Effect of Consumption of Cocoa-Derived Products on Uric Acid Crystallization in Urine of Healthy Volunteers
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Participants
2.3. Nutritional Intervention and Urine Collection
2.4. Sample Analysis
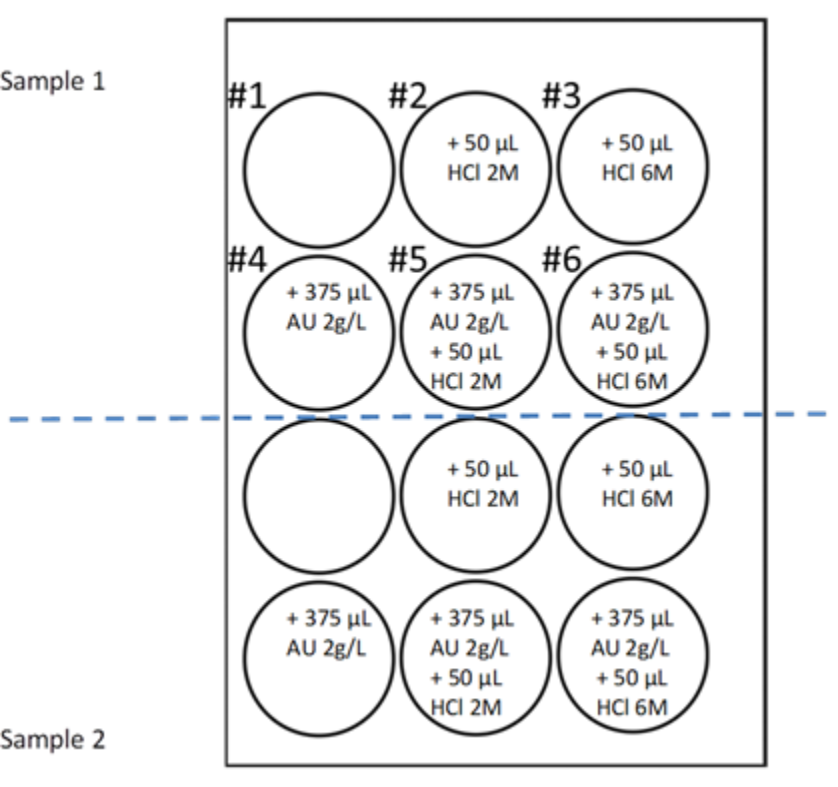
2.5. UAC Test
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ramello, A.; Vitale, C.; Marangella, M. Epidemiology of nephrolithiasis. J. Nephrol. 2000, 13, S65–S70. [Google Scholar]
- Brikowski, T.H.; Lotan, Y.; Pearle, M.S. Climate-related increase in the prevalence of urolithiasis in the United States. Proc. Natl. Acad. Sci. USA 2008, 105, 9841–9846. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Costa-Bauzá, A.; Ramis, M.; Montesinos, V.; Conte, A. Simple classification of renal calculi closely related to their micromorphology and etiology. Clin. Chim. Acta 2002, 322, 29–36. [Google Scholar] [CrossRef]
- Mandel, N.S.; Mandel, G.S. Urinary tract stone disease in the United States veteran population. I. Geographical frequency of occurrence. J. Urol. 1989, 142, 1513–1515. [Google Scholar] [CrossRef]
- Mandel, N.S.; Mandel, G.S. Urinary tract stone disease in the United States veteran population. II. Geographical analysis of variations in composition. J. Urol. 1989, 142, 1516–1521. [Google Scholar] [CrossRef]
- Gault, M.H.; Chafe, L. Relationship of frequency, age, sex, stone weight and composition in 15,624 stones: Comparison of resutls for 1980 to 1983 and 1995 to 1998. J. Urol. 2000, 164, 302–307. [Google Scholar] [CrossRef]
- Costa-Bauzá, A.; Ramis, M.; Montesinos, V.; Grases, F.; Conte, A.; Pizá, P.; Pieras, E.; Grases, F. Type of renal calculi: Variation with age and sex. World J. Urol. 2007, 25, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Daudon, M.; Traxer, O.; Conort, P.; Lacour, B.; Jungers, P. Type 2 diabetes increases the risk for uric acid stones. J. Am. Soc. Nephrol. 