Health Benefits of Bioactive Compounds from the Genus Ilex, a Source of Traditional Caffeinated Beverages
Abstract
1. Introduction
2. Distribution
3. Bioactive Compounds
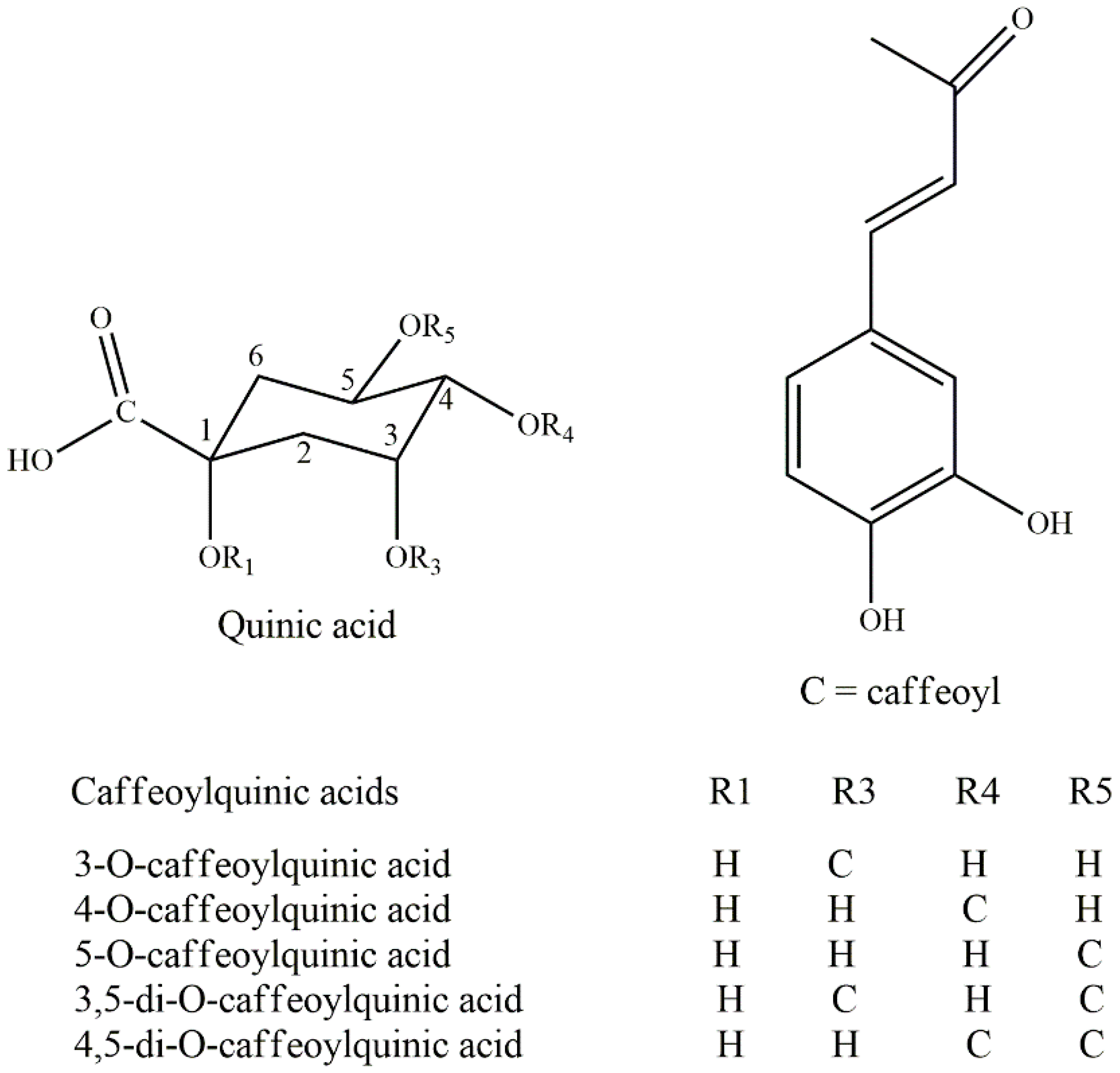
3.1. Polyphenols
3.1.1. Polyphenols in Large-Leaved Kudingcha
3.1.2. Polyphenols in Yerba Mate
3.1.3. Polyphenols in Yaupon Holly
3.1.4. Polyphenols in Guayusa
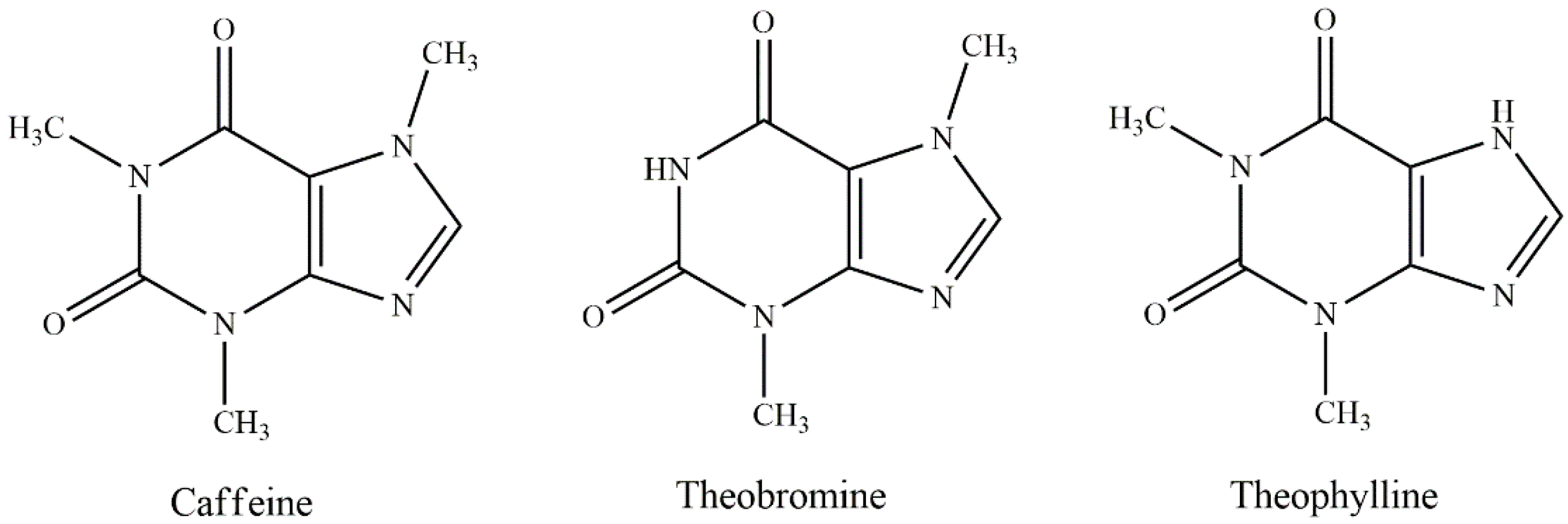
3.2. Alkaloids
3.2.1. Alkaloids in Large-Leaved Kudingcha
3.2.2. Alkaloids in Yerba Mate
3.2.3. Alkaloids in Yaupon Holly
3.2.4. Alkaloids in Guayusa
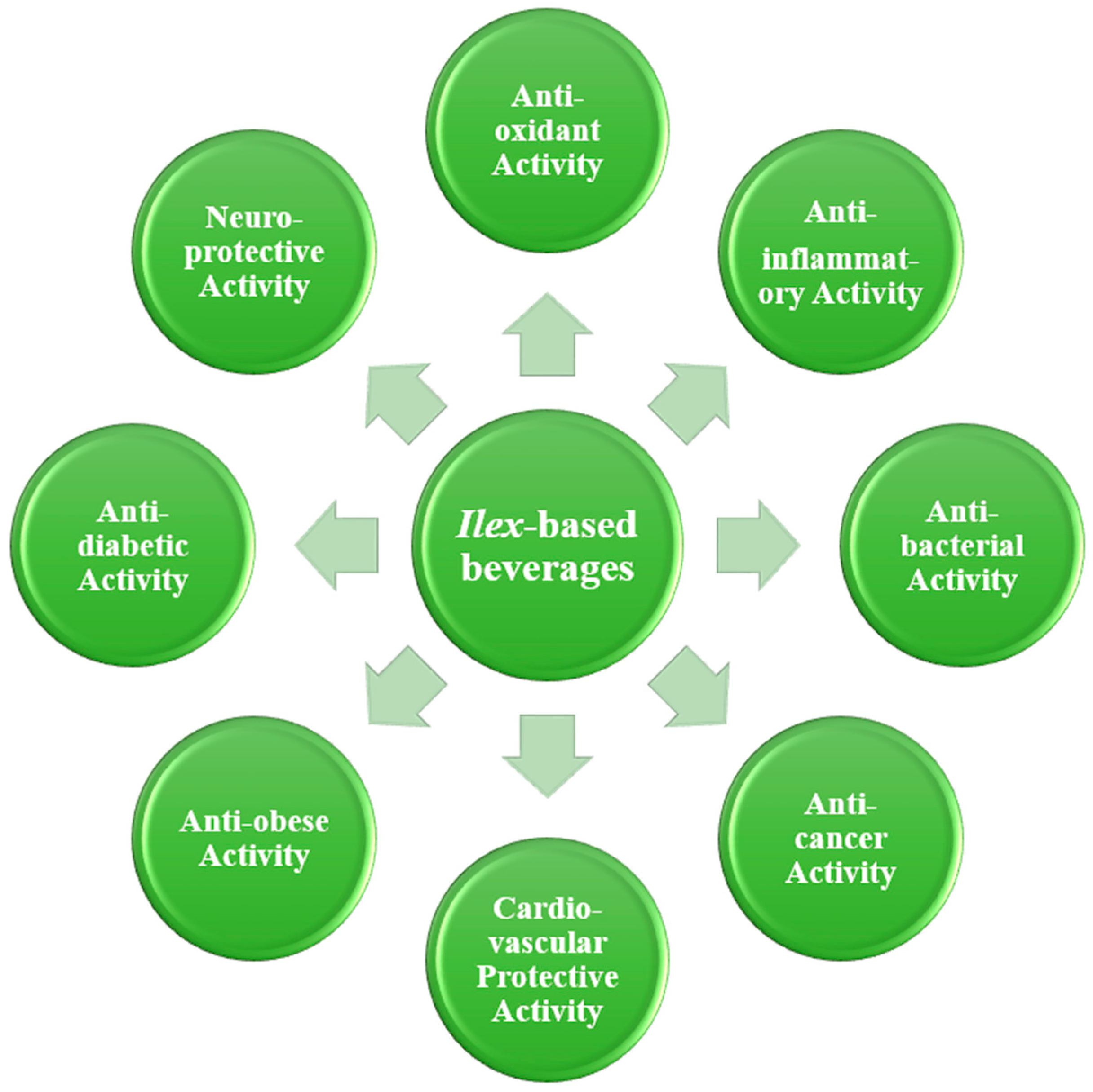
4. Health Benefits
4.1. Antioxidant Activity
4.2. Anti-Inflammatory Activity
4.3. Antibacterial Activity
4.4. Lipid-Reducing Activity
4.5. Regulation of Gut Microbiota
4.6. Anti-Cancer Activity
4.7. Cardiovascular Protective Activity
4.8. Anti-Obesity Activity
4.9. Anti-Diabetic Activity
4.10. Neuroprotective Activity
4.11. Other Health Benefits
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mohanpuria, P.; Kumar, V.; Yadav, S.K. Tea caffeine: Metabolism, functions, and reduction strategies. Food Sci. Biotechnol. 2010, 19, 275–287. [Google Scholar] [CrossRef]
- Zhang, C.; Suen, C.L.C.; Yang, C.; Quek, S.Y. Antioxidant capacity and major polyphenol composition of teas as affected by geographical location, plantation elevation and leaf grade. Food Chem. 2018, 244, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xu, L.J.; Ma, G.Z.; Dong, Y.M.; Peng, Y.; Xiao, P.G. The large-leaved kudingcha (Ilex latifolia thunb and Ilex kudingcha C.J. Tseng): A traditional Chinese tea with plentiful secondary metabolites and potential biological activities. J. Nat. Med. 2013, 67, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.L.; Wu, Z.W.; Zhao, T.H.; Sun, Y.; Ye, H.; Xu, R.J.; Zeng, X.X. Characterization, antioxidant and hepatoprotective activities of polysaccharides from Ilex latifolia Thunb. Carbohyd. Polym. 2014, 101, 990–997. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; He, X.W.; Jiang, J.G. Functional analyses on antioxidant, anti-inflammatory, and antiproliferative effects of extracts and compounds from Ilex latifolia thunb., a Chinese bitter tea. J. Agric. Food Chem. 2014, 62, 8608–8615. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Li, G.; Sun, P.; Wang, R.; Qian, Y.; Zhao, X. In vitro and in vivo anti-cancer activities of kuding tea (Ilex kudingcha C.J. Tseng) against oral cancer. Exp. Ther. Med. 2014, 7, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Q.; Li, M.M.; Zhang, W.; Wang, L.; Fan, C.L.; Feng, R.B.; Zhang, X.Q.; Ye, W.C. Four new triterpenes and triterpene glycosides from the leaves of Ilex latifolia and their inhibitory activity on triglyceride accumulation. Fitoterapia 2015, 106, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Wang, Q.; Zhang, W.; Hu, B.; Zhou, L.; Zeng, X.; Sun, Y. Inhibitory activities of caffeoylquinic acid derivatives from Ilex kudingcha C.J. Tseng on α-glucosidase from Saccharomyces cerevisiae. J. Agric. Food Chem. 2015, 63, 3694–3703. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Chen, G.; Wan, P.; Dai, Z.; Hu, B.; Chen, L.; Ou, S.; Zeng, X.; Sun, Y. Modulating effects of dicaffeoylquinic acids from Ilex kudingcha on intestinal microecology in vitro. J. Agric. Food Chem. 2017, 65, 10185–10196. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.-T.; Hu, T.; Jiang, J.-G.; Zhao, J.-W.; Zhu, W. Antioxidant and anti-inflammatory effects of polyphenols extracted from Ilex latifolia thunb. RSC Adv. 2018, 8, 7134–7141. [Google Scholar] [CrossRef]
- Burris, K.P.; Davidson, P.M.; Stewart, C.N., Jr.; Zivanovic, S.; Harte, F.M. Aqueous extracts of yerba mate (Ilex paraguariensis) as a natural antimicrobial against Escherichia coli O157:H7 in a microbiological medium and pH 6.0 apple juice. J. Food Prot. 2012, 75, 753–757. [Google Scholar] [CrossRef] [PubMed]
- Bassani, D.C.; Nunes, D.S.; Granato, D. Optimization of phenolics and flavonoids extraction conditions and antioxidant activity of roasted yerba-mate leaves (Ilex paraguariensis a. St.-Hil., aquifoliaceae) using response surface methodology. An. Acad. Bras. Cienc. 2014, 86, 923–933. [Google Scholar] [CrossRef]
