Dietary Choline Intake: Current State of Knowledge Across the Life Cycle
Abstract
:1. Introduction
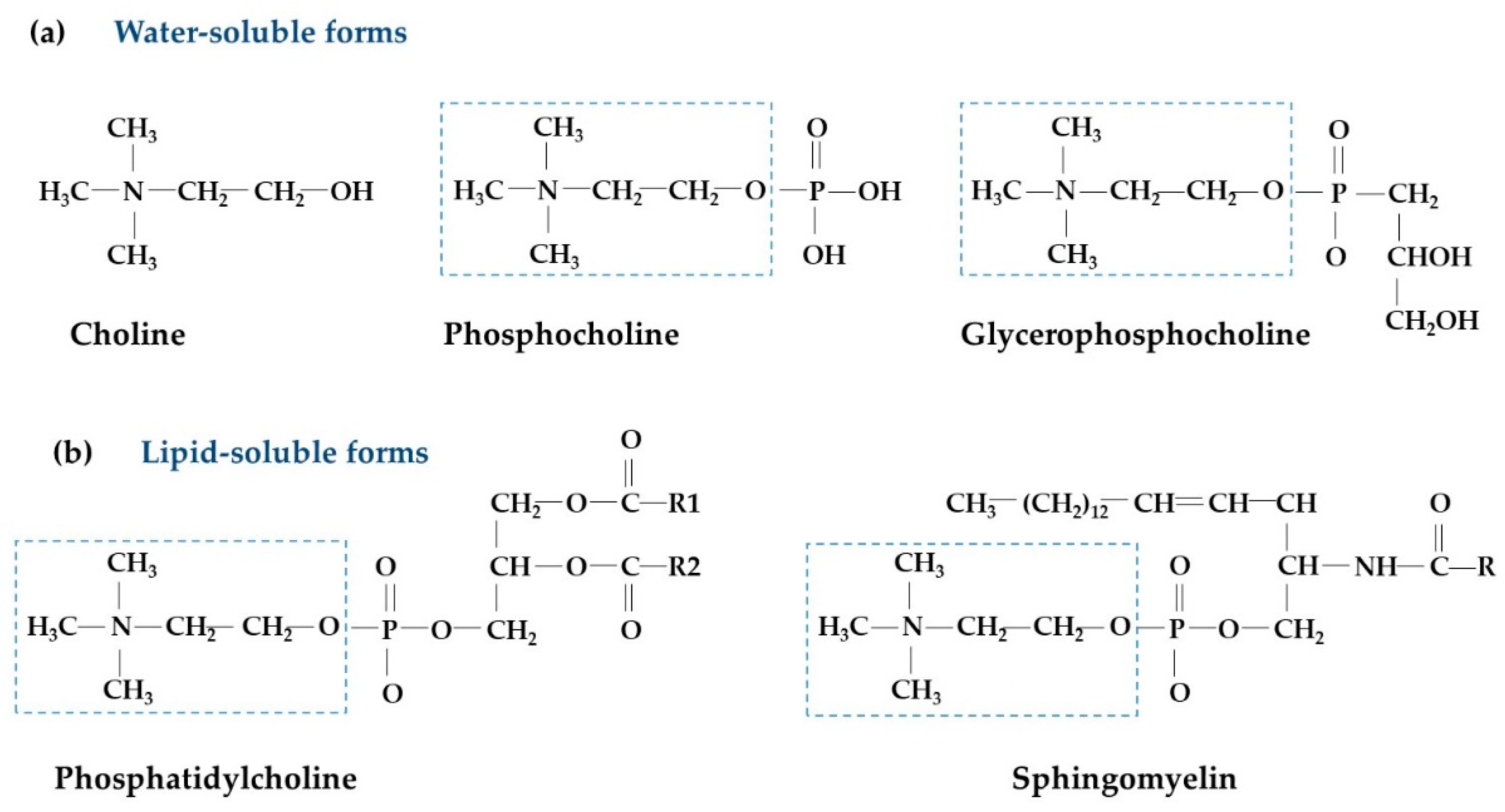
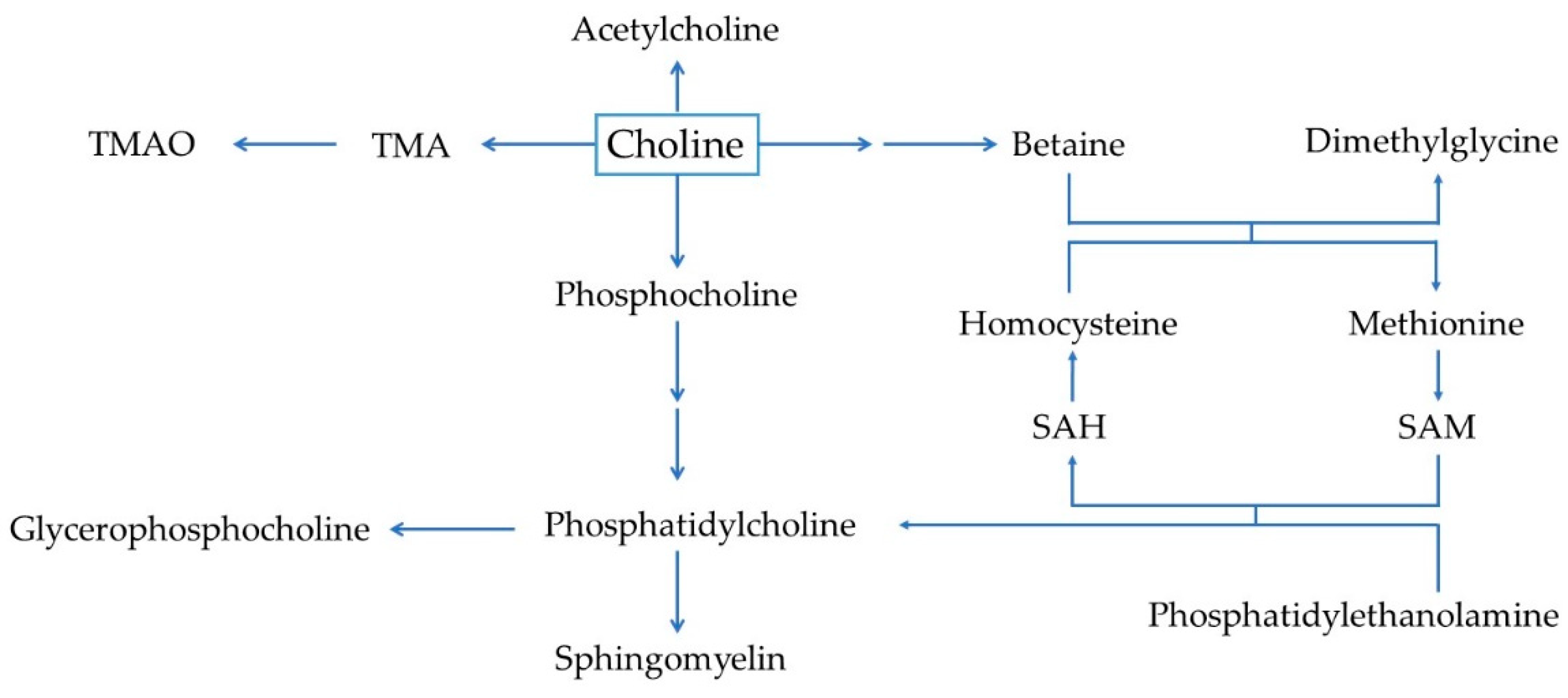
2. Overview of Choline Metabolism and Biological Functions
2.1. Choline Metabolism
2.2. Biological Functions of Choline
3. Choline Content in Dietary Food Sources
3.1. Choline Concentration in Human Milk
3.2. Choline Concentration in Infant Formulas
3.3. Choline Content in Dietary Food Sources
4. Dietary Recommendation for Choline
4.1. Adequate Intake Recommendation by Stage of the Life Cycle
4.2. Tolerable Upper Intake Levels
5. Dietary Assessment Methods and Validation
5.1. Dietary Assessment Methods
5.2. Dietary Assessment Validation
6. Dietary Choline Intake by Stage of Life Cycle
6.1. Dietary Choline Intakes in Adults
6.2. Dietary Choline Intakes in Other Age Groups
7. Summary and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Vance, D.E.; Ridgway, N.D. The methylation of phosphatidylethanolamine. Prog. Lipid Res. 1988, 27, 61–79. [Google Scholar] [CrossRef]
- Patterson, Y.K.; Bhagwat, A.S.; Williams, R.J.; Howe, C.J.; Holden, M.J. USD Database for The Choline Content of Common Foods, Release 2; Agricultural Research Service: Washington, DC, USA, 2008.