2006, 17, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Trinchieri, A.; Montanari, E. Prevalence of renal uric acid stones in the adult. Urolithiasis 2017, 45, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Hesse, A.; Brandle, E.; Wilbert, D.; Köhrmann, K.U.; Alken, P. Study on the prevalence and incidence of urolithiasis in Germany comparing the years 1979 vs. 2000. Eur. Urol. 2003, 44, 709–713. [Google Scholar] [CrossRef]
- Grases, F.; Costa-Bauzá, A.; Ramis, M.; Montesinos, V.; Conte, A. Recurrence of renal lithiasis. Scand. J. Urol. Nephrol. 2003, 37, 482–486. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, P.M.; Curhan, G.C.; D’Addessi, A.; Gambaro, G. Risk of recurrence of idiopathic calcium kidney stones: Analysis of data from the literature. J. Nephrol. 2017, 30, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Villacampa, A.I.; Costa-Bauzá, A.; Söhnel, O. Uric acid calculi: Types, etiology and mechanisms of formation. Clin. Chim. Acta 2000, 302, 89–104. [Google Scholar] [CrossRef]
- Sakhaee, K.; Adams-Huet, B.; Moe, O.W.; Pak, C.Y. Pathophysiologic basis for normouricosuric uric acid nephrolithiasis. Kidney Int. 2002, 62, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, P.; Bonny, O. Diagnosis and prevention of uric acid stones. Ther. Umsch. 2004, 61, 571–574. [Google Scholar] [CrossRef] [PubMed]
- Hess, B.; Jaeger, P. Physiopathology, etiology and medical treatment of non-calcium lithiasis. Rev. Prat. 1991, 41, 2037–2042. [Google Scholar] [PubMed]
- Heilberg, I.P. Treatment of patients with uric acid stones. Urolithiasis 2016, 44, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.C.; Assimos, D.G. Uric acid nephrolithiasis: Recent progress and future directions. Rev. Urol. 2007, 9, 17–27. [Google Scholar] [PubMed]
- Grases, F.; Rodriguez, A.; Costa-Bauza, A. Theobromine inhibits uric acid crystallization. A potential application in the treatment of uric acid nephrolithiasis. PLoS ONE 2014, 9, e111184. [Google Scholar] [CrossRef] [PubMed]
