Post-Game High Protein Intake May Improve Recovery of Football-Specific Performance during a Congested Game Fixture: Results from the PRO-FOOTBALL Study
Abstract
1. Introduction
2. Materials and Methods
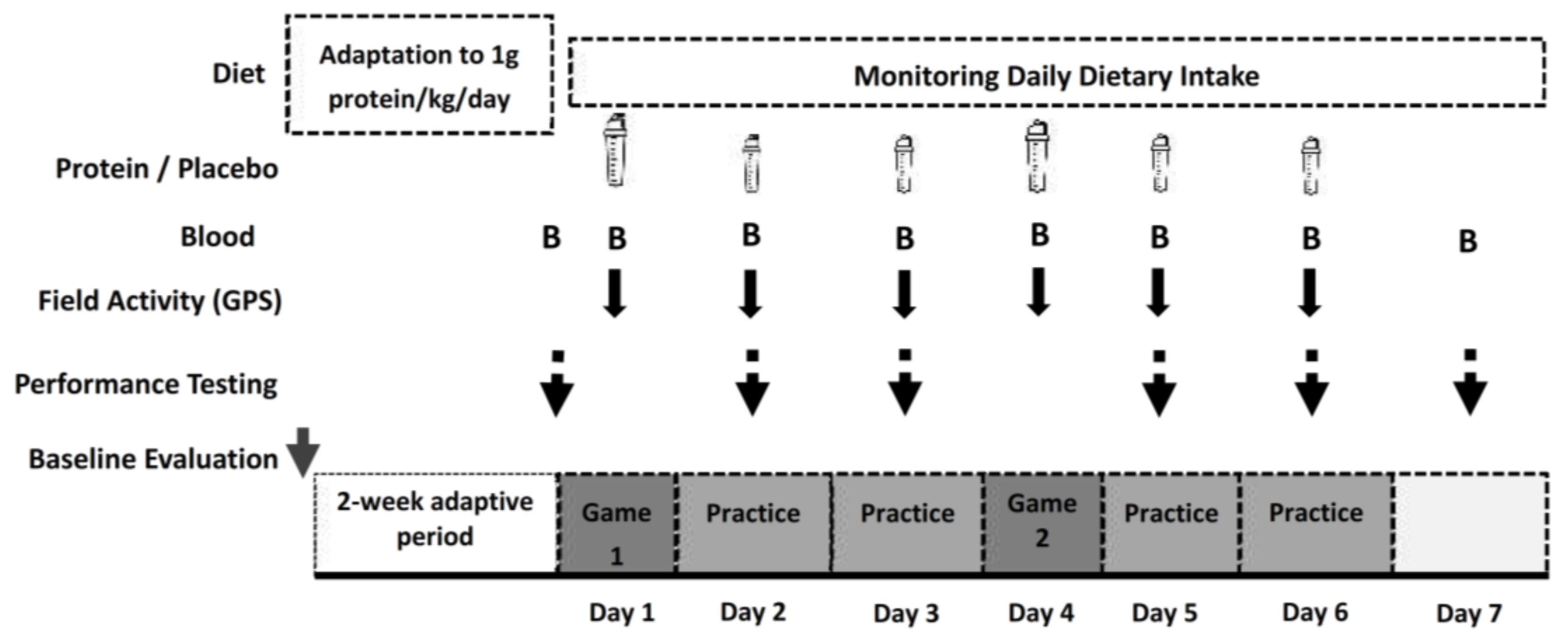
2.1. Experimental Design
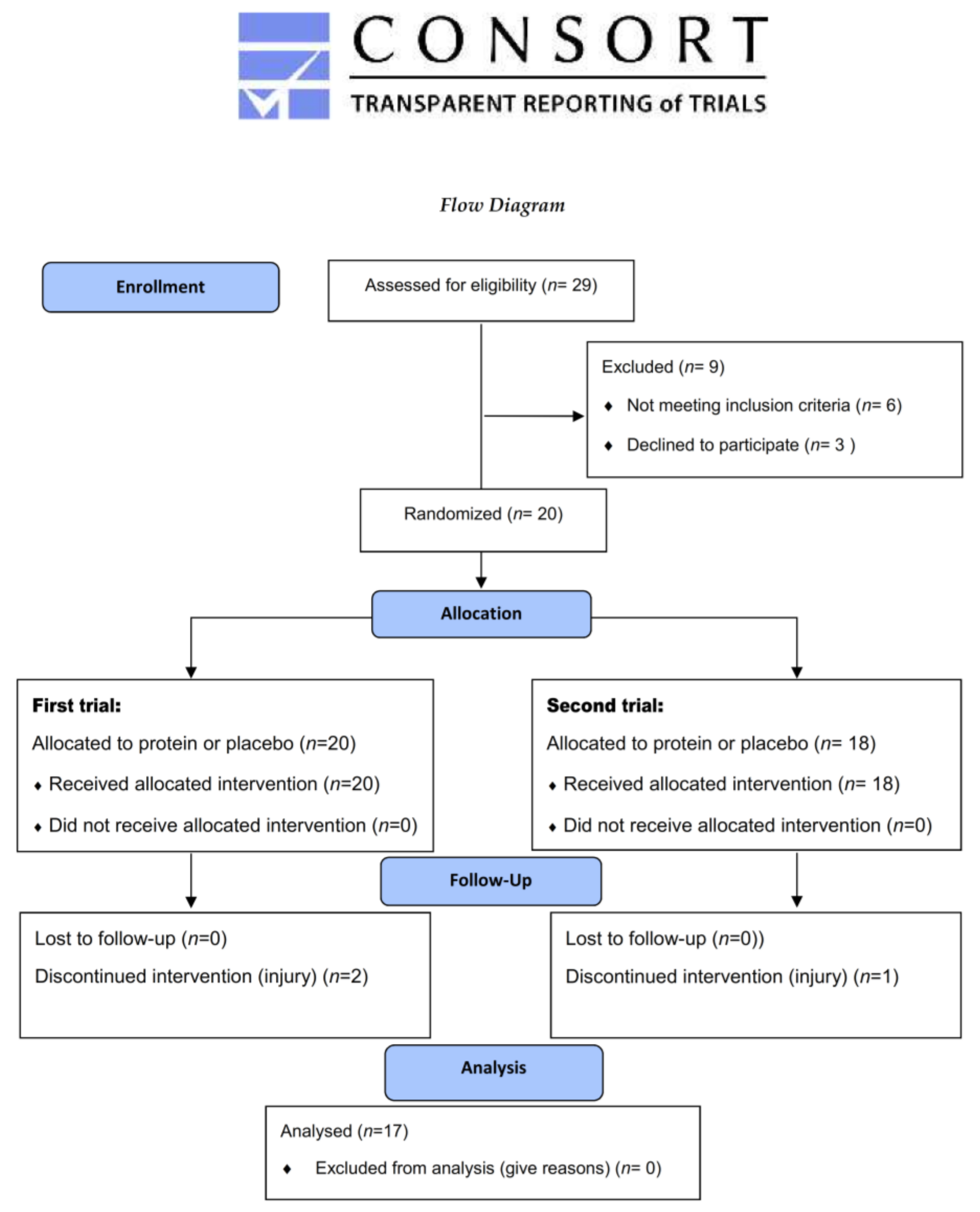
2.2. Participants
2.3. Supplementation Protocol
2.4. Diet Monitoring
2.5. Measurement of Game/Practice Locomotor Activity Pattern
2.6. Descriptives
2.7. Performance Measurements
2.8. Blood Sampling
2.9. Statistics
3. Results
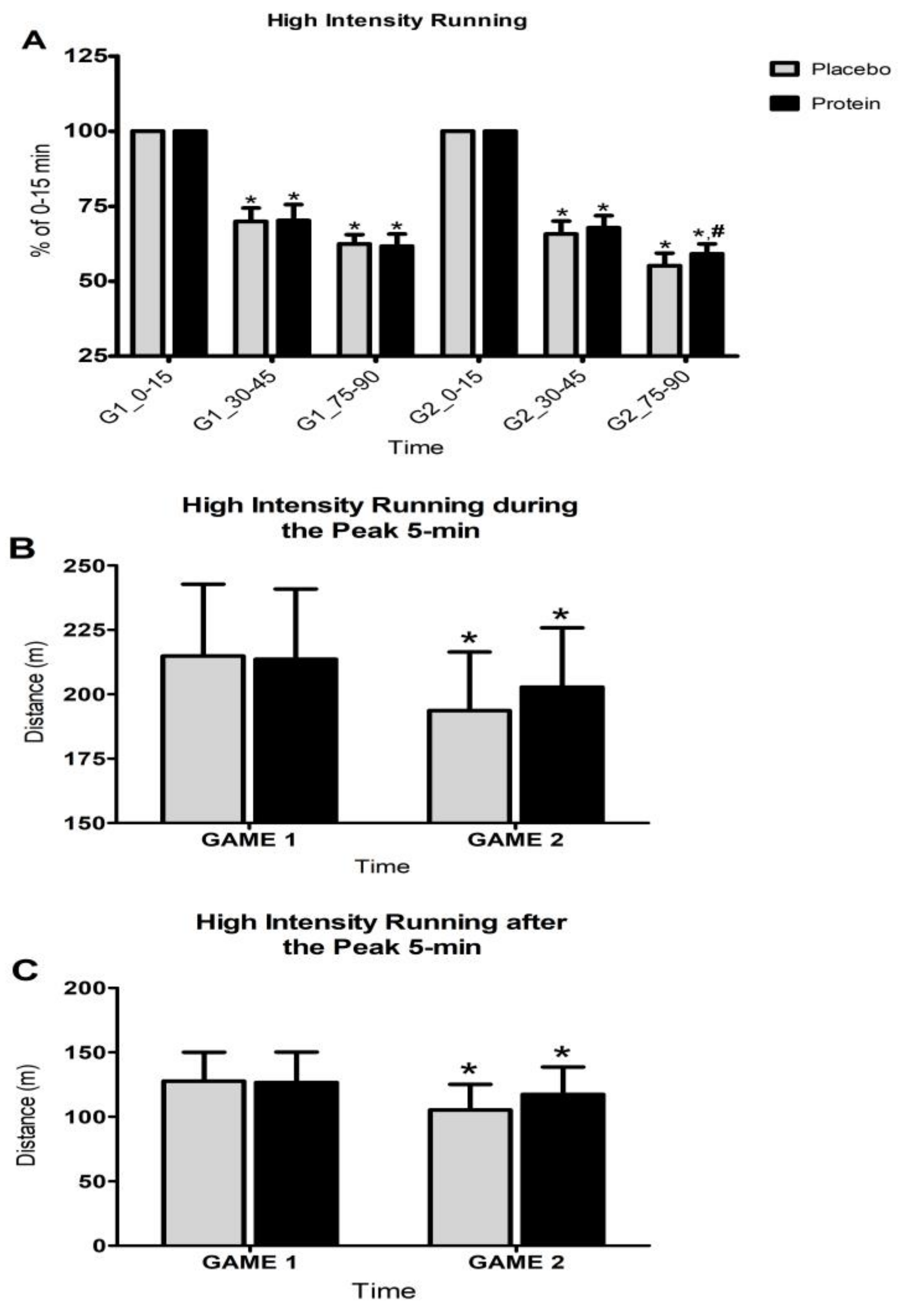
3.1. Game Activity Pattern
3.2. Activity Pattern During Training
3.3. Performance
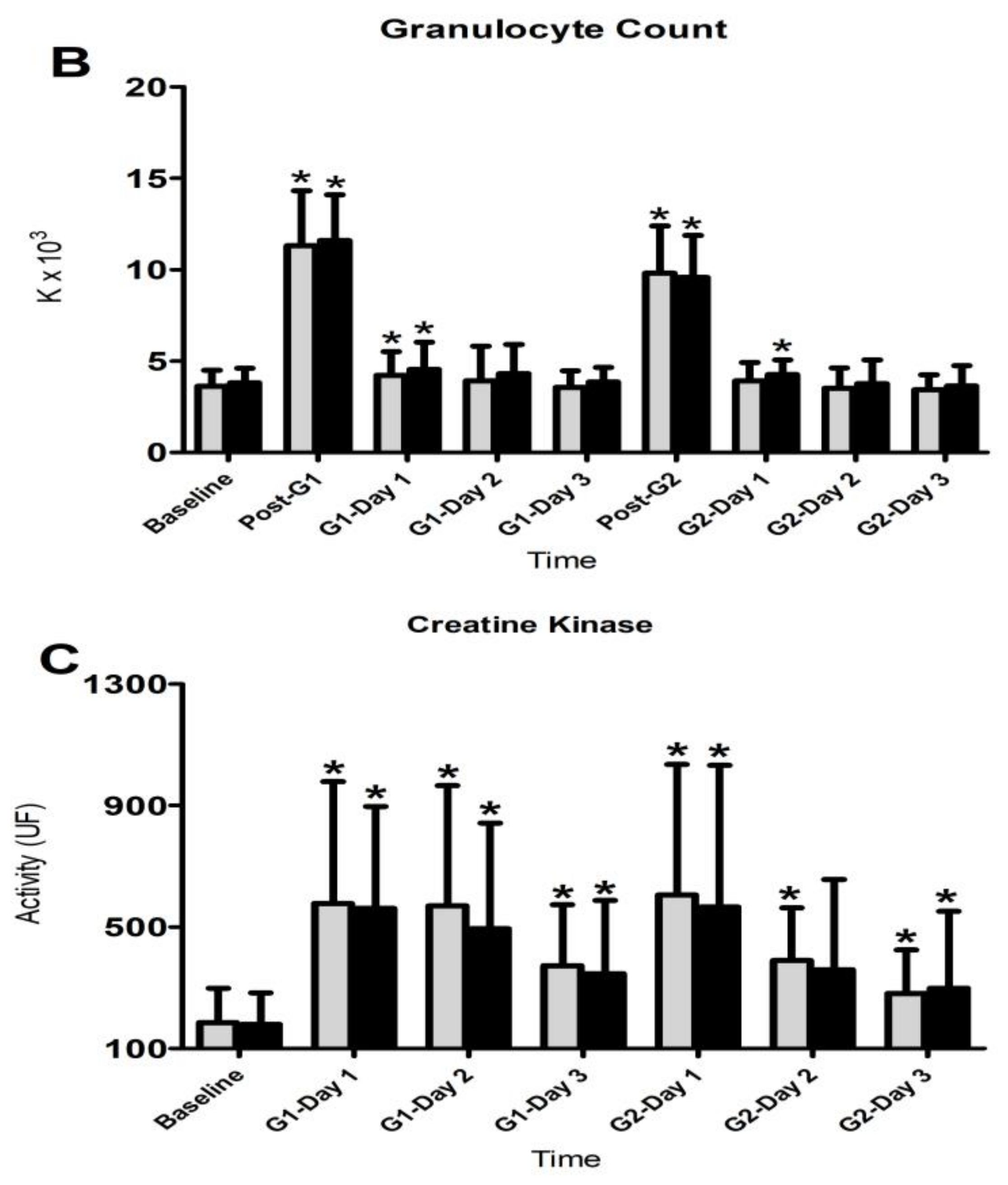
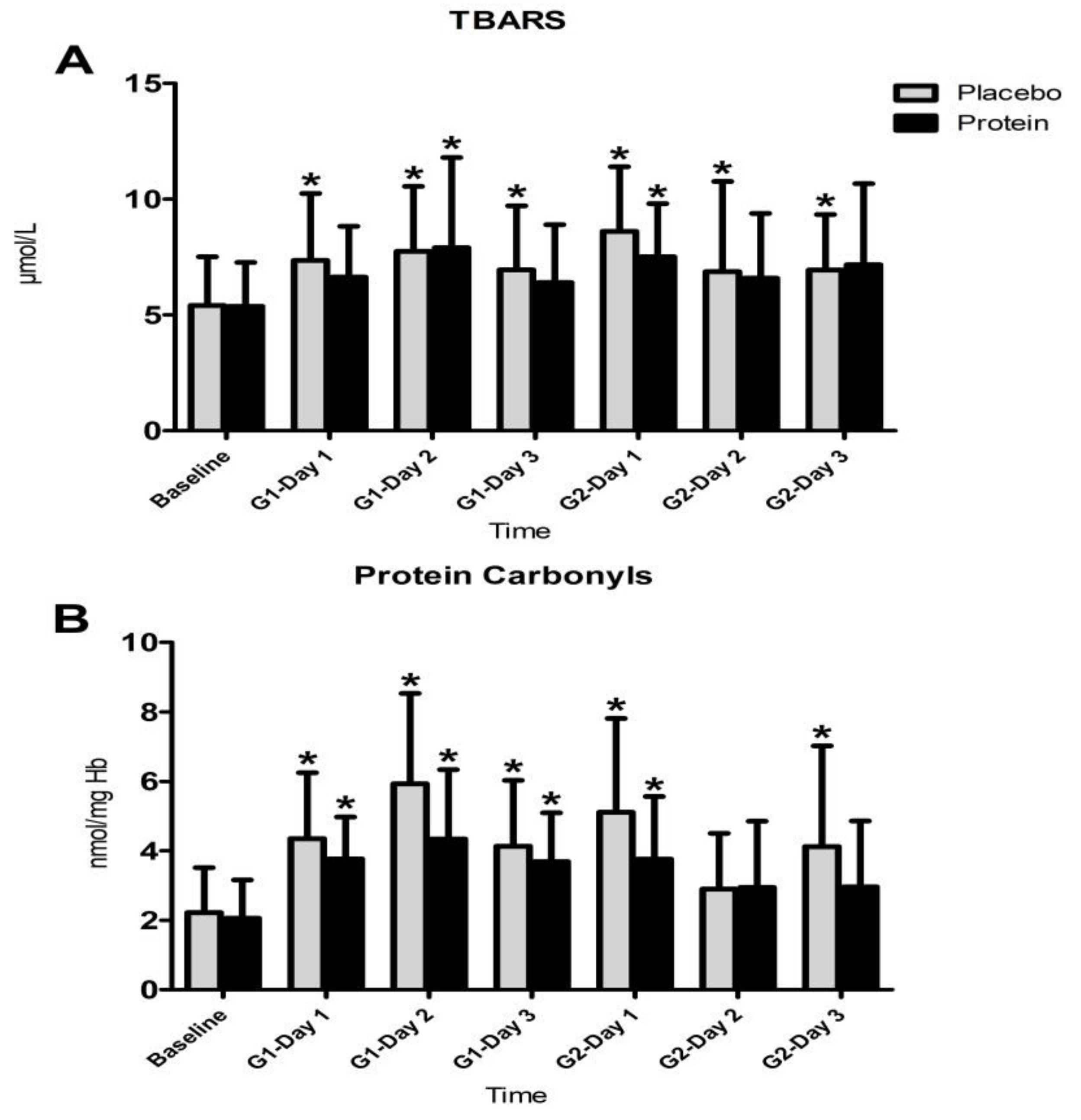
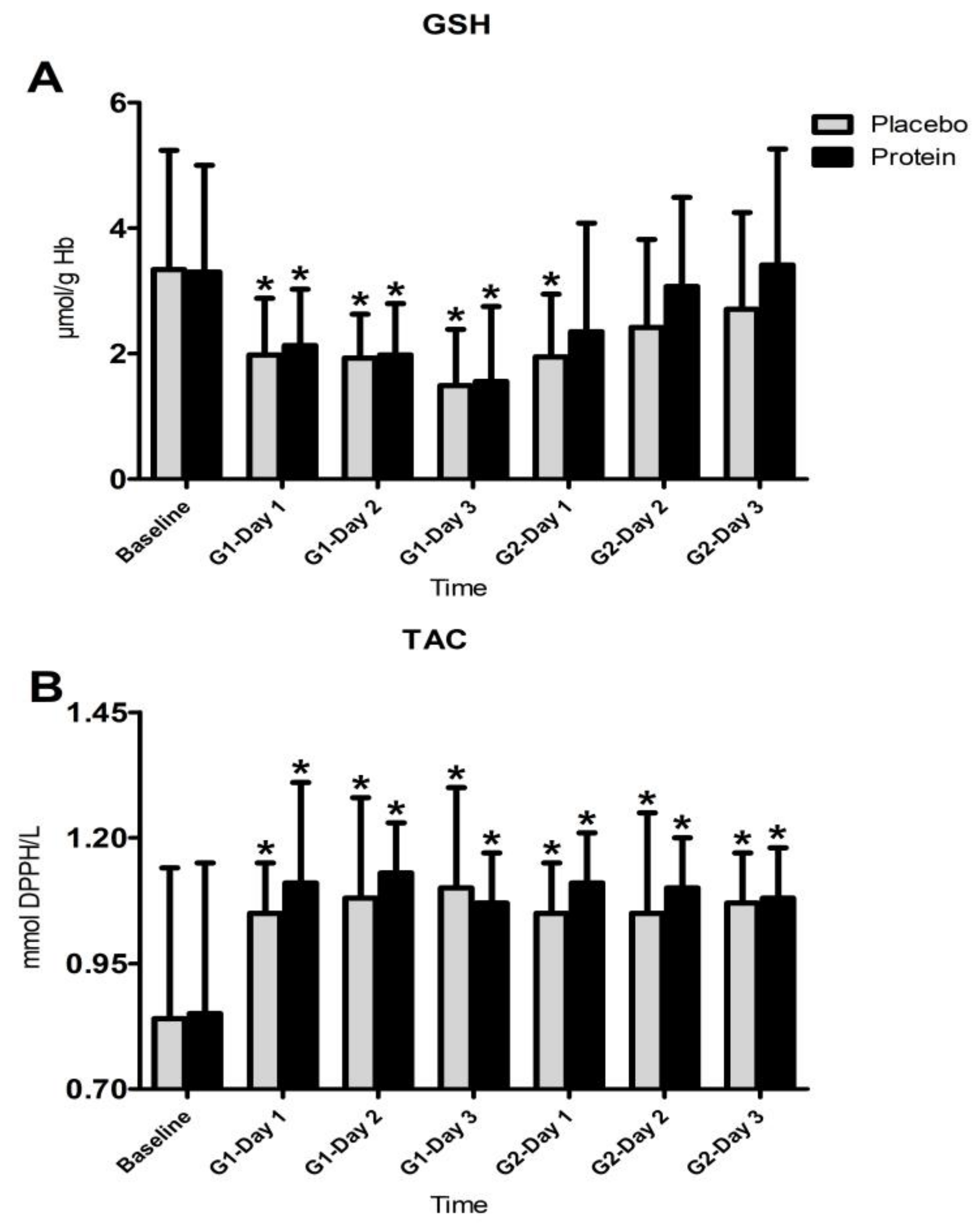
3.4. Inflammatory Responses
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bangsbo, J. The physiology of soccer-with special reference to intense intermittent exercise. Acta Physiol. Scand. Suppl. 1994, 619, 1–155. [Google Scholar] [PubMed]
- Mohr, M.; Krustrup, P.; Bangsbo, J. Game performance of high standard soccer players with special reference to development of fatigue. J. Sports Sci. 2003, 21, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Bradley, S.P.; Sheldon, W.; Wooster, B.; Olsen, P.; Boanas, P.; Krustrup, P. High-intensity running in English FA Premier League soccer games. J. Sports Sci. 2009, 27, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Fatouros, I.G.; Chatzinikolaou, A.; Douroudos, I.I.; Nikolaidis, M.G.; Kyparos, A.; Margonis, K.; Michailidis, Y.; Vantarakis, A.; Taxildaris, K.; Katrabasas, I.; et al. Time course of changes in oxidative stress and antioxidant status responses following a soccer game. J. Strength Cond. Res. 2010, 24, 3278–3286. [Google Scholar] [CrossRef] [PubMed]
- Ispirlidis, I.; Fatouros, I.G.; Jamurtas, A.Z.; Nikolaidis, M.G.; Michailidis, I.; Douroudos, I.; Margonis, K.; Chatzinikolaou, A.; Kalistratos, E.; Katrabasas, I.; et al. Time course of changes in inflammatory and performance responses following a soccer game. Clin. J. Sports Med. 2008, 18, 428–431. [Google Scholar] [CrossRef] [PubMed]
- Jamurtas, A.Z.; Douroudos, I.I.; Deli, C.K.; Draganidis, D.; Chatzinikolaou, A.; Mohr, M.; Avloniti, A.; Barbero-Álvarez, J.C.; Margonis, K.; Mavropalias, G.; et al. Iron status markers are only transiently affected by a football game. J. Sports Sci. 2015, 33, 2088–2099. [Google Scholar] [CrossRef] [PubMed]
- Varley, M.C.; Aughey, R.J. Acceleration profiles in elite Australian soccer. Int. J. Sports Med. 2013, 34, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Andersson, H.; Ekblom, B.; Krustrup, P. Elite football on artificial turf versus natural grass: Movement patterns, technical standards, and player impressions. J. Sports Sci. 2008, 26, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Draganidis, D.; Chatzinikolaou, A.; Barbero-Álvarez, J.C.; Castagna, C.; Douroudos, I.; Avloniti, A.; Margeli, A.; Papassotiriou, I.; Flouris, A.D.; et al. Muscle damage, inflammatory, immune and performance responses to three football games in one week in competitive male players. Eur. J. Appl. Physiol. 2016, 116, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Draganidis, D.; Chatzinikolaou, A.; Avloniti, A.; Barbero-Álvarez, J.C.; Mohr, M.; Malliou, P.; Gourgoulis, V.; Deli, C.K.; Douroudos, I.I.; Margonis, K.; et al. Recovery kinetics of knee flexor and extensor strength after a football game. PLoS ONE 2015, 10, e0128072. [Google Scholar] [CrossRef]
- Fransson, D.; Vigh-Larsen, J.F.; Fatouros, I.G.; Krustrup, P.; Mohr, M. Performance responses in various muscle groups in well-trained competitive male players after a simulated soccer game. J. Hum. Kinet. 2018, 61, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Nedelec, M.; McCall, A.; Carling, C.; Legall, F.; Berthoin, S.; Dupont, G. Recovery in soccer: Part I—post-game fatigue and time course of recovery. Sports Med. 2012, 42, 997–1015. [Google Scholar] [CrossRef] [PubMed]
