Diet Quality and Sarcopenia in Older Adults: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search and Eligibility Criteria
2.1.1. Inclusion and Exclusion Criteria
2.1.2. Search Strategy
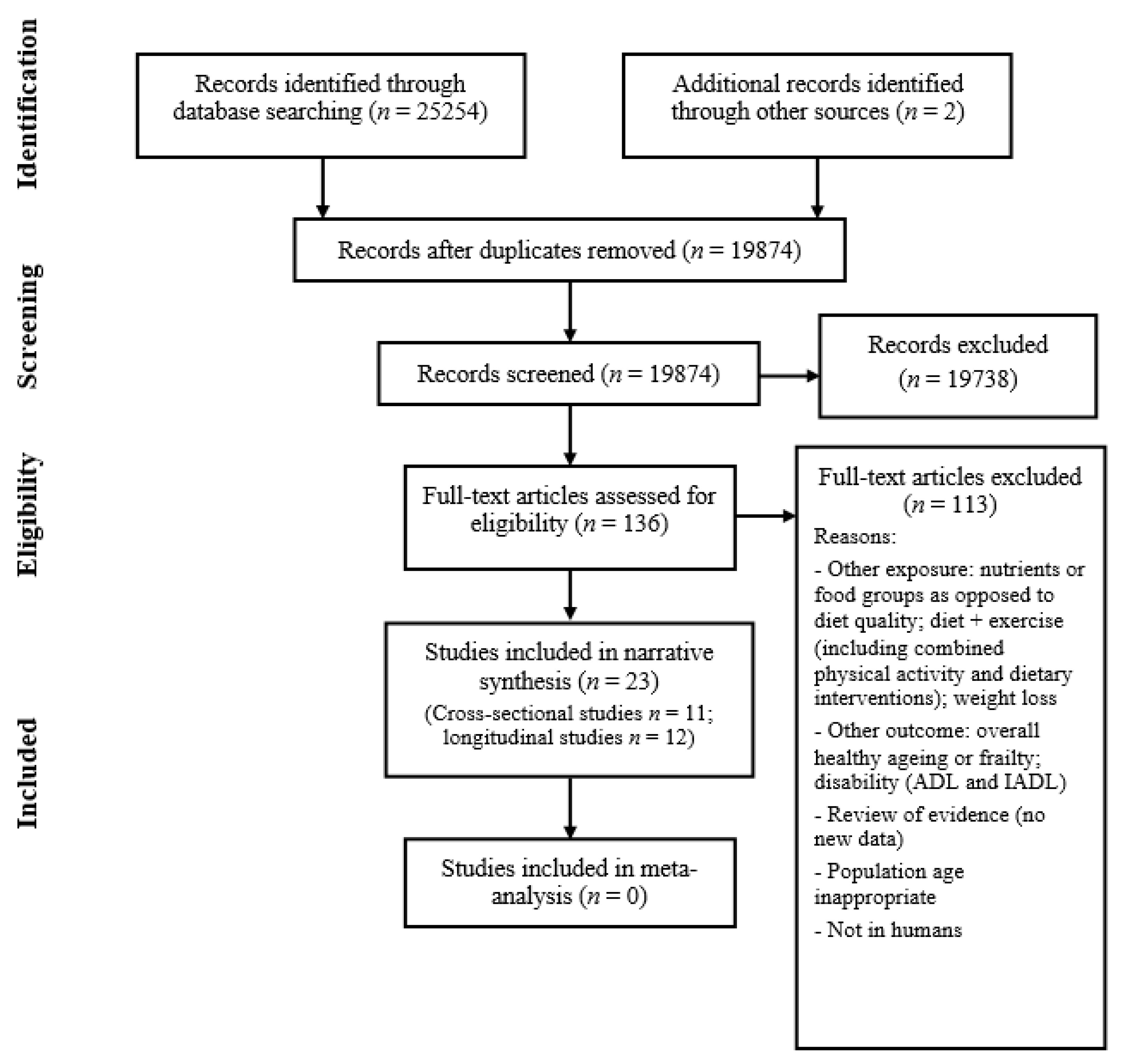
2.2. Study Selection
2.3. Data Extraction and Assessment of Risk of Bias
2.4. Data Synthesis
3. Results
3.1. Muscle Mass
3.2. Muscle Strength
3.3. Physical Performance
3.4. Sarcopenia
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shaw, S.C.; Dennison, E.M.; Cooper, C. Epidemiology of Sarcopenia: Determinants Throughout the Lifecourse. Calcif. Tissue Int. 2017, 101, 229–247. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Sayer, A.A. Sarcopenia the new geriatric giant: Time to translate research findings into clinical practice. Age Ageing 2014, 43, 736–737. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.S.; Guerra, R.S.; Fonseca, I.; Pichel, F.; Ferreira, S.; Amaral, T.F. Financial impact of sarcopenia on hospitalization costs. Eur. J. Clin. Nutr. 2016, 70, 1046–1051. [Google Scholar] [CrossRef] [PubMed]
- Dodds, R.M.; Syddall, H.E.; Cooper, R.; Benzeval, M.; Deary, I.J.; Dennison, E.M.; Der, G.; Gale, C.R.; Inskip, H.M.; Jagger, C.; et al. Grip Strength across the Life Course: Normative Data from Twelve British Studies. PLoS ONE 2014, 9, e113637. [Google Scholar] [CrossRef] [PubMed]
- Maynard, M.; Gunnell, D.; Ness, A.R.; Abraham, L.; Bates, C.J.; Blane, D. What influences diet in early old age? Prospective and cross-sectional analyses of the Boyd Orr cohort. Eur. J. Public Health 2006, 16, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Russell, C.A.; Stratton, R.J. Malnutrition in the UK: Policies to address the problem. Proc. Nutr. Soc. 2010, 69, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.E.; Donkin, A.J.; Morgan, K.; Neale, R.J.; Page, R.M.; Silburn, R.L. Fruit and vegetable consumption in later life. Age Ageing 1998, 27, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Margetts, B.M.; Thompson, R.L.; Elia, M.; Jackson, A.A. Prevalence of risk of undernutrition is associated with poor health status in older people in the UK. Eur. J. Clin. Nutr. 2003, 57, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.; Cooper, C.; Aihie Sayer, A. Nutrition and sarcopenia: A review of the evidence and implications for preventive strategies. J. Aging Res. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Waijers, P.M.; Feskens, E.J.; Ocké, M.C. A critical review of predefined diet quality scores. Br. J. Nutr. 2007, 97, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Scapagnini, G.; Willcox, B.J. Healthy aging diets other than the Mediterranean: A focus on the Okinawan diet. Mech. Ageing Dev. 2014, 136–137, 148–162. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Bogensberger, B.; Hoffmann, G. Diet Quality as Assessed by the Healthy Eating Index, Alternate Healthy Eating Index, Dietary Approaches to Stop Hypertension Score, and Health Outcomes: An Updated Systematic Review and Meta-Analysis of Cohort Studies. J. Acad. Nutr. Diet. 2018, 118, 74–100. [Google Scholar] [CrossRef] [PubMed]
- Schwedhelm, C.; Boeing, H.; Hoffmann, G.; Aleksandrova, K.; Schwingshackl, L. Effect of diet on mortality and cancer recurrence among cancer survivors: A systematic review and meta-analysis of cohort studies. Nutr. Rev. 2016, 74, 737–748. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.A.; Bates, C.J.; Mishra, G.D. Diet quality is associated with all-cause mortality in adults aged 65 years and older. J. Nutr. 2012, 142, 320–325. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.A.; Dunstan, D.W.; Ball, K.; Shaw, J.; Crawford, D. Dietary Quality Is Associated with Diabetes and Cardio-Metabolic Risk Factors. J. Nutr. 2009, 139, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.M.; Reginster, J.Y.; Rizzoli, R.; Shaw, S.C.; Kanis, J.A.; Bautmans, I.; Bischoff-Ferrari, H.; Bruyère, O.; Cesari, M.; Dawson-Hughes, B.; et al. Does nutrition play a role in the prevention and management of sarcopenia? Clin. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; McNaughton, S.A. Dietary patterns and successful ageing: A systematic review. Eur. J. Nutr. 2016, 55, 4234–4250. [Google Scholar] [CrossRef] [PubMed]
- Craig, J.V.; Bunn, D.K.; Hayhoe, R.P.; Appleyard, W.O.; Lenaghan, E.A.; Welch, A.A. Relationship between the Mediterranean dietary pattern and musculoskeletal health in children, adolescents, and adults: Systematic review and evidence map. Nutr. Rev. 2017, 75, 830–857. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.; Pizato, N.; Da Mata, F.; Figueiredo, A.; Ito, M.; Pereira, M.G. Mediterranean diet and musculoskeletal-functional outcomes in community-dwelling older people: A systematic review and meta-analysis. J. Nutr. Health Aging 2017. [Google Scholar] [CrossRef]
- Centre for Reviews and Dissemination, Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care. 2009. Available online: https://www.york.ac.uk/media/crd/Systematic_Reviews.pdf (accessed on 12 September 2017).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, M.; Berkman, N.D. Development of the RTI item bank on risk of bias and precision of observational studies. J. Clin. Epidemiol. 2012, 65, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, M.A.; Tucker, K.L.; Ryan, N.D.; O’Neill, E.F.; Clements, K.M.; Nelson, M.E.; Evans, W.J.; Singh, M.A.F. Higher dietary variety is associated with better nutritional status in frail elderly people. J. Am. Diet. Assoc. 2002, 102, 1096–1104. [Google Scholar] [CrossRef]