- Ptolemy, A.S.; Tzioumis, E.; Thomke, A.; Rifai, S.; Kellogg, M. Quantification of theobromine and caffeine in saliva, plasma and urine via liquid chromatography-tandem mass spectrometry: A single analytical protocol applicable to cocoa intervention studies. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2010, 878, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.; Costa-Bauzá, A.; Saez-Torres, C.; Rodrigo, D.; Grases, F. HPLC method for urinary theobromine determination: Effect of consumption of cocoa products on theobromine urinary excretion in children. Clin. Biochem. 2015, 48, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Craig, W.J.; Nguyen, T.T. Caffeine and theobromine levels in cocoa and carob products. J. Food Sci. 1984, 49, 302–303. [Google Scholar] [CrossRef]
- Lelo, A.; Birkett, D.J.; Robson, R.A.; Miners, J.O. Comparative pharmacokinetics of caffeine and its primary demethylated metabolites paraxanthine, theobromine and theophylline in man. Br. J. Clin. Pharmacol. 1986, 22, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Tarka, S.M.; Arnaud, M.J.; Kvorchik, B.H.; Vesell, E.S. Theobromine kinetics and metabolic disposition. Clin. Pharmacol. Ther. 1983, 34, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Tang-Liu, D.S.; Williams, R.L.; Riegelman, S. Disposition of caffeine and its metabolites in man. J. Pharm. Exp. Ther. 1983, 224, 180–185. [Google Scholar]
- Marshall, R.W.; Robertson, W.G. Nomograms for the estimation of the saturation of urine with calcium oxalate, calcium phosphate, magnesium ammonium phosphate, uric acid, sodium acid urate, ammonium acid urate and cystine. Clin. Chim. Acta 1976, 72, 253–260. [Google Scholar] [CrossRef]
