The Food-Specific Serum IgG Reactivity in Major Depressive Disorder Patients, Irritable Bowel Syndrome Patients and Healthy Controls
Abstract
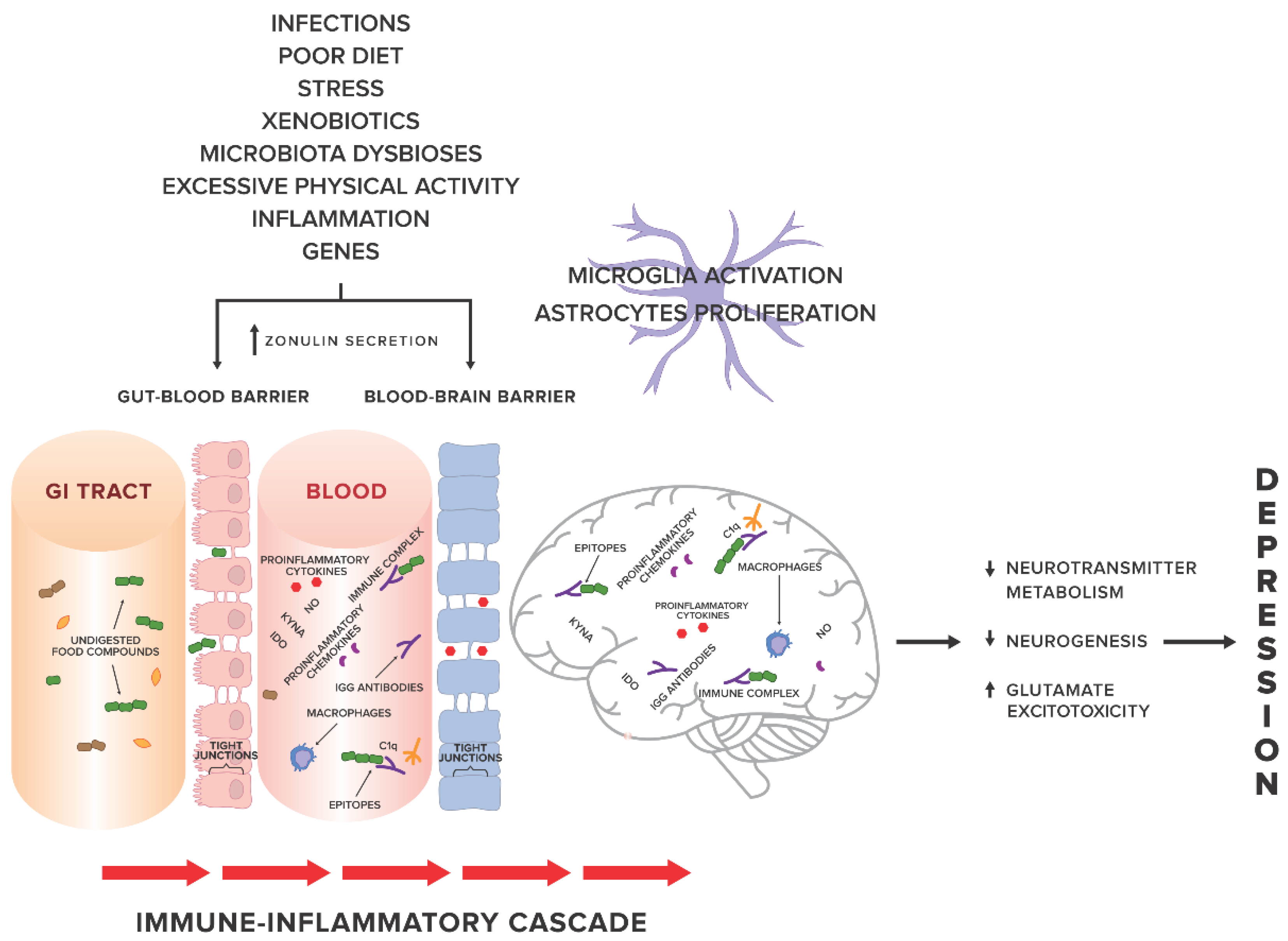
1. Introduction
- (1)
- increasing evidence linking IBS and food IgG hyperreactivity, and
- (2)
- a suggested relationship between food IgG reactivity and MDD.
