Dietary Intake of Magnesium or Calcium and Chemotherapy-Induced Peripheral Neuropathy in Colorectal Cancer Patients
Abstract
1. Introduction
2. Materials and Methods
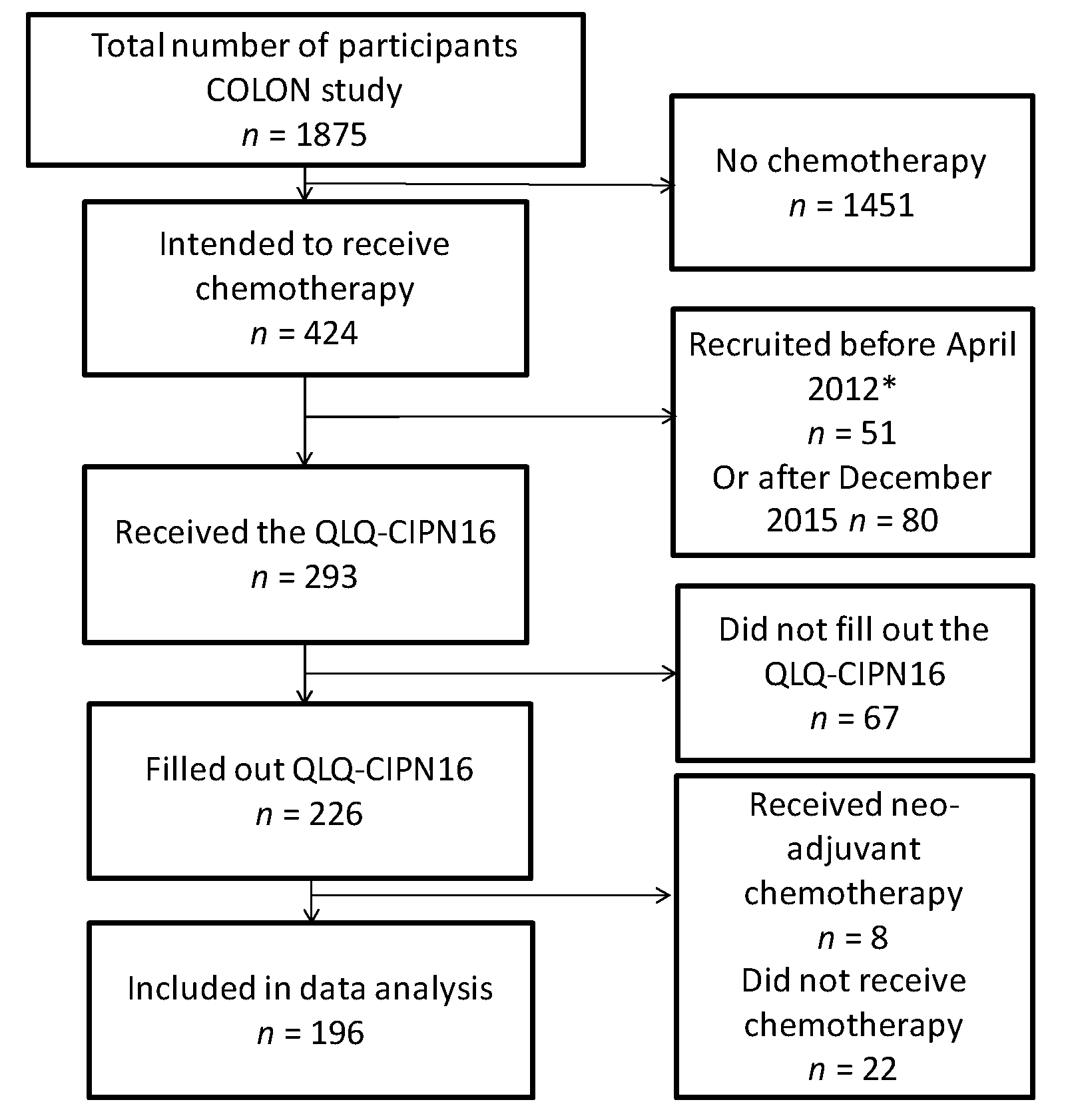
2.1. Patients
2.2. Magnesium and Calcium Intake
2.3. Chemotherapy-Induced Peripheral Neuropathy
2.4. Data Analysis
3. Results
3.1. Patients Characteristics
3.2. Dietary Magnesium or Calcium Intake and CIPN
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Argyriou, A.A.; Polychronopoulos, P.; Iconomou, G.; Chroni, E.; Kalofonos, H.P. A review on oxaliplatin-induced peripheral nerve damage. Cancer Treat. Rev. 2008, 34, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Staff, N.P.; Grisold, A.; Grisold, W.; Windebank, A.J. Chemotherapy-Induced Peripheral Neuropathy: A Current Review. Ann. Neurol. 2017, 81, 772–781. [Google Scholar] [CrossRef] [PubMed]
- Vincenzi, B.; Frezza, A.M.; Schiavon, G.; Spoto, C.; Silvestris, N.; Addeo, R.; Catalano, V.; Graziano, F.; Santini, D.; Tonini, G. Identification of clinical predictive factors of oxaliplatin-induced chronic peripheral neuropathy in colorectal cancer patients treated with adjuvant Folfox IV. Support. Care Cancer 2013, 21, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Attal, N.; Bouhassira, D.; Gautron, M.; Vaillant, J.N.; Mitry, E.; Lepère, C.; Rougier, P.; Guirimand, F. Thermal hyperalgesia as a marker of oxaliplatin neurotoxicity: A prospective quantified sensory assessment study. Pain 2009, 144, 245–252. [Google Scholar] [CrossRef] [PubMed]
- André, T.; de Gramont, A.; Vernerey, D.; Chibaudel, B.; Bonnetain, F.; Tijeras-Raballand, A.; Scriva, A.; Hickish, T.; Tabernero, J.; Van Laethem, J.L.; et al. Adjuvant Fluorouracil, Leucovorin, and Oxaliplatin in Stage II to III Colon Cancer: Updated 10-Year Survival and Outcomes According to BRAF Mutation and Mismatch Repair Status of the MOSAIC Study. J. Clin. Oncol. 2015, 33, 4176–4187. [Google Scholar] [CrossRef] [PubMed]
- De Gramont, A.; Figer, A.; Seymour, M.; Homerin, M.; Hmissi, A.; Cassidy, J.; Boni, C.; Cortes-Funes, H.; Cervantes, A.; Freyer, G.; et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J. Clin. Oncol. 2000, 18, 2938–2947. [Google Scholar] [CrossRef] [PubMed]
- Extra, J.M.; Marty, M.; Brienza, S.; Misset, J.L. Pharmacokinetics and safety profile of oxaliplatin. Semin. Oncol. 1998, 25, 13–22. [Google Scholar] [PubMed]
- Laires, M.J.; Monteiro, C.P.; Bicho, M. Role of cellular magnesium in health and human disease. Front. Biosci. 2004, 9, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Gamelin, L.; Boisdron-Celle, M.; Delva, R.; Guérin-Meyer, V.; Ifrah, N.; Morel, A.; Gamelin, E. Prevention of oxaliplatin-related neurotoxicity by calcium and magnesium infusions: A retrospective study of 161 patients receiving oxaliplatin combined with 5-Fluorouracil and leucovorin for advanced colorectal cancer. Clin. Cancer Res. 2004, 10, 4055–4061. [Google Scholar] [CrossRef] [PubMed]
- Knijn, N.; Tol, J.; Koopman, M.; Werter, M.J.B.P.; Imholz, A.L.T.; Valster, F.A.A.; Mol, L.; Vincent, A.D.; Teerenstra, S.; Punt, C.J.A. The effect of prophylactic calcium and magnesium infusions on the incidence of neurotoxicity and clinical outcome of oxaliplatin-based systemic treatment in advanced colorectal cancer patients. Eur. J. Cancer 2011, 47, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Kono, T.; Mamiya, N.; Chisato, N.; Ebisawa, Y.; Yamazaki, H.; Watari, J.; Yamamoto, Y.; Suzuki, S.; Asama, T.; Kamiya, K. Efficacy of Goshajinkigan for Peripheral Neurotoxicity of Oxaliplatin in Patients with Advanced or Recurrent Colorectal Cancer. Evid.-Based Complement. Altern. Med. 2011, 2011, 418481. [Google Scholar] [CrossRef] [PubMed]
- Grothey, A.; Nikcevich, D.A.; Sloan, J.A.; Kugler, J.W.; Silberstein, P.T.; Dentchev, T.; Wender, D.B.; Novotny, P.J.; Chitaley, U.; Alberts, S.R.; et al. Intravenous calcium and magnesium for oxaliplatin-induced sensory neurotoxicity in adjuvant colon cancer: NCCTG N04C7. J. Clin. Oncol. 2011, 29, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Hochster, H.S.; Grothey, A.; Hart, L.; Rowland, K.; Ansari, R.; Alberts, S.; Chowhan, N.; Ramanathan, R.K.; Keaton, M.; Hainsworth, J.D.; et al. Improved time to treatment failure with an intermittent oxaliplatin strategy: Results of CONcePT. Ann. Oncol. 2014, 25, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, C.L.; Qin, R.; Dakhil, S.R.; Fehrenbacher, L.; Flynn, K.A.; Atherton, P.; Seisler, D.; Qamar, R.; Lewis, G.C.; Grothey, A. Phase III randomized, placebo-controlled, double-blind study of intravenous calcium and magnesium to prevent oxaliplatin-induced sensory neurotoxicity (N08CB/Alliance). J. Clin. Oncol. 2014, 32, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, K.; Okada, N.; Miyazaki, T.; Sano, M.; Ishida, H. Effect of calcium and magnesium on neurotoxicity and blood platinum concentrations in patients receiving mFOLFOX6 therapy: A prospective randomized study. Int. J. Clin. Oncol. 2010, 15, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Chay, W.-Y.; Tan, S.-H.; Lo, Y.-L.; Ong, S.Y.-K.; Ng, H.-C.; Gao, F.; Koo, W.-H.; Choo, S.-P. Use of calcium and magnesium infusions in prevention of oxaliplatin induced sensory neuropathy. Asia Pac. J. Clin. Oncol. 2010, 6, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Albers, J.W.; Chaudhry, V.; Cavaletti, G.; Donehower, R.C. Interventions for preventing neuropathy caused by cisplatin and related compounds. In Cochrane Database of Systematic Reviews; Albers, J.W., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2014; p. CD005228. [Google Scholar]
- Winkels, R.M.; Heine-Bröring, R.C.; van Zutphen, M.; van Harten-Gerritsen, S.; Kok, D.E.G.; van Duijnhoven, F.J.B.; Kampman, E. The COLON study: Colorectal cancer: Longitudinal, Observational study on Nutritional and lifestyle factors that may influence colorectal tumour recurrence, survival and quality of life. BMC Cancer 2014, 14, 374. [Google Scholar] [CrossRef] [PubMed]
- Van Leersum, N.J.; Snijders, H.S.; Henneman, D.; Kolfschoten, N.E.; Gooiker, G.A.; ten Berge, M.G.; Eddes, E.H.; Wouters, M.W.J. M.; Tollenaar, R.A.E.M.; Bemelman, W.A.; et al. The Dutch Surgical Colorectal Audit. Eur. J. Surg. Oncol. 2013, 39, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Netherlands Nutrition Center: NEVO Nederlandse Voedingsmiddelen Tabel 2011 (In Englisch: Dutch Food Composition Table). Available online: http://nevo-online.rivm.nl/ (accessed on 10 May 2016).
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1228S. [Google Scholar] [CrossRef] [PubMed]
- Lavoie Smith, E.M.; Barton, D.L.; Qin, R.; Steen, P.D.; Aaronson, N.K.; Loprinzi, C.L. Assessing patient-reported peripheral neuropathy: The reliability and validity of the European Organization for Research and Treatment of Cancer QLQ-CIPN20 Questionnaire. Qual. Life Res. 2013, 22, 2787–2799. [Google Scholar] [CrossRef] [PubMed]
- Mols, F.; van de Poll-Franse, L.V.; Vreugdenhil, G.; Beijers, A.J.; Kieffer, J.M.; Aaronson, N.K.; Husson, O. Reference data of the European Organisation for Research and Treatment of Cancer (EORTC) QLQ-CIPN20 Questionnaire in the general Dutch population. Eur. J. Cancer 2016, 69, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Roth, P.L. Missing Data in Multiple Item Scales: A Monte Carlo Analysis of Missing Data Techniques. Organ. Res. Methods 1999, 2, 211–232. [Google Scholar] [CrossRef]
- Tamhane, A.R.; Westfall, A.O.; Burkholder, G.A.; Cutter, G.R. Prevalence odds ratio versus prevalence ratio: Choice comes with consequences. Stat. Med. 2016, 35, 5730–5735. [Google Scholar] [CrossRef] [PubMed]
- Bewick, V.; Cheek, L.; Ball, J. Statistics review 8: Qualitative data—Tests of association. Crit. Care 2004, 8, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US). Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Chapter 6 Magnesium; In Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press US: Washington, DC, USA, 1997; pp. 71–145. ISBN 978-0-309-06403-3. [Google Scholar]
- Institute of Medicine (US). Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Chapter 4 Calcium; In Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press US: Washington, DC, USA, 1997; pp. 190–249. ISBN 978-0-309-06403-3. [Google Scholar]
- Alberti, P.; Rossi, E.; Cornblath, D.R.; Merkies, I.S.J.; Postma, T.J.; Frigeni, B.; Bruna, J.; Velasco, R.; Argyriou, A.A.; Kalofonos, H.P.; et al. Physician-assessed and patient-reported outcome measures in chemotherapy-induced sensory peripheral neurotoxicity: Two sides of the same coin. Ann. Oncol. 2014, 25, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Le-Rademacher, J.; Kanwar, R.; Seisler, D.; Pachman, D.R.; Qin, R.; Abyzov, A.; Ruddy, K.J.; Banck, M.S.; Lavoie Smith, E.M.; Dorsey, S.G.; et al. Patient-reported (EORTC QLQ-CIPN20) versus physician-reported (CTCAE) quantification of oxaliplatin- and paclitaxel/carboplatin-induced peripheral neuropathy in NCCTG/Alliance clinical trials. Support. Care Cancer 2017, 25, 3537–3544. [Google Scholar] [CrossRef] [PubMed]
- Cavaletti, G.; Cornblath, D.R.; Merkies, I.S.J.; Postma, T.J.; Rossi, E.; Frigeni, B.; Alberti, P.; Bruna, J.; Velasco, R.; Argyriou, A.A.; et al. The chemotherapy-induced peripheral neuropathy outcome measures standardization study: From consensus to the first validity and reliability findings. Ann. Oncol. 2013, 24, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Hoenderop, J.G.J.; Bindels, R.J.M. Epithelial Ca2+ and Mg2+ Channels in Health and Disease. J. Am. Soc. Nephrol. 2004, 16, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Gamelin, E.; Gamelin, L.; Bossi, L.; Quasthoff, S. Clinical aspects and molecular basis of oxaliplatin neurotoxicity: Current management and development of preventive measures. Semin. Oncol. 2002, 29, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Bara, M.; Guiet-Bara, A.; Durlach, J. Regulation of sodium and potassium pathways by magnesium in cell membranes. Magnes. Res. 1993, 6, 167–177. [Google Scholar] [PubMed]
- Miltenburg, N.C.; Boogerd, W. Chemotherapy-induced neuropathy: A comprehensive survey. Cancer Treat. Rev. 2014, 40, 872–882. [Google Scholar] [CrossRef] [PubMed]
- De Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J.M. Regulation of magnesium balance: Lessons learned from human genetic disease. Clin. Kidney J. 2012, 5, i15–i24. [Google Scholar] [CrossRef] [PubMed]
- Elin, R.J. Magnesium: The Fifth but Forgotten Electrolyte. Am. J. Clin. Pathol. 1994, 102, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Leurs, J.L.; Schouten, L.J.; Mons, M.N.; Goldbohm, R.A.; van den Brandt, P. Relationship between Tap Water Hardness, Magnesium, and Calcium Concentration and Mortality Due to Ischemic Heart Disease or Stroke in the Netherlands. Environ. Health Perspect. 2010, 118, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.; Barton, D.; Kottschade, L.; Grothey, A.; Loprinzi, C. Chemotherapy-induced peripheral neuropathy: Prevention and treatment strategies. Eur. J. Cancer 2008, 44, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
| CIPN | |||
|---|---|---|---|
| Total Population n = 196 | CIPN No n = 36 (18%) | Yes 1 n = 160 (82%) | |
| Gender, women | 71 (36%) | 12 (33%) | 59 (37%) |
| Age (years) | 64.0 (59.8–68.1) | 65.4 (59.0–68.2) | 63.7 (59.9–68.0) |
| Diabetes mellitus (yes) 2 | 15 (8%) | 3 (8%) | 12 (8%) |
| Physical activity (meeting norm) 3 | 143 (73%) | 25 (69%) | 118 (74%) |
| Tumor stage | |||
| Stage II | 15 (8%) | 2 (6%) | 13 (8%) |
| Stage III | 145 (74%) | 28 (76%) | 117 (73%) |
| Stage IV | 21 (10%) | 3 (9%) | 18 (11%) |
| Missing | 15 (8%) | 3 (9%) | 12 (8%) |
| Cancer site | |||
| Colon | 181 (93%) | 33 (92%) | 148 (93%) |
| Rectum | 14 (7%) | 3 (8%) | 11 (7%) |
| Missing | 1 (0%) | 0 (0%) | 1 (0%) |
| Type of chemotherapy | |||
| Oxaliplatin-containing (OX) | 166 (85%) | 27 (75%) | 139 (87%) |
| Capecitabine monotherapy | 23 (12%) | 9 (25%) | 14 (9%) |
| Other | 2 (1%) | 0 (0%) | 2 (1%) |
| Missing | 5 (2%) | 0 (0%) | 5 (3%) |
| Dietary factors | |||
| Magnesium intake from diet (mg/day) 2,4 | 317 (261–383) | 325 (261–396) | 313 (261–380) |
| Use of magnesium supplements (yes) | 36 (18%) | 6 (17%) | 30 (18%) |
| Calcium intake from diet (mg/day) 2,4 | 851 (621–1143) | 789 (597–1155) | 861 (631–1123) |
| Use of calcium supplements (yes) | 35 (18%) | 6 (17%) | 29 (18%) |
| Vitamin D intake from diet (mg/day) 2,4 | 3.1 (2.4–4.1) | 2.8 (2.2–3.7) | 3.2 (2.4–4.2) |
| Total energy intake (kcal/day) 2,4 | 1893 (1534–2265) | 1898 (1547–2230) | 1893 (1534–2285) |
| CIPN 5 | |||
| Total score 6 | 14.6 (6.3–26.4) | 0.0 (0–2.1) | 16.7 (10.4–29.2) |
| Sensory score 7 | 16.7 (4.2–37.5) | 0.0 (0–0) | 20.8 (12.5–37.5) |
| Motor score 8 | 8.3 (4.2–20.8) | 0.0 (0–0) | 12.5 (8.3–20.8) |
| n/Events | Prevalence Ratio (95% CI) Total Score CIPN16 1 | n/Events | Sensory Symptoms 1 | n/Events | Motor Symptoms 1 | |
|---|---|---|---|---|---|---|
| Magnesium intake | ||||||
| Crude model | ||||||
| At diagnosis | 194/158 | 0.81 (0.51, 1.29) | 194/157 | 0.98 (0.64, 1.52) | 194/147 | 0.95 (0.54, 1.67) |
| During chemotherapy | 192/156 | 0.57 (0.35, 0.92) | 192/156 | 0.70 (0.44, 1.12) | 192/145 | 0.59 (0.33, 1.05) |
| After chemotherapy | 181/147 | 0.67 (0.43, 1.05) | 181/146 | 0.86 (0.57, 1.31) | 181/136 | 0.61 (0.36, 1.03) |
| Full model | ||||||
| At diagnosis | 194/158 | 0.69 (0.42, 1.13) | 194/157 | 0.84 (0.51, 1.39) | 194/147 | 0.81 (0.43, 1.52) |
| During chemotherapy | 192/156 | 0.53 (0.32, 0.90) | 192/156 | 0.67 (0.40, 1.10) | 192/145 | 0.57 (0.30, 1.09) |
| After chemotherapy | 181/147 | 0.61 (0.37, 1.03) | 181/146 | 0.84 (0.51, 1.37) | 181/136 | 0.51 (0.28, 0.93) |
| Calcium intake | ||||||
| Crude model | ||||||
| At diagnosis | 194/158 | 1.15 (0.93, 1.41) | 194/157 | 1.20 (0.98, 1.46) | 194/147 | 1.12 (0.89, 1.41) |
| During chemotherapy | 192/156 | 0.97 (0.83, 1.13) | 192/156 | 1.02 (0.87, 1.19) | 192/145 | 0.89 (0.73, 1.09) |
| After Chemotherapy | 181/147 | 1.03 (0.86, 1.23) | 181/146 | 1.08 (0.90, 1.30) | 181/136 | 0.96 (0.78, 1.17) |
| Full model | ||||||