- Zeisel, S.H. The fetal origins of memory: The role of dietary choline in optimal brain development. J Pediatr. 2006, 149, S131136. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Choline: Critical role during fetal development and dietary requirements in adults. Annu. Rev. Nutr. 2006, 26, 229–250. [Google Scholar] [CrossRef] [PubMed]
- Leermakers, E.T.; Moreira, E.M.; Kiefte-de, J.C.; Darweesh, S.K.; Visser, T.; Voortman, T.; Bautista, P.K.; Chowdhury, R.; Gorman, D.; Bramer, W.M.; et al. Effects of choline on health across the life course: A systematic review. Nutr. Rev. 2015, 73, 500–522. [Google Scholar] [CrossRef] [PubMed]
- Buchman, A.L.; Moukarzel, A.; Jenden, D.J.; Roch, M.; Rice, K.; Ament, M.E. Low plasma free choline is prevalent in patients receiving long term parenteral nutrition and is associated with hepatic aminotransferase abnormalities. Clin. Nutr. 1993, 12, 33–37. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Da Costa, K.A.; Franklin, P.D.; Alexander, E.A.; Lamont, J.T.; Sheard, N.F.; Beiser, A. Choline, an essential nutrient for humans. FASEB J. 1991, 5, 2093–2098. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; The National Academy Press: Washington, DC, USA, 1998. [Google Scholar]
- European Food Safety Authority. Dietary reference values for choline. EFSA J. 2016, 14, e04484. [Google Scholar] [CrossRef]
- Zeisel, S.H. Dietary choline: Biochemistry, physiology, and pharmacology. Annu. Rev. Nutr. 1981, 1, 95–121. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Field, C.J.; Jacobs, R.L. Should the forms of dietary choline also be considered when estimating dietary intake and the implications for health? Lipid Technol. 2015, 27, 227–230. [Google Scholar] [CrossRef]
- Cheng, W.-L.; Holmes-McNary, M.Q.; Mar, M.-H.; Lien, E.L.; Zeisel, S.H. Bioavailability of choline and choline esters from milk in rat pups. J. Nutr. Biochem. 1996, 7, 457–464. [Google Scholar] [CrossRef]
- Lewis, E.D.; Richard, C.; Goruk, S.; Dellschaft, N.S.; Curtis, J.M.; Jacobs, R.L.; Field, C.J. The form of choline in the maternal diet affects immune development in suckled rat offspring. J. Nutr. 2016, 146, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Richard, C.; Lewis, E.D.; Goruk, S.; Wadge, E.; Curtis, J.M.; Jacobs, R.L.; Field, C.J. Feeding a mixture of choline forms to lactating dams improves the development of the immune system in sprague-dawley rat offspring. Nutrients 2017, 9, 567. [Google Scholar] [CrossRef] [PubMed]
- Dellschaft, N.S.; Richard, C.; Lewis, E.D.; Goruk, S.; Jacobs, R.L.; Curtis, J.M.; Field, C.J. The dietary form of choline during lactation affects maternal immune function in rats. Eur. J. Nutr. 2018, 57, 2189–2199. [Google Scholar] [CrossRef] [PubMed]
- Sarter, M.; Parikh, V. Choline transporters, cholinergic transmission and cognition. Nat. Rev. Neurosci. 2005, 6, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Varoqui, H.; Erickson, J.D. The cytoplasmic tail of the vesicular acetylcholine transporter contains a synaptic vesicle targeting signal. J. Biol. Chem. 1998, 273, 9094–9098. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, K.; Fujii, T. Basic and clinical aspects of non-neuronal acetylcholine: Overview of non-neuronal cholinergic systems and their biological significance. J. Pharmacol. Sci. 2008, 106, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Wessler, I.; Kirkpatrick, C.J. Acetylcholine beyond neurons: The non-neuronal cholinergic system in humans. Br. J. Pharmacol. 2008, 154, 1558–1571. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Wishnok, J.S.; Blusztajn, J.K. Formation of methylamines from ingested choline and lecithin. J. Pharmacol. Exp. Ther. 1983, 225, 320–324. [Google Scholar] [PubMed]
- Romano, K.A.; Vivas, E.I.; Amador-Noguez, D.; Rey, F.E. Intestinal microbiota composition modulates choline bioavailability from diet and accumulation of the proatherogenic metabolite trimethylamine-N-oxide. MBio 2015, 6, e02481. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.R.; Chaykin, S. The biosynthesis of trimethylamine-N-oxide. J. Biol. Chem. 1962, 237, 1309–1313. [Google Scholar] [CrossRef]
- Bianchi, G.; Azzone, G.F. Oxidation of choline in rat liver mitochondria. J. Biol. Chem. 1964, 239, 3947–3955. [Google Scholar] [PubMed]
- Munoz-Clares, R.A.; Diaz-Sanchez, A.G.; Gonzalez-Segura, L.; Montiel, C. Kinetic and structural features of betaine aldehyde dehydrogenases: Mechanistic and regulatory implications. Arch. Biochem. Biophys. 2010, 493, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Garrow, T.A. Purification, kinetic properties, and cdna cloning of mammalian betaine-homocysteine methyltransferase. J. Biol. Chem. 1996, 271, 22831–22838. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Reyes, C.V.; Wallig, M.A.; Garrow, T.A. Immunohistochemical detection of betaine-homocysteine S-methyltransferase in human, pig, and rat liver and kidney. Arch. Biochem. Biophys. 2001, 393, 184–186. [Google Scholar] [CrossRef] [PubMed]
- Lever, M.; Slow, S. The clinical significance of betaine, an osmolyte with a key role in methyl group metabolism. Clin. Biochem. 2010, 43, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Kalari, K.; Fridley, B.L.; Jenkins, G.; Ji, Y.; Abo, R.; Hebbring, S.; Zhang, J.; Nye, M.D.; Leeder, J.S.; et al. Betaine-homocysteine methyltransferase: Human liver genotype-phenotype correlation. Mol. Genet. Metab. 2011, 102, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Pajares, M.A.; Perez-Sala, D. Betaine homocysteine S-methyltransferase: Just a regulator of homocysteine metabolism? Cell Mol. Life Sci. 2006, 63, 2792–2803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeisel, S.H. Dietary choline deficiency causes DNA strand breaks and alters epigenetic marks on DNA and histones. Mutat. Res. 2012, 733, 34–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haubrich, D.R.; Gerber, N.H. Choline dehydrogenase. Assay, properties and inhibitors. Biochem. Pharmacol. 1981, 30, 2993–3000. [Google Scholar] [CrossRef]
- Grossman, E.B.; Hebert, S.C. Renal inner medullary choline dehydrogenase activity: Characterization and modulation. Am. J. Physiol. 1989, 256, F107–F112. [Google Scholar] [CrossRef] [PubMed]
- McKeever, M.P.; Weir, D.G.; Molloy, A.; Scott, J.M. Betaine-homocysteine methyltransferase: Organ distribution in man, pig and rat and subcellular distribution in the rat. Clin. Sci. (Lond.) 1991, 81, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Soloway, S.; Stetten, D., Jr. The metabolism of choline and its conversion to glycine in the rat. J. Biol. Chem. 1953, 204, 207–214. [Google Scholar] [PubMed]
- Li, Z.; Vance, D.E. Phosphatidylcholine and choline homeostasis. J. Lipid Res. 2008, 49, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, E.P. The synthesis of cytidine diphosphate choline, cytidine diphosphate ethanolamine, and related compounds. J. Biol. Chem. 1956, 222, 185–191. [Google Scholar] [PubMed]
- Sundler, R.; Akesson, B. Biosynthesis of phosphatidylethanolamines and phosphatidylcholines from ethanolamine and choline in rat liver. Biochem. J. 1975, 146, 309–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeLong, C.J.; Shen, Y.J.; Thomas, M.J.; Cui, Z. Molecular distinction of phosphatidylcholine synthesis between the CDP-choline pathway and phosphatidylethanolamine methylation pathway. J. Biol. Chem. 1999, 274, 29683–29688. [Google Scholar] [CrossRef] [PubMed]
- Bremer, J.; Greenberg, D.M. Biosynthesis of choline in vitro. Biochim. Biophys. Acta 1960, 37, 173–175. [Google Scholar] [CrossRef]
