The Role of Diet, Micronutrients and the Gut Microbiota in Age-Related Macular Degeneration: New Perspectives from the Gut–Retina Axis
Abstract
1. Introduction
2. Literature Review Method
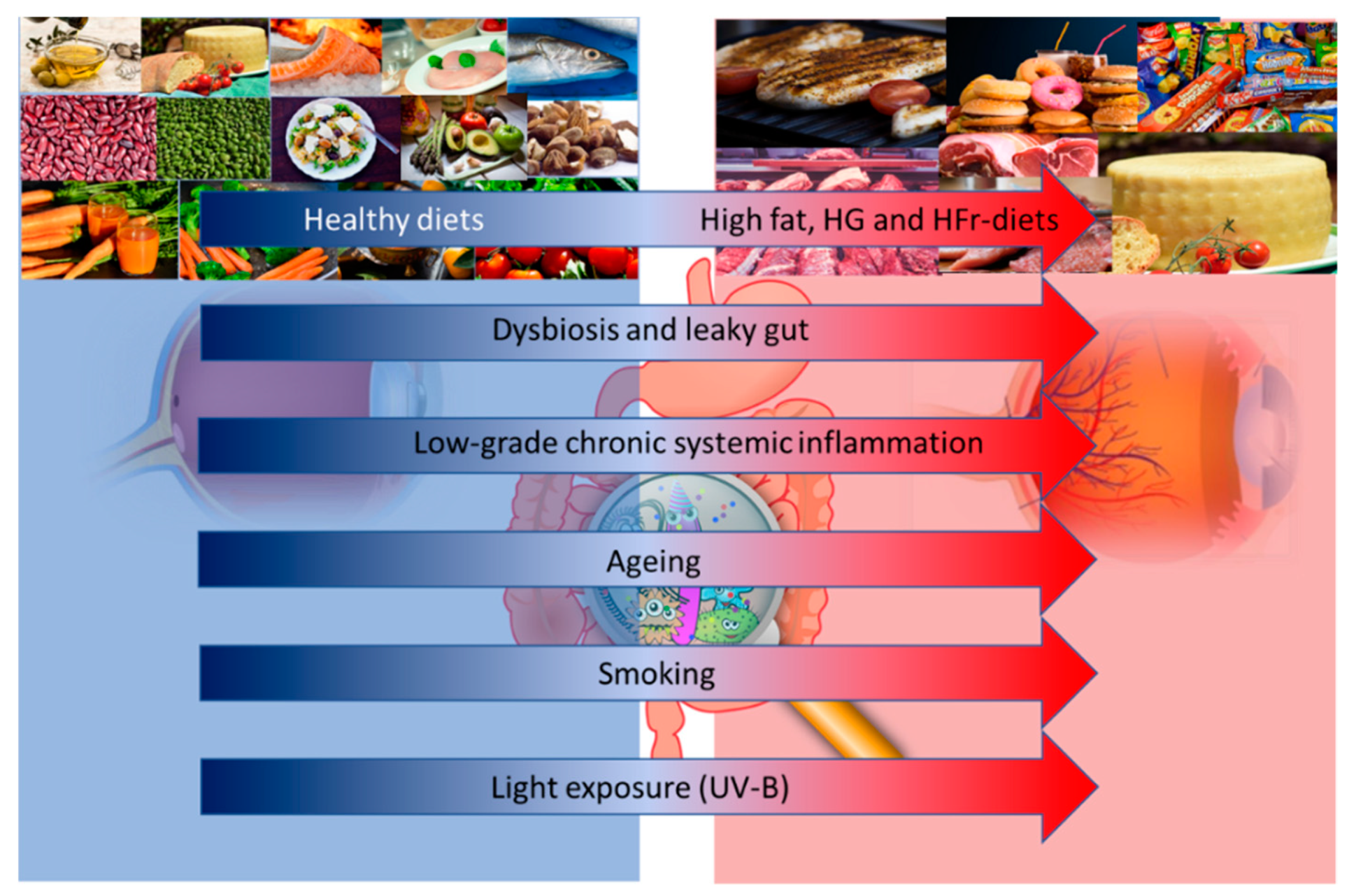
3. Diet and Gut-Microbiota in Age-Related Macular Degeneration (AMD)
3.1. Gut Microbiota: A Diet-Driven Ecosystem
3.2. High-Fat Diets and the Gut Microbiota in AMD
3.3. High-Glucose or -Fructose Diets and the Gut Microbiota in AMD
4. Micronutrients
4.1. Vitamins C and E
4.2. Zinc
4.3. Carotenoids
4.3.1. Beta-Carotene
4.3.2. Lutein and Zeaxanthin
4.4. Vitamin D
4.5. Omega-3 Fatty Acids
5. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health. 2014, 2, e106–e116. [Google Scholar] [CrossRef]
- Friedman, D.S.; O’Colmain, B.J.; Muñoz, B.; Tomany, S.C.; McCarty, C.; de Jong, P.T.; Nemesure, B.; Mitchell, P.; Kempen, J. Prevalence of age-related macular degeneration in the United States. Arch. Ophthalmol. 2004, 122, 564–572. [Google Scholar] [PubMed]
- Klein, R. Overview of progress in the epidemiology of age-related macular degeneration. Ophthalmic Epidemiol. 2007, 14, 184–187. [Google Scholar] [CrossRef] [PubMed]
- The Global Economic Cost of Visual Impairment, March 2010. Available online: http://www.icoph.org/dynamic/attachments/resources/globalcostofvi_finalreport.pdf. (accessed on 18 June 2018).
