Probiotics in Celiac Disease
Abstract
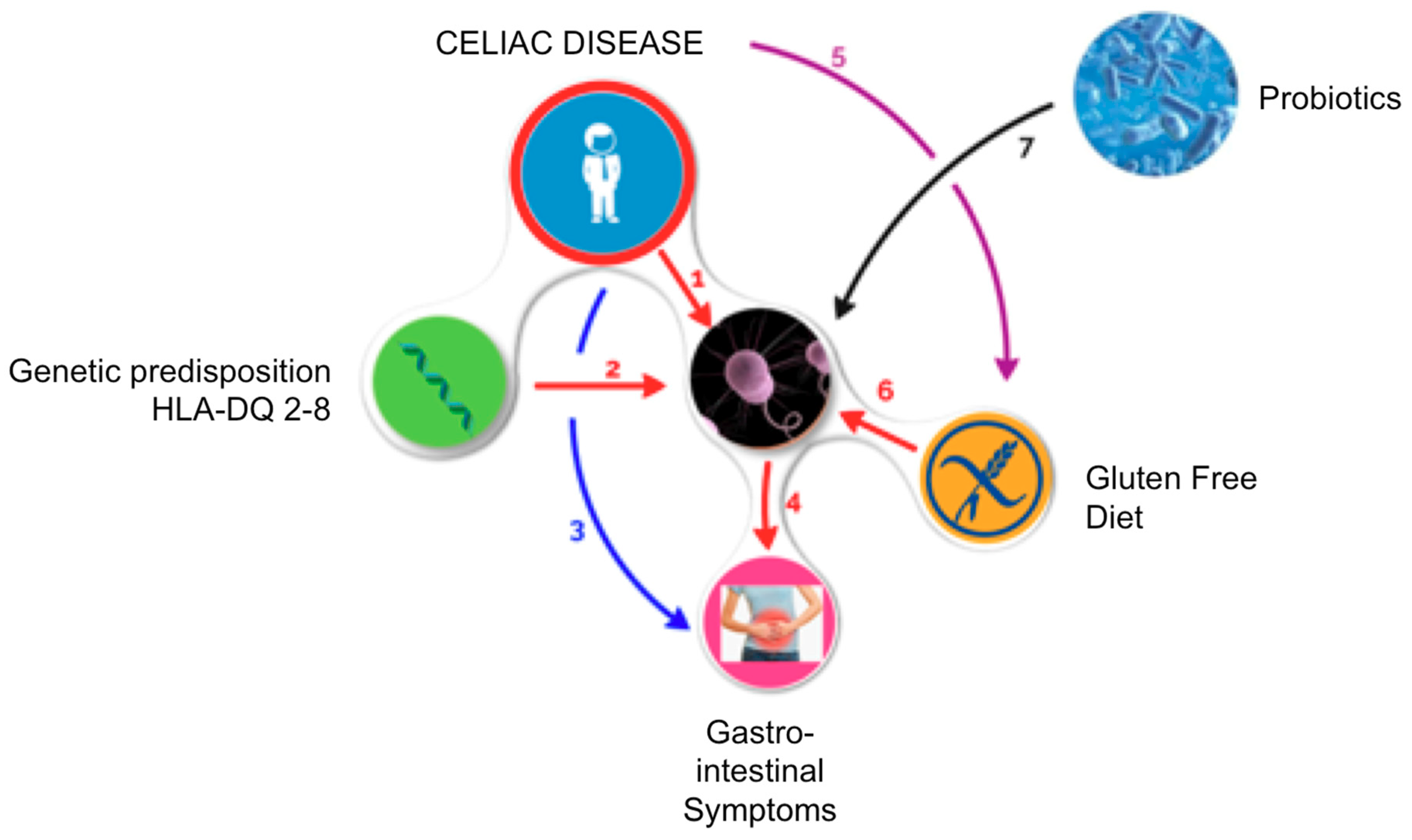
1. Introduction
2. Gut Microbiota and Risk of Developing Celiac Disease
3. Microbiota in Celiac Patient
4. Gluten-Free Diet and Gut Microbiota
5. Probiotics Supplementation
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Green, P.H.; Cellier, C. Celiac disease. N. Engl. J. Med. 2007, 357, 1731–1743. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Rubio-Tapia, A.; van Dyke, C.T.; Melton, L.J., 3rd; Zinsmeister, A.R.; Lahr, B.D.; Murray, J.A. Increasing incidence of celiac disease in a North American population. Am. J. Gastroenterol. 2013, 108, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.A.; Van Dyke, C.; Plevak, M.F.; Dierkhising, R.A.; Zinsmeister, A.R.; Melton, L.J., 3rd. Trends in the identification and clinical features of celiac disease in a North American community, 1950–2001. Clin. Gastroenterol. Hepatol. 2003, 1, 19–27. [Google Scholar] [CrossRef] [PubMed]
- White, L.E.; Merrick, V.M.; Bannerman, E.; Russell, R.K.; Basude, D.; Henderson, P.; Wilson, D.C.; Gillett, P.M. The rising incidence of celiac disease in Scotland. Pediatrics 2013, 132, e924–e931. [Google Scholar] [CrossRef] [PubMed]
- Galipeau, H.J.; McCarville, J.L.; Huebener, S.; Litwin, O.; Meisel, M.; Jabri, B.; Sanz, Y.; Murray, J.A.; Jordana, M.; Alaedini, A.; et al. Intestinal microbiota modulates gluten-induced immunopathology in humanized mice. Am. J. Pathol. 2015, 185, 2969–2982. [Google Scholar] [CrossRef] [PubMed]
- Sellitto, M.; Bai, G.; Serena, G.; Fricke, W.F.; Sturgeon, C.; Gajer, P.; White, J.R.; Koenig, S.S.; Sakamoto, J.; Boothe, D.; et al. Proof of concept of microbiome-metabolome analysis and delayed gluten exposure on celiac disease autoimmunity in genetically at-risk infants. PLoS ONE 2012, 7, 33387. [Google Scholar] [CrossRef] [PubMed]
- Conte, M.P.; Schippa, S.; Zamboni, I.; Penta, M.; Chiarini, F.; Seganti, L.; Osborn, J.; Falconieri, P.; Borrelli, O.; Cucchiara, S. Gut-associated bacterial microbiota in paediatric patients with inflammatory bowel disease. Gut 2006, 55, 1760–1767. [Google Scholar] [CrossRef] [PubMed]
- Maynard, C.L.; Elson, C.O.; Hatton, R.D.; Weaver, C.T. Reciprocal interactions of the intestinal microbiota and immune system. Nature 2012, 489, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Oelschlaeger, T.A.; Wullaert, A.; Vlantis, K.; Pasparakis, M.; Wehkamp, J.; Stange, E.F.; Gersemann, M. Bacteria regulate intestinal epithelial cell differentiation factors both in vitro and in vivo. PLoS ONE 2013, 8, e55620. [Google Scholar] [CrossRef]
- Cénit, M.C.; Olivares, M.; Codoner-Franch, P.; Sanz, Y. Intestinal microbiota and celiac disease: Cause, consequence or coevolution? Nutrients 2015, 7, 6900–6923. [Google Scholar] [CrossRef] [PubMed]
- Verdu, E.F.; Galipeau, H.J.; Jabri, B. Novel players in coeliac disease pathogenesis: Role of the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Sommer, F.; BaÅNckhed, F. The gut microbiota—Masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Belkaid, Y.; Hand, T.W. Role of the microbiota in immunity and inflammation. Cell 2014, 157, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Neu, J.; Rushing, J. Cesarean versus vaginal delivery: Long-term infant outcomes and the hygiene hypothesis. Clin. Perinatol. 2011, 38, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 2010, 107, 11971–11975. [Google Scholar] [CrossRef] [PubMed]