2. Material and Methods
2.1. Participants
2.2. Concentration of Specific IgG Antibodies
2.3. Statistical Analysis
3. Results
3.1. Characteristics of the Examined Groups
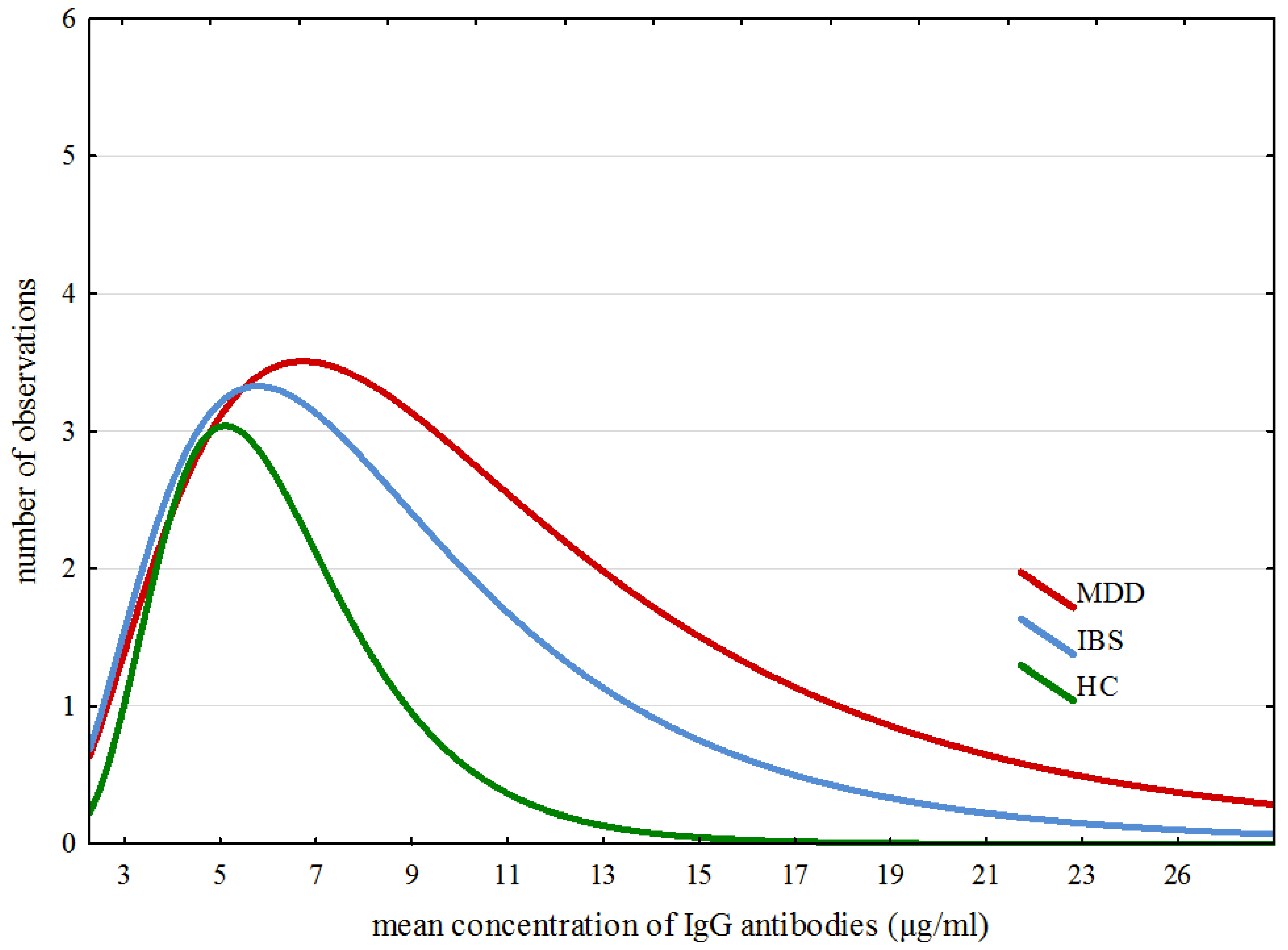
3.2. Seroreactivity to Food Antigens in the Examined Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Klein, D.N.; Hajcak, G. Heterogeneity of Depression: Clinical Considerations and Psychophysiological Measures. Psychol. Inq. 2015, 26, 247–252. [Google Scholar] [CrossRef]
- World Health Organization. Depression. Available online: http://www.who.int/mediacentre/factsheets/fs369/en/ (accessed on 5 December 2017).