- de Jong, P.T. Age-related macular degeneration. N. Engl. J. Med. 2008, 358, 2606–2617. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.R.; Lawrenson, J.G. Antioxidant vitamin and mineral supplements for slowing the progression of age-related macular degeneration. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Carneiro, Â.; Andrade, J.P. Nutritional and Lifestyle Interventions for Age-Related Macular Degeneration: A Review. Oxid. Med. Cell Longev. 2017. [Google Scholar] [CrossRef]
- Margrain, T.H.; Boulton, M.; Marshall, J.; Sliney, D.H. Do blue light filters confer protection against age-related macular degeneration? Prog. Retin. Eye Res. 2004, 23, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Y.; Tie, L.J.; Wu, S.S.; Lv, P.L.; Huang, H.W.; Wang, W.Q.; Wang, H.; Ma, L. Overweight, obesity, and risk of age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2016, 57, 1276–1283. [Google Scholar] [CrossRef] [PubMed]
- Peeters, A.; Magliano, D.J.; Stevens, J.; Duncan, B.B.; Klein, R.; Wong, T.Y. Changes in abdominal obesity and age-related macular degeneration: The Atherosclerosis Risk in Communities Study. Arch. Ophthalmol. 2008. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.J.; Taylor, A. Dietary hyperglycemia, glycemic index and metabolic retinal diseases. Prog. Retin. Eye Res. 2011, 30, 18–53. [Google Scholar] [CrossRef] [PubMed]
- Lau, K.; Srivatsav, V.; Rizwan, A.; Nashed, A.; Liu, R.; Shen, R.; Akhtar, M. Bridging the gap between gut microbial dysbiosis and cardiovascular diseases. Nutrients 2017, 9, 859. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhou, Q.; Li, H. Gut microbiota and nonalcoholic fatty liver disease: insights on mechanisms and therapy. Nutrients 2017, 9, 1124. [Google Scholar] [CrossRef] [PubMed]
- Ponziani, F.R.; Bhoori, S.; Castelli, C.; Putignani, L.; Rivoltini, L.; Del Chierico, F.; Sanguinetti, M.; Morelli, D.; Paroni Sterbini, F.; Petito, V.; et al. Hepatocellular carcinoma is associated with gut microbiota profile and inflammation in nonalcoholic fatty liver disease. Hepatology 2018. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Andriessen, E.M.; Wilson, A.M.; Mawambo, G.; Dejda, A.; Miloudi, K.; Sennlaub, F.; Sapieha, P. Gut microbiota influences pathological angiogenesis in obesity-driven choroidal neovascularization. EMBO Mol. Med. 2016, 8, 1366–1379. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Lauretani, F.; Milani, C.; Nouvenne, A.; Tana, C.; Del Rio, D.; Maggio, M.; Ventura, M.; Meschi, T. Aging gut microbiota at the cross-road between nutrition, physical frailty, and sarcopenia: is there a gut-muscle axis? Nutrients 2017, 9, 1303. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Fuchs, S.; Milo, R. Are we really vastly outnumbered? Revisiting the ratio of bacterial to host cells in humans. Cell 2016, 164, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Laterza, L.; Rizzatti, G.; Gaetani, E.; Chiusolo, P.; Gasbarrini, A. The gut microbiota and immune system relationship in human graft-versus-host disease. Mediterr. J. Hematol. Infect Dis. 2016, 8, e2016025. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.F.; Murphy, E.F.; Nilaweera, K.; Ross, P.R.; Shanahan, F.; O’Toole, P.W.; Cotter, P.D. The gut microbiota and its relationship to diet and obesity: New insights. Gut Microbes 2012, 3, 186–202. [Google Scholar] [CrossRef] [PubMed]
- Rabot, S.; Membrez, M.; Bruneau, A.; Gérard, P.; Harach, T.; Moser, M.; Raymond, F.; Mansourian, R.; Chou, C.J. Germ-free C57BL/6J mice are resistant to high-fat-diet-induced insulin resistance and have altered cholesterol metabolism. FASEB J. 2010, 24, 4948–4959. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.S.; Mitchell, P.; Seddon, J.M.; Holz, F.G.; Wong, T.Y. Age-related macular degeneration. Lancet 2012, 379, 1728–1738. [Google Scholar] [CrossRef]
- Chiu, C.J.; Chang, M.L.; Zhang, F.F.; Li, T.; Gensler, G.; Schleicher, M.; Taylor, A. The relationship of major American dietary patterns to age-related macular degeneration. Am. J. Ophthalmol. 2014, 158, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.K.; Simpson, J.A.; Aung, K.Z.; Makeyeva, G.A.; Giles, G.G.; English, D.R.; Hopper, J.; Guymer, R.H.; Baird, P.N.; Robman, L.D. Abdominal obesity and age-related macular degeneration. Am. J. Epidemiol. 2011, 173, 1246–1255. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L. Sugar consumption, metabolic disease and obesity: The state of the controversy. Crit. Rev. Clin. Lab. Sci. 2016, 53, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.M.; Campbell, F.M.; Drew, J.E.; Koch, C.; Hoggard, N.; Rees, W.D.; Kamolrat, T.; Ngo, H.T.; Steffensen, I.-L.; Gray, S.R. The development of diet-induced obesity and glucose intolerance in c57bl/6 mice on a high-fat diet consists of distinct phases. PLoS ONE 2014, 9, e106159. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.M.; Jeong, J.J.; Woo, K.H.; Han, M.J.; Kim, D.H. Lactobacillus sakei ok67 ameliorates high-fat diet–induced blood glucose intolerance and obesity in mice by inhibiting gut microbiota lipopolysaccharide production and inducing colon tight junction protein expression. Nutr. Res. 2016, 36, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Schleicher, M.; Weikel, K.; Garber, C.; Taylor, A. Diminishing risk for age-related macular degeneration with nutrition: A current view. Nutrients 2013, 5, 2405–2456. [Google Scholar] [CrossRef] [PubMed]
- Do, M.H.; Lee, E.; Oh, M.J.; Kim, Y.; Park, H.Y. High-Glucose or -Fructose Diet Cause Changes of the Gut Microbiota and Metabolic Disorders in Mice without Body Weight Change. Nutrients 2018, 10, 761. [Google Scholar] [CrossRef] [PubMed]
- Rizzatti, G.; Lopetuso, L.R.; Gibino, G.; Binda, C.; Gasbarrini, A. Proteobacteria: A common factor in human diseases. Biomed. Res. Int. 2017. [Google Scholar] [CrossRef] [PubMed]
- Rowan, S.; Jiang, S.; Korem, T.; Szymanski, J.; Chang, M.L.; Szelog, J.; Cassalman, C.; Dasuri, K.; McGuire, C.; Nagai, R.; et al. Involvement of a gut-retina axis in protection against dietary glycemia-induced age-related macular degeneration. Proc. Natl. Acad. Sci. USA 2017, 114, E4472–E4481. [Google Scholar] [CrossRef] [PubMed]
- Schmidl, D.; Garhöfer, G.; Schmetterer, L. Nutritional supplements in age-related macular degeneration. Acta Ophthalmol. 2015, 93, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss. Arch. Ophthalmol. 2001, 119, 1417–1436. [Google Scholar] [CrossRef]
