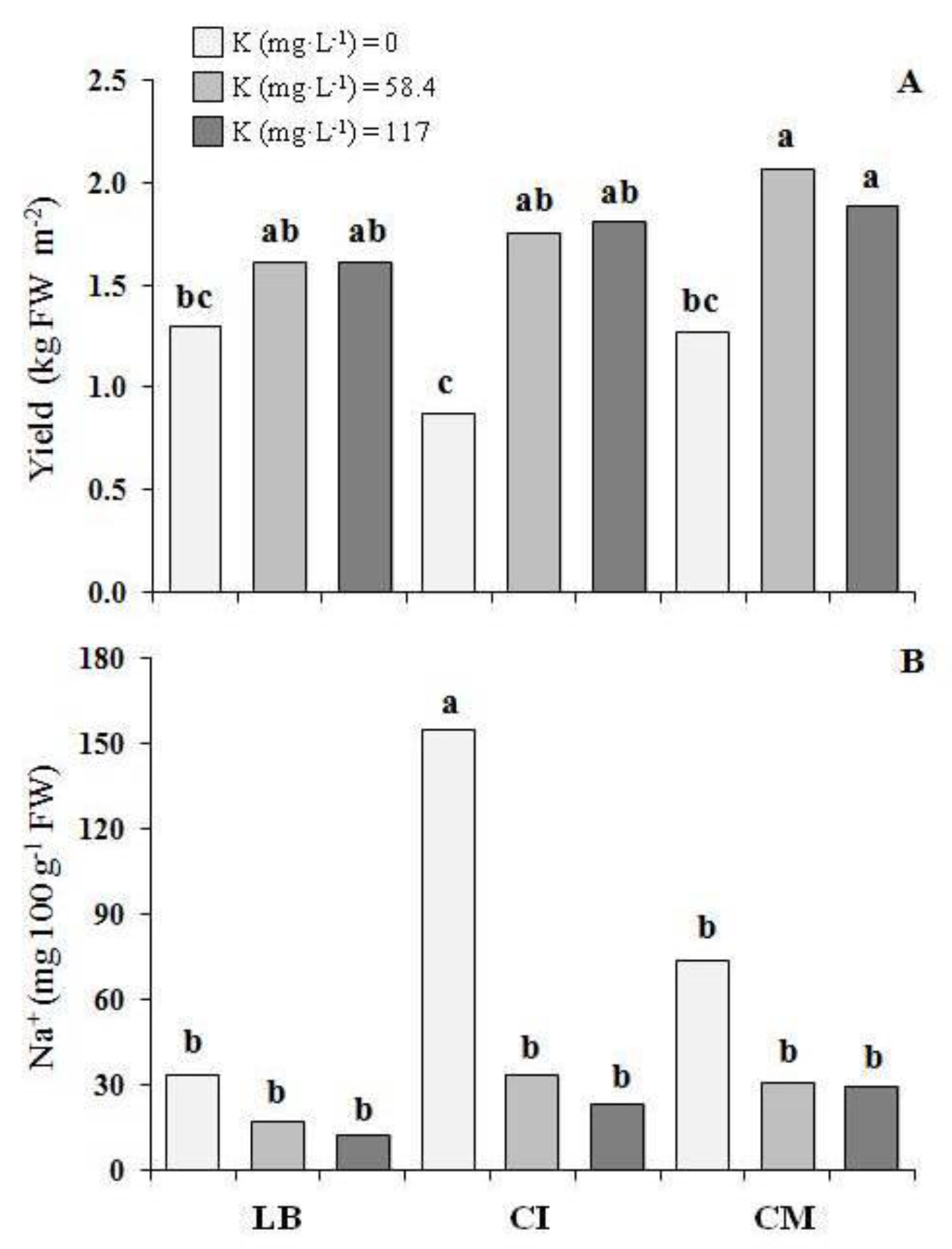
3.1. Crop Performance
3.2. Cation Contents
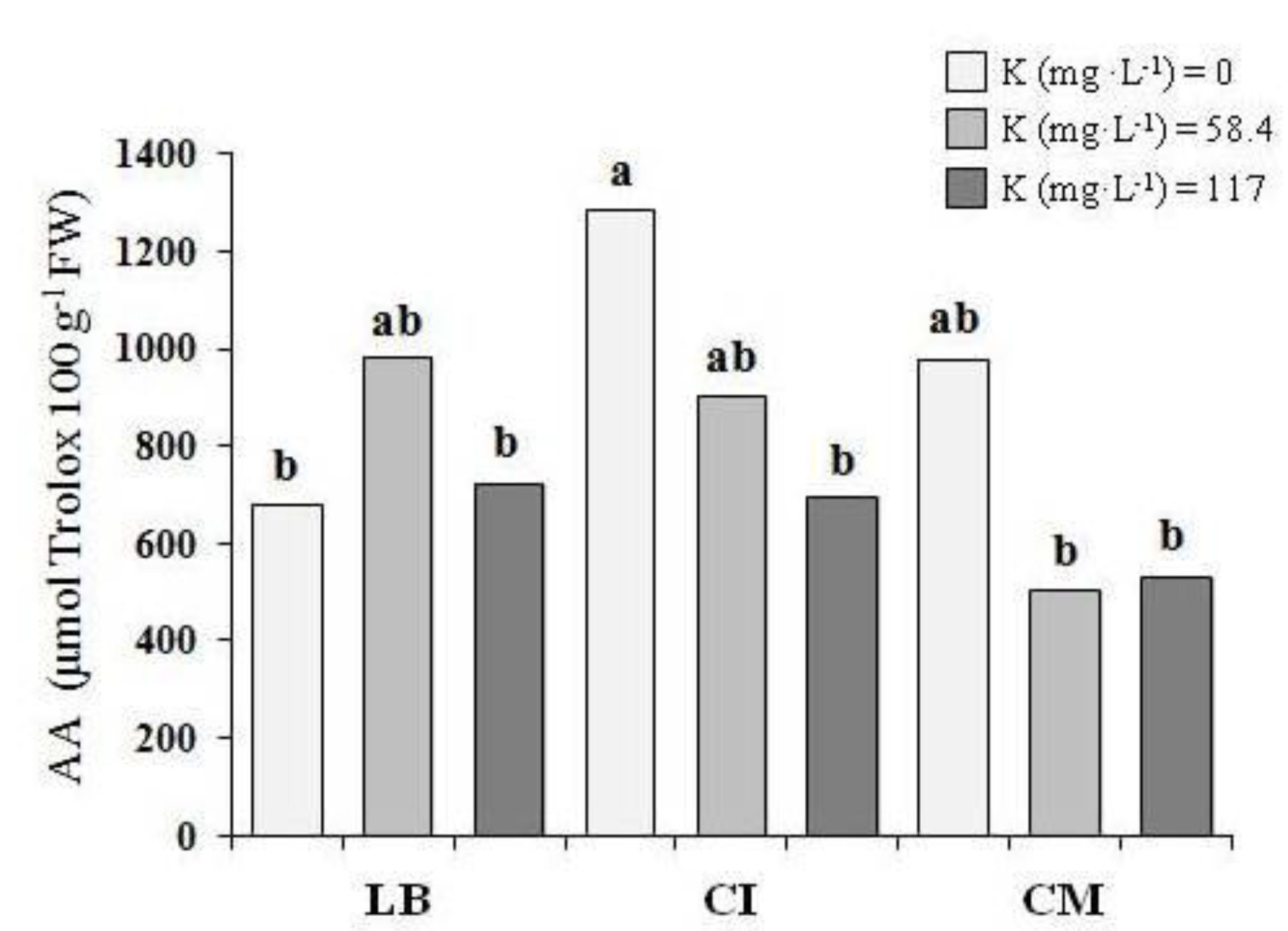
3.3. Proximate Analysis and Antioxidant Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Choi, H.Y.; Ha, S.K. Potassium Balances in Maintenance Hemodialysis. Electrol. Blood Press. 2013, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Guideline: Potassium Intake for Adults and Children; WHO: Geneva, Switzerland, 2012; pp. 1–42. [Google Scholar]
- European Food Safety Authority. Tolerable Upper Intake Levels for Vitamins and Minerals; European Food Safety Authority: Parma, Italy, 2006; ISBN 9291990140. [Google Scholar]
- Ikegami, F.; Wang, Y.; Kaneko, M.; Sumino, M.; Tsukagoshi, S. Human health sciences—From cultivation to utilization of medicinal plants. Open J. Prev. Med. 2012, 2, 214–224. [Google Scholar] [CrossRef]
- Fresenius Medical Care. Annual Report 2013; Fresenius Medical Care: Bad Homburg vor der Höhe, Germany, 2013. [Google Scholar]
- Committee for Revision of Diet Therapy. Guideline Dietary Recommendations for Chronic Kidney Disease. Nihon Jinzo Gakkai Shi 2007, 49, 871–878. [Google Scholar]
- Putcha, N.; Allon, M. Management of Hyperkalemia in Dialysis Patients. Semin. Dial. 2007, 20, 431–439. [Google Scholar] [CrossRef] [PubMed]
- St-Jules, D.E.; Goldfarb, D.S.; Sevick, M.A. Nutrient Non-equivalence: Does Restricting High-Potassium Plant Foods Help to Prevent Hyperkalemia in Hemodialysis Patients? J. Ren. Nutr. 2016, 26, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Weiner, I.D.; Wingo, C.S. Hyperkalemia: A Potential Silent Killer. J. Am. Soc. Nephrol. 1998, 9, 1535–1543. [Google Scholar] [PubMed]