- Vance, J.E. Phosphatidylserine and phosphatidylethanolamine in mammalian cells: Two metabolically related aminophospholipids. J. Lipid Res. 2008, 49, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- Ridgway, N.D.; Yao, Z.; Vance, D.E. Phosphatidylethanolamine levels and regulation of phosphatidylethanolamine N-methyltransferase. J. Biol. Chem. 1989, 264, 1203–1207. [Google Scholar] [PubMed]
- Cantoni, G.L. Biological methylation: Selected aspects. Annu. Rev. Biochem. 1975, 44, 435–451. [Google Scholar] [CrossRef] [PubMed]
- Infante, J.P.; Kinsella, J.E. Phospholipid synthesis in mammary tissue. Choline and ethanolamine kinases: Kinetic evidence for two discrete active sites. Lipids 1976, 11, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Bjornstad, P. Phospholipase activity in rat liver mitochondria studied by the use of endogenous substrates. J. Lipid Res. 1966, 7, 612–620. [Google Scholar] [PubMed]
- Noga, A.A.; Vance, D.E. Insights into the requirement of phosphatidylcholine synthesis for liver function in mice. J. Lipid Res. 2003, 44, 1998–2005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horl, G.; Wagner, A.; Cole, L.K.; Malli, R.; Reicher, H.; Kotzbeck, P.; Kofeler, H.; Hofler, G.; Frank, S.; Bogner-Strauss, J.G.; et al. Sequential synthesis and methylation of phosphatidylethanolamine promote lipid droplet biosynthesis and stability in tissue culture and in vivo. J. Biol. Chem. 2011, 286, 17338–17350. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, K.A.; Sanders, L.M.; Fischer, L.M.; Zeisel, S.H. Docosahexaenoic acid in plasma phosphatidylcholine may be a potential marker for in vivo phosphatidylethanolamine N-methyltransferase activity in humans. Am. J. Clin. Nutr. 2011, 93, 968–974. [Google Scholar] [CrossRef] [PubMed]
- West, A.A.; Yan, J.; Jiang, X.; Perry, C.A.; Innis, S.M.; Caudill, M.A. Choline intake influences phosphatidylcholine DHA enrichment in nonpregnant women but not in pregnant women in the third trimester. Am. J. Clin. Nutr. 2013, 97, 718–727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, G.M.; Carmichael, S.L.; Yang, W.; Selvin, S.; Schaffer, D.M. Periconceptional dietary intake of choline and betaine and neural tube defects in offspring. Am. J. Epidemiol. 2004, 160, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Detopoulou, P.; Panagiotakos, D.B.; Antonopoulou, S.; Pitsavos, C.; Stefanadis, C. Dietary choline and betaine intakes in relation to concentrations of inflammatory markers in healthy adults: The ATTICA study. Am. J. Clin. Nutr. 2008, 87, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Gammon, M.D.; Zeisel, S.H.; Lee, Y.L.; Wetmur, J.G.; Teitelbaum, S.L.; Bradshaw, P.T.; Neugut, A.I.; Santella, R.M.; Chen, J. Choline metabolism and risk of breast cancer in a population-based study. FASEB J. 2008, 22, 2045–2052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, G.M.; Finnell, R.H.; Blom, H.J.; Carmichael, S.L.; Vollset, S.E.; Yang, W.; Ueland, P.M. Choline and risk of neural tube defects in a folate-fortified population. Epidemiology 2009, 20, 714–719. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.T.; Dyer, R.A.; King, D.J.; Richardson, K.J.; Innis, S.M. Early second trimester maternal plasma choline and betaine are related to measures of early cognitive development in term infants. PLoS ONE 2012, 7, e43448. [Google Scholar] [CrossRef] [PubMed]
- Boeke, C.E.; Gillman, M.W.; Hughes, M.D.; Rifas-Shiman, S.L.; Villamor, E.; Oken, E. Choline intake during pregnancy and child cognition at age 7 years. Am. J. Epidemiol. 2013, 177, 1338–1347. [Google Scholar] [CrossRef] [PubMed]
- Strain, J.J.; McSorley, E.M.; van Wijngaarden, E.; Kobrosly, R.W.; Bonham, M.P.; Mulhern, M.S.; McAfee, A.J.; Davidson, P.W.; Shamlaye, C.F.; Henderson, J.; et al. Choline status and neurodevelopmental outcomes at 5 years of age in the Seychelles child development nutrition study. Br. J. Nutr. 2013, 110, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Shu, X.O.; Xiang, Y.B.; Li, H.; Yang, G.; Gao, Y.T.; Zheng, W.; Zhang, X. Higher dietary choline intake is associated with lower risk of nonalcoholic fatty liver in normal-weight chinese women. J. Nutr. 2014, 144, 2034–2040. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Li, X.; Ren, A.; Du, M.; Du, H.; Shu, Y.; Zhu, L.; Wang, W. Choline and betaine consumption lowers cancer risk: A meta-analysis of epidemiologic studies. Sci. Rep. 2016, 6, 35547. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.F.; Chen, X.L.; Zhou, Z.G.; Zhang, Y.J.; Lan, Q.Y.; Liao, G.C.; Chen, Y.M.; Zhu, H.L. Higher dietary intakes of choline and betaine are associated with a lower risk of primary liver cancer: A case-control study. Sci. Rep. 2017, 7, 679. [Google Scholar] [CrossRef] [PubMed]
- Obeid, R.; Awwad, H.M.; Knell, A.I.; Hubner, U.; Geisel, J. Glucose and fat tolerance tests induce differential responses in plasma choline metabolites in healthy subjects. Nutrients 2018, 10, 1209. [Google Scholar] [CrossRef] [PubMed]
- Rees, W.D.; Hay, S.M.; Cruickshank, M. An imbalance in the methionine content of the maternal diet reduces postnatal growth in the rat. Metabolism 2006, 55, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Luka, Z.; Mudd, S.H.; Wagner, C. Glycine N-methyltransferase and regulation of S-adenosylmethionine levels. J. Biol. Chem. 2009, 284, 22507–22511. [Google Scholar] [CrossRef] [PubMed]
- Niculescu, M.D.; Craciunescu, C.N.; Zeisel, S.H. Dietary choline deficiency alters global and gene-specific DNA methylation in the developing hippocampus of mouse fetal brains. FASEB J. 2006, 20, 43–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovacheva, V.P.; Mellott, T.J.; Davison, J.M.; Wagner, N.; Lopez-Coviella, I.; Schnitzler, A.C.; Blusztajn, J.K. Gestational choline deficiency causes global and igf2 gene DNA hypermethylation by up-regulation of dnmt1 expression. J. Biol. Chem. 2007, 282, 31777–31788. [Google Scholar] [CrossRef] [PubMed]
- Davison, J.M.; Mellott, T.J.; Kovacheva, V.P.; Blusztajn, J.K. Gestational choline supply regulates methylation of histone h3, expression of histone methyltransferases g9a (kmt1c) and suv39h1 (kmt1a), and DNA methylation of their genes in rat fetal liver and brain. J. Biol. Chem. 2009, 284, 1982–1989. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Yan, J.; West, A.A.; Perry, C.A.; Malysheva, O.V.; Devapatla, S.; Pressman, E.; Vermeylen, F.; Caudill, M.A. Maternal choline intake alters the epigenetic state of fetal cortisol-regulating genes in humans. FASEB J. 2012, 26, 3563–3574. [Google Scholar] [CrossRef] [PubMed]
- Shaw, G.M.; Carmichael, S.L.; Laurent, C.; Rasmussen, S.A. Maternal nutrient intakes and risk of orofacial clefts. Epidemiology 2006, 17, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Shaw, G.M.; Carmichael, S.L.; Rasmussen, S.A.; Waller, D.K.; Pober, B.R.; Anderka, M.; National Birth Defects Prevention Study. Nutrient intakes in women and congenital diaphragmatic hernia in their offspring. Birth Defects Res. A Clin. Mol. Teratol. 2008, 82, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, S.L.; Yang, W.; Correa, A.; Olney, R.S.; Shaw, G.M.; National Birth Defects Prevention Study. Hypospadias and intake of nutrients related to one-carbon metabolism. J. Urol. 2009, 181, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.; Deng, L.; Mikael, L.G.; Yan, J.; Pickell, L.; Wu, Q.; Caudill, M.A.; Rozen, R. Low dietary choline and low dietary riboflavin during pregnancy influence reproductive outcomes and heart development in mice. Am. J. Clin. Nutr. 2010, 91, 1035–1043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meck, W.H.; Williams, C.L. Metabolic imprinting of choline by its availability during gestation: Implications for memory and attentional processing across the lifespan. Neurosci. Biobehav. Rev. 2003, 27, 385–399. [Google Scholar] [CrossRef]
