The Effect of Branched-Chain Amino Acids, Citrulline, and Arginine on High-Intensity Interval Performance in Young Swimmers
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
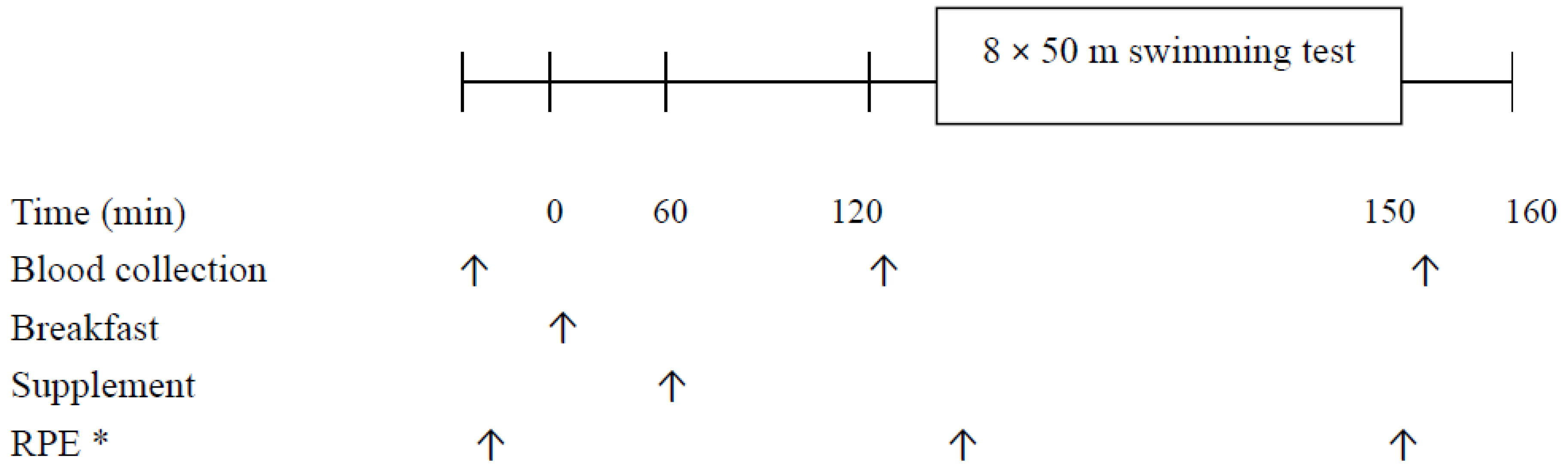
2.2. Study Design
2.3. Dietary Control
2.4. Supplementation
2.5. High-Intensity Interval Swimming Test
2.6. Stroke Characteristics
2.7. Measurement of Blood Biochemical Parameters
2.8. Statistical Analysis
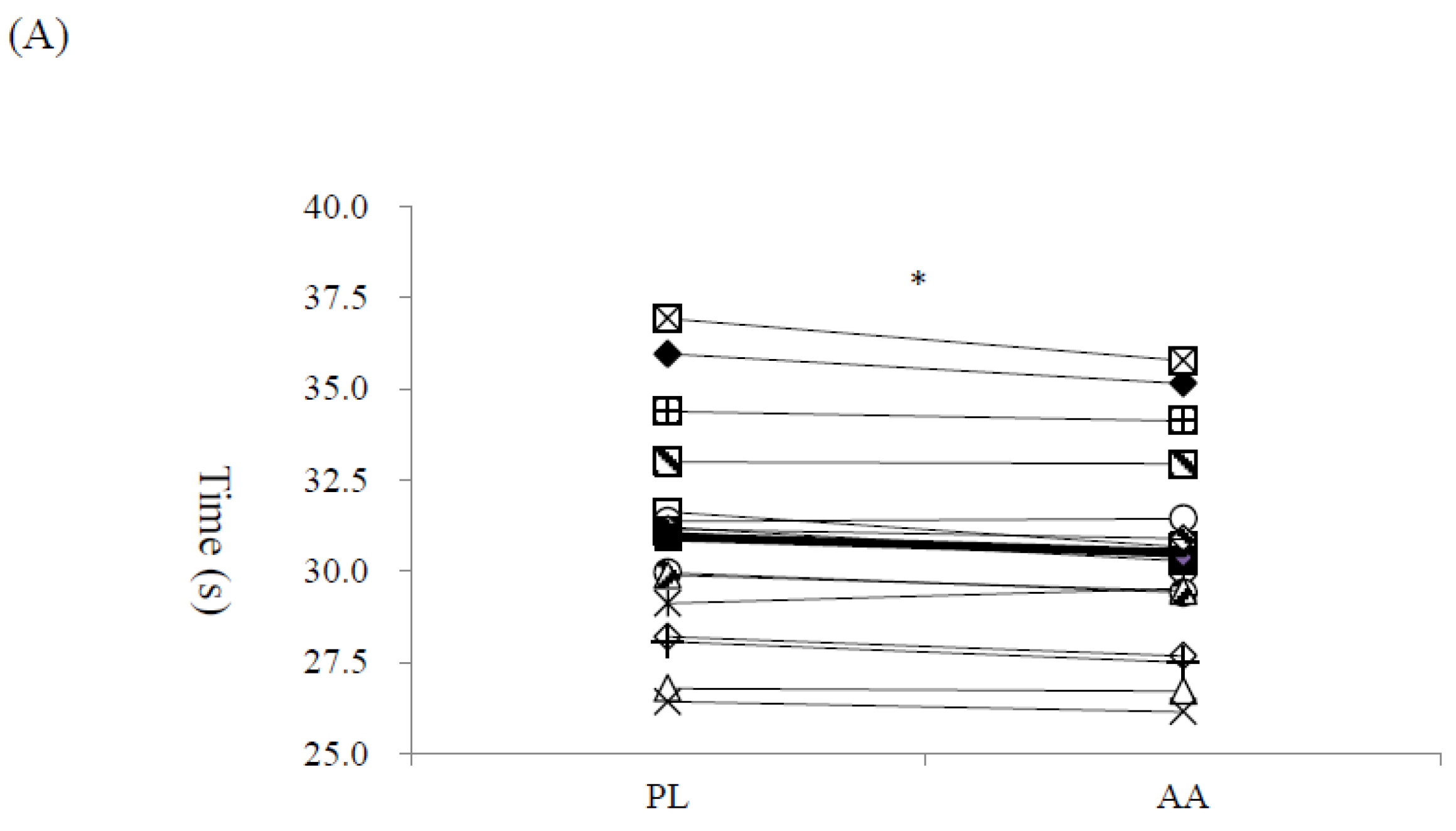
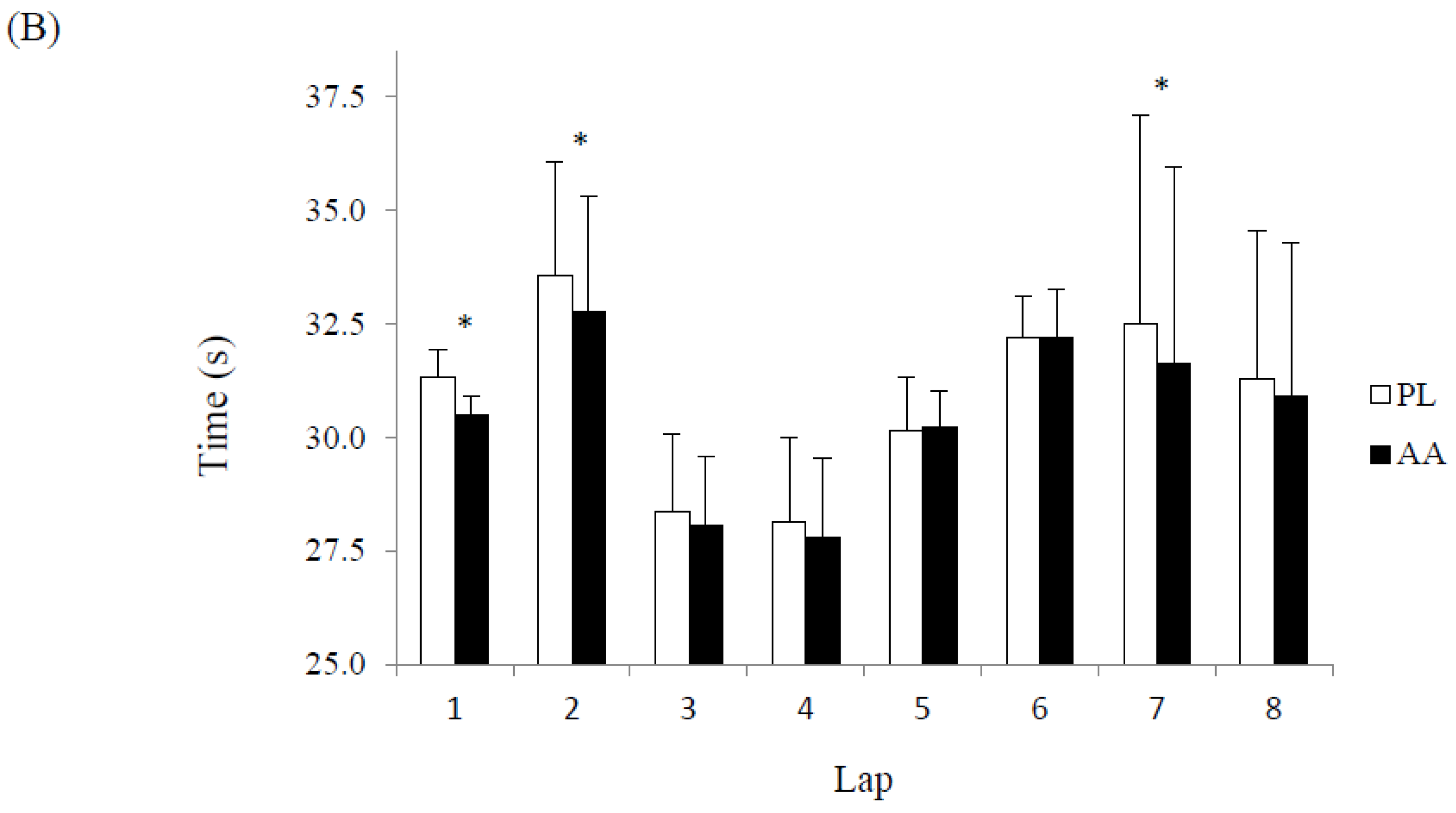
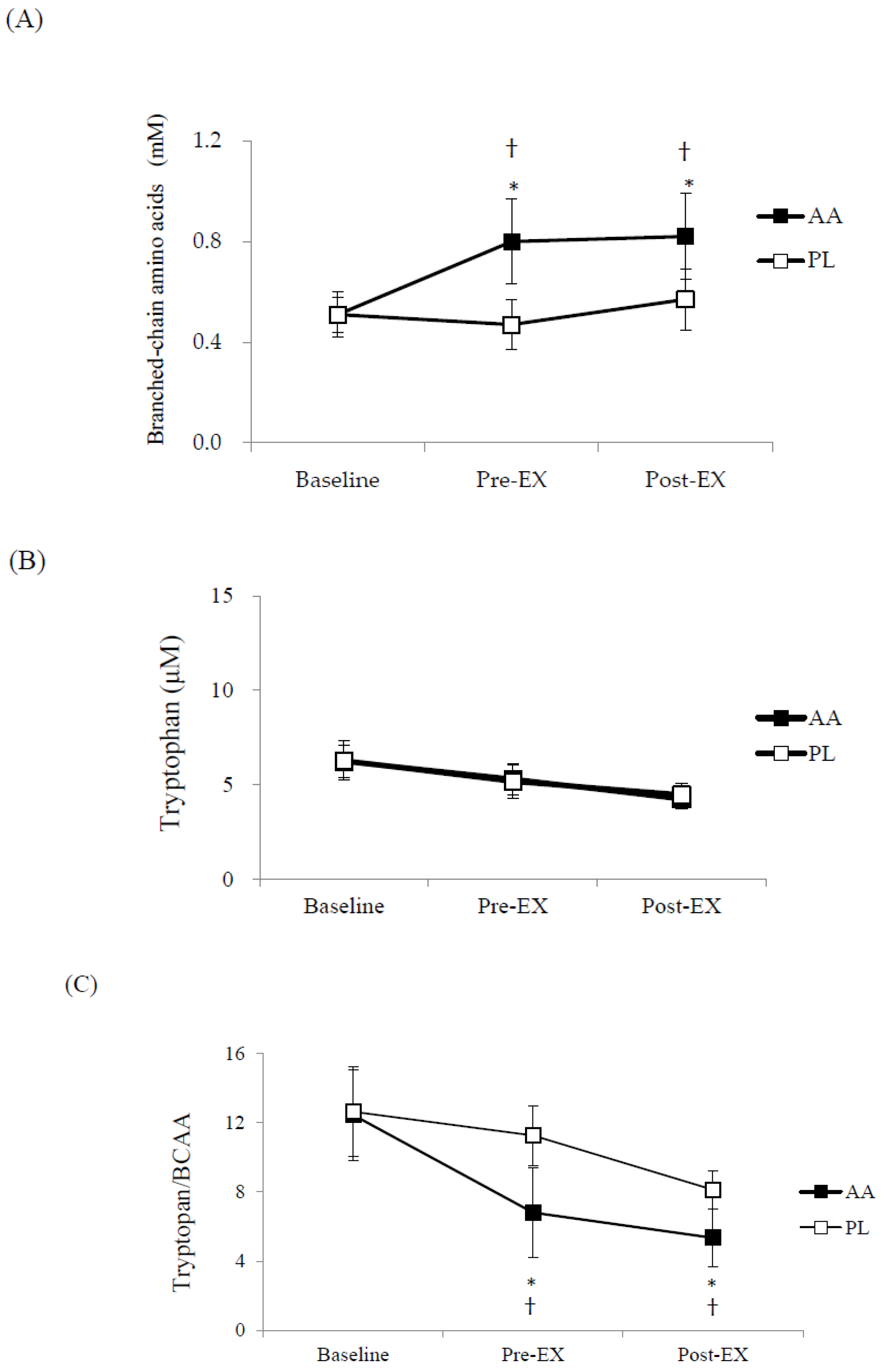
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle: Part I: Cardiopulmonary emphasis. Sports Med. 2013, 43, 313–338. [Google Scholar] [CrossRef] [PubMed]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle. Part II: Anaerobic energy, neuromuscular load and practical applications. Sports Med. 2013, 43, 927–954. [Google Scholar] [CrossRef] [PubMed]
- Altenburg, T.M.; Degens, H.; van Mechelen, W.; Sargeant, A.J.; de Haan, A. Recruitment of single muscle fibers during submaximal cycling exercise. J. Appl. Physiol. (1985) 2007, 103, 1752–1756. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Hermoso, A.; Cerrillo-Urbina, A.J.; Herrera-Valenzuela, T.; Cristi-Montero, C.; Saavedra, J.M.; Martinez-Vizcaino, V. Is high-intensity interval training more effective on improving cardiometabolic risk and aerobic capacity than other forms of exercise in overweight and obese youth? A meta-analysis. Obes. Rev. 2016, 17, 531–540. [Google Scholar] [CrossRef]
- Midgley, A.W.; McNaughton, L.R.; Carroll, S. Reproducibility of time at or near VO2max during intermittent treadmill running. Int. J. Sports Med. 2007, 28, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Faude, O.; Meyer, T.; Scharhag, J.; Weins, F.; Urhausen, A.; Kindermann, W. Volume vs. intensity in the training of competitive swimmers. Int. J. Sports Med. 2008, 29, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Sperlich, B.; Zinner, C.; Heilemann, I.; Kjendlie, P.L.; Holmberg, H.C.; Mester, J. High-intensity interval training improves VO(2peak), maximal lactate accumulation, time trial and competition performance in 9-11-year-old swimmers. Eur. J. Appl. Physiol. 