Resveratrol and Pterostilbene Exhibit Anticancer Properties Involving the Downregulation of HPV Oncoprotein E6 in Cervical Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Determination of IC50 Using WST-1 Assay
2.3. Live Imaging
2.4. Clonogenic Assay
2.5. Scratch Assay
2.6. Flow Cytometry
2.7. Western Blot Analysis
2.8. Immunocytochemistry
2.9. Statistical Analysis
3. Results
3.1. Pterostilbene Is More Potent in Eliminating HPV+ HeLa Cells Compared to Resveratrol
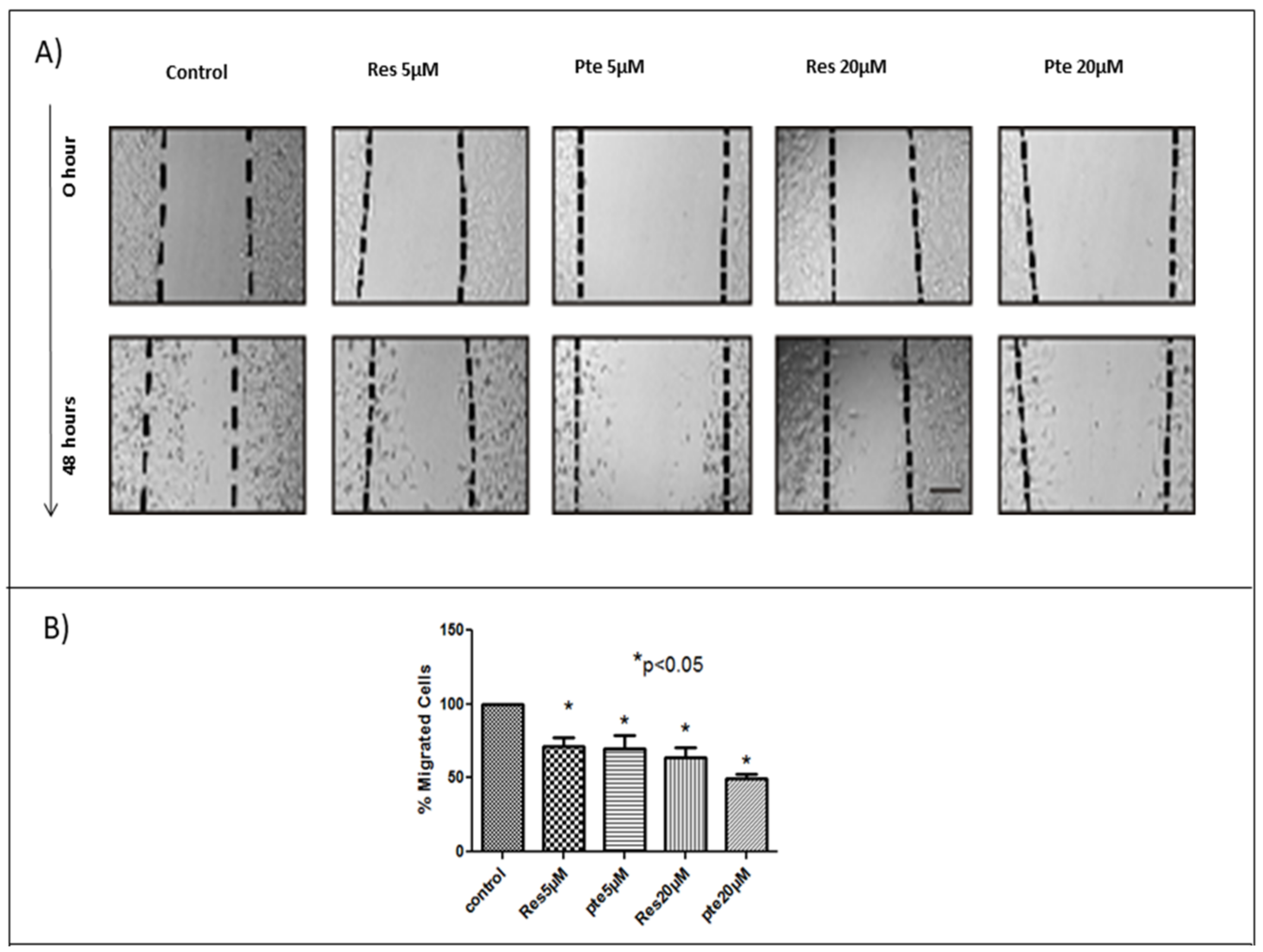
3.2. Inhibition of Cell Migration of HeLa Cells Treated with Pterostilbene and Resveratrol
3.3. Cell Cycle Arrest at S-Phase in HeLa Cells Treated with Low Concentrations of Resveratrol and Pterostilbene
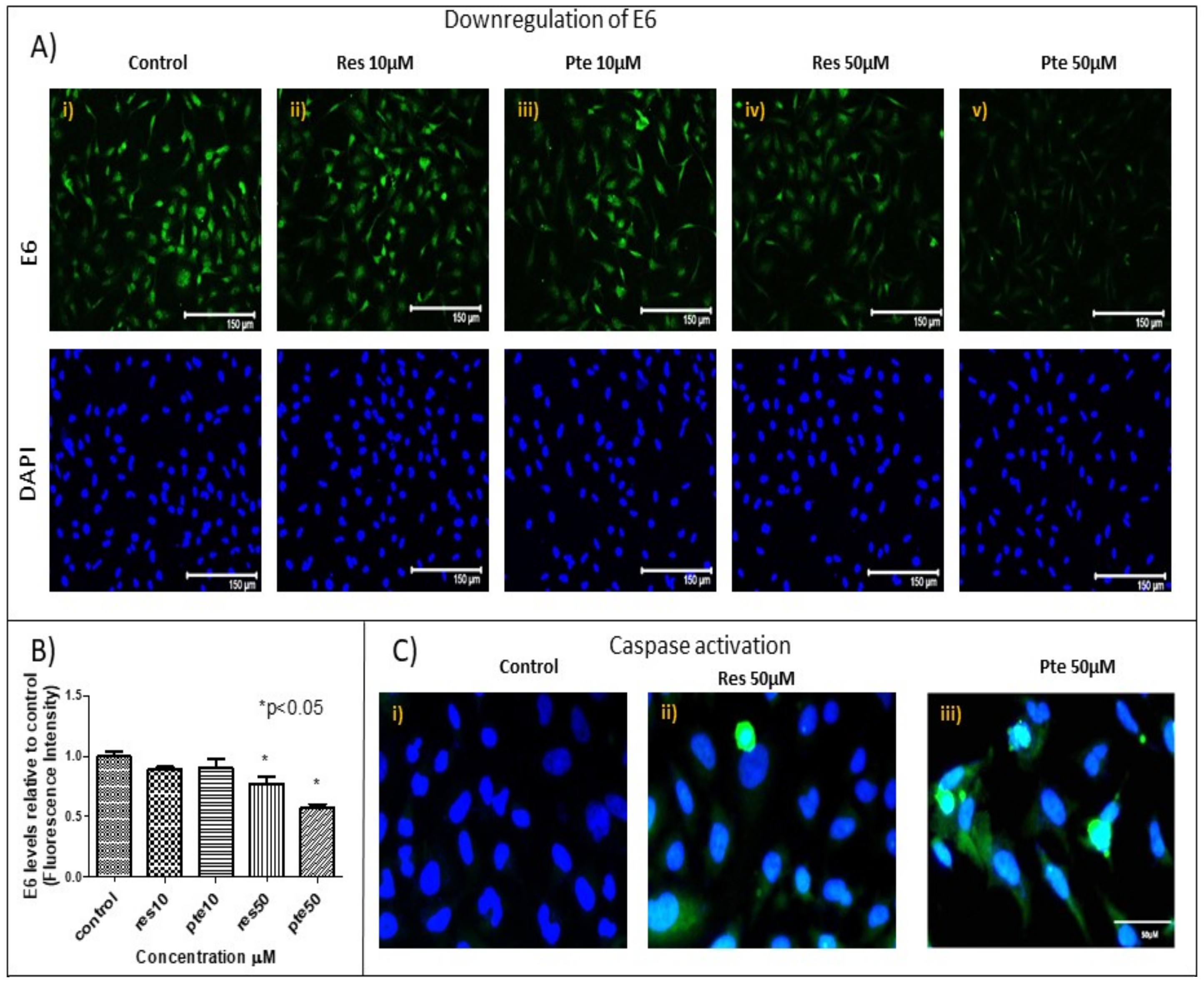
3.4. Downregulation of Viral Oncoprotein E6 and Upregulation of Active-Caspase-3 in HeLa Cells Treated with Pterostilbene and Resveratrol
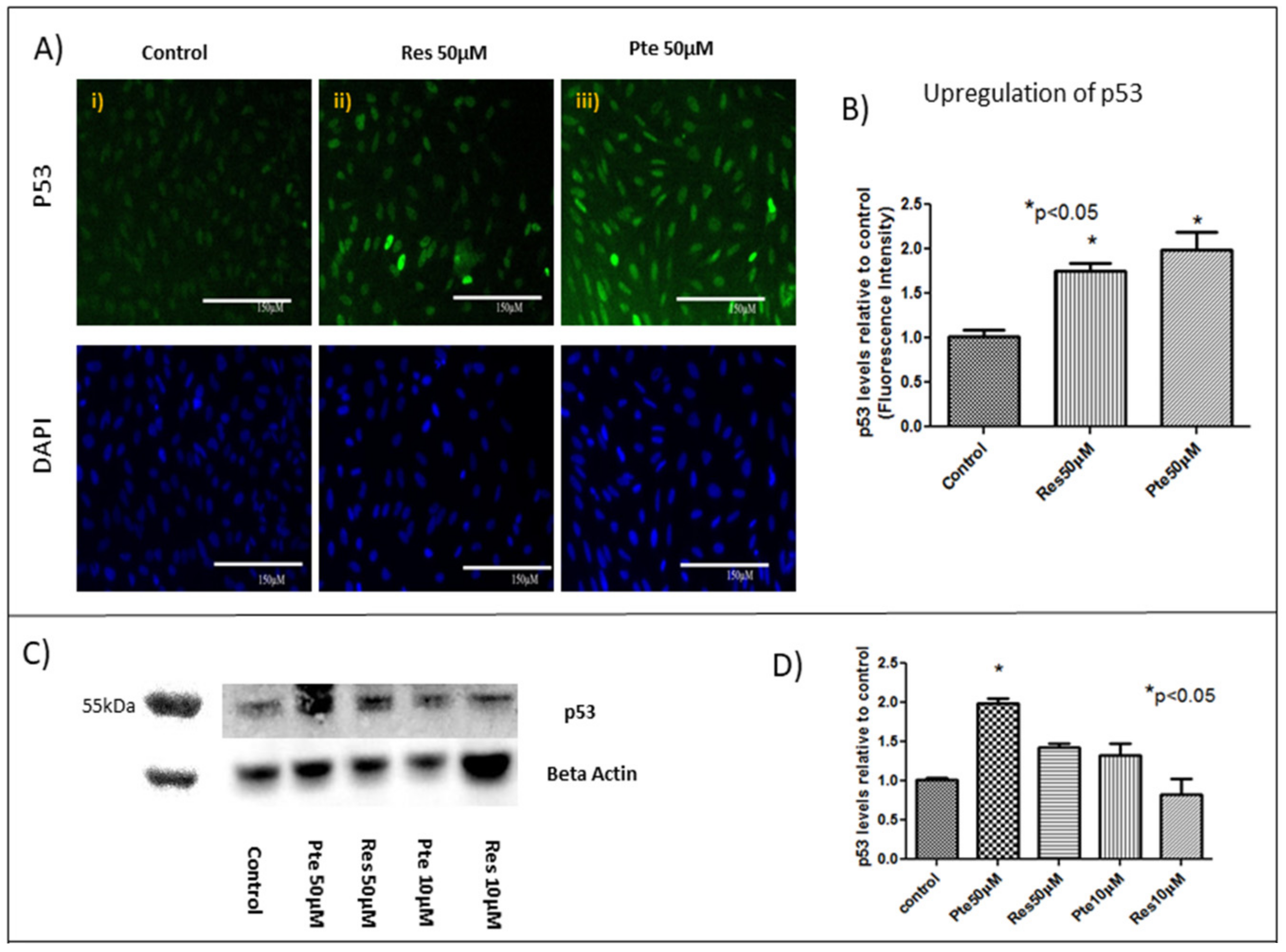
3.5. Upregulation of Tumor Suppressor Protein p53 in HeLa Cells Treated with Pterostilbene and Resveratrol
4. Discussion and Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Safaeian, M.; Solomon, D.; Castle, P.E. Cervical cancer prevention—Cervical screening: Science in evolution. Obstet. Gynecol. Clin. N. Am. 2007, 34, 739–760. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Burd, E.M. Human papillomavirus and cervical cancer. Clin. Microbiol. Rev. 2003, 16, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Parkin, D.M.; Bray, F. Chapter 2: The burden of HPV-related cancers. Vaccine 2006, 24. [Google Scholar] [CrossRef] [PubMed]
- Palefsky, J.M. Human papillomavirus-related disease in men: Not just a women’s issue. J. Adolesc. Health 2010, 46, S12–S19. [Google Scholar] [CrossRef] [PubMed]