- Francavilla, R.; Cristofori, F.; Tripaldi, M.E.; Indrio, F. Intervention for Dysbiosis in Children Born by C-Section. Ann. Nutr. Metab. 2018, 73 (Suppl. 3), 33–39. [Google Scholar] [CrossRef] [PubMed]
- Decker, E.; Hornef, M.; Stockinger, S. Cesarean delivery is associated with celiac disease but not inflammatory bowel disease in children. Gut Microbes 2011, 2, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Adlercreutz, E.H.; Wingren, C.J.; Vincente, R.P.; Merlo, J.; Agardh, D. Perinatal risk factors increase the risk of being affected by both type 1 diabetes and coeliac disease. Acta Paediatr. 2015, 104, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Akobeng, A.K.; Ramanan, A.V.; Buchan, I.; Heller, R.F. Effect of breast feeding on risk of coeliac disease: A systematic review and meta-analysis of observational studies. Arch. Dis. Child. 2006, 91, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Dydensborg Sander, S.; Hansen, A.V.; Størdal, K.; Andersen, A.N.; Murray, J.A.; Husby, S. Mode of delivery is not associated with celiac disease. Clin. Epidemiol. 2018, 10, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Aronsson, C.A.; Lee, H.-S.; Liu, E.; Uusitalo, U.; Hummel, S.; Yang, J.; Hummel, M.; Rewers, M.; She, J.-X.; Simell, O.; et al. Age at gluten introduction and risk of celiac disease. Pediatrics 2015, 135, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Størdal, K.; White, R.A.; Eggesbø, M. Early feeding and risk of celiac disease in a prospective birth cohort. Pediatrics 2013, 132, e1202–e1209. [Google Scholar] [CrossRef] [PubMed]
- Palma, G.D.; Capilla, A.; Nova, E.; Castillejo, G.; Varea, V.; Pozo, T.; Garrote, J.A.; Polanco, I.; López, A.; Ribes-Koninckx, C.; et al. Influence of milk-feeding type and genetic risk of developing coeliac disease on intestinalbmicrobiota of infants: The PROFICEL study. PLoS ONE 2012, 7, e30791. [Google Scholar] [CrossRef] [PubMed]
- Olivares, M.; Albrecht, S.; De Palma, G.; Ferrer, MD.; Castillejo, G.; Schols, HA.; Sanz, Y. Human milk composition differs in healthy mothers and mothers with celiac disease. Eur. J. Nutr. 2015, 54, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Olivares, M.; Neef, A.; Castillejo, G.; Palma, G.D.; Varea, V.; Capilla, A.; Palau, F.; Nova, E.; Marcos, A.; Polanco, I.; et al. The HLA-DQ2 genotype selects for early intestinal microbiota composition in infants at high risk of developing coeliac disease. Gut 2015, 64, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Dieli-Crimi, R.; Cénit, M.C.; Núñez, C. The genetics of celiac disease: A comprehensive review of clinical implications. J. Autoimmun. 2015, 64, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.M.; Camhi, S.; Huedo-Medina, T.B.; Fasano, A. Celiac Disease Genomic, Environmental, Microbiome, and Metabolomic (CDGEMM) Study Design: Approach to the Future of Personalized Prevention of Celiac Disease. Nutrients 2015, 7, 9325–9336. [Google Scholar] [CrossRef] [PubMed]
- Collado, M.C.; Calabuig, M.; Sanz, Y. Differences between the fecal microbiota of coeliac infants and healthy controls. Curr. Issues Intest. Microbiol. 2007, 8, 9–14. [Google Scholar] [PubMed]
