Maternal Anemia and Low Birth Weight: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Registration and Protocol
2.2. Eligibility Criteria for the Studies
2.3. Information Sources
2.4. Search Strategies
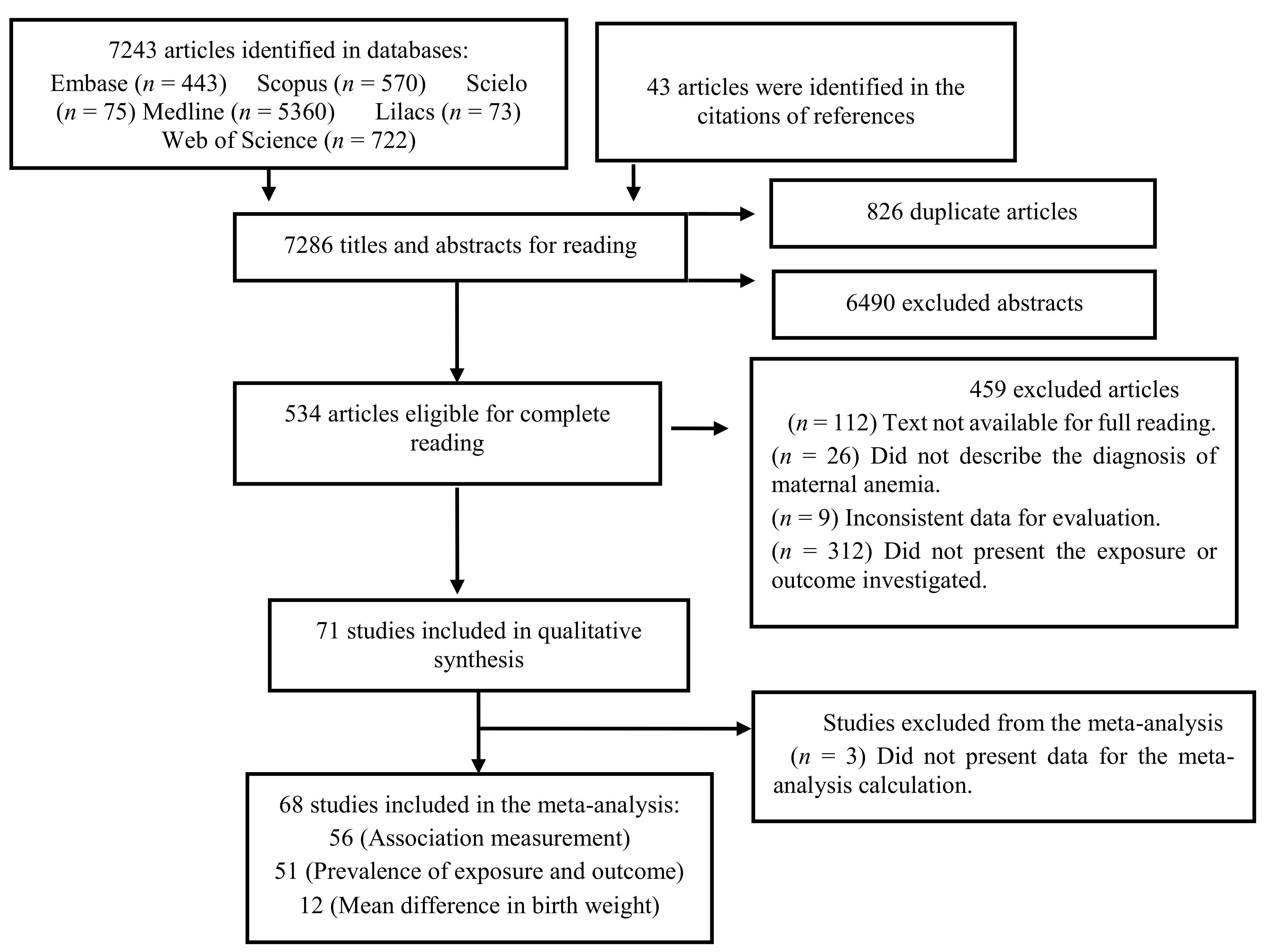
2.5. Studies Selection
2.6. Extraction of Data
2.7. Evaluation of Study Quality
2.8. Data Analysis
3. Results
3.1. Selected Studies
3.2. General Characteristics and Quality of Studies
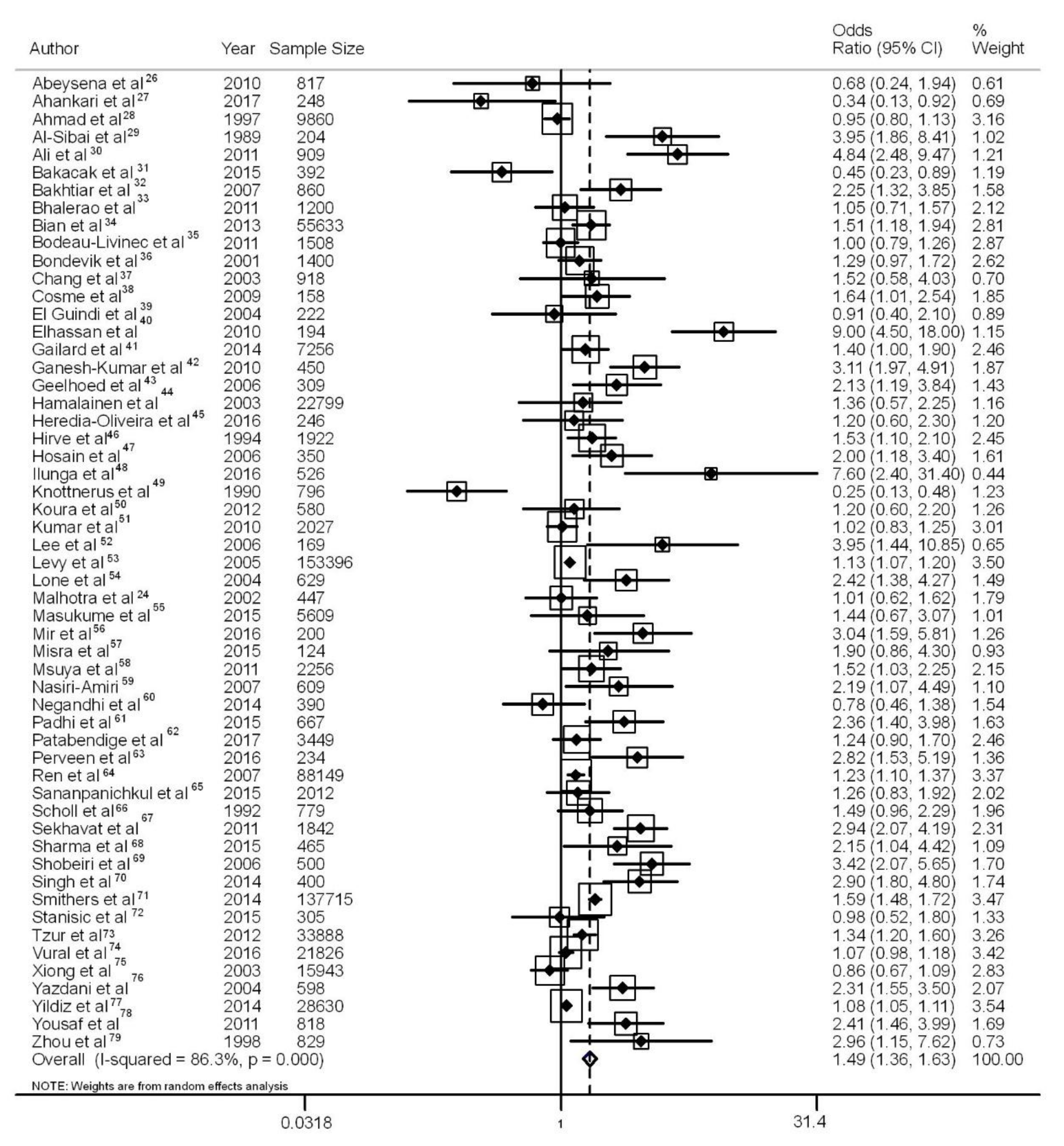
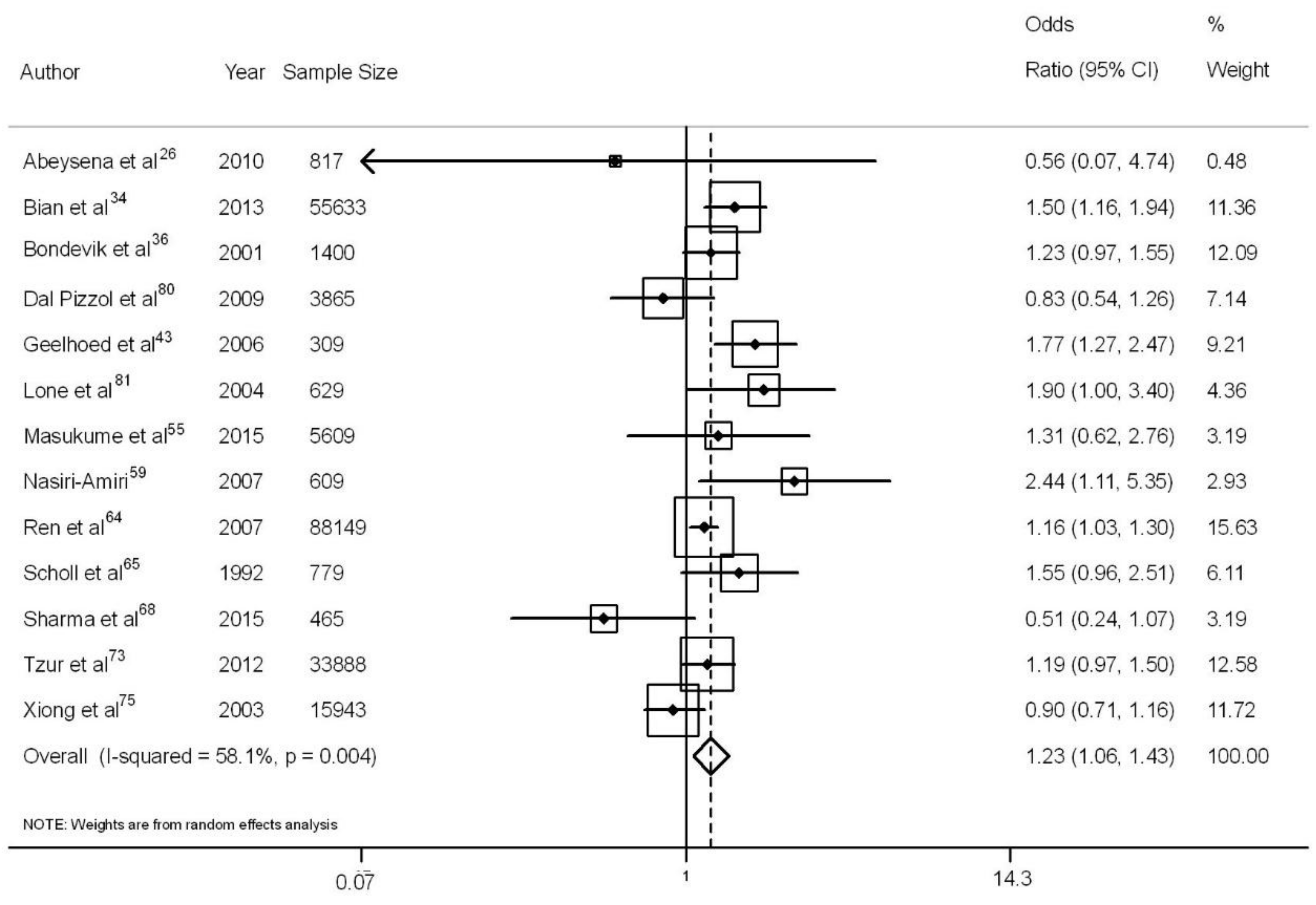
3.3. Maternal Anemia and Low Birth Weight
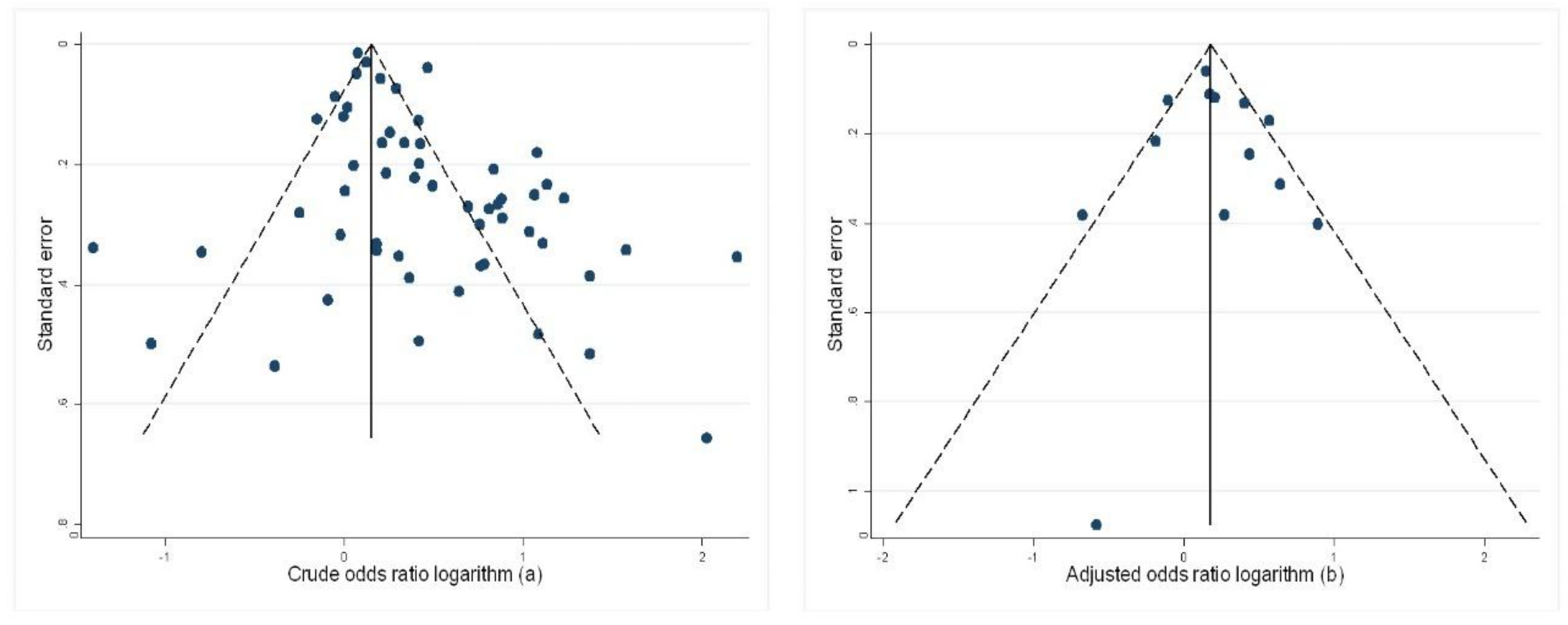
4. Discussion
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- UNICEF. Low Birthweight: Country, Regional and Global Estimates; UNICEF: New York, NY, USA, 2004. [Google Scholar]
- Lawani, L.O.; Iyoke, C.A.; Onoh, R.C.; Nkwo, P.O.; Ibrahim, I.A.; Ekwedigwe, K.C.; Ekine, A.A. Obstetric benefits of health insurance: A comparative analysis of obstetric indices and outcome of enrollees and non-enrollees in southeast Nigeria. J. Obstet. Gynaecol. 2016, 36, 946–949. [Google Scholar] [CrossRef] [PubMed]
- Petrou, S. Economic consequences of preterm birth and low birthweight. Int. J. Obstet. Gynaecol. 2003, 110 (Suppl. 20), 17–23. [Google Scholar] [CrossRef]
- Bird, A.L.; Grant, C.C.; Bandara, D.K.; Mohal, J.; Atatoa-Carr, P.E.; Wise, M.R.; Inskip, H.; Miyahara, M.; Morton, S.M. Maternal health in pregnancy and associations with adverse birth outcomes: Evidence from growing up in New Zealand. Aust. N. Z. J. Obstet. Gynaecol. 2017, 57, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Harvey, E.M.; Strobino, D.; Sherrod, L.; Webb, M.C.; Anderson, C.; White, J.A.; Atlas, R. Community-Academic Partnership to Investigate Low Birth Weight Deliveries and Improve Maternal and Infant Outcomes at a Baltimore City Hospital. Matern. Child Health J. 2017, 21, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Brannon, P.M.; Taylor, C.L. Iron Supplementation during Pregnancy and Infancy: Uncertainties and Implications for Research and Policy. Nutrients 2017, 9, 1327. [Google Scholar] [CrossRef] [PubMed]
- Grotto, H.Z.W. Metabolismo do ferro: Uma revisão sobre os principais mecanismos envolvidos em sua homeostase. Rev. Bras. Hematol. Hemoter. 2008, 30, 390–397. [Google Scholar] [CrossRef]
- World Health Organization (WHO). The Global Prevalence of Anaemia in 2011; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Recommendations to prevent and control iron deficiency in the United States. Centers for Disease Control and Prevention. MMWR Recomm. Rep. 1998, 47, 1–29. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). CDC criteria for anemia in children and childbearing-aged women. MMWR Morb. Mortal. Wkly. Rep. 1989, 38, 400–404. [Google Scholar]