- Chew, E.Y.; Clemons, T.E.; SanGiovanni, J.P.; Danis, R.; Ferris, F.L.; Elman, M.; Antoszyk, A.; Ruby, A.; Orth, D.; Bressler, S.; et al. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA 2013, 309, 2005–2015. [Google Scholar]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report No. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Delcourt, C.; Carrière, I.; Delage, M.; Barberger-Gateau, P.; Schalch, W. Plasma Lutein and Zeaxanthin and Other Carotenoids as Modifiable Risk Factors for Age-Related Maculopathy and Cataract: The POLA Study. Investig. Ophthalmol. Vis. Sci. 2006, 47, 2329–2335. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; George, S.; Rosner, B. Cigarette, smoking, fish consumption, omega 3 fatty acid intake and association with age-related macular degeneration: The US Twin Study of age-related macular degeneration. Arch. Ophthlmol. 2006, 124, 995–1001. [Google Scholar] [CrossRef] [PubMed]
- Chua, B.; Flood, V.; Rochtchina, E.; Wang, J.J.; Smith, W.; Mitchell, P. Dietary fatty acids and the 5-year incidence of age-related maculopathy. Arch. Ophthalmol. 2006, 124, 981–986. [Google Scholar] [CrossRef] [PubMed]
- SanGiovanni, J.P.; Chew, E.Y.; Clemons, T.E.; Davis, M.D.; Ferris, F.L. 3rd; Gensler, G.R.; Kurinij, N.; Lindblad, A.S.; Milton, R.C.; Seddon, J.M.; et al. The relationship of dietary lipid intake and age-related macular degeneration in a case-control study: AREDS Report No. 20. Arch. Ophthalmol. 2007, 125, 671–679. [Google Scholar] [PubMed]
- Age-Related Eye Disease Study Research Group. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study: AREDS Report No. 22. Arch. Ophthalmol. 2007, 125, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- SanGiovanni, J.P.; Chew, E.Y.; Agrón, E.; Clemons, T.E.; Ferris, F.L., 3rd; Gensler, G.; Lindblad, A.S.; Milton, R.C.; Seddon, J.M.; Klein, R.; et al. The relationship of dietary omega-3 long-chain polyunsaturated fatty acid intake with incident age-related macular degeneration: AREDS report No. 23. Arch. Ophthalmol. 2008, 126, 1274–1279. [Google Scholar] [PubMed]
- Stringham, J.M.; Hammond, B.R. Macular pigment and visual performance under glare conditions. Optom. Vis. Sci. 2008, 85, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Hankinson, S.E.; Rosner, B.; Willett, W.C.; Colditz, G.A. Prospective study of lutein/zeaxanthin intake and risk of age-related macular degeneration. Am. J. Clin. Nutr. 2008, 87, 1837–1843. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.S.; Wang, J.J.; Flood, V.; Rochtchina, E.; Smith, W.; Mitchell, P. Dietary antioxidants and the long-term incidence of age-related macular degeneration: The Blue Mountains eye study. Ophthalmology 2008, 115, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Newsome, D.A. A randomized, prospective, placebo-controlled clinical trial of a novel zinc-monocysteine compound in age-related macular degeneration. Curr. Eye Res. 2008, 33, 591–598. [Google Scholar] [CrossRef] [PubMed]
- SanGiovanni, J.P.; Agron, E.; Clemons, T.E.; Chew, E.Y. Omega-3 long-chain polyunsaturated fatty acid intake inversely associated with 12-year progression to advanced age-related macular degeneration. Arch. Ophthalmol. 2009, 127, 110–112. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.; van Leeuwen, R.; Witteman, J.C.; van Duijn, C.M.; Uitterlinden, A.G.; Hofman, A.; de Jong, P.T.; Vingerling, J.R.; Klaver, C.C. Reducing the genetic risk of age-related macular degeneration with dietary antioxidants, zinc, and omega-3 fatty acids: The Rotterdam study. Arch. Ophthalmol. 2011, 129, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Snellen, E.L.M.; Verbeek, A.L.M.; van den Hoogen, G.W.P.; Cruysberg, J.R.M.; Hoyng, C.B. Neovascular age-related macular degeneration and its relationship to antioxidant intake. Acta Ophthalmol. Scand. 2002, 80, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Akkali, M.C.; Loughman, J.; Howard, A.N.; Beatty, S. Macular carotenoid supplementation in subjects with atypical spatial profiles of macular pigment. Exp. Eye Res. 2012, 101, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Souied, E.H.; Delcourt, C.; Querques, G.; Bassols, A.; Merle, B.; Zourdani, A.; Smith, T.; Benlian, P. Nutritional AMD Treatment 2 Study Group. Oral docosahexaenoic acid in the prevention of exudative age-related macular degeneration: The Nutritional AMD Treatment 2 study. Ophthalmology 2013, 120, 1619–1631. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R.; Fletcher, L.M.; Roos, F. A double-blind, placebo- controlled study on the effects of lutein and zeaxanthin on photostress recovery, glare disability, and chromatic contrast. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8583–8589. [Google Scholar] [CrossRef] [PubMed]
- Aoki, A.; Inoue, M.; Nguyen, E. Dietary n-3 Fatty Acid, α-Tocopherol, Zinc, vitamin D, vitamin C, and β-carotene are Associated with Age-Related Macular Degeneration in Japan. Sci. Rep. 2016, 6, 20723. [Google Scholar] [CrossRef] [PubMed]
- Braakhuis, A.; Raman, R.; Vaghefi, E. The Association between Dietary Intake of Antioxidants and Ocular Disease. Diseases 2017, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Pizzoferrato, M.; Cintoni, M.; Servidei, S.; Mele, M.C. Nutritional support in mitochondrial diseases: The state of the art. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 4288–4298. [Google Scholar] [PubMed]
- Ambati, J.; Fowler, B.J. Mechanisms of age-related macular degeneration. Neuron 2012, 75, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Schwenger, K.J.; Allard, J.P. Clinical approaches to non-alcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 1712–1723. [Google Scholar] [CrossRef] [PubMed]
- Singh, U.; Devaraj, S.; Jialal, I. Vitamin E, oxidative stress, and inflammation. Annu Rev. Nutr. 2005, 25, 151–174. [Google Scholar] [CrossRef] [PubMed]
- Westergren, T.; Kalikstad, B. Dosage and formulation issues: Oral vitamin E therapy in children. Eur. J. Clin. Pharmacol. 2010, 66, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, V.S.; Srinivasan, P.; Wildman, A.; Marchant, J.S.; Said, H.M. Molecular mechanism(s) involved in differential expression of vitamin C transporters along the intestinal tract. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 312, G340–G347. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Y.; Tso, M.O.; Wang, H.M.; Organisciak, D.T. Amelioration of photic injury in rat retina by ascorbic acid: A histopathologic study. Investig. Ophthalmol. Vis. Sci. 1985, 26, 1589–1598. [Google Scholar]
- VandenLangerberg, G.M.; Mares-Perlman, J.A.; Klein, R. Associations between antioxidant and zinc intake and the 5-years incidence of early age-related maculopathy in the Beaver Dam Eye Study. Am. J. Epidemiol. 1998, 148, 204–214. [Google Scholar] [CrossRef]
- Qiao, Y.; Sun, J.; Ding, Y.; Le, G.; Shi, Y. Alterations of the gut microbiota in high-fat diet mice is strongly linked to oxidative stress. Appl. Microbiol. Biotechnol. 2013, 97, 1689–1697. [Google Scholar] [CrossRef] [PubMed]