- Signore, C.; Ueland, P.M.; Troendle, J.; Mills, J.L. Choline concentrations in human maternal and cord blood and intelligence at 5 y of age. Am. J. Clin. Nutr. 2008, 87, 896–902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheatham, C.L.; Goldman, B.D.; Fischer, L.M.; da Costa, K.A.; Reznick, J.S.; Zeisel, S.H. Phosphatidylcholine supplementation in pregnant women consuming moderate-choline diets does not enhance infant cognitive function: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2012, 96, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Villamor, E.; Rifas-Shiman, S.L.; Gillman, M.W.; Oken, E. Maternal intake of methyl-donor nutrients and child cognition at 3 years of age. Paediatr. Perinat. Epidemiol. 2012, 26, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Wiedeman, A.M.; Chau, C.M.Y.; Grunau, R.E.; McCarthy, D.; Yurko-Mauro, K.; Dyer, R.A.; Innis, S.M.; Devlin, A.M. Plasma betaine is positively associated with developmental outcomes in healthy toddlers at age 2 years who are not meeting the recommended adequate intake for dietary choline. J. Nutr. 2018, 148, 1309–1314. [Google Scholar] [CrossRef] [PubMed]
- Poly, C.; Massaro, J.M.; Seshadri, S.; Wolf, P.A.; Cho, E.; Krall, E.; Jacques, P.F.; Au, R. The relation of dietary choline to cognitive performance and white-matter hyperintensity in the Framingham offspring cohort. Am. J. Clin. Nutr. 2011, 94, 1584–1591. [Google Scholar] [CrossRef] [PubMed]
- Nurk, E.; Refsum, H.; Bjelland, I.; Drevon, C.A.; Tell, G.S.; Ueland, P.M.; Vollset, S.E.; Engedal, K.; Nygaard, H.A.; Smith, D.A. Plasma free choline, betaine and cognitive performance: The Hordaland health study. Br. J. Nutr. 2013, 109, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Ladd, S.L.; Sommer, S.A.; LaBerge, S.; Toscano, W. Effect of phosphatidylcholine on explicit memory. Clin. Neuropharmacol. 1993, 16, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Spiers, P.A.; Myers, D.; Hochanadel, G.S.; Lieberman, H.R.; Wurtman, R.J. Citicoline improves verbal memory in aging. Arch. Neurol. 1996, 53, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Donohoe, R.T. The influence on cognition of the interactions between lecithin, carnitine and carbohydrate. Psychopharmacology (Berl.) 2004, 175, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Knott, V.; de la Salle, S.; Choueiry, J.; Impey, D.; Smith, D.; Smith, M.; Beaudry, E.; Saghir, S.; Ilivitsky, V.; Labelle, A. Neurocognitive effects of acute choline supplementation in low, medium and high performer healthy volunteers. Pharmacol. Biochem. Behav. 2015, 131, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Naber, M.; Hommel, B.; Colzato, L.S. Improved human visuomotor performance and pupil constriction after choline supplementation in a placebo-controlled double-blind study. Sci. Rep. 2015, 5, 13188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lippelt, D.P.; van der Kint, S.; van Herk, K.; Naber, M. No acute effects of choline bitartrate food supplements on memory in healthy, young, human adults. PLoS ONE 2016, 11, e0157714. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, K.A.; Badea, M.; Fischer, L.M.; Zeisel, S.H. Elevated serum creatine phosphokinase in choline-deficient humans: Mechanistic studies in C2C12 mouse myoblasts. Am. J. Clin. Nutr. 2004, 80, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, K.W.; Zilversmit, D.B. Exchange of phospholipids between liver mitochondria and microsomes in vitro. J. Biol. Chem. 1968, 243, 3596–3602. [Google Scholar] [PubMed]
- Devaux, P.F. Static and dynamic lipid asymmetry in cell membranes. Biochemistry 1991, 30, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Agellon, L.B.; Allen, T.M.; Umeda, M.; Jewell, L.; Mason, A.; Vance, D.E. The ratio of phosphatidylcholine to phosphatidylethanolamine influences membrane integrity and steatohepatitis. Cell Metab. 2006, 3, 321–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ling, J.; Chaba, T.; Zhu, L.F.; Jacobs, R.L.; Vance, D.E. Hepatic ratio of phosphatidylcholine to phosphatidylethanolamine predicts survival after partial hepatectomy in mice. Hepatology 2012, 55, 1094–1102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, J.A.; Fieldsend, J.K. Phosphatidylcholine synthesis for incorporation into membranes or for secretion as plasma lipoproteins by golgi membranes of rat liver. J. Lipid Res. 1987, 28, 268–278. [Google Scholar] [PubMed]
- Vance, D.E. Role of phosphatidylcholine biosynthesis in the regulation of lipoprotein homeostasis. Curr. Opin. Lipidol. 2008, 19, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Corbin, K.D.; Zeisel, S.H. Choline metabolism provides novel insights into nonalcoholic fatty liver disease and its progression. Curr. Opin. Gastroenterol. 2012, 28, 159–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guerrerio, A.L.; Colvin, R.M.; Schwartz, A.K.; Molleston, J.P.; Murray, K.F.; Diehl, A.; Mohan, P.; Schwimmer, J.B.; Lavine, J.E.; Torbenson, M.S.; et al. Choline intake in a large cohort of patients with nonalcoholic fatty liver disease. Am. J. Clin. Nutr. 2012, 95, 892–900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeisel, S.H. Choline: An important nutrient in brain development, liver function and carcinogenesis. J. Am. Coll. Nutr. 1992, 11, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Buchman, A.L.; Ament, M.E.; Sohel, M.; Dubin, M.; Jenden, D.J.; Roch, M.; Pownall, H.; Farley, W.; Awal, M.; Ahn, C. Choline deficiency causes reversible hepatic abnormalities in patients receiving parenteral nutrition: Proof of a human choline requirement: A placebo-controlled trial. J. Parenter. Enteral. Nutr. 2001, 25, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Gerhard, G.T.; Duell, P.B. Homocysteine and atherosclerosis. Curr. Opin. Lipidol. 1999, 10, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Leach, N.V.; Dronca, E.; Vesa, S.C.; Sampelean, D.P.; Craciun, E.C.; Lupsor, M.; Crisan, D.; Tarau, R.; Rusu, R.; Para, I.; et al. Serum homocysteine levels, oxidative stress and cardiovascular risk in non-alcoholic steatohepatitis. Eur. J. Intern. Med. 2014, 25, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Dalmeijer, G.W.; Olthof, M.R.; Verhoef, P.; Bots, M.L.; van der Schouw, Y.T. Prospective study on dietary intakes of folate, betaine, and choline and cardiovascular disease risk in women. Eur. J. Clin. Nutr. 2008, 62, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Konstantinova, S.V.; Tell, G.S.; Vollset, S.E.; Nygard, O.; Bleie, O.; Ueland, P.M. Divergent associations of plasma choline and betaine with components of metabolic syndrome in middle age and elderly men and women. J. Nutr. 2008, 138, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.A.; Shea, J.W. Dietary choline and betaine and risk of CVD: A systematic review and meta-analysis of prospective studies. Nutrients 2017, 9, 711. [Google Scholar] [CrossRef] [PubMed]
- Schwab, U.; Torronen, A.; Toppinen, L.; Alfthan, G.; Saarinen, M.; Aro, A.; Uusitupa, M. Betaine supplementation decreases plasma homocysteine concentrations but does not affect body weight, body composition, or resting energy expenditure in human subjects. Am. J. Clin. Nutr. 2002, 76, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Steenge, G.R.; Verhoef, P.; Katan, M.B. Betaine supplementation lowers plasma homocysteine in healthy men and women. J. Nutr. 2003, 133, 1291–1295. [Google Scholar] [CrossRef] [PubMed]