- Serrano, B.; Brotons, M.; Bosch, F.X.; Bruni, L. Epidemiology and burden of HPV-related disease. Best Pract. Res. Clin. Obstet. Gynaecol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Wigle, J.; Coast, E.; Watson-Jones, D. Human papillomavirus (HPV) vaccine implementation in low and middle-income countries (LMICs): Health system experiences and prospects. Vaccine 2013, 31, 3811–3817. [Google Scholar] [CrossRef] [PubMed]
- Stanley, M. Potential mechanisms for HPV vaccine-induced long-term protection. Gynecol. Oncol. 2017, 118, S2–S7. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, R.; Katsumata, N.; Shibata, T.; Kamura, T.; Kasamatsu, T.; Nakanishi, T.; Nishimura, S.; Ushijima, K.; Takano, M.; Satoh, T.; et al. Paclitaxel Plus Carboplatin Versus Paclitaxel Plus Cisplatin in Metastatic or Recurrent Cervical Cancer: The Open-Label Randomized Phase III Trial JCOG0505. J. Clin. Oncol. 2015, 33, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Astolfi, L.; Ghiselli, S.; Guaran, V.; Chicca, M.; Simoni, E.; Olivetto, E.; Lelli, G.; Martini, A. Correlation of adverse effects of cisplatin administration in patients affected by solid tumours: A retrospective evaluation. Oncol. Rep. 2013, 29, 1285–1292. [Google Scholar] [CrossRef] [PubMed]
- Stoner, G.D.; Mukhtar, H. Polyphenols as cancer chemopreventive agents. J. Cell. Biochem. 1995, 59, 169–180. [Google Scholar] [CrossRef]
- Mukherjee, S.; Ranjan Debata, P.; Hussaini, R.; Chatterjee, K.; NE Baidoo, J.; Sampat, S.; Navarra, J.P.; Fata, J.; Severinova, E.; Banerjee, P.; et al. Unique synergistic formulation of curcumin, epicatechin gallate and resveratrol, tricurin, suppresses HPV E6, eliminates HPV+ cancer cells, and inhibits tumor progression. Oncotarget 2017, 8, 60904–60916. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-G.; Liu, W.-Y.; Chen, G.-T. A simple method for the isolation and purification of resveratrol from Polygonum cuspidatum. J. Pharm. Anal. 2013, 3, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Dudley, J.I.; Das, D.K. Dose-dependency of resveratrol in providing health benefits. Dose Response 2010, 8, 478–500. [Google Scholar] [CrossRef] [PubMed]
- Nutakul, W.; Sobers, H.S.; Qiu, P.; Dong, P.; Decker, E.A.; McClements, D.J.; Xiao, H. Inhibitory effects of resveratrol and pterostilbene on human colon cancer cells: A side-by-side comparison. J. Agric. Food Chem. 2011, 59, 10964–10970. [Google Scholar] [CrossRef] [PubMed]
- Francioso, A.; Mastromarino, P.; Masci, A.; d’Erme, M.; Mosca, L. Chemistry, stability and bioavailability of resveratrol. Med. Chem. 2014, 10, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S. Resveratrol and derivatives for the prevention and treatment of cancer. Drug Discov. Today 2010, 15, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Langcake, P.; Cornford, C.A.; Pryce, R.J. Identification of pterostilbene as a phytoalexin from Vitis vinifera leaves. Phytochemistry 1979, 18, 1025–1027. [Google Scholar] [CrossRef]
- McCormack, D.; McFadden, D. Pterostilbene and cancer: Current review. J. Surg. Res. 2012, 173. [Google Scholar] [CrossRef] [PubMed]
- Riche, D.M.; McEwen, C.L.; Riche, K.D.; Sherman, J.J.; Wofford, M.R.; Deschamp, D.; Griswold, M. Analysis of safety from a human clinical trial with pterostilbene. J. Toxicol. 2013, 2013, 463595. [Google Scholar] [CrossRef] [PubMed]