- Kruidenier, L.; Kuiper, I.; van Duijn, W.; Marklund, S.L.; van Hogezand, R.A.; Lamers, C.B.; Verspaget, H.W. Differential mucosal expression of three superoxide dismutase isoforms in inflammatory bowel disease. J. Pathol. 2003, 201, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, K.; Kanai, T. The gut microbiota and inflammatory bowel disease. Semin. Immunopathol. 2015, 37, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.K.; Kamm, M.A.; Teo, S.M.; Inouye, M.; Wagner, J.; Kirkwood, C.D. Recent advances in characterizing the gastrointestinal microbiome in Crohn’s disease: A systematic review. Inflamm. Bowel Dis. 2015, 21, 1219–1228. [Google Scholar] [PubMed]
- Yan, H.; Wang, H.; Zhang, X.; Li, X.; Yu, J. Ascorbic acid ameliorates oxidative stress and inflammation in dextran sulfate sodium-induced ulcerative colitis in mice. Int. J. Clin. Exp. Med. 2015, 8, 20245–20253. [Google Scholar] [PubMed]
- Tahan, G.; Aytac, E.; Aytekin, H.; Gunduz, F.; Dogusoy, G.; Aydin, S.; Tahan, V.; Uzun, H. Vitamin E has a dual effect of anti-inflammatory and antioxidant activities in acetic acid-induced ulcerative colitis in rats. Can. J. Surg. 2011, 54, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Maggini, M.; Eva, S.W.; Beveridge, S.; Dietrich, H.H. Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Br. J. Nutr. 2007, 98, S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Pierre, J.F.; Hinterleitner, R.; Bouziat, R.; Hubert, N.A.; Leone, V.; Miyoshi, J.; Jabri, B.; Chang, E.B. Dietary antioxidant micronutrients alter mucosal inflammatory risk in a murine model of genetic and microbial susceptibility. J. Nutr. Biochem. 2018, 54, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Godfrey, K.M.; McDonald, D.; Treuren, W.V.; Bjørnholt, J.V.; Midtvedt, T.; Moen, B.; Rudi, K.; Knight, R.; Brantsæter, A.L.; et al. Fat and vitamin intakes during pregnancy have stronger relations with a pro-inflammatory maternal microbiota than does carbohydrate intake. Microbiome 2016, 4, 55. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Krause, L.; Somerset, S. Associations between micronutrient intakes and gut microbiota in a group of adults with cystic fibrosis. Clin. Nutr. 2017, 36, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Frank, D.N.; Sherlock, L.; Ir, D.; Robertson, C.E.; Krebs, N.F. Effect of vitamin E with therapeutic iron supplementation on iron repletion and gut microbiome in US iron deficient infants and toddlers. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xu, C.; Chen, X. Regulation of an antioxidant blend on intestinal redox status and major microbiota in early weaned piglets. Nutrition 2014, 30, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Packer, L.; Colman, C. The Antioxidant Miracle; Wiley & Sons: New York, NY, USA, 1999. [Google Scholar]
- Skrypnik, K.; Suliburska, J. Association between the gut microbiota and mineral metabolism. J. Sci. Food Agric. 2017, 98, 2449–2460. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yi, L.; Zhao, M.L. Effects of zinc–methionine on growth performance, intestinal flora and immune function in pigeon squabs. Br. Poult. Sci. 2014, 3, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Leure-duPree, A.E.; McClain, C.J. The Effect of severe zinc deficiency on the morphology of the rat retinal pigment epithelium. Investig. Ophthalmol. Vis. Sci. 1982, 23, 425–434. [Google Scholar]
- Smailhodzic, D.; van Asten, F.; Blom, A.M.; Mohlin, F.C.; den Hollander, A.I.; van de Ven, J.P.; van Huet, R.A.; Groenewoud, J.M.; Tian, Y.; Berendschot, T.T.; et al. Zinc supplementation inhibits complement activation in age-related macular degeneration. PLoS ONE 2014, 9, e112682. [Google Scholar] [CrossRef] [PubMed]
- Zampatti, S.; Ricci, F.; Cusumano, A.; Marsella, L.T.; Novelli, G.; Giardina, E. Review of nutrient actions on age-related macular degeneration. Nutr. Res. 2014, 34, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Hernandez, J.; Mullins, R.F. Effects of antioxidant components of AREDS vitamins and zinc ions on endothelial cell activation: Implications for macular degeneration. Investig. Ophthalmol. Vis. Sci. 2012, 53, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Organisciak, D.; Wong, P.; Rapp, C.; Darrow, R.; Ziesel, A.; Rangarajan, R.; Lang, J. Light-induced retinal degeneration is prevented by zinc, a component in the agerelated eye disease study formulation. Photochem. Photobiol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.P.; Osborne, N.N. Zinc and energy requirements in induction of oxidative stress to retinal pigmented epithelial cells. Neurochem. Res. 2003, 28, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Gielda, L.M.; DiRita, V.J. Zinc competition among the intestinal microbiota. MBio 2012. [Google Scholar] [CrossRef] [PubMed]
- Zackular, J.; Moore, A.J. Dietary Zinc Alters the Microbiota and Decreases Resistance to Clostridium difficile Infection. Nat. Med. 2016, 22, 1330–1334. [Google Scholar] [CrossRef] [PubMed]
- Reed, S.; Neuman, H.; Moscovich, S. Chronic Zinc Deficiency Alters Chick Gut Microbiota Composition and Function. Nutrients 2015, 7, 9768–9784. [Google Scholar] [CrossRef] [PubMed]
- Watson, H.; Mitra, S.; Croden, F.C.; Taylor, M.; Wood, H.M.; Perry, S.L.; Spencer, J.A.; Quirke, P.; Toogood, G.J.; Lawton, C.L.; et al. A randomised trial of the effect of omega-3 polyunsaturated fatty acid supplements on the human intestinal microbiota. Gut 2017. [Google Scholar] [CrossRef] [PubMed]
- Engberg, R.M.; Hedemann, M.S.; Leser, T.D.; Jensen, B.B. Effect of zinc bacitracin and salinomycin on intestinal microflora and performance of broilers. Poult. Sci. 2000, 79, 1311–1319. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Zhao, Z.; Wang, Y.; Huang, K. Preparation of selenium/zinc-enriched probiotics and their effect on blood selenium and zinc concentrations, antioxidant capacities, and intestinal microflora in canine. Biol. Trace Elem. Res. 2011, 141, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Chhikara, N.; Kushwaha, K.; Sharma, P.; Gat, Y.; Panghal, A. Bioactive compounds of beetroot and utilization in food processing industry: A critical review. Food Chem. 2019, 272, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Castenmiller, J.J.; West, C.E. Bioavailability and bioconversion of carotenoids. Annu. Rev. Nutr. 1998, 18, 19–38. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Abou, L.; Mikail, C.; Ghiringhelli, O.; André, M.; Portugal, H.; Jourdheuil-Rahmani, D.; Amiot, M.J.; Lairon, D.; Borel, P. Lutein transport by Caco-2 TC-7 cells occurs partly by a facilitated process involving the scavenger receptor class B type I (SR-BI). Biochem. J. 2005, 387, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Van Bennekum, A.; Werder, M.; Thuahnai, S.T.; Han, C.H.; Duong, P.; Williams, D.L.; Wettstein, P.; Schulthess, G.; Philips, M.C.; Hauser, H. Class B scavenger receptor-mediated intestinal absorption of dietary beta-carotene and cholesterol. Biochemistry 2005, 44, 4517–4525. [Google Scholar] [CrossRef] [PubMed]