- Fatouros, I.G.; Jamurtas, A.Z. Insights into the molecular etiology of exercise-induced inflammation: Opportunities for optimizing sport performance. J. Inflamm. Res. 2016, 9, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Krustrup, P.; Nybo, L.; Nielsen, J.J.; Bangsbo, J. Muscle temperature and sprint performance during soccer matches—Beneficial effect of re-warm-up at half-time. Scand. J. Med. Sci. Sports 2004, 14, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Nybo, L.; Grantham, J.; Racinais, S. Physiological responses and physical performance during football in the heat. PLoS ONE 2012, 7, e39202. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Mujika, I.; Santisteban, J.; Randers, M.B.; Bischoff, R.; Solano, R.; Hewitt, A.; Zubillaga, A.; Peltola, E.; Krustrup, P. Examination of fatigue development in elite soccer in a hot environment: A multi-experimental approach. Scand. J. Med. Sci. Sports 2010, 20, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Krustrup, P. Heat stress impairs repeated jump ability after competitive elite soccer games. J. Strength Cond. Res. 2013, 27, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Krustrup, P.; Ortenblad, N.; Nielsen, J.; Nybo, L.; Gunnarsson, T.P.; Iaia, F.M.; Madsen, K.; Stephens, F.; Greenhaff, P.; Bangsbo, J. Maximal voluntary contraction force, SR function and glycogen resynthesis during the first 72 h after a high-level competitive soccer game. Eur. J. Appl. Physiol. 2011, 111, 2987–2995. [Google Scholar] [CrossRef] [PubMed]
- Krustrup, P.; Mohr, M.; Steensberg, A.; Bencke, J.; Kjaer, M.; Bangsbo, J. Muscle and blood metabolites during a soccer game: Implications for sprint performance. Med. Sci. Sports Exerc. 2006, 38, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Marshall, P.W.; Lovell, R.; Jeppesen, G.K.; Andersen, K.; Siegler, J.C. Hamstring muscle fatigue and central motor output during a simulated soccer game. PLoS ONE 2014, 9, e102753. [Google Scholar] [CrossRef] [PubMed]
- Michailidis, Y.; Karagounis, L.G.; Terzis, G.; Jamurtas, A.Z.; Spengos, K.; Tsoukas, D.; Chatzinikolaou, A.; Mandalidis, D.; Stefanetti, R.J.; Papassotiriou, I.; et al. Evidence of potential redox-sensitive regulation of human skeletal muscle’s performance and intracellular signaling following aseptic inflammation induced by damaging Exercise. Am. J. Clin. Nutr. 2013, 98, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Fatouros, I.G.; Jamurtas, A.Z.; Nikolaidis, M.G.; Destouni, A.; Michailidis, Y.; Vrettou, C.; Douroudos, I.I.; Avloniti, A.; Chatzinikolaou, A.; Taxildaris, K.; et al. Time of sampling is crucial for measurement of cell free plasma DNA following acute aseptic inflammation induced by exercise. Clin. Biochem. 2010, 43, 1368–1370. [Google Scholar] [CrossRef] [PubMed]
- Tidball, J.G.; Villalta, S.A. Regulatory interactions between muscle and the immune system during muscle regeneration. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1173–R1187. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.; Nosaka, K.; Suzuki, K. Characterization of inflammatory responses to eccentric exercise in humans. Exerc. Immunol. Rev. 2005, 11, 64–85. [Google Scholar] [PubMed]
- Fatouros, I.G.; Douroudos, I.; Panagoutsos, S.; Pasadakis, P.; Nikolaidis, M.G.; Chatzinikolaou, A.; Sovatzidis, A.; Michailidis, Y.; Jamurtas, A.Z.; Mandalidis, D.; et al. Effects of l-carnitine on oxidative stress responses in patients with renal disease. Med. Sci. Sports Exerc. 2010, 42, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.; Kruger, M.J.; Smith, R.M.; Myburgh, K.H. The inflammatory response to skeletal muscle injury: Illuminating complexities. Sports Med. 2008, 38, 947–969. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, R.; Sunderland, C. Muscle damage, endocrine, and immune marker response to a soccer game. J. Strength Cond. Res. 2012, 26, 2783–2790. [Google Scholar] [CrossRef] [PubMed]
- Heisterberg, M.F.; Fahrenkruq, J.; Krustrup, P.; Storskov, A.; Kjaer, M.; Andersen, J.L. Extensive monitoring through multiple blood samples in professional soccer players. J. Strength Cond. Res. 2013, 27, 1260–1271. [Google Scholar] [CrossRef] [PubMed]
- Ekstand, J.; Walden, M.; Haqqlund, M.A. Congested football calendar and the wellbeing of players: Correlation between game exposure of European footballers before the World Cup 2002 and their injuries and performances during that World Cup. Br. J. Sports Med. 2004, 38, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Dupont, G.; Nedelec, M.; McCall, A.; McCormack, D.; Berthoin, S.; Wisloff, U. Effect of 2 soccer games in a week on physical performance and injury rate. Am. J. Sports Med. 2010, 38, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Minett, G.M.; Costello, J.T. Specificity and context in post-exercise recovery: It is not a one-size-fits-all approach. Front. Physiol. 2015, 3, 130. [Google Scholar] [CrossRef] [PubMed]