- Sanz, Y.; Sanchez, E.; Marzotto, M.; Calabuig, M.; Torriani, S.; Dellaglio, F. Differences in faecal bacterial communities in coeliac and healthy children as detected by PCR and denaturing gradient gel electrophoresis. FEMS Immunol. Med. Microbiol. 2007, 51, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Nadal, I.; Donat, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Imbalance in the composition of the duodenal microbiota of children with coeliac disease. J. Med. Microbiol. 2007, 56, 1669–1674. [Google Scholar] [CrossRef] [PubMed]
- Collado, MC.; Donat, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Specific duodenal and faecal bacterial groups associated with paediatric coeliac disease. J. Clin. Pathol. 2009, 62, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Di Cagno, R.; Rizzello, C.G.; Gagliardi, F.; Ricciuti, P.; Ndagijimana, M.; Francavilla, R.; Guerzoni, M.E.; Crecchio, C.; Gobbetti, M.; De Angelis, M. Different fecal microbiotas and volatile organic compounds in treated and untreated children with celiac disease. Appl. Environ. Microbiol. 2009, 75, 3963–3971. [Google Scholar] [CrossRef] [PubMed]
- Ou, G.; Hedberg, M.; Horstedt, P.; Baranov, V.; Forsberg, G.; Drobni, M.; Sandström, O.; Wai, S.N.; Johansson, I.; Hammarström, M.L.; et al. Proximal small intestinal microbiota and identification of rod-shaped bacteria associated with childhood celiac disease. Am. J. Gastroenterol. 2009, 104, 3058–3067. [Google Scholar] [CrossRef] [PubMed]
- Schippa, S.; Iebba, V.; Barbato, M.; Di Nardo, G.; Totino, V.; Checchi, M.P.; Longhi, C.; Maiella, G.; Cucchiara, S.; Conte, M.P. A distinctive ‘‘microbial signature’’ in celiac pediatric patients. BMC Microbiol. 2010, 10, 175. [Google Scholar] [CrossRef] [PubMed]
- De Palma, G.; Nadal, I.; Medina, M.; Donat, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Intestinal dysbiosis and reduced immunoglobulin-coated bacteria associated with coeliac disease in children. BMC Microbiol. 2010, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, E.; Donat, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Intestinal Bacteroides species associated with coeliac disease. J. Clin. Pathol. 2010, 63, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Di Cagno, R.; De Angelis, M.; De Pasquale, I.; Ndagijimana, M.; Vernocchi, P.; Ricciuti, P.; Gagliardi, F.; Laghi, L.; Crecchio, C.; Guerzoni, M.E.; et al. Duodenal and faecal microbiota of celiac children: Molecular, phenotype and metabolome characterization. BMC Microbiol. 2011, 11, 219. [Google Scholar] [CrossRef] [PubMed]
- Nistal, E.; Caminero, A.; Vivas, S.; Ruiz de Morales, J.M.; Sáenz de Miera, L.E.; Rodríguez-Aparicio, L.B.; Casqueiro, J. Differences in faecal bacteria populations and faecal bacteria metabolism in healthy adults and celiac disease patients. Biochimie 2012, 94, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Nistal, E.; Caminero, A.; Herran, A.R.; Arias, L.; Vivas, S.; de Morales, J.M.; Calleja, S.; de Miera, L.E.; Arroyo, P.; Casqueiro, J. Differences of small intestinal bacteria populations in adults and children with/ without celiac disease: Effect of age, gluten diet, and disease. Inflamm. Bowel Dis. 2012, 18, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Intestinal Staphylococcus spp. and virulent features associated with coeliac disease. J. Clin. Pathol. 2012, 65, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Acar, S.; Yetkiner, A.A.; Ersin, N.; Oncag, O.; Aydogdu, S.; Arikan, C. Oral findings and salivary parameters in children with celiac disease: A preliminary study. Med. Princ. Pract. 2012, 21, 129–133. [Google Scholar] [CrossRef] [PubMed]