- Stangret, A.; Wnuk, A.; Szewczyk, G.; Pyzlak, M.; Szukiewicz, D. Maternal hemoglobin concentration and hematocrit values may affect fetus development by influencing placental angiogenesis. J. Matern. Fetal Neonatal Med. 2017, 30, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Abe, S.K.; Rahman, M.S.; Kanda, M.; Narita, S.; Bilano, V.; Ota, E.; Gilmour, S.; Shibuya, K. Maternal anemia and risk of adverse birth and health outcomes in low-and middle-income countries: Systematic review and meta-analysis. Am. J. Clin. Nutr. 2016, 103, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Sukrat, B.; Wilasrusmee, C.; Siribumrungwong, B.; McEvoy, M.; Okascharoen, C.; Attia, J.; Thakkinstian, A. Hemoglobin concentration and pregnancy outcomes: A systematic review and meta-analysis. Biomed. Res. Int. 2013, 2013, 769057. [Google Scholar] [CrossRef] [PubMed]
- Peña-Rosas, J.P.; De-Regil, L.M.; Garcia-Casal, M.N.; Dowswell, T. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015, CD004736. [Google Scholar] [CrossRef] [PubMed]
- Peña-Rosas, J.P.; De-Regil, L.M.; Gomez Malave, H.; Flores-Urrutia, M.C.; Dowswell, T. Intermittent oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015, CD009997. [Google Scholar] [CrossRef] [PubMed]
- Peña-Rosas, J.P.; De-Regil, L.M.; Dowswell, T.; Viteri, F.E. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2012, 12, CD004736. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; The Ottawa Hospital: Ottawa, ON, Canada, 2014. [Google Scholar]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.F.; Tukey, J.W. Transformations related to the angular and the square root. Ann. Math. Stat. 1950, 21, 607–611. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, K.F. What’s the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. J. Am. Med. Assoc. 1998, 280, 1690–1691. [Google Scholar] [CrossRef]
- Buzyan, L.O. Mild anemia as a protective factor against pregnancy loss. Int. J. Risk Saf. Med. 2015, 27 (Suppl. 1), S7–S8. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, S.A.; Hatcher, J.; Jehan, I.; Qureshi, R. Maternal risk factors associated with low birth weight in Karachi: A case-control study. East. Mediterr. Health J. 2007, 13, 1343–1352. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, O.T.S.; Szarfarc, S.C.; d’Aquino Benicio, M.H. Anemia e desnutrição maternas e sua relação com o peso ao nascer. Rev. Saúde Pública 1991, 25, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, M.; Sharma, J.B.; Batra, S.; Sharma, S.; Murthy, N.S.; Arora, R. Maternal and perinatal outcome in varying degrees of anemia. Int. J. Gynaecol. Obstet. 2002, 79, 93–100. [Google Scholar] [CrossRef]
- Duthie, S.J.; King, P.A.; To, W.K.; Lopes, A.; Ma, H.K. A case controlled study of pregnancy complicated by severe maternal anaemia. Aust. N. Z. J. Obstet. Gynaecol. 1991, 31, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Abeysena, C.; Jayawardana, P.; de A Seneviratne, R. Maternal haemoglobin level at booking visit and its effect on adverse pregnancy outcome. Aust. N. Z. J. Obstet. Gynaecol. 2010, 50, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Ahankari, A.S.; Myles, P.R.; Dixit, J.V.; Tata, L.J.; Fogarty, A.W. Risk factors for maternal anaemia and low birth weight in pregnant women living in rural India: A prospective cohort study. Public Health. 2017, 151, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Jaafar, R.; Hassan, M.M.; Othman, M.S.; Hashim, A. Anaemia during pregnancy in rural Kelantan. Malays. J. Nutr. 1997, 3, 83–90. [Google Scholar] [PubMed]