- Olthof, M.R.; Brink, E.J.; Katan, M.B.; Verhoef, P. Choline supplemented as phosphatidylcholine decreases fasting and postmethionine-loading plasma homocysteine concentrations in healthy men. Am. J. Clin. Nutr. 2005, 82, 111–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, T.; Chen, Y.; Yang, B.; Yang, J.; Wahlqvist, M.L.; Li, D. Meta-analysis of B vitamin supplementation on plasma homocysteine, cardiovascular and all-cause mortality. Clin. Nutr. 2012, 31, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Guo, L.L.; Cai, L.L.; Zhu, X.J.; Shu, J.L.; Liu, X.L.; Jin, H.M. Homocysteine-lowering therapy does not lead to reduction in cardiovascular outcomes in chronic kidney disease patients: A meta-analysis of randomised, controlled trials. Br. J. Nutr. 2012, 108, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Mente, A.; Chalcraft, K.; Ak, H.; Davis, A.D.; Lonn, E.; Miller, R.; Potter, M.A.; Yusuf, S.; Anand, S.S.; McQueen, M.J. The relationship between trimethylamine-N-oxide and prevalent cardiovascular disease in a multiethnic population living in Canada. Can. J. Cardiol. 2015, 31, 1189–1194. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Gregory, J.C.; Org, E.; Buffa, J.A.; Gupta, N.; Wang, Z.; Li, L.; Fu, X.; Wu, Y.; Mehrabian, M.; et al. Gut microbial metabolite TMAO enhances platelet hyperreactivity and thrombosis risk. Cell 2016, 165, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.A.; Corbin, K.D.; da Costa, K.A.; Zhang, S.; Zhao, X.; Galanko, J.A.; Blevins, T.; Bennett, B.J.; O’Connor, A.; Zeisel, S.H. Effect of egg ingestion on trimethylamine-N-oxide production in humans: A randomized, controlled, dose-response study. Am. J. Clin. Nutr. 2014, 100, 778–786. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Missimer, A.; Murillo, A.G.; Lemos, B.S.; Malysheva, O.V.; Caudill, M.A.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 eggs/day increases hdl cholesterol and plasma choline while plasma trimethylamine-N-oxide is unchanged in a healthy population. Lipids 2017, 52, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Mueller, D.M.; Allenspach, M.; Othman, A.; Saely, C.H.; Muendlein, A.; Vonbank, A.; Drexel, H.; von Eckardstein, A. Plasma levels of trimethylamine-N-oxide are confounded by impaired kidney function and poor metabolic control. Atherosclerosis 2015, 243, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Romano, K.A.; Martinez-Del, C.A.; Kasahara, K.; Chittim, C.L.; Vivas, E.I.; Amador-Noguez, D.; Balskus, E.P.; Rey, F.E. Metabolic, epigenetic, and transgenerational effects of gut bacterial choline consumption. Cell Host Microbe 2017, 22, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Landfald, B.; Valeur, J.; Berstad, A.; Raa, J. Microbial trimethylamine-N-oxide as a disease marker: Something fishy? Microb. Ecol. Health Dis. 2017, 28, 1327309. [Google Scholar] [CrossRef] [PubMed]
- Kruger, R.; Merz, B.; Rist, M.J.; Ferrario, P.G.; Bub, A.; Kulling, S.E.; Watzl, B. Associations of current diet with plasma and urine TMAO in the KarMeN study: Direct and indirect contributions. Mol. Nutr. Food Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The World Health Organization’s Infant Feeding Recommendation; WHO: Geneva, Switzerland, 2001. [Google Scholar]
- Zeisel, S.H.; Wurtman, R.J. Developmental changes in rat blood choline concentration. Biochem. J. 1981, 198, 565–570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmes, H.C.; Snodgrass, G.J.; Iles, R.A. Changes in the choline content of human breast milk in the first 3 weeks after birth. Eur. J. Pediatr. 2000, 159, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Ilcol, Y.O.; Ozbek, R.; Hamurtekin, E.; Ulus, I.H. Choline status in newborns, infants, children, breast-feeding women, breast-fed infants and human breast milk. J. Nutr. Biochem. 2005, 16, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T.; Furukawa, M.; Asoh, M.; Kanno, T.; Kojima, T.; Yonekubo, A. Fat-soluble and water-soluble vitamin contents of breast milk from Japanese women. J. Nutr. Sci. Vitaminol. (Tokyo) 2005, 51, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Hanok, J.Y.S.; Chung, Y.-J. Choline and betaine concentrations in breast milk of Korean lactating women and the choline and betaine intakes of their infants. Korean J. Nutr. 2010, 43, 588–596. [Google Scholar]
- Fischer, L.M.; da Costa, K.A.; Galanko, J.; Sha, W.; Stephenson, B.; Vick, J.; Zeisel, S.H. Choline intake and genetic polymorphisms influence choline metabolite concentrations in human breast milk and plasma. Am. J. Clin. Nutr. 2010, 92, 336–346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davenport, C.; Yan, J.; Taesuwan, S.; Shields, K.; West, A.A.; Jiang, X.; Perry, C.A.; Malysheva, O.V.; Stabler, S.P.; Allen, R.H.; et al. Choline intakes exceeding recommendations during human lactation improve breast milk choline content by increasing pemt pathway metabolites. J. Nutr. Biochem. 2015, 26, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Holmes-McNary, M.Q.; Cheng, W.L.; Mar, M.H.; Fussell, S.; Zeisel, S.H. Choline and choline esters in human and rat milk and in infant formulas. Am. J. Clin. Nutr. 1996, 64, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Bitman, J.; Wood, D.L.; Mehta, N.R.; Hamosh, P.; Hamosh, M. Comparison of the phospholipid composition of breast milk from mothers of term and preterm infants during lactation. Am. J. Clin. Nutr. 1984, 40, 1103–1119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patton, S.; Keenan, T.W. The milk fat globule membrane. Biochim. Biophys. Acta. 1975, 415, 273–309. [Google Scholar] [CrossRef]
- Chao, C.K.; Pomfret, E.A.; Zeisel, S.H. Uptake of choline by rat mammary-gland epithelial cells. Biochem. J. 1988, 254, 33–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, E.K.; Blusztajn, J.K.; Pomfret, E.A.; Zeisel, S.H. Rat and human mammary tissue can synthesize choline moiety via the methylation of phosphatidylethanolamine. Biochem. J. 1988, 256, 821–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmes, H.C.; Snodgrass, G.J.; Iles, R.A. The choline content of human breast milk expressed during the first few weeks of lactation. Biochem. Soc. Trans. 1996, 24, 350S. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Char, D.; Sheard, N.F. Choline, phosphatidylcholine and sphingomyelin in human and bovine milk and infant formulas. J. Nutr. 1986, 116, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Cheatham, C.L.; Sheppard, K.W. Synergistic effects of human milk nutrients in the support of infant recognition memory: An observational study. Nutrients 2015, 7, 9079–9095. [Google Scholar] [CrossRef] [PubMed]
- Ozarda, Y.; Cansev, M.; Ulus, I.H. Breast milk choline contents are associated with inflammatory status of breastfeeding women. J. Hum. Lact. 2014, 30, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Domellof, M.; Zivkovic, A.M.; Larsson, G.; Ohman, A.; Nording, M.L. NMR-based metabolite profiling of human milk: A pilot study of methods for investigating compositional changes during lactation. Biochem. Biophys. Res. Commun. 2016, 469, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Strecker, A. Notiz über die zusammensetzung des leucins. Liebigs Ann. Chem. 1842, 72, 89–91. [Google Scholar] [CrossRef]
- Allen, L.H. B vitamins in breast milk: Relative importance of maternal status and intake, and effects on infant status and function. Adv. Nutr. 2012, 3, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Stanbury, J.B.; Wurtman, R.J.; Brigida, M.; Fierro-Benitez, R. Choline content of mothers’ milk in Ecuador and Boston. N. Engl. J. Med. 1982, 306, 175–176. [Google Scholar] [PubMed]
- Food and Agriculture Organization of the United Nations, Statistical Division. FAOSTAT Statistics Database. 2013. Available online: www.fao.org/faostat/en/#compare (accessed on 20 August 2017).