- De Meij, T.G.J.; Budding, A.E.; Grasman, M.E.; Kneepkens, C.M.F.; Savelkoul, P.H.M.; Mearin, M.L. Composition and diversity of the duodenal mucosa-associated microbiome in children with untreated coeliac disease. Scand. J. Gastroenterol. 2013, 48, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, E.; Donat, E.; Ribes-Koninckx, C.; Fernandez-Murga, M.L.; Sanz, Y. Duodenal-mucosal bacteria associated with celiac disease in children. Appl. Environ. Microbiol. 2013, 79, 5472–5479. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, P.; Kaukinen, K.; Tuovinen, E.; Collin, P.; Lindfors, K.; Partanen, J.; Mäki, M.; Mättö, J. The duodenal microbiota composition of adult celiac disease patients is associated with the clinical manifestation of the disease. Inflamm. Bowel Dis. 2013, 19, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Kalliomaki, M.; Heilig, H.G.H.J.; Palva, A.; Lähteenoja, H.; de Vos, W.M.; Salojärvi, J.; Satokari, R. Duodenal microbiota composition and mucosal homeostasis in pediatric celiac disease. BMC Gastroenterol. 2013, 13, 113. [Google Scholar] [CrossRef] [PubMed]
- Francavilla, R.; Ercolini, D.; Piccolo, M.; Vannini, L.; Siragusa, S.; De Filippis, F.; De Pasquale, I.; Di Cagno, R.; Di Toma, M.; Gozzi, G.; et al. Salivary microbiota and metabolome associated with celiac disease. Appl. Environ. Microbiol. 2014, 80, 3416–3425. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, P.; Laurikka, P.; Lindfors, K.; Collin, P.; Salmi, T.; Lähdeaho, M.L.; Saavalainen, P.; Mäki, M.; Mättö, J.; Kurppa, K.; et al. Altered duodenal microbiota composition in celiac disease patients suffering from persistent symptoms on a long-term gluten-free diet. Am. J. Gastroenterol. 2014, 109, 1933–1941. [Google Scholar] [CrossRef] [PubMed]
- Giron Fernandez-Crehuet, F.; Tapia-Paniagua, S.; Morinigo Gutierrez, M.A.; Navas-López, V.M.; Juliana Serrano, M.; Blasco-Alonso, J.; Sierra Salinas, C. The duodenal microbiota composition in children with active coeliac disease is influenced by the degree of enteropathy. An. Pediatr. (Barc.) 2016, 84, 224–230. [Google Scholar] [CrossRef] [PubMed]
- D’Argenio, V.; Casaburi, G.; Precone, V.; Pagliuca, C.; Colicchio, R.; Sarnataro, D.; Discepolo, V.; Kim, S.M.; Russo, I.; Del Vecchio Blanco, G.; et al. Metagenomics Reveals Dysbiosis and a Potentially Pathogenic, N. flavescens Strain in Duodenum of Adult Celiac Patients. Am. J. Gastroenterol. 2016, 111, 879–890. [Google Scholar] [CrossRef] [PubMed]
- Quagliariello, A.; Aloisio, I.; Bozzi Cionci, N.; Luiselli, D.; D’Auria, G.; Martinez-Priego, L.; Pérez-Villarroya, D.; Langerholc, T.; Primec, M.; Mičetić-Turk, D.; et al. Effect of Bifidobacterium breve on the Intestinal Microbiota of Coeliac Children on a Gluten Free Diet: A Pilot Study. Nutrients 2016, 8, 660. [Google Scholar] [CrossRef] [PubMed]
- Tian, N.; Faller, L.; Leffler, D.A.; Kelly, C.P.; Hansen, J.; Bosch, J.A.; Wei, G.; Paster, B.J.; Schuppan, D.; Helmerhorst, E.J. Salivary Gluten Degradation and Oral Microbial Profiles in Healthy Individuals and Celiac Disease Patients. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef] [PubMed]
- Sanz, Y. Microbiome and gluten. Ann. Nutr. Metab. 2015, 2, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Guandalini, S.; Assiri, A. Celiac disease: A review. JAMA Pediatr. 2014, 168, 272–278. [Google Scholar] [CrossRef] [PubMed]
- De Palma, G.; Nadal, I.; Collado, M.C.; Sanz, Y. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects. Br. J. Nutr. 2009, 102, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Jackson, F.W. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects-comment by Jackson. Br. J. Nutr. 2010, 104, 773. [Google Scholar] [CrossRef] [PubMed]
- Galipeau, H.; McCarville, J.L.; Moeller, S.; Murray, J.; Alaedini, A.; Jabri, B.; Verdu, E. Gluten-induced responses in NOD/DQ8 mice are influenced by bacterial colonization. Gastroenterology 2014, 146 (Suppl. 1). [Google Scholar] [CrossRef]
- D’Arienzo, R.; Maurano, F.; Luongo, D.; Mazzarella, G.; Stefanile, R.; Troncone, R.; Auricchio, S.; Ricca, E.; David, C.; Rossi, M. Adjuvant effect of Lactobacillus casei in a mouse model of gluten sensitivity. Immunol. Lett. 2008, 119, 78–83. [Google Scholar] [CrossRef] [PubMed]
- D’Arienzo, R.; Stefanile, R.; Maurano, F.; Mazzarella, G.; Ricca, E.; Troncone, R.; Auricchio, S.; Rossi, M. Immunomodulatory effects of Lactobacillus casei administration in a mouse model of gliadin-sensitive enteropathy. Scand. J. Immunol. 2011, 74, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Papista, C.; Gerakopoulos, V.; Kourelis, A.; Sounidaki, M.; Kontana, A.; Berthelot, L.; Moura, I.C.; Monteiro, R.C.; Yiangou, M. Gluten induces coeliac-like disease in sensitised mice involving IgA, CD71 and transglutaminase 2 interactions that are prevented by probiotics. Lab. Investig. 2012, 92, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Lindfors, K.; Blomqvist, T.; Juuti-Uusitalo, K.; Stenman, S.; Venäläinen, J.; Mäki, M.; Kaukinen, K. Live probiotic Bifidobacterium lactis bacteria inhibit the toxic effects induced by wheat gliadin in epithelial cell culture. Clin. Exp. Immunol. 2008, 152, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Laparra, J.M.; Olivares, M.; Gallina, O.; Sanz, Y. Bifidobacterium longum CECT 7347 modulates immune responses in a gliadin-induced enteropathy animal model. PLoS ONE 2012, 7, e30744. [Google Scholar] [CrossRef] [PubMed]
- D’Arienzo, R.; Maurano, F.; Lavermicocca, P.; Ricca, E.; Rossi, M. Modulation of the immune response by probiotic strains in a mouse model of gluten sensitivity. Cytokine 2009, 48, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Smecuol, E.; Hwang, H.J.; Sugai, E.; Corso, L.; Cherñavsky, A.C.; Bellavite, F.P.; González, A.; Vodánovich, F.; Moreno, M.L.; Vázquez, H.; et al. Exploratory, randomized, double-blind, placebo-controlled study on the effects of Bifidobacterium infantis Natren life start strain super strain in active celiac disease. J. Clin. Gastroenterol. 2013, 47, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Pinto-Sánchez, M.I.; Smecuol, E.C.; Temprano, M.P.; Sugai, E.; González, A.; Moreno, M.L.; Huang, X.; Bercik, P.; Cabanne, A.; Vázquez, H.; et al. Bifidobacterium infantis NLS Super Strain Reduces the Expression of α-Defensin-5, a Marker of Innate Immunity, in the Mucosa of Active Celiac Disease Patients. J. Clin. Gastroenterol. 2017, 51, 814–817. [Google Scholar] [CrossRef] [PubMed]