- Fink, H.A.; Wilt, T.J.; Eidman, K.E.; Garimella, P.S.; MacDonald, R.; Rutks, I.R.; Brasure, M.; Kane, R.L.; Ouellette, J.; Monga, M. Medical management to prevent recurrent nephrolithiasis in adults: A systematic review for an American College of Physicians Clinical Guideline. Ann. Intern. Med. 2013, 158, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Spatola, L.; Ferraro, P.M.; Gambaro, G.; Badalamenti, S.; Dauriz, M. Metabolic syndrome and uric acid nephrolithiasis: Insulin resistance in focus. Metabolism 2018, 83, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zisman, A.L.; Coe, F.L.; Worcester, E.M. Kidney stones: An update on current pharmacological management and future directions. Expert Opin. Pharmacother. 2013, 14, 435–447. [Google Scholar] [CrossRef] [PubMed]

| Volunteer | pH | Uric Acid (mg/L) | Oxalate (mg/L) | |||||||||
| 1 | B | MC | PC | DC | B | MC | PC | DC | B | MC | PC | DC |
| 2 | 5.85 | 6.05 | 5.68 | 6.39 | 404 | 460 | 461 | 393 | 16 | 17 | 15 | 16 |
| 3 | 6.44 | 6.03 | 5.54 | 6.33 | 346 | 623 | 478 | 555 | 11 | 22 | 10 | 18 |
| 4 | 7.05 | 5.73 | 6.58 | 5.53 | 207 | 142 | 210 | 336 | 37 | 39 | 40 | 34 |
| 5 | 5.00 | 6.25 | 6.58 | 5.72 | 273 | 679 | 398 | 367 | 16 | 25 | 13 | 16 |
| 6 | 4.90 | 5.40 | 5.04 | 5.15 | 453 | 396 | 391 | 525 | 20 | 33 | 19 | 39 |
| 7 | 5.02 | 5.60 | 5.40 | 5.74 | 562 | 709 | 571 | 857 | 37 | 34 | 38 | 58 |
| 8 | 5.35 | 5.22 | 5.39 | 5.10 | 393 | 549 | 156 | 270 | 15 | 29 | 8 | 27 |
| 9 | 5.65 | 5.43 | 6.35 | 5.81 | 326 | 608 | 276 | 516 | 15 | 25 | 10 | 50 |
| 10 | 5.84 | 5.56 | 5.66 | 5.60 | 537 | 558 | 725 | 489 | 36 | 9 | 45 | 26 |
| 11 | 5.54 | 5.72 | 6.35 | 6.60 | 404 | 400 | 317 | 539 | 16 | 39 | 12 | 29 |
| 12 | 5.66 | 5.92 | 5.68 | 5.44 | 469 | 688 | 528 | 405 | 24 | 12 | 28 | 44 |
| 13 | 6.20 | 5.62 | 5.86 | 5.89 | 847 | 281 | 303 | 561 | 31 | 36 | 15 | 23 |
| 14 | 5.33 | 5.32 | 5.04 | 5.53 | 847 | 951 | 389 | 891 | 23 | 24 | 22 | 40 |
| 15 | 5.29 | 5.23 | 5.15 | 5.20 | 728 | 757 | 594 | 610 | 27 | 42 | 31 | 25 |
| 16 | 5.62 | 5.49 | 6.07 | 5.86 | 313 | 113 | 277 | 230 | 23 | 6 | 14 | 18 |
| 17 | 6.15 | 5.33 | 6.23 | 7.03 | 316 | 813 | 283 | 496 | 16 | 24 | 16 | 27 |
| 18 | 5.42 | 5.64 | 6.22 | 5.50 | 392 | 285 | 139 | 508 | 22 | 10 | 10 | 36 |
| 19 | 6.40 | 6.58 | 6.96 | 5.64 | 286 | 720 | 311 | 706 | 24 | 28 | 27 | 42 |
| 20 | 6.06 | 5.40 | 5.20 | 5.53 | 188 | 178 | 252 | 169 | 11 | 10 | 14 | 13 |
| mean | 5.69 | 5.65 | 5.80 | 5.72 | 425 | 507 | 387 | 483 | 22 | 24 | 21 | 32* |
| median | 5.64 | 5.58 | 5.68 | 5.59 | 393 | 554 | 353 | 502 | 21 | 25 | 16 | 30 |
| SD | 0.56 | 0.36 | 0.59 | 0.48 | 195 | 244 | 167 | 191 | 8 | 11 | 11 | 12 |
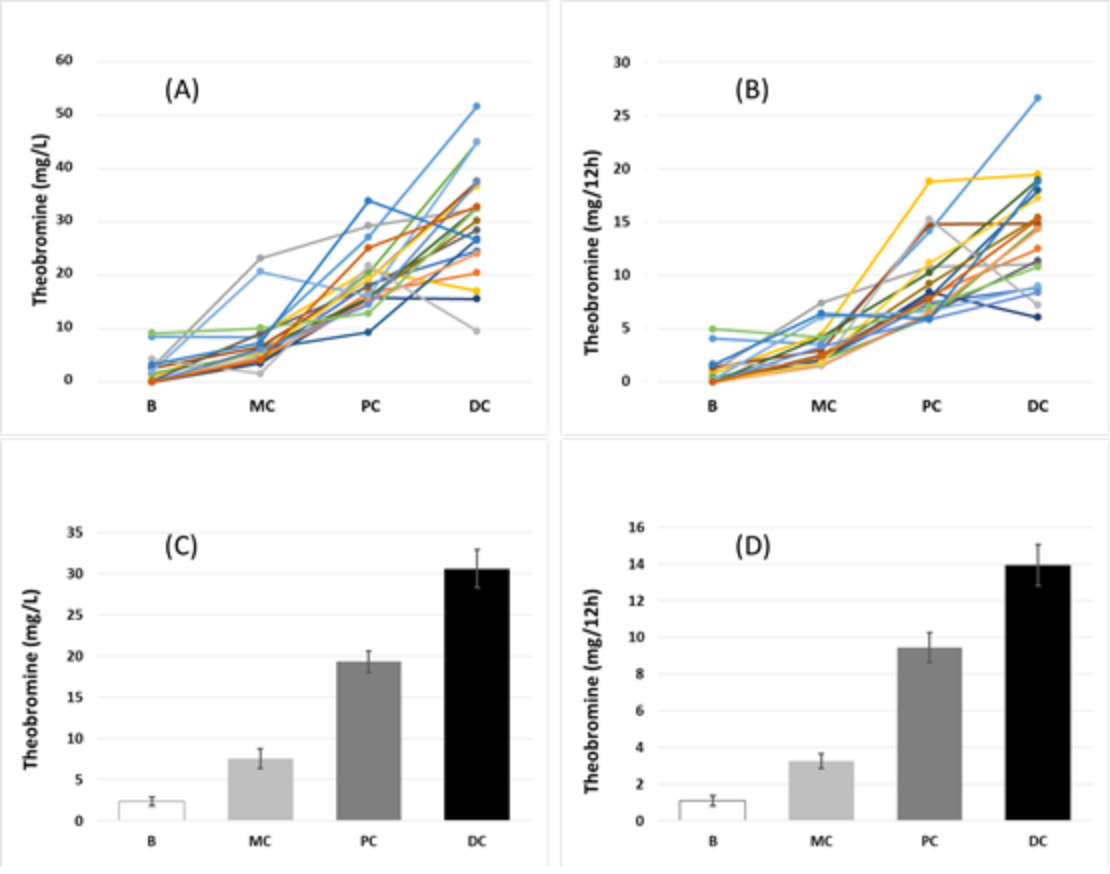
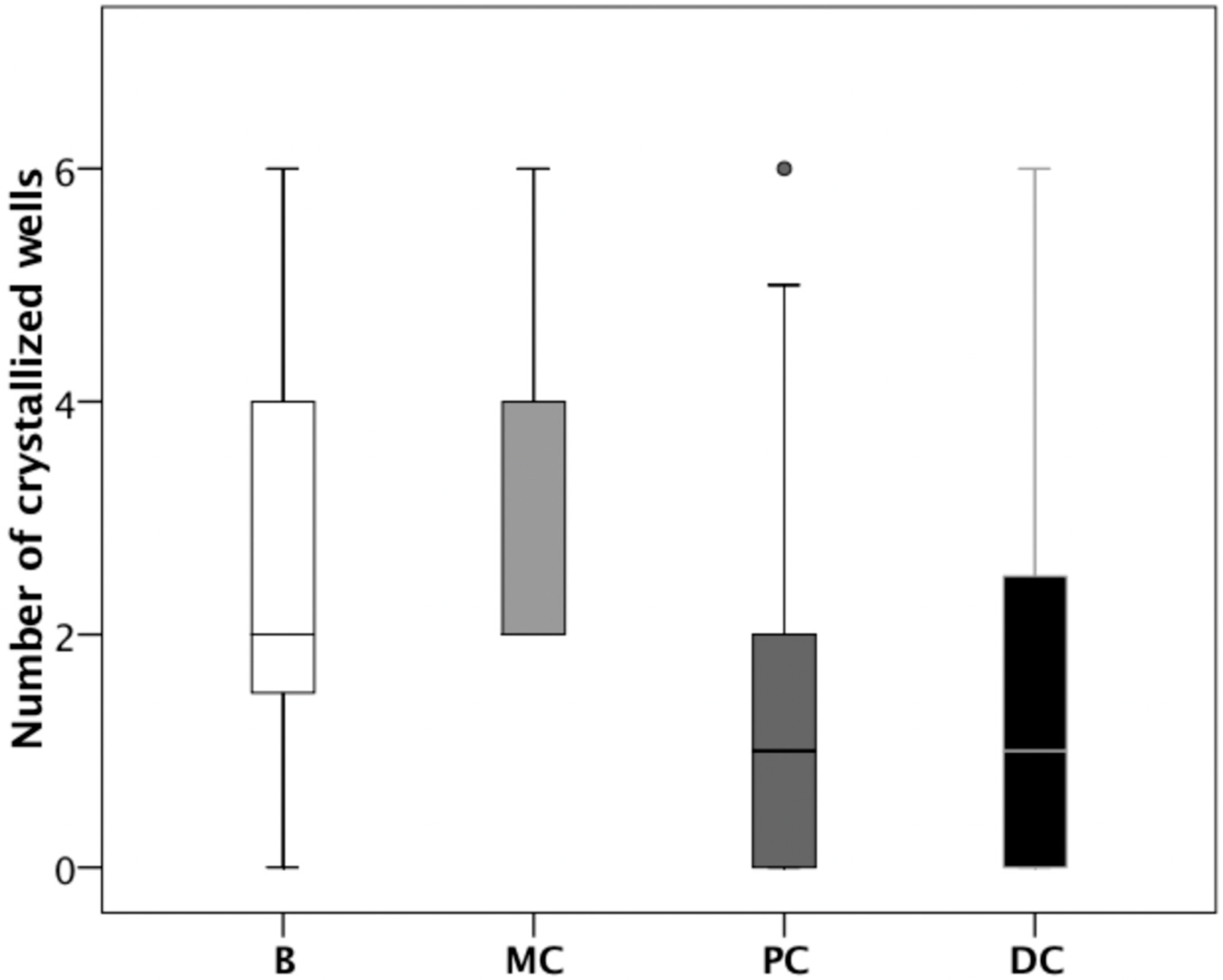
| Volunteer | Theobromine (mg/L) | UAC test | Uric acid RSS | |||||||||
| 1 | B | MC | PC | DC | B | MC | PC | DC | B | MC | PC | DC |
| 2 | 8.5 | 8.5 | 27.2 | 51.7 | 1 | 2 | 0 | 0 | −0.37 | −0.65 | 0.15 | −1.52 |
| 3 | 0.0 | 4.2 | 16.8 | 20.5 | 2 | 4 | 2 | 3 | −1.76 | −0.28 | 0.51 | −1.02 |
| 4 | 2.7 | 23.2 | 29.3 | 32.5 | 1 | 1 | 1 | 3 | −3.47 | −1.25 | 2.58 | 0.14 |
| 5 | 0.9 | 9.0 | 21.0 | 17.1 | 4 | 2 | 0 | 1 | 1.20 | −0.64 | −1.88 | −0.19 |
| 6 | 0.0 | 4.8 | 18.3 | 24.6 | 1 | 4 | 0 | 0 | 2.01 | 0.63 | 1.49 | 1.54 |
| 7 | 1.6 | 5.3 | 20.5 | 45.0 | 3 | 4 | 0 | 1 | 1.94 | 0.81 | 1.03 | 0.70 |
| 8 | 0.0 | 6.2 | 9.4 | 26.8 | 2 | 2 | 0 | 1 | 0.74 | 1.42 | −0.37 | 0.93 |
| 9 | 2.6 | 6.7 | 19.2 | 37.6 | 4 | 3 | 0 | 0 | −0.16 | 1.03 | −1.83 | −0.01 |
| 10 | 0.2 | 9.1 | 17.9 | 28.5 | 2 | 2 | 3 | 1 | −0.03 | 0.63 | 0.69 | 0.40 |
| 11 | 0.0 | 4.2 | 15.9 | 30.3 | 3 | 2 | 1 | 0 | 0.32 | −0.09 | −1.68 | −1.58 |
| 12 | 0.0 | 3.5 | 15.8 | 15.6 | 1 | 4 | 0 | 0 | 0.22 | 0.07 | 0.30 | 0.56 |
| 13 | 0.0 | 6.3 | 15.8 | 32.7 | 2 | 2 | 1 | 2 | −0.29 | −0.26 | −0.71 | −0.09 |
| 14 | 2.0 | 20.7 | 16.2 | 45.0 | 6 | 6 | 2 | 4 | 1.63 | 1.78 | 1.48 | 1.22 |
| 15 | 0.0 | 4.6 | 15.2 | 24.1 | 6 | 6 | 6 | 4 | 1.56 | 1.75 | 1.67 | 1.58 |
