Dietary Neurotransmitters: A Narrative Review on Current Knowledge
Abstract
:1. Introduction
2. Overview of Dietary Neurotransmitters
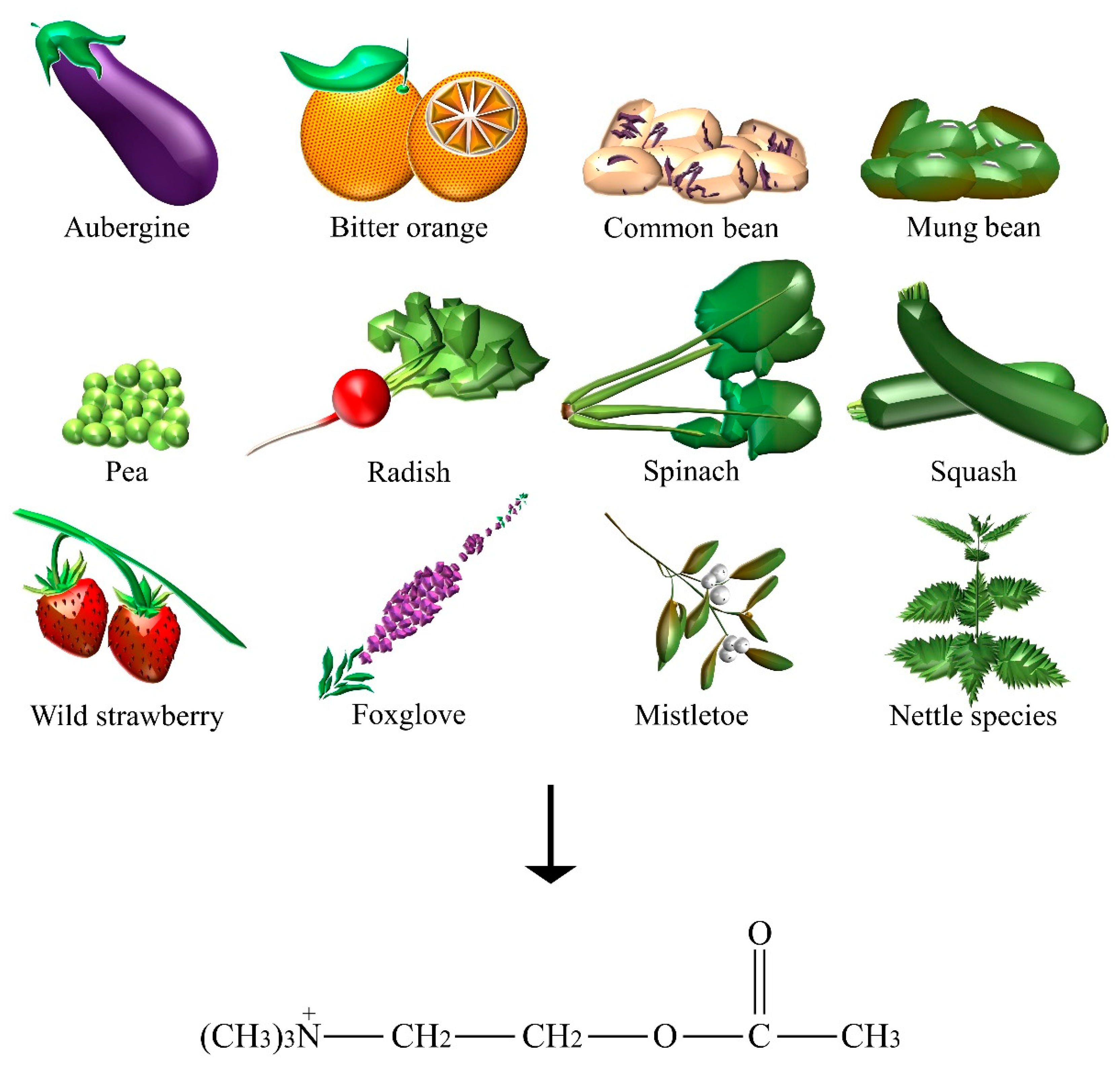
2.1. Acetylcholine
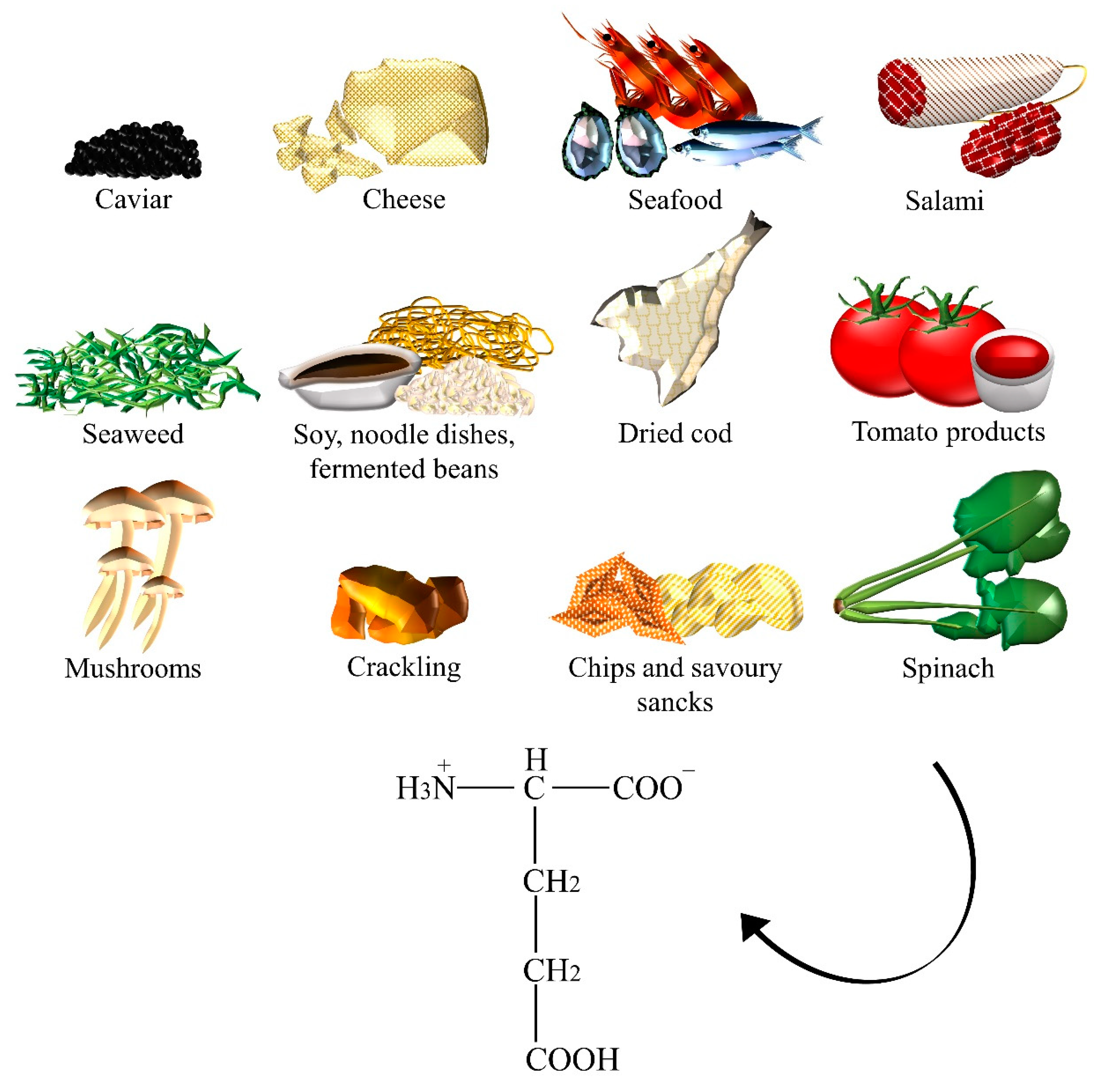
2.2. Glutamate
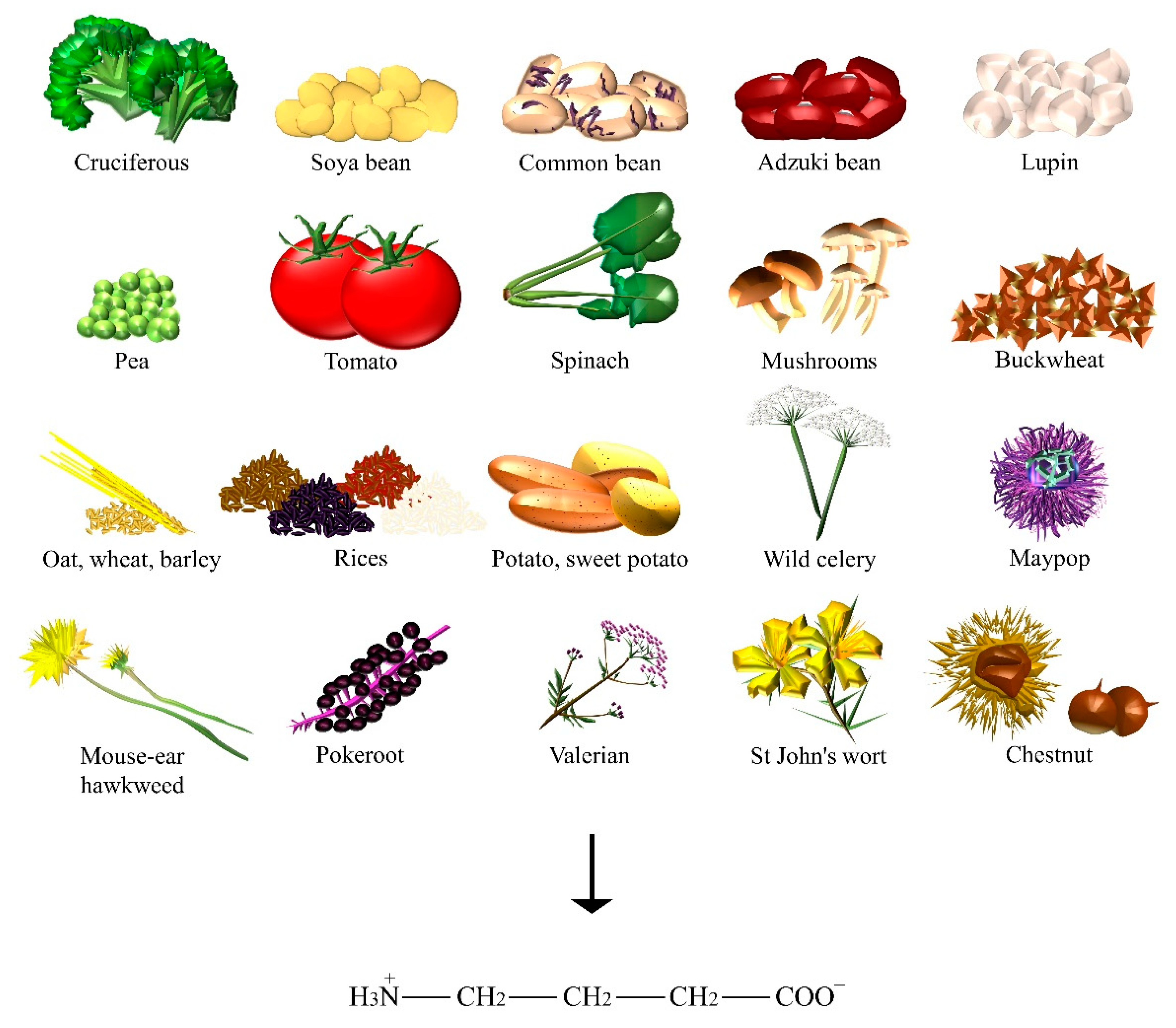
2.3. Gamma-Aminobutyric Acid
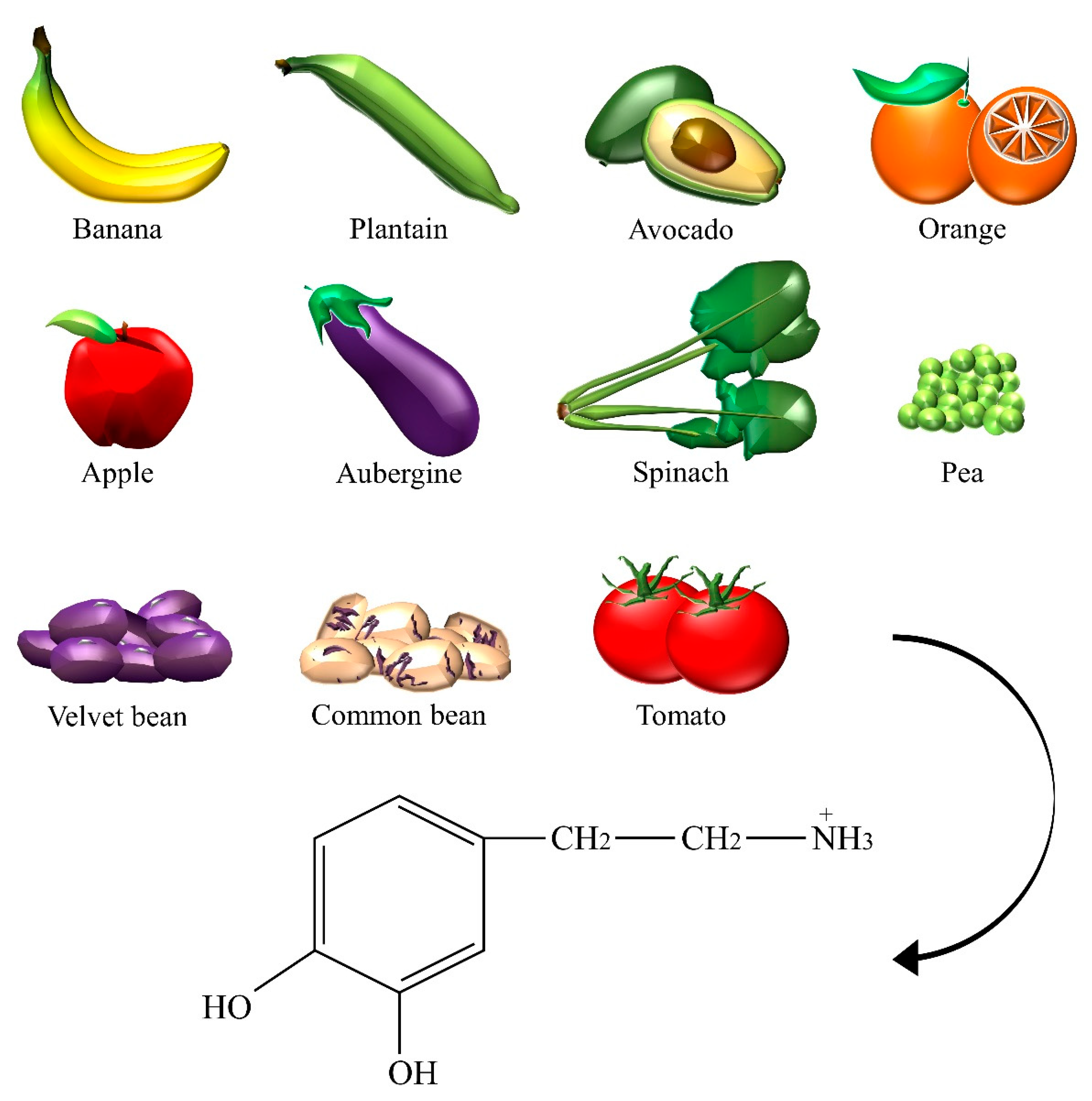
2.4. Dopamine
2.5. Serotonin
2.6. Histamine
2.7. What about Microorganism-Derived NTs?
3. Discussion
- Monosodium glutamate was hypothesized to be associated with the Chinese restaurant syndrome [93] (for example, numbness, weakness, and heart palpitations), but also with vertigo [94]. However, the lack of significant evidence [95] may be explained by the fact that trials always enrolled patients with no particular sensitivity to glutamate, while positive cases reported in the literature referred to fragile or glutamate-sensitive individuals [96]. Dietary glutamate could exert central nervous system effects only during neonatal development: a subcutaneous injection of monosodium glutamate caused adult mice to be more prone to anxiety and depression-like behaviors [97]. Actually, facilitative glutamate transporters through the blood-brain barrier were found only on the luminal membrane [98].
- GABA was proven to have central nervous system action after an oral administration of 800 mg by modulating fronto-striatal networks [35]. Moreover, the benefit from the consumption of GABA-containing vegetables showed the importance of dietary GABA on the sympathetic nerve activity [99]. Conversely, there is still discordance over the alleged GABA capacity to cross the blood-brain barrier [100].
- The increase of the histamine concentration in the plasma was shown to be due to both the consumption of specific foods rich in histamine and to foods with histamine-releasing capacities (for example, citrus fruit, tomatoes). The positive results were demonstrated by diets with low histamine levels [101]. The intestinal diamine oxidase (DAO) enzyme normally prevent dietary histamine uptake into the blood circulation. However, excessive ingestion, the use of DAO inhibitor drugs, alcohol consumption, and cases of concomitant gastrointestinal diseases (for example, gastritis, irritable bowel syndrome, Crohn’s disease, and ulcers) or enzyme-deficiency, may compromise the catabolic capacity of this enzyme.
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Millstine, D.; Chen, C.; Bauer, B. Complementary and integrative medicine in the management of headache. BMJ 2017, 357, j1805. [Google Scholar] [CrossRef] [PubMed]
- Pasca, L.; De Giorgis, V.; Macasaet, J.A.; Trentani, C.; Tagliabue, A.; Veggiotti, P. The changing face of dietary therapy for epilepsy. Eur. J. Pediatr. 2016, 175, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xiong, N.; Huang, J.; Guo, S.; Liu, L.; Han, C.; Zhang, G.; Jiang, H.; Ma, K.; Xia, Y.; et al. Protein-restricted diets for ameliorating motor fluctuations in Parkinson’s disease. Front. Aging Neurosci. 2017, 9, 206. [Google Scholar] [CrossRef] [PubMed]
- Briguglio, M.; Dell’Osso, B.; Galentino, R.; Zanaboni Dina, C.; Banfi, G.; Porta, M. Tics and obsessive-compulsive disorder in relation to diet: Two case reports. L'Encephale 2017. [Google Scholar] [CrossRef] [PubMed]