- Olivares, M.; Castillejo, G.; Varea, V.; Sanz, Y. Double-blind, randomised, placebo-controlled intervention trial to evaluate the effects of Bifidobacterium longum CECT 7347 in children with newly diagnosed coeliac disease. Br. J. Nutr. 2014, 112, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Harnett, J.; Myers, S.P.; Rolfe, M. Probiotics and the microbiome in celiac disease: A randomised controlled trial. Evid. Based. Complement. Altern. Med. 2016, 2016, 9048574. [Google Scholar] [CrossRef] [PubMed]
- Klemenak, M.; Dolinšek, J.; Langerholc, T.; Di Gioia, D. Administration of Bifidobacterium breve Decreases the Production of TNF-α in Children with Celiac Disease. Dig. Dis. Sci. 2015, 60, 3386–3392. [Google Scholar] [CrossRef] [PubMed]
- Primec, M.; Klemenak, M.; Di Gioia, D.; Aloisio, I.; Bozzi Cionci, N.; Quagliariello, A.; Gorenjak, M.; Mičetić-Turk, D.; Langerholc, T. Clinical intervention using Bifidobacterium strains in celiac disease children reveals novel microbial modulators of TNF-α and short-chain fatty acids. Clin. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Francavilla, R.; Piccolo, M.; Francavilla, A.; Polimeno, L.; Semeraro, F.; Cristofori, F.; Castellaneta, S.; Barone, M.; Indrio, F.; Gobbetti, M.; et al. Clinical and Microbiological Effect of a Multispecies Probiotic Supplementation in Celiac Patients with Persistent IBS-type Symptoms: A Randomized, Double-Blind, Placebo-controlled, Multicenter Trial. J. Clin. Gastroenterol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Losurdo, G.; Marra, A.; Shahini, E.; Girardi, B.; Giorgio, F.; Amoruso, A.; Pisani, A.; Piscitelli, D.; Barone, M.; Principi, M.; et al. Small intestinal bacterial overgrowth and celiac disease: A systematic review with pooled-data analysis. Neurogastroenterol. Motil. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
| Author | Population | Age | Saliva Samples | Duodenal Biopsies | Fecal Samples | Methods | Results in CD Patients |
|---|---|---|---|---|---|---|---|
| Collado et al. [28] | 26 CD vs. 23 HC | Children | No | No | Yes | Colture and FISH | ↑ Bacteroides–Prevotella, Clostriudium hystoliticum, Eubacterium rectale–C. coccoides, Atopobium and Staphylococcus |
| Sanz et al. [29] | 10 CD vs. 10 HC | Children | No | No | Yes | Culture DGGE | L. curvatus, Leuconostoc mesenteroides only in CD |
| Nadal et al. [30] | 20 CD vs. 10 CD-GFD vs. 8 HC | Children | No | Yes | No | FISH Flow citometry | ↓ Ratio of Lactobacillus–Bifidobacterium to Bacteroides–E. coli ↑ Gram-negative |
| Collado et al. [31] | 8 CD vs. 8 CD vs. 8 HC | Children | No | Yes | Yes | real-time PCR | ↑ Bacteroides, C. leptum, E. coli, Staphylococcus ↓ Bifidobacteria |
| Di Cagno et al. [32] | 7 CD vs. 7 CD-GFD vs. 7 HC | Children | No | No | Yes | real time PCR DGGE | ↓ Ratio of cultivable lactic acid bacteria and Bifidobacterium to Bacteroides and enterobacteria ↓ Lactobacillus |
| Ou et al [33] | 45 CD vs. 18 HC | Children | No | Yes | No | 16S rDNA sequencing | ↑ Haemophilus, Streptococcus, Neisseria |
| Schippa et al. [34] | 20 CD before and after GFD vs. 10 HC | Children | No | Yes | No | 16S rDNA sequencing TTGE | ↑ Bacteroides vulgatus and Escherichia coli |
| De Palma et al. [35] | 24 CD vs. 18 CD-GFD vs. 20 HC | Children | No | No | Yes | FISH flow cytometry | ↓ Gram-positive to Gram-negative bacteria ratio ↓ Bifidobacterium, Clostridium histolyticum, C. lituseburense and Faecalibacterium prausnitzii ↑ Bacteroides–Prevotella |
| Sanchez et al. [36] | 20 CD vs. 12 CD-GFD vs. 8 HC | Children | No | Yes | No | DGGE | ↑ Bacteroides dorei ↓ Bacteroides distasonis, Bacteroides fragilis/Bacteroides thetaiotaomicron, Bacteroides uniformis, and Bacteroides ovatus ↑ Bifidobacterium adolescentis Bifidobacterium animalis subsp lactis |
| Di Cagno et al. [37] | 19 CD vs. 15 HC | Children | No | Yes | Yes | DGGE | ↓ Lactobacillus, Enterococcus, and Bifidobacteria |
| Nistal et al. [38] | 10 CD vs. 11 CD-GFD vs. 11 HC | Adults | No | No | Yes | DGGE | ↑ B. bifidum and catenulatum |
| Nistal et al. [39] | 13 CD vs. 5 CD-GFD vs. 10 HC | Children Adults | No | Yes | No | 16SrRNA gene sequencing | ↓ Streptococcus and Prevotella |
| Sanchez et al. [40] | 20 CD vs. 20 CD-GFD vs. 20 HC | Children | No | No | Yes | PCR DNA sequencing | ↑ Staphylococcus epidermidis Staphylococcus haemolyticus ↓ S. aureus |
| Acar et al. [41] | 35 CD vs. 35 HC | Children | Yes | No | No | CRT Bacteria | ↓ Salivary mutans streptococci and lactobacilli colonization |
| De Meij et al. [42] | 21 CD vs. 21 HC | Children | No | Yes | No | IS-pro, profiling method | No differences |
| Sanchez et al. [43] | 32 CD vs. 17 CD-GFD vs. 8 HC | Children | No | Yes | No | Colture 16S rRNA gene sequencing | ↑ Proteobacteria, Enterobacteriaceae, and Staphylococcaceae ↓ Streptococcaceae, Firmicutes |
| Wacklin et al. [44] | 33 CD (either symptomatic or asymptomatic) vs. 18 HC | Adults | No | Yes | No | 16S rRNA gene sequencing | ↑ Proteobacteria, such as Acinetobacter and Neisseria, in patient with GI symptoms. ↓ microbial diversity in GI symptoms or anemia |
| Cheng et al [45] | 10 CD vs. 9 HC | Children | No | Yes | No | qRT-PCR | No differences Haemophilus ssp. and Serratia ssp. had relatively higher abundance in CD |
| Francavilla et al. [46] | 13 CD-GFD vs. 13 HC | Children | Yes | No | No | 16S rRNA gene sequencing | ↑ Lachnospiraceae, Gemellaceae, and Streptococcus sanguinis Bacteroidetes ↓ Streptococcus thermophilus |
| Wacklin et al. [47] | 18 CD-GFD symptomatic vs. 18 CD-GFD asymptomatic | Adults | No | Yes | No | 16S rRNA gene sequencing | ↑ Proteobacteria ↓ Bacteroides and Firmicutes |
| Giron-Fernandez Crehuet et al. [48] | 11 A-CD vs. 11 HC | Children | No | Yes | No | DGGE | Lactobacillus genus |
| D’Argenio et al. [49] | 20 A-CD vs. 6 CD-GFD vs. 15 HC | Adults | No | Yes | No | 16S rRNA gene sequencing metagenomics | ↑ Proteobacteria ↓ Firmicutes and Actinobacteria ↑ Neisseria genus (Neisseria flavescens) |
| Quagliariello et al. [50] | 40 A-CD vs. 16 HC | Children | No | No | Yes | 16S rRNA gene sequencing Quantitative PCR (qPCR) | ↓ Firmicutes/Bacteroidetes ratio, ↓ Actinobacteria and Euryarchaeota |
| Tian et al. [51] | 21 CD-GFD vs. 8 RCD vs. 20 HC | Adults | Yes | No | No | 16S rRNA gene sequencing | Bacteroidetes (CD > RCD), Actinobacteria (CD < RCD), Fusobacteria (CD > RCD) |