- Estrela, J.M.; Ortega, A.; Mena, S.; Rodriguez, M.L.; Asensi, M. Pterostilbene: Biomedical applications. Crit. Rev. Clin. Lab. Sci. 50 2013, 50, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Rimando, A.M.; Lee, H.J.; Ji, Y.; Reddy, B.S.; Suh, N. Anti-inflammatory Action of Pterostilbene Is Mediated through the p38 Mitogen-Activated Protein Kinase Pathway in Colon Cancer Cells. Cancer Prev. Res. 2009, 2, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Jena, L.; Sahoo, M.; Kakde, M.; Daf, S.; Varma, A.K.; Hoory, T.; Monie, A.; Gravitt, P.; Wu, T.; et al. In Silico Docking to Explicate Interface between Plant-Originated Inhibitors and E6 Oncogenic Protein of Highly Threatening Human Papillomavirus 18. Genom. Inform. 2015, 13, 60. [Google Scholar] [CrossRef] [PubMed]
- Scheffner, M.; Huibregtse, J.M.; Vierstra, R.D.; Howley, P.M. The HPV-16 E6 and E6-AP complex functions as a ubiquitin-protein ligase in the ubiquitination of p53. Cell 2017, 75, 495–505. [Google Scholar] [CrossRef]
- Chung, E.; Prelli, F.; Dealler, S.; Lee, W.S.; Chang, Y.-T.; Wisniewski, T. Styryl-based and tricyclic compounds as potential anti-prion agents. PLoS ONE 2011, 6, e24844. [Google Scholar] [CrossRef] [PubMed]
- Kusaczuk, M.; Krętowski, R.; Stypułkowska, A.; Cechowska-Pasko, M. Molecular and cellular effects of a novel hydroxamate-based HDAC inhibitor—belinostat—in glioblastoma cell lines: A preliminary report. Invest. New Drugs 2016, 34, 552–564. [Google Scholar] [CrossRef] [PubMed]
- Munshi, A.; Hobbs, M.; Meyn, R.E. Clonogenic Cell Survival Assay. In Chemosensitivity; Humana Press: Totowa, NJ, USA, 2005; Volume 110, pp. 021–028. [Google Scholar]
