Polyunsaturated Fatty Acids and Their Potential Therapeutic Role in Cardiovascular System Disorders—A Review
Abstract
:1. Introduction
2. Results
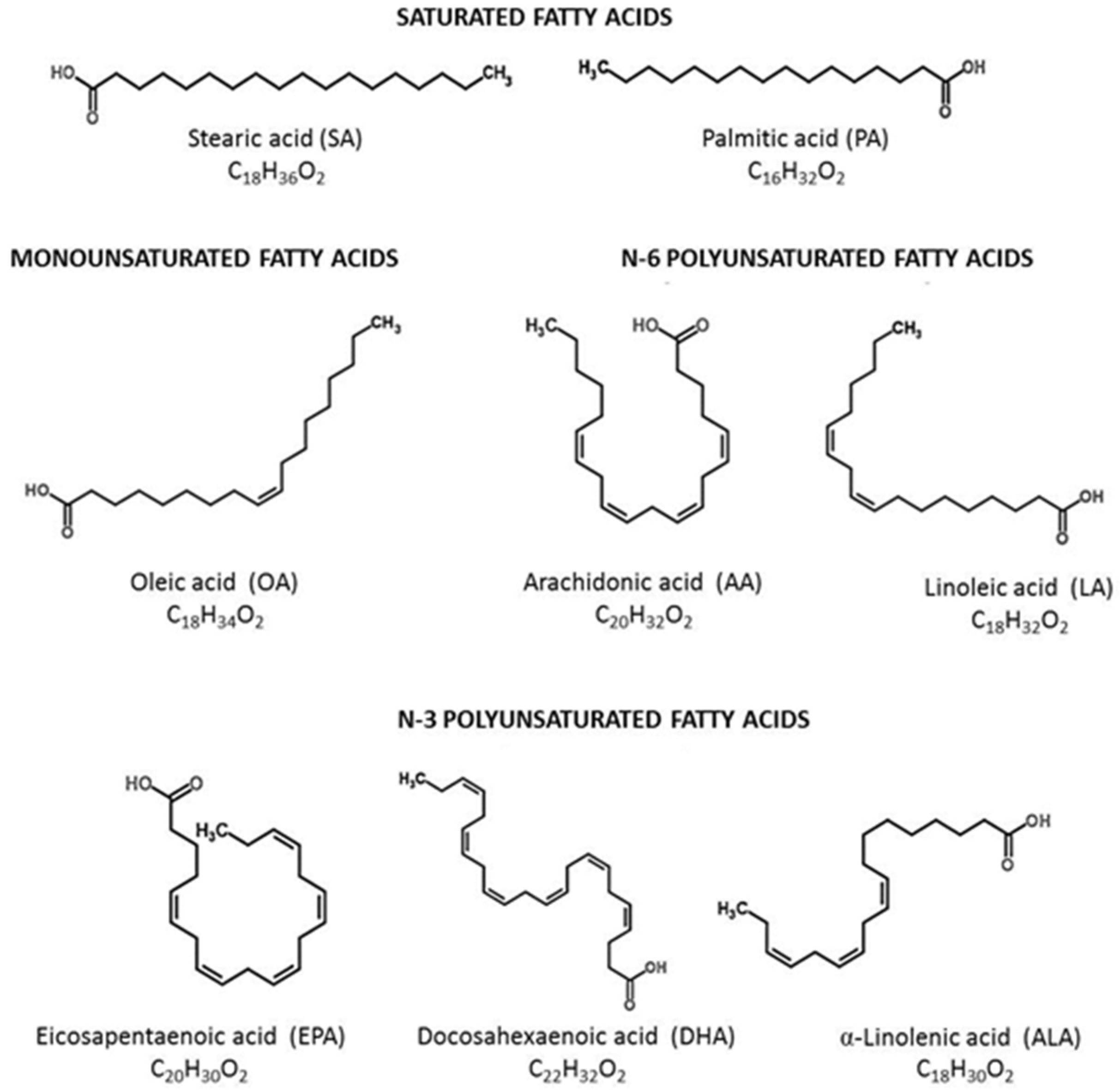
2.1. Classification of Unsaturated Fatty Acids
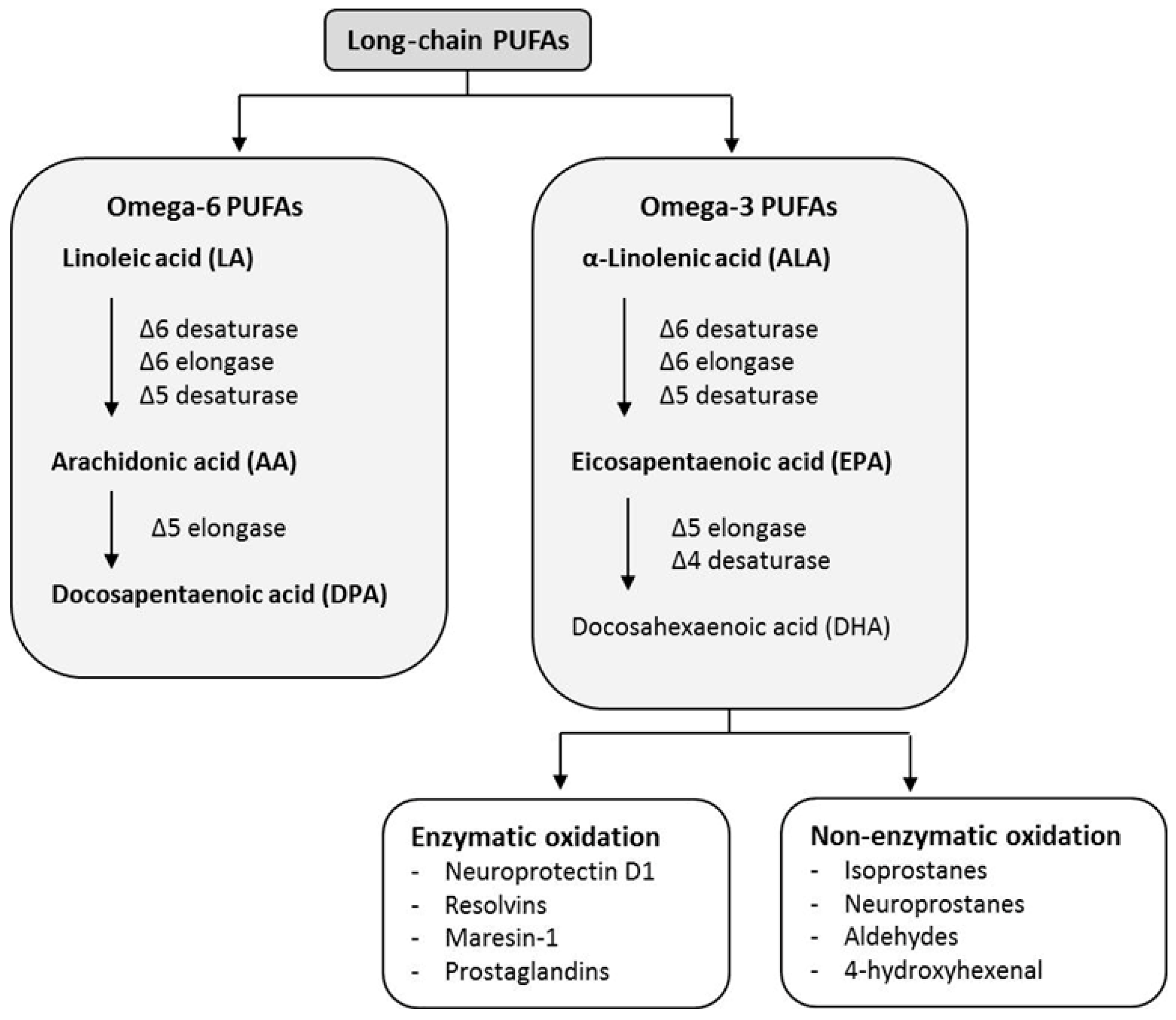
2.2. EFAs Conversion
2.3. CVD Risk Factors
2.4. Modern Diet as a Precursor for Inflammation Progression
2.5. Lipid Profile
2.6. Atherosclerosis
2.7. Cardioprotective Effects via Antithrombotic Activity
2.8. Antiarrhythmic Properties
2.9. Myocardial Infarction
2.10. Atrial Fibrillation
2.11. Stroke
3. Safety Concerns
4. Perspectives
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of interest
Abbreviations
| AA | arachidonic acid (20:4, n-6) |
| AF | atrial fibrillation |
| ALA | alpha-/α-linolenic acid (18:3, n-3) |
| AMI | acute myocardial infarction |
| CAD | coronary artery disease |
| CVD | cardiovascular disease |
| DHA | docosahexaenoic acid (22:6, n-3) |
| EFA | essential fatty acid |
| EPA | eicosapentaenoic acid (20:5, n-3) |
| GLA | gamma-/γ-linolenic acid (18:3, n-6) |
| HDL | high-density lipoprotein |
| IHD | ischemic heart disease |
| LA | linoleic acid (18:2, n-6) |
| LDL | low-density lipoprotein |
| MI | myocardial infarction |
| MUFA | monounsaturated fatty acid |
| PUFA | polyunsaturated fatty acid |
| TGs | triglycerides |
| VLDL | very low-density lipoprotein |
| WHF | World Health Federation |
| WHO | World Health Organization |
References
- Bodkowski, R.; Patkowska-Sokola, B.; Filip-Psurska, B.; Kempinska, K.; Wietrzyk, J.; Czyz, K.; Walisiewicz-Niedbalska, W.; Usydus, Z. Evaluation of the anti-proliferative activity of natural lipid preparations against tumor cell lines. J. Anim. Vet. Adv. 2014, 13, 257–266. [Google Scholar]
- Fabian, C.J.; Kimler, B.F.; Hursting, S.D. Omega-3 fatty acids for breast cancer prevention and survivorship. Breast Cancer Res. 2015, 17, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.; Ralston, J.; Taubert, K. Urbanization and Cardiovascular Disease: Raising Heart-Healthy Children in Today’s Cities; The World Heart Federation: Geneva, Switzerland, 2012. [Google Scholar]
- Lim, S.S.; Vos, T.; Flaxman, A.D. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Tvermosegaard, M.; Dahl-Petersen, I.K.; Nielsen, N.O.; Bjerregaard, P.; Jørgensen, M.E. Cardiovascular Disease Susceptibility and Resistance in Circumpolar Inuit Populations. Can. J. Cardiol. 2015, 31, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.E.; Moustgaard, H.; Bjerregaard, P.; Borch-Johnsen, K. Gender differences in the association between westernization and metabolic risk among Greenland Inuit. Eur. J. Epidemiol. 2006, 21, 741–748. [Google Scholar] [CrossRef] [PubMed]
- Fodor, J.G.; Helis, E.; Yazdekhasti, N.; Vohnout, B. “Eskimos and heart disease” story: Facts or wishful thinking? Can. J. Cardiol. 2014, 30, 864–868. [Google Scholar] [CrossRef] [PubMed]
- Marchioli, R.; Schweiger, C.; Tavazzi, L.; Valagussa, F. Efficacy of n-3 polyunsaturated fatty acids after myocardial infarction: Results of GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico. Lipids 2001, 36 (Suppl. 1), 119–126. [Google Scholar] [CrossRef]
- Marchioli, R.; Barzi, F.; Bomba, E.; Chieffo, C.; Di Gregorio, D.; Di Mascio, R.; Franzosi, M.G.; Geraci, E.; Levantesi, G.; Maggioni, A.P.; et al. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: Time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)-Prevenzione. Circulation 2002, 105, 1897–1903. [Google Scholar] [CrossRef] [PubMed]
- Dalal, J.J.; Kasliwal, R.R.; Dutta, A.L.; Sawhney, J.P.S.; Iyengar, S.S.; Dani, S.; Desai, N.; Sathyamurthy, I.; Rao, D.; Menon, A.; et al. Role of omega-3 ethyl ester concentrate in reducing sudden cardiac death following myocardial infarction and in management of hypertriglyceridemia: An Indian consensus statement. Indian Heart J. 2012, 64, 503–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marchioli, R.; Levantesi, G. N-3 PUFAs and heart failure. Int. J. Cardiol. 2013, 170 (Suppl. 1), 28–32. [Google Scholar] [CrossRef] [PubMed]
- Patkowska-Sokoła, B.; Usydus, Z.; Szlinder-Richert, J.; Bodkowski, R. Technology for recovering omega-3 fatty acids from family from fish oils and protecting them against oxidative changes. Przem. Chem. 2009, 88, 548–552. [Google Scholar]
- Bodkowski, R.; Szlinder-Richert, J.; Usydus, Z.; Patkowska-Sokoła, B. An attempt of optimization of fish oil crystallization at low temperature. Przem. Chem. 2011, 90, 703–706. [Google Scholar]