- Al-Sibai, M.H.; Khwaja, S.S.; al-Suleiman, S.A.; Magbool, G. The low birth-weight infants of Saudi adolescents: Maternal implications. Aust. N. Z. J. Obstet. Gynaecol. 1987, 27, 320–322. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A.; Rayis, D.A.; Abdallah, T.M.; Elbashir, M.I.; Adam, I. Severe anaemia is associated with a higher risk for preeclampsia and poor perinatal outcomes in Kassala hospital, eastern Sudan. BMC Res. Notes 2011, 4, 311. [Google Scholar] [CrossRef] [PubMed]
- Bakacak, M.; Avci, F.; Ercan, O.; Köstü, B.; Serin, S.; Kiran, G.; Bostanci, M.S.; Bakacak, Z. The effect of maternal hemoglobin concentration on fetal birth weight according to trimesters. J. Matern. Fetal Neonatal Med. 2015, 28, 2106–2110. [Google Scholar] [CrossRef] [PubMed]
- Bakhtiar, U.J.; Khan, Y.; Nasar, R. Relationship between maternal hemoglobin and Perinatal outcome. Rawal Med. J. 2007, 32, 102–104. [Google Scholar]
- Bhalerao, A.; Kawthalkar, A.; Ghike, S.; Joshi, S. Anemia during Pregnancy: Most Preventableyet most Prevalent. J. South Asian Fed. Obstet. Gynaecol. 2011, 3, 75–77. [Google Scholar] [CrossRef]
- Bian, Y.; Zhang, Z.; Liu, Q.; Wu, D.; Wang, S. Maternal risk factors for low birth weight for term births in a developed region in China: A hospital-based study of 55,633 pregnancies. J. Biomed. Res. 2013, 27, 14–22. [Google Scholar] [PubMed]
- Bodeau-Livinec, F.; Briand, V.; Berger, J.; Xiong, X.; Massougbodji, A.; Day, K.P.; Cot, M. Maternal anemia in Benin: Prevalence, risk factors, and association with low birth weight. Am. J. Trop. Med. Hyg. 2011, 85, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Bondevik, G.T.; Lie, R.T.; Ulstein, M.; Kvåle, G. Maternal hematological status and risk of low birth weight and preterm delivery in Nepal. Acta Obstet. Gynecol. Scand. 2001, 80, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.C.; O’Brien, K.O.; Nathanson, M.S.; Mancini, J.; Witter, F.R. Hemoglobin concentrations influence birth outcomes in pregnant African-American adolescents. J. Nutr. 2003, 133, 2348–2355. [Google Scholar] [CrossRef] [PubMed]
- Cosme, J.A.G.; Hechavarría, E.M.; Miralles, R.M.F.; Isaac, R.C.; Blanco, Y.V. Factores de riesgo del bajo peso al nacer en el hospital materno de Palma Soriano durante un trienio Risk factors of low birth weight in Palma Soriano Maternal Hospital during a triennium. Medisan 2009, 13, 1–7. [Google Scholar]
- El Guindi, W.; Pronost, J.; Carles, G.; Largeaud, M.; El Gareh, N.; Montoya, Y.; Arbeille, P. Anémies maternelles sévèreset issues de grossesse. J. Gynecol. Obstet. Biol. Reprod. 2004, 33, 506–509. [Google Scholar] [CrossRef]
- Elhassan, E.M.; Abbaker, A.O.; Haggaz, A.D.; Abubaker, M.S.; Adam, I. Anaemia and low birth weight in Medani, Hospital Sudan. BMC Res. Notes 2010, 3, 181. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, R.; Eilers, P.H.; Yassine, S.; Hofman, A.; Steegers, E.A.; Jaddoe, V.W. Risk factors and consequences of maternal anaemia and elevated haemoglobin levels during pregnancy: A population-based prospective cohort study. Paediatr. Perinat. Epidemiol. 