Selenium Status Is Not Associated with Cognitive Performance: A Cross-Sectional Study in 154 Older Australian Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Health and Demographics
2.4. Biological Samples
2.5. Plasma Selenium
2.6. Inflammatory and Neurotrophic Markers
2.7. Cognitive Function
2.8. Physical Activity
2.9. Anthropometry
2.10. Selenium Intake
2.11. APOE Genotype
2.12. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Harada, C.N.; Natelson Love, M.C.; Triebel, K. Normal cognitive aging. Clin. Geriatr. Med. 2013, 29, 737–752. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer’s Association. 2018 alzheimer’s disease facts and figures. Alzheimers Dement. 2018, 14, 367–429. [Google Scholar] [CrossRef]
- De Bruijn, R.F.; Bos, M.J.; Portegies, M.L.; Hofman, A.; Franco, O.H.; Koudstaal, P.J.; Ikram, M.A. The potential for prevention of dementia across two decades: The prospective, population-based rotterdam study. BMC Med. 2015, 13, 132. [Google Scholar] [CrossRef] [PubMed]
- Ashby-Mitchell, K.; Burns, R.; Shaw, J.; Anstey, K.J. Proportion of dementia in australia explained by common modifiable risk factors. Alzheimers Res. Ther. 2017, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; de la Torre, R.; Martínez-González, M.Á.; Martínez-Lapiscina, E.H.; Fitó, M.; Pérez-Heras, A.; Salas-Salvadó, J.; et al. Mediterranean diet and age-related cognitive decline: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Barnes, L.L.; Bennett, D.A.; Aggarwal, N.T. Mind diet slows cognitive decline with aging. Alzheimers Dement. 2015, 11, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Roberts, B.R.; Bush, A.I.; Hare, D.J. Selenium, selenoproteins and neurodegenerative diseases. Metallomics 2015, 7, 1213–1228. [Google Scholar] [CrossRef] [PubMed]
- Abedelhaffez, A.S.; Hassan, A. Brain derived neurotrophic factor and oxidative stress index in pups with developmental hypothyroidism: Neuroprotective effects of selenium. Acta Physiol. Hung. 2013, 100, 197–210. [Google Scholar] [CrossRef] [PubMed]
- Nazıroğlu, M.; Senol, N.; Ghazizadeh, V.; Yürüker, V. Neuroprotection induced by n-acetylcysteine and selenium against traumatic brain injury-induced apoptosis and calcium entry in hippocampus of rat. Cell Mol. Neurobiol. 2014, 34, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.L.; Mendelev, N.; Kumari, S.; Andy Li, P. Overexpression of human selenoprotein h in neuronal cells enhances mitochondrial biogenesis and function through activation of protein kinase a, protein kinase b, and cyclic adenosine monophosphate response element-binding protein pathway. Int. J. Biochem. Cell Biol. 2013, 45, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Mendelev, N.; Mehta, S.L.; Witherspoon, S.; He, Q.; Sexton, J.Z.; Li, P.A. Upregulation of human selenoprotein h in murine hippocampal neuronal cells promotes mitochondrial biogenesis and functional performance. Mitochondrion 2011, 11, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Mehta, S.L.; Li, P.A. Glutamate induces mitochondrial dynamic imbalance and autophagy activation: Preventive effects of selenium. PLoS ONE 2012, 7, e39382. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Hare, D.J.; Bush, A.I.; Roberts, B.R. Glutathione peroxidase 4: A new player in neurodegeneration? Mol. Psychiatry 2017, 22, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef] [PubMed]