- Rahi, B.; Morais, J.A.; Dionne, I.J.; Gaudreau, P.; Payette, H.; Shatenstein, B. The combined effects of diet quality and physical activity on maintenance of muscle strength among diabetic older adults from the NuAge cohort. Exp. Gerontol. 2014, 49, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Milaneschi, Y.; Bandinelli, S.; Corsi, A.M.; Lauretani, F.; Paolisso, G.; Dominguez, L.J.; Semba, R.D.; Tanaka, T.; Abbatecola, A.M.; Talegawkar, S.A.; et al. Mediterranean diet and mobility decline in older persons. Exp. Gerontol. 2011, 46, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Talegawkar, S.A.; Bandinelli, S.; Bandeen-Roche, K.; Chen, P.; Milaneschi, Y.; Tanaka, T.; Semba, R.D.; Guralnik, J.M.; Ferrucci, L. A higher adherence to a Mediterranean-style diet is inversely associated with the development of frailty in community-dwelling elderly men and women. J. Nutr. 2012, 142, 2161–2166. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, Y.; Nishi, M.; Murayama, H.; Amano, H.; Taniguchi, Y.; Nofuji, Y.; Narita, M.; Matsuo, E.; Seino, S.; Kawano, Y.; et al. Dietary variety and decline in lean mass and physical performance in community-dwelling older Japanese: A 4-year follow-up study. J. Nutr. Health Aging 2017, 21, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Smee, D.; Pumpa, K.; Falchi, M.; Lithander, F.E. The Relationship between Diet Quality and Falls Risk, Physical Function and Body Composition in Older Adults. J. Nutr. Health Aging 2015, 19, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Chan, R.; Leung, J.; Woo, J. A Prospective Cohort Study to Examine the Association Between Dietary Patterns and Sarcopenia in Chinese Community-Dwelling Older People in Hong Kong. J. Am. Med. Dir. Assoc. 2016, 17, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.; No, J.K.; Kim, H.S. Dietary pattern classifications with nutrient intake and body composition changes in Korean elderly. Nutr. Res. Pract. 2014, 8, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Kojima, N.; Kim, M.; Saito, K.; Yoshida, H.; Yoshida, Y.; Hirano, H.; Obuchi, S.; Shimada, H.; Suzuki, T.; Kim, H. Lifestyle-Related Factors Contributing to Decline in Knee Extension Strength among Elderly Women: A Cross-Sectional and Longitudinal Cohort Study. PLoS ONE 2015, 10, e0132523. [Google Scholar] [CrossRef] [PubMed]
- Shahar, D.R.; Houston, D.K.; Hue, T.F.; Lee, J.S.; Sahyoun, N.R.; Tylavsky, F.A.; Geva, D.; Vardi, H.; Harris, T.B. Adherence to mediterranean diet and decline in walking speed over 8 years in community-dwelling older adults. J. Am. Geriatr. Soc. 2012, 60, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Leon-Munoz, L.M.; Guallar-Castillon, P.; Lopez-Garcia, E.; Rodriguez-Artalejo, F. Mediterranean diet and risk of frailty in community-dwelling older adults. J. Am. Med. Dir. Assoc. 2014, 15, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, R.; Motlagh, A.D.; Heshmat, R.; Esmaillzadeh, A.; Payab, M.; Yousefinia, M.; Siassi, F.; Pasalar, P.; Baygi, F. Diet and its relationship to sarcopenia in community dwelling Iranian elderly: A cross sectional study. Nutrition 2015, 31, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Nikolov, J.; Spira, D.; Aleksandrova, K.; Otten, L.; Meyer, A.; Demuth, I.; Steinhagen-Thiessen, E.; Eckardt, R.; Norman, K. Adherence to a Mediterranean-Style Diet and Appendicular Lean Mass in Community-Dwelling Older People: Results From the Berlin Aging Study II. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.M.; Jameson, K.A.; Batelaan, S.F.; Martin, H.J.; Syddall, H.E.; Dennison, E.M.; Cooper, C.; Sayer, A.A.; Hertfordshire Cohort Study Group. Diet and its relationship with grip strength in community-dwelling older men and women: The Hertfordshire cohort study. J. Am. Geriatr. Soc. 2008, 56, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Akbaraly, T.; Sabia, S.; Hagger-Johnson, G.; Tabak, A.G.; Shipley, M.J.; Jokela, M.; Brunner, E.J.; Hamer, M.; Batty, G.D.; Singh-Manoux, A.; et al. Does Overall Diet in Midlife Predict Future Aging Phenotypes? A Cohort Study. Am. J. Med. 2013, 126, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Leon-Munoz, L.M.; Garcia-Esquinas, E.; Lopez-Garcia, E.; Banegas, J.R.; Rodriguez-Artalejo, F. Major dietary patterns and risk of frailty in older adults: A prospective cohort study. BMC Med. 2015, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Perala, M.M.; Von Bonsdorff, M.; Mannisto, S.; Salonen, M.K.; Simonen, M.; Kanerva, N.; Pohjolainen, P.; Kajantie, E.; Rantanen, T.; Eriksson, J.G. A healthy Nordic diet and physical performance in old age: Findings from the longitudinal Helsinki Birth Cohort Study. Br. J. Nutr. 2016, 115, 878–886. [Google Scholar] [CrossRef] [PubMed]
- Granic, A.; Jagger, C.; Davies, K.; Adamson, A.; Kirkwood, T.; Hill, T.R.; Siervo, M.; Mathers, J.C.; Sayer, A.A. Effect of Dietary Patterns on Muscle Strength and Physical Performance in the Very Old: Findings from the Newcastle 85+ Study. PLoS ONE 2016, 11, e0149699. [Google Scholar] [CrossRef] [PubMed]
- Martin, H.; Aihie Sayer, A.; Jameson, K.; Syddall, H.; Dennison, E.M.; Cooper, C.; Robinson, S. Does diet influence physical performance in community-dwelling older people? Findings from the Hertfordshire Cohort Study. Age Ageing 2011, 40, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Houston, D.K.; Locher, J.L.; Ellison, K.J.; Gropper, S.; Buys, D.R.; Zizza, C.A. Higher Healthy Eating Index-2005 scores are associated with better physical performance. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2012, 67, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Zbeida, M.; Goldsmith, R.; Shimony, T.; Vardi, H.; Naggan, L.; Shahar, D.R. Mediterranean diet and functional indicators among older adults in non-Mediterranean and Mediterranean countries. J. Nutr. Health Aging 2014, 18, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Bollwein, J.; Diekmann, R.; Kaiser, M.J.; Bauer, J.M.; Uter, W.; Sieber, C.C.; Volkert, D. Dietary quality is related to frailty in community-dwelling older adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2013, 68, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Fougere, B.; Mazzuco, S.; Spagnolo, P.; Guyonnet, S.; Vellas, B.; Cesari, M.; Gallucci, M. Association between the Mediterranean-style dietary pattern score and physical performance: Results from TRELONG study. J. Nutr. Health Aging 2016, 20, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Heymsfield, S.B.; Smith, R.; Aulet, M.; Bensen, B.; Lichtman, S.; Wang, J.; Pierson, R.N., Jr. Appendicular skeletal muscle mass: measurement by dual-photon absorptiometry. Am. J. Clin. Nutr. 1990, 52, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, S.; Watanabe, S.; Shibata, H.; Amano, H.; Fujiwara, Y.; Shinkai, S.; Yoshida, H.; Suzuki, T.; Yukawa, H.; Yasumura, S.; et al. Effects of dietary variety on declines in high-level functional capacity in elderly people living in a community. Nihon Koshu Eisei Zasshi 2003, 50, 1117–1124. [Google Scholar] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 26, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; McCullough, M.L.; Newby, P.K.; Manson, J.E.; Meigs, J.B.; Rifai, N.; Willett, W.C.; Hu, F.B. Diet-quality scores and plasma concentrations of markers of inflammation and endothelial dysfunction. Am. J. Clin. Nutr. 2005, 82, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Perälä, M.-M.; Von Bonsdorff, M.B.; Männistö, S.; Salonen, M.K.; Simonen, M.; Kanerva, N.; Rantanen, T.; Pohjolainen, P.; Eriksson, J.G. The healthy Nordic diet predicts muscle strength 10 years later in old women, but not old men. Age Ageing 2017, 46, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo-López, L.; Maseda, A.; De Labra, C.; Regueiro-Folgueira, L.; Rodríguez-Villamil, J.L.; Millán-Calenti, J.C. Nutritional determinants of frailty in older adults: A systematic review. BMC Geriatr. 2017, 17, 108. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Cardiovascular Hlth Study, C.; Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Morley, J.E.; Malmstrom, T.K.; Miller, D.K. A simple frailty questionnaire (FRAIL) predicts outcomes in middle aged African Americans. J. Nutr. Health Aging 2012, 16, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Rimm, E.; Smith-Warner, S.A.; Feskanich, D.; Stampfer, M.J.; Ascherio, A.; Sampson, L.; Willett, W.C. Reproducibility and validity of dietary patterns assessed with a food-frequency questionnaire. Am. J. Clin. Nutr. 1999, 69, 243–249. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.A.; Mishra, G.D.; Bramwell, G.; Paul, A.A.; Wadsworth, M.E.J. Comparability of dietary patterns assessed by multiple dietary assessment methods: Results from the 1946 British Birth Cohort. Eur. J. Clin. Nutr. 2005, 59, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Crozier, S.R.; Inskip, H.M.; Godfrey, K.M.; Robinson, S.M. Dietary patterns in pregnant women: A comparison of food frequency questionnaires and four-day prospective diaries. Br. J. Nutr. 2008, 99, 869–875. [Google Scholar] [CrossRef] [PubMed]
- Ford, D.W.; Jensen, G.L.; Hartman, T.J.; Wray, L.; Smiciklas-Wright, H. Association Between Dietary Quality and Mortality in Older Adults: A Review of the Epidemiological Evidence. J. Nutr. Gerontol. Geriatr. 2013, 32, 85–105. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K. Dietary patterns: Biomarkers and chronic disease risk. Appl. Physiol. Nutr. Metab. 2010, 35, 199–206. [Google Scholar] [CrossRef] [PubMed]
| Outcome | Muscle Mass | Muscle Strength | Physical Performance | Sarcopenia |
|---|---|---|---|---|
| Acceptable measures | Anthropometry a Dual energy X-ray absorptiometry (DXA) Bio impedance analysis (BIA) Computed tomography (CT) Magnetic resonance imaging (MRI) | Handgrip strength Quadriceps strength Muscle quality index | Short Physical Performance Battery (SPPB) Gait/walking speed Timed get-up-and-go test Balance Stair climb power test | Combined outcomes of muscle mass, muscle strength or physical performance |
| Muscle Mass | Muscle Strength | Physical Performance | Physical Performance + Muscle Strength | Physical Performance + Muscle Strength + Muscle Mass | Physical Performance + Muscle Mass | Sarcopenia |
|---|---|---|---|---|---|---|
| Bernstein, 2002 [33] | Rahi, 2014 [35] | Milaneschi, 2011 [37] | Talegawkar, 2012 [41] | Yokoyama, 2017 [46] | Smee, 2015 [47] | Chan, 2016 [29] |
| Cross-sectional | Longitudinal; FU: 3 years | Longitudinal; FU: 3 years, 6 years and 9 years | Longitudinal; FU: 3 years and 6 years | Longitudinal; FU: 4 years | Cross-sectional | Longitudinal; FU: 4 years |
| Risk of bias: High | Risk of bias: Medium | Risk of bias: Medium | Risk of bias: Medium | Risk of bias: Medium | Risk of bias: High | Risk of bias: Low |
| Oh, 2014 [31] | Kojima, 2015 [36] | Shahar, 2012 [38] | León-Muñoz, 2014 [42] | Hashemi, 2015 [30] | ||
| Cross-sectional | Longitudinal; FU: 4 years | Longitudinal; FU: 8 years | Longitudinal; FU: 3.5 years | Cross-sectional | ||
| Risk of bias: Medium | Risk of bias: Medium | Risk of bias: Low | Risk of bias: Low | Risk of bias: Medium | ||
| Nikolov, 2016 [34] | Robinson, 2008 [25] | Akbaraly, 2013 [26] | León-Muñoz, 2015 [28] | |||
| Cross-sectional | Cross-sectional | Longitudinal; FU: 16 years | Longitudinal; FU: 3.5 years | |||
| Risk of bias: Medium | Risk of bias: Low | Risk of bias: Medium | Risk of bias: Low | |||
| Perälä, 2016 [39] | Granic, 2016 [32] | |||||
| Longitudinal; FU: 10 years | Longitudinal; FU: 5 years | |||||
| Risk of bias: Low | Risk of bias: Medium | |||||
| Martin, 2011 [27] | Xu, 2012 [43] | |||||
| Cross-sectional | Cross-sectional | |||||
| Risk of bias: Medium | Risk of bias: Low | |||||
| Zbeida, 2014 [40] | Bollwein, 2013 [44] | |||||
| Cross-sectional | Cross-sectional | |||||
| Risk of bias: Medium | Risk of bias: Low | |||||
| Fougère, 2016 [45] | ||||||
| Cross-sectional | ||||||
| Risk of bias: Medium |
| First Author, Year | Setting | Study Participants | Study Design | Measure of Physical Function | Assessment of Dietary Intake + Measure of Diet Quality (DQ) | Association of Outcome with Exposure | Risk of Bias a |
|---|---|---|---|---|---|---|---|
| Muscle Mass | |||||||
| Bernstein, 2002 [33] | The Boston FICSIT Study, Boston, MA, USA | 98 men and women older than 70 years (aged 72 to 98 years) were recruited among residents of a nursing home Mean ± SD age: 87.1 ± 0.6 years | CS | Mean arm muscle area (MAMA) was calculated and thigh muscle area was measured using computerized tomography (CT) scanning. | 3-day weighed food records on 3 consecutive days of the week Dietary indices: 2 dietary variety scores. | −4: High | |
| (1) Dietary variety score equal to the number of different foods eaten over 3 days. | MAMA approached a significant relationship with dietary variety score (p = 0.06) in both men and women. No association with thigh muscle area was reported. | ||||||
| (2) Fruit and vegetable variety score equal to the number of different fruits and vegetables consumed over the 3 days. | In women, high fruit and vegetable variety score only was associated with higher MAMA (β = 2.94) (p ≤ 0.03). Thigh muscle area was tested and no significant association was found. Conclusions: Results suggest that a highly varied diet in elderly nursing home residents is associated with higher MAMA. | ||||||
| Oh, 2014 [31] | The KNHANES 2011, Korea | 1435 non-institutionalized Korean people who were aged 65 years or more Mean ± SD age: not given | CS | Appendicular skeletal muscle mass (ASM) was measured by DXA. ASM was defined as the sum of lean soft tissue masses for the arms and legs, after the method of Heymsfield et al. [48]. | Single 24-h dietary recall Data-driven: cluster analysis. | +3: Med | |
| (1) “Traditional Korean” dietary pattern b, in which consumption of white rice accounted for 76% of total energy intake. | Compared with the “Traditional Korean” pattern, the “Westernized Korean” pattern was associated with a 74% increased abnormality of ASM/Wt (kg) by logistics analysis. | ||||||
| (2) “Meat and Alcohol” dietary pattern, with a higher consumption of meat and alcohol. | No association was observed. | ||||||
| (3) “Westernized Korean” dietary pattern, based on a rice and vegetable diet but characterized by a variety of food groups such as other grains, fruit, bread, eggs, fish, milk, and alcohol. | See above. Conclusions: A “Westernized Korean” pattern was associated with a markedly increased abnormality of muscle mass, compared to the “Traditional Korean” pattern. | ||||||
| Nikolov, 2016 [34] | BASE-II, Berlin, Germany | 1509 community-dwelling, well-functioning older men and women between 60 and 80 years. Mean ± SD age: 68.2 ± 3.7 years | CS | Body composition was assessed by using DXA. ALM was calculated as the sum of bone-free lean mass of arms and legs and related to height and weight (ALM/BMI). The proportion of ALM to whole body fat mass (FM) was defined as the ALM/FM ratio. | Self-administered EPIC-FFQ. Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the Mediterranean-type diet score (mMedTypeDiet) suggested by Grosso and colleagues. | A higher adherence to the mMedTypeDiet was associated with higher ALM/BMI in women and better ALM/FM ratio when compared to a medium and a low diet quality. No significant association was found in men. Conclusions: Higher adherence to a Mediterranean-style diet was associated with a positive effect on ALM/BMI in women. | +3: Med |
| Muscle Strength | |||||||
| Rahi, 2014 [35] | Secondary analysis of the NuAge cohort, QC, Canada. | 156 community-dwelling men and women with type 2 diabetes Mean ± SD age: at baseline 74.6 ± 4.2 years | LS | Handgrip, knee extensor and elbow flexor strengths, were measured at recruitment and at the 3-year follow-up. Crude change was calculated by subtracting values at recruitment from values at the 3-year follow-up. In order to show the yearly MS decline, the percentage relative change per year was adjusted for baseline value. | Three non-consecutive 24-h dietary recalls (on two randomly-chosen weekdays and one weekend day). Dietary indices: DQ was evaluated at recruitment using the Canadian Healthy Eating Index (C-HEI). | There was no effect of DQ at baseline on maintenance of the three measures of muscle strength, in either males or females. Likewise, DQ, which was dichotomized based on the median or categorized into quartiles, showed no significant effects on MS maintenance. Conclusions: DQ alone had no effect on MS maintenance in this sample of diabetic older men and women. However, when good DQ was combined with stable or increased PA, MS losses were minimal in diabetic older males over the 3-year follow-up, despite some discordance between changes in MS in the upper and lower extremities. | +2: Med |
| Kojima, 2015 [36] | Itabashi Ward of Tokyo, Japan | 575 community-dwelling women from the Itabashi Ward of Tokyo Mean ± SD age: ages ranged between 75 and 85 years (78.07 ± 2.56) in 2008 and between 78 and 89 years (82.07 ± 2.55) in 2012 | LS | Isometric knee extension strength (KES, in N) was measured in the dominant leg using a hand-held dynamometer incorporated into a custom-made frame. | Participants were asked closed-ended questions about intake frequencies of 10 food groups Dietary indices: A DVS, an index of dietary variety introduced by Kumagai et al. [49], was calculated. | There was no significant cross-sectional relationship between KES and DVS. Longitudinal analysis showed that except for 3 food groups, no lifestyle-related variables at baseline were related to changes in KES over 4 years. Conclusions: The age-related decline in muscle strength was lower in people who frequently ate soy products or green and yellow vegetables, but no association was found with DVS. | +1: Med |
| Robinson, 2008 [25] * | HCS, UK | 2983 community-dwelling men and women aged 59 to 73 years Mean ± SD age: Men: 65.7 ± 2.9 years; Women: 66.6 ± 2.7 years | CS | Maximum grip strength was measured using a handgrip Jamar dynamometer. | Administered FFQ based on EPIC questionnaire, pertaining to 3-month period preceding the interview. Data-driven: PCA. Using PCA, a “prudent” dietary pattern c was identified. | Men and women with high prudent diet scores had higher grip strength. In men, the association was no longer evident when fatty fish consumption was accounted for. In women, independent associations between grip strength and prudent diet score and fatty fish consumption remained, although the size of the effect was markedly reduced (regression coefficient of 0.17, 95% CI = 0.00 to 0.34 kg per unit change in score, p = 0.044). Conclusions: Whilst a healthier pattern of eating was associated with higher grip strength, this effect was at least partly explained by more prudent diets also being characterised by greater consumption of fatty fish. | +5: Low |
| Physical Performance | |||||||
| Milaneschi, 2011 [37] * | InCHIANTI (Invecchiare in Chianti), study, Tuscany, Italy | Older men and women: 705 participants had available data on lower body mobility at 3-year follow-up, 614 at 6-year follow-up and 486 at 9-year follow-up. Mean ± SD age: at baseline 74.1 ± 6.8 years | LS | Lower extremity function was measured at baseline, and at the 3-, 6- and 9-year follow-up visits using the SPPB, which was derived from three objective tests: 4-m walking speed, repeated chair rises and standing balance in progressively more challenging positions. | FFQ created for EPIC, validated in this population. Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the MDS by Trichopoulou et al. [50]. | At baseline, higher adherence to Mediterranean diet was associated with better lower body performance. Participants with higher adherence experienced less decline in SPPB score, which was of 0.9 points higher (p < 0.0001) at the 3-year-follow, 1.1 points higher (p = 0.0004) at the 6-year follow-up and 0.9 points higher (p = 0.04) at the 9-year follow-up compared to those with lower adherence. Among participants free of mobility disability at baseline, those with higher adherence had a lower risk (HR (hazard ratio) = 0.71, 95% CI = 0.51–0.98, p = 0.04) of developing mobility disability (defined as SPPB ≤ 9 points). Conclusions: High adherence to a Mediterranean-style diet is associated with a slower decline of mobility over time in community-dwelling older persons. | +2: Med |
| Shahar, 2012 [38] | Health, Aging, and Body Composition cohort study, USA | 1201 participants Mean ± SD age: at baseline 74.6 ± 2.9 years (middle category of MD score) | LS | Performance-based evaluations included usual and rapid walking speed assessed over a 20-metre course. | Administered FFQ Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the MDS by Trichopoulou et al. [50]. | Higher MD adherence was an independent predictor of less decline in usual 20 m walking speed (p = 0.049). The effect decreased after adding total body-fat-percent to the model (p = 0.134). Similar results were observed for MD .adherence and rapid 20 m walking speed; the association remained significant after adjustment for total body-fat-percent (p = 0.012). Conclusions: Walking speed over 8 years was faster among those with higher MD adherence at baseline. The differences remained significant over 8 year, suggesting a long-term effect of diet on mobility performance with aging. | +4: Low |
| Akbaraly, 2013 [26] | Whitehall II study (London-based office staff), UK | 5350 men and women aged 60 years or older at the final follow-up Mean ± SD age: at baseline 51.3 ± 5.3 years | LS | Walking speed over a 8-feet walking course. | Semi-quantitative FFQ Data-driven: PCA. Two dietary patterns were identified. | +2: Med | |
| (1) “Healthy-foods” dietary pattern. | No association was reported | ||||||
| (2) “Western-type” dietary pattern. | Participants in the highest tertile of “Western-type” dietary pattern, compared with those in the bottom tertile, were more likely to have poorer musculoskeletal functioning (OR (odds ratio) = 1.45; 95% CI = 1.14–1.84). | ||||||
| Dietary indices: Adherence to the Alternative Healthy Eating Index (AHEI) was calculated. | No association was reported Conclusions: Avoidance of “Western type foods” might increase the possibility of achieving older ages with better musculoskeletal functioning (faster walking speed). | ||||||
| Perälä, 2016 [39] | Helsinki Birth Cohort Study, Finland | 1072 men and women Mean ± SD age: 61.3 ± 0.2 (SE) years | LS | Physical performance was assessed using the validated Senior Fitness Test (SFT) battery d. | Self-administered FFQ pertaining to the previous 12 months Dietary indices: An a priori-defined Nordic diet score (NDS) was calculated, as a measure of a healthy Nordic diet. | In a fully adjusted model, the overall Senior Fitness Test (SFT) score was 0.55 (95% CI = 0.22, 0.88) points higher per 1 unit increase in the NDS. Women in the highest fourth of the NDS had on average 5 points higher SFT score compared with those in the lowest fourth (p for trend 0.005). No such association was observed in men. Women with the highest score had 17% better result in the walk test, 16% better arm curl and 20% better chair stand results compared with those with the lowest score (all p values < 0.01). Conclusions: The study indicates that among women a healthy Nordic diet predicts better physical performance, and especially better aerobic endurance and upper- and lower-body strength 10 years later. | +5: Low |
| Martin, 2011 [27] * | HCS, UK | 628 community-dwelling men and women Mean ± SD age: 68.0 ± 2.5 years | CS | Participants completed a short physical performance battery. This included measures of time taken to complete a 3-m customary pace walk and 5 sit–stand chair rises; balance performance was assessed by measurement of one-legged timed standing balance. | Administered FFQ pertaining to 3-month period preceding the interview Data-driven: PCA. A “prudent” dietary pattern was identified c. | In men, no independent associations were found between 3-m walk time and diet. For women, a higher prudent diet score was associated with shorter 3-m walk time (p = 0.016), although this association was not robust to adjustment for confounding factors. In men, there were no associations between diet and chair-rise time. Among the women, univariate comparisons showed that shorter chair-rise times were associated with higher prudent diet scores (p = 0.011). However, this association was not robust to adjustment. Higher prudent diet scores in the women were associated with better balance (p = 0.033) but this was not robust to adjustment for confounders. This association was not observed in men. Conclusions: There were no independent associations between the dietary pattern and physical performance. | +3: Med |
| Zbeida, 2014 [40] * | US NHANES and the Israeli MABAT ZAHAV survey e | NHANES: 2791 people aged 60 years and older. Mean age: 71.2 years. | CS | Observed timed 20-feet walk. | 24-h multiple-pass dietary recall interview on a random day of the week. Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the MDS, similar to that constructed by Trichopoulou et al. [50]. | MDS (high vs. low) was associated with faster walking speed after adjusting for confounders in a logistic regression model (OR = 0.71, p = 0.034, 95% CI = 0.511–0.974]. Conclusions: In a secondary analysis of the national health and nutrition survey data from the US, adherence to the Mediterranean diet was significantly associated with better physical functional abilities among older people. | +2: Med |
| Physical Performance + Muscle Strength | |||||||
| Talegawkar, 2012 [41] * | InCHIANTI (Invecchiare in Chianti), study, Tuscany, Italy | 690 older men and women (people ≥ 65 years) Mean ± SD age: at baseline 73.0 ± 6.24 years | LS | MS: grip strength. PP: walking speed (time to walk 15 feet). | FFQ created for EPIC, validated in this population. Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the MDS by Trichopoulou et al. [50]. | MS: No association was observed for grip strength. | +2: Med |
| PP: After a 6-year follow-up, higher adherence to a MD at baseline was associated with a lower risk of low walking speed (OR = 0.48 (95% CI = 0.27, 0.86)). Conclusions: In community-dwelling older adults, higher adherence to a Mediterranean-style diet was associated with a lower risk of low walking speed. | |||||||
| León-Muñoz, 2014 [42] * | ENRICA cohort, Spain | 1815 community-dwelling people aged ≥ 60 years Mean ± SD age: at baseline 68.5 ± 0.3 years | LS | MS: measured with a Jamar dynamometer on the dominant hand. PP: walking speed was assessed using the 3-metre walking speed test. | Validated computerized diet history. Dietary indices: Adherence to a Mediterranean dietary pattern. | +4: Low | |
| (1) Mediterranean Diet Adherence Screener (MEDAS). | MS: No significant association was observed. | ||||||
| PP: Being in the highest tertile of the MEDAS score (highest MD adherence) was associated with reduced risk of slow walking (OR = 0.53; 95% CI = 0.35–0.79). | |||||||
| (2) MDS. | MS: Participants in the highest tertile of the MDS had lower risk of low grip strength, but the association was not statistically significant. | ||||||
| PP: No association was observed. Conclusions: Among community-dwelling older adults a higher adherence to the MD was associated with reduced risk of slow walking. | |||||||
| León-Muñoz, 2015 [28] | ENRICA cohort, Spain | 1872 community-dwelling people aged ≥ 60 years Mean ± SD age: at baseline: 68.7 ± 0.3 years | LS | MS: strength on the dominant hand was measured with a Jamar dynamometer. PP: walking speed was assessed with the 3-m walking speed test. | Validated computerized diet history. Data-driven: Factor analysis. Two patterns were identified. | +5: Low | |
| (1) The first was called the “prudent” pattern due to the high consumption of olive oil, vegetables, potatoes, legumes, blue fish, pasta, and meat. | MS: No association was observed | ||||||
| PP: A greater adherence to the prudent pattern showed a non-statistically significant tendency to a lower risk of slow walking speed. | |||||||
| (2) The second was called the “Westernized” pattern because of the high consumption of refined bread, whole dairy products, and red and processed meat, as well as the low intake of whole grains, fruit, low-fat dairy, and vegetables. | MS: No association was observed. | ||||||
| PP: The westernized pattern showed an association with an increasing risk of slow walking speed. Specifically, the OR (95% CI) of slow walking speed across tertiles of the WP were 1, 1.15 (0.74–1.76), and 1.85 (1.19–2.87); p-trend = 0.007. Conclusions: Greater adherence to the “Westernized” pattern was associated with increased risk of slow walking speed. | |||||||
| Granic, 2016 [32] | The Newcastle 85+ Study, UK | 791 men and women (living either at home or in a care facility) were followed-up for change in hand grip strength (HGS) and Timed Up-and Go (TUG) test over 5 years. Participants with DP and HGS data 5 years later (n = 291). Participants with DP and TUG data 5 years later (n = 271) Mean age: the Newcastle 85+ Study targeted 85 years old at baseline | LS | MS: hand grip strength (HGS) was assessed using a hand-held dynamometer. PP: assessed by the Timed Up-and-Go (TUG) test. | 24-h multiple-pass dietary recall on two different days of the week, at least one week apart. Data-driven: Cluster analysis. Three dietary patterns (DP) were identified. | +3: Med | |
| (1) DP1 (“High Red Meat”). | MS: Men in DP1 (“High Red Meat”) had worse overall HGS (β = −1.70, p = 0.05) compared with DP2 (“Low Meat”). No association between DP and HGS was observed in women. | ||||||
| PP: Men in DP1 and women in DP3 had overall slower TUG than those in DP2 (β = 0.08, p = 0.001 and β = 0.06, p = 0.01, respectively), but similar rate of decline. | |||||||
| (2) DP2 (“Low Meat”). | MS: See above and below. | ||||||
| PP: See above. | |||||||
| (3) DP3 (“High Butter”). | MS: Men in DP3 (“High Butter”) had a steeper decline in HGS (β = −0.63, p = 0.05) than men in DP2 (“Low Meat”). No association between DP and HGS was observed in women. | ||||||
| PP: See above. Conclusions: DP high in red meats, potato and gravy (DP1), or butter (DP3) may adversely affect muscle strength and physical performance in later life. | |||||||
| Xu, 2012 [43] * | 1999–2002 NHANES, USA | The final sample size was 2132 for gait speed and 1392 for knee extensor power. Men and women aged 60 years or older Mean ± SD age: 70.4 ± 0.3 (SE) years | CS | MS: knee extensor power. Right knee extensor force production was measured using an isokinetic dynamometer. PP: gait speed; the timed 20-feet walk test was performed at the participant’s usual pace. | 24-h multiple-pass dietary recall interview. Dietary indices: The Healthy Eating Index-2005 (HEI-2005), a composite score assessing an individual’s adherence to the 2005 Dietary Guidelines for Americans, was used to measure an individual’s overall diet quality. | MS: Total HEI-2005 scores were positively associated with knee extensor power (p for trend = 0.05). Those with HEI-2005 scores in Quartile 4 had a greater knee extensor power compared with those with HEI-2005 scores in the lowest quartile (p = 0.04). The associations were no longer statistically significant after further adjustment for PA. | +4: Low |
| PP: Total HEI-2005 scores were positively associated with gait speed (p for trend = 0.02). Older adults with higher HEI-2005 scores had a faster gait speed (p = 0.03 for both Quartile 3 and Quartile 4 versus Quartile 1) compared with those with HEI-2005 scores in the lowest quartile. The associations were no longer statistically significant after further adjustment for PA. Conclusions: Adherence to overall dietary recommendations was associated with better physical performance among older adults. | |||||||
| Bollwein, 2013 [44] | Region of Nürnberg, Germany | 192 community-dwelling older men and women, aged 75 years and older Mean ± SD age: 83 ± 4 years | CS | MS: grip strength was measured with a dynamometer. PP: walking speed. | Administered FFQ of the German part of the EPIC study. Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the MD score used was the alternate MED score of Fung et al. [51] who adapted the original score of Trichopoulou et al. [50] for a non-Mediterranean population. | MS: No association was observed. | +4: Low |
| PP: There was a significant inverse association between “low walking speed” and the MED score; there was an association between a high diet quality and a lower risk of low walking speed. Compared with the lowest quartile (least healthy diet), the participants in the highest quartile (most healthy diet) had a significantly decreased risk of low walking speed (OR (95% CI) = 0.29 (0.09–1.00), p for trend = 0.043). Conclusions: There was an association between adherence to a healthy dietary pattern and low walking speed. | |||||||
| Fougère, 2016 [45] | TRELONG study, Northeast Italy | 304 men and women, over 70 years of age at baseline (aged 77 years and over). Mean ± SD age: 86.3 ± 6.8 years | CS | MS: hand grip strength was measured using a dynamometer with the stronger hand. PP: The SPPB was used: standing balance, walking, and chair stand tests. Standing balance tests included tandem, semi tandem and side-by-side stands. | Unclear how dietary data were collected. Dietary indices: Adherence to a Mediterranean dietary pattern was assessed using the Mediterranean Style Dietary Pattern Score (MSDPS). | MS: No correlation was found for hand grip strength. | +1: Med |
| PP: A statistically significant association (Regression coefficient = 1.0006; Std. Error = 0.4780; p-value = 0.0363) between participants with the highest adherence to the Mediterranean diet (fourth quartile) and high physical performance (SPPB > 7) was found. Conclusions: A statistically significant association between a high adherence to the Mediterranean diet and higher physical performance (SPPB) was found. These findings suggest that MD does not improve the muscle (hand grip) but rather improve global function (indicated by SPPB). | |||||||
| Physical Performance + Muscle Strength + Muscle Mass | |||||||
| Yokoyama, 2017 [46] | Kusatsu Longitudinal Study, and the Hatoyama Cohort Study, Japan | Community-dwelling Japanese aged 65 years or older. Grip strength: n = 781; Gait speed: n = 772; Body composition: n = 542 Mean ± SD age: according to DVS categories: Lowest: 71.1 ± 4.9; Middle: 71.8 ± 5.1; Highest: 72.8 ± 5.6 | LS | MM: Body composition was measured using the InBody 720 device. In this study, lean body mass refers to bone-free lean mass. The sum of non-fat, non-bone tissue in both arms and legs was used to represent ALM. MS: grip strength was measured using hand dynamometers, with the dominant hand.PP: usual gait speed was measured over a straight 11-m walkway. | Participants were asked about consumption frequencies during 1 week for 10 food items. Dietary indices: Dietary variety was assessed at the baseline survey using the DVS (a method of assessing dietary quality based on a count of the number of foods consumed). DVS was categorized into 3 groups. | MM: Dietary variety was not significantly associated with changes in lean body mass or ALM. However, the OR for decline in ALM tended to decrease with increasing DVS at baseline; the multivariable-adjusted OR for decline in ALM was 0.28 (0.07–1.07) for the highest DVS category as compared with the lowest DVS category (p for trend = 0.068). | +3: Med |
| MS: ORs for decline in grip strength was 0.43 (95% CI = 0.19–0.99), for the highest category of dietary variety score as compared with the lowest category. | |||||||
| PP: ORs for decline in usual gait speed was 0.43 (confidence interval, 0.19–0.99), respectively, for the highest category of dietary variety score as compared with the lowest category. Conclusions: Greater dietary variety was associated with lower risk for future declines in physical function (muscle strength and gait speed), but the associations with lean body mass and ALM were less clear. The findings indicate that among older adults, greater dietary variety may help maintain physical performance and muscle strength but not lean mass. | |||||||
| Physical Performance + Muscle Mass | |||||||