2014, 28, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Ganesh Kumar, S.; Harsha Kumar, H.N.; Jayaram, S.; Kotian, M.S. Determinants of low birth weight: A case control study in a district hospital in Karnataka. Indian J. Pediatr. 2010, 77, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Geelhoed, D.; Agadzi, F.; Visser, L.; Ablordeppey, E.; Asare, K.; O’Rourke, P.; van Leeuwen, J.S.; van Roosmalen, J. Maternal and fetal outcome after severe anemia in pregnancy in rural Ghana. Acta Obstet. Gynecol. Scand. 2006, 85, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Hämäläinen, H.; Hakkarainen, K.; Heinonen, S. Anaemia in the first but not in the second or third trimester is a risk factor for low birth weight. Clin. Nutr. 2003, 22, 271–275. [Google Scholar] [CrossRef]
- Heredia-Olivera, K.; Munares-García, O. Factores maternos asociados al bajo peso al nacer. Rev. Med. Inst. Mex. Seguro Soc. 2016, 54, 562–567. [Google Scholar] [PubMed]
- Hirve, S.S.; Ganatra, B.R. Determinants of low birth weight: A community based prospective cohort study. Indian Pediatr. 1994, 31, 1221–1225. [Google Scholar] [PubMed]
- Hosain, G.M.; Chatterjee, N.; Begum, A.; Saha, S.C. Factors associated with low birthweight in rural Bangladesh. J. Trop. Pediatr. 2006, 52, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Ilunga, P.M.; Mukuku, O.; Mawaw, P.M.; Mutombo, A.M.; Lubala, T.K.; Shongo Ya Pongombo, M.; Kakudji Luhete, P.; Wembonyama, S.O.; Mutombo Kabamba, A.; Luboya Numbi, O. Risk factors for low birth weight in Lubumbashi, Democratic Republic of the Congo. Med. Sante Trop. 2016, 26, 386–390. [Google Scholar] [PubMed]
- Knottnerus, J.A.; Delgado, L.R.; Knipschild, P.G.; Essed, G.G.; Smits, F. Haematologic parameters and pregnancy outcome. A prospective cohort study in the third trimester. J. Clin. Epidemiol. 1990, 43, 461–466. [Google Scholar] [CrossRef]
- Koura, G.K.; Ouedraogo, S.; Le Port, A.; Watier, L.; Cottrell, G.; Guerra, J.; Massougbodji, A.; Garcia, A. Anaemia during pregnancy: Impact on birth outcome and infant haemoglobin level during the first 18 months of life. Trop. Med. Int. Health 2012, 17, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Chaudhary, K.; Prasad, S. Maternal indicators and obstetric outcome in the north Indian population: A hospital-based study. J. Postgrad. Med. 2010, 56, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Kim, M.S.; Kim, M.H.; Kim, Y.J.; Kim, W.Y. Iron status and its association with pregnancy outcome in Korean pregnant women. Eur. J. Clin. Nutr. 2006, 60, 1130–1135. [Google Scholar] [CrossRef] [PubMed]
- Levy, A.; Fraser, D.; Katz, M.; Mazor, M.; Sheiner, E. Maternal anemia during pregnancy is an independent risk factor for low birthweight and preterm delivery. Eur. J. Obstet. Gynecol. Reprod. Biol. 2005, 122, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Lone, F.W.; Qureshi, R.N.; Emanuel, F. Maternal anaemia and its impact on perinatal outcome. Trop. Med. Int. Health 2004, 9, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Masukume, G.; Khashan, A.S.; Kenny, L.C.; Baker, P.N.; Nelson, G.; Consortium, S. Risk factors and birth outcomes of anaemia in early pregnancy in a nulliparous cohort. PLoS ONE 2015, 10, e0122729. [Google Scholar] [CrossRef] [PubMed]