2010, 110, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Spencer, M.; Bishop, D.; Dawson, B.; Goodman, C. Physiological and metabolic responses of repeated-sprint activities:specific to field-based team sports. Sports Med. 2005, 35, 1025–1044. [Google Scholar] [CrossRef] [PubMed]
- Girard, O.; Mendez-Villanueva, A.; Bishop, D. Repeated-sprint ability—Part I: Factors contributing to fatigue. Sports Med. 2011, 41, 673–694. [Google Scholar] [CrossRef]
- Sidhu, S.K.; Bentley, D.J.; Carroll, T.J. Locomotor exercise induces long-lasting impairments in the capacity of the human motor cortex to voluntarily activate knee extensor muscles. J. Appl. Physiol. (1985) 2009, 106, 556–565. [Google Scholar] [CrossRef]
- Davis, J.M.; Bailey, S.P. Possible mechanisms of central nervous system fatigue during exercise. Med. Sci. Sports Exerc. 1997, 29, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Newsholme, E.A.; Blomstrand, E. Branched-chain amino acids and central fatigue. J. Nutr. 2006, 136, 274S–276S. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.D.; Coimbra, C.C.; Marubayashi, U. Tryptophan-induced central fatigue in exercising rats is related to serotonin content in preoptic area. Neurosci. Lett. 2007, 415, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Azechi, H.; Board, M. Essential role of excessive tryptophan and its neurometabolites in fatigue. Can. J. Neurol. Sci. 2012, 39, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Blood-brain barrier carrier-mediated transport and brain metabolism of amino acids. Neurochem. Res. 1998, 23, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Fernstrom, J.D. Branched-chain amino acids and brain function. J. Nutr. 2005, 135, 1539S–1546S. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.D.; Srinivasan, M.; Hogikyan, R.V.; Dengel, D.R.; Glickman, S.G.; Galecki, A.; Supiano, M.A. Nitric oxide biomarkers increase during exercise-induced vasodilation in the forearm. Int. J. Sports Med. 2000, 21, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Vanhatalo, A.; Winyard, P.G.; Jones, A.M. The nitrate-nitrite-nitric oxide pathway: Its role in human exercise physiology. Eur. J. Sport Sci. 2012, 12, 309–320. [Google Scholar] [CrossRef]
- Yavuz, H.U.; Turnagol, H.; Demirel, A.H. Pre-exercise arginine supplementation increases time to exhaustion in elite male wrestlers. Biol. Sport 2014, 31, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Blackwell, J.R.; Lord, T.; Vanhatalo, A.; Winyard, P.G.; Jones, A.M. l-Citrulline supplementation improves O2 uptake kinetics and high-intensity exercise performance in humans. J. Appl. Physiol. (1985) 2015, 119, 385–395. [Google Scholar] [CrossRef]
- Chang, C.K.; Chang Chien, K.M.; Chang, J.H.; Huang, M.H.; Liang, Y.C.; Liu, T.H. Branched-chain amino acids and arginine improve performance in two consecutive days of simulated handball games in male and female athletes: A randomized trial. PLoS ONE 2015, 10, e0121866. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.C.; Wu, C.L.; Chen, I.F.; Chang, C.K. Prevention of perceptual-motor decline by branched-chain amino acids, arginine, citrulline after tennis match. Scand. J. Med. Sci. Sports 2017, 29, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.F.; Wu, H.J.; Chen, C.Y.; Chou, K.M.; Chang, C.K. Branched-chain amino acids, arginine, citrulline alleviate central fatigue after 3 simulated matches in taekwondo athletes: A randomized controlled trial. J. Int. Soc. Sports Nutr. 2016, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Morita, M.; Hayashi, T.; Kamimura, A. The effects on plasma L-arginine levels of combined oral L-citrulline and L-arginine supplementation in healthy males. Biosci. Biotechnol. Biochem. 