- McDonald, C.M.; McLean, J.; Kroeun, H.; Talukder, A.; Lynd, L.D.; Green, T.J. Household food insecurity and dietary diversity as correlates of maternal and child undernutrition in rural Cambodia. Eur. J. Clin. Nutr. 2015, 69, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Mark, H.E.; Houghton, L.A.; Gibson, R.S.; Monterrosa, E.; Kraemer, K. Estimating dietary micronutrient supply and the prevalence of inadequate intakes from national food balance sheets in the South Asia regiona. Asia Pac. J. Clin. Nutr. 2016, 25, 368–376. [Google Scholar] [PubMed]
- Wiedeman, A.M.; Whitfield, K.C.; March, K.M.; Chen, N.N.; Kroeun, H.; Sokhoing, L.; Sophonneary, P.; Dyer, R.A.; Xu, Z.; Kitts, D.D.; et al. Concentrations of water-soluble forms of choline in human milk from lactating women in Canada and Cambodia. Nutrients 2018, 10, 381. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.M.; Aiello, M.O.; Fujita, M.; Hinde, K.; Milligan, L.; Quinn, E.A. Field and laboratory methods in human milk research. Am. J. Hum. Biol. 2013, 25, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Koc, H.; Mar, M.H.; Ranasinghe, A.; Swenberg, J.A.; Zeisel, S.H. Quantitation of choline and its metabolites in tissues and foods by liquid chromatography/electrospray ionization-isotope dilution mass spectrometry. Anal. Chem. 2002, 74, 4734–4740. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.M. Analytical approaches to determination of total choline in foods and dietary supplements. Anal. Bioanal. Chem. 2012, 403, 2103–2112. [Google Scholar] [CrossRef] [PubMed]
- Hampel, D.; Allen, L.H. Analyzing B-vitamins in human milk: Methodological approaches. Crit. Rev. Food Sci. Nutr. 2016, 56, 494–511. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization and United Nations International Children’s Emergency. Acceptable Medical Reasons for Use of Breastmilk Substitutes; WHO: Geneva, Switzerland, 2008. [Google Scholar]
- Koletzko, B.; Baker, S.; Cleghorn, G.; Neto, U.F.; Gopalan, S.; Hernell, O.; Hock, Q.S.; Jirapinyo, P.; Lonnerdal, B.; Pencharz, P.; et al. Global standard for the composition of infant formula: Recommendations of an ESPGHAN coordinated international expert group. J. Pediatr. Gastroenterol. Nutr. 2005, 41, 584–599. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius Commission. CODEX STAN 72-1981 for Infant Formula and Formulas for Special Medical Purposes Intended for Infants; Codex Alimentarius: Geneva, Switzerland, 2007. [Google Scholar]
- Fu, S.; Tao, B.; Lai, S.; Zhang, J.; Ren, Y.P. Determination of total choline by liquid chromatography-electrospray ionization-tandem mass spectrometry in infant formulas. AOAC Int. 2012, 95, 157–162. [Google Scholar] [CrossRef]
- Pardini, R.S.; Sapien, R.E. Trimethylaminuria (fish odor syndrome) related to the choline concentration of infant formula. Pediatr. Emerg. Care 2003, 19, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Thompson, J.J.; Jacobs, W.A.; Salvati, L.M. Determination of free and total carnitine and choline in infant formulas and adult nutritional products by UPLC/MS/MS: Single-laboratory validation, first action 2014.04. AOAC Int. 2015, 98, 1395–1406. [Google Scholar] [CrossRef] [PubMed]
- United Stated Department of Agriculture. Database for the Choline Content of Common Foods, Release 1; Agricultural Research Service: Washington, DC, USA, 2004.