- Renna, M.; Cocozza, C.; Gonnella, M.; Abdelrahman, H.; Santamaria, P. Elemental characterization of wild edible plants from countryside and urban areas. Food Chem. 2015, 177, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Burrowes, J.D.; Ramer, N.J. Changes in potassium content of different potato varieties after cooking. J. Ren. Nutr. 2008, 18, 530–534. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, L.M.; Valente Soares, L.M. Mineral profile of raw and cooked leafy vegetables consumed in Southern Brazil. J. Food Compos. Anal. 2003, 16, 605–611. [Google Scholar] [CrossRef]
- Mazzeo, T.; N’Dri, D.; Chiavaro, E.; Visconti, A.; Fogliano, V.; Pellegrini, N. Effect of two cooking procedures on phytochemical compounds, total antioxidant capacity and colour of selected frozen vegetables. Food Chem. 2011, 128, 627–633. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Artés-Hernández, F.; Colares-Souza, F.; Gómez, P.A.; García-Gómez, P.; Artés, F. Innovative Cooking Techniques for Improving the Overall Quality of a Kailan-Hybrid Broccoli. Food Bioprocess Technol. 2013, 6, 2135–2149. [Google Scholar] [CrossRef]
- Ogawa, A.; Egughi, T.; Toyofoku, K. Cultivation Methods for Leafy Vegetables and Tomatoes with Low Potassium Content for Dialysis Patients. Environ. Control Biol. 2012, 50, 407–414. [Google Scholar] [CrossRef]
- Asao, T.; Asaduzzaman, M.; Mondal, M.F.; Tokura, M.; Adachi, F.; Ueno, M.; Kawaguchi, M.; Yano, S.; Ban, T. Impact of reduced potassium nitrate concentrations in nutrient solution on the growth, yield and fruit quality of melon in hydroponics. Sci. Hortic. (Amst.) 2013, 164, 221–231. [Google Scholar] [CrossRef]
- Tsukagoshi, S.; Hamano, E.; Hohjo, M.; Ikegami, F. Hydroponic Production of Low-Potassium Tomato Fruit for Dialysis Patients. Int. J. Veg. Sci. 2016, 22, 451–460. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; Rouphael, Y.; Di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; De Pascale, S.; Santamaria, P. Micro-scale vegetable production and the rise of microgreens. Trends Food Sci. Technol. 2016, 57, 103–115. [Google Scholar] [CrossRef]
- Renna, M.; Di Gioia, F.; Leoni, B.; Mininni, C.; Santamaria, P. Culinary Assessment of Self-Produced Microgreens as Basic Ingredients in Sweet and Savory Dishes. J. Culin. Sci. Technol. 2017, 15, 126–142. [Google Scholar] [CrossRef]
- Di Gioia, F.; Renna, M.; Santamaria, P. Sprouts, Microgreens and “Baby Leaf” Vegetables. In Minimally Processed Refrigerated Fruits and Vegetables; Springer: Boston, MA, USA, 2017; pp. 403–432. ISBN 978-1-4939-7016-2. [Google Scholar]
- Mir, S.A.; Shah, M.A.; Mir, M.M. Microgreens: Production, shelf life, and bioactive components. Crit. Rev. Food Sci. Nutr. 2017, 57, 2730–2736. [Google Scholar] [CrossRef] [PubMed]
- Kyriacou, M.C.; De Pascale, S.; Kyratzis, A.; Rouphael, Y. Microgreens as a Component of Space Life Support Systems: A Cornucopia of Functional Food. Front. Plant Sci. 2017, 8, 1587. [Google Scholar] [CrossRef] [PubMed]
- AOAC. Official Methods of Analysis of AOAC International, 16th ed.; AOAC International: Rockville, MD, USA, 1995; Volume 1. [Google Scholar]
- Difonzo, G.; Russo, A.; Trani, A.; Paradiso, V.M.; Ranieri, M.; Pasqualone, A.; Summo, C.; Tamma, G.; Silletti, R.; Caponio, F. Green extracts from Coratina olive cultivar leaves: Antioxidant characterization and biological activity. J. Funct. Foods 2017, 31, 63–70. [Google Scholar] [CrossRef]
- D’Imperio, M.; Renna, M.; Cardinali, A.; Buttaro, D.; Santamaria, P.; Serio, F. Silicon biofortification of leafy vegetables and its bioaccessibility in the edible parts. J. Sci. Food Agric. 2016, 96, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil, 2nd ed.; California Agricultural Experiment Station: Davis, CA, USA, 1950; Volume 347. [Google Scholar]
- Cakmak, I. The role of potassium in alleviating detrimental effects of abiotic stresses in plants. J. Plant Nutr. Soil Sci. 2005, 168, 521–530. [Google Scholar] [CrossRef]
- Food and Nutritional Board. Dietary Reference Intakes for Water, Potassium, Sodium, Chloride, and Sulfate; National Academies Press: Washington, DC, USA, 2005; ISBN 978-0-309-09169-5. [Google Scholar]
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.; Ferreira, I.M.P. Comparison between the mineral profile and nitrate content of microgreens and mature lettuces. J. Food Compos. Anal. 2015, 37, 38–43. [Google Scholar] [CrossRef]
- Xiao, Z.; Codling, E.E.; Luo, Y.; Nou, X.; Lester, G.E.; Wang, Q. Microgreens of Brassicaceae: Mineral composition and content of 30 varieties. J. Food Compos. Anal. 2016, 49, 87–93. [Google Scholar] [CrossRef]
- United States Department of Agriculture National Nutrient Database for Standard Reference Release 28. Available online: https://ndb.nal.usda.gov/ndb/search/list?SYNCHRONIZER_TOKEN=c42319cd-7b79-4908-b3e8-5bb7ca42944c&SYNCHRONIZER_URI=%2Fndb%2Fsearch%2Flist&qt=&ds=Standard+Reference&qlookup=&manu= (accessed on 10 April 2018).