- Molin, R.F.; Dartora, N.; Borges, A.C.P.; Gonçalves, I.L.; Di Luccio, M.; Valduga, A.T. Total phenolic contents and antioxidant activity in oxidized leaves of mate (Ilex paraguariensis St. Hil). Braz. Arch. Biol. Technol. 2014, 57, 997–1003. [Google Scholar] [CrossRef]
- Boado, L.S.; Fretes, R.M.; Brumovsky, L.A. Bioavailability and antioxidant effect of the Ilex paraguariensis polyphenols. Nutr. Food Sci. 2015, 45, 326–335. [Google Scholar] [CrossRef]
- Souza, A.H.P.; Correa, R.C.G.; Barros, L.; Calhelha, R.C.; Santos-Buelga, C.; Peralta, R.M.; Bracht, A.; Matsushita, M.; Ferreira, I.C.F.R. Phytochemicals and bioactive properties of Ilex paraguariensis: An in-vitro comparative study between the whole plant, leaves and stems. Food Res. Int. 2015, 78, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Colpo, A.C.; de Lima, M.E.; Maya-Lopez, M.; Rosa, H.; Marquez-Curiel, C.; Galvan-Arzate, S.; Santamaria, A.; Folmer, V. Compounds from Ilex paraguariensis extracts have antioxidant effects in the brains of rats subjected to chronic immobilization stress. Appl. Physiol. Nutr. Metab. 2017, 42, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Konieczynski, P.; Viapiana, A.; Wesolowski, M. Comparison of infusions from black and green teas (Camellia sinensis l. Kuntze) and erva-mate (Ilex paraguariensis a. St.-Hil.) based on the content of essential elements, secondary metabolites, and antioxidant activity. Food Anal. Method 2017, 10, 3063–3070. [Google Scholar] [CrossRef]
- Mateos, R.; Baeza, G.; Sarria, B.; Bravo, L. Improved LC-MSn characterization of hydroxycinnamic acid derivatives and flavonols in different commercial mate (Ilex paraguariensis) brands. Quantification of polyphenols, methylxanthines, and antioxidant activity. Food Chem. 2018, 241, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, M.J.; Talcott, S.T.; Putz, F.E. Ilex vomitoria ait. (yaupon): A native north American source of a caffeinated and antioxidant-rich tea. Econ. Bot. 2009, 63, 130–137. [Google Scholar] [CrossRef]
- Noratto, G.D.; Kim, Y.; Talcott, S.T.; Mertens-Talcott, S.U. Flavonol-rich fractions of yaupon holly leaves (Ilex vomitoria, aquifoliaceae) induce microRNA-146a and have anti-inflammatory and chemopreventive effects in intestinal myofribroblast CCD-18Co cells. Fitoterapia 2011, 82, 557–569. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Talcott, S.T. Tea creaming in nonfermented teas from Camellia sinensis and Ilex vomitoria. J. Agric. Food Chem. 2012, 60, 11793–11799. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Ruiz, A.; Baenas, N.; Benitez-Gonzalez, A.M.; Stinco, C.M.; Melendez-Martinez, A.J.; Moreno, D.A.; Ruales, J. Guayusa (Ilex guayusa L.) new tea: Phenolic and carotenoid composition and antioxidant capacity. J. Sci. Food Agric. 2017, 97, 3929–3936. [Google Scholar] [CrossRef] [PubMed]
- Pardau, M.D.; Pereira, A.S.P.; Apostolides, Z.; Serem, J.C.; Bester, M.J. Antioxidant and anti-inflammatory properties of Ilex guayusa tea preparations: A comparison to Camellia sinensis teas. Food Funct. 2017, 8, 4601–4610. [Google Scholar] [CrossRef] [PubMed]
- Villacis-Chiriboga, J.; Garcia-Ruiz, A.; Baenas, N.; Moreno, D.A.; Melendez-Martinez, A.J.; Stinco, C.M.; Jerves-Andrade, L.; Leon-Tamariz, F.; Ortiz-Ulloa, J.; Ruales, J. Changes in phytochemical composition, bioactivity and in vitro digestibility of guayusa leaves (Ilex guayusa loes.) in different ripening stages. J. Sci. Food Agric. 2018, 98, 1927–1934. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Cai, Y.Z.; Sun, M.; Ke, J.X.; Lu, D.Y.; Corke, H. Comparison of major phenolic constituents and in vitro antioxidant activity of diverse kudingcha genotypes from Ilex kudingcha, Ilex cornuta, and Ligustrum robustum. J. Agric. Food Chem. 2009, 57, 6082–6089. [Google Scholar] [CrossRef] [PubMed]
- Heck, C.I.; De Mejia, E.G. Yerba mate tea (Ilex paraguariensis): A comprehensive review on chemistry, health implications, and technological considerations. J. Food Sci. 2007, 72, R138–R151. [Google Scholar] [CrossRef] [PubMed]
- Burris, K.P.; Harte, F.M.; Davidson, P.M.; Stewart, C.N.; Zivanovic, S. Composition and bioactive properties of yerba mate (Ilex paraguariensis a. St.-Hil.): A review. Chil. J. Agric. Res. 2012, 72, 268–274. [Google Scholar] [CrossRef]
- Marcelo, M.C.A.; Martins, C.A.; Pozebon, D.; Dressler, V.L.; Ferrao, M.F. Classification of yerba mate (Ilex paraguariensis) according to the country of origin based on element concentrations. Microchem. J. 2014, 117, 164–171. [Google Scholar] [CrossRef]
- Palumbo, M.J.; Putz, F.E.; Talcott, S.T. Nitrogen fertilizer and gender effects on the secondary metabolism of yaupon, a caffeine-containing north American holly. Oecologia 2007, 151, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.; Zhou, J.; Shang, X.Y.; Zhao, Z.X.; Peng, Q.; Zhu, M.J.; Zhu, C.C.; Lin, C.Z.; Liu, Q.D.; Liao, Q.F.; et al. Multi-component analysis of Ilex kudingcha C. J. Tseng by a single marker quantification method and chemometric discrimination of HPLC fingerprints. Molecules 2018, 23, 854. [Google Scholar] [CrossRef] [PubMed]
- Mazur, L.; Peralta-Zamora, P.G.; Demczuk, B.; Ribani, R.H. Application of multivariate calibration and NIR spectroscopy for the quantification of methylxanthines in yerba mate (Ilex paraguariensis). J. Food Compos. Anal. 2014, 35, 55–60. [Google Scholar] [CrossRef]
- Pinto, R.M.C.; Lemes, B.M.; Zielinski, A.A.F.; Klein, T.; de Paula, F.; Kist, A.; Marques, A.S.F.; Nogueira, A.; Demiate, I.M.; Beltrame, F.L. Detection and quantification of phytochemical markers of Ilex paraguariensis by liquid chromatography. Quim. Nova 2015, 38, 1219–1225. [Google Scholar] [CrossRef]
- Mateos, R.; Baeza, G.; Martinez-Lopez, S.; Sarria, B.; Bravo, L. LC-MSn characterization of saponins in mate (Ilex paraguariens, St. Hil) and their quantification by HPLC-DAD. J. Food Compos. Anal. 2017, 63, 164–170. [Google Scholar] [CrossRef]
- King, A.; Powis, T.G.; Cheong, K.F.; Gaikwad, N.W. Cautionary tales on the identification of caffeinated beverages in north America. J. Archaeol. Sci. 2017, 85, 30–40. [Google Scholar] [CrossRef]
- Duenas, J.F.; Jarrett, C.; Cummins, I.; Logan-Hines, E. Amazonian guayusa (Ilex guayusa Loes.): A historical and ethnobotanical overview. Econ. Bot. 2016, 70, 85–91. [Google Scholar] [CrossRef]
- Kapp, R.W.; Mendes, O.; Roy, S.; McQuate, R.S.; Kraska, R. General and genetic toxicology of guayusa concentrate (Ilex guayusa). Int. J. Toxicol. 2016, 35, 222–242. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yi, H.; Zhao, Z.X.; Shang, X.Y.; Zhu, M.J.; Kuang, G.J.; Zhu, C.C.; Zhang, L. Simultaneous qualitative and quantitative evaluation of Ilex kudingcha C.J. Tseng by using UPLC and UHPLC-QTOE-MS/MS. J. Pharm. Biomed. 2018, 155, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; He, X.W.; Jiang, J.G.; Xu, X.L. Efficacy evaluation of a Chinese bitter tea (Ilex latifolia thunb.) via analyses of its main components. Food Funct. 2014, 5, 876–881. [Google Scholar] [CrossRef] [PubMed]
- Zhong, T.; Piao, L.H.; Kim, H.J.; Liu, X.D.; Jiang, S.N.; Liu, G.M. Chlorogenic acid-enriched extract of Ilex kudingcha C.J. Tseng inhibits angiogenesis in zebrafish. J. Med. Food 2017, 20, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Che, Y.Y.; Wang, Z.B.; Zhu, Z.Y.; Ma, Y.Y.; Zhang, Y.Q.; Gu, W.; Zhang, J.Y.; Rao, G.X. Simultaneous qualitation and quantitation of chlorogenic acids in kuding tea using ultra-high-performance liquid chromatography-diode array detection coupled with linear ion trap-orbitrap mass spectrometer. Molecules 2016, 21, 1728. [Google Scholar] [CrossRef] [PubMed]
- Zwyrzykowska, A.; Kupczynski, R.; Jarosz, B.; Szumny, A.; Kucharska, A.Z. Qualitative and quantitative analysis of polyphenolic compounds in Ilex sp. Open Chem. 2015, 13, 1303–1312. [Google Scholar] [CrossRef]
- da Silveira, T.F.F.; Meinhart, A.D.; Coutinho, J.P.; de Souza, T.C.L.; Cunha, E.C.E.; de Moraes, M.R.; Godoy, H.T. Content of lutein in aqueous extracts of yerba mate (Ilex paraguariensis St. Hil). Food Res. Int. 2016, 82, 165–171. [Google Scholar] [CrossRef]
- Kim, Y.; Welt, B.A.; Talcott, S.T. The impact of packaging materials on the antioxidant phytochemical stability of aqueous infusions of green tea (Camellia sinensis) and yaupon holly (Ilex vomitoria) during cold storage. J. Agric. Food Chem. 2011, 59, 4676–4683. [Google Scholar] [CrossRef] [PubMed]
- Boaventura, B.C.; Di Pietro, P.F.; Stefanuto, A.; Klein, G.A.; de Morais, E.C.; de Andrade, F.; Wazlawik, E.; da Silva, E.L. Association of mate tea (Ilex paraguariensis) intake and dietary intervention and effects on oxidative stress biomarkers of dyslipidemic subjects. Nutrition 2012, 28, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Barg, M.; Rezin, G.T.; Leffa, D.D.; Balbinot, F.; Gomes, L.M.; Carvalho-Silva, M.; Vuolo, F.; Petronilho, F.; Dal-Pizzol, F.; Streck, E.L.; et al. Evaluation of the protective effect of Ilex paraguariensis and Camellia sinensis extracts on the prevention of oxidative damage caused by ultraviolet radiation. Environ. Toxicol. Pharmacol. 2014, 37, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.A.F.; Tirapeli, K.G.; Chaves-Neto, A.H.; da Silva Brasilino, M.; da Rocha, C.Q.; Bello-Klein, A.; Llesuy, S.F.; Dornelles, R.C.M.; Nakamune, A. Ilex paraguariensis supplementation may be an effective nutritional approach to modulate oxidative stress during perimenopause. Exp. Gerontol. 2017, 90, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Boaventura, B.C.B.; Di Pietro, P.F.; Klein, G.A.; Stefanuto, A.; de Morais, E.C.; de Andrade, F.; Wazlawik, E.; da Silva, E.L. Antioxidant potential of mate tea (Ilex paraguariensis) in type 2 diabetic mellitus and pre-diabetic individuals. J. Funct. Foods 2013, 5, 1057–1064. [Google Scholar] [CrossRef]
- Bremer Boaventura, B.C.; da Silva, E.L.; Liu, R.H.; Prudêncio, E.S.; Di Pietro, P.F.; Becker, A.M.; Amboni, R.D.d.M.C. Effect of yerba mate (Ilex paraguariensis a. St. Hil.) infusion obtained by freeze concentration technology on antioxidant status of healthy individuals. LWT Food Sci. Technol. 2015, 62, 948–954. [Google Scholar] [CrossRef]
- Baeza, G.; Sarria, B.; Mateos, R.; Bravo, L. Dihydrocaffeic acid, a major microbial metabolite of chlorogenic acids, shows similar protective effect than a yerba mate phenolic extract against oxidative stress in HepG2 cells. Food Res. Int. 2016, 87, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Song, J.L.; Yi, R.; Li, G.; Sun, P.; Park, K.Y.; Suo, H. Comparison of antioxidative effects of insect tea and its raw tea (kuding tea) polyphenols in Kunming mice. Molecules 2018, 23, 204. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, C.C.; Calado, V.M.; Ares, G.; Granato, D. Statistical approaches to assess the association between phenolic compounds and the in vitro antioxidant activity of Camellia sinensis and Ilex paraguariensis teas. Crit. Rev. Food Sci. Nutr. 2015, 55, 1456–1473. [Google Scholar] [CrossRef] [PubMed]
- Yonny, M.E.; Medina, A.V.; Nazareno, M.A.; Chaillou, L.L. Enhancement in the oxidative stability of green peas by Ilex paraguariensis addition in a blanching process before their refrigerated and frozen storage. LWT 2018, 91, 315–321. [Google Scholar] [CrossRef]
- Song, J.L.; Qian, Y.; Li, G.J.; Zhao, X. Anti-inflammatory effects of kudingcha methanol extract (Ilex kudingcha c.J. Tseng) in dextran sulfate sodium-induced ulcerative colitis. Mol. Med. Rep. 2013, 8, 1256–1262. [Google Scholar] [CrossRef] [PubMed]
- Luz, A.B.G.; da Silva, C.H.B.; Nascimento, M.; de Campos Facchin, B.M.; Baratto, B.; Frode, T.S.; Reginatto, F.H.; Dalmarco, E.M. The anti-inflammatory effect of Ilex paraguariensis a. St. Hil (mate) in a murine model of pleurisy. Int. Immunopharmacol. 2016, 36, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Lanzetti, M.; Bezerra, F.S.; Romana-Souza, B.; Brando-Lima, A.C.; Koatz, V.L.; Porto, L.C.; Valenca, S.S. Mate tea reduced acute lung inflammation in mice exposed to cigarette smoke. Nutrition 2008, 24, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, G.D.; Lira, F.S.; Rosa, J.C.; Caris, A.V.; Pinheiro, F.; Ribeiro, E.B.; Oller do Nascimento, C.M.; Oyama, L.M. Yerba mate extract (Ilex paraguariensis) attenuates both central and peripheral inflammatory effects of diet-induced obesity in rats. J. Nutr. Biochem. 2013, 24, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Culla, M.; Saenz-Cuesta, M.; Guereca-Barandiaran, M.J.; Ribeiro, M.L.; Otaegui, D. Yerba mate (Ilex paraguariensis) inhibits lymphocyte activation in vitro. Food Funct. 2016, 7, 4556–4563. [Google Scholar] [CrossRef] [PubMed]
- Puangpraphant, S.; Dia, V.P.; de Mejia, E.G.; Garcia, G.; Berhow, M.A.; Wallig, M.A. Yerba mate tea and mate saponins prevented azoxymethane-induced inflammation of rat colon through suppression of NF-κB p65ser(311) signaling via IκB-α and GSK-3β reduced phosphorylation. Biofactors 2013, 39, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Schinella, G.; Neyret, E.; Console, G.; Tournier, H.; Prieto, J.M.; Rios, J.L.; Giner, R.M. An aqueous extract of Ilex paraguariensis reduces carrageenan-induced edema and inhibits the expression of cyclooxygenase-2 and inducible nitric oxide synthase in animal models of inflammation. Planta Med. 2014, 80, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Petrilli, A.A.; Souza, S.J.; Teixeira, A.M.; Pontilho, P.M.; Souza, J.M.P.; Luzia, L.A.; Rondo, P.H.C. Effect of Chocolate and Yerba Mate Phenolic Compounds on Inflammatory and Oxidative Biomarkers in HIV/AIDS Individuals. Nutrients 2016, 8, 132. [Google Scholar] [CrossRef] [PubMed]
- Prado Martin, J.G.; Porto, E.; de Alencar, S.M.; da Glória, E.M.; Corrêa, C.B.; Ribeiro Cabral, I.S. Antimicrobial activity of yerba mate (Ilex paraguariensis St. Hil.) against food pathogens. Rev. Argent. Microbiol. 2013, 45, 93–98. [Google Scholar] [CrossRef]
- Burris, K.P.; Davidson, P.M.; Stewart, C.N., Jr.; Harte, F.M. Antimicrobial activity of yerba mate (Ilex paraguariensis) aqueous extracts against Escherichia coli O157:H7 and Staphylococcus aureus. J. Food Sci. 2011, 76, M456–M462. [Google Scholar] [CrossRef] [PubMed]
- Burris, K.P.; Higginbotham, K.L.; Stewart, C.N. Aqueous extracts of yerba mate as bactericidal agents against methicillin-resistant Staphylococcus aureus in a microbiological medium and ground beef mixtures. Food Control. 2015, 50, 748–753. [Google Scholar] [CrossRef]
- Correa, V.G.; Goncalves, G.A.; de Sa-Nakanishi, A.B.; Ferreira, I.; Barros, L.; Dias, M.I.; Koehnlein, E.A.; de Souza, C.G.M.; Bracht, A.; Peralta, R.M. Effects of in vitro digestion and in vitro colonic fermentation on stability and functional properties of yerba mate (Ilex paraguariensis a. St. Hil.) beverages. Food Chem. 2017, 237, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Rempe, C.S.; Burris, K.P.; Woo, H.L.; Goodrich, B.; Gosnell, D.K.; Tschaplinski, T.J.; Stewart, C.N., Jr. Computational ranking of yerba mate small molecules based on their predicted contribution to antibacterial activity against methicillin-resistant Staphylococcus aureus. PLoS ONE 2015, 10, e0123925. [Google Scholar] [CrossRef] [PubMed]
- Rempe, C.S.; Lenaghan, S.C.; Burris, K.P.; Stewart, C.N. Metabolomic analysis of the mechanism of action of yerba mate aqueous extract on Salmonella enterica serovar Typhimurium. Metabolomics 2017, 13. [Google Scholar] [CrossRef]
- Song, C.; Yu, Q.; Li, X.; Jin, S.; Li, S.; Zhang, Y.; Jia, S.; Chen, C.; Xiang, Y.; Jiang, H. The hypolipidemic effect of total saponins from kuding tea in high-fat diet-induced hyperlipidemic mice and its composition characterized by UPLC-QTOF-MS/MS. J. Food Sci. 2016, 81, H1313–H1319. [Google Scholar] [CrossRef] [PubMed]
- Balzan, S.; Hernandes, A.; Reichert, C.L.; Donaduzzi, C.; Pires, V.A.; Gasparotto, A., Jr.; Cardozo, E.L., Jr. Lipid-lowering effects of standardized extracts of Ilex paraguariensis in high-fat-diet rats. Fitoterapia 2013, 86, 115–122. [Google Scholar] [CrossRef] [PubMed]
- de Resende, P.E.; Kaiser, S.; Pittol, V.; Hoefel, A.L.; D’Agostini Silva, R.; Vieira Marques, C.; Kucharski, L.C.; Ortega, G.G. Influence of crude extract and bioactive fractions of Ilex paraguariensis a. St. Hil. (yerba mate) on the Wistar rat lipid metabolism. J. Funct. Foods 2015, 15, 440–451. [Google Scholar] [CrossRef]
- Gao, H.; Long, Y.; Jiang, X.; Liu, Z.; Wang, D.; Zhao, Y.; Li, D.; Sun, B.L. Beneficial effects of yerba mate tea (Ilex paraguariensis) on hyperlipidemia in high-fat-fed hamsters. Exp. Gerontol. 2013, 48, 572–578. [Google Scholar] [CrossRef] [PubMed]
- de Morais, E.C.; Stefanuto, A.; Klein, G.A.; Boaventura, B.C.; de Andrade, F.; Wazlawik, E.; Di Pietro, P.F.; Maraschin, M.; da Silva, E.L. Consumption of yerba mate (Ilex paraguariensis) improves serum lipid parameters in healthy dyslipidemic subjects and provides an additional LDL-cholesterol reduction in individuals on statin therapy. J. Agric. Food Chem. 2009, 57, 8316–8324. [Google Scholar] [CrossRef] [PubMed]
- Messina, D.; Soto, C.; Mendez, A.; Corte, C.; Kemnitz, M.; Avena, V.; Del Balzo, D.; Perez Elizalde, R. Lipid—Lowering effect of mate tea intake in dyslipidemic subjects. Nutr. Hosp. 2015, 31, 2131–2139. [Google Scholar] [CrossRef] [PubMed]
- Chaves, G.; Britez, N.; Oviedo, G.; Gonzalez, G.; Italiano, C.; Blanes, M.; Sandoval, G.; Mereles, D. Heavy drinkers of Ilex paraguariensis beverages show lower lipid profiles but higher body weight. Phytother. Res. 2018, 32, 1030–1038. [Google Scholar] [CrossRef] [PubMed]
- Feng, R.B.; Fan, C.L.; Liu, Q.; Liu, Z.; Zhang, W.; Li, Y.L.; Tang, W.; Wang, Y.; Li, M.M.; Ye, W.C. Crude triterpenoid saponins from Ilex latifolia (Da Ye Dong Qing) ameliorate lipid accumulation by inhibiting SREBP expression via activation of AMPK in a non-alcoholic fatty liver disease model. Chin. Med. 2015, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Che, Y.Y.; Wang, Q.H.; Xiao, R.Y.; Zhang, J.Y.; Zhang, Y.Q.; Gu, W.; Rao, G.X.; Wang, C.F.; Kuang, H.X. Kudinoside-d, a triterpenoid saponin derived from Ilex kudingcha suppresses adipogenesis through modulation of the AMPK pathway in 3T3-L1 adipocytes. Fitoterapia 2018, 125, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Arcari, D.P.; Santos, J.C.; Gambero, A.; Ribeiro, M.L. The in vitro and in vivo effects of yerba mate (Ilex paraguariensis) extract on adipogenesis. Food Chem. 2013, 141, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gil, F.; Diaz-Sanchez, S.; Pendleton, S.; Andino, A.; Zhang, N.; Yard, C.; Crilly, N.; Harte, F.; Hanning, I. Yerba mate enhances probiotic bacteria growth in vitro but as a feed additive does not reduce Salmonella enteritidis colonization in vivo. Poult. Sci. 2014, 93, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xie, M.; Dai, Z.; Wan, P.; Ye, H.; Zeng, X.; Sun, Y. Kudingcha and fuzhuan brick tea prevent obesity and modulate gut microbiota in high-fat diet fed mice. Mol. Nutr. Food Res. 2018, 62, e1700485. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Chen, G.; Hu, B.; Zhou, L.; Ou, S.; Zeng, X.; Sun, Y. Hydrolysis of dicaffeoylquinic acids from Ilex kudingcha happens in the colon by intestinal microbiota. J. Agric. Food Chem. 2016, 64, 9624–9630. [Google Scholar] [CrossRef] [PubMed]
- Dasanayake, A.P.; Silverman, A.J.; Warnakulasuriya, S. Mate drinking and oral and oro-pharyngeal cancer: A systematic review and meta-analysis. Oral. Oncol. 2010, 46, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Amigo-Benavent, M.; Wang, S.; Mateos, R.; Sarria, B.; Bravo, L. Antiproliferative and cytotoxic effects of green coffee and yerba mate extracts, their main hydroxycinnamic acids, methylxanthine and metabolites in different human cell lines. Food Chem. Toxicol. 2017, 106, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Juaristi, M.; Martinez-Lopez, S.; Sarria, B.; Bravo, L.; Mateos, R. Absorption and metabolism of yerba mate phenolic compounds in humans. Food Chem. 2018, 240, 1028–1038. [Google Scholar] [CrossRef] [PubMed]
- Murad, L.D.; Soares Nda, C.; Brand, C.; Monteiro, M.C.; Teodoro, A.J. Effects of caffeic and 5-caffeoylquinic acids on cell viability and cellular uptake in human colon adenocarcinoma cells. Nutr. Cancer 2015, 67, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Ronco, A.L.; De Stefani, E.; Mendoza, B.; Deneo-Pellegrini, H.; Vazquez, A.; Abbona, E. Mate intake and risk of breast cancer in uruguay: A. case-control study. Asian Pac. J. Cancer Prev. 2016, 17, 1453–1461. [Google Scholar] [CrossRef] [PubMed]
- Puangpraphant, S.; Berhow, M.A.; Vermillion, K.; Potts, G.; Gonzalez de Mejia, E. Dicaffeoylquinic acids in yerba mate (Ilex paraguariensis St. Hilaire) inhibit NF-κB nucleus translocation in macrophages and induce apoptosis by activating caspases-8 and -3 in human colon cancer cells. Mol. Nutr. Food Res. 2011, 55, 1509–1522. [Google Scholar] [CrossRef] [PubMed]
- Zhai, X.; Ren, D.; Luo, Y.; Hu, Y.; Yang, X. Chemical characteristics of an Ilex kuding tea polysaccharide and its protective effects against high fructose-induced liver injury and vascular endothelial dysfunction in mice. Food Funct. 2017, 8, 2536–2547. [Google Scholar] [CrossRef] [PubMed]
- Cardozo Junior, E.L.; Morand, C. Interest of mate (Ilex paraguariensis a. St.-Hil.) as a new natural functional food to preserve human cardiovascular health—A review. J. Funct. Foods 2016, 21, 440–454. [Google Scholar] [CrossRef]
- Yu, S.; Yue, S.; Liu, Z.; Zhang, T.; Xiang, N.; Fu, H. Yerba mate (Ilex paraguariensis) improves microcirculation of volunteers with high blood viscosity: A. randomized, double-blind, placebo-controlled trial. Exp. Gerontol. 2015, 62, 14–22. [Google Scholar] [CrossRef] [PubMed]
- da Veiga, D.T.A.; Bringhenti, R.; Copes, R.; Tatsch, E.; Moresco, R.N.; Comim, F.V.; Premaor, M.O. Protective effect of yerba mate intake on the cardiovascular system: A post hoc analysis study in postmenopausal women. Braz. J. Med. Biol. Res. 2018, 51, e7253. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Chen, Y.L.; Yu, Y.; Zang, J.; Wu, Y.; He, Z. Ilex latifolia thunb protects mice from HFD-induced body weight gain. Sci. Rep. 2017, 7, 14660. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.D.; Bueno, A.L.; Gallon, C.W.; Gomes, L.F.; Kaiser, S.; Pavei, C.; Ortega, G.G.; Kucharski, L.C.; Jahn, M.P. The effect of aqueous extract of gross and commercial yerba mate (Ilex paraguariensis) on intra-abdominal and epididymal fat and glucose levels in male Wistar rats. Fitoterapia 2011, 82, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Lima Nda, S.; Franco, J.G.; Peixoto-Silva, N.; Maia, L.A.; Kaezer, A.; Felzenszwalb, I.; de Oliveira, E.; de Moura, E.G.; Lisboa, P.C. Ilex paraguariensis (yerba mate) improves endocrine and metabolic disorders in obese rats primed by early weaning. Eur. J. Nutr. 2014, 53, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Gambero, A.; Ribeiro, M.L. The positive effects of yerba mate (Ilex paraguariensis) in obesity. Nutrients 2015, 7, 730–750. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, E.; Lima, N.S.; Conceicao, E.P.S.; Peixoto-Silva, N.; Moura, E.G.; Lisboa, P.C. Treatment with Ilex paraguariensis (yerba mate) aqueous solution prevents hepatic redox imbalance, elevated triglycerides, and microsteatosis in overweight adult rats that were precociously weaned. Braz. J. Med. Biol. Res. 2018, 51, e7342. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.R.; Lee, H.Y.; Kim, J.H.; Moon, D.I.; Seo, M.Y.; Park, S.H.; Choi, K.H.; Kim, C.R.; Kim, S.H.; Oh, J.H.; et al. Anti-obesity and anti-diabetic effects of yerba mate (Ilex paraguariensis) in C57BL/6J mice fed a high-fat diet. Lab. Anim. Res. 2012, 28, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Hussein, G.M.E.; Matsuda, H.; Nakamura, S.; Hamao, M.; Akiyama, T.; Tamura, K.; Yoshikawa, M. Mate tea (Ilex paraguariensis) promotes satiety and body weight lowering in mice: Involvement of glucagon-like peptide-1. Biol. Pharm. Bull. 2011, 34, 1849–1855. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Oh, M.R.; Kim, M.G.; Chae, H.J.; Chae, S.W. Anti-obesity effects of yerba mate (Ilex paraguariensis): A randomized, double-blind, placebo-controlled clinical trial. BMC Complement Altern. Med. 2015, 15, 338. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, E.P.; Torezan, G.A.; Goncalves, L.D.; Corrente, J.E.; McLellan, K.C.P.; Burini, R.C. Acute intake of yerba mate increases energy expenditure of health young men: A. pilot study. Rbone-Rev. Bras. Obes. 2016, 10, 242–249. [Google Scholar]
- Maufrais, C.; Sarafian, D.; Dulloo, A.; Montani, J.P. Cardiovascular and metabolic responses to the ingestion of caffeinated herbal tea: Drink it hot or cold? Front. Physiol. 2018, 9, 315. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Clifford, M.N.; Sharp, P. Analysis of chlorogenic acids in beverages prepared from Chinese health foods and investigation, in vitro, of effects on glucose absorption in cultured Caco-2 cells. Food Chem. 2008, 108, 369–373. [Google Scholar] [CrossRef]