| Smee, 2015 [47] | Canberra, Australia | 171 cognitively unimpaired, community-dwelling men and women aged 60 years and over Mean ± SD age: 68.12 ± 6.21 years | CS | MM: Lean mass was assessed by DXA. PP: measured using the SPPB. | Valid semi-quantitative self-administered questionnaire. Dietary indices: Diet quality was determined using two measures. | −4: High | |
| (1) Healthy Eating Index (HEI) | MM: Lean mass was not significantly associated with the HEI-total score. | ||||||
| PP: SPPB was not significantly associated with the HEI-total score. | |||||||
| (2) Healthy Diet Indicator (HDI) | MM: In women, there was a weak positive association between HDI score and % lean mass (r = 0.20, p = 0.03). When controlling for age, there was a weak negative correlation (r = 1.19, p = 0.03). | ||||||
| PP: Men showed weak positive associations between HDI score and SPPB (r = 0.26, p = 0.04). Conclusions: In women, there was a weak association between better diet quality and higher lean mass, which disappeared when controlling for age. In men there was a weak association between better diet quality and better physical function (SPPB). | |||||||
| Sarcopenia | |||||||
| Chan, 2016 [29] | Hong Kong, China | Chinese community-dwelling men and women, aged 65 years or older. The final sample size for the cross-sectional analyses was 3957 and for the prospective analyses was 2948 Mean ± SD age: At baseline: Non-sarcopenic participants (n = 3667): 72.2 ± 5.0 years; Sarcopenic participants (n = 290): 76.2 ± 6.1 years | LS | Sarcopenia was defined according to the AWGS algorithm. An individual with low muscle mass, low muscle strength, and/or low physical performance was categorized as having sarcopenia. | Validated semi-quantitative FFQ Data-driven: factor analysis. | +5: Low | |
| (1) “Vegetables-fruits” dietary pattern. | At baseline, men with higher “vegetables-fruits” dietary pattern score, and higher “snacks-drinks-milk products” dietary pattern score had lower likelihood of being sarcopenic. Men in the highest quartile of “vegetables-fruits” pattern score (adjusted OR = 0.60, 95% CI = 0.36–0.99, p for trend = 0.034) showed reduced likelihood of sarcopenia compared with men in the lowest quartile. No such associations were observed in women. No significant associations were found between any of the dietary patterns and 4-year incident sarcopenia. | ||||||
| (2) “Snacks-drinks-milk products” dietary pattern. | See above. Higher quartile of “snacks-drinks-milk products” pattern score was associated with lower likelihood of sarcopenia in men (adjusted OR = 0.41, 95% CI = 0.24–0.70, p for trend < 0.001). No such associations were observed in women. | ||||||
| (3) “meat-fish” dietary pattern. | Sarcopenia: No association was observed. | ||||||
| Dietary indices. | |||||||
| (1) Adherence to a Mediterranean dietary pattern was assessed using the MDS by Trichopoulou et al. [50] | Sarcopenia: No associations were found between MDS and sarcopenia. | ||||||
| (2) The Diet Quality Index-International (DQI-I) was calculated. | Sarcopenia: At baseline, men with higher DQI-I score had lower likelihood of being sarcopenic. Men in the highest quartile of DQI-I had reduced likelihood of sarcopenia (adjusted OR = 0.50, 95% CI = 0.31–0.81, p for trend = 0.004) compared with men in the lowest quartile. No such associations were observed in women. No significant associations were found between dietary patterns and 4-year incident sarcopenia. Conclusions: Higher DQI-I score, higher “vegetables-fruits” dietary pattern score, and higher “snacks-drinks-milk products” dietary pattern score were associated with lower likelihood of prevalent sarcopenia in Chinese older men. | ||||||
| Hashemi, 2015 [30] | Tehran, Iran | 300 elderly men and women (55 years old and older) who lived in the sixth district of Tehran Mean ± SD age: 66.8 ± 7.72 years | CS | Sarcopenia was defined according to EWGSOP criteria, based on a combination of relatively low appendicular muscle mass with either low muscle strength or low muscle performance. | Administered FFQ. Data-driven: PCA. Three major dietary patterns were identified. | +2: Med | |
| (1) Mediterranean, defined as a dietary pattern with high factor loadings (>0.4) in food groups such as olives and olive oil, low and high carotenoid vegetables, tomatoes, whole grains, nuts, fish, fresh and dried fruits, and pickles. | Participants in the highest tertile of the MD pattern had a lower odds ratio for sarcopenia than those in the lowest tertile (OR = 0.42; 95% CI = 0.18–0.97; p for trend = 0.04). | ||||||
| (2) Western, defined as a dietary pattern with high factor loadings in tea, soy, sweets, desserts, sugars, and fast foods. | Adherence to the Western dietary pattern was not associated with sarcopenia (OR = 0.51; 95% CI = 0.21–1.24; p for trend = 0.13). | ||||||
| (3) Mixed, identified as a pattern with high factor loadings in the following food groups: animal proteins, legumes, potatoes, and refined grains. | Adherence to the Mixed dietary pattern did not affect the odds of sarcopenia (OR = 1.45; 95% CI = 0.66–3.19; p for trend = 0.95). Conclusions: The study suggests that adherence to the MD is associated with lower odds of sarcopenia among Iranian older adults. | ||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bloom, I.; Shand, C.; Cooper, C.; Robinson, S.; Baird, J. Diet Quality and Sarcopenia in Older Adults: A Systematic Review. Nutrients 2018, 10, 308. https://doi.org/10.3390/nu10030308
Bloom I, Shand C, Cooper C, Robinson S, Baird J. Diet Quality and Sarcopenia in Older Adults: A Systematic Review. Nutrients. 2018; 10(3):308. https://doi.org/10.3390/nu10030308
Chicago/Turabian StyleBloom, Ilse, Calum Shand, Cyrus Cooper, Sian Robinson, and Janis Baird. 2018. "Diet Quality and Sarcopenia in Older Adults: A Systematic Review" Nutrients 10, no. 3: 308. https://doi.org/10.3390/nu10030308
APA StyleBloom, I., Shand, C., Cooper, C., Robinson, S., & Baird, J. (2018). Diet Quality and Sarcopenia in Older Adults: A Systematic Review. Nutrients, 10(3), 308. https://doi.org/10.3390/nu10030308