- Guallar, E.; Stranges, S.; Mulrow, C.; Appel, L.J.; Miller, E.R., 3rd. Enough is enough: Stop wasting money on vitamin and mineral supplements. Ann. Intern. Med. 2013, 159, 850–851. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; DiSilvio, B.; Fernstrom, M.H.; Fernstrom, J.D. Effect of chronic protein ingestion on tyrosine and tryptophan levels and catecholamine and serotonin synthesis in rat brain. Nutr. Neurosci. 2011, 14, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Fallon, S.; Shearman, E.; Sershen, H.; Lajtha, A. Food reward-induced neurotransmitter changes in cognitive brain regions. Neurochem. Res. 2007, 32, 1772–1782. [Google Scholar] [CrossRef] [PubMed]
- Bender, A.; Hagan, K.E.; Kingston, N. The association of folate and depression: A meta-analysis. J. Psychiatr. Res. 2017, 95, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Roshchina, V. Neurotransmitters in Plant Life; Science Publishers: Dubai, UAE, 2001. [Google Scholar]
- Naila, A.; Flint, S.; Fletcher, G.; Bremer, P.; Meerdink, G. Control of biogenic amines in food—Existing and emerging approaches. J. Food Sci. 2010, 75, R139–R150. [Google Scholar] [CrossRef] [PubMed]
- Odjakova, M.; Hadjiivanova, C. Animal neurotransmitter substances in plants. Bulg. J. Plant Physiol. 1997, 23, 94–102. [Google Scholar]
- Hartmann, E.; Kilbinger, H. Occurrence of light-dependent acetylcholine concentrations in higher plants. Experientia 1974, 30, 1397–1398. [Google Scholar] [CrossRef] [PubMed]
- Fluck, R.A.; Jaffe, M.J. The acetylcholine system in plants. In Commentaries in Plant Science; Smith, H., Ed.; Pergamon Press: Oxford, UK, 1976; pp. 119–136. [Google Scholar]
- Fryer, A.; Christopoulos, A.; Nathanson, N. Muscarinic Receptors, 1st ed.; Springer: Berlin, Germany, 2012. [Google Scholar]
- Wessler, I.; Kilbinger, H.; Bittinger, F.; Kirkpatrick, C.J. The biological role of non-neuronal acetylcholine in plants and humans. Jpn. J. Pharmacol. 2001, 85, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Wessler, I.; Kirkpatrick, C.J.; Racke, K. Non-neuronal acetylcholine, a locally acting molecule, widely distributed in biological systems: Expression and function in humans. Pharmacol. Ther. 1998, 77, 59–79. [Google Scholar] [CrossRef]
- Barnes, J.; Anderson, L.; Phillipson, D. Herbal Medicines, 3rd ed.; Pharmaceutical Press: London, UK, 2007; 440p. [Google Scholar]
- European Pharmacopoeia Commission. European Pharmacopoeia: Published under the Direction of the Council of Europe (Partial Agreement) in Accordance with the Convention on the Elaboration of a European Pharmacopoeia, 2nd ed.; Maisonneuve: Sainte Ruffine, France, 1981. [Google Scholar]
- Rangan, C.; Barceloux, D.G. Food additives and sensitivities. Dis. Mon. 2009, 55, 292–311. [Google Scholar] [CrossRef] [PubMed]
- Jinap, S.; Hajeb, P. Glutamate. Its applications in food and contribution to health. Appetite 2010, 55, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Venkitasamy, C.; Pan, Z.; Liu, W.; Zhao, L. Novel umami ingredients: Umami peptides and their taste. J. Food Sci. 2017, 82, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Skypala, I.J.; Williams, M.; Reeves, L.; Meyer, R.; Venter, C. Sensitivity to food additives, vaso-active amines and salicylates: A review of the evidence. Clin. Transl. Allergy 2015, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.; Lui, W.; Wu, K.; Chan, C.; Dai, S.; Sui, Z.; Corke, H. Bioactive compounds and bioactivities of germinated edible seeds and sprouts: An updated review. Trends Food Sci. Technol. 2017, 59, 1–14. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Kuo, Y.-H.; Lambein, F.; Frias, J.; Vidal-Valverde, C. Kinetics of free protein amino acids, free non-protein amino acids and trigonelline in soybean (Glycine max L.) and lupin (Lupinus angustifolius L.) sprouts. Eur. Food Res. Technol. 2006, 224, 177–186. [Google Scholar] [CrossRef]
- Li, L.; Liu, B.; Zheng, X. Bioactive ingredients in adzuki bean sprouts. J. Med. Plants Res. 2011, 5, 5894–5898. [Google Scholar]
- Xu, J.; Hu, Q. Changes in γ-aminobutyric acid content and related enzyme activities in Jindou 25 soybean (Glycine max L.) seeds during germination. LWT Food Sci. Technol. 2014, 55, 341–346. [Google Scholar] [CrossRef]
- Kuo, Y.; Rozan, P.; Lambein, F.; Frias, J.; Vidal-Valverde, C. Effects of different germination conditions on the contents of free protein and non-protein amino acids of commercial legumes. Food Chem. 2004, 86, 537–545. [Google Scholar] [CrossRef]
- Xu, J.G.; Hu, Q.P.; Duan, J.L.; Tian, C.R. Dynamic changes in gamma-aminobutyric acid and glutamate decarboxylase activity in oats (Avena nuda L.) during steeping and germination. J. Agric. Food Chem. 2010, 58, 9759–9763. [Google Scholar] [CrossRef] [PubMed]
- Van Hung, P.; Maeda, T.; Morita, N. Improvement of nutritional composition and antioxidant capacity of high-amylose wheat during germination. J. Food Sci. Technol. 2015, 52, 6756–6762. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Moon, Y.; Oh, C. γ-Aminobutyric Acid (GABA) Content of Selected Uncooked Foods. Prev. Nutr. Food Sci. 2003, 8, 75–78. [Google Scholar] [CrossRef]
- Lin, L.Y.; Peng, C.C.; Yang, Y.L.; Peng, R.Y. Optimization of bioactive compounds in buckwheat sprouts and their effect on blood cholesterol in hamsters. J. Agric. Food Chem. 2008, 56, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Akihiro, T.; Koike, S.; Tani, R.; Tominaga, T.; Watanabe, S.; Iijima, Y.; Aoki, K.; Shibata, D.; Ashihara, H.; Matsukura, C.; et al. Biochemical mechanism on GABA accumulation during fruit development in tomato. Plant Cell Physiol. 2008, 49, 1378–1389. [Google Scholar] [CrossRef] [PubMed]
- Park, K.B.; Oh, S.H. Production of yogurt with enhanced levels of gamma-aminobutyric acid and valuable nutrients using lactic acid bacteria and germinated soybean extract. Bioresour. Technol. 2007, 98, 1675–1679. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Lee, M.Y.; Ji, G.E.; Lee, Y.S.; Hwang, K.T. Production of gamma-aminobutyric acid in black raspberry juice during fermentation by Lactobacillus brevis GABA100. Int. J. Food Microbiol. 2009, 130, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Steenbergen, L.; Sellaro, R.; Stock, A.K.; Beste, C.; Colzato, L.S. gamma-Aminobutyric acid (GABA) administration improves action selection processes: A randomised controlled trial. Sci. Rep. 2015, 5, 12770. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Ma, Y.; Wei, Z.Z.; Yuan, W.X.; Li, Y.L.; Zhang, C.H.; Xue, X.T.; Zhou, H.J. Determination and comparison of gamma-aminobutyric acid (GABA) content in pu-erh and other types of Chinese tea. J. Agric. Food Chem. 2011, 59, 3641–3648. [Google Scholar] [CrossRef] [PubMed]
- Funayama, S.; Hikino, H. Hypotensive principles of Phytolacca roots. J. Nat. Prod. 1979, 42, 672–674. [Google Scholar] [CrossRef] [PubMed]
- Carratu, B.; Boniglia, C.; Giammarioli, S.; Mosca, M.; Sanzini, E. Free amino acids in botanicals and botanical preparations. J. Food Sci. 2008, 73, C323–C328. [Google Scholar] [CrossRef] [PubMed]
- Feldman, J.M.; Lee, E.M.; Castleberry, C.A. Catecholamine and serotonin content of foods: Effect on urinary excretion of homovanillic and 5-hydroxyindoleacetic acid. J. Am. Diet Assoc. 1987, 87, 1031–1035. [Google Scholar] [PubMed]
- Kanazawa, K.; Sakakibara, H. High content of dopamine, a strong antioxidant, in Cavendish banana. J. Agric. Food Chem. 2000, 48, 844–848. [Google Scholar] [CrossRef] [PubMed]