- Liang, C.-C.; Park, A.Y.; Guan, J.-L. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Aumailley, L.; Warren, A.; Garand, C.; Dubois, M.J.; Paquet, E.R.; Le Couteur, D.G.; Marette, A.; Cogger, V.C.; Lebel, M. Vitamin C modulates the metabolic and cytokine profiles, alleviates hepatic endoplasmic reticulum stress, and increases the life span of Gulo-/- mice. Aging (Albany NY) 2016, 8, 458–483. [Google Scholar] [CrossRef] [PubMed]
- Correa-Costa, M.; Braga, T.T.; Semedo, P.; Hayashida, C.Y.; Bechara, L.R.G.; Elias, R.M.; Barreto, C.R.; Silva-Cunha, C.; Hyane, M.I.; Gonçalves, G.M.; et al. Pivotal role of Toll-like receptors 2 and 4, its adaptor molecule MyD88, and inflammasome complex in experimental tubule-interstitial nephritis. PLoS ONE 2011, 6, e29004. [Google Scholar] [CrossRef] [PubMed]
- DeFilippis, R.A.; Goodwin, E.C.; Wu, L.; DiMaio, D. Endogenous human papillomavirus E6 and E7 proteins differentially regulate proliferation, senescence, and apoptosis in HeLa cervical carcinoma cells. J. Virol. 2003, 77, 1551–1563. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K.; Mukherjee, S.; Vanmanen, J.; Banerjee, P.; Fata, J.E. CUNY Graduate Center, College of Staten Island. Unpublished work. 2018. [Google Scholar]
- Pajonk, F.; Pajonk, K.; McBride, W.H. Inhibition of NF-κB, Clonogenicity, and Radiosensitivity of Human Cancer Cells. JNCI J. Natl. Cancer Inst. 1999, 91, 1956–1960. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, H.; Wyckoff, J.; Condeelis, J. Cell migration in tumors. Curr. Opin. Cell Biol. 2005, 17, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Sull, J.W.; Sung, H.J. Suppressing effect of resveratrol on the migration and invasion of human metastatic lung and cervical cancer cells. Mol. Biol. Rep. 2012, 39, 8709–8716. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.-H.; Chiou, Y.-S.; Chen, W.-J.; Wang, J.-M.; Badmaev, V.; Ho, C.-T. Pterostilbene inhibited tumor invasion via suppressing multiple signal transduction pathways in human hepatocellular carcinoma cells. Carcinogenesis 2009, 30, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.P.; Wesierska-Gadek, J. Monitoring of long-term effects of resveratrol on cell cycle progression of human HeLa cells after administration of a single dose. Ann. N. Y. Acad. Sci. 2009, 1171, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Zoberi, I.; Bradbury, C.M.; Curry, H.A.; Bisht, K.S.; Goswami, P.C.; Roti Roti, J.L.; Gius, D. Radiosensitizing and anti-proliferative effects of resveratrol in two human cervical tumor cell lines. Cancer Lett. 2002, 175, 165–173. [Google Scholar] [CrossRef]
- Pan, M.-H.; Chang, Y.-H.; Badmaev, V.; Nagabhushanam, K.; Ho, C.-T. Pterostilbene Induces Apoptosis and Cell Cycle Arrest in Human Gastric Carcinoma Cells. J. Agric. Food Chem. 2007, 55, 7777–7785. [Google Scholar] [CrossRef] [PubMed]
- Veras, E.; Malpica, A.; Deavers, M.T.; Silva, E.G. Mitosis-specific Marker Phospho-histone H3 in the Assessment of Mitotic Index in Uterine Smooth Muscle Tumors: A Pilot Study. Int. J. Gynecol. Pathol. 2009, 28, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, X.-Q.; Chen, H.-Y.; Liu, B.-H. Involvement of the Nrf2 Pathway in the Regulation of Pterostilbene-Induced Apoptosis in HeLa Cells via ER Stress. J. Pharmacol. Sci. 2014, 126, 216–229. [Google Scholar] [CrossRef] [PubMed]
- Yim, E.-K.; Park, J.-S. The role of HPV E6 and E7 oncoproteins in HPV-associated cervical carcinogenesis. Cancer Res. Treat. 2005, 37, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Athar, M.; Back, J.H.; Kopelovich, L.; Bickers, D.R.; Kim, A.L. Multiple molecular targets of resveratrol: Anti-carcinogenic mechanisms. Arch. Biochem. Biophys. 2009, 486, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; McKalip, A.; Herman, B. Human papillomavirus type 16 E6 and HPV-16 E6/E7 sensitize human keratinocytes to apoptosis induced by chemotherapeutic agents: Roles of p53 and caspase activation. J. Cell. Biochem. 2000, 78, 334–349. [Google Scholar] [CrossRef]
- Kapetanovic, I.M.; Muzzio, M.; Huang, Z.; Thompson, T.N.; McCormick, D.L. Pharmacokinetics, oral bioavailability, and metabolic profile of resveratrol and its dimethylether analog, pterostilbene, in rats. Cancer Chemother. Pharmacol. 2011, 68, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Shu, X.-H.; Li, H.; Sun, X.-X.; Wang, Q.; Sun, Z.; Wu, M.-L.; Chen, X.-Y.; Li, C.; Kong, Q.-Y.; Liu, J. Metabolic patterns and biotransformation activities of resveratrol in human glioblastoma cells: Relevance with therapeutic efficacies. PLoS ONE 2011, 6, e27484. [Google Scholar] [CrossRef] [PubMed]
- Gosslau, A.; Chen, M.; Ho, C.-T.; Chen, K.Y. A methoxy derivative of resveratrol analogue selectively induced activation of the mitochondrial apoptotic pathway in transformed fibroblasts. Br. J. Cancer 2005, 92, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Dewi, N.I.; Yagasaki, K.; Miura, Y. Anti-proliferative effect of pterostilbene on rat hepatoma cells in culture. Cytotechnology 2015, 67, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.R.; Scott, E.; Brown, V.A.; Gescher, A.J.; Steward, W.P.; Brown, K. Clinical trials of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 161–169. [Google Scholar] [CrossRef] [PubMed]

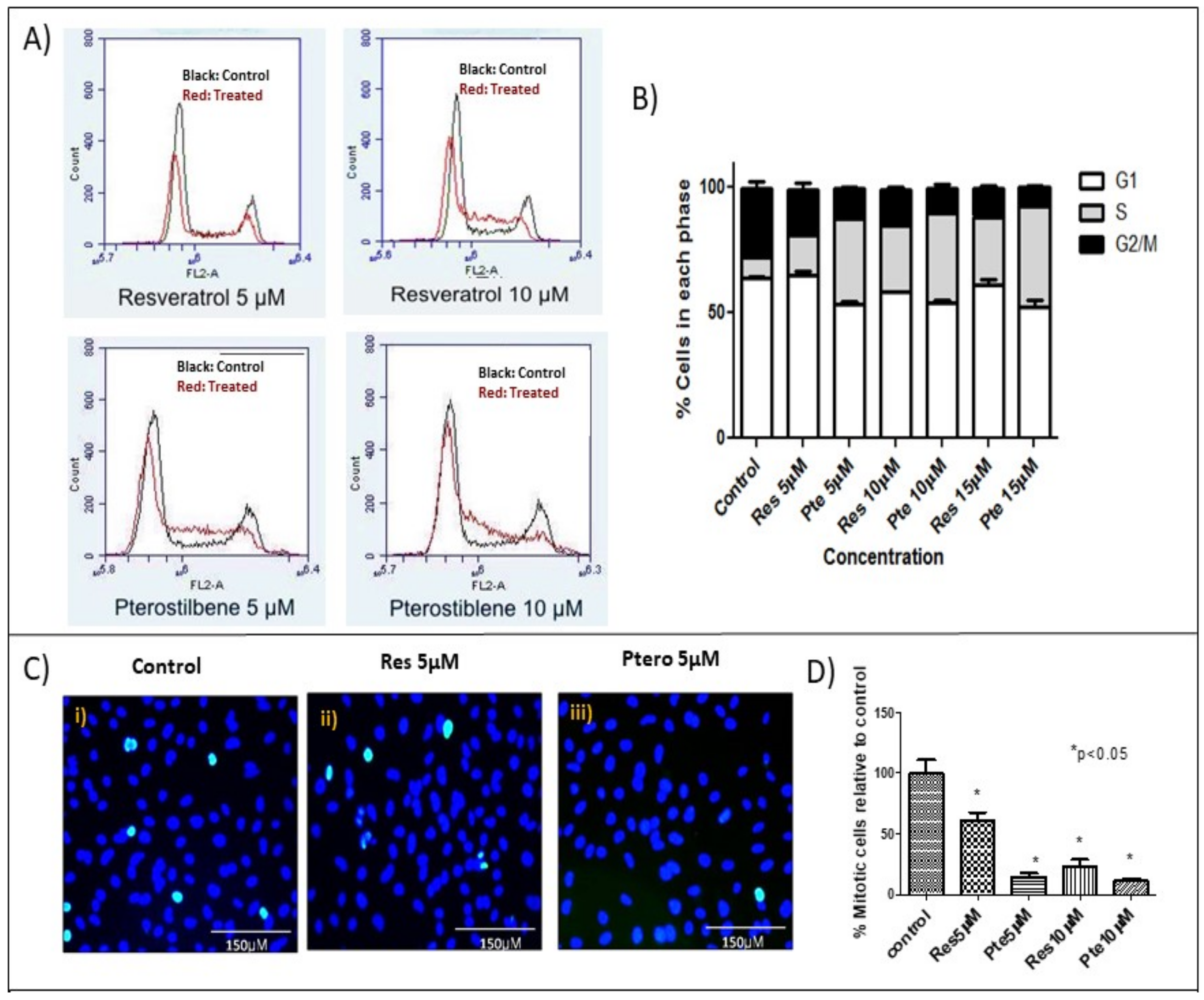
| G1 ± S.E.M. | S ± S.E.M. | G2 ± S.E.M. | |
|---|---|---|---|
| Control | 64.1 ± 0.4 | 8.00 ± 2.5 | 27.7 ± 2.4 |
| Res 5 µM | 64.8 ± 2.0 | 16.3 ± 1.0 | 18.3 ± 2.3 ^ |
| Pte 5 µM | 53.4 ± 1.4 + | 34.0 ± 1.4 * | 12.5 ± 0.2 ^ |
| Res 10 µM | 58.5 ± 0.2 | 26.5 ± 0.2 * | 14.4 ± 1.0 ^ |
| Pte 10 µM | 54.3 ± 0.8 + | 35.6 ± 2.4 * | 10.1 ± 1.5 ^ |
| Res 15 µM | 61.3 ± 1.9 | 27.1 ± 0.8 *,# | 11.5 ± 1.2 ^ |
| Pte 15 µM | 52.3 ± 2.0 + | 40.1 ± 3.4 *,# | 7.7 ± 0.5 ^ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chatterjee, K.; AlSharif, D.; Mazza, C.; Syar, P.; Al Sharif, M.; Fata, J.E. Resveratrol and Pterostilbene Exhibit Anticancer Properties Involving the Downregulation of HPV Oncoprotein E6 in Cervical Cancer Cells. Nutrients 2018, 10, 243. https://doi.org/10.3390/nu10020243
Chatterjee K, AlSharif D, Mazza C, Syar P, Al Sharif M, Fata JE. Resveratrol and Pterostilbene Exhibit Anticancer Properties Involving the Downregulation of HPV Oncoprotein E6 in Cervical Cancer Cells. Nutrients. 2018; 10(2):243. https://doi.org/10.3390/nu10020243
Chicago/Turabian StyleChatterjee, Kaushiki, Dina AlSharif, Christina Mazza, Palwasha Syar, Mohamed Al Sharif, and Jimmie E. Fata. 2018. "Resveratrol and Pterostilbene Exhibit Anticancer Properties Involving the Downregulation of HPV Oncoprotein E6 in Cervical Cancer Cells" Nutrients 10, no. 2: 243. https://doi.org/10.3390/nu10020243
APA StyleChatterjee, K., AlSharif, D., Mazza, C., Syar, P., Al Sharif, M., & Fata, J. E. (2018). Resveratrol and Pterostilbene Exhibit Anticancer Properties Involving the Downregulation of HPV Oncoprotein E6 in Cervical Cancer Cells. Nutrients, 10(2), 243. https://doi.org/10.3390/nu10020243