2016, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Naderi, A.; Earnest, C.P.; Lowery, R.P.; Wilson, J.M.; Willems, M.E. Co-ingestion of Nutritional Ergogenic Aids and High-Intensity Exercise Performance. Sports Med. 2016, 46, 1407–1418. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Costill, D.L.; Fink, W.J. Plasma volume changes following exercise and thermal dehydration. J. Appl. Physiol. 1974, 37, 521–525. [Google Scholar] [CrossRef]
- Atkinson, G. Analysis of repeated measurements in physical therapy research: Multiple comparisons amongst level means and multi-factorial designs. Phys. Ther. Sport 2002, 3, 191–203. [Google Scholar] [CrossRef]
- Tan, X.R.; Low, I.C.C.; Stephenson, M.C.; Soong, T.W.; Lee, J.K.W. Neural Basis of Exertional Fatigue in the Heat: A Review of Magnetic Resonance Imaging Methods. Scand. J. Med. Sci. Sports 2017. [CrossRef]
- Benwell, N.M.; Mastaglia, F.L.; Thickbroom, G.W. Changes in the functional MR signal in motor and non-motor areas during intermittent fatiguing hand exercise. Exp. Brain Res. 2007, 182, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Blomstrand, E.; Hassmen, P.; Ek, S.; Ekblom, B.; Newsholme, E.A. Influence of ingesting a solution of branched-chain amino acids on perceived exertion during exercise. Acta Physiol. Scand. 1997, 159, 41–49. [Google Scholar] [CrossRef]
- Cheng, I.S.; Wang, Y.W.; Chen, I.F.; Hsu, G.S.; Hsueh, C.F.; Chang, C.K. The supplementation of branched-chain amino acids, arginine, and citrulline improves endurance exercise performance in two consecutive days. J. Sports Sci. Med. 2016, 15, 509–515. [Google Scholar] [PubMed]
- Perez-Guisado, J.; Jakeman, P.M. Citrulline malate enhances athletic anaerobic performance and relieves muscle soreness. J. Strength Cond. Res. 2010, 24, 1215–1222. [Google Scholar] [CrossRef] [PubMed]
- Cuenca, E.; Jodra, P.; Perez-Lopez, A.; Gonzalez-Rodriguez, L.G.; Fernandes da Silva, S.; Veiga-Herreros, P.; Dominguez, R. Effects of Beetroot Juice Supplementation on Performance and Fatigue in a 30-s All-Out Sprint Exercise: A Randomized, Double-Blind Cross-Over Study. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Dominguez, R.; Mate-Munoz, J.L.; Cuenca, E.; Garcia-Fernandez, P.; Mata-Ordonez, F.; Lozano-Estevan, M.C.; Veiga-Herreros, P.; da Silva, S.F.; Garnacho-Castano, M.V. Effects of beetroot juice supplementation on intermittent high-intensity exercise efforts. J. Int. Soc. Sports Nutr. 2018, 15, 2. [Google Scholar] [CrossRef]
- Peyrebrune, M.C.; Toubekis, A.G.; Lakomy, H.K.; Nevill, M.E. Estimating the energy contribution during single and repeated sprint swimming. Scand. J. Med. Sci. Sports 2014, 24, 369–376. [Google Scholar] [CrossRef]
- Peyrebrune, M.C.; Nevill, M.E.; Donaldson, F.J.; Cosford, D.J. The effects of oral creatine supplementation on performance in single and repeated sprint swimming. J. Sports Sci. 1998, 16, 271–279. [Google Scholar] [CrossRef]
- Cunniffe, B.; Papageorgiou, M.; O’Brien, B.; Davies, N.A.; Grimble, G.K.; Cardinale, M. Acute Citrulline-Malate Supplementation and High-Intensity Cycling Performance. J. Strength Cond. Res. 2016, 30, 2638–2647. [Google Scholar] [CrossRef]
- Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; DiMenna, F.J.; Pavey, T.G.; Wilkerson, D.P.; Benjamin, N.; Winyard, P.G.; Jones, A.M. Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R1121–R1131. [Google Scholar] [CrossRef]
- Glenn, J.M.; Gray, M.; Jensen, A.; Stone, M.S.; Vincenzo, J.L. Acute citrulline-malate supplementation improves maximal strength and anaerobic power in female, masters athletes tennis players. Eur. J. Sport Sci. 2016, 16, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.J.; Weitzberg, E.; Lundberg, J.O.; Ekblom, B. Dietary nitrate reduces maximal oxygen consumption while maintaining work performance in maximal exercise. Free Radic. Biol. Med. 2010, 48, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Kilen, A.; Larsson, T.H.; Jorgensen, M.; Johansen, L.; Jorgensen, S.; Nordsborg, N.B. Effects of 12 weeks high-intensity & reduced-volume training in elite athletes. PLoS ONE 2014, 9, e95025. [Google Scholar] [CrossRef]
- Maglischo, E.W. Swimming Fastest; Human Kinetics: Champaign, IL, USA, 2003. [Google Scholar]
- Theodorou, A.S.; Havenetidis, K.; Zanker, C.L.; O’Hara, J.P.; King, R.F.; Hood, C.; Paradisis, G.; Cooke, C.B. Effects of acute creatine loading with or without carbohydrate on repeated bouts of maximal swimming in high-performance swimmers. J. Strength Cond. Res. 2005, 19, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Toubekis, A.G.; Adam, G.V.; Douda, H.T.; Antoniou, P.D.; Douroundos, I.I.; Tokmakidis, S.P. Repeated sprint swimming performance after low- or high-intensity active and passive recoveries. J. Strength Cond. Res. 2011, 25, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Avlonitou, E. Maximal lactate values following competitive performance varying according to age, sex and swimming style. J. Sports Med. Phys. Fitness 1996, 36, 24–30. [Google Scholar]
- Craig, A.B., Jr.; Pendergast, D.R. Relationships of stroke rate, distance per stroke, and velocity in competitive swimming. Med. Sci. Sports 1979, 11, 278–283. [Google Scholar] [CrossRef]
- Craig, A.B., Jr.; Skehan, P.L.; Pawelczyk, J.A.; Boomer, W.L. Velocity, stroke rate, and distance per stroke during elite swimming competition. Med. Sci. Sports Exerc. 1985, 17, 625–634. [Google Scholar] [CrossRef]
| Gender * | Age (Year) | Height (m) | Weight (kg) | Body Mass Index | Best Style | Personal Best † 50 m (s) | Personal Best † 100 m (s) |
|---|---|---|---|---|---|---|---|
| M | 15 | 1.78 | 67.1 | 21.18 | Backstroke | 29.8 | 62.2 |
| M | 17 | 1.78 | 71.1 | 22.44 | Breaststroke | 31.2 | 69.1 |
| M | 17 | 1.77 | 70.2 | 22.41 | Front crawl | 25.7 | 54.9 |
| M | 17 | 1.78 | 70.5 | 22.25 | Front crawl | 25.6 | 52.7 |
| M | 15 | 1.69 | 53.6 | 18.77 | Front crawl | 27.3 | 56.2 |
| M | 14 | 1.68 | 52.6 | 18.64 | Butterfly | 28.9 | 57.3 |
| M | 16 | 1.69 | 68.8 | 24.09 | Butterfly | 26.8 | 57.3 |
| M | 14 | 1.71 | 61.1 | 20.90 | Front crawl | 26.2 | 54.8 |
| Mean ‡ | 15.6 | 1.74 | 64.4 | 21.33 | |||
| SD ‡ | 1.3 | 0.05 | 7.6 | 1.89 | |||
| F | 16 | 1.46 | 46.3 | 21.72 | Front crawl | 29.7 | 65.2 |
| F | 15 | 1.59 | 45.4 | 17.96 | Front crawl | 29.8 | 66.1 |
| F | 17 | 1.60 | 51.4 | 20.08 | Front crawl | 28.2 | 59.9 |
| F | 16 | 1.64 | 60.9 | 22.64 | Front crawl | 28.0 | 61.2 |
| F | 15 | 1.53 | 44.1 | 18.84 | Front crawl | 29.5 | 61.1 |
| F | 16 | 1.58 | 56.5 | 22.63 | Butterfly | 31.3 | 69.2 |
| F | 16 | 1.65 | 60.9 | 22.37 | Butterfly | 33.5 | 71.8 |
| F | 14 | 1.60 | 66.6 | 26.02 | Butterfly | 31.7 | 68.9 |
| Mean § | 15.6 | 1.58 | 54.0 | 21.53 | |||
| SD § | 0.9 | 0.06 | 8.4 | 2.55 |
| Trial | Baseline | Pre-Ex | Post-Ex | |
|---|---|---|---|---|
| NOx (μM) | AA | 11.61 ± 4.94 | 31.65 ± 29.56 | 23.06 ± 15.28 |
| PL | 12.68 ± 7.42 | 25.84 ± 29.39 | 18.36 ± 11.71 | |
| NH3 (μM) | AA | 115.20 ± 103.49 | 123.72 ± 66.29 | 123.86 ± 65.35 |
| PL | 116.59 ± 40.69 | 109.02 ± 50.89 | 114.81 ± 50.31 | |
| Urea (mM) | AA | 4.64 ± 0.77 | 5.38 ± 0.90 | 5.11 ± 0.91 |
| PL | 4.77 ± 0.65 | 5.01 ± 0.87 | 4.63 ± 0.73 | |
| Lactate (mM) | AA | 1.36 ± 0.56 | 1.91 ± 0.59 | 14.75 ± 4.43 *,† |
| PL | 1.43 ± 0.34 | 1.71 ± 0.38 | 14.12 ± 2.79 *,† | |
| Glycerol (μM) | AA | 46.00 ± 21.00 | 81.89 ± 28.83 * | 240.89 ± 96.61 *,† |
| PL | 44.63 ± 25.27 | 71.35 ± 26.45 | 227.44 ± 58.36 *,† | |
| NEFA (mM) 1 | AA | 0.49 ± 0.27 | 0.42 ± 0.16 | 0.22 ± 0.09 † |
| PL | 0.44 ± 0.25 | 0.38 ± 0.10 | 0.20 ± 0.08 † | |
| RPE 2 | AA | 9.1 ± 2.4 | 15.0 ± 1.8 * | 17.2 ± 2.3 * |
| PL | 9.7 ± 2.4 | 14.8 ± 2.2 * | 17.3 ± 1.8 * |
| Trial | Lap | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | Mean | ||
| Stroke rate 1 (count/min) | PL | 70.6 ± 19.5 | 71.2 ± 19.3 | 72.3 ± 19.0 | 71.4 ± 20.2 | 70.2 ± 18.7 | 69.2 ± 18.5 | 69.4 ± 18.2 | 69.4 ± 18.3 | 70.5 ± 19.0 |
| AA | 74.5 ± 18.5 | 76.0 ± 18.3 | 72.7 ± 19.5 | 72.4 ± 18.4 | 70.7 ± 16.5 | 71.4 ± 18.0 | 70.1± 17.4 | 71.9 ± 18.2 | 72.7 ± 18.1 | |
| Stroke count 2 | PL | 30.7 ± 9.0 a,b | 30.6 ± 9.5 a,b | 30.8 ± 9.1 a | 30.8 ± 9.2 a,b | 31.1 ± 9.4 a,b | 31.7 ± 9.5 a,b | 31.6 ± 9.4 a,b | 31.9 ± 9.0 b | 31.1 ± 9.3 |
| (count/lap) | AA | 31.1 ± 8.5 a,b | 30.6 ± 8.2 a,b | 30.6 ± 8.2 a | 31.1 ± 8.6 a | 31.2 ± 8.3 a,b | 31.3 ± 8.1 a,b | 31.5 ± 8.4 a,b | 32.1 ± 8.4 b | 31.2 ± 8.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsueh, C.-F.; Wu, H.-J.; Tsai, T.-S.; Wu, C.-L.; Chang, C.-K. The Effect of Branched-Chain Amino Acids, Citrulline, and Arginine on High-Intensity Interval Performance in Young Swimmers. Nutrients 2018, 10, 1979. https://doi.org/10.3390/nu10121979
Hsueh C-F, Wu H-J, Tsai T-S, Wu C-L, Chang C-K. The Effect of Branched-Chain Amino Acids, Citrulline, and Arginine on High-Intensity Interval Performance in Young Swimmers. Nutrients. 2018; 10(12):1979. https://doi.org/10.3390/nu10121979
Chicago/Turabian StyleHsueh, Chun-Fang, Huey-June Wu, Tzu-Shiou Tsai, Ching-Lin Wu, and Chen-Kang Chang. 2018. "The Effect of Branched-Chain Amino Acids, Citrulline, and Arginine on High-Intensity Interval Performance in Young Swimmers" Nutrients 10, no. 12: 1979. https://doi.org/10.3390/nu10121979
APA StyleHsueh, C.-F., Wu, H.-J., Tsai, T.-S., Wu, C.-L., & Chang, C.-K. (2018). The Effect of Branched-Chain Amino Acids, Citrulline, and Arginine on High-Intensity Interval Performance in Young Swimmers. Nutrients, 10(12), 1979. https://doi.org/10.3390/nu10121979