- Udenfriend, S.; Lovenberg, W.; Sjoerdsma, A. Physiologically active amines in common fruits and vegetables. Arch. Biochem. Biophys. 1959, 85, 487–490. [Google Scholar] [CrossRef]
- Wichers, H.; Visser, J.; Huizing, H.; Pras, N. Occurrence of L-DOPA and dopamine in plants and cell cultures of Mucuna pruriens and effects of 2,4-D and NaCl on these compounds. Plant Cell Tissue Organ Cult. 1993, 33, 259–264. [Google Scholar] [CrossRef]
- Cassani, E.; Cilia, R.; Laguna, J.; Barichella, M.; Contin, M.; Cereda, E.; Isaias, I.U.; Sparvoli, F.; Akpalu, A.; Budu, K.O.; et al. Mucuna pruriens for Parkinson’s disease: Low-cost preparation method, laboratory measures and pharmacokinetics profile. J. Neurol. Sci. 2016, 365, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Gerrard, J.W.; Richardson, J.S.; Donat, J. Neuropharmacological evaluation of movement disorders that are adverse reactions to specific foods. Int. J. Neurosci. 1994, 76, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Mazza, G. Application of LC and LC-MS to the analysis of melatonin and serotonin in edible plants. Crit. Rev. Food Sci. Nutr. 2011, 51, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Adão, R.C.; Glória, M.B.A. Bioactive amines and carbohydrate changes during ripening of ‘Prata’ banana (Musa acuminata × M. balbisiana). Food Chem. 2005, 90, 705–711. [Google Scholar] [CrossRef]
- Kang, S.; Back, K. Enriched production of N-hydroxycinnamic acid amides and biogenic amines in pepper (Capsicum annuum) flowers. Sci. Hortic. 2006, 108, 337–341. [Google Scholar] [CrossRef]
- Ly, D.; Kang, K.; Choi, J.Y.; Ishihara, A.; Back, K.; Lee, S.G. HPLC analysis of serotonin, tryptamine, tyramine, and the hydroxycinnamic acid amides of serotonin and tyramine in food vegetables. J. Med. Food 2008, 11, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Lavizzari, T.; Teresa Veciana-Nogues, M.; Bover-Cid, S.; Marine-Font, A.; Carmen Vidal-Carou, M. Improved method for the determination of biogenic amines and polyamines in vegetable products by ion-pair high-performance liquid chromatography. J. Chromatogr. A 2006, 1129, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Foy, J.M.; Parratt, J.R. 5-Hydroxytryptamine in pineapples. J. Pharm. Pharmacol. 1961, 13, 382–383. [Google Scholar] [CrossRef] [PubMed]
- Council, N.R. Toxicants Occurring Naturally in Foods; National Academy of Sciences: Washington, DC, USA, 1973. [Google Scholar]
- Feldman, J.M.; Lee, E.M. Serotonin content of foods: Effect on urinary excretion of 5-hydroxyindoleacetic acid. Am. J. Clin. Nutr. 1985, 42, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Bowden, K.; Brown, B.G.; Batty, J.E. 5-Hydroxytryptamine: Its occurrence in cowhage. Nature 1954, 174, 925–926. [Google Scholar] [CrossRef] [PubMed]
- Engstrom, K.; Lundgren, L.; Samuelsson, G. Bioassay-guided isolation of serotonin from fruits of Solanum tuberosum L. Acta Pharm. Nord. 1992, 4, 91–92. [Google Scholar] [PubMed]
- Kang, S.; Kang, K.; Lee, K.; Back, K. Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants. Plant Cell Rep. 2007, 26, 2009–2015. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishna, A.; Giridhar, P.; Sankar, K.U.; Ravishankar, G.A. Melatonin and serotonin profiles in beans of Coffea species. J. Pineal Res. 2012, 52, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Badria, F. Melatonin, serotonin, and tryptamine in some egyptian food and medicinal plants. J. Med. Food 2002, 5, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Collier, H.O.; Chesher, G.B. Identification of 5-hydroxytryptamine in the sting of the nettle (urtica dioica). Br. J. Pharmacol. Chemother. 1956, 11, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, G.; Di Viesti, V.; Zavatti, M.; Zanoli, P. Anxiolytic-like effect of Griffonia simplicifolia Baill. seed extract in rats. Phytomedicine 2011, 18, 848–851. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, I.M.; Pinho, O. Biogenic amines in Portuguese traditional foods and wines. J. Food Prot. 2006, 69, 2293–2303. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, A. Significance of biogenic amines to food safety and human health. Food Res. Int. 1996, 29, 675–690. [Google Scholar] [CrossRef]
- Emmelin, N.; Feldberg, W. Pharmacologically active substances in the fluid of nettle hairs (Urtica urens). J. Physiol. 1947, 106, 14. [Google Scholar] [PubMed]
- Ough, C.S. Measurement of histamine in California wines. J. Agric. Food Chem. 1971, 19, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, S.; Imark, C.; Kneubuhl, M. Biogenic amines in foods: Histamine and food processing. Inflamm. Res. 1999, 48, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Latorre-Moratalla, M.L.; Bover-Cid, S.; Veciana-Nogues, M.T.; Vidal-Carou, M.C. Control of biogenic amines in fermented sausages: Role of starter cultures. Front. Microbiol. 2012, 3, 169. [Google Scholar] [CrossRef] [PubMed]
- Kalac, P.; Svecova, S.; Pelikanova, T. Levels of biogenic amines in typical vegetable products. Food Chem. 2002, 77, 349–351. [Google Scholar] [CrossRef]
- Shukla, S.; Kim, J.; Kim, M. Occurrence of Biogenic Amines in Soybean Food Products. In Soybean and Health; El-Shemy, H., Ed.; InTech: Shanghai, China, 2011; pp. 189–190. [Google Scholar]
- Roshchina, V.V. The Fluorescence Methods to Study Neurotransmitters (Biomediators) in Plant Cells. J. Fluoresc. 2016, 26, 1029–1043. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Droz, S.; Patsenker, E.; Bogli-Stuber, K.; Aebi, B.; Leib, S.L. Severe hepatotoxicity following ingestion of herbalife nutritional supplements contaminated with Bacillus subtilis. J. Hepatol. 2009, 50, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Lyte, M. Probiotics function mechanistically as delivery vehicles for neuroactive compounds: Microbial endocrinology in the design and use of probiotics. Bioessays 2011, 33, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Barrett, E.; Ross, R.P.; O’Toole, P.W.; Fitzgerald, G.F.; Stanton, C. gamma-Aminobutyric acid production by culturable bacteria from the human intestine. J. Appl. Microbiol. 2012, 113, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Wei, L.; Liu, J. Biotechnological advances and perspectives of gamma-aminobutyric acid production. World J. Microbiol. Biotechnol. 2017, 33, 64. [Google Scholar] [CrossRef] [PubMed]
- Siragusa, S.; De Angelis, M.; Di Cagno, R.; Rizzello, C.G.; Coda, R.; Gobbetti, M. Synthesis of gamma-aminobutyric acid by lactic acid bacteria isolated from a variety of Italian cheeses. Appl. Environ. Microbiol. 2007, 73, 7283–7290. [Google Scholar] [CrossRef] [PubMed]
- Moo-Chang, K.; Seok-Cheol, C. Production of GABA (gamma amino butyric acid) by lactic acid bacteria. Korean J. Food Sci. Anim. Resour. 2013, 33, 377–389. [Google Scholar]
- Kadir, S.A.; Wan-Mohtar, W.; Mohammad, R.; Lim, S.; Mohammed, A.; Saari, N. Evaluation of commercial soy sauce koji strains of Aspergillus oryzae for gamma-aminobutyric acid (GABA) production. J. Ind. Microbiol. Biotechnol. 2016, 43, 1387–1395. [Google Scholar] [CrossRef] [PubMed]
- Nikmaram, N.; Dar, B.N.; Roohinejad, S.; Koubaa, M.; Barba, F.J.; Greiner, R.; Johnson, S.K. Recent advances in gamma-aminobutyric acid (GABA) properties in pulses: An overview. J. Sci. Food Agric. 2017, 97, 2681–2689. [Google Scholar] [CrossRef] [PubMed]
- Asano, Y.; Hiramoto, T.; Nishino, R.; Aiba, Y.; Kimura, T.; Yoshihara, K.; Koga, Y.; Sudo, N. Critical role of gut microbiota in the production of biologically active, free catecholamines in the gut lumen of mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G1288–G1295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Dinan, T.G. Mind-altering microorganisms: The impact of the gut microbiota on brain and behaviour. Nat. Rev. Neurosci. 2012, 13, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.M.; Hong, T.; van Pijkeren, J.P.; Hemarajata, P.; Trinh, D.V.; Hu, W.; Britton, R.A.; Kalkum, M.; Versalovic, J. Histamine derived from probiotic Lactobacillus reuteri suppresses TNF via modulation of PKA and ERK signaling. PLoS ONE 2012, 7, e31951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iyer, L.M.; Aravind, L.; Coon, S.L.; Klein, D.C.; Koonin, E.V. Evolution of cell-cell signaling in animals: Did late horizontal gene transfer from bacteria have a role? Trends Genet. 2004, 20, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Yunes, R.A.; Poluektova, E.U.; Dyachkova, M.S.; Klimina, K.M.; Kovtun, A.S.; Averina, O.V.; Orlova, V.S.; Danilenko, V.N. GABA production and structure of gadB/gadC genes in Lactobacillus and Bifidobacterium strains from human microbiota. Anaerobe 2016, 42, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Tetel, M.J.; de Vries, G.J.; Melcangi, R.C.; Panzica, G.; O’Mahony, S.M. Steroids, stress and the gut microbiome-brain axis. J. Neuroendocrinol. 2018, 30. [Google Scholar] [CrossRef] [PubMed]
- Javurek, A.B.; Spollen, W.G.; Johnson, S.A.; Bivens, N.J.; Bromert, K.H.; Givan, S.A.; Rosenfeld, C.S. Effects of exposure to bisphenol A and ethinyl estradiol on the gut microbiota of parents and their offspring in a rodent model. Gut Microbes 2016, 7, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Eisenhofer, G.; Aneman, A.; Friberg, P.; Hooper, D.; Fandriks, L.; Lonroth, H.; Hunyady, B.; Mezey, E. Substantial production of dopamine in the human gastrointestinal tract. J. Clin. Endocrinol. Metab. 1997, 82, 3864–3871. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.K.; Park, K.J.; Hennig, G.W. Colonic migrating motor complexes, high amplitude propagating contractions, neural reflexes and the importance of neuronal and mucosal serotonin. J. Neurogastroenterol. Motil. 2014, 20, 423–446. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N. Inflammatory mediators and modulation of blood-brain barrier permeability. Cell Mol. Neurobiol. 2000, 20, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, Y.; Sato-Suzuki, I.; Tsujino, N.; Nakasato, A.; Seki, Y.; Fumoto, M.; Arita, H. Augmented brain 5-HT crosses the blood-brain barrier through the 5-HT transporter in rat. Eur. J. Neurosci. 2008, 27, 2466–2472. [Google Scholar] [CrossRef] [PubMed]
- Young, L.W.; Darios, E.S.; Watts, S.W. An immunohistochemical analysis of SERT in the blood-brain barrier of the male rat brain. Histochem. Cell Biol. 2015, 144, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Benarroch, E.E. Circumventricular organs: Receptive and homeostatic functions and clinical implications. Neurology 2011, 77, 1198–1204. [Google Scholar] [CrossRef] [PubMed]
- Alstadhaug, K. Histamine in migraine and brain. Headache 2014, 54, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Konig, J.; Wells, J.; Cani, P.D.; Garcia-Rodenas, C.L.; MacDonald, T.; Mercenier, A.; Whyte, J.; Troost, F.; Brummer, R.-J. Human Intestinal Barrier Function in Health and Disease. Clin. Transl. Gastroenterol. 2016, 7, e196. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.H. Chinese-restaurant syndrome. N. Engl. J. Med. 1968, 278, 796. [Google Scholar] [PubMed]
- Leussink, V.I.; Hartung, H.P.; Stuve, O.; Kieseier, B.C. Vestibular hypofunction after monosodium glutamate ingestion: Broadening the spectrum of ‘Chinese restaurant syndrome’. J. Neurol. 2016, 263, 1027–1028. [Google Scholar] [CrossRef] [PubMed]
- Obayashi, Y.; Nagamura, Y. Does monosodium glutamate really cause headache? A systematic review of human studies. J. Headache Pain 2016, 17, 54. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.H.; Drouin, M.A.; Herbert, M.; Mao, Y.; Karsh, J. The monosodium glutamate symptom complex: Assessment in a double-blind, placebo-controlled, randomized study. J. Allergy Clin. Immunol. 1997, 99 (6 Pt 1), 757–762. [Google Scholar] [CrossRef]
- Quines, C.B.; Rosa, S.G.; Da Rocha, J.T.; Gai, B.M.; Bortolatto, C.F.; Duarte, M.M.; Nogueira, C.W. Monosodium glutamate, a food additive, induces depressive-like and anxiogenic-like behaviors in young rats. Life Sci. 2014, 107, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, R.A.; O’Kane, R.L.; Simpson, I.A.; Vina, J.R. Structure of the blood-brain barrier and its role in the transport of amino acids. J. Nutr. 2006, 136 (Suppl. S1), 218S–226S. [Google Scholar] [CrossRef] [PubMed]
- Okita, Y.; Nakamura, H.; Kouda, K.; Takahashi, I.; Takaoka, T.; Kimura, M.; Sugiura, T. Effects of vegetable containing gamma-aminobutyric acid on the cardiac autonomic nervous system in healthy young people. J. Physiol. Anthropol. 2009, 28, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Takanaga, H.; Ohtsuki, S.; Hosoya, K.; Terasaki, T. GAT2/BGT-1 as a system responsible for the transport of gamma-aminobutyric acid at the mouse blood-brain barrier. J. Cereb. Blood Flow Metab. 2001, 21, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Zhuang, Y.; Gomez-Pinilla, F. High-fat diet transition reduces brain DHA levels associated with altered brain plasticity and behaviour. Sci. Rep. 2012, 2, 431. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.L.; Biesiekierski, J.R.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Randomised clinical trial: Gluten may cause depression in subjects with non-coeliac gluten sensitivity—An exploratory clinical study. Aliment. Pharmacol. Ther. 2014, 39, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Lopez, R.; Perea-Milla, E.; Garcia, C.R.; Rivas-Ruiz, F.; Romero-Gonzalez, J.; Moreno, J.L.; Faus, V.; del Castillo Aguas, G.; Ramos Diaz, J.C. New therapeutic approach to Tourette Syndrome in children based on a randomized placebo-controlled double-blind phase IV study of the effectiveness and safety of magnesium and vitamin B6. Trials 2009, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Belzeaux, R.; Annweiler, C.; Bertrand, J.A.; Beauchet, O.; Pichet, S.; Jollant, F.; Turecki, G.; Richard-Devantoy, S. Association between hypovitaminosis D and cognitive inhibition impairment during major depression episode. J. Affect. Disord. 2017, 225, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Blood-brain barrier biology and methodology. J. Neurovirol. 1999, 5, 556–569. [Google Scholar] [CrossRef] [PubMed]