- Uher, R. Gene-environment interactions in common mental disorders: An update and strategy for a genome-wide search. Soc. Psychiatry Psychiatr. Epidemiol. 2014, 49, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Dunn, E.C.; Brown, R.C.; Dai, Y.; Rosand, J.; Nugent, N.R.; Amstadter, A.B.; Smoller, J.W. Genetic determinants of depression: Recent findings and future directions. Harv. Rev. Psychiatry 2015, 23, 1. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.S. The macrophage theory of depression. Med. Hypotheses 1991, 35, 298–306. [Google Scholar] [CrossRef]
- Dantzer, R. Role of the Kynurenine metabolism pathway in inflammation-induced depression: Preclinical approaches. In Inflammation-Associated Depression: Evidence, Mechanisms and Implications; Springer: Berlin, Germany, 2016; pp. 117–138. [Google Scholar]
- Dahl, J.; Ormstad, H.; Aass, H.; Sandvik, L.; Malt, U.; Andreassen, O. Recovery from major depressive disorder episode after non-pharmacological treatment is associated with normalized cytokine levels. Acta Psychiatr. Scand. 2016, 134, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Maker, G.L.; Hood, S.D.; Drummond, P.D. A review of peripheral biomarkers in major depression: The potential of inflammatory and oxidative stress biomarkers. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 48, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Young, J.J.; Bruno, D.; Pomara, N. A review of the relationship between proinflammatory cytokines and major depressive disorder. J. Affect. Disord. 2014, 169, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Köhler, O.; Benros, M.E.; Nordentoft, M.; Farkouh, M.E.; Iyengar, R.L.; Mors, O.; Krogh, J. Effect of anti-inflammatory treatment on depression, depressive symptoms, and adverse effects: A systematic review and meta-analysis of randomized clinical trials. JAMA Psychiatry 2014, 71, 1381–1391. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Núñez, B.; Pruimboom, L.; Dijck-Brouwer, D.J.; Muskiet, F.A. Lifestyle and nutritional imbalances associated with Western diseases: Causes and consequences of chronic systemic low-grade inflammation in an evolutionary context. J. Nutr. Biochem. 2013, 24, 1183–1201. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Kubera, M.; Leunis, J.-C. The gut-brain barrier in major depression: Intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuroendocrinol. Lett. 2008, 29, 117–124. [Google Scholar] [CrossRef]
- Thevaranjan, N.; Puchta, A.; Schulz, C.; Naidoo, A.; Szamosi, J.; Verschoor, C.P.; Loukov, D.; Schenck, L.P.; Jury, J.; Foley, K.P. Age-Associated Microbial Dysbiosis Promotes Intestinal Permeability, Systemic Inflammation, and Macrophage Dysfunction. Cell Host Microbe 2017, 21, 455–466.e454. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A. Zonulin and its regulation of intestinal barrier function: The biological door to inflammation, autoimmunity, and cancer. Physiol. Rev. 2011, 91, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Zhou, L.; Zhang, J.; Wang, B. Abnormal intestinal permeability and microbiota in patients with autoimmune hepatitis. Int. J. Clin. Exp. Pathol. 2015, 8, 5153. [Google Scholar] [PubMed]
- Clairembault, T.; Leclair-Visonneau, L.; Coron, E.; Bourreille, A.; Le Dily, S.; Vavasseur, F.; Heymann, M.-F.; Neunlist, M.; Derkinderen, P. Structural alterations of the intestinal epithelial barrier in Parkinson’s disease. Acta Neuropathol. Commun. 2015, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Szachta, P.; Bartnicka, A.; Galecka, M.; Skonieczna-Zydecka, K. Microbiota disorders and food hypersensitivity in autism spectrum disorders; what do we know? J. Exp. Integr. Med. 2015, 5, 117–120. [Google Scholar] [CrossRef]
- Esnafoglu, E.; Cırrık, S.; Ayyıldız, S.N.; Erdil, A.; Ertürk, E.Y.; Daglı, A.; Noyan, T. Increased serum zonulin levels as an intestinal permeability marker in autistic subjects. J. Pediatr. 2017, 188, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Karakuła-Juchnowicz, H.; Szachta, P.; Opolska, A.; Morylowska-Topolska, J.; Gałęcka, M.; Juchnowicz, D.; Krukow, P.; Lasik, Z. The role of IgG hypersensitivity in the pathogenesis and therapy of depressive disorders. Nutr. Neurosci. 2017, 20, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Lovell, R.M.; Ford, A.C. Global prevalence of and risk factors for irritable bowel syndrome: A meta-analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721.e714. [Google Scholar] [CrossRef] [PubMed]