- Pitts, M.W.; Kremer, P.M.; Hashimoto, A.C.; Torres, D.J.; Byrns, C.N.; Williams, C.S.; Berry, M.J. Competition between the brain and testes under selenium-compromised conditions: Insight into sex differences in selenium metabolism and risk of neurodevelopmental disease. J. Neurosci. 2015, 35, 15326–15338. [Google Scholar] [CrossRef] [PubMed]
- Dregan, A.; Chowienczyk, P.; Gulliford, M.C. Are inflammation and related therapy associated with all-cause dementia in a primary care population? J. Alzheimers Dis. 2015, 46, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- McGeer, P.L.; Rogers, J.; McGeer, E.G. Inflammation, antiinflammatory agents, and alzheimer’s disease: The last 22 years. J. Alzheimers Dis. 2016, 54, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Liu, G.A.; Perez, E.; Rainer, R.D.; Febo, M.; Cruz-Almeida, Y.; Ebner, N.C. Systemic inflammation mediates age-related cognitive deficits. Front. Aging Neurosci. 2018, 10, 236. [Google Scholar] [CrossRef] [PubMed]
- Savaskan, N.E.; Brauer, A.U.; Kuhbacher, M.; Eyupoglu, I.Y.; Kyriakopoulos, A.; Ninnemann, O.; Behne, D.; Nitsch, R. Selenium deficiency increases susceptibility to glutamate-induced excitotoxicity. Faseb. J. 2003, 17, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, K.S.; Zamamiri-Davis, F.; Stewart, J.B.; Thompson, J.T.; Sordillo, L.M.; Reddy, C.C. Selenium deficiency increases the expression of inducible nitric oxide synthase in raw 264.7 macrophages: Role of nuclear factor-kappab in up-regulation. Biochem. J. 2002, 366, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Christensen, M.J.; Nartey, E.T.; Hada, A.L.; Legg, R.L.; Barzee, B.R. High selenium reduces nf-kappab-regulated gene expression in uninduced human prostate cancer cells. Nutr. Cancer 2007, 58, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.; Méplan, C.; Gautrey, H.; Hall, J.; Hesketh, J.E. Differential effects of selenium and knock-down of glutathione peroxidases on tnfα and flagellin inflammatory responses in gut epithelial cells. Genes Nutr. 2012, 7, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, L.F.; Stockler-Pinto, M.B.; Mafra, D. Brazil nut consumption modulates nrf2 expression in hemodialysis patients: A pilot study. Mol. Nutr. Food Res. 2016, 60, 1719–1724. [Google Scholar] [CrossRef] [PubMed]
- Stockler-Pinto, M.B.; Mafra, D.; Moraes, C.; Lobo, J.; Boaventura, G.T.; Farage, N.E.; Silva, W.S.; Cozzolino, S.F.; Malm, O. Brazil nut (bertholletia excelsa, h.B.K.) improves oxidative stress and inflammation biomarkers in hemodialysis patients. Biol. Trace Elem. Res. 2014, 158, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Jin, Y.; Hall, K.S.; Liang, C.; Unverzagt, F.W.; Ji, R.; Murrell, J.R.; Cao, J.; Shen, J.; Ma, F.; et al. Selenium level and cognitive function in rural elderly Chinese. Am. J. Epidemiol. 2007, 165, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.S.; Bukke, S.; Dutt, N.; Rana, P.; Pandey, A.K. A systematic review and meta-analysis of the circulatory, erythrocellular and csf selenium levels in alzheimer’s disease: A metal meta-analysis (amma study-i). J. Trace Elem. Med. Biol. 2017, 42, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Hare, D.J.; Lind, M.; McLean, C.A.; Volitakis, I.; Laws, S.M.; Masters, C.L.; Bush, A.I.; Roberts, B.R. The apoe ε4 allele is associated with lower selenium levels in the brain: Implications for alzheimer’s disease. ACS Chem. Neurosci. 2017, 8, 1459–1464. [Google Scholar] [CrossRef] [PubMed]