- Howatson, F.; van Someren, K.A. The prevention and treatment of exercise-induced muscle damage. Sports Med. 2008, 38, 483–503. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Domenech, E.; Romagnoli, M.; Arduini, A.; Borras, C.; Pallardo, F.V.; Sastre, J.; Vina, J. Oral administration of vitamin C decreases muscle mitochondrial biogenesis and hampers training-induced adaptations in endurance performance. Am. J. Clin. Nutr. 2008, 87, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; Montain, S.J.; Young, A.J. Protein supplementation in U.S. Military personnel. J. Nutr. 2013, 143, 1815S–1819S. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; Lieberman, H.R.; McLellan, T.M. Effects of protein supplements on muscle damage, soreness and recovery of muscle function and physical performance: A systematic review. Sports Med. 2014, 44, 655–670. [Google Scholar] [CrossRef] [PubMed]
- Draganidis, D.; Chatzinikolaou, A.; Jamurtas, A.Z.; Barbero, J.C.; Tsoukas, D.; Theodorou, A.S.; Margonis, K.; Michailidis, Y.; Avloniti, A.; Theodorou, A.; et al. The time-frame of acute resistance exercise effects on football skill performance: The impact of exercise intensity. J. Sport Sci. 2013, 31, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Lunn, W.R.; Pasiakos, S.M.; Colletto, M.R.; Karfonta, K.E.; Carbone, J.W.; Anderson, J.M.; Rodriguez, N.R. Chocolate milk and endurance exercise recovery: Protein balance, glycogen, and performance. Med. Sci. Sports Exerc. 2012, 44, 682–691. [Google Scholar] [CrossRef] [PubMed]
- Breen, L.; Philip, A.; Witard, O.C.; Jackman, S.R.; Selby, A.; Smith, K.; Baar, K.; Tipton, K.D. The influence of carbohydrate–protein co-ingestion following endurance exercise on myofibrillar and mitochondrial protein synthesis. J. Physiol. 2011, 589, 4011–4025. [Google Scholar] [CrossRef] [PubMed]
- Koopman, R.; Wagenmakers, A.J.M.; Manders, R.J.F.; Zorenc, A.H.; Senden, J.M.; Gorselink, M.; Keizer, H.A.; van Loon, L.J. Combined ingestion of protein and free leucine with carbohydrate increases postexercise muscle protein synthesis in vivo in male subjects. Am. J. Physiol. 2005, 288, E645–E653. [Google Scholar] [CrossRef] [PubMed]
- Gilson, S.F.; Saunders, M.J.; Moran, C.W.; Moore, R.W.; Womack, C.J.; Todd, M.K. Effects of chocolate milk consumption on markers of muscle recovery following soccer training: A randomized cross-over study. J. Int. Soc. Sports Nutr. 2010, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, T.P.; Bendiksen, M.; Bischoff, R.; Christensen, P.M.; Lesivig, B.; Madsen, K.; Stephens, F.; Greenhaff, P.; Krustrup, P.; Bangsbo, J. Effect of whey protein- and carbohydrate-enriched diet on glycogen resynthesis during the first 48 h after a soccer game. Scand. J. Med. Sci. Sports 2013, 23, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Highton, J.; Twist, C.; Lamb, K.; Nicholas, C. Carbohydrate-protein coingestion improves multiple-sprint running performance. J. Sports Sci. 2013, 31, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Naclerio, F.; Larumbe-Zabala, E.; Cooper, R.; Allgrove, J.; Earnest, C.P. A multi-ingredient containing carbohydrate, proteins l-glutamine and l-carnitine attenuates fatigue perception with no effect on performance, muscle damage or immunity in soccer players. PLoS ONE 2015, 10, e0125188. [Google Scholar] [CrossRef] [PubMed]
- Naclerio, F.; Larumbe-Zabala, E.; Cooper, R.; Jimenez, A.; Goss-Sampson, M. Effect of a carbohydrate-protein multi-ingredient supplement on intermittent sprint performance and muscle damage in recreational athletes. Appl. Physiol. Nutr. Metab. 2014, 39, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Packer, J.E.; Wooding, D.J.; Kato, H.; Courtney-Martin, G.; Pencharz, P.B.; Moore, D.R. Variable-intensity simulated team-sport exercise increases daily protein requirements in active males. Front. Nutr. 2017, 4, 64. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Wilborn, C.D.; Taylor, L.; Campbell, B.; Almada, A.L.; Collins, R.; Cooke, M.; Earnest, C.P.; Greenwood, M.; Kalman, D.S.; et al. ISNN exercise & sport nutrition review: Research & recommendations. J. Int. Soc. Sports Nutr. 2010, 7, 7. [Google Scholar] [CrossRef]
- Elango, R.; Humayun, M.A.; Ballm, R.O.; Pencharz, P.B. Evidence that protein requirements have been significantly underestimated. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Draganidis, D.; Chondrogianni, N.; Chatzinikolaou, A.; Terzis, G.; Karagounis, L.G.; Sovatzidis, A.; Avloniti, A.; Lefaki, M.; Protopapa, M.; Deli, C.K.; et al. Protein ingestion preserves proteasome activity during intense aseptic inflammation and facilitates skeletal muscle recovery in humans. Br. J. Nutr. 2017, 118, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Areta, J.L.; Burke, L.M.; Ross, M.L.; Camera, D.M.; West, D.W.; Broad, E.M.; Jeacocke, N.A.; Moore, D.R.; Stellingwerff, T.; Phillips, S.M.; et al. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J. Physiol. 2013, 591, 2319–2331. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Areta, J.; Coffey, V.G.; Stellingwerff, T.; Phillips, S.M.; Burke, L.M.; Cléroux, M.; Godin, J.P.; Hawley, J.A. Daytime pattern of post-exercise protein intake affects whole-body protein turnover in resistance-trained males. Nutr. Metab. 2012, 9, 91. [Google Scholar] [CrossRef] [PubMed]
- Sakelliou, A.; Fatouros, I.; Athanailidis, I.; Tsoukas, D.; Chatzinikolaou, A.; Draganidis, D.; Liacos, C.; Papassotiriou, I.; Mandalidis, D.; Jamurtas, A.Z.; et al. Evidence of a redox-dependent regulation of immune responses to aseptic inflammation induced by exercise. Oxid. Med. Cell. Longev. 2016, 2016, 2840643. [Google Scholar] [CrossRef] [PubMed]
- Fatouros, I.G.; Chatzinikolaou, A.; Tournis, S.; Nikolaidis, M.G.; Jamurtas, A.Z.; Douroudos, I.I.; Papassotiriou, I.; Thomakos, P.M.; Taxildaris, K.; Mastorakos, G.; et al. Intensity of resistance exercise determines adipokine and resting energy expenditure responses in overweight elderly individuals. Diabetes Care 2009, 32, 2161–2167. [Google Scholar] [CrossRef] [PubMed]
- Bangsbo, J.; Mohr, M. Fitness Testing in Football. Fitness Training in Soccer II; Bangsbosport: Copenhagen, Denmark, 2012. [Google Scholar]
- Barbas, I.; Fatouros, I.G.; Douroudos, I.I.; Chatzinikolaou, A.; Michailidis, Y.; Jamurtas, A.Z.; Draganidis, D.; Nikolaidis, M.G.; Parotsidis, C.; Theodorou, A.T.; et al. Physiological and performance adaptations of elite Greco-Roman wrestlers during a one-day tournament. Eur. J. Appl. Physiol. 2011, 111, 1421–1436. [Google Scholar] [CrossRef] [PubMed]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, A.A.; Nikolaidis, M.G.; Paschalis, V.; Sakellariou, G.K.; Fatouros, I.G.; Koutedakis, Y.; Jamurtas, A.Z. Comparison between glucose-6-phosphate dehydrogenase-defcient and normal individuals after eccentric exercise. Med. Sci. Sports Exerc. 2010, 42, 1113–1121. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, J.; Rebelo, A.; Oliveira, E.; Silva, J.R.; Marques, F.; Ascensao, A. Impact of Loughborough intermittent shuttle test versus soccer game on physiological, biochemical and neuromuscular parameters. Eur. J. Appl. Physiol. 2010, 108, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Randers, M.B.; Mujika, I.; Hewitt, A.; Santisteban, J.; Bischoff, R.; Solano, R.; Zubillaga, A.; Peltola, E.; Krustrup, P.; Mohr, M. Application of four different football game analysis systems: A comparative study. J. Sports Sci. 2010, 28, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.R.; Ascensão, A.; Marques, F.; Seabra, A.; Rebelo, A.; Magalhães, J. Neuromuscular function, hormonal and redox status and muscle damage of professional soccer players after a high-level competitive game. Eur. J. Appl. Physiol. 2013, 113, 2193–2201. [Google Scholar] [CrossRef] [PubMed]
- Gentle, H.L.; Love, T.D.; Howe, A.S.; Black, K.E. A randomised trial of pre-exercise meal composition on performance and muscle damage in well-trained basketball players. J. Int. Soc. Sports Nutr. 2014, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Arent, S.M.; Pellegrino, J.K.; Williams, C.A.; DiFabio, D.A.; Greenwood, J.C. Nutritional supplementation, performance, and oxidative stress in college soccer players. J. Strength Cond. Res. 2010, 24, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, E.; Bell, P.G.; Stevenson, E. Effect of milk on team sport performance after exercise-induced muscle damage. Med. Sci. Sports Exerc. 2013, 45, 1585–1592. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.A.; Toone, R.J.; Stokes, K.A.; Thompson, D. Systemic indices of skeletal muscle damage and recovery of muscle function after exercise: Effect of combined carbohydrate-protein ingestion. Appl. Physiol. Nutr. Metab. 2009, 34, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Foure, A.; Wegrzyk, J.; Le Fur, Y.; Mattei, J.P.; Boudinet, H.; Vilmen, C.; Bendahan, D.; Gondin, J. Impaired mitochondrial function and reduced energy cost as a result of muscle damage. Med. Sci. Sports Exerc. 2015, 47, 1135–1144. [Google Scholar] [CrossRef] [PubMed]
- Foure, A.; Nosaka, K.; Gastaldi, M.; Mattei, J.P.; Boudinet, H.; Guye, M.; Vilmen, C.; Le Fur, Y.; Bendahan, D.; Gondin, J. Effects of branched-chain amino acids supplementation on both plasma amino acids concentration and muscle energetics changes resulting from muscle damage: A randomized placebo controlled trial. Clin. Nutr. 2016, 35, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Tatpati, L.L.; Irving, B.A.; Tom, A.; Bigelow, M.L.; Klaus, K.; Short, K.R.; Nair, K.S. The effect of branched chain amino acids on skeletal muscle mitochondrial function in young and elderly adults. J. Clin. Endocrinol. Metab. 2010, 95, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.; Bartlett, J.D.; van de Graaf, C.S.; Louhelainen, J.; Coyne, V.; Iqbal, Z.; Maclaren, D.P.; Gregson, W.; Close, G.L.; Morton, J.P. Protein ingestion does not impair exercise-induced AMPK signalling when in a glycogen-depleted state: Implications for train-low compete-high. Eur. J. Appl. Physiol. 2013, 113, 1457–1468. [Google Scholar] [CrossRef] [PubMed]
- Babault, N.; Deley, G.; Le Ruyet, P.; Morgan, F.; Allaert, F.A. Effects of soluble milk protein or casein supplementation on muscle fatigue following resistance training program: A randomized, double-blind, and placebo-controlled study. J. Int. Soc. Sports Nutr. 2014, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Lv, J.; You, S.; Zhao, Q.; Chen, X.; Hu, X. Supplementation with oat protein ameliorates exercise-induced fatigue in mice. Food Funct. 2013, 4, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.B.; Rybalka, E.; Stathis, C.G.; Cribb, P.J.; Hayes, A. Whey protein isolate attenuates strength decline after eccentrically-induced muscle damage in healthy individuals. J. Int. Soc. Sports Nutr. 2010, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Buckley, J.D.; Thomson, R.L.; Coates, A.M.; Howe, P.R.; DeNichilo, M.O.; Rowney, M.K. Supplementation with a whey protein hydrolysate enhances recovery of muscle force-generating capacity following eccentric exercise. J. Sci. Med. Sport 2010, 13, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Derave, W.; Ozdemir, M.S.; Harris, R.C.; Pottier, A.; Reyngoudt, H.; Koppo, K.; Wise, J.A.; Achten, E. beta-Alanine supplementation augments muscle carnosine content and attenuates fatigue during repeated isokinetic contraction bouts in trained sprinters. J. Appl. Physiol. 2007, 103, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.E.; Moon, J.R.; Kendall, K.L.; Graef, J.L.; Lockwood, C.M.; Walter, A.A.; Beck, T.W.; Cramer, J.T.; Stout, J.R. The effects of beta-alanine supplementation and high-intensity interval training on neuromuscular fatigue and muscle function. Eur. J. Appl. Physiol. 2009, 105, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Jackman, S.R.; Witard, O.C.; Jeukendrup, A.E.; Tipton, K.D. Branched-chain amino acid ingestion can ameliorate soreness from eccentric exercise. Med. Sci. Sports Exerc. 2010, 42, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, S.; Dhawan, M.; Singh Sandhu, J. Four weeks of supplementation with isolated soy protein attenuates exercise-induced muscle damage and enhances muscle recovery in well trained athletes: A randomized trial. Asian J. Sports Med. 2016, 7, e33528. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; Hoad, M.; Goodall, S.; Tallent, J.; Bell, P.G.; French, D.N. Exercise-induced muscle damage is reduced in resistance-trained males by branched chain amino acids: A randomized, double-blind, placebo controlled study. J. Int. Soc. Sports Nutr. 2012, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Rahbek, S.K.; Farup, J.; de Paoli, F.; Vissing, K. No differential effects of divergent isocaloric supplements on signaling for muscle protein turnover during recovery from muscle-damaging eccentric exercise. Amino Acids 2015, 47, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Rössler, K.; Thorp, R.M.; Graham, D.F.; Timmons, B.W.; Stannard, S.R.; Tarnopolsky, M.A. Effect of dietary protein content during recovery from high-intensity cycling on subsequent performance and markers of stress, inflammation, and muscle damage in well-trained men. Appl. Physiol. Nutr. Metab. 2008, 33, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.R.; Ratamess, N.A.; Tranchina, C.P.; Rashti, S.L.; Kang, J.; Faigenbaum, A.D. Effect of proprietary protein supplement on recovery indices following resistance exercise in strength/power athletes. Amino Acids 2010, 38, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Farup, J.; Rahbek, S.K.; Knudsen, I.S.; de Paoli, F.; Mackey, A.L.; Vissing, K. Whey protein supplementation accelerates satellite cell proliferation during recovery from eccentric exercise. Amino Acids 2014, 46, 2503–2516. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.J.S.; Oliver, S.J.; Laing, S.J.; Walters, R.; Bilzon, J.L.J.; Walsh, N.P. Influence of timing of postexercise carbohydrate-protein ingestion on selected immune indices. Int. J. Sport Nutr. Exerc. Metab. 2009, 19, 366–385. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.J.S.; Walters, R.; Bilzon, J.L.J.; Walsh, N.P. Effects of immediate postexercise carbohydrate ingestion with and without protein on neutrophil degranulation. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 205–213. [Google Scholar] [CrossRef]
- Powell, J.D.; Delgoffe, G.M. The mammalian target of rapamycin: Linking T cell differentiation, function, and metabolism. Immunity 2010, 33, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Moinard, C.; Caldefie-Chezet, F.; Walrand, S.; Vasson, M.P.; Cynober, L. Evidence that glutamine modulates respiratory burst in stressed rat polymorphonuclear cells through its metabolism into arginine. Br. J. Nutr. 2002, 88, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Kurihara, S.; Koikawa, N.; Nakamura, A.; Aoki, K.; Yosigi, H.; Sawaki, K.; Ohtani, M. Effects of oral supplementation with cystine and theanine on the immune function of athletes in endurance exercise: Randomized, double-blind, placebo-controlled trial. Biosci. Biotechnol. Biochem. 2009, 73, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Naclerio, F.; Larumbe Zabala, E.; Ashrafi, N.; Seijo, M.; Nielsen, B.; Allgrove, J.; Earnest, C.P. Effects of protein–carbohydrate supplementation on immunity and resistance training outcomes: A double blind, randomized, controlled clinical trial. Eur. J. Appl. Physiol. 2017, 117, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.R.; Jackson, L.; Clarke, J.; Stellingwerff, T.; Broadbent, S.; Rowlands, D.S. Effect of post-exercise protein–leucine feeding on neutrophil function, immunomodulatory plasma metabolites and cortisol during a 6-day block of intense cycling. Eur. J. Appl. Physiol. 2013, 113, 2211–2222. [Google Scholar] [CrossRef] [PubMed]
- Middleton, N.; Jelen, P.; Bell, G. Whole blood and mononuclear cell glutathione response to dietary whey protein supplementation in sedentary and trained male human subjects. Int. J. Food Sci. Nutr. 2004, 55, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Cury-Boaventura, M.F.; Levada-Pires, A.C.; Gorjão, A.F.R.; Alba-Loureiro, T.C.; Hirabara, S.M.; Peres, F.P.; Silva, P.R.S.; Curi, R.; Pithon-Curi, T.C. Effects of exercise on leukocyte death: Prevention by hydrolyzed whey protein enriched with glutamine dipeptide. Eur. J. Appl. Physiol. 2008, 103, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Kuang, S.; Kuroda, K.; Le Grand, F.; Rudnicki, M.A. Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell 2007, 129, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Farup, J.; Rahbek, S.K.; Riis, S.; Vendelbo, M.H.; Paoli, F.D.; Vissing, K. Influence of exercise contraction mode and protein supplementation on human skeletal muscle satellite cell content and muscle fiber growth. J. Appl. Phyiol. 2014, 117, 898–909. [Google Scholar] [CrossRef] [PubMed]
- Reidy, P.T.; Fry, C.S.; Igbinigie, S.; Deer, R.R.; Jennings, K.; CopE, M.B.; Mukherjea, R.; Volpi, E.; Rasmussen, B.B. Protein Supplementation does not affect myogenic adaptations to resistance training. Med. Sci. Sports Exerc. 2017, 49, 1197–1208. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.G.; Silva, M.T.; da Cunha, F.M.; Moriscot, A.S.; Aoki, M.S.; Miyabara, E.H. Leucine supplementation improves regeneration of skeletal muscles from old rats. Exp. Gerontol. 2015, 72, 269–277. [Google Scholar] [CrossRef] [PubMed]
- West, D.W.D.; Sawan, S.A.; Mazzulla, M.; Williamson, E.; Moore, D.R. Whey protein supplementation enhances whole body protein metabolism and performance recovery after resistance exercise: A double-blind crossover study. Nutrients 2017, 9, 735. [Google Scholar] [CrossRef] [PubMed]
- Nicastro, H.; da Luz, C.R.; Chaves, D.F.S.; Bechara, L.R.G.; Voltarelli, V.A.; Rogero, M.M.; Lancha, A.H. Does branched-chain amino acids supplementation modulate skeletal muscle remodeling through inflammation modulation? Possible mechanisms of action. J. Nutr. Metabol. 2012, 2012, 136937. [Google Scholar] [CrossRef] [PubMed]

| Male (n = 20) | |
|---|---|
| Age (years) | 20.6 ± 1.1 |
| Height (cm) | 179.1 ± 4.6 |
| Weight (kg) | 77.6 ± 5.8 |
| BMI (kg/m2) | 24.0 ± 1.0 |
| Body fat (%) | 9.9 ± 2.2 |
| RMR (kj/day) | 6099.8 ± 641.40 |
| VO2max (mL/kg/min) | 57.7 ± 3.4 |
| Maximal heart rate (bpm) | 196.8 ± 6.3 |
| Yo-Yo IE2 (m) | 2890.5 ± 303.7 |
| Yo-Yo IR2 (m) | 1638.44 ± 153.8 |
| PLA | PRO | |||||
|---|---|---|---|---|---|---|
| Diet | Placebo | Total | Diet | Supplement | Total | |
| Daily Energy Intake (kJ/kg BM/day) | ||||||
| Adaptive period | 119.1 ± 8.1 | N/A | 119.1 ± 8.1 | 120.1 ± 8.0 | N/A | 120.1 ± 8.0 |
| Game days | 191.1 ± 8.2 | 22.8 ± 0.0 | 213.9 ± 8.2 | 191.2 ± 7.4 | 23.0 ± 0.0 | 214.2 ± 7.4 |
| Training days | 152.4 ± 6.4 | 5.2 ± 0.0 | 157.6 ± 6.4 | 152.0 ± 6.3 | 5.25 ± 0.0 | 157.2 ± 6.3 |
| Carbohydrate intake | ||||||
| Adaptive period (g/kg BM) | 3.72 ± 0.2 | N/A | 3.72 ± 0.2 | 3.78 ± 0.2 | N/A | 3.78 ± 0.2 |
| Adaptive period (% of total intake) | 52.2 ± 2.4 | 52.6 ± 2.1 | ||||
| Game days: pre-game period (g/kg BM) | 2.27 ± 0.4 | N/A | 2.27 ± 0.4 | 2.28 ± 0.4 | N/A | 2.28 ± 0.4 |