| Author | RCT | Population | Used Strain | Time of Administration | Findings in Probiotics Group |
|---|---|---|---|---|---|
| Smecuol et al. [63] | Yes | 22 A-CD (12 probiotic vs. 10 placebo) | Bifidobacterium infantis Natren life start | 3 weeks | Improvement in GI symptoms (indigestion, constipation, and gastroesophageal reflux) ↓ Final/baseline IgA tTG and IgA DGP antibody concentration ratios ↑ Serum macrophage inflammatory protein-1β No differences in intestinal permeability No significant changes in cytokines and chemokines production |
| Pinto-Sánchez et al. [64] | No | 24 A-CD no treatment vs. 12 A-CD probiotic treatment vs. 5 CD-GFD | Bifidobacterium infantis Natren life start | 3 weeks | ↓ Paneth cell counts ↓ α-defensin-5 |
| Olivares et al. [65] | Yes | 36 A-CD (18 B. longum + GFD vs. 18 placebo + GFD) | Bifidobacterium longum CECT 7347 | 3 months | ↑ Height percentile ↓ Peripheral CD3+ T lymphocytes concentration ↓ TNF-α levels ↓ Bacteroides fragilis and Enterobacteriaceae ↑ Harmless to potentially harmful bacteria ratio No differences in GI symptoms |
| Quagliarello et al. [50] | Yes | 40 A-CD children (20 probiotic and 20 placebo) vs. 16 HC | Bifidobacterium breve strains (B632 and BR03) | 3 months | ↑ Actinobacteria Re-establishment Firmicutes/Bacteroidetes ratio. |
| Harnett et al. [66] | Yes | 45 CD-GFD with symptoms (23 probiotic and 22 placebo) | multispecies probiotic VSL#3 (450 billion viable lyophilized bacteria Streptococcus thermophilus, Bifidobacterium breve, Bifidobacterium longum, Bifidobacterium infantis, Lactobacillus acidophilus, Lactobacillus plantarum, Lactobacillus paracasei, and Lactobacillus delbrueckii subsp. Bulgaricus) | 12 weeks | No differences in the fecal microbiota counts No differences in symptoms severity |
| Klemenak et al. [67] | Yes | 49 CD-GFD (24 probiotic and 25 placebo) 18 HC | Bifidobacterium breve strains (BR03 and B632) | 3 months | ↓ TNF-alpha levels (not persistent) |
| Primec et al. [68] | Yes | 40 CD (20 probiotic and 20 placebo) 16 HC | Bifidobacterium breve strains (BR03 and B632) | 3 months | Negative relationship between Firmicutes and pro-inflammatory TNF-α. |
| Francavilla et al. [69] | Yes | 109 CD-GFD with IBS symptoms (54 probiotic vs. 55 placebo) | mixture of 5 Lactobacillus casei LMG 101/37 P-17504 Lactobacillus plantarum CECT 4528, Bifidobacterium animalis subsp. lactis Bi1 LMG P-17502, Bifidobacterium breve Bbr8 LMG P-17501 Bifidobacterium breve Bl10 LMG P-17500 | 6 weeks | Improvement in GI symptoms ↑ Bifidobacteria (persistent) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cristofori, F.; Indrio, F.; Miniello, V.L.; De Angelis, M.; Francavilla, R. Probiotics in Celiac Disease. Nutrients 2018, 10, 1824. https://doi.org/10.3390/nu10121824
Cristofori F, Indrio F, Miniello VL, De Angelis M, Francavilla R. Probiotics in Celiac Disease. Nutrients. 2018; 10(12):1824. https://doi.org/10.3390/nu10121824
Chicago/Turabian StyleCristofori, Fernanda, Flavia Indrio, Vito Leonardo Miniello, Maria De Angelis, and Ruggiero Francavilla. 2018. "Probiotics in Celiac Disease" Nutrients 10, no. 12: 1824. https://doi.org/10.3390/nu10121824
APA StyleCristofori, F., Indrio, F., Miniello, V. L., De Angelis, M., & Francavilla, R. (2018). Probiotics in Celiac Disease. Nutrients, 10(12), 1824. https://doi.org/10.3390/nu10121824