- Sinagra, E.; Pompei, G.; Tomasello, G.; Cappello, F.; Morreale, G.C.; Amvrosiadis, G.; Rossi, F.; Monte, A.I.L.; Rizzo, A.G.; Raimondo, D. Inflammation in irritable bowel syndrome: Myth or new treatment target? World J. Gastroenterol. 2016, 22, 2242. [Google Scholar] [CrossRef] [PubMed]
- Spiller, R.C. Infection, immune function, and functional gut disorders. Clin. Gastroenterol. Hepatol. 2004, 2, 445–455. [Google Scholar] [CrossRef]
- Li, F.X.; Patten, S.B.; Hilsden, R.J.; Sutherland, L.R. Irritable bowel syndrome and health-related quality of life: A population-based study in Calgary, Alberta. Can. J. Gastroenterol. Hepatol. 2003, 17, 259–263. [Google Scholar] [CrossRef][Green Version]
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional bowel disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, I.B.; O’toole, P.W.; Öhman, L.; Claesson, M.J.; Deane, J.; Quigley, E.M.; Simrén, M. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 2012, 61, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Foxx-Orenstein, A.E. New and emerging therapies for the treatment of irritable bowel syndrome: An update for gastroenterologists. Ther. Adv. Gastroenterol. 2016, 9, 354–375. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, W.; Sheldon, T.; Shaath, N.; Whorwell, P. Food elimination based on IgG antibodies in irritable bowel syndrome: A randomised controlled trial. Gut 2004, 53, 1459–1464. [Google Scholar] [CrossRef] [PubMed]
- Shih, D.Q.; Kwan, L.Y. All roads lead to Rome: Update on Rome III criteria and new treatment options. Gastroenterol. Rep. 2007, 1, 56. [Google Scholar] [PubMed]
- Marsh, A.; Eslick, E.M.; Eslick, G.D. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur. J. Nutr. 2016, 55, 897–906. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, Y.; Bowyer, R.; Leach, H.; Gulia, P.; Horobin, J.; O’sullivan, N.; Pettitt, C.; Reeves, L.; Seamark, L.; Williams, M. British Dietetic Association systematic review and evidence-based practice guidelines for the dietary management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 2016, 29, 549–575. [Google Scholar] [CrossRef] [PubMed]
- Barmeyer, C.; Schumann, M.; Meyer, T.; Zielinski, C.; Zuberbier, T.; Siegmund, B.; Schulzke, J.-D.; Daum, S.; Ullrich, R. Long-term response to gluten-free diet as evidence for non-celiac wheat sensitivity in one third of patients with diarrhea-dominant and mixed-type irritable bowel syndrome. Int. J. Colorectal Dis. 2017, 32, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Alaedini, A.; Bojarski, C.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; De Magistris, L.; Dieterich, W.; Di Liberto, D. The Overlapping Area of Non-Celiac Gluten Sensitivity (NCGS) and Wheat-Sensitive Irritable Bowel Syndrome (IBS): An Update. Nutrients 2017, 9, 1268. [Google Scholar] [CrossRef] [PubMed]
- Böhmer, C.J.; Tuynman, H.A. The effect of a lactose-restricted diet in patients with a positive lactose tolerance test, earlier diagnosed as irritable bowel syndrome: A 5-year follow-up study. Eur. J. Gastroenterol. Hepatol. 2001, 13, 941–944. [Google Scholar] [CrossRef] [PubMed]
- Defrees, D.N.; Bailey, J. Irritable Bowel Syndrome: Epidemiology, Pathophysiology, Diagnosis, and Treatment. Prim. Care Clin. Off. Pract. 2017, 44, 655–671. [Google Scholar] [CrossRef] [PubMed]
- Halmos, E.P. When the low FODMAP diet does not work. J. Gastroenterol. Hepatol. 2017, 32, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Hill, P.; Muir, J.G.; Gibson, P.R. Controversies and Recent Developments of the Low-FODMAP Diet. Gastroenterol. Hepatol. 2017, 13, 36. [Google Scholar]
- Zar, S.; Mincher, L.; Benson, M.J.; Kumar, D. Food-specific IgG4 antibody-guided exclusion diet improves symptoms and rectal compliance in irritable bowel syndrome. Scand. J. Gastroenterol. 2005, 40, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Drisko, J.; Bischoff, B.; Hall, M.; McCallum, R. Treating irritable bowel syndrome with a food elimination diet followed by food challenge and probiotics. J. Am. Coll. Nutr. 2006, 25, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Monsbakken, K.; Vandvik, P.; Farup, P. Perceived food intolerance in subjects with irritable bowel syndrome–etiology, prevalence and consequences. Eur. J. Clin. Nutr. 2006, 60, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Böhn, L.; Störsrud, S.; Törnblom, H.; Bengtsson, U.; Simrén, M. Self-reported food-related gastrointestinal symptoms in IBS are common and associated with more severe symptoms and reduced quality of life. Am. J. Gastroenterol. 2013, 108, 634–641. [Google Scholar]
- Fritscher-Ravens, A.; Schuppan, D.; Ellrichmann, M.; Schoch, S.; Röcken, C.; Brasch, J.; Bethge, J.; Böttner, M.; Klose, J.; Milla, P.J. Confocal endomicroscopy shows food-associated changes in the intestinal mucosa of patients with irritable bowel syndrome. Gastroenterology 2014, 147, 1012–1020.e1014. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association (APA). Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Arlington, VA, USA, 2013. [Google Scholar]
- Hossain, M.I.; Nahar, B.; Hamadani, J.D.; Ahmed, T.; Roy, A.K.; Brown, K.H. Intestinal mucosal permeability of severely underweight and nonmalnourished Bangladeshi children and effects of nutritional rehabilitation. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Gummesson, A.; Carlsson, L.M.; Storlien, L.H.; Backhed, F.; Lundin, P.; Lofgren, L.; Stenlof, K.; Lam, Y.Y.; Fagerberg, B.; Carlsson, B. Intestinal permeability is associated with visceral adiposity in healthy women. Obesity 2011, 19, 2280–2282. [Google Scholar] [CrossRef] [PubMed]
- Genton, L.; Cani, P.D.; Schrenzel, J. Alterations of gut barrier and gut microbiota in food restriction, food deprivation and protein-energy wasting. Clin. Nutr. 2015, 34, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Karakula-Juchnowicz, H.; Pankowicz, H.; Juchnowicz, D.; Valverde Piedra, J.L.; Malecka-Massalska, T. Intestinal microbiota—A key to understanding the pathophysiology of anorexia nervosa? Psychiatr. Pol. 2017, 51, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Seganfredo, F.B.; Blume, C.A.; Moehlecke, M.; Giongo, A.; Casagrande, D.S.; Spolidoro, J.V.N.; Padoin, A.V.; Schaan, B.D.; Mottin, C.C. Weight-loss interventions and gut microbiota changes in overweight and obese patients: A systematic review. Obes. Rev. 2017, 18, 832–851. [Google Scholar] [CrossRef] [PubMed]
- Alpay, K.; Ertaş, M.; Orhan, E.K.; Üstay, D.K.; Lieners, C.; Baykan, B. Diet restriction in migraine, based on IgG against foods: A clinical double-blind, randomised, cross-over trial. Cephalalgia 2010, 30, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Dong, S.Y.; Wu, L.X.; Li, H.; Sun, Z.J.; Li, J.B.; Jiang, H.X.; Chen, Z.H.; Wang, Q.B.; Chen, W.W. Variable food-specific IgG antibody levels in healthy and symptomatic Chinese adults. PLoS ONE 2013, 8, e53612. [Google Scholar] [CrossRef] [PubMed]
- Lacouture, Y.; Cousineau, D. How to use MATLAB to fit the ex-Gaussian and other probability functions to a distribution of response times. Tutor. Quant. Methods Psychol. 2008, 4, 35–45. [Google Scholar] [CrossRef]
- Field, A.; Hole, G. How to Design and Report Experiments; Sage: Newcastle, UK, 2002. [Google Scholar]
- Zuo, X.; Li, Y.; Li, W.; Guo, Y.; Lu, X.; Li, J.; Desmond, P. Alterations of food antigen-specific serum immunoglobulins G and E antibodies in patients with irritable bowel syndrome and functional dyspepsia. Clin. Exp. Allergy 2007, 37, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Isolauri, E.; Rautava, S.; Kalliomäki, M. Food allergy in irritable bowel syndrome: New facts and old fallacies. Gut 2004, 53, 1391–1393. [Google Scholar] [CrossRef] [PubMed]
- Whorwell, P.; Lea, R. Dietary treatment of the irritable bowel syndrome. Curr. Treat. Opt. Gastroenterol. 2004, 7, 307–316. [Google Scholar] [CrossRef]
- Anthoni, S.; Savilahti, E.; Rautelin, H.; Kolho, K.-L. Milk protein IgG and IgA: The association with milk-induced gastrointestinal symptoms in adults. World J. Gastroenterol. WJG 2009, 15, 4915. [Google Scholar] [CrossRef] [PubMed]
- Aydinlar, E.I.; Dikmen, P.Y.; Tiftikci, A.; Saruc, M.; Aksu, M.; Gunsoy, H.G.; Tozun, N. IgG-based elimination diet in migraine plus irritable bowel syndrome. Headache J. Head Face Pain 2013, 53, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Jiang, T.; Wang, J.; Chang, Y.; Guo, H.; Zhang, W. The value of eliminating foods according to food-specific immunoglobulin G antibodies in irritable bowel syndrome with diarrhoea. J. Int. Med. Res. 2012, 40, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Roque, M.I.; Camilleri, M.; Smyrk, T.; Murray, J.A.; Marietta, E.; O’Neill, J.; Carlson, P.; Lamsam, J.; Janzow, D.; Eckert, D. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: Effects on bowel frequency and intestinal function. Gastroenterology 2013, 144, 903–911.e903. [Google Scholar] [CrossRef] [PubMed]
- Mansueto, P.; D’Alcamo, A.; Seidita, A.; Carroccio, A. Food allergy in irritable bowel syndrome: The case of non-celiac wheat sensitivity. World J. Gastroenterol. WJG 2015, 21, 7089. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Sun, J.; Du, J.; Wang, F.; Fang, R.; Yu, C.; Xiong, J.; Chen, W.; Lu, Z.; Liu, J. Clostridium butyricum exerts a neuroprotective effect in a mouse model of traumatic brain injury via the gut-brain axis. Neurogastroenterol. Motil. 2017. [Google Scholar] [CrossRef] [PubMed]