- Chern, M.K.; Gage, D.A.; Pietruszko, R. Betaine aldehyde, betaine, and choline levels in rat livers during ethanol metabolism. Biochem. Pharmacol. 2000, 60, 1629–1637. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Mar, M.H.; Howe, J.C.; Holden, J.M. Concentrations of choline-containing compounds and betaine in common foods. J. Nutr. 2003, 133, 1302–1307. [Google Scholar] [CrossRef] [PubMed]
- Dilger, R.N.; Garrow, T.A.; Baker, D.H. Betaine can partially spare choline in chicks but only when added to diets containing a minimal level of choline. J. Nutr. 2007, 137, 2224–2228. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Kosik, S.J.; Zhao, Y.Y.; Jacobs, R.L.; Curtis, J.M.; Field, C.J. Total choline and choline-containing moieties of commercially available pulses. Plant Foods Hum. Nutr. 2014, 69, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Zhao, Y.Y.; Richard, C.; Bruce, H.L.; Jacobs, R.L.; Field, C.J.; Curtis, J.M. Measurement of the abundance of choline and the distribution of choline-containing moieties in meat. Int. J. Food Sci. Nutr. 2015, 66, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Hofvander, Y.; Hagman, U.; Hillervik, C.; Sjolin, S. The amount of milk consumed by 1–3 months old breast- or bottle-fed infants. Acta Paediatr. Scand. 1982, 71, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Butte, N.F.; Garza, C.; Smith, E.O.; Nichols, B.L. Human milk intake and growth in exclusively breast-fed infants. J. Pediatr. 1984, 104, 187–195. [Google Scholar] [CrossRef]
- Widdowson, E.M.; McCance, R.A. The effect of finite periods of undernutrition at different ages on the composition and subsequent development of the rat. Proc. R. Soc. Lond. B Biol. Sci. 1963, 158, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Welsch, F. Studies on accumulation and metabolic fate of (N-Me3H)choline in human term placenta fragments. Biochem. Pharmacol. 1976, 25, 1021–1030. [Google Scholar] [CrossRef]
- Pomfret, E.A.; daCosta, K.A.; Schurman, L.L.; Zeisel, S.H. Measurement of choline and choline metabolite concentrations using high-pressure liquid chromatography and gas chromatography-mass spectrometry. Anal. Biochem. 1989, 180, 85–90. [Google Scholar] [CrossRef]
- Pauwels, S.; Dopere, I.; Huybrechts, I.; Godderis, L.; Koppen, G.; Vansant, G. Reproducibility and validity of an FFQ to assess usual intake of methyl-group donors. Public Health Nutr. 2015, 18, 2530–2539. [Google Scholar] [CrossRef] [PubMed]
- Vennemann, F.B.; Ioannidou, S.; Valsta, L.M.; Dumas, C.; Ocke, M.C.; Mensink, G.B.; Lindtner, O.; Virtanen, S.M.; Tlustos, C.; D’Addezio, L.; et al. Dietary intake and food sources of choline in European populations. Br. J. Nutr. 2015, 114, 2046–2055. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.M.; daCosta, K.A.; Kwock, L.; Stewart, P.W.; Lu, T.S.; Stabler, S.P.; Allen, R.H.; Zeisel, S.H. Sex and menopausal status influence human dietary requirements for the nutrient choline. Am. J. Clin. Nutr. 2007, 85, 1275–1285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyd, W.D.; Graham-White, J.; Blackwood, G.; Glen, I.; McQueen, J. Clinical effects of choline in Alzheimer senile dementia. Lancet 1977, 2, 711. [Google Scholar] [CrossRef]
- Fischer, L.M.; da Costa, K.A.; Kwock, L.; Galanko, J.; Zeisel, S.H. Dietary choline requirements of women: Effects of estrogen and genetic variation. Am. J. Clin. Nutr. 2010, 92, 1113–1119. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, K.A.; Kozyreva, O.G.; Song, J.; Galanko, J.A.; Fischer, L.M.; Zeisel, S.H. Common genetic polymorphisms affect the human requirement for the nutrient choline. FASEB J. 2006, 20, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Veenema, K.; Solis, C.; Li, R.; Wang, W.; Maletz, C.V.; Abratte, C.M.; Caudill, M.A. Adequate intake levels of choline are sufficient for preventing elevations in serum markers of liver dysfunction in Mexican American men but are not optimal for minimizing plasma total homocysteine increases after a methionine load. Am. J. Clin. Nutr. 2008, 88, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Kroke, A.; Klipstein-Grobusch, K.; Voss, S.; Moseneder, J.; Thielecke, F.; Noack, R.; Boeing, H. Validation of a self-administered food-frequency questionnaire administered in the European prospective investigation into cancer and nutrition (EPIC) study: Comparison of energy, protein, and macronutrient intakes estimated with the doubly labeled water, urinary nitrogen, and repeated 24-h dietary recall methods. Am. J. Clin. Nutr. 1999, 70, 439–447. [Google Scholar] [PubMed]
- Yonemori, K.M.; Lim, U.; Koga, K.R.; Wilkens, L.R.; Au, D.; Boushey, C.J.; Le Marchand, L.; Kolonel, L.N.; Murphy, S.P. Dietary choline and betaine intakes vary in an adult multiethnic population. J. Nutr. 2013, 143, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Thompson, F.E.; Byers, T. Dietary assessment resource manual. J. Nutr. 1994, 124, 2245S–2317S. [Google Scholar] [PubMed]
- Cade, J.; Thompson, R.; Burley, V.; Warm, D. Development, validation and utilisation of food-frequency questionnaires—A review. Public Health Nutr. 2002, 5, 567–587. [Google Scholar] [CrossRef] [PubMed]
- Bingham, S.A.; Gill, C.; Welch, A.; Day, K.; Cassidy, A.; Khaw, K.T.; Sneyd, M.J.; Key, T.J.; Roe, L.; Day, N.E. Comparison of dietary assessment methods in nutritional epidemiology: Weighed records v. 24 h recalls, food-frequency questionnaires and estimated-diet records. Br. J. Nutr. 1994, 72, 619–643. [Google Scholar] [CrossRef] [PubMed]
- Block, G. A review of validations of dietary assessment methods. Am. J. Epidemiol. 1982, 115, 492–505. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.L.; Margetts, B.M. Comparison of a food frequency questionnaire with a 10-day weighed record in cigarette smokers. Int. J. Epidemiol. 1993, 22, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Schatzkin, A.; Kipnis, V.; Carroll, R.J.; Midthune, D.; Subar, A.F.; Bingham, S.; Schoeller, D.A.; Troiano, R.P.; Freedman, L.S. A comparison of a food frequency questionnaire with a 24-h recall for use in an epidemiological cohort study: Results from the biomarker-based observing protein and energy nutrition (OPEN) study. Int. J. Epidemiol. 2003, 32, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Basiotis, P.P.; Welsh, S.O.; Cronin, F.J.; Kelsay, J.L.; Mertz, W. Number of days of food intake records required to estimate individual and group nutrient intakes with defined confidence. J. Nutr. 1987, 117, 1638–1641. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Olendzki, B.C.; Pagoto, S.L.; Hurley, T.G.; Magner, R.P.; Ockene, I.S.; Schneider, K.L.; Merriam, P.A.; Hebert, J.R. Number of 24-h diet recalls needed to estimate energy intake. Ann. Epidemiol. 2009, 19, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Lavery, A.M.; Brender, J.D.; Zhao, H.; Sweeney, A.; Felkner, M.; Suarez, L.; Canfield, M.A. Dietary intake of choline and neural tube defects in Mexican Americans. Birth Defects Res. A Clin. Mol. Teratol. 2014, 100, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.F.; Luo, W.P.; Lin, F.Y.; Lian, Z.Q.; Mo, X.F.; Yan, B.; Xu, M.; Huang, W.Q.; Huang, J.; Zhang, C.X. Dietary choline and betaine intake, choline-metabolising genetic polymorphisms and breast cancer risk: A case-control study in china. Br. J. Nutr. 2016, 116, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Bidulescu, A.; Chambless, L.E.; Siega-Riz, A.M.; Zeisel, S.H.; Heiss, G. Usual choline and betaine dietary intake and incident coronary heart disease: The atherosclerosis risk in communities (ARIC) study. BMC Cardiovasc. Disord. 2007, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Weighted kappa: Nominal scale agreement with provision for scaled disagreement or partial credit. Psychol. Bull. 1968, 70, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Beaton, G.H.; Milner, J.; Corey, P.; McGuire, V.; Cousins, M.; Stewart, E.; de Ramos, M.; Hewitt, D.; Grambsch, P.V.; Kassim, N.; et al. Sources of variance in 24-h dietary recall data: Implications for nutrition study design and interpretation. Am. J. Clin. Nutr. 1979, 32, 2546–2559. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sampson, L.; Stampfer, M.J.; Rosner, B.; Bain, C.; Witschi, J.; Hennekens, C.H.; Speizer, F.E. Reproducibility and validity of a semiquantitative food frequency questionnaire. Am. J. Epidemiol. 1985, 122, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999, 8, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Willett, W. Nutritional Epidemiology, 3rd ed.; Oxford University Press: Cary, NC, USA, 2013. [Google Scholar]
- Cho, E.; Zeisel, S.H.; Jacques, P.; Selhub, J.; Dougherty, L.; Colditz, G.A.; Willett, W.C. Dietary choline and betaine assessed by food-frequency questionnaire in relation to plasma total homocysteine concentration in the Framingham offspring study. Am. J. Clin. Nutr. 2006, 83, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Brunst, K.J.; Kannan, S.; Ni, Y.M.; Gennings, C.; Ganguri, H.B.; Wright, R.J. Validation of a food frequency questionnaire for estimating micronutrient intakes in an urban US sample of multi-ethnic pregnant women. Matern Child Health J. 2016, 20, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Coathup, V.; Wheeler, S.; Smith, L. A method comparison of a food frequency questionnaire to measure folate, choline, betaine, vitamin c and carotenoids with 24-h dietary recalls in women of reproductive age. Eur. J. Clin. Nutr. 2016, 70, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Fayet, F.; Flood, V.; Petocz, P.; Samman, S. Relative and biomarker-based validity of a food frequency questionnaire that measures the intakes of vitamin B(12), folate, iron, and zinc in young women. Nutr. Res. 2011, 31, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K.A.; Byrne, N.M.; Magarey, A.M.; Hills, A.P. Minimizing random error in dietary intakes assessed by 24-h recall, in overweight and obese adults. Eur. J. Clin. Nutr. 2008, 62, 537–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1228S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, L.M.; Scearce, J.A.; Mar, M.H.; Patel, J.R.; Blanchard, R.T.; Macintosh, B.A.; Busby, M.G.; Zeisel, S.H. Ad libitum choline intake in healthy individuals meets or exceeds the proposed adequate intake level. J. Nutr. 2005, 135, 826–829. [Google Scholar] [CrossRef] [PubMed]
- Chester, D.N.; Goldman, J.D.; Ahuja, J.K.; Moshfegh, A. Dietary Intakes of Choline: What We Eat in America, NHANES 2007–2008, 2011. Food Surveys Research Group Web Site. Available online: www.ars.usda.gov/Services/docs.html?docid=19476 (accessed on 18 September 2017).