- During, A.; Dawson, H.D.; Harrison, E.H. Carotenoid transport is decreased and expression of the lipid transporters SR-BI, NPC1L1, and ABCA1 is downregulated in Caco-2 cells treated with ezetimibe. J. Nutr. 2005, 135, 2305–2312. [Google Scholar] [CrossRef] [PubMed]
- Binxing, L.; Preejith, P.V.; Zhengqing, S. Retinal Accumulation of Zeaxanthin, Lutein, and β-Carotene in Mice Deficient in Carotenoid Cleavage Enzymes. Exp. Eye Res. 2017, 159, 123–131. [Google Scholar]
- Leung, I.Y.; Sandstrom, M.M.; Zucker, C.L.; Neuringer, M.; Max Snodderly, D. Nutritional manipulation of primate retinas. IV Effects of n--3 fatty acids, lutein, and zeaxanthin on S-cones and rods in the foveal region. Exp. Eye Res. 2005, 81, 513–529. [Google Scholar] [CrossRef] [PubMed]
- Trevithick-Sutton, C.C.; Foote, C.S.; Collins, M.; Trevithick, J.R. The retinal carotenoids, zeaxanthin and lutein scavenge superoxide and hydroxyl radicals: A chemiluminescence and ESR study. Mol. Vis. 2006, 12, 1127–1135. [Google Scholar] [PubMed]
- Vu, H.T.; Robman, L.; McCarty, C.A.; Taylor, H.R.; Hodge, A. Does dietary lutein and zeaxanthin increase the risk of age related macular degeneration? The Melbourne Visual Impairment Project. Br. J. Ophthalmol. 2006, 90, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Landrum, J.T.; Bone, R.A.; Joa, H.; Kilburn, M.D.; Moore, L.L.; Sprague, K.E. A one year study of the macular pigment: The effect of 140 days of a lutein supplement. Exp. Eye Res. 1997, 65, 57–62. [Google Scholar] [CrossRef] [PubMed]
- LaRowe, T.L.; Mares, J.A.; Snodderly, D.M.; Klein, M.L.; Wooten, B.R.; Chappell, R. Macular pigment density and age-related maculopathy in the Carotenoids in Age-Related Eye Disease Study. An ancillary study of the women’s health initiative. Ophthalmology 2008, 115, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Power, R.; Stringham, J.; Dennison, J.; Stack, J.; Kelly, D.; Moran, R.; Akuffo, K.O.; Corcoran, L.; Beatty, S. Enrichment of Macular Pigment Enhances Contrast Sensitivity in Subjects Free of Retinal Disease: Central Retinal Enrichment Supplementation Trials-Report 1. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3429–3439. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y. Nutrition Effects on Ocular Diseases in the Aging Eye. Investig. Ophthalmol. Vis. Sci. 2013, 54, ORSF42–ORSF47. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, F.H.; Fåk, F.; Nookaew, I.; Tremaroli, V.; Fagerberg, B.; Petranovic, D.; Bäckhed, F.; Nielsen, J. Symptomatic atherosclerosis is associated with an altered gut metagenome. Nat. Commun. 2012, 3, 1245. [Google Scholar] [CrossRef] [PubMed]
- Arteni, A.A.; Fradot, M.; Galzerano, D.; Mendes-Pinto, M.M.; Sahel, J.A.; Picaud, S.; Robert, B.; Pascal, A.A. Structure and conformation of the carotenoids in human retinal macular pigment. PLoS ONE 2015, 10, e0135779. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, H.; Howells, O.; Eperjesi, F. The role of macular pigment assessment in clinical practice: A review. Clin. Exp. Optom. 2010, 93, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Meagher, K.; Kashani, S.; Beatty, S. What is meso-zeaxanthin, and where does it come from? Eye 2013, 27, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Hadden, W.L.; Watkins, R.H.; Levy, L.W.; Regalado, E.; Rivadeneira, D.M.; van Breemen, R.B.; Schwartz, S.J. Carotenoids composition of marigold (Tagetes erecta) flower extract used as nutritional supplement. J. Agric. Food Chem. 1999, 47, 4189–4194. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Stack, J.; O’ Donovan, O.; Loane, E.; Beatty, S. Risk factors for age-related maculopathy are associated with a relative lack of macular pigment. Exp. Eye Res. 2007, 84, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, B.; Natoli, S.; Liew, G.; Flood, V.M. Lutein and Zeaxanthin food sources, bioavailability and dietary variety in age related macular degeneration protection. Nutrients 2017, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Yonekura, L.; Nagao, A. Intestinal absorption of dietary carotenoids. Mol. Nutr. Food Res. 2007, 51, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.J.; Buitendijk, G.H.; Rochtchina, E.; Lee, K.E.; Klein, B.E.; van Duijn, C.M.; Flood, V.M.; Meuer, S.M.; Attia, J.; Myers, C.; et al. Genetic susceptibility, dietary antioxidants, and long-term incidence of age-related macular degeneration in two populations. Ophthalmology 2014, 121, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Meyers, K.J.; Mares, J.A.; Igo, R.P.Jr.; Truitt, B.; Liu, Z.; Millen, A.E.; Klein, M.; Johnson, E.J.; Engelman, C.D.; Karki, C.K.; et al. Genetic evidence for role of carotenoids in age-related macular degeneration in the Carotenoids in Age-related Eye Disease study (CAREDS). Investig. Ophthalmol. Vis. Sci. 2014, 55, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S.; Delori, F.C.; Richer, S.; van Kuijk, F.J.; Wenzel, A.J. The Value of Measurement of Macular Carotenoid Pigment Optical Densities and Distributions in Age-Related Macular Degeneration and Other Retinal Disorders. Vis. Res. 2010, 50, 716–728. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Liu, R.; Du, J.H.; Liu, T.; Wu, S.S.; Liu, X.H. Lutein, Zeaxanthin and Meso-zeaxanthin Supplementation Associated with Macular Pigment Optical Density. Nutrients. 2016, 8, 426. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Wang, X. Enhancement of bifidobacteria from human gut contents by oligofructose using continuous culture. FEMS Microbiol. Lett. 1994, 118, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Burns, A.J.; Rowland, I.R. Anti-carcinogenicity of probiotics and prebiotics. Curr. Issues Intest. Microbiol. 2000, 1, 13–24. [Google Scholar] [PubMed]
- Gill, H.S. Stimulation of the immune system by lactic cultures. Int. Dairy J. 1998, 8, 535–544. [Google Scholar] [CrossRef]
- Molan, A.L.; Liu, Z.; Plimmer, G. Evaluation of the Effect of Blackcurrant Products on Gut Microbiota and on Markers of Risk for Colon Cancer in Humans. Phytother. Res. 2014, 28, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Gill, C.I.; Rowland, I.R. Diet and cancer: Assessing the risk. Br. J. Nutr. 2002, 88, S73–S87. [Google Scholar] [CrossRef] [PubMed]
- Bakke, D.; Sun, J. Ancient nuclear receptor vdr with new functions: Microbiome and inflammation. Inflamm. Bowel Dis. 2018, 24, 1149–1154. [Google Scholar] [CrossRef] [PubMed]
- Kassi, E.; Adamopoulos, C.; Basdra, E.K.; Papavassiliou, A.G. Role of vitamin D in atherosclerosis. Circulation 2013, 128, 2517–2531. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Vitamin D metabolism, mechanism of action, and clinical applications. Chem. Biol. 2014, 21, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.A.; Grande, J.P.; Roche, P.C.; Campbell, R.J.; Kumar, R. Immuno-localization of the calcitriol receptor, calbindin-D28k and the plasma membrane calcium pump in the human eye. Curr. Eye Res. 1995, 14, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Morrison, M.A.; Silveira, A.C.; Huynh, N.; Jun, G.; Smith, S.E.; Zacharaki, F.; Sato, H.; Loomis, S.; Andreoli, M.T.; Adams, S.M.; et al. Systems biology-based analysis implicates a novel role for vitamin D metabolism in the pathogenesis of age-related macular degeneration. Hum. Genom. 2011, 5, 538–568. [Google Scholar] [CrossRef]
- Choi, D.; Appukuttan, B.; Binek, S.J.; Planck, S.R.; Stout, J.T.; Rosenbaum, J.T.; Smith, J.R. Prediction of cis-regulatory elements controlling genes differentially expressed by retinal and choroidal vascular endothelial cells. J. Ocul. Biol. Dis. Inform. 2008, 1, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Layana, A.G.; Minnella, A.M.; Garhöfer, G.; Aslam, T.; Holz, F.G.; Leys, A.; Silva, R.; Delcourt, C.; Souied, E.; Seddon, J.M. Vitamin D and Age-Related Macular Degeneration. Nutrients 2017, 9, E1120. [Google Scholar] [CrossRef] [PubMed]
- Riccio, P.; Rossano, R. Diet, Gut Microbiota, and Vitamins D + A in Multiple Sclerosis. Neurotherapeutics 2018, 15, 75–91. [Google Scholar] [CrossRef] [PubMed]
- Gubatan, J.; Moss, A.C. Vitamin D in inflammatory bowel disease: More than just a supplement. Curr. Opin. Gastroenterol. 2018, 34, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Villa, C.R.; Taibi, A.; Chen, J.; Ward, W.E.; Comelli, E.M. Colonic Bacteroides are positively associated with trabecular bone structure and programmed by maternal vitamin D in male but not female offspring in an obesogenic environment. Int. J. Obes. 2018, 42, 696–703. [Google Scholar] [CrossRef] [PubMed]
- Talsness, C.E.; Penders, J.; Jansen, E.H.J.M.; Damoiseaux, J.; Thijs, C.; Mommers, M. Influence of vitamin D on key bacterial taxa in infant microbiota in the KOALA Birth Cohort Study. PLoS ONE 2017, 12, e0188011. [Google Scholar] [CrossRef] [PubMed]
- Luthold, R.V.; Fernandes, G.R.; Franco-de-Moraes, A.C.; Folchetti, L.G.; Ferreira, S.R. Gut microbiota interactions with the immunomodulatory role of vitamin D in normal individuals. Metabolism 2017, 69, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Bashir, M.; Prietl, B.; Tauschmann, M.; Mautner, S.I.; Kump, P.K.; Treiber, G.; Wurm, P.; Gorkiewicz, G.; Högenauer, C.; Pieber, T.R. Effects of high doses of vitamin D3 on mucosa-associated gut microbiome vary between regions of the human gastrointestinal tract. Eur. J. Nutr. 2016, 55, 1479–1489. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.; Hendy, P.; Ding, J.N.; Shaw, S.; Hold, G.; Hart, A. The effect of vitamin D on intestinal inflammation and faecal microbiota in patients with ulcerative colitis. J. Crohn’s Colitis 2018, 12, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Schäffler, H.; Herlemann, D.P.; Klinitzke, P.; Berlin, P.; Kreikemeyer, B.; Jaster, R.; Lamprecht, G. Vitamin D administration leads to a shift of the intestinal bacterial composition in Crohn’s disease patients, but not in healthy controls. J. Dig. Dis. 2018, 19, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Kanhere, M.; He, J.; Chassaing, B.; Ziegler, T.R.; Alvarez, J.A.; Ivie, E.A.; Hao, L.; Hanfelt, J.; Gewirtz, A.T.; Tangpricha, V. Bolus Weekly Vitamin D3 Supplementation Impacts Gut and Airway Microbiota in Adults with Cystic Fibrosis: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial. J. Clin. Endocrinol. MeTable 2018, 103, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Tatsuno, I. Omega-3 polyunsaturated fatty acids for cardiovascular diseases: Present, past and future. Expert Rev. Clin. Pharmacol. 2017, 10, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Miles, E.A.; Calder, P.C. Influence of marine n-3 polyunsaturated fatty acids on immune function and a systematic review of their effects on clinical outcomes in rheumatoid arthritis. Br. J. Nutr. 2012, 107, S171–S184. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Fatty acids and immune function: Relevance to inflammatory bowel diseases. Int. Rev. Immunol. 2009, 28, 506–534. [Google Scholar] [CrossRef] [PubMed]
- Arnold, L.E.; Young, A.S.; Belury, M.A.; Cole, R.M.; Gracious, B.; Seidenfeld, A.M.; Wolfson, H.; Fristad, M.A. Omega-3 fatty acids plasma levels before and after supplementation: Correlation with mood and clinical outcomes in the omega-3 and therapy studies. J. Child Adolesc. Psychopharmacol. 2017, 27, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Merendino, N.; Costantini, L.; Manzi, L.; Molinari, R.; D’Eliseo, D.; Velotti, F. Dietary omega-3 polyunsaturated fatty acid DHA: A potential adjuvant in the treatment of cancer. Biomed. Res. Int. 2013. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, M.; Bonafè, S.; Valensin, F.; Olivieri, M.; de Luca, E.; Ottaviani, G.; de Benedictis, G. Inflammaging: An evolutionary perspective on immunosenescence. Ann. N.Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, M.; Capri, D.; Monti, S.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and anti-inflammaging: A systemic perspective on aging and longevity emerged from studies in humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Fu, Z.; Liegl, R.; Chen, J.; Hellström, A.; Smith, L.E. ω-3 and ω-6 long-chain PUFAs and their enzymatic metabolites in neovascular eye diseases. Am. J. Clin. Nutr. 2017, 106, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Lawrenson, J.G.; Evans, J.R. Omega 3 fatty acids for preventing or slowing the progression of age-related macular degeneration. Cochrane Database Syst. Rev. 2015, 9, CD010015. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill-Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 2009, 137, 1716–1724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, M.; Wang, S.; Han, R.; Cao, Y.; Hua, W.; Mao, Y.; Zhang, X.; Pang, X.; Wei, C.; et al. Interaction between gut microbiota, host genetics and diet relevant to development of metabolic syndrome in mice. ISME J. 2010, 4, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Devkota, S.; Wang, Y.; Musch, M.W.; Leone, V.; Fehlner-Peach, H.; Nadimpalli, A.; Antonopoulos, D.A.; Jabri, B.; Chang, E.B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10−/− mice. Nature 2012, 487, 104–108. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Noriega, B.S.; Sanchez-Gonzalez, M.A.; Salyakina, D.; Coffman, J. Understanding the impact of omega-3 rich diet on the gut microbiota. Case Rep. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Zierer, J.; Pallister, T.; Jackson, M.A.; Long, T.; Mohney, R.P.; Steves, C.J.; Spector, T.D.; Valdes, A.M. Omega-3 fatty acids correlate with gut microbiome diversity and production of N-carbamylglutamate in middle aged and elderly women. Sci. Rep. 2017, 7, 11079. [Google Scholar] [CrossRef] [PubMed]
- Mokkala, K.; Roytio, H.; Munukka, E.; Pietila, S.; Ekblad, U.; Ronnemaa, T.; Eerola, E.; Laiho, A.; Laitinen, K. Gut Microbiota Richness and Composition and Dietary Intake of Overweight Pregnant Women Are Related to Serum Zonulin Concentration, a Marker for Intestinal Permeability. J. Nutr. 2016, 146, 1694–1700. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.C.; Kaliannan, K.; Strain, C.R.; Ross, R.P.; Stanton, C.; Kang, J.X. Maternal omega-3 fatty acids regulate offspring obesity through persistent modulation of gut microbiota. Microbiome 2018, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef] [PubMed]
- Bidu, C.; Escoula, Q.; Bellenger, S.; Spor, A.; Galan, M.; Geissler, A.; Bouchot, A.; Dardevet, D.; Morio-Liondor, B.; Cani, P.D.; et al. The transplantation of ω3 pufa-altered gut microbiota of fat-1 mice to wild-type littermates prevents obesity and associated metabolic disorders. Diabetes 2018, 67, 1512–1523. [Google Scholar] [CrossRef] [PubMed]
| Year; Author [Ref.] | Study Design | Population | Micronutrients | Aims | Results |
|---|---|---|---|---|---|
| 1994; Seddon J.M. et al. [38] | Case-control | 356 subjects with advanced AMD and 520 control subjects | Vitamins A, C, E, Carotenoids | To assess association between dietary intake of carotenoids and vitamins A, C, and E and the risk of AMD. | Carotenoid intake reduces the risk of AMD (OR: 0.57; 95% CI, 0.35–0.92; p = 0.02); L and Z were most strongly associated with a reduced risk of AMD (p = 0.001). The intake of retinol was not appreciably related to AMD. Vitamin E or total vitamin C consumption or the intake of retinol were not associated with a reduced risk of AMD. |
| 2004; Richer S. et al. (the LAST study) [39] | RCT | 90 patients affected by atrophic AMD followed up in a period of 12 months: Group 1, L 10 mg; Group 2, L 10 mg + antioxidants/vitamins and minerals Group 3, placebo | L | To determine whether nutritional suppl. with L or L with antioxidants, vitamins, and minerals improves visual function in atrophic AMD. | Visual function and eye MPOD improved with L alone or L together with other nutrients. Patients who received the placebo had no significant changes in the measured findings. |
| 2006; Delcourt C. et al. (the POLA Study) [40] | Cohort study | 899 subjects ≥60 years, resident in Sète (Southern France) | L, Z | To assess association of plasma L/Z with the risk of AMD and cataract in the POLA Study. | The highest quintile of plasma Z was significantly associated with reduced risks of AMD (OR: 0.07; 95% CI, 0.01–0.58; p = 0.005), nuclear cataract (OR: 0.23; 95% CI, 0.08–0.68; p = 0.003) and any cataract (OR: 0.53; 95% CI, 0.31–0.89; p = 0.01). AMD was significantly associated with combined plasma L/Z (OR: 0.21; 95% CI, 0.05–0.79; p = 0.01) and tended to be associated with plasma L (OR: 0.31; 95% CI, 0.09–1.07; p = 0.04), whereas cataract showed no associations. Among other carotenoids, only beta-carotene showed a significant negative association with nuclear cataract, but not AMD. |
| 2006; Seddon et al. (US Twin Study) [41] | Case-control | 681 twins: 222 with AMD (intermediate or late stages) and 459 with no maculopathy | omega-3 PUFA | To evaluate modifiable risk and protective factors for AMD among elderly twins. | Dietary omega-3 PUFA intake inversely associated with AMD (OR: 0.55; 95% CI, 0.32–0.95). Cigarette smoking increases risk, while fish consumption and omega-3 fatty acid intake reduce risk of AMD. |
| 2006; Chua B. et al. [42] | Cohort study | 2335 subjects ≥49 years, participated in the Blue Mountains Eye Study (1992–1994) and re-examined after 5 years (1997–1999) | omega-3 PUFA | To assess association between dietary fat intake in the older cohort and incident AMD. | Participants with omega-3 PUFA intake had a lower risk of incident early AMD (OR: 0.41; 95% CI, 0.22–0.75). A 40% reduction of incident early AMD was associated with fish consumption at least once per week (OR: 0.58; 95% CI, 0.37–0.90), whereas fish consumption at least 3 times per week could reduce the incidence of late AMD (OR: 0.25; 95% CI, 0.06–1.00). |
| 2007; SanGiovanni et al. (AREDS Study Report 20) [43] | Case-control | 4519 AREDS Study participants: 4 AMD severity groups and a control group | omega-3 PUFA | To assess association of lipid intake with AMD in AREDS Study. | Dietary total omega-3 PUFA intake was inversely associated with neovascular AMD (OR, 0.61; 95% CI, 0.41–0.90). Higher intake of omega-3 PUFAs and fish was associated with decreased likelihood of having neovascular AMD. |
| 2007; SanGiovanni et al. (AREDS Study Report 22 [44] | Case-control | 4519 AREDS participants: 4 AMD severity groups and a control group | L/Z Vitamins A and C, alpha-tocopherol | To evaluate the relationship among dietary carotenoids, vitamins A and C and alpha-tocopherol with prevalent AMD. | Dietary L/Z intake was inversely associated with neovascular AMD (OR: 0.65; 95% CI, 0.45–0.93), geographic atrophy (OR: 0.45; 95% CI, 0.24–0.86), and large or extensive intermediate drusen (OR: 0.7; 95% CI, 0.56–0.96), comparing the highest vs lowest quintiles of intake. |
| 2008; SanGiovanni et al. (AREDS Study Report 23) [45] | Cohort study | 2132 subjects from the clinical trial AREDS | omega-3 PUFA | To examine the association of neovascular AMD and CGA. | Decreased risk of progression from bilateral drusen to CGA among individuals who reported the highest levels of EPA (OR: 0.44, 95% CI, 0.23–0.87) and EPA+DHA (OR: 0.45, 95% CI, 0.23–0.90) consumption. |
| 2008; Stringham, J.M. et al. [46] | Cohort study | 40 healthy subjects | L, Z | To measure MPOD after L and Z supplementation for 6 months and evaluate visual improvement. | After 6 months, daily L/Z supplementation significantly increased MPOD and improved visual performance in glare for most subjects. At the 2-month time point, average MPOD had increased from 0.41 at baseline to 0.46. MPOD continued to increase at 4-month (p = 0.032) and 6-month (p = 0.003) time points, with increases from baseline of 0.10 and 0.16, respectively. |
| 2008; Cho E. et al. [47] | Cohort study | 71494 women and 41564 men with no diagnosis of AMD or cancer | L/Z | To assess association between L/Z intake and AMD risk by smoking status, vitamin C and E intakes, and body fatness. | L and Z intake was not associated with the risk of self-reported early AMD. There was a non-significant association between lutein and zeaxanthin intake and neovascular AMD risk. |
| 2008; Tan J.S. et al. (the Blue Mountains Eye Study) [48] | Cohort study | 3654 participants | Carotenoids, Vitamins A, C, E, Iron, Zinc | To assess incidence of early, late, and any AMD. | L/Z: participants in the top tertile of intake had a reduced risk of incident neovascular AMD (RR: 0.35; 95% CI, 0.13–0.92), and individuals with above median intakes had a reduced risk of indistinct soft or reticular drusen (RR: 0.66; 95% CI, 0.48–0.92). Beta-carotene: the highest compared with the lowest tertile of total beta-carotene intake predicted incident neovascular AMD (RR, 2.68; 95% CI, 1.03–6.96). Zinc: the RR that compared the top decile intake with the remaining population was 0.56 (95% CI, 0.32–0.97) for any AMD and 0.54 (95% CI, 0.30–0.97) for early AMD. Vitamin E: higher intakes predicted late AMD (RR compared with the lowest tertile, 2.83; 95% CI, 1.28–6.23; and RR, 2.55; 95% CI, 1.14–5.70 for the middle and highest tertiles, respectively). |