- Mir, M.K.; Yousaf, S.; Ilyas, A.; Tahir, S. Maternal anemia is a risk factor for low birth weight babies at Term. Pak. J. Med. Health Sci. 2016, 10, 741–743. [Google Scholar]
- Misra, A.; Ray, S.; Patrikar, S. A longitudinal study to determine association of various maternal factors with neonatal birth weight at a tertiary care hospital. Med. J. Armed Forces India 2015, 71, 270–273. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Msuya, S.E.; Hussein, T.H.; Uriyo, J.; Sam, N.E.; Stray-Pedersen, B. Anaemia among pregnant women in northern Tanzania: Prevalence, risk factors and effect on perinatal outcomes. Tanzan. J. Health Res. 2011, 13, 33–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nasiri-Amiri, F.; Hajiahmadi, M.; Basirat, Z. Maternal hematocrit status affecting pregnancy outcome. Iran. Red Crescent Med. J. 2007, 9, 104–108. [Google Scholar]
- Negandhi, P.H.; Negandhi, H.N.; Zodpey, S.P.; Ughade, S.N.; Biranjan, J.R. Risk factors for low birth weight in an Indian urban setting: A nested case control study. Asia Pac. J. Public Health 2014, 26, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Padhi, B.K.; Baker, K.K.; Dutta, A.; Cumming, O.; Freeman, M.C.; Satpathy, R.; Das, B.S.; Panigrahi, P. Risk of adverse pregnancy outcomes among women practicing poor sanitation in rural India: A population-based prospective cohort study. PLoS Med. 2015, 12, e1001851. [Google Scholar] [CrossRef] [PubMed]
- Patabendige, M.; Dias, T.; Gunasena, J.; Motha, C.M.B.; Palihawadana, T.; Herath, R.P.; Wijesinghe, P.S. Retrospective study on anaemia in pregnancy: Effect on fetal and labour outcomes. In Proceedings of the RCOG World Congress 2017, Cape Town, South Africa, 20–22 March 2017. [Google Scholar]
- Perveen, S.; Soomro, T.K. Sideropaenic anaemia: Impact on perinatal outcome at tertiary care hospital. J. Pak. Med. Assoc. 2016, 66, 952–956. [Google Scholar] [PubMed]
- Ren, A.; Wang, J.; Ye, R.W.; Li, S.; Liu, J.M.; Li, Z. Low first-trimester hemoglobin and low birth weight, preterm birth and small for gestational age newborns. Int. J. Gynaecol. Obstet. 2007, 98, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Sananpanichkul, P.; Rujirabanjerd, S. Association between maternal body mass index and weight gain with low birth weight in eastern Thailand. Southeast Asian J. Trop. Med. Public Health 2015, 46, 1085–1091. [Google Scholar] [PubMed]
- Scholl, T.O.; Hediger, M.L.; Fischer, R.L.; Shearer, J.W. Anemia vs iron deficiency: Increased risk of preterm delivery in a prospective study. Am. J. Clin. Nutr. 1992, 55, 985–988. [Google Scholar] [CrossRef] [PubMed]
- Sekhavat, L.; Davar, R.; Hosseinidezoki, S. Relationship between maternal hemoglobin concentration and neonatal birth weight. Hematology 2011, 16, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.R.; Giri, S.; Timalsina, U.; Bhandari, S.S.; Basyal, B.; Wagle, K.; Shrestha, L. Low birth weight at term and its determinants in a tertiary hospital of Nepal: A case-control study. PLoS ONE 2015, 10, e0123962. [Google Scholar] [CrossRef] [PubMed]
- Shobeiri, F.; Begumb, K.; Nazari, M. A prospective study of maternal hemoglobin status of Indian women during pregnancy and pregnancy outcome. Nutr. Res. 2006, 26, 209–213. [Google Scholar] [CrossRef]
- Singh, A.; Arya, S.; Chellani, H.; Aggarwal, K.C.; Pandey, R.M. Prediction model for low birth weight and its validation. Indian J. Pediatr. 2014, 81, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Smithers, L.G.; Gialamas, A.; Scheil, W.; Brinkman, S.; Lynch, J.W. Anaemia of pregnancy, perinatal outcomes and children’s developmental vulnerability: A whole-of-population study. Paediatr. Perinat. Epidemiol. 2014, 28, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Stanisic, D.I.; Moore, K.A.; Baiwog, F.; Ura, A.; Clapham, C.; King, C.L.; Siba, P.M.; Beeson, J.G.; Mueller, I.; Fowkes, F.J.; et al. Risk factors for malaria and adverse birth outcomes in a prospective cohort of pregnant women resident in a high malaria transmission area of Papua New Guinea. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Tzur, T.; Weintraub, A.Y.; Sergienko, R.; Sheiner, E. Can anemia in the first trimester predict obstetrical complications later in pregnancy? J. Matern. Fetal Neonatal Med. 2012, 25, 2454–2457. [Google Scholar] [CrossRef] [PubMed]
- Vural, T.; Toz, E.; Ozcan, A.; Biler, A.; Ileri, A.; Inan, A.H. Can anemia predict perinatal outcomes in different stages of pregnancy? Pak. J. Med. Sci. 2016, 32, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Buekens, P.; Fraser, W.D.; Guo, Z. Anemia during pregnancy in a Chinese population. Int. J. Gynaecol. Obstet. 2003, 83, 159–164. [Google Scholar] [CrossRef]
- Yazdani, M.; Tadbiri, M.; Shakeri, S. Maternal hemoglobin level, prematurity, and low birth weight. Int. J. Gynaecol. Obstet. 2004, 85, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, Y.; Özgü, E.; Unlu, S.B.; Salman, B.; Eyi, E.G. The relationship between third trimester maternal hemoglobin and birth weight/length; results from the tertiary center in Turkey. J. Matern. Fetal Neonatal Med. 2014, 27, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, F.; Haider, G.; Shaikh, R.B.; Haider, A.; Muhammad, N. Impact of maternal anemia on perinatal outcome. Pak. Armed Forces Med. J. 2011, 60, 1–3. [Google Scholar]
- Zhou, L.M.; Yang, W.W.; Hua, J.Z.; Deng, C.Q.; Tao, X.; Stoltzfus, R.J. Relation of hemoglobin measured at different times in pregnancy to preterm birth and low birth weight in Shanghai, China. Am. J. Epidemiol. 1998, 148, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Dal Pizzol, T.S.; Giugliani, E.R.J.; Mengue, S.S. Associação entre o uso de sais de ferro durante a gestação e nascimento pré-termo, baixo peso ao nascer e muito baixo peso ao nascer. Cad. Saúde Pública 2009, 25, 106–168. [Google Scholar] [CrossRef]
- Lone, F.W.; Qureshi, R.N.; Emmanuel, F. Maternal anaemia and its impact on perinatal outcome in a tertiary care hospital in Pakistan. East. Mediterr. Health J. 2004, 10, 801–807. [Google Scholar] [PubMed]
- Human Development Report, 2016. Available online: http://hdr.undp.org/sites/default/files/2016_human_development_report.pdf (accessed on 11 May 2018).