- Stevens, B.R.; Goel, R.; Seungbum, K.; Richards, E.M.; Holbert, R.C.; Pepine, C.J.; Raizada, M.K. Increased human intestinal barrier permeability plasma biomarkers zonulin and FABP2 correlated with plasma LPS and altered gut microbiome in anxiety or depression. Gut 2017. gutjnl-2017-314759. [Google Scholar] [CrossRef] [PubMed]
- Katzenberger, R.J.; Ganetzky, B.; Wassarman, D.A. The gut reaction to traumatic brain injury. Fly 2015, 9, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Ma, E.L.; Smith, A.D.; Desai, N.; Cheung, L.; Hanscom, M.; Stoica, B.A.; Loane, D.J.; Shea-Donohue, T.; Faden, A.I. Bidirectional brain-gut interactions and chronic pathological changes after traumatic brain injury in mice. Brain Behav. Immun. 2017, 66, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Torres-Platas, S.G.; Cruceanu, C.; Chen, G.G.; Turecki, G.; Mechawar, N. Evidence for increased microglial priming and macrophage recruitment in the dorsal anterior cingulate white matter of depressed suicides. Brain Behav. Immun. 2014, 42, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Setiawan, E.; Wilson, A.A.; Mizrahi, R.; Rusjan, P.M.; Miler, L.; Rajkowska, G.; Suridjan, I.; Kennedy, J.L.; Rekkas, P.V.; Houle, S. Role of translocator protein density, a marker of neuroinflammation, in the brain during major depressive episodes. JAMA Psychiatry 2015, 72, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Hart, G.R. Food-specific IgG guided elimination diet; a role in mental health? BAOJ Nutr. 2017, 3, 045. [Google Scholar]
- Okusaga, O.; Fuchs, D.; Reeves, G.; Giegling, I.; Hartmann, A.M.; Konte, B.; Friedl, M.; Groer, M.; Cook, T.B.; Stearns-Yoder, K.A. Kynurenine and Tryptophan Levels in Patients With Schizophrenia and Elevated Antigliadin Immunoglobulin G Antibodies. Psychosom. Med. 2016, 78, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Severance, E.G.; Gressitt, K.L.; Yang, S.; Stallings, C.R.; Origoni, A.E.; Vaughan, C.; Khushalani, S.; Alaedini, A.; Dickerson, F.B.; Yolken, R.H. Seroreactive marker for inflammatory bowel disease and associations with antibodies to dietary proteins in bipolar disorder. Bipolar Disord. 2014, 16, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Rudzki, L.; Pawlak, D.; Pawlak, K.; Waszkiewicz, N.; Małus, A.; Konarzewska, B.; Gałęcka, M.; Bartnicka, A.; Ostrowska, L.; Szulc, A. Immune suppression of IgG response against dairy proteins in major depression. BMC Psychiatry 2017, 17, 268. [Google Scholar] [CrossRef] [PubMed]
- Köhler, C.; Freitas, T.; Maes, M.; Andrade, N.; Liu, C.; Fernandes, B.; Stubbs, B.; Solmi, M.; Veronese, N.; Herrmann, N. Peripheral cytokine and chemokine alterations in depression: A meta-analysis of 82 studies. Acta Psychiatr. Scand. 2017, 135, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, A.E.; Chapman, K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol. Cell. Endocrinol. 2011, 335, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Zunszain, P.A.; Anacker, C.; Cattaneo, A.; Carvalho, L.A.; Pariante, C.M. Glucocorticoids, cytokines and brain abnormalities in depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Pariante, C.M. Why are depressed patients inflamed? A reflection on 20 years of research on depression, glucocorticoid resistance and inflammation. Eur. Neuropsychopharmacol. 2017, 27, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Lamers, F.; Vogelzangs, N.; Merikangas, K.; De Jonge, P.; Beekman, A.; Penninx, B. Evidence for a differential role of HPA-axis function, inflammation and metabolic syndrome in melancholic versus atypical depression. Mol. Psychiatry 2013, 18, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Shen, J.; Zhao, D.; Qiao, Y.; Xu, A.; Jin, S.; Ran, Z.; Zheng, Q. Serological investigation of food specific immunoglobulin G antibodies in patients with inflammatory bowel diseases. PLoS ONE 2014, 9, e112154. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Lee, K.J. Alterations of Food-specific Serum IgG4 Titers to Common Food Antigens in Patients With Irritable Bowel Syndrome. J. Neurogastroenterol. Motil. 2017, 23, 578. [Google Scholar] [CrossRef] [PubMed]
- Ikechi, R.; Fischer, B.D.; DeSipio, J.; Phadtare, S. Irritable Bowel Syndrome: Clinical Manifestations, Dietary Influences, and Management; Healthcare, Multidisciplinary Digital Publishing Institute: Basel, Switzerland, 2017; p. 21. [Google Scholar]