- Blasbalg, T.L.; Hibbeln, J.R.; Ramsden, C.R.; Majchrzak, S.F.; Rawlings, R.R. Changes in consumption of omega-3 and omega-6 fatty acids in the United States during the 20th century. Am. J. Clin. Nutr. 2011, 93, 950–962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.H.; O’Keefe, J.H.; Lavie, C.J.; Harris, W.S. Omega-3 fatty acids: Cardiovascular benefits, sources and sustainability. Nat. Rev. Cardiol. 2009, 6, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Walisiewicz-Niedbalska, W.; Patkowska-Sokoła, B.; Gwardiak, H.; Szulc, T.; Bodkowski, R.; Różycki, K. Potential raw materials in synthesis of bioactive fat derivatives. Przem. Chem. 2012, 91, 1058–1063. [Google Scholar]
- Bodkowski, R.; Czyż, K.; Kupczyński, R.; Patkowska-Sokoła, B.; Nowakowski, P.; Wiliczkiewicz, A. Lipid complex effect on fatty acid profile and chemical composition of cow milk and cheese. J. Dairy Sci. 2016, 99, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Sokoła-Wysoczańska, E.; Wysoczański, T.; Czyż, K.; Vogt, A.; Patkowska-Sokoła, B.; Sokoła, K.; Bodkowski, R.; Wyrostek, A.; Roman, K. Characteristics of polyunsaturated fatty acids ethyl esters with high alpha-linolenic acid content as a component of biologically active health-promoting supplements. Przem. Chem. 2014, 93, 1923–1927. [Google Scholar]
- Usydus, Z.; Bodkowski, R.; Szlinder-Richert, J.; Patkowska-Sokoła, B.; Dobrzański, Z. Use of aminopropyl column extraction for fish oil enrichment in omega-3 acids. Przem. Chem. 2012, 91, 1043–1048. [Google Scholar]
- Rahmawaty, S.; Charlton, K.; Lyons-Wall, P.; Meyer, B.J. Dietary intake and food sources of EPA, DPA and DHA in Australian children. Lipids 2013, 48, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.J.; Neil, J.; Mann, N.J.; Lewis, J.L.; Milligan, G.C.; Sinclair, A.J.; Howe, P.R.C. Dietary intakes and food sources of omega-6 and omega-3 polyunsaturated fatty acids. Lipids 2003, 38, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Arnoldussen, I.A.; Kiliaan, A.J. Impact of DHA on metabolic diseases from womb to tomb. Mar. Drugs 2014, 12, 6190–6212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finch, J.; Munhutu, M.N.; Whitaker-Worth, D.L. Atopic dermatitis and nutrition. Clin. Dermatol. 2010, 28, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Johnston, D.T.; Deuster, P.A.; Harris, W.S.; MacRae, H.; Dretsch, M.N. Red blood cell omega-3 fatty acid levels and neurocognitive performance in deployed U.S. Servicemembers. Nutr. Neurosci. 2013, 16, 30–38. [Google Scholar] [CrossRef] [PubMed]
- García-de-Lorenzo, A.; Denia, R.; Atlan, P.; Martinez-Ratero, S.; Le Brun, A.; Evard, D.; Bereziat, G. Parenteral nutrition providing a restricted amount of linoleic acid in severely burned patients: A randomised double-blind study of an olive oil-based lipid emulsion v. medium/long-chain triacylglycerols. Br. J. Nutr. 2005, 94, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, I.; Hussein, N.; Baumann, M.H.; Di Martino, C.; Salem, N. An n-3 fatty acid deficiency impairs rat spatial learning in the Barnes maze. Behav. Neurosci. 2009, 123, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Pallebage-Gamarallage, M.M.; Lam, V.; Takechi, R.; Galloway, S.; Mamo, J.C.L. A diet enriched in docosahexanoic acid exacerbates brain parenchymal extravasation of apo B lipoproteins induced by chronic ingestion of saturated fats. Int. J. Vasc. Med. 2012, 647689, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Haast, R.A.; Kiliaan, A.J. Impact of fatty acids on brain circulation, structure and function. Prostaglandins Leukot. Essent. Fatty Acids 2015, 92, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Wysoczański, T.; Sokoła-Wysoczańska, E.; Pękala, J.; Lochyński, St.; Czyż, K.; Bodkowski, R.; Herbinger, G.; Patkowska-Sokoła, B.; Librowski, T. Omega-3 fatty acids and their role in central nervous system—A Review. Curr. Med. Chem. 2016, 23, 816–831. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.H. Omega-3 fatty acids: New insights into the pharmacology and biology of docosahexaenoic acid, docosapentaenoic acid, and eicosapentaenoic acid. Curr. Opin. Lipidol. 2013, 24, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.T.; Nara, T.Y. Structure, function, and dietary regulation of delta6, delta5, and delta9 desaturases. Annu. Rev. Nutr. 2004, 24, 345–376. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Green, P.; Mann, J.J.; Rapoport, S.I.; Sublette, M.E. Pathways of polyunsaturated fatty acid utilization: Implications for brain function in neuropsychiatric health and disease. Brain Res. 2015, 1597, 220–246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demar, J.C.; Ma, K.; Chang, L.; Bell, J.M.; Rapoport, S.I. Alpha-Linolenic acid does not contribute appreciably to docosahexaenoic acid within brain phospholipids of adult rats fed a diet enriched in docosahexaenoic acid. J. Neurochem. 2005, 94, 1063–1076. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serhan, C.N.; Yang, R.; Martinod, K.; Kasuga, K.; Pillai, P.S.; Porter, T.F.; Oh, S.F.; Spite, M. Maresins: Novel macrophage mediators with potent antiinflammatory and proresolving actions. J. Exp. Med. 2009, 206, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, M.; Mirakaj, V.; Serhan, C.N. Functional metabolomics reveals novel active products in the DHA metabolome. Front Immunol. 2012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burdge, G.C.; Jones, A.E.; Wootton, S.A. Eicosapentaenoic and docosapentaenoic acids are the principal products of alpha-linolenic acid metabolism in young men. Br. J. Nutr. 2002, 88, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Abedi, E.; Sahari, M.A. Long-chain polyunsaturated fatty acid sources and evaluation of their nutritional and functional properties. Food Sci. Nutr. 2014, 2, 443–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talahalli, R.R.; Vallikannan, B.; Sambaiah, K.; Lokesh, B.R. Lower efficacy in the utilization of dietary ALA as compared to preformed EPA + DHA on long chain n-3 PUFA levels in rats. Lipids 2010, 45, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Pischon, T.; Hankinson, S.E.; Rifai, N.; Joshipura, K.; Willett, W.C.; Rimm, E.B. Dietary intake of trans fatty acids and systemic inflammation in women. Am. J. Clin. Nutr. 2004, 79, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, S.; Rise, P.; Ceruti, S.; Galli, C. Effects of cigarette smoke on cell viability, linoleic acid metabolism and cholesterol synthesis, in THP-1 cells. Lipids 2007, 42, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Essential fatty acids: Biochemistry, physiology and pathology. Biotechnol. J. 2006, 1, 420–439. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Cho, H.P.; Nakamura, M.T.; Clarke, S.D. Regulation of human delta-6 desaturase gene transcription: Identification of a functional direct repeat-1 element. J. Lipid Res. 2003, 44, 686–695. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Kushiro, M.; Shinohara, K.; Ide, T. Activity and mRNA levels of enzymes involved in hepatic fatty acid synthesis and oxidation in mice fed conjugated linoleic acid. Biochim. Biophys. Acta 2003, 1631, 265–273. [Google Scholar] [CrossRef]
- Kromhout, D.; Yasuda, S.; Geleijnse, J.M.; Shimokawa, H. Fish oil and omega-3 fatty acids in cardiovascular disease: Do they really work? Eur. Heart J. 2012, 33, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Hadzhieva, B.; Dimitrov, M.; Obreshkova, D.; Petkova, V.; Atanasov, P.; Kasnakova, P. Omega-3 polyunsaturated fatty acids metabolism and prevention of some socially significant diseases world. J. Pharm. Pharm. Sci. 2016, 5, 304–316. [Google Scholar]
- Mendis, S.; Puska, P.; Norrving, B. Global Atlas on Cardiovascular Disease Prevention and Control; WHO: Geneva, Switzerland, 2011; p. 164. [Google Scholar]
- Teo, K.K.; Ounpuu, S.; Hawken, S.; Pandey, M.R.; Valentin, V.; Hunt, D.; Diaz, R.; Rashed, W.; Freeman, R.; Jiang, L.; et al. Tobacco use and risk of myocardial infarction in 52 countries in the INTERHEART study: A case-control study. Lancet 2006, 368, 647–658. [Google Scholar] [CrossRef]
- Chiao, Y.A.; Rabinovitch, P.S. The aging heart. Cold Spring Harb. Perspect. Med. 2015, 5, a025148. [Google Scholar] [CrossRef] [PubMed]
- Rota, M.; Goichberg, P.; Anversa, P.; Leri, A. Aging effects on cardiac progenitor cell physiology. Compr. Physiol. 2015, 5, 1705–1750. [Google Scholar]
- Simopoulos, A.P. Evolutionary aspects of diet: The omega-6/omega-3 ratio and the brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, M.; Yao, Z. Recent progress in understanding protein and lipid factors affecting hepatic VLDL assembly and secretion. Nutr. Metab. 2010, 7, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandasescu, S.; Mocanu, V.; Dascalita, A.M.; Haliga, R.; Nestian, I.; Stitt, P.A.; Luca, V. Flaxseed supplementation in hyperlipidemic patients. Rev. Med. Chir. Soc. Med. Nat. Iasi. 2005, 109, 502–506. [Google Scholar] [PubMed]
- Egert, S.; Somoza, V.; Kannenberg, F.; Fobker, M.; Krome, K.; Erbersdobler, H.F.; Wahrburg, U. Influence of three rapeseed oil-rich diets, fortified with alpha-linolenic acid, eicosapentaenoic acid or docosahexaenoic acid on the composition and oxidizability of low-density lipoproteins: Results of a controlled study in healthy volunteers. Eur. J. Clin. Nutr. 2007, 61, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Mustad, V.A.; Demichele, S.; Huang, Y.S.; Mika, A.; Lubbers, N.; Berthiaume, N.; Polakowski, J.; Zinker, B. Differential effects of n-3 polyunsaturated fatty acids on metabolic control and vascular reactivity in the type 2 diabetic ob/ob mouse. Metabolism 2006, 55, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Stark, A.H.; Reifen, R.; Crawford, M.A. Past and present insights on alpha linolenic acid and the omega-3 fatty acid family. Crit. Rev. Food Sci. Nutr. 2016, 56, 2261–2267. [Google Scholar] [CrossRef] [PubMed]