| 2008; Newsome D.A. [49] | RCT | 40 subjects randomly assigned to ZMC (25 mg) or placebo | Zinc (ZMC) | To assess association between use of ZMC and macular function in individuals with dry AMD. | After 6 months, the ZMC group showed improved visual acuity (p < 0.0001). ZMC (25 mg) twice daily was well tolerated and improved with macular function AMD. |
| 2009; San Giovanni et al. (AREDS Study Report 30) [50] | Cohort study | 1837 AREDS participants | omega-3 PUFA | To assess the association of dietary omega-3 PUFAs with progression to advanced AMD in subjects with a moderate risk of developing AMD. | Participants who reported highest omega-3 PUFA intake were 30% less likely than their peers to develop central geographic atrophy and neovascular AMD. Respective multivariate ORs are 0.65 (95% CI, 0.45–0.92; p = 0.02) and 0.68 (95% CI, 0.49–0.94; p = 0.02). |
| 2011; Ho L. et al. (the Rotterdam Study) [51] | Case control | 2167 individuals from the population-based Rotterdam Study | Zinc, beta-carotene, L/Z, EPA/DHA | To investigate the role of dietary nutrients in reducing the genetic risk of AMD conferred by the genetic variants CFH Y402H and LOC387715 A69S. | High dietary intake of nutrients with antioxidant properties reduces the risk of early AMD in individuals at high genetic risk. The results supported the possibility of biological interactions among LOC387715 A69S and CFH Y402H and zinc, beta-carotene, lutein/zeaxanthin, and EPA/DHA (all p < 0.05). |
| 2012; Snellen E.L.M. et al. [52] | Case-control | 72 cases and 66 controls | L | To assess the association between low antioxidant intake and neovascular AMD. | The prevalence rate of AMD in patients with low antioxidant intake and low L intake was approximately twice as high as that in patients with high intake: OR: 1.7; 95% CI, 0.8–3.7 and OR: 2.4; 95% CI, 1.1–5.1, respectively. |
| 2012; Nolan J.M. et al. [53] | Cohort study | 828 healthy subjects | L/Z | To investigate MPOD with respect to risk factors for AMD. | A statistically significant age-related reduction in MPOD was present in current and past smokers (p < 0.01), with a family history of AMD (p < 0.01). The enhanced risk that these variables represent for AMD may be attributable, at least in part, to a parallel deficiency of macular carotenoids. |
| 2013; Souied E.H. et al. [54] | RCT | 263 patients with early lesions of AMD received 840 mg/day of DHA and 270 mg/day of EPA or placebo | EPA, DHA | To evaluate the efficacy of DHA-enriched oral supplementation in preventing exudative AMD (time to occurrence of CNV, incidence of CNV developing in patients, changes in visual acuity, occurrence and progression of drusen, and changes in EPA plus DHA level in red blood cell membrane in RBCM). | Time to occurrence and incidence of CNV in the study eye were not significantly different between the DHA group (19.5 ± 10.9 months and 28.4%, respectively) and the placebo group (18.7 ± 10.6 months and 25.6%, respectively). In the DHA group, EPA plus DHA levels significantly increased in RBCM (+ 70%; p < 0.001). In the DHA- allocated group, patients steadily achieving the highest tertile of EPA plus DHA levels in RBCM had a significantly lower risk (−68%; p = 0.047; HR: 0.32; 95% CI, 0.10–0.99) of CNV developing over 3 years. No marked changes from baseline in best-corrected visual acuity, drusen progression, or geographic atrophy in the study eye were observed throughout the study in either group. |
| 2014; Hammond, B.R. et al. [55] | RCT | 115 young, healthy subjects (58 assigned to placebo and 57 assigned to L/Z (10/2 mg/day) | L, Z | To assess correlation between L and Z supplementation with MPOD, glare disability, photostress recovery, and chromatic contrast. | MPOD significantly increased in L/Z group versus placebo (p < 0.001 for both L and Z). Serum L and Z significantly increased by the first follow-up visit (at 3 months) and remained elevated throughout the intervention period of 1 year. There was a significant correlation between MPOD levels over time and visual performance. |
| 2016; Aoki A. et al. [56] | Case -control | 161 neovascular AMD cases and 369 control subjects | omega-3 PUFA, -tocopherol, Zinc, Vitamins D and C, beta-carotene | To assess association between micronutrient intake and neovascular AMD. | Low intakes of omega-3 PUFA, alpha-tocopherol, zinc, vitamin D, vitamin C, and beta-carotene were associated with neovascular AMD (p < 0.0001 for n-3 fatty acid, p < 0.0001 for alpha-tocopherol, p < 0.0001 for zinc, p = 0.002 for vitamin D, p = 0.04 for vitamin C, and p = 0.0004 for beta-carotene). |
| 2017; Braakhuis A. et al. [57] | Case-control | 149 controls; 42 cases with oxidative stress-related AMD | beta-carotene, Vitamin C | To assess association between the intake of dietary antioxidants and incidence of AMD. | Protective associations with higher consumption of vitamin C (OR: 0.63; 95% CI, 0.23–1.03; p = 0.022) and beta-carotene (OR: 0.56; 95% CI, 0.15–0.98; p = 0.007). |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rinninella, E.; Mele, M.C.; Merendino, N.; Cintoni, M.; Anselmi, G.; Caporossi, A.; Gasbarrini, A.; Minnella, A.M. The Role of Diet, Micronutrients and the Gut Microbiota in Age-Related Macular Degeneration: New Perspectives from the Gut–Retina Axis. Nutrients 2018, 10, 1677. https://doi.org/10.3390/nu10111677
Rinninella E, Mele MC, Merendino N, Cintoni M, Anselmi G, Caporossi A, Gasbarrini A, Minnella AM. The Role of Diet, Micronutrients and the Gut Microbiota in Age-Related Macular Degeneration: New Perspectives from the Gut–Retina Axis. Nutrients. 2018; 10(11):1677. https://doi.org/10.3390/nu10111677
Chicago/Turabian StyleRinninella, Emanuele, Maria Cristina Mele, Nicolò Merendino, Marco Cintoni, Gaia Anselmi, Aldo Caporossi, Antonio Gasbarrini, and Angelo Maria Minnella. 2018. "The Role of Diet, Micronutrients and the Gut Microbiota in Age-Related Macular Degeneration: New Perspectives from the Gut–Retina Axis" Nutrients 10, no. 11: 1677. https://doi.org/10.3390/nu10111677
APA StyleRinninella, E., Mele, M. C., Merendino, N., Cintoni, M., Anselmi, G., Caporossi, A., Gasbarrini, A., & Minnella, A. M. (2018). The Role of Diet, Micronutrients and the Gut Microbiota in Age-Related Macular Degeneration: New Perspectives from the Gut–Retina Axis. Nutrients, 10(11), 1677. https://doi.org/10.3390/nu10111677