- Hayes, P.; Corish, C.; Vny, E.; Quigley, E.M.M. A dietary survey of patients with irritable bowel syndrome. J. Hum. Nutr. Dietet. 2014, 27, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Ligaarden, S.C.; Lydersen, S.; Farup, P.G. IgG and IgG4 antibodies in subjects with irritable bowel syndrome: A case control study in the general population. BMC Gastroenterol. 2012, 12, 166. [Google Scholar] [CrossRef] [PubMed]
- Rees, T.; Watson, D.; Lipscombe, S.; Speight, H.; Cousins, P.; Hardman, G.; Dowson, A.J. A prospective audit of food intolerance among migraine patients in primary care clinical practice. Headache Care 2005, 2, 105–110. [Google Scholar]
- Wilders-Truschnig, M.; Mangge, H.; Lieners, C.; Gruber, H.-J.; Mayer, C.; März, W. IgG antibodies against food antigens are correlated with inflammation and intima media thickness in obese juveniles. Exp. Clin. Endocrinol. Diabet. 2008, 116, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Bentz, S.; Hausmann, M.; Piberger, H.; Kellermeier, S.; Paul, S.; Held, L.; Falk, W.; Obermeier, F.; Fried, M.; Schölmerich, J. Clinical relevance of IgG antibodies against food antigens in Crohn’s disease: A double-blind cross-over diet intervention study. Digestion 2010, 81, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Piche, T. Tight junctions and IBS-the link between epithelial permeability, low-grade inflammation, and symptom generation? Neurogastroenterol. Motil. 2014, 26, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Sentsova, T.; Vorozhko, I.; Isakov, V.; Morozov, S.; Shakhovskaia, A. Immune status estimation algorithm in irritable bowel syndrome patients with food intolerance. Exp. Clin. Gastroenterol. 2014, 7, 13–17. [Google Scholar]
- Stapel, S.O.; Asero, R.; Ballmer-Weber, B.; Knol, E.; Strobel, S.; Vieths, S.; Kleine-Tebbe, J. Testing for IgG4 against foods is not recommended as a diagnostic tool: EAACI Task Force Report. Allergy 2008, 63, 793–796. [Google Scholar] [CrossRef] [PubMed]
- Carr, S.; Chan, E.; Lavine, E.; Moote, W. CSACI Position statement on the testing of food-specific IgG. Allergy Asthma Clin. Immunol. 2012, 8, 12. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Allen, K.; Lack, G.; Taylor, S.L.; Donovan, S.M.; Oria, M. Critical Issues in Food Allergy: A National Academies Consensus Report. Pediatrics 2017, 140, e20170194. [Google Scholar] [CrossRef] [PubMed]
- Chabane, H.; Doyen, V.; Bienvenu, F.; Adel-Patient, K.; Vitte, J.; Mariotte, D.; Bienvenu, J. Les dosages d’IgG anti-aliments: Méthodes et pertinence clinique des résultats. Position du groupe de travail de biologie de la Société française d’allergologie. Revue Française d’Allergologie 2018. Available online: https://www.sciencedirect.com/science/article/pii/S1877032018300289 (accessed on 11 April 2018). [CrossRef]
- Engelhart, S.; Glynn, R.J.; Schur, P.H. Disease associations with isolated elevations of each of the four IgG subclasses. Semin. Arthritis and Rheum. 2017, 47, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Aljada, A.; Mohanty, P.; Ghanim, H.; Abdo, T.; Tripathy, D.; Chaudhuri, A.; Dandona, P. Increase in intranuclear nuclear factor κB and decrease in inhibitor κB in mononuclear cells after a mixed meal: Evidence for a proinflammatory effect. Am. J. Clin. Nutr. 2004, 79, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Abautret-Daly, Á.; Dempsey, E.; Parra-Blanco, A.; Medina, C.; Harkin, A. Gut-brain actions underlying comorbid anxiety and depression associated with inflammatory bowel disease. Acta Neuropsychiatr. 2017, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Powell, N.; Walker, M.M.; Talley, N.J. The mucosal immune system: Master regulator of bidirectional gut-brain communications. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 143–159. [Google Scholar] [CrossRef] [PubMed]
- Delaney, S.; Hornig, M. Environmental Exposures and Neuropsychiatric Disorders: What Role Does the Gut–Immune–Brain Axis Play? Curr. Environ. Health Rep. 2018, 5, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Rios-Covian, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; de Los Reyes-Gavilan, C.G.; Salazar, N. Intestinal Short Chain Fatty Acids and their Link with Diet and Human Health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. The Gut-Brain Axis and the Microbiome: Clues to Pathophysiology and Opportunities for Novel Management Strategies in Irritable Bowel Syndrome (IBS). J. Clin. Med. 2018, 7, 6. [Google Scholar] [CrossRef] [PubMed]