- Goyens, P.L.; Spilker, M.E.; Zock, P.L.; Katan, M.B.; Mensink, R.P. Conversion of alpha-linolenic acid in humans is influenced by the absolute amounts of alpha-linolenic acid and linoleic acid in the diet and not by their ratio. Am. J. Clin. Nutr. 2006, 84, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Goyens, P.L.; Mensink, R.P. Effects of alpha-linolenic acid versus those of EPA/DHA on cardiovascular risk markers in healthy elderly subjects. Eur. J. Clin. Nutr. 2006, 60, 978–984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weber, P.; Raederstorff, D. Triglyceride-lowering effect of omega-3 LC-polyunsaturated fatty acids—A review. Nutr. Metab. Cardiovasc. Dis. 2000, 10, 28–37. [Google Scholar] [PubMed]
- Alexander, D.D.; Miller, P.E.; Van Elswyk, M.E.; Kuratko, C.N.; Bylsma, L.C. A meta-analysis of randomized controlled trials and prospective cohort studies of eicosapentaenoic and docosahexaenoic long-chain omega-3 fatty acids and coronary heart disease risk. Mayo Clin. Proc. 2017, 92, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Nambi, V.; Ballantyne, C.M. Combination therapy with statins and omega-3 fatty acids. Am. J. Cardiol. 2006, 98, 34i–38i. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Harris, W.S. Omega-3 fatty acid supplementation accelerates chylomicron triglyceride clearance. J. Lipid Res. 2003, 44, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.H.; Stein, E.A.; Bays, H.E.; Maki, K.C.; Doyle, R.T.; Shalwitz, R.A.; Ballantyne, C.M.; Ginsberg, H.N. Efficacy and tolerability of adding prescription Omega-3 fatty acids 4 g/d to Simvastatin 40 mg/d in hypertriglyceridemic patients: An 8-week, randomized, double-blind, placebo-controlled study. Clin. Ther. 2007, 29, 1354–1367. [Google Scholar] [CrossRef] [PubMed]
- Micallef, M.A.; Garga, M.L. Beyond blood lipids: Phytosterols, statins and omega-3 polyunsaturated fatty acid therapy for hyperlipidemia. J. Nutr. Biochem. 2009, 20, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, L.J.; Balestrieri, M.L.; Napoli, C. Nutrition, physical activity, and cardiovascular disease: An update. Cardiovasc. Res. 2007, 73, 326–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badimon, L.; Vilahur, G.; Padro, T. Nutraceuticals and atherosclerosis: Human trials. Cardiovasc. Ther. 2010, 28, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Torres, N.; Guevara-Cruz, M.; Velazquez-Villegas, L.A.; Tovar, A.R. Nutrition and atherosclerosis. Arch. Med. Res. 2015, 46, 408–426. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Yan, P.; Chen, L.; Luo, C.; Gao, H.; Deng, Q.; Zheng, M.; Shi, Y.; Liu, L. Flaxseed oil containing alpha-linolenic acid ester of plant sterol improved atherosclerosis in ApoE deficient mice. Oxid. Med. Cell. Longev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, M.S.; Kessuane, M.C.; Lobo Ladd, A.A.; Lobo Ladd, F.V.; Cogliati, B.; Castro, I.A. Effect of long-term ingestion of weakly oxidised flaxseed oil on biomarkers of oxidative stress in LDL-receptor knockout mice. Br. J. Nutr. 2016, 116, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Flaxseed and cardiovascular health. J. Cardiovasc. Pharmacol. 2009, 54, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Din, J.N.; Newby, D.E.; Flapan, A.D. Omega 3 fatty acids and cardiovascular disease—Fishing for a natural treatment. BMJ 2004, 328, 30–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mostowik, M.; Gajos, G.; Zalewski, J.; Nessler, J.; Undas, A. Omega-3 polyunsaturated fatty acids increase plasma adiponectin to leptin ratio in stable coronary artery disease. Cardiovasc. Drugs Ther. 2013, 27, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A. Dietary n-3 PUFA and CVD: A review of the evidence. Proc. Nutr. Soc. 2014, 73, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Albert, B.B.; Cameron-Smith, D.; Hofman, P.L.; Cutfield, W.S. Oxidation of marine omega-3 supplements and human health. Biomed. Res. Int. 2013, 2013, 464921. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Zhao, J.; Wanga, Y.; Liu, W.; Wang, Z.; Shanga, Y.; Zhang, W.; Zhang, Y.; Zhong, M. The limited effect of omega-3 polyunsaturated fatty acids on cardiovascular risk in patients with impaired glucose metabolism: A meta-analysis. Clin. Biochem. 2014, 47, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Gajos, G.; Zalewski, J.; Rostoff, P.; Nessler, J.; Piwowarska, W.; Undas, A. Reduced thrombin formation and altered fibrin clot properties induced by polyunsaturated omega-3 fatty acids on top of dual antiplatelet therapy in patients undergoing percutaneous coronary intervention (OMEGA-PCI clot). Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1696–1702. [Google Scholar] [CrossRef] [PubMed]