| 16 | 4.3 | 1.6 | 21.8 | 9.6 | 2 | 2 | 2 | 1 | −0.14 | −0.96 | −1.25 | −1.01 |
| 17 | 0.0 | 5.7 | 19.3 | 36.7 | 4 | 4 | 2 | 6 | −1.27 | 1.58 | −1.56 | −2.48 |
| 18 | 0.0 | 6.1 | 15.4 | 37.5 | 2 | 2 | 0 | 0 | 0.57 | −0.29 | −2.32 | 0.67 |
| 19 | 9.2 | 10.1 | 12.9 | 32.7 | 2 | 2 | 1 | 1 | −1.89 | −1.23 | −2.86 | 0.71 |
| 20 | 3.3 | 7.3 | 34.0 | 26.6 | 1 | 2 | 0 | 1 | −1.66 | −0.25 | 0.61 | −0.61 |
| mean | 2.3 | 7.6 | 19.3 | 30.6 | 2.6 | 3.0 | 1.3 | 1.6 | −0.01 | 0.18 | −0.35 | −0.00 |
| median | 1.0 | 6.2 | 18.1 | 31.4 | 2.0 | 2.0 | 1.0 | 1.0 | 0.09 | −0.15 | −0.11 | 0.08 |
| SD | 2.4 | 5.4 | 5.9 | 10.3 | 1.7 | 1.3 | 1.7 | 1.7 | 1.44 | 0.97 | 1.55 | 1.09 |
| Urine Sample | UAC Test | |||
|---|---|---|---|---|
| Uric acid (mg/L) | pH | mean | SD | |
| Sample 1 | 608 | 5.76 | 4 | 0 |
| Sample 2 | 210 | 5.69 | 2 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa-Bauza, A.; Grases, F.; Calvó, P.; Rodriguez, A.; Prieto, R.M. Effect of Consumption of Cocoa-Derived Products on Uric Acid Crystallization in Urine of Healthy Volunteers. Nutrients 2018, 10, 1516. https://doi.org/10.3390/nu10101516
Costa-Bauza A, Grases F, Calvó P, Rodriguez A, Prieto RM. Effect of Consumption of Cocoa-Derived Products on Uric Acid Crystallization in Urine of Healthy Volunteers. Nutrients. 2018; 10(10):1516. https://doi.org/10.3390/nu10101516
Chicago/Turabian StyleCosta-Bauza, Antonia, Felix Grases, Paula Calvó, Adrian Rodriguez, and Rafael M. Prieto. 2018. "Effect of Consumption of Cocoa-Derived Products on Uric Acid Crystallization in Urine of Healthy Volunteers" Nutrients 10, no. 10: 1516. https://doi.org/10.3390/nu10101516
APA StyleCosta-Bauza, A., Grases, F., Calvó, P., Rodriguez, A., & Prieto, R. M. (2018). Effect of Consumption of Cocoa-Derived Products on Uric Acid Crystallization in Urine of Healthy Volunteers. Nutrients, 10(10), 1516. https://doi.org/10.3390/nu10101516