| MDD (n = 22) | IBS (n = 22) | HC (n = 21) | Analysis | Post-hoc Analysis | |
|---|---|---|---|---|---|
| Age | 31.5 (14.5) | 38.5 (15.3) | 34 (25) | H = 1.36, p = 0.506 | – |
| Gender (% male) | 50 | 45.4 | 57.1 | χ2 = 1.17, p = 0.554 | – |
| BMI | 28.6 (3.2) | 22.8 (5.5) | 25.8 (5.9) | H = 9.59, p = 0.005 | MDD > IBS |
| Physical activity * | 1 (0) | 2 (0.3) | 2 (1.5) | H = 7.77, p = 0.021 | MDD < IBS |
| Number of cigarettes per day | 0 (2.25) | 0 (0) | 0 (0) | H = 2.45, p = 0.294 | – |
| Number of cups of coffee per day | 2 (2) | 0 (0) | 0 (2) | H = 22.32, p <0.001 | MDD > IBS MDD > HC |
| Intake of alcohol | 4 (7.5) | 4 (1) | 3.5 (3.3) | H = 2.11, p = 0.348 | – |
| Gastric complaints * | 6.5 (6) | 5 (5.5) | 1 (3) | H = 16.25, p <0.001 | MDD > HC IBS > HC |
| Duration of illness | 5 (8) | 3 (11.5) | – | Z = 0.94, p = 0.35 | – |
| ex-Gaussian Parameters | MDD M | IBS M | HC M | H (2, 65) | p | Post-hoc Analysis |
|---|---|---|---|---|---|---|
| µ | 1.640 | 1.820 | 2.027 | 1.178 | 0.554 | – |
| σ | 0.001 | 0.175 | 0.463 | 0.883 | 0.642 | – |
| τ | 8.088 | 5.692 | 2.616 | 19.389 | 0.0001 | MDD > HC IBS > HC |
| IgG | G | M | IQR | Min | Max | Kruskal–Wallis H Test for 3 Groups | Post-hoc Analysis | ||
|---|---|---|---|---|---|---|---|---|---|
| H | p | Groups | p | ||||||
| Total IgG | MDD | 403.9 | 365.1 | 109.68 | 1075.11 | 7.90 | 0.019 | MDD > HC | 0.004 |
| IBS | 308.6 | 282.1 | 108.07 | 1041.08 | |||||
| HC | 219.3 | 70.2 | 130.20 | 657.15 | |||||
| Broccoli | MDD | 5.29 | 3.16 | 1.52 | 21.85 | 6.20 | 0.045 | MDD > HC | 0.039 |
| IBS | 4.73 | 2.72 | 1.79 | 10.43 | |||||
| HC | 3.56 | 1.04 | 1.62 | 6.40 | |||||
| Celery | MDD | 6.15 | 6.80 | 1.58 | 28.20 | 8.03 | 0.017 * | MDD > HC | 0.019 |
| IBS | 4.53 | 4.74 | 1.52 | 12.43 | |||||
| HC | 2.87 | 2.14 | 1.14 | 7.32 | |||||
| Horseradish | MDD | 4.08 | 3.66 | 1.42 | 48.10 | 6.98 | 0.030 | MDD > HC | 0.024 |
| IBS | 4.02 | 2.68 | 1.51 | 11.43 | |||||
| HC | 2.77 | 0.89 | 1.08 | 6.79 | |||||
| Garlic | MDD | 6.33 | 7.02 | 1.01 | 75.64 | 7.88 | 0.017 * | MDD > HC | 0.015 |
| IBS | 2.92 | 4.98 | 0.75 | 15.83 | |||||
| HC | 2.49 | 1.19 | 0.95 | 10.77 | |||||
| Gluten | MDD | 16.44 | 16.10 | 5.26 | 112.61 | 10.37 | 0.005 * | MDD > HC | 0.025 |
| IBS | 8.87 | 10.43 | 2.26 | 117.87 | |||||
| HC | 11.74 | 7.40 | 3.94 | 43.01 | MDD > IBS | 0.010 | |||
| Wheat | MDD | 15.26 | 16.38 | 4.56 | 99.42 | 6.79 | 0.033 | MDD > HC | 0.043 |
| IBS | 9.31 | 8.05 | 2.28 | 122.13 | |||||
| HC | 8.60 | 5.53 | 3.28 | 47.22 | |||||
| Rye | MDD | 11.41 | 8.42 | 3.73 | 108.92 | 7.23 | 0.026 | MDD > HC | 0.032 |
| IBS | 7.38 | 8.14 | 2.10 | 28.52 | |||||
| HC | 5.59 | 4.94 | 2.08 | 38.05 | |||||
| Sunflower seed | MDD | 6.22 | 5.84 | 1.43 | 46.15 | 7.07 | 0.029 | MDD > IBS | 0.038 |
| IBS | 3.06 | 2.94 | 0.91 | 68.05 | |||||
| HC | 3.13 | 2.45 | 0.74 | 61.62 | |||||
| Milk products | MDD | 27.57 | 39.82 | 1.62 | 110.57 | 7.59 | 0.025 | MDD > HC | 0.019 |
| IBS | 20.00 | 42.78 | 1.24 | 119.30 | |||||
| HC | 6.86 | 5.46 | 0.96 | 86.24 | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karakula-Juchnowicz, H.; Gałęcka, M.; Rog, J.; Bartnicka, A.; Łukaszewicz, Z.; Krukow, P.; Morylowska-Topolska, J.; Skonieczna-Zydecka, K.; Krajka, T.; Jonak, K.; et al. The Food-Specific Serum IgG Reactivity in Major Depressive Disorder Patients, Irritable Bowel Syndrome Patients and Healthy Controls. Nutrients 2018, 10, 548. https://doi.org/10.3390/nu10050548
Karakula-Juchnowicz H, Gałęcka M, Rog J, Bartnicka A, Łukaszewicz Z, Krukow P, Morylowska-Topolska J, Skonieczna-Zydecka K, Krajka T, Jonak K, et al. The Food-Specific Serum IgG Reactivity in Major Depressive Disorder Patients, Irritable Bowel Syndrome Patients and Healthy Controls. Nutrients. 2018; 10(5):548. https://doi.org/10.3390/nu10050548
Chicago/Turabian StyleKarakula-Juchnowicz, Hanna, Mirosława Gałęcka, Joanna Rog, Anna Bartnicka, Zuzanna Łukaszewicz, Pawel Krukow, Justyna Morylowska-Topolska, Karolina Skonieczna-Zydecka, Tomasz Krajka, Kamil Jonak, and et al. 2018. "The Food-Specific Serum IgG Reactivity in Major Depressive Disorder Patients, Irritable Bowel Syndrome Patients and Healthy Controls" Nutrients 10, no. 5: 548. https://doi.org/10.3390/nu10050548
APA StyleKarakula-Juchnowicz, H., Gałęcka, M., Rog, J., Bartnicka, A., Łukaszewicz, Z., Krukow, P., Morylowska-Topolska, J., Skonieczna-Zydecka, K., Krajka, T., Jonak, K., & Juchnowicz, D. (2018). The Food-Specific Serum IgG Reactivity in Major Depressive Disorder Patients, Irritable Bowel Syndrome Patients and Healthy Controls. Nutrients, 10(5), 548. https://doi.org/10.3390/nu10050548