- Gajos, G.; Rostoff, P.; Undas, A.; Piwowarska, W. Effects of polyunsaturated omega-3 fatty acids on responsiveness to dual antiplatelet therapy in patients undergoing percutaneous coronary intervention: The OMEGA-PCI (OMEGA-3 fatty acids after pci to modify responsiveness to dual antiplatelet therapy) study. J. Am. Coll. Cardiol. 2010, 55, 1671–1678. [Google Scholar] [PubMed]
- Gajos, G.; Zalewski, J.; Nessler, J.; Zmudka, K.; Undas, A.; Piwowarska, W. Polyunsaturated omega-3 fatty acids improve responsiveness to clopidogrel after percutaneous coronary intervention in patients with cytochrome P450 2C19 loss-of-function polymorphism. Kardiol. Pol. 2012, 70, 439–445. [Google Scholar] [PubMed]
- Sorich, M.J.; Vitry, A.; Ward, M.B.; Horowitz, J.D.; McKinnon, R.A. Prasugrel vs. clopidogrel for cytochrome P450 2C19-genotyped subgroups: Integration of the TRITON-TIMI 38 trial data. J. Thromb. Haemost. 2010, 8, 1678–1684. [Google Scholar] [CrossRef] [PubMed]
- Din, J.N.; Harding, S.A.; Valerio, C.J.; Sarma, J.; Lyall, K.; Riemersma, R.A.; Newby, D.E.; Flapan, A.D. Dietary intervention with oil rich fish reduces platelet-monocyte aggregation in man. Atherosclerosis 2008, 197, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Gamez, R.; Maz, R.; Arruzazabala, M.L.; Mendoza, S.; Castano, G. Effects of concurrent therapy with policosanol and omega-3 fatty acids on lipid profile and platelet aggregation in rabbits. Drugs RD 2005, 6, 11–19. [Google Scholar] [CrossRef]
- Castano, G.; Fernandez, L.; Mas, R.; Illnait, J.; Games, R.; Mendoza, S.; Mesa, M.; Fernandez, J. Effects of addition of policosanol to omega-3 fatty acid therapy on the lipid profile of patients with type II hypercholesterolaemia. Drugs RD 2005, 6, 207–219. [Google Scholar] [CrossRef]
- Castano, G.; Arruzazabala, M.L.; Fernandez, L.; Mas, R.; Carbajal, D.; Molina, V.; Illnait, J.; Mendoza, S.; Games, R.; Mesa, M.; et al. Effects of combination treatment with policosanol and omega-3 fatty acids on platelet aggregation: A randomized, double-blind clinical study. Curr. Ther. Res. Clin. Exp. 2006, 67, 174–192. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Wu, J.H. Omega-3 fatty acids and cardiovascular disease: Effects on risk factors, molecular pathways, and clinical events. J. Am. Coll. Cardiol. 2011, 58, 2047–2067. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C. Omega-3 fatty acids: Antiarrhythmic, proarrhythmic or both? Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 94–99. [Google Scholar] [CrossRef] [PubMed]
- de Leiris, J.; de Lorgeril, M.; Boucher, F. Fish oil and heart health. J. Cardiovasc. Pharmacol. 2009, 54, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Saremi, A.; Arora, R. The utility of omega-3 fatty acids in cardiovascular disease. Am. J. Ther. 2009, 16, 421–436. [Google Scholar] [CrossRef] [PubMed]
- Bodkowski, R.; Patkowska-Sokoła, B.; Walisiewicz-Niedbalska, W.; Usydus, Z. The composition of bioactive lipid complex reducing the level of blood atherogenic lipid indices. Przem. Chem. 2010, 89, 304–310. [Google Scholar]
- Bianconi, L.; Calò, L.; Mennuni, M.; Santini, L.; Morosetti, P.; Azzolini, P.; Barbato, G.; Biscione, F.; Romano, P.; Santini, M. N-3 Polyunsaturated fatty acids for the prevention of arrhythmia recurrence after electrical cardioversion of chronic persistent atrial fibrillation: A randomized, double-blind, multicentre study. EP Europace 2011, 13, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Endo, J.; Arita, M. Cardioprotective mechanism of omega-3 polyunsaturated fatty acids. J. Cardiol. 2016, 67, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Reiffel, J.A.; McDonald, A. Antiarrhythmic effects of omega-3 fatty acids. Am. J. Cardiol. 2006, 98, 50i–60i. [Google Scholar] [CrossRef] [PubMed]
- Billman, G.E. The effects of omega-3 polyunsaturated fatty acids on cardiac rhythm: A critical reassessment. Pharmacol. Ther. 2013, 140, 53–80. [Google Scholar] [CrossRef] [PubMed]
- Bhupathiraju, S.N.; Tucker, K.L. Coronary heart disease prevention: Nutrients, foods, and dietary patterns. Clin. Chim. Acta 2011, 412, 1493–1514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grosso, G.; Marventano, S.; Yang, J.; Micek, A.; Pajak, A.; Scalfi, L.; Galvano, F.; Kales, S.N. A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: Are individual components equal? Crit. Rev. Food Sci. Nutr. 2017, 57, 3218–3232. [Google Scholar] [CrossRef] [PubMed]
- Dhein, S.; Michaelis, B.; Mohr, F.W. Antiarrhythmic and electrophysiological effects of long-chain omega-3 polyunsaturated fatty acids. Naunyn Schmiedebergs Arch. Pharmacol. 2005, 371, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Guizy, M.; David, M.; Arias, C.; Zhang, L.; Cofán, M.; Ruiz-Gutiérrez, V.; Ros, E.; Pilar Lillo, M.; Martens, J.R.; Valenzuela, C. Modulation of the atrial specific Kv1.5 channel by the n-3 polyunsaturated fatty acid, α-linolenic acid. J. Mol. Cell. Cardiol. 2008, 44, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Assmann, G.; Gotto, A.M., Jr. HDL cholesterol and protective factors in atherosclerosis. Circulation 2004, 109, III8–III14. [Google Scholar] [CrossRef] [PubMed]
- Rye, K.A.; Barter, P.J. Cardioprotective functions of HDLs. J. Lipid Res. 2014, 55, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Annema, W.; Tietge, U.J.F. Regulation of reverse cholesterol transport—A comprehensive appraisal of available animal studies. Nutr. Metab. 2012, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Demonty, I.; Chan, Y.M.; Pelled, D.; Jones, P.J. Fish-oil esters of plant sterols improve the lipid profile of dyslipidemic subjects more than do fish-oil or sunflower oil esters of plant sterols. Am. J. Clin. Nutr. 2006, 84, 1534–1542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palade, F.; Alexa, I.D.; Azoicai, D.; Panaghiu, L.; Ungureanu, G. Oxidative stress in atherosclerosis. Rev. Med. Chir. Soc. Med. Nat. Iasi 2003, 107, 502–511. [Google Scholar] [PubMed]
- Lichtenstein, A.H. Dietary fat and cardiovascular disease risk: Quantity or quality? J. Womens Health 2003, 12, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Berneis, K. Who needs to care about small, dense low-density lipoproteins? Int. J. Clin. Pract. 2007, 61, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, I.A.; Katan, M.B.; Zock, P.L. Dietary alpha-linolenic acid is associated with reduced risk of fatal coronary heart disease, but increased prostate cancer risk: A meta-analysis. J. Nutr. 2004, 134, 919–922. [Google Scholar] [CrossRef] [PubMed]
- Weitz, D.; Weintraub, H.; Fisher, E.; Schwartzbard, A.Z. Fish oil for the treatment of cardiovascular disease. Cardiol. Rev. 2010, 18, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Koh, W.P.; Yuan, J.M.; Choi, H.; Su, J.; Ong, C.N.; van Dam, R.M. Plasma alpha-linolenic and long-chain omega-3 fatty acids are associated with a lower risk of acute myocardial infarction in Singapore Chinese adults. J. Nutr. 2016, 146, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Derbali, A.; Mnafgui, K.; Affes, M.; Derbali, F.; Hajji, R.; Gharsallah, N.; Allouche, N.; El Feki, A. Cardioprotective effect of linseed oil against isoproterenol-induced myocardial infarction in Wistar rats: A biochemical and electrocardiographic study. J. Physiol. Biochem. 2015, 71, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Zoni-Berisso, M.; Lercari, F.; Carazza, T.; Domenicucci, S. Epidemiology of atrial fibrillation: European perspective. Clin. Epidemiol. 2014, 6, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Fretts, A.M.; Mozaffarian, D.; Siscovick, D.S.; Heckbert, S.R.; McKnight, B.; King, I.B.; Rimm, E.B.; Psaty, B.M.; Sacks, F.M.; Song, X.; et al. Associations of plasma phospholipid and dietary alpha linolenic acid with incident atrial fibrillation in older adults: The Cardiovascular Health Study. J. Am. Heart Assoc. 2013, 2, e003814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kastner, D.W.; Van Wagoner, D.R. Diet and atrial fibrillation: Does α-linolenic acid, a plant derived essential fatty acid, have an impact? J. Am. Heart Assoc. 2013, 2, e000030. [Google Scholar] [CrossRef] [PubMed]
- Wilk, J.B.; Tsai, M.Y.; Hanson, N.Q.; Gaziano, J.M.; Djousse, L. Plasma and dietary omega-3 fatty acids, fish intake, and heart failure risk in the Physicians’ Health Study. Am. J. Clin. Nutr. 2012, 96, 882–888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calo, L.; Bianconi, L.; Colivicchi, F.; Lamberti, F.; Loricchio, M.L.; de Ruvo, E.; Meo, A.; Pandozi, C.; Staibano, M.; Santini, M. N-3 Fatty acids for the prevention of atrial fibrillation after coronary artery bypass surgery: A randomized, controlled trial. J. Am. Coll. Cardiol. 2005, 45, 1723–1728. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, A.A. Atrial fibrillation post cardiac surgery trends toward management. Heart Views 2010, 11, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Folino, A.; Sprio, A.E.; Di Scipio, F.; Berta, G.N.; Rastaldo, R. Alpha-linolenic acid protects against cardiac injury and remodelling induced by beta-adrenergic overstimulation. Food Funct. 2015, 6, 2231–2239. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shi, Y.; Zhang, L.; Zhang, F.; Hu, X.; Zhang, W.; Leak, R.K.; Gao, Y.; Chen, L.; Chen, J. Omega-3 polyunsaturated fatty acids enhance cerebral angiogenesis and provide long-term protection after stroke. Neurobiol. Dis. 2014, 68, 91–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demaerschalk, B.M.; Hwang, H.M.; Leung, G. US cost burden of ischemic stroke: A systematic literature review. Am. J. Manag. Care 2010, 16, 525–533. [Google Scholar] [PubMed]
- Wiltrout, C.; Lang, B.; Yan, Y.; Dempsey, R.J.; Vemuganti, R. Repairing brain after stroke: A review on post-ischemic neurogenesis. Neurochem. Int. 2007, 50, 1028–1041. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.L.; Chopp, M.; Roberts, C.; Jia, L.; Wei, M.; Lu, M.; Wang, X.; Pourabdollah, S.; Zhang, Z.G. Ascl1 lineage cells contribute to ischemia-induced neurogenesis and oligodendrogenesis. J. Cereb. Blood Flow MeTable 2011, 31, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Hermann, D.M.; Chopp, M. Promoting brain remodelling and plasticity for stroke recovery: Therapeutic promise and potential pitfalls of clinical translation. Lancet Neurol. 2012, 11, 369–380. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Y.; Akamatsu, Y.; Lee, C.C.; Stetler, R.A.; Lawton, M.T.; Yang, G.Y. Vascular remodeling after ischemic stroke: Mechanisms and therapeutic potentials. Prog. Neurobiol. 2014, 115, 138–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLaughlin, B.; Gidday, J.M. Poised for success: Implementation of sound conditioning strategies to promote endogenous protective responses to stroke in patients. Transl. Stroke Res. 2013, 4, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Belayev, L.; Khoutorova, L.; Atkins, K.D.; Bazan, N.G. Robust docosahexaenoic acid-mediated neuroprotection in a rat model of transient, focal cerebral ischemia. Stroke 2009, 40, 3121–3126. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zhang, F.; Leak, R.K.; Zhang, W.; Iwai, M.; Stetler, R.A.; Dai, Y.; Zhao, A.; Gao, Y.; Chen, J. Transgenic overproduction of omega-3 polyunsaturated fatty acids provides neuroprotection and enhances endogenous neurogenesis after stroke. Curr. Mol. Med. 2013, 13, 1465–1473. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Hu, X.; Yang, W.; Gao, Y.; Chen, J. Omega-3 polyunsaturated fatty acid supplementation confers long-term neuroprotection against neonatal hypoxic-ischemic brain injury through anti-inflammatory actions. Stroke 2010, 41, 2341–2347. [Google Scholar] [CrossRef] [PubMed]
- Musiek, E.S.; Brooks, J.D.; Joo, M.; Brunoldi, E.; Porta, A.; Zanoni, G.; Vidari, G.; Blackwell, T.S.; Montine, T.J.; Milne, G.L.; et al. Electrophilic cyclopentenone neuroprostanes are anti-inflammatory mediators formed from the peroxidation of the omega-3 polyunsaturated fatty acid docosahexaenoic acid. J. Biol. Chem. 2008, 283, 19927–19935. [Google Scholar] [CrossRef] [PubMed]
- Bazan, N.G. Neuroprotectin D1 (NPD1): A DHA-derived mediator that protects brain and retina against cell injury-induced oxidative stress. Brain Pathol. 2005, 15, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, S.; Mao, L.; Leak, R.K.; Shi, Y.; Zhang, W.; Hu, X.; Sun, B.; Cao, G.; Gao, Y.; et al. Omega-3 fatty acids protect the brain against ischemic injury by activating Nrf2 and upregulating heme oxygenase 1. J. Neurosci. 2014, 34, 1903–1915. [Google Scholar] [CrossRef] [PubMed]
- Palmer, T.D.; Willhoite, A.R.; Gage, F.H. Vascular niche for adult hippocampal neurogenesis. J. Comp. Neurol. 2000, 425, 479–494. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, H.; Zhang, H.; Leak, R.K.; Shi, Y.; Hu, X.; Gao, Y.; Chen, J. Dietary supplementation with omega-3 polyunsaturated fatty acids robustly promotes neurovascular restorative dynamics and improves neurological functions after stroke. Exp. Neurol. 2015, 272, 170–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mozaffarian, D.; Lemaitre, R.N.; King, I.B.; Song, X.; Huang, H.; Sacks, F.M.; Rimm, E.B.; Wang, M.; Siscovick, D.S. Plasma phospholipid long-chain omega-3 fatty acids and total and causespecific mortality in older adults: A cohort study. Ann. Intern. Med. 2013, 158, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Orsini, N. Fish consumption and the risk of stroke: A dose-response meta-analysis. Stroke 2011, 42, 3621–3623. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Song, Y.; Daviglus, M.L.; Liu, K.; Van Horn, L.; Dyer, A.R.; Goldbourt, U.; Greenland, P. Fish consumption and incidence of stroke A meta-analysis of cohort studies. Stroke 2004, 35, 1538–1542. [Google Scholar] [CrossRef] [PubMed]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Siscovick, D.S.; Barringer, T.A.; Amanda, M.; Fretts, A.M.; Wu, J.H.Y.; Lichtenstein, A.H.; Costello, R.B.; Kris-Etherton, P.M.; Jacobson, T.A.; Engler, M.B.; et al. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease a science advisory from the American Heart Association. Circulation 2017, 135, e867–e884. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.M.; Myung, S.K.; Lee, Y.J.; Seo, H.G. Efficacy of omega-3 fatty acid supplements (eicosapentaenoic acid and docosahexaenoic acid) in the secondary prevention of cardiovascular disease: A meta-analysis of randomized, double-blind, placebo-controlled trials. Arch. Intern. Med. 2012, 172, 686–694. [Google Scholar] [PubMed]
- Kotwal, S.; Jun, M.; Sullivan, D.; Perkovic, V.; Neal, B. Omega 3 fatty acids and cardiovascular outcomes: Systematic review and meta-analysis. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Stevens, S.; Gorman, D.; Pan, A.; Warnakula, S.; Chowdhury, S.; Ward, H.; Johnson, L.; Crowe, F.; Hu, F.B.; et al. Association between fish consumption, long chain omega 3 fatty acids, and risk of cerebrovascular disease: Systematic review and meta-analysis. BMJ 2012, 345, e6698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nestel, P.; Clifton, P.; Colquhoun, D.; Noakes, M.; Mori, T.A.; Sullivan, D.; Thomas, B. Indications for omega-3 long chain polyunsaturated fatty acid in the prevention and treatment of cardiovascular disease. Heart Lung Circ. 2015, 24, 769–779. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Rimm, E.B.; Merchant, A.; Rosner, B.A.; Stampfer, M.J.; Willet, W.C.; Ascherio, A. Fish consumption and risk of stroke in men. JAMA 2002, 288, 3130–3136. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Harris, W.S.; Chung, M.; Lichtenstein, A.H.; Balk, E.M.; Kupelnick, B.; Jordan, H.S.; Lau, J. n-3 Fatty acids from fish or fish-oil supplements, but not alpha-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: A systematic review. Am. J. Clin. Nutr. 2006, 84, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Myint, P.K.; Welch, A.A.; Bingham, S.A.; Luben, R.N.; Wareham, N.J.; Day, N.E.; Khaw, K.T. Habitual fish consumption and risk of incident stroke: The European Prospective Investigation into Cancer (EPIC)-Norfolk prospective population study. Public Health Nutr. 2006, 9, 882–888. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Longstreth, W.T., Jr.; Lemaitre, R.N.; Manolio, T.A.; Kuller, L.H.; Burke, G.L.; Siscovick, D.S. Fish consumption and stroke risk in elderly individuals: The cardiovascular health study. Arch. Intern. Med. 2005, 165, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Yashodhara, B.M.; Umakanth, S.; Pappachan, J.M.; Bhat, S.K.; Kamath, R.; Choo, B.H. Omega-3 fatty acids: A comprehensive review of their role in health and disease. Postgrad. Med. J. 2009, 85, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Bays, H.E. Safety considerations with omega-3 fatty acid therapy. Am. J. Cardiol. 2007, 99, 35C–43C. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. Expert opinion: Omega-3 fatty acids and bleeding—Cause for concern? Am. J. Cardiol. 2007, 99, 44C–46C. [Google Scholar] [CrossRef] [PubMed]
- Watson, P.D.; Joy, P.S.; Nkonde, C.; Hessen, S.E.; Karalis, D.G. Comparison of bleeding complications with omega-3 fatty acids + aspirin + clopidogrel—versus—aspirin + clopidogrel in patients with cardiovascular disease. Am. J. Cardiol. 2009, 104, 1052–1054. [Google Scholar] [CrossRef] [PubMed]
- Salisbury, A.C.; Harris, W.S.; Amin, A.P.; Reid, K.J.; James, H.; O’Keefe, J.H., Jr.; Spertus, J.A. Relation between red blood cell omega-3 fatty acid index and bleeding during acute myocardial infarction. Am. J. Cardiol. 2012, 109, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Covington, M.B. Omega-3 fatty acids. Am. Fam. Physician 2004, 70, 133–140. [Google Scholar] [PubMed]
- Bays, H. Clinical overview of Omacor: A concentrated formulation of omega-3 polyunsaturated fatty acids. Am. J. Cardiol. 2006, 98, 71i–76i. [Google Scholar] [CrossRef] [PubMed]
- Myhre, A.M.; Carlsen, M.H.; Bøhn, S.K.; Wold, H.L.; Laake, P.; Blomhoff, R. Water-miscible, emulsified, and solid forms of retinol supplements are more toxic than oil-based preparations. Am. J. Clin. Nutr. 2003, 78, 1152–1159. [Google Scholar] [CrossRef] [PubMed]
- Despres, C.; Beuter, A.; Richer, F.; Poitras, K.; Veilleux, A.; Ayotte, P.; Dewailly, E.; Saint-Amour, D.; Muckle, G. Neuromotor functions in Inuit preschool children exposed to Pb, PCBs, and Hg. Neurotoxicol. Teratol. 2005, 27, 245–257. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokoła-Wysoczańska, E.; Wysoczański, T.; Wagner, J.; Czyż, K.; Bodkowski, R.; Lochyński, S.; Patkowska-Sokoła, B. Polyunsaturated Fatty Acids and Their Potential Therapeutic Role in Cardiovascular System Disorders—A Review. Nutrients 2018, 10, 1561. https://doi.org/10.3390/nu10101561
Sokoła-Wysoczańska E, Wysoczański T, Wagner J, Czyż K, Bodkowski R, Lochyński S, Patkowska-Sokoła B. Polyunsaturated Fatty Acids and Their Potential Therapeutic Role in Cardiovascular System Disorders—A Review. Nutrients. 2018; 10(10):1561. https://doi.org/10.3390/nu10101561
Chicago/Turabian StyleSokoła-Wysoczańska, Ewa, Tomasz Wysoczański, Jolanta Wagner, Katarzyna Czyż, Robert Bodkowski, Stanisław Lochyński, and Bożena Patkowska-Sokoła. 2018. "Polyunsaturated Fatty Acids and Their Potential Therapeutic Role in Cardiovascular System Disorders—A Review" Nutrients 10, no. 10: 1561. https://doi.org/10.3390/nu10101561