| At diagnosis | 194/158 | 1.21 (0.95, 1.56) | 194/157 | 1.24 (0.97, 1.58) | 194/147 | 1.15 (0.88, 1.50) |
| During chemotherapy | 192/156 | 1.03 (0.86, 1.23) | 192/156 | 1.06 (0.89, 1.28) | 192/145 | 0.92 (0.73, 1.16) |
| After Chemotherapy | 181/147 | 1.08 (0.89, 1.32) | 181/146 | 1.10 (0.89, 1.35) | 181/136 | 0.99 (0.79, 1.24) |
| Beta (95% CI) | ||||||
|---|---|---|---|---|---|---|
| n | Total Score CIPN16 | n | Sensory Symptoms | n | Motor Symptoms | |
| Magnesium intake | ||||||
| Crude model | ||||||
| At diagnosis | 158 | −0.68 (−1.45, 0.08) | 157 | −1.09 (−1,90 −0.27) | 147 | −0.45 (−1.33, 0.43) |
| During chemotherapy | 156 | −1.21 (−2.01, −0.41) | 156 | −1.43 (−2.30, −0.57) | 145 | −1.25 (−2.16, −0.35) |
| After chemotherapy | 147 | −0.81 (−1.59, −0.04) | 146 | −1.05 (−1.87, −0.22) | 136 | −0.64 (−1.48, 0.21) |
| Full model | ||||||
| At diagnosis | 158 | −0.65 (−1,52, 0.20) | 157 | −1.10 (−2.01, −0.18) | 147 | −0.36 (−1.36, 0.63) |
| During chemotherapy | 156 | −1.08 (−1.95, −0.22) | 156 | −1.24 (−2.17, −0.32) | 145 | −1.25 (−2.24, −0.27) |
| After chemotherapy | 147 | −0.93 (−1.81, −0.06) | 146 | −1.11 (−2.04, −0.19) | 136 | −0.76 (−1.73, 0.21) |
| Calcium intake | ||||||
| Crude model | ||||||
| At diagnosis | 158 | −0.20 (−0.55, 0.14) | 157 | −0.31 (−0.68, 0.06) | 147 | −0.19 (−0.56, 0.18) |
| During chemotherapy | 156 | −0.31 (−0.62, 0.00) | 156 | −0.39 (−0.74, −0.04) | 145 | −0.31 (−0.66, 0.04) |
| After chemotherapy | 147 | −0.19 (−0.55, 0.17) | 146 | −0.36 (−0.75, 0.03) | 136 | −0.04 (−0.42, 0.35) |
| Full model | ||||||
| At diagnosis | 158 | −0.05 (−0.45, 0.35) | 157 | 0.03 (−0.40, 0.45) | 147 | −0.19 (−0.61, 0.23) |
| During chemotherapy | 156 | −0.16 (−0.51, 0.19) | 156 | −0.14 (−0.52, 0.23) | 145 | −0.19 (−0.57, 0.19) |
| After chemotherapy | 147 | −0.03 (−0.42, 0.35) | 146 | −0.10 (−0.52, 0.32) | 136 | 0.01 (−0.40, 0.43) |
| Beta (95% CI) | ||||||
|---|---|---|---|---|---|---|
| n | Total Score CIPN16 | n | Sensory Symptoms | n | Motor Symptoms | |
| Patients receiving oxaliplatin-containing chemotherapy | ||||||
| Magnesium | ||||||
| At diagnosis | 137 | −0.68 (−1.58, 0.22) | 138 | −1.01 (−1.97, −0.05) | 126 | −0.48 (−0.53, 0.57) |
| During chemotherapy | 136 | −0.90 (−1.80, −0.00) | 137 | −1.10 (−2.06, −0.14) | 125 | −1.28 (−2.33, −0.24) |
| After chemotherapy | 132 | −0.94 (−1.85, −0.02) | 133 | −1.10 (−2.07, −0.12) | 121 | −0.94 (−1.98, 0.10) |
| Calcium | ||||||
| At diagnosis | 137 | 0.00 (−0.42, 0.42) | 138 | 0.08 (−0.36, 0.53) | 132 | −0.19 (−0.63, 0.26) |
| During chemotherapy | 136 | −0.18 (−0.54; 0.18) | 137 | −0.11 (−0.51; 0.28) | 125 | −0.28 (−0.69; 0.13) |
| After chemotherapy | 132 | 0.00 (−0.39, 0.41) | 133 | −0.06 (−0.49, 0.38) | 121 | 0.01 (−0.43, 0.45) |
| Not supplement users | ||||||
| Magnesium | ||||||
| At diagnosis | 128 | −0.80 (−1.80, 0.20) | 126 | −0.99 (−2.08, 0.10) | 119 | −0.68 (−1.83, 0.48) |
| During chemotherapy | 136 | −1.24 (−2.21, −0.27) | 134 | −1.20 (−2.27, −0.13) | 128 | −1.81 (−2.90, −0.71) |
| After chemotherapy | 129 | −1.34 (−2.30, −0.37) | 126 | −1.29 (−2.37, −0.21) | 119 | −1.38 (−2.44, −0.32) |
| Calcium | ||||||
| At diagnosis | 127 | −0.06 (−0.50, 0.37) | 127 | 0.06 (−0.41, 0.53) | 119 | −0.08 (−0.56, 0.38) |
| During chemotherapy | 140 | −0.12 (−0.48, 0.23) | 138 | −0.07 (−0.45, 0.32) | 132 | −0.20 (−0.60, 0.20) |
| After chemotherapy | 127 | −0.00 (−0.41, 0.42) | 130 | −0.09 (−0.54, 0.36) | 119 | 0.09 (−0.35, 0.53) |
| Colon cancer patients | ||||||
| Magnesium | ||||||
| At diagnosis | 146 | −0.66 (−1.54, 0.22) | 145 | −1.11 (−2.04, −0.18) | 135 | −0.34 (−1.37, 0.68) |
| During chemotherapy | 145 | −1.13 (−2.03, −0.23) | 145 | −1.34 (−2.30, −0.38) | 134 | −1.30 (−2.33, −0.26) |
| After chemotherapy | 139 | −0.80 (−1.71, 0.11) | 137 | −0.98 (−1.95, −0.02) | 128 | −0.66 (−1.67, 0.36) |
| Calcium | ||||||
| At diagnosis | 146 | −0.00 (−0.42, 0.41) | 145 | 0.07 (−0.37, 0.51) | 135 | −0.15 (−0.58, 0.28) |
| During chemotherapy | 145 | −0.11 (−0.48, 0.25) | 145 | −0.07 (−0.47, 0.35) | 134 | −0.16 (−0.57, 0.26) |
| After chemotherapy | 139 | −0.03 (−0.45, 0.39) | 137 | −0.11 (−0.56, 0.34) | 128 | 0.04 (−0.41, 0.50) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wesselink, E.; Winkels, R.M.; Van Baar, H.; Geijsen, A.J.M.R.; Van Zutphen, M.; Van Halteren, H.K.; Hansson, B.M.E.; Radema, S.A.; De Wilt, J.H.W.; Kampman, E.; et al. Dietary Intake of Magnesium or Calcium and Chemotherapy-Induced Peripheral Neuropathy in Colorectal Cancer Patients. Nutrients 2018, 10, 398. https://doi.org/10.3390/nu10040398
Wesselink E, Winkels RM, Van Baar H, Geijsen AJMR, Van Zutphen M, Van Halteren HK, Hansson BME, Radema SA, De Wilt JHW, Kampman E, et al. Dietary Intake of Magnesium or Calcium and Chemotherapy-Induced Peripheral Neuropathy in Colorectal Cancer Patients. Nutrients. 2018; 10(4):398. https://doi.org/10.3390/nu10040398
Chicago/Turabian StyleWesselink, Evertine, Renate M. Winkels, Harm Van Baar, Anne J. M. R. Geijsen, Moniek Van Zutphen, Henk K. Van Halteren, Bibi M. E. Hansson, Sandra A. Radema, Johannes H. W. De Wilt, Ellen Kampman, and et al. 2018. "Dietary Intake of Magnesium or Calcium and Chemotherapy-Induced Peripheral Neuropathy in Colorectal Cancer Patients" Nutrients 10, no. 4: 398. https://doi.org/10.3390/nu10040398
APA StyleWesselink, E., Winkels, R. M., Van Baar, H., Geijsen, A. J. M. R., Van Zutphen, M., Van Halteren, H. K., Hansson, B. M. E., Radema, S. A., De Wilt, J. H. W., Kampman, E., & Kok, D. E. G. (2018). Dietary Intake of Magnesium or Calcium and Chemotherapy-Induced Peripheral Neuropathy in Colorectal Cancer Patients. Nutrients, 10(4), 398. https://doi.org/10.3390/nu10040398