- Rukuni, R.; Bhattacharya, S.; Murphy, M.F.; Roberts, D.; Stanworth, S.J.; Knight, M. Maternal and neonatal outcomes of antenatal anemia in a Scottish population: A retrospective cohort study. Acta Obstet. Gynecol. Scand. 2016, 95, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Steer, P.; Alam, M.A.; Wadsworth, J.; Welch, A. Relation between maternal haemoglobin concentration and birth weight in different ethnic groups. Br. Med. J. 1995, 310, 489–491. [Google Scholar] [CrossRef]
- López, J.I.; Botell, M.L.; Echevarría, S.M.M.; Pérez, C.G.; Valdés-Dapena, D.P. Algunos factores de riesgo relacionados con el bajo peso al nacer. Rev. Cubana Ginecol. Obstet. 2012, 38, 45–55. [Google Scholar]
- Bánhidy, F.; Acs, N.; Puhó, E.H.; Czeizel, A.E. Iron deficiency anemia: Pregnancy outcomes with or without iron supplementation. Nutrition 2011, 27, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.; Jo, L.-B. Maternal hemoglobin and birth weight: Systematic review and meta-analysis. Int. J. Med. Sci. Public Health 2015, 4, 435–445. [Google Scholar]
- Rahmati, S.; Delpishe, A.; Azami, M.; Hafezi Ahmadi, M.R.; Sayehmiri, K. Maternal Anemia during pregnancy and infant low birth weight: A systematic review and Meta-analysis. Int. J. Reprod. Biomed. 2017, 15, 125–134. [Google Scholar] [CrossRef]
- WHO. WHA Global Nutrition Targets 2025: Low Birth Weight Policy Brief; World Health Organization: Geneva, Switzerland, 2014; p. 7. [Google Scholar]
- Friedrisch, J.R.; Friedrisch, B.K. Prophylactic Iron Supplementation in Pregnancy: A Controversial Issue. Biochem. Insights 2017. [Google Scholar] [CrossRef] [PubMed]
- Balshem, H.; Helfand, M.; Schünemann, H.J.; Oxman, A.D.; Kunz, R.; Brozek, J.; Vist, G.E.; Falck-Ytter, Y.; Meerpohl, J.; Norris, S.; et al. GRADE guidelines: 3. Rating the quality of evidence. J. Clin. Epidemiol. 2011, 64, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Woodcock, J.; Brozek, J.; Helfand, M.; Alonso-Coello, P.; Falck-Ytter, Y.; Jaeschke, R.; Vist, G.; et al. GRADE guidelines: 8. Rating the quality of evidence—Indirectness. J. Clin. Epidemiol. 2011, 64, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Montori, V.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Djulbegovic, B.; Atkins, D.; Falck-Ytter, Y.; et al. GRADE guidelines: 5. Rating the quality of evidence—Publication bias. J. Clin. Epidemiol. 2011, 64, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Schünemann, H.J.; Tugwell, P.; Knottnerus, A. GRADE guidelines: A new series of articles. J. Clin. Epidemiol. 2011, 64, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Montori, V.; Akl, E.A.; Djulbegovic, B.; Falck-Ytter, Y.; et al. GRADE guidelines: 4. Rating the quality of evidence—Study limitations (risk of bias). J. Clin. Epidemiol. 2011, 64, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. J. Am. Med. Assoc. 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
| Characteristic | N | % |
|---|---|---|
| Study design | ||
| Prospective cohort | 38 | 53.6 |
| Retrospective cohort | 16 | 22.5 |
| Case-control | 17 | 23.9 |
| Location of data collection | ||
| Hospital | 58 | 81.7 |
| Primary health units | 7 | 9.9 |
| Community | 3 | 8.4 |
| Differential diagnosis of exposure | ||
| Maternal anemia | 65 | 91.6 |
| Iron deficiency anemia | 6 | 8.4 |
| Hemoglobin levels * | ||
| <11 g/dL | 54 | 77.1 |
| <10 g/dL | 10 | 14.3 |
| <8 g/dL | 6 | 8.6 |
| Geographic region | ||
| America | 9 | 12.7 |
| Africa | 6 | 8.4 |
| Asia | 44 | 62.0 |
| Europe | 8 | 11.3 |
| Oceania | 4 | 5.6 |
| Sample size | ||
| ≤1000 | 43 | 60.6 |
| >1000 | 28 | 39.4 |
| Methodological quality of the studies | ||
| Moderate | 40 | 56.3 |
| High | 31 | 43.7 |
| Year the research was initiated ** | ||
| Before 1990 | 8 | 14.0 |
| 1990–2009 | 12 | 21.0 |
| 2000–2009 | 25 | 44.0 |
| 2010–2017 | 12 | 21.0 |
| Control for confounding | ||
| Yes | 20 | 28.2 |
| No | 51 | 71.8 |
| Variable | N | Crude OR (95% CI) | Heterogeneity (I2) | Meta-Regression p Value * | N | Adjusted OR (95% CI) | Heterogeneity (I2) | Meta-Regression p Value * |
|---|---|---|---|---|---|---|---|---|
| Study design | ||||||||
| Prospective cohort | 30 | 1.51 (1.29–1.76) | 83.8% | 6 | 1.38 (0.96–2.01) | 46.7% | ||