- Berr, C.; Balansard, B.; Arnaud, J.; Roussel, A.M.; Alperovitch, A. Cognitive decline is associated with systemic oxidative stress: The eva study. Etude du vieillissement arteriel. J. Am. Geriatr. Soc. 2000, 48, 1285–1291. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.D. Selenium and iodine intakes and status in new zealand and australia. Br. J. Nutr. 2004, 91, 661–672. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.; Colebourne, K.; Faddy, H.M.; Flower, R.; Fraser, J.F. Plasma selenium status in a group of australian blood donors and fresh blood components. J. Trace Elem. Med. Biol. 2013, 27, 352–354. [Google Scholar] [CrossRef] [PubMed]
- Daly, R.M.; Gianoudis, J.; Prosser, M.; Kidgell, D.; Ellis, K.A.; O’Connell, S.; Nowson, C.A. The effects of a protein enriched diet with lean red meat combined with a multi-modal exercise program on muscle and cognitive health and function in older adults: Study protocol for a randomised controlled trial. Trials 2015, 16, 339. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, J.; Yesavage, J. Geriatric depression scale (gds) recent evidence and development of a shorter version. In Clinical Gerontology: A Guide to Assessment And Intervention; The Haworth Press: New York, NY, USA, 1986; pp. 165–173. [Google Scholar]
- Pfeiffer, E. A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients. J. Am. Geriatr. Soc. 1975, 23, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Hare, D.J.; Bush, A.I.; Li, Q.X.; Fowler, C.J.; Masters, C.L.; Martins, R.N.; Ganio, K.; Lothian, A.; Mukherjee, S.; et al. Selenium levels in serum, red blood cells, and cerebrospinal fluid of alzheimer’s disease patients: A report from the australian imaging, biomarker & lifestyle flagship study of ageing (aibl). J. Alzheimers Dis. 2017, 57, 183–193. [Google Scholar] [PubMed]
- Maruff, P.; Lim, Y.Y.; Darby, D.; Ellis, K.A.; Pietrzak, R.H.; Snyder, P.J.; Bush, A.I.; Szoeke, C.; Schembri, A.; Ames, D.; et al. Clinical utility of the cogstate brief battery in identifying cognitive impairment in mild cognitive impairment and alzheimer’s disease. BMC Psychol. 2013, 1, 30. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.Y.; Ellis, K.A.; Harrington, K.; Ames, D.; Martins, R.N.; Masters, C.L.; Rowe, C.; Savage, G.; Szoeke, C.; Darby, D.; et al. Use of the cogstate brief battery in the assessment of alzheimer’s disease related cognitive impairment in the australian imaging, biomarkers and lifestyle (aibl) study. J. Clin. Exp. Neuropsychol. 2012, 34, 345–358. [Google Scholar] [CrossRef] [PubMed]
- Maruff, P.; Thomas, E.; Cysique, L.; Brew, B.; Collie, A.; Snyder, P.; Pietrzak, R.H. Validity of the cogstate brief battery: Relationship to standardized tests and sensitivity to cognitive impairment in mild traumatic brain injury, schizophrenia, and aids dementia complex. Arch. Clin. Neuropsychol. 2009, 24, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Baron, I.S. Test review: Behavior rating inventory of executive function. Child Neuropsychol. 2000, 6, 4. [Google Scholar]
- Stewart, A.L.; Mills, K.M.; King, A.C.; Haskell, W.L.; Gillis, D.; Ritter, P.L. Champs physical activity questionnaire for older adults: Outcomes for interventions. Med. Sci. Sports Exerc. 2001, 33, 1126–1141. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic: Report of a Who Consultation; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Australian National Health and Medical Research Council (NHMRC); New Zealand Ministry of Health (MoH). Nutrient Reference Values for Australia and New Zealand—Including Recommended Dietary Intakes; Commonwealth of Australia: Camberra, Australia, 2006.