- European Parliament and The Council of the European Union. REGULATION (EC) No 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods. Off. J. Eur. Union 2006, 49, 9–25. [Google Scholar]
- Diem, B.; Godbold, D.L. Potassium, calcium and magnesium antagonism in clones of Populus trichocarpa. Plant Soil 1993, 155–156, 411–414. [Google Scholar] [CrossRef]
- Pujos, A.; Morard, P. Effects of potassium deficiency on tomato growth and mineral nutrition at the early production stage. Plant Soil 1997, 189, 189–196. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants; Academic Press: Cambridge, MA, USA, 1995; ISBN 9780080571874. [Google Scholar]
- Flowers, T.J.; Lauchli, A. Sodium versus potassium: Substitution and compartmentation. Encycl. Plant Physiol. 1983, 15, 651–681. [Google Scholar]
- Ogawa, A.; Yamauchi, A. Root Osmotic Adjustment under Osmotic Stress in Maize Seedlings. 2. Mode of Accumulation of Several Solutes for Osmotic Adjustment in the Root. Plant Prod. Sci. 2006, 9, 39–46. [Google Scholar] [CrossRef]
- Delian, E.; Chira, A.; Bădulescu, L.; Chira, L. Scientific Papers Series B Horticulture; University of Agronomic Sciences and Veterinary Medicine: București, Romania, 2012. [Google Scholar]
- Buttaro, D.; Renna, M.; Gerardi, C.; Blando, F.; Santamaria, P.; Serio, F. Soilless production of wild rocket as affected by greenhouse coverage with photovoltaic modules. Acta Sci. Pol. Cultus 2016, 15, 129–142. [Google Scholar]
- Samuolienė, G.; Brazaitytė, A.; Sirtautas, R.; Sakalauskienė, S.; Jankauskienė, J.; Duchovskis, P.; Novičkovas, A. The Impact of supplementary short-term red led lighting on the antioxidant properties of microgreens. Acta Hortic. 2012, 649–656. [Google Scholar] [CrossRef]
- Brazaitytė, A.; Jankauskienė, J.; Novičkovas, A. The effects of supplementary short-term red LEDs lighting on nutritional quality of Perilla frutescens L. Rural Dev. 2013, 2013, 54–58. [Google Scholar]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Xie, Z.; Yu, L.; Wang, Q. Effect of light exposure on sensorial quality, concentrations of bioactive compounds and antioxidant capacity of radish microgreens during low temperature storage. Food Chem. 2014, 151, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.M.; Kwee, E.M.; Niemeyer, E.D. Potassium rate alters the antioxidant capacity and phenolic concentration of basil (Ocimum basilicum L.) leaves. Food Chem. 2010, 123, 1235–1241. [Google Scholar] [CrossRef]

| Yield | Shoot Height | Dry Matter | Na+ | K+ | Mg2+ | Ca2+ | |
|---|---|---|---|---|---|---|---|
| kg FW m−2 | cm | g 100 g−1 FW | mg 100 g−1 FW | ||||
| Genotype (G) | |||||||
| LB | 1.49 b | 3.9 | 6.33 | 20.8 b | 194.4 | 30.7 | 99.2 b |
| CI | 1.44 b | 4.3 | 7.93 | 42.8 a | 224.5 | 30.8 | 127.9 a |
| CM | 1.73 a | 4.0 | 6.88 | 44.5 a | 195.4 | 25.6 | 95.5 b |
| K (mg L−1) | |||||||
| 0 | 1.15 b | 3.4 | 8.28 | 61.5 a | 128.6 b | 38.0 a | 126.8 a |
| 58.4 | 1.81 a | 4.4 | 6.88 | 27.0 b | 224.9 a | 29.8 b | 111.4 ab |
| 117 | 1.77 a | 4.5 | 5.99 | 21.6 b | 250.2 a | 20.1 c | 84.3 b |
| Significance | |||||||