| Retrospective cohort | 13 | 1.18 (1.08–1.28) | 72.7% | 0.03 | 5 | 1.24 (1.04–1.49) | 70.8% | 0.56 |
| Case-control | 12 | 2.29 (1.51–3.47) | 82.8% | 2 | 0.85 (0.37–2.00) | 79.4% | ||
| Data collection location | ||||||||
| Hospital | 44 | 1.54 (1.39–1.71) | 85.4% | 10 | 1.30 (1.06–1.59) | 63.4% | ||
| Primary health units | 6 | 1.28 (1.02–1.61) | 68.9% | 0.28 | 2 | 1.05 (0.77–1.42) | 55.2% | 0.50 |
| Community | 5 | 1.40 (1.09–1.78) | 60.4% | 1 | - | - | ||
| Differential diagnosis of exposure | ||||||||
| Maternal anemia | 51 | 1.47 (1.34–1.61) | 86.8% | 0.54 | 12 | 1.21 (1.04–1.42) | 60.0% | 0.53 |
| Iron deficiency anemia | 4 | 1.89 (1.10–3.27) | 60.9% | 1 | - | - | ||
| Hemoglobin levels (g/dL) | ||||||||
| <11 | 33 | 1.48 (1.32–1.66) | 86.7% | 10 | 1.25 (1.04–1.49) | 50.3% | ||
| <10.6 | 5 | 2.58 (1.69–3.94) | 72.4% | 0.87 | 1 | - | - | 0.84 |
| <10 | 7 | 1.25 (1.02–1.52) | 86.2% | 2 | 1.04 (0.79–1.37) | 64.0% | ||
| <8 | 3 | 1.72 (1.10–2.69) | 33.6% | 0 | - | - | ||
| Severity levels of maternal anemia | ||||||||
| Mild | 7 | 1.14 (0.99–1.31) | 66.3% | 0 | - | - | ||
| Moderate | 9 | 1.39 (1.11–1.74) | 49.0% | 0.61 | 2 | 1.16 (1.04–1.29) | 0.0% | - |
| Severe | 9 | 2.31 (1.47–3.63) | 82.7% | 4 | 2.24 (1.35–3.71) | 0.0% | ||
| Odds ratio | ||||||||
| <2 | 34 | 1.15 (1.06–1.25) | 80.1% | <0.01 | 10 | 1.14 (0.99–1.30) | 47.8% | 0.02 |
| ≥2 | 21 | 2.85 (2.48–3.27) | 16.0% | 3 | 1.87 (1.42–2.45) | 0.0% | ||
| Maternal anemia by gestational trimester | ||||||||
| First | 3 | 1.51 (0.94–2.42) | 55.7% | 0 | - | - | ||
| Second | 3 | 0.98 (0.56–1.68) | 61.5% | - | 0 | - | - | - |
| Third | 3 | 0.88 (0.53–1.48) | 72.2% | 0 | - | - | ||
| Human Development Index ** | ||||||||
| Very high | 12 | 1.24 (1.03–1.49) | 88.4% | 3 | 1.25 (1.03–1.51) | 0.0% | ||
| High | 15 | 1.30 (1.14–1.49) | 82.0% | 0.02 | 6 | 1.15 (0.92–1.44) | 64.8% | 0.92 |
| Medium | 21 | 1.82 (1.46–2.27) | 77.3% | 4 | 1.30 (0.87–1.94) | 72.4% | ||
| Low | 7 | 2.28 (1.23–4.21) | 89.5% | 1 | - | - | ||
| Geographic region | ||||||||
| America | 4 | 1.38 (1.00–1.91) | 0.0% | 1 | - | - | ||
| Africa | 6 | 3.07 (1.60–5.89) | 83.9% | 1 | - | - | 0.76 | |
| Asia | 38 | 1.44 (1.31–1.58) | 83.7% | 0.30 | 9 | 1.20 (1.02–1.40) | 58.2% | |
| Europe | 4 | 0.94 (0.47–1.89) | 87.4% | 1 | - | - | ||
| Oceania | 3 | 1.51 (1.23–1.85) | 16.0% | 1 | - | - | ||
| Sample size | ||||||||
| ≤1000 | 34 | 1.84 (1.46–2.33) | 79.0% | 0.02 | 6 | 1.46 (1.00–2.16) | 57.2% | 0.15 |
| >1000 | 21 | 1.24 (1.13–1.35) | 86.8% | 7 | 1.15 (1.02–1.31) | 44.1% | ||
| Year the research was initiated | ||||||||
| Before 1990 | 4 | 0.96 (0.58–1.60) | 91.5% | 1 | - | - | ||
| 1990–1999 | 9 | 1.34 (1.10–1.64) | 81.1% | 0.36 | 4 | 1.22 (1.00–1.50) | 64.6% | 0.08 |
| 2000–2009 | 22 | 1.75 (1.44–2.13) | 82.3% | 6 | 1.40 (1.16–1.69) | 16.0% | ||
| 2010–2017 | 11 | 1.32 (1.09–1.60) | 87.2% | 0 | - | - | ||
| Methodological quality of the studies | ||||||||
| Moderate | 30 | 1.65 (1.42–1.91) | 83.2% | 0.25 | 2 | 0.52 (0.26–1.04) | 0.0% | 0.05 |
| High | 25 | 1.49 (1.36–1.63) | 87.0% | 13 | 1.27 (1.10–1.46) | 56.6% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Figueiredo, A.C.M.G.; Gomes-Filho, I.S.; Silva, R.B.; Pereira, P.P.S.; Mata, F.A.F.D.; Lyrio, A.O.; Souza, E.S.; Cruz, S.S.; Pereira, M.G. Maternal Anemia and Low Birth Weight: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 601. https://doi.org/10.3390/nu10050601
Figueiredo ACMG, Gomes-Filho IS, Silva RB, Pereira PPS, Mata FAFD, Lyrio AO, Souza ES, Cruz SS, Pereira MG. Maternal Anemia and Low Birth Weight: A Systematic Review and Meta-Analysis. Nutrients. 2018; 10(5):601. https://doi.org/10.3390/nu10050601
Chicago/Turabian StyleFigueiredo, Ana C. M. G., Isaac S. Gomes-Filho, Roberta B. Silva, Priscilla P. S. Pereira, Fabiana A. F. Da Mata, Amanda O. Lyrio, Elivan S. Souza, Simone S. Cruz, and Mauricio G. Pereira. 2018. "Maternal Anemia and Low Birth Weight: A Systematic Review and Meta-Analysis" Nutrients 10, no. 5: 601. https://doi.org/10.3390/nu10050601
APA StyleFigueiredo, A. C. M. G., Gomes-Filho, I. S., Silva, R. B., Pereira, P. P. S., Mata, F. A. F. D., Lyrio, A. O., Souza, E. S., Cruz, S. S., & Pereira, M. G. (2018). Maternal Anemia and Low Birth Weight: A Systematic Review and Meta-Analysis. Nutrients, 10(5), 601. https://doi.org/10.3390/nu10050601