- Rawle, M.J.; Davis, D.; Bendayan, R.; Wong, A.; Kuh, D.; Richards, M. Apolipoprotein-e (apoe) ε4 and cognitive decline over the adult life course. Transl. Psychiatry 2018, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.D. Assessment of requirements for selenium and adequacy of selenium status: A review. Eur. J. Clin. Nutr. 2004, 58, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Fayet-Moore, F.; Petocz, P.; Samman, S. Micronutrient status in female university students: Iron, zinc, copper, selenium, vitamin b12 and folate. Nutrients 2014, 6, 5103–5116. [Google Scholar] [CrossRef] [PubMed]
- Fardy, J.J.; McOrist, G.D.; Farrar, Y.J. The determination of selenium status in the australian diet using neutron activation analysis. J. Radioanal. Nuclear Chem. 1989, 133, 397–405. [Google Scholar] [CrossRef]
- Hurst, R.; Collings, R.; Harvey, L.J.; King, M.; Hooper, L.; Bouwman, J.; Gurinovic, M.; Fairweather-Tait, S.J. Eurreca-estimating selenium requirements for deriving dietary reference values. Crit. Rev. Food Sci. Nutr. 2013, 53, 1077–1096. [Google Scholar] [CrossRef] [PubMed]
- Shahar, A.; Patel, K.V.; Semba, R.D.; Bandinelli, S.; Shahar, D.R.; Ferrucci, L.; Guralnik, J.M. Plasma selenium is positively related to performance in neurological tasks assessing coordination and motor speed. Mov. Disord. 2010, 25, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Kryscio, R.J.; Abner, E.L.; Caban-Holt, A.; Lovell, M.; Goodman, P.; Darke, A.K.; Yee, M.; Crowley, J.; Schmitt, F.A. Association of antioxidant supplement use and dementia in the prevention of alzheimer’s disease by vitamin e and selenium trial (preadvise). JAMA Neurol. 2017, 74, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Apolinario, D.; da Silva Bandeira, V.; Busse, A.L.; Magaldi, R.M.; Jacob-Filho, W.; Cozzolino, S.M. Effects of brazil nut consumption on selenium status and cognitive performance in older adults with mild cognitive impairment: A randomized controlled pilot trial. Eur. J. Nutr. 2016, 55, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F., Jr. Biomarkers of selenium status. Nutrients 2015, 7, 2209–2236. [Google Scholar] [CrossRef] [PubMed]
- Cornejo, F.; von Bernhardi, R. Age-dependent changes in the activation and regulation of microglia. In Glial Cells in Health and Disease of the CNS; von Bernhardi, R., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 205–226. [Google Scholar]
- Zhang, S.; Rocourt, C.; Cheng, W.-H. Selenoproteins and the aging brain. Mech. Ageing Dev. 2010, 131, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Stranahan, A.M.; Mattson, M.P. Metabolic reserve as a determinant of cognitive aging. J. Alzheimers Dis. 2012, 30, S5–S13. [Google Scholar] [CrossRef] [PubMed]
- Solovyev, N.; Drobyshev, E.; Bjorklund, G.; Dubrovskii, Y.; Lysiuk, R.; Rayman, M.P. Selenium, selenoprotein p, and alzheimer’s disease: Is there a link? Free Radic. Biol. Med. 2018, 127, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Lewczuk, P.; Riederer, P.; O’Bryant, S.E.; Verbeek, M.M.; Dubois, B.; Visser, P.J.; Jellinger, K.A.; Engelborghs, S.; Ramirez, A.; Parnetti, L.; et al. Cerebrospinal fluid and blood biomarkers for neurodegenerative dementias: An update of the consensus of the task force on biological markers in psychiatry of the world federation of societies of biological psychiatry. World J. Biol. Psychiatry 2018, 19, 244–328. [Google Scholar] [CrossRef] [PubMed]
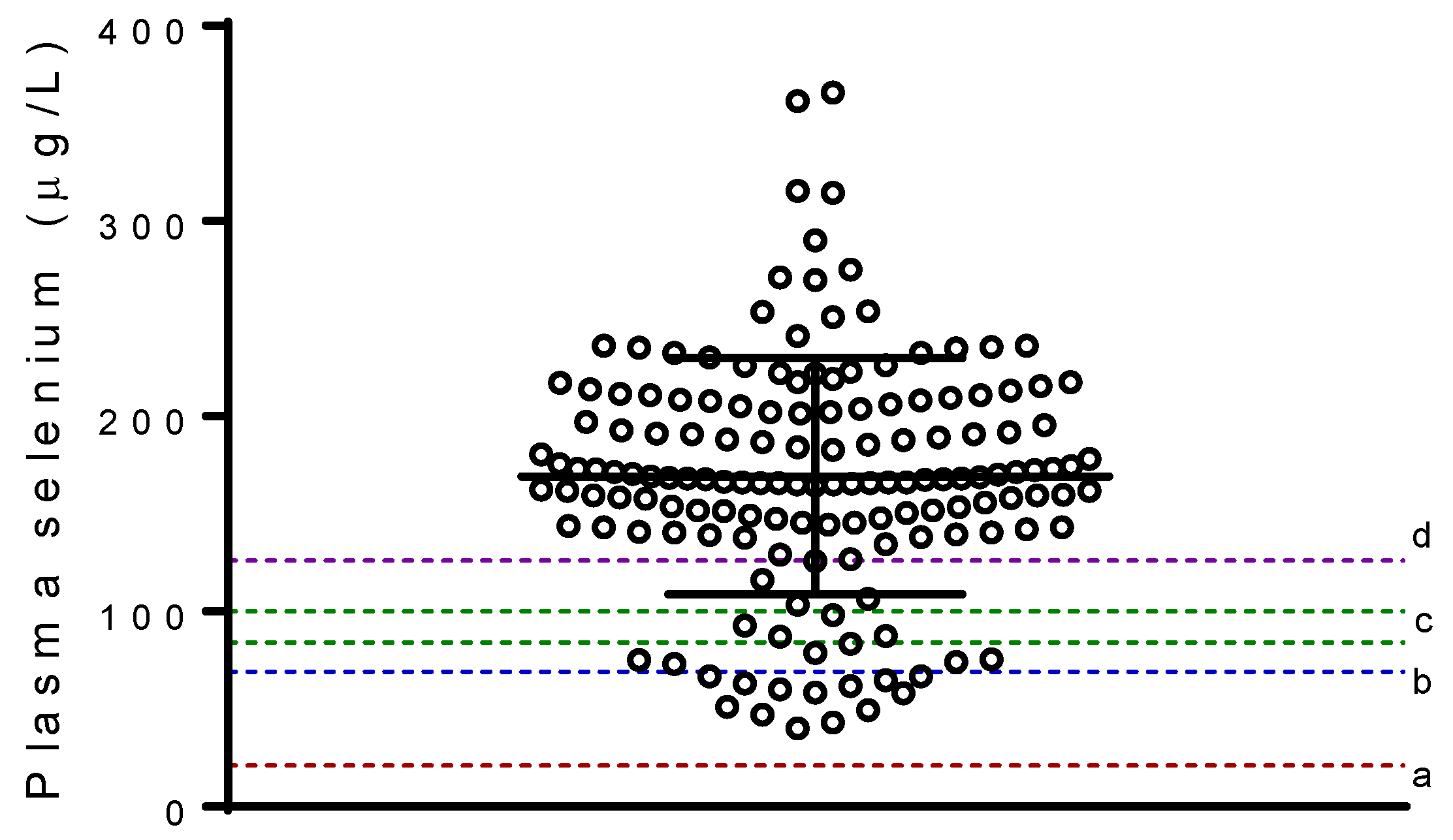
| Parameter | Values |
|---|---|
| Age (year) | 70.7 (4.1) |
| Women, n (%) a | 96 (62.3) |
| APOEɛ4 carriers, n (%) a | 33 (21.4) |
| BMI (kg m2) | 27.9 (5.3) |
| Overweight, n (%) a | 54 (35.1) |
| Obese, n (%) a | 49 (31.8) |
| Education | |
| Primary/high school, n (%) a | 57 (37) |
| Technical certificate, n (%) a | 25 (16.2) |
| University, n (%) a | 72 (46.8) |
| Habitual physical activity (kJ week−1) | 7957.3 (7526.5) |
| History of CVD, n (%) a | 23 (14.9) |
| Selenium intake (µg day−1) | 93.1 (53.6) |
| Plasma selenium (µg L−1) | 169.3 (60.4) |
| Serum IGF-1 (nmol L−1) | 17.1 (4.7) |
| Serum BDNF (ng mL−1) | 21.3 (7.7) |
| Serum VEGF (pg mL−1) | 407.5 (455.1) |
| Serum hs-CRP (mg/mL) | 2.1 (2.6) |
| Serum IL-6 (pg/mL) | 4.6 (7.9) |
| Serum IL-8 (pg/mL) | 15.6 (10.5) |
| TNF-alpha (pg/mL) | 10.2 (3.9) |
| IL-10 (pg/mL) | 10.7 (17.1) |
| Adiponectin (µg/mL) | 5.1 (3.5) |
| Composite inflammatory index b | 0.00 (2.4) |
| Outcomes | β | 95% CI | p |
|---|---|---|---|
| Cogstate | |||
| Global cognitive function composite a | 0.001 | −0.001; 0.003 | 0.198 |
| Working memory/learning composite a | 0.002 | 0.000; 0.004 | 0.095 |
| Attention/psychomotor composite a | 0.002 | −0.001; 0.004 | 0.149 |
| BRIEF-A | |||
| Behavioural regulation index b | −0.016 | −0.042; 0.010 | 0.214 |
| Metacognition index b | −0.013 | −0.041; 0.015 | 0.369 |
| Global executive composite b | −0.015 | −0.041; 0.011 | 0.255 |
| Outcomes | β | 95% CI | p |
|---|---|---|---|
| Composite inflammatory index a | 0.002 | −0.005; 0.009 | 0.550 |
| IGF-1 (nmol L−1) b | 0.000 | −0.001; 0.000 | 0.238 |
| BDNF (ng mL−1) b | 0.000 | −0.001; 0.001 | 0.799 |
| VEGF (pg mL−1) b | −0.001 | −0.003; 0.002 | 0.687 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardoso, B.R.; Szymlek-Gay, E.A.; Roberts, B.R.; Formica, M.; Gianoudis, J.; O’Connell, S.; Nowson, C.A.; Daly, R.M. Selenium Status Is Not Associated with Cognitive Performance: A Cross-Sectional Study in 154 Older Australian Adults. Nutrients 2018, 10, 1847. https://doi.org/10.3390/nu10121847
Cardoso BR, Szymlek-Gay EA, Roberts BR, Formica M, Gianoudis J, O’Connell S, Nowson CA, Daly RM. Selenium Status Is Not Associated with Cognitive Performance: A Cross-Sectional Study in 154 Older Australian Adults. Nutrients. 2018; 10(12):1847. https://doi.org/10.3390/nu10121847
Chicago/Turabian StyleCardoso, Barbara R., Ewa A. Szymlek-Gay, Blaine R. Roberts, Melissa Formica, Jenny Gianoudis, Stella O’Connell, Caryl A. Nowson, and Robin M. Daly. 2018. "Selenium Status Is Not Associated with Cognitive Performance: A Cross-Sectional Study in 154 Older Australian Adults" Nutrients 10, no. 12: 1847. https://doi.org/10.3390/nu10121847
APA StyleCardoso, B. R., Szymlek-Gay, E. A., Roberts, B. R., Formica, M., Gianoudis, J., O’Connell, S., Nowson, C. A., & Daly, R. M. (2018). Selenium Status Is Not Associated with Cognitive Performance: A Cross-Sectional Study in 154 Older Australian Adults. Nutrients, 10(12), 1847. https://doi.org/10.3390/nu10121847