- Riachi, L.G.; De Maria, C.A.B. Yerba mate: An overview of physiological effects in humans. J. Funct. Foods 2017, 38, 308–320. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, H.K.; Hwang, B.Y.; Kim, S.; Yoo, J.K.; Seong, Y.H. Neuroprotection of Ilex latifolia and caffeoylquinic acid derivatives against excitotoxic and hypoxic damage of cultured rat cortical neurons. Arch. Pharm. Res. 2012, 35, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Lee, H.K.; Jang, J.Y.; Yoo, J.K.; Seong, Y.H. Ilex latifolia prevents amyloid beta protein (25–35)-induced memory impairment by inhibiting apoptosis and tau phosphorylation in mice. J. Med. Food 2015, 18, 1317–1326. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Jeong, H.Y.; Lee, H.K.; Yoo, J.K.; Bae, K.; Seong, Y.H. Protective effect of Ilex latifolia, a major component of “kudingcha”, against transient focal ischemia-induced neuronal damage in rats. J. Ethnopharmacol. 2011, 133, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Gatto, E.M.; Melcon, C.; Parisi, V.L.; Bartoloni, L.; Gonzalez, C.D. Inverse association between yerba mate consumption and idiopathic Parkinson’s disease. A case-control study. J. Neurol. Sci. 2015, 356, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Conforti, A.S.; Gallo, M.E.; Saravi, F.D. Yerba mate (Ilex paraguariensis) consumption is associated with higher bone mineral density in postmenopausal women. Bone 2012, 50, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Brasilino, M.D.S.; Stringhetta-Garcia, C.T.; Pereira, C.S.; Pereira, A.A.F.; Stringhetta, K.; Leopoldino, A.M.; Crivelini, M.M.; Ervolino, E.; Dornelles, R.C.M.; de Melo Stevanato Nakamune, A.C.; et al. Mate tea (Ilex paraguariensis) improves bone formation in the alveolar socket healing after tooth extraction in rats. Clin. Oral. Investig. 2018, 22, 1449–1461. [Google Scholar] [CrossRef] [PubMed]
| Common Name | Species | Distribution | References |
|---|---|---|---|
| Large-leaved Kudingcha | I. kudingcha C.J. Tseng | China: Guangxi; Guangdong; Hainan | [3,25,30] |
| I. latifolia Thunb. | China: Zhejiang; Jiangsu; Fujian; Anhui; Hainan | [3,6,7,10] | |
| Yerba Mate | Ilex paraguariensis A. St.-Hil | South America: Argentina; Brazil; Paraguay; Uruguay | [26,27,31,32,33] |
| Yaupon Holly | Ilex vomitoria | Southeastern United States | [19,21,29,34] |
| Guayusa | Ilex guayusa Loes | South America: Argentina, Southern of Brazil, Paraguay and Uruguay | [22,24,35,36] |
| Tea Name | Species | Main Polyphenols | Reference |
|---|---|---|---|
| Large-leaved Kudingcha | Ilex kudingcha C. J. Tseng | Neochlorogenic acid | [30,37] |
| Chlorogenic acid | |||
| Cryptochlorogenic acid | |||
| Protocatechuic acid | [37] | ||
| Caffeic acid | |||
| Isochlorogenic acid | |||
| Rutin | |||
| I. latifolia | Caffeic acid derivatives | [3] | |
| Ethyl caffeate | [5] | ||
| 3,4-di-O-caffeoylquinic acid methyl ester | |||
| 3,5-di-O-caffeoylquinic acid methyl ester | |||
| Chlorogenic acid | |||
| Yerba Mate | Ilex paraguariensis A. St.-Hil | Hydroxycinnamic acid derivatives | [18] |
| Flavonols | |||
| 3-caffeoylquinic acid | |||
| 5-caffeoylquinic acid | |||
| 4-caffeoylquinic acid | |||
| 3, 5-dicaffeoylquinic acid | |||
| Rutin | |||
| Yaupon holly | I. vomitoria | Rutin | [20,21] |
| Chlorogenic acid | [21] | ||
| Neochlorogenic acid | |||
| Cryptochlorogenic acid | |||
| Guayusa | I. guayusa | Chlorogenic acid | [22,24] |
| Isochlorogenic acid | [22] | ||
| Neochlorogenic acid |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gan, R.-Y.; Zhang, D.; Wang, M.; Corke, H. Health Benefits of Bioactive Compounds from the Genus Ilex, a Source of Traditional Caffeinated Beverages. Nutrients 2018, 10, 1682. https://doi.org/10.3390/nu10111682
Gan R-Y, Zhang D, Wang M, Corke H. Health Benefits of Bioactive Compounds from the Genus Ilex, a Source of Traditional Caffeinated Beverages. Nutrients. 2018; 10(11):1682. https://doi.org/10.3390/nu10111682
Chicago/Turabian StyleGan, Ren-You, Dan Zhang, Min Wang, and Harold Corke. 2018. "Health Benefits of Bioactive Compounds from the Genus Ilex, a Source of Traditional Caffeinated Beverages" Nutrients 10, no. 11: 1682. https://doi.org/10.3390/nu10111682
APA StyleGan, R.-Y., Zhang, D., Wang, M., & Corke, H. (2018). Health Benefits of Bioactive Compounds from the Genus Ilex, a Source of Traditional Caffeinated Beverages. Nutrients, 10(11), 1682. https://doi.org/10.3390/nu10111682