- Logan, A.C.; Jacka, F.N. Nutritional psychiatry research: An emerging discipline and its intersection with global urbanization, environmental challenges and the evolutionary mismatch. J. Physiol. Anthropol. 2014, 33, 22. [Google Scholar] [CrossRef] [PubMed]
- Institute of Food Technologists Foundation (IFT). Making Decisions about the Risks of Chemicals in Foods with Limited Scientific Information; Institute of Food Technologists Foundation: Chicago, IL, USA, 2009. [Google Scholar]
- Roshchina, V.V.; Bezuglov, V.V.; Markova, L.N.; Sakharova, N.Y.; Buznikov, G.A.; Karnaukhov, V.N.; Chailakhyan, L.M. Interaction of living cells with fluorescent derivatives of biogenic amines. Dokl. Biochem. Biophys. 2003, 393, 346–349. [Google Scholar] [CrossRef] [PubMed]
- Briguglio, M.; Dina, C.Z.; Servello, D.; Porta, M. Tourette Syndrome and Nutritional Implications. In Proceedings of the Annual Meeting of the European Society for the Study of Tourette Syndrome—9th European Conference on Tourette Syndrome and Tic Disorders, Warsaw, Poland, 9–10 June 2016. [Google Scholar]
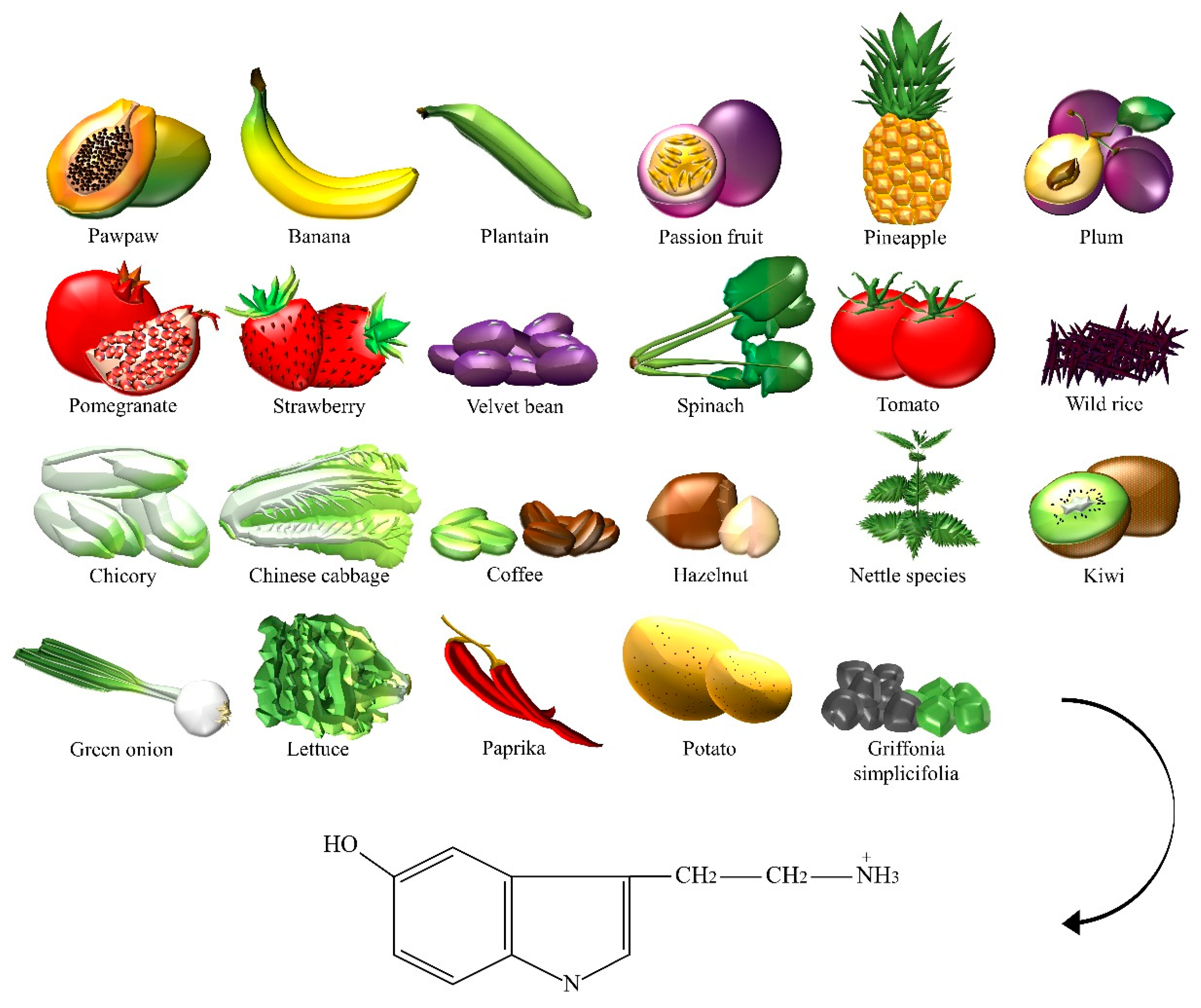
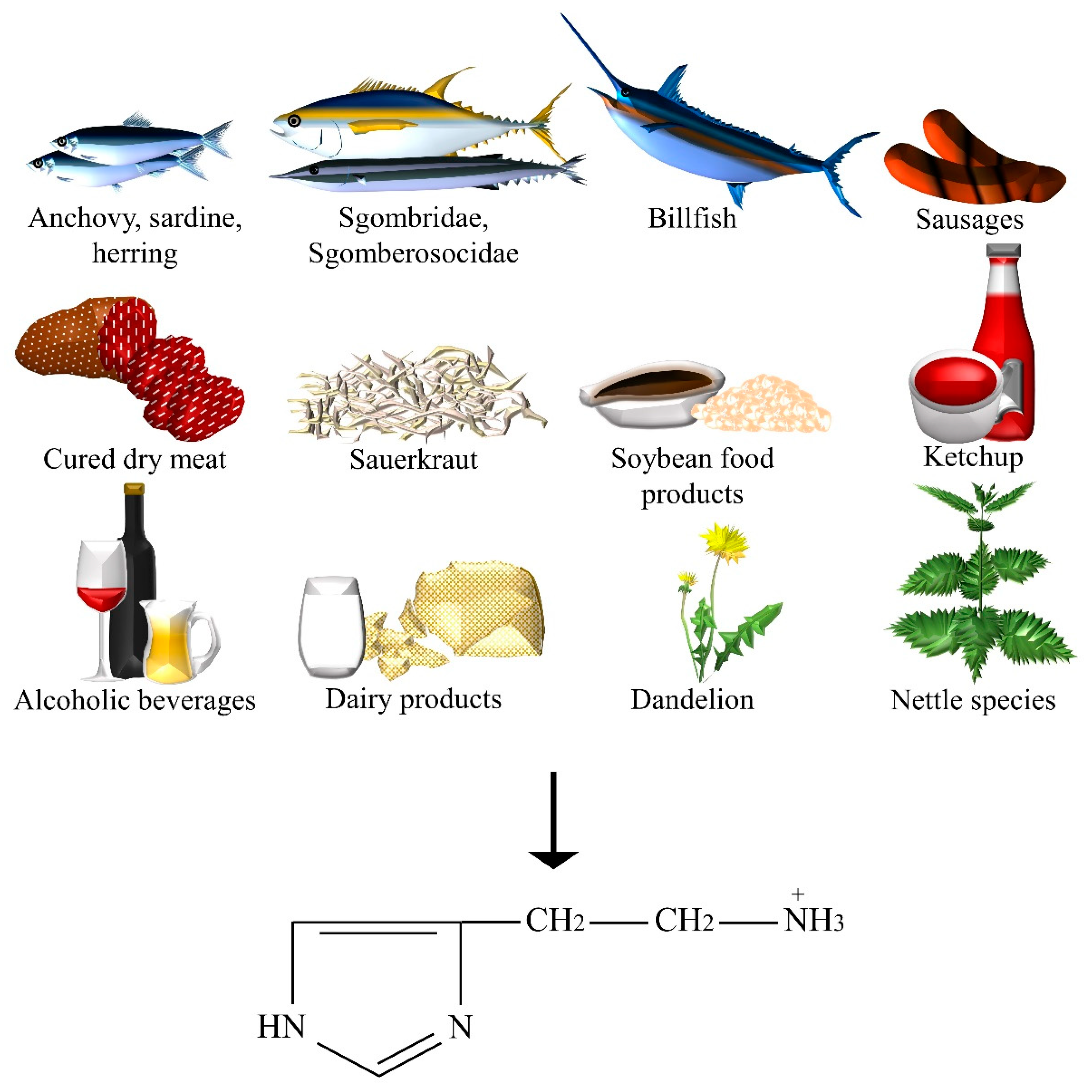
| Dietary Neurotransmitter | Foods and Botanicals |
|---|---|
| Acetylcholine | Aubergine, bitter orange, common bean, foxglove, mistletoe, mung bean, nettle species, pea, radish, spinach, squash, wild strawberry. |
| Glutamate | Caviar, cheese, crackling, chips, dried cod, fermented beans, fish sauces, gravies, instant coffee powder, meats, miso, mushrooms, noodle dishes, oyster sauce, Parmesan cheese, ready-to-eat meals, salami, savory snacks, seafood, seaweeds, soups, soy sauces, spinach, stews, tomato, tomato sauce. |
| GABA | Adzuki bean, barley, broccoli, buckwheat, chestnut, common bean, kale, lupin, maypop, mouse-ear hawkweed, oat, pea, pokeroot, potato, rice, shiitake, soya bean, spinach, St John’s wort, sweet potato, tea, tomato, valerian, wheat, wild celery. |
| Dopamine | Aubergine, avocado, banana, common bean, apple, orange, pea, plantain, spinach, tomato, velvet bean. |
| Serotonin | Bananas, chicory, Chinese cabbage, coffee powders, green coffee bean, green onion, hazelnut, kiwi, lettuce, nettle, Griffonia simplicifolia, paprika, passion fruit, pawpaw, pepper, pineapple, plantain, plum, pomegranate, potato, spinach, strawberry, tomato, velvet bean, wild rice. |
| Histamine | Anchovy, beer, billfish, Champagne and Sherry, dandelion, fermented sausages, ham and other cured dry meat products, herring, ketchup, aged cheeses, nettle, red, white and dessert wines, sardine, sauerkraut, Scomberesocidae (for example, sauries), Scombridae (for example, tuna, mackerel, and bonitos), soybean food products (for example, soy, tempeh, soy sauce, soya bean milk, doenjang, doufuru, and nattō), sweet or sour cream, UHT, pasteurized, and fresh milk, yoghurt. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Briguglio, M.; Dell’Osso, B.; Panzica, G.; Malgaroli, A.; Banfi, G.; Zanaboni Dina, C.; Galentino, R.; Porta, M. Dietary Neurotransmitters: A Narrative Review on Current Knowledge. Nutrients 2018, 10, 591. https://doi.org/10.3390/nu10050591
Briguglio M, Dell’Osso B, Panzica G, Malgaroli A, Banfi G, Zanaboni Dina C, Galentino R, Porta M. Dietary Neurotransmitters: A Narrative Review on Current Knowledge. Nutrients. 2018; 10(5):591. https://doi.org/10.3390/nu10050591
Chicago/Turabian StyleBriguglio, Matteo, Bernardo Dell’Osso, Giancarlo Panzica, Antonio Malgaroli, Giuseppe Banfi, Carlotta Zanaboni Dina, Roberta Galentino, and Mauro Porta. 2018. "Dietary Neurotransmitters: A Narrative Review on Current Knowledge" Nutrients 10, no. 5: 591. https://doi.org/10.3390/nu10050591