| G | * | NS | NS | *** | NS | NS | * |
| K | * | NS | NS | * | ** | ** | * |
| G*K | * | NS | NS | * | NS | NS | NS |
| Yield | Shoot Height | Dry Matter | Na+ | K+ | Mg2+ | Ca2+ | |
|---|---|---|---|---|---|---|---|
| kg FW m−2 | cm | g 100 g−1 FW | mg 100 g−1 FW | ||||
| Genotype (G) | |||||||
| LB | 1.83 c | 4.6 b | 5.85 | 13.5 b | 154.4 | 31.6 | 101.1 |
| CI | 2.27 b | 5.1 a | 6.24 | 31.1 a | 168.3 | 29.7 | 110.4 |
| CM | 2.53 a | 5.0 a | 5.29 | 32.1 a | 157.4 | 28.7 | 88.1 |
| K (mg L−1) | |||||||
| 0 | 2.08 b | 4.5 | 6.01 | 32.5 | 103.2 b | 33.2 | 107.3 |
| 29.1 | 2.04 b | 4.9 | 5.06 | 20.6 | 129.7 b | 26.2 | 90.4 |
| 58.4 | 2.52 a | 5.3 | 6.30 | 23.7 | 247.1 a | 30.6 | 101.8 |
| Significance | |||||||
| G | *** | ** | NS | *** | NS | NS | NS |
| K | * | NS | NS | NS | * | NS | NS |
| G*K | NS | NS | NS | NS | NS | NS | NS |
| Total Lipid | Protein | Total Carbohydrate | Fiber, Total Dietary | Ashes | Trolox | |
|---|---|---|---|---|---|---|
| g 100 g−1 FW | μmol 100 g−1 FW | |||||
| Genotype (G) | ||||||
| LB | 0.40 | 1.75 | 3.47 b | 0.49 b | 0.81 b | 793.2 ab |
| CI | 0.34 | 2.11 | 4.55 a | 0.79 a | 1.04 a | 959.4 a |
| CM | 0.36 | 1.93 | 3.79 b | 0.80 a | 0.88 ab | 668.5 b |
| K (mg L−1) | ||||||
| 0 | 0.42 | 2.17 | 4.86 | 0.82 | 0.88 | 978.9 |
| 58.4 | 0.37 | 1.93 | 3.74 | 0.65 | 0.95 | 794.9 |
| 117 | 0.32 | 1.68 | 3.20 | 0.60 | 0.90 | 647.3 |
| Significance | ||||||
| G | NS | NS | * | *** | * | * |
| K | NS | NS | NS | NS | NS | NS |
| G*K | NS | NS | NS | NS | NS | * |
| Total Lipid | Protein | Total Carbohydrate | Fiber, Total Dietary | Ashes | Trolox | |
|---|---|---|---|---|---|---|
| g 100 g−1 FW | μmol 100 g−1 FW | |||||
| Genotype (G) | ||||||
| LB | 0.32 | 1.87 | 3.09 | 0.64 b | 0.73 | 457.0 b |
| CI | 0.26 | 1.89 | 3.26 | 0.80 a | 0.83 | 663.0 a |
| CM | 0.30 | 1.64 | 2.78 | 0.64 b | 0.78 | 344.1 b |
| K (mg L−1) | ||||||
| 0 | 0.30 | 1.80 | 3.28 | 0.69 | 0.69 | 531.5 |
| 29.1 | 0.25 | 1.59 | 2.52 | 0.61 | 0.69 | 500.7 |
| 58.4 | 0.31 | 1.97 | 3.19 | 0.77 | 0.93 | 489.0 |
| Significance | ||||||
| G | NS | NS | NS | ** | NS | ** |
| K | NS | NS | NS | NS | NS | NS |
| G*K | NS | NS | NS | NS | NS | NS |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renna, M.; Castellino, M.; Leoni, B.; Paradiso, V.M.; Santamaria, P. Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function. Nutrients 2018, 10, 675. https://doi.org/10.3390/nu10060675
Renna M, Castellino M, Leoni B, Paradiso VM, Santamaria P. Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function. Nutrients. 2018; 10(6):675. https://doi.org/10.3390/nu10060675
Chicago/Turabian StyleRenna, Massimiliano, Maria Castellino, Beniamino Leoni, Vito Michele Paradiso, and Pietro Santamaria. 2018. "Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function" Nutrients 10, no. 6: 675. https://doi.org/10.3390/nu10060675
APA StyleRenna, M., Castellino, M., Leoni, B., Paradiso, V. M., & Santamaria, P. (2018). Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function. Nutrients, 10(6), 675. https://doi.org/10.3390/nu10060675