| Game days: post-game period (g/kg BM) | 4.46 ± 0.2 | 1.37 ± 0.0 | 5.83 ± 0.2 | 4.50 ± 0.1 | 0.19 ± 0.0 | 4.69 ± 0.1* |
| Game days, total (total g/kg BM) | 6.74 ± 0.3 | 1.37 ± 0.0 | 8.11 ± 0.3 | 6.78 ± 0.4 | 0.19 ± 0.0 | 6.97 ± 0.4* |
| Game days (% of total intake) | 71.0 ± 1.3 | 61.0 ± 1.4 | ||||
| Training days (g/kg BM) | 5.11 ± 0.3 | 0.31 ± 0.0 | 5.44 ± 0.3 | 5.17 ± 0.2 | 0.05 ± 0.0 | 5.22 ± 0.2 |
| Training days (% of total intake) | 59.7 ± 3.8 | 57.4 ± 3.9 | ||||
| Protein intake | ||||||
| Adaptive period (g/kg BM) | 1.03 ± 0.05 | N/A | 1.03 ± 0.05 | 1.05 ± 0.04 | N/A | 1.05 ± 0.04 |
| Adaptive period (g/kg FFM) | 1.15 ± 0.06 | N/A | 1.15 ± 0.06 | 1.17 ± 0.05 | N/A | 1.17 ± 0.05 |
| Adaptive period (% of total intake) | 14.5 ± 1.2 | 14.6 ± 1.1 | ||||
| Game days: pre-game period (g/kg BM) | 0.85 ± 0.05 | N/A | 0.85 ± 0.05 | 0.86 ± 0.05 | N/A | 0.86 ± 0.05 |
| Game days: pre-game period (g/kg FFM) | 0.90 ± 0.2 | N/A | 0.90 ± 0.2 | 0.95 ± 0.05 | N/A | 0.95 ± 0.05 |
| Game days: post-game recovery (g/kg BM) | 0.47 ± 0.04 | 0.0 ± 0.0 | 0.47 ± 0.04 | 0.46 ± 0.04 | 1.03 ± 0.0 | 1.49 ± 0.04 * |
| Game days: post-game recovery (g/kg FFM) | 0.53 ± 0.05 | 0.0 ± 0.0 | 0.53 ± 0.05 | 0.52 ± 0.05 | 1.15 ± 0.0 | 1.67 ± 0.05 |
| Game days, total (g/kg BM) | 1.33 ± 0.1 | 0.0 ± 0.0 | 1.33 ± 0.1 | 1.32 ± 0.07 | 1.03 ± 0.0 | 2.35 ± 0.07 * |
| Game days (g/kg FFM) | 1.48 ± 0.1 | 0.0 ± 0.0 | 1.48 ± 0.1 | 1.47 ± 0.07 | 1.15 ± 0.0 | 2.62 ± 0.07 * |
| Game days (% of total intake) | 13.0 ± 1.1 | 22.9 ± 0.4 | ||||
| Training days (g/kg BM) | 1.21 ± 0.04 | 0.0 ± 0.0 | 1.21 ± 0.04 | 1.22 ± 0.04 | 0.26 ± 0.0 | 1.48 ± 0.04 |
| Training days (g/kg FFM) | 1.35 ± 0.06 | 0.0 ± 0.0 | 1.35 ± 0.06 | 1.36 ± 0.06 | 0.29 ± 0.0 | 1.65 ± 0.06 |
| Training days (% of total intake) | 14.8 ± 0.9 | 18.2 ± 1.0 | ||||
| Fat intake (g/kg) | ||||||
| Adaptive period (g/kg BM) | 1.05 ± 0.1 | N/A | 1.05 ± 0.1 | 1.06 ± 0.1 | N/A | 1.06 ± 0.1 |
| Game days (g/kg BM) | 1.50 ± 0.1 | 0.0 ± 0.0 | 1.50 ± 0.1 | 1.49 ± 0.1 | 0.06 ± 0.0 | 1.55 ± 0.1 |
| Game days (% of total intake) | 29.6 ± 1.7 | 30.5 ± 1.5 | ||||
| Selenium (μg/day) | ||||||
| Adaptive period | 43.9 ± 9.6 | 0.0 ± 0.0 | 43.9 ± 9.6 | 44.5 ± 10.3 | 0.0 ± 0.0 | 44.5 ± 10.3 |
| Game days | 48.8 ± 12.4 | 0.0 ± 0.0 | 47.4 ± 12.4 | 46.2 ± 9.7 | 0.0 ± 0.0 | 46.2 ± 9.7 |
| Training days | 46.7 ± 11.3 | 0.0 ± 0.0 | 46.7 ± 11.3 | 45.9 ± 10.9 | 0.0 ± 0.0 | 45.9 ± 10.9 |
| Zinc (mg/day) | ||||||
| Adaptive period | 10.7 ± 2.8 | 0.0 ± 0.0 | 10.7 ± 2.8 | 10.9 ± 3.3 | 0.0 ± 0.0 | 10.9 ± 3.3 |
| Game days | 11.1 ± 3.2 | 0.0 ± 0.0 | 11.1 ± 3.2 | 11.8 ± 3.6 | 0.0 ± 0.0 | 11.8 ± 3.6 |
| Training days | 11.0 ± 2.9 | 0.0 ± 0.0 | 11.0 ± 2.9 | 11.5 ± 3.6 | 0.0 ± 0.0 | 11.5 ± 3.6 |
| Vitamin C (mg/day) | ||||||
| Adaptive period | 123.6 ± 9.4 | 0.0 ± 0.0 | 123.6 ± 9.4 | 121.9 ± 9.1 | 0.0 ± 0.0 | 121.9 ± 9.1 |
| Game days | 128.3 ± 11.8 | 0.0 ± 0.0 | 128.8 ± 11.8 | 124.7± 8.1 | 0.0 ± 0.0 | 124.7± 8.1 |
| Training days | 126.7 ± 11.2 | 0.0 ± 0.0 | 126.7 ± 11.2 | 125.5 ± 11.0 | 0.0 ± 0.0 | 125.5 ± 11.0 |
| Vitamin E (mg/day, α-TE) | ||||||
| Adaptive period | 8.7 ± 4.1 | 0.0 ± 0.0 | 8.7 ± 4.1 | 8.9 ± 4.2 | 0.0 ± 0.0 | 8.9 ± 4.2 |
| Game days | 9.1 ± 4.5 | 0.0 ± 0.0 | 9.1 ± 4.5 | 9.3 ± 4.7 | 0.0 ± 0.0 | 9.3 ± 4.7 |
| Training days | 8.8 ± 4.4 | 0.0 ± 0.0 | 8.8 ± 4.4 | 9.2 ± 4.3 | 0.0 ± 0.0 | 9.2 ± 4.3 |
| Game 1 | Game 2 | |
|---|---|---|
| Average HR (beats/min) | ||
| PLA | 166.9 ± 6.6 | 157.4 ± 8.51 1 |
| PRO | 167.2 ± 6.9 | 161.5 ± 8.81 |
| Total distance (m) | ||
| PLA | 10,048.1 ± 748.5 | 9448.8 ± 545.2 1 |
| PRO | 10,032.2 ± 768.6 | 9756.6 ± 553.8 |
| Distance at >14 km/h | ||
| PLA | 1890.5 ± 245.4 | 1683.8 ± 232,9 1 |
| PRO | 1909.0 ± 240.1 | 1800.3 ± 235.7 1 |
| Peak speed (km/h) | ||
| PLA | 27.8 ± 2.6 | 27.4 ± 1.7 |
| PRO | 27.1 ± 3.0 | 27.0 ± 2.4 |
| Accelerations (no) | ||
| PLA | 268.0 ± 18.9 | 262.9 ± 19.7 1 |
| PRO | 267.9 ± 20.5 | 265.0 ± 20.4 1 |
| Decelerations (no) | ||
| PLA | 266.8 ± 18.1 | 259.8 ± 19.2 1 |
| PRO | 265.3 ± 20.3 | 262.5 ± 20.0 1 |
| Game 1-D1 | Game 1-D2 | Game 2-D1 | Game 2-D2 | |
|---|---|---|---|---|
| Average HR (beats/min) | ||||
| PLA | 125.5 ± 8.2 1 | 132.8 ± 6.8 1 | 123.8 ± 5.9 1 | 154.2 ± 5.9 1 |
| PRO | 126.4 ± 10.2 1 | 131.9 ± 9.3 1 | 124.4 ± 3.9 1 | 161.0 ± 6.7 1 |
| Total distance (m) | ||||
| PLA | 2543.1 ± 776.3 1 | 4414.0 ± 485.8 1 | 2341.1 ± 392.9 1 | 5366.4 ± 367.4 1 |
| PRO | 2562.8 ± 883.6 1 | 4337.4 ± 446.9 1 | 2385.2 ± 342.8 1 | 5565.2 ± 383.3 1 |
| High-intensity running (m) | ||||
| PLA | 172.4 ± 26.3 1 | 947.7 ± 93.2 1 | 153.8 ± 26.4 1 | 1223.1 ± 379.8 1 |
| PRO | 170.8 ± 28.1 1 | 941.2 ± 116.5 1 | 155.8 ± 29.7 1 | 1343.8 ± 366.6 1 |
| Baseline | Game 1-D1 | Game 1-D2 | Game 1-D3 | Game 2-D1 | Game 2-D2 | Game 2-D3 | |
|---|---|---|---|---|---|---|---|
| 10-m sprint (s) | |||||||
| PLA | 1.81 ± 0.06 | 2.00 ± 0.07 1 | 1.93 ± 0.07 1 | 1.87 ± 0.08 1 | 2.09 ± 0.13 1 | 1.97 ± 0.06 1 | 1.88 ± 0.06 1 |
| PRO | 1.80 ± 0.06 | 2.01 ± 0.07 1 | 1.93 ± 0.06 1,2 | 1.86 ± 0.07 1 | 2.09 ± 0.08 1 | 1.98 ± 0.06 1,2 | 1.88 ± 0.08 1 |
| 30-m sprint (s) | |||||||
| PLA | 4.18 ± 0.18 | 4.70 ± 0.21 1 | 4.54 ± 0.18 1 | 4.38 ± 0.14 1 | 4.90 ± 0.28 1 | 4.62 ± 0.22 1 | 4.48 ± 0.2 1 |
| PRO | 4.19 ± 0.16 | 4.67 ± 0.19 1 | 4.53 ± 0.17 1 | 4.38 ± 0.13 1 | 4.89 ± 0.26 1 | 4.62 ± 0.19 1 | 4.45 ± 0.15 1 |
| CMJ (cm) | |||||||
| PLA | 48.6 ± 4.3 | 43.9 ± 3.9 1 | 45.5 ± 4.5 1 | 47.1 ± 4.3 1 | 43.6 ± 4.2 1 | 43.9 ± 3.4 1 | 45.1 ± 3.0 1 |
| PRO | 48.7 ± 4.4 | 44.2 ± 4.5 1 | 45.7 ± 3.4 1 | 47.2 ± 4.5 1 | 44.5 ± 3.5 1 | 44.6 ± 3.3 1 | 45.8 ± 3.1 1 |
| KE concentric strength/Dominant limb (Nm/kg) | |||||||
| PLA | 3.31 ± 0.7 | 3.05 ± 0.6 1 | 3.13 ± 0.6 1 | 3.20 ± 0.6 | 3.00 ± 0.6 1 | 3.02 ± 0.6 1 | 3.12 ± 0.7 1 |
| PRO | 3.33 ± 0.7 | 3.11 ± 0.7 1 | 3.15 ± 0.7 | 3.22 ± 0.7 | 3.05 ± 0.6 1 | 3.12 ± 0.7 1 | 3.17 ± 0.6 |
| KE concentric strength/Non-Dominant limb (Nm/kg) | |||||||
| PLA | 3.34 ± 0.7 | 3.10 ± 0.6 1 | 3.15 ± 0.7 1 | 3.22 ± 0.7 | 3.12 ± 0.6 1 | 3.14 ± 0.6 | 3.23 ± 0.7 1 |
| PRO | 3.33 ± 0.7 | 3.11 ± 0.6 1 | 3.16 ± 0.6 | 3.20 ± 0.7 | 3.12 ± 0.6 1 | 3.13 ± 0.7 1 | 3.21 ± 0.6 |
| KF eccentric strength/Dominant limb (Nm/kg) | |||||||
| PLA | 3.20 ± 0.5 | 2.66 ± 0.4 1 | 2.91 ± 0.4 1 | 2.92 ± 0.3 1 | 2.72 ± 0.3 1 | 2.73 ± 0.4 1 | 2.96 ± 0.4 1 |
| PRO | 3.17 ± 0.5 | 2.80 ± 0.3 1 | 2.82 ± 0.4 1 | 2.93 ± 0.3 1 | 2.75 ± 0.4 1 | 2.78 ± 0.4 1 | 3.02 ± 0.4 |
| KF eccentric strength/non-dominant limb (Nm/kg) | |||||||
| PLA | 3.23 ± 0.5 | 2.69 ± 0.5 1 | 2.90 ± 0.4 1 | 3.03 ± 0.5 1 | 2.49 ± 0.6 1 | 2.73 ± 0.4 1 | 2.98 ± 0.4 1 |
| PRO | 3.20 ± 0.4 | 2.73 ± 0.3 1 | 2.89 ± 0.4 1 | 3.04 ± 0.3 1 | 2.53 ± 0.5 1 | 2.73 ± 0.5 1 | 3.02 ± 0.3 |
| DOMS, KE/Dominant limb | |||||||
| PLA | 1.0 ± 0.0 | 4.3 ± 1.7 1 | 3.6 ± 1.5 1 | 3.3 ± 1.2 1 | 4.2 ± 1.3 1 | 3.1 ± 1.4 1 | 3.0 ± 1.3 1 |
| PRO | 1.0 ± 0.0 | 3.2 ± 1.6 1 | 3.3 ± 1.2 1 | 2.9 ± 1.2 1 | 3.7 ± 1.1 1 | 3.2 ± 1.6 1 | 2.9 ± 1.2 1 |
| DOMS, KE/non-dominant limb | |||||||
| PLA | 1.0 ± 0.0 | 4.5 ± 1.3 1 | 3.8 ± 1.8 1 | 3.7 ± 1.4 1 | 4.2 ± 1.6 1 | 3.4 ± 1.9 1 | 2.9 ± 1.3 1 |
| PRO | 1.0 ± 0.0 | 3.6 ± 2.0 1 | 3.7 ± 1.7 1 | 3.5 ± 2.0 1 | 4.5 ± 1.7 1 | 3.6 ± 1.9 1,5 | 3.0 ± 1.3 1 |
| DOMS, KF/Dominant limb | |||||||
| PLA | 1.0 ± 0.0 | 4.7 ± 1.5 1 | 4.3 ± 1.3 1 | 3.7 ± 1.2 1 | 4.6 ± 1.1 1 | 4.4 ± 1.3 1 | 3.7 ± 1.2 1 |
| PRO | 1.0 ± 0.0 | 3.8 ± 1.7 1 | 4.0 ± 1.4 1 | 3.4 ± 1.2 1 | 4.7 ± 1.1 1 | 4.5 ± 1.2 1 | 3.5 ± 1.2 1 |
| DOMS, KF/non-dominant limb | |||||||
| PLA | 1.0 ± 0.0 1 | 4.4 ± 1.4 1 | 4.7 ± 1.1 1 | 3.8 ± 1.1 1 | 4.8 ± 1.0 1 | 4.1 ± 1.2 1 | 4.0 ± 0.7 1 |
| PRO | 1.0 ± 0.0 | 3.7 ± 1.6 1 | 4.1 ± 1.2 1 | 3.7 ± 1.0 1 | 4.7 ± 1.1 1 | 4.1 ± 1.0 1 | 3.8 ± 0.7 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poulios, A.; Fatouros, I.G.; Mohr, M.; Draganidis, D.; Deli, C.K.; Papanikolaou, K.; Sovatzidis, A.; Nakopoulou, T.; Ermidis, G.; Tzatzakis, T.; et al. Post-Game High Protein Intake May Improve Recovery of Football-Specific Performance during a Congested Game Fixture: Results from the PRO-FOOTBALL Study. Nutrients 2018, 10, 494. https://doi.org/10.3390/nu10040494
Poulios A, Fatouros IG, Mohr M, Draganidis D, Deli CK, Papanikolaou K, Sovatzidis A, Nakopoulou T, Ermidis G, Tzatzakis T, et al. Post-Game High Protein Intake May Improve Recovery of Football-Specific Performance during a Congested Game Fixture: Results from the PRO-FOOTBALL Study. Nutrients. 2018; 10(4):494. https://doi.org/10.3390/nu10040494
Chicago/Turabian StylePoulios, Athanasios, Ioannis G. Fatouros, Magni Mohr, Dimitrios Draganidis, Chariklia K. Deli, Konstantinos Papanikolaou, Apostolos Sovatzidis, Theofano Nakopoulou, Georgios Ermidis, Theofanis Tzatzakis, and et al. 2018. "Post-Game High Protein Intake May Improve Recovery of Football-Specific Performance during a Congested Game Fixture: Results from the PRO-FOOTBALL Study" Nutrients 10, no. 4: 494. https://doi.org/10.3390/nu10040494
APA StylePoulios, A., Fatouros, I. G., Mohr, M., Draganidis, D., Deli, C. K., Papanikolaou, K., Sovatzidis, A., Nakopoulou, T., Ermidis, G., Tzatzakis, T., Laschou, V. C., Georgakouli, K., Koulouris, A., Tsimeas, P., Chatzinikolaou, A., Karagounis, L. G., Batsilas, D., Krustrup, P., & Jamurtas, A. Z. (2018). Post-Game High Protein Intake May Improve Recovery of Football-Specific Performance during a Congested Game Fixture: Results from the PRO-FOOTBALL Study. Nutrients, 10(4), 494. https://doi.org/10.3390/nu10040494