- Wallace, T.C.; Fulgoni, V.L., 3rd. Assessment of total choline intakes in the United States. J. Am. Coll. Nutr. 2016, 35, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Fulgoni, V.L. Usual choline intakes are associated with egg and protein food consumption in the United States. Nutrients 2017, 9, 839. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wang, Y.; Randell, E.; Pedram, P.; Yi, Y.; Gulliver, W.; Sun, G. Higher dietary choline and betaine intakes are associated with better body composition in the adult population of Newfoundland, Canada. PLoS ONE 2016, 11, e0155403. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Carrillo, L.; Gamboa-Loira, B.; Becerra, W.; Hernandez-Alcaraz, C.; Hernandez-Ramirez, R.U.; Gandolfi, A.J.; Franco-Marina, F.; Cebrian, M.E. Dietary micronutrient intake and its relationship with arsenic metabolism in Mexican women. Environ. Res. 2016, 151, 445–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.P.; Chen, C.H.; Kuo, C.S.; Kuo, H.T.; Huang, K.T.; Shen, Y.L.; Chang, C.H.; Huang, R.F.S. Dietary choline and folate relationships with serum hepatic inflammatory injury markers in Taiwanese adults. Asia Pac. J. Clin. Nutr. 2017, 26, 642–649. [Google Scholar] [PubMed]
- Institute of Medicine. Dietary Reference Intakes: Applications in Dietary Assessment; The National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Nagata, C.; Wada, K.; Tamura, T.; Konishi, K.; Kawachi, T.; Tsuji, M.; Nakamura, K. Choline and betaine intakes are not associated with cardiovascular disease mortality risk in Japanese men and women. J. Nutr. 2015, 145, 1787–1792. [Google Scholar] [CrossRef] [PubMed]
- Mygind, V.L.; Evans, S.E.; Peddie, M.C.; Miller, J.C.; Houghton, L.A. Estimation of usual intake and food sources of choline and betaine in New Zealand reproductive age women. Asia Pac. J. Clin. Nutr. 2013, 22, 319–324. [Google Scholar] [PubMed]
- Chiuve, S.E.; Giovannucci, E.L.; Hankinson, S.E.; Zeisel, S.H.; Dougherty, L.W.; Willett, W.C.; Rimm, E.B. The association between betaine and choline intakes and the plasma concentrations of homocysteine in women. Am. J. Clin. Nutr. 2007, 86, 1073–1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, E.; Willett, W.C.; Colditz, G.A.; Fuchs, C.S.; Wu, K.; Chan, A.T.; Zeisel, S.H.; Giovannucci, E.L. Dietary choline and betaine and the risk of distal colorectal adenoma in women. J. Natl. Cancer Inst. 2007, 99, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Gammon, M.D.; Zeisel, S.H.; Bradshaw, P.T.; Wetmur, J.G.; Teitelbaum, S.L.; Neugut, A.I.; Santella, R.M.; Chen, J. High intakes of choline and betaine reduce breast cancer mortality in a population-based study. FASEB J. 2009, 23, 4022–4028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.E.; Giovannucci, E.; Fuchs, C.S.; Willett, W.C.; Zeisel, S.H.; Cho, E. Choline and betaine intake and the risk of colorectal cancer in men. Cancer Epidemiol. Biomarkers Prev. 2010, 19, 884–887. [Google Scholar] [CrossRef] [PubMed]
- Richman, E.L.; Kenfield, S.A.; Stampfer, M.J.; Giovannucci, E.L.; Zeisel, S.H.; Willett, W.C.; Chan, J.M. Choline intake and risk of lethal prostate cancer: Incidence and survival. Am. J. Clin. Nutr. 2012, 96, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Lewis, E.D.; Subhan, F.B.; Bell, R.C.; McCargar, L.J.; Curtis, J.M.; Jacobs, R.L.; Field, C.J.; APrON Team. Estimation of choline intake from 24 h dietary intake recalls and contribution of egg and milk consumption to intake among pregnant and lactating women in Alberta. Br. J. Nutr. 2014, 112, 112–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunst, K.J.; Wright, R.O.; DiGioia, K.; Enlow, M.B.; Fernandez, H.; Wright, R.J.; Kannan, S. Racial/ethnic and sociodemographic factors associated with micronutrient intakes and inadequacies among pregnant women in an urban US population. Public Health Nutr. 2014, 17, 1960–1970. [Google Scholar] [CrossRef] [PubMed]
- Masih, S.P.; Plumptre, L.; Ly, A.; Berger, H.; Lausman, A.Y.; Croxford, R.; Kim, Y.I.; O’Connor, D.L. Pregnant Canadian women achieve recommended intakes of one-carbon nutrients through prenatal supplementation but the supplement composition, including choline, requires reconsideration. J. Nutr. 2015, 145, 1824–1834. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, A.M.; Szwengiel, A.; Chmurzynska, A. Dietary, anthropometric, and biochemical factors influencing plasma choline, carnitine, trimethylamine, and trimethylamine-N-oxide concentrations. Int. J. Food Sci. Nutr. 2017, 68, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, S.; Ghosh, M.; Duca, R.C.; Bekaert, B.; Freson, K.; Huybrechts, I.; Langie, S.A.S.; Koppen, G.; Devlieger, R.; Godderis, L. Maternal intake of methyl-group donors affects DNA methylation of metabolic genes in infants. Clin. Epigenetics 2017, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Bar, H.Y.; Yan, J.; Jones, S.; Brannon, P.M.; West, A.A.; Perry, C.A.; Ganti, A.; Pressman, E.; Devapatla, S.; et al. A higher maternal choline intake among third-trimester pregnant women lowers placental and circulating concentrations of the antiangiogenic factor fms-like tyrosine kinase-1 (sflt1). FASEB J. 2013, 27, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- American Medical Association. Busines of the American Medical Association House of Delegates Annual Meeting. 2017. Available online: https://www.Ama-assn.Org/aboutus/business-ama-house-delegates-2017-annualmeeting#annotated%20reference%20committee%20reports (accessed on 18 September 2017).
- Wu, B.T. Choline Nutrition, Choline Status, and Developmental Outcome in Early Childhood. Master’s Thesis, The University of British Columbia, Vancouver, BC, Canada, 2014. [Google Scholar]
- Reinhard, P.C.; Lotrean, L.M. Choline intake and its food sources in the diet of Romanian kindergarten children. Nutrients 2017, 9, 896. [Google Scholar]
- Al-Daghri, N.M.; Al-Othman, A.; Alkharfy, K.M.; Alokail, M.S.; Khan, N.; Alfawaz, H.A.; Aiswaidan, I.A.; Chrousos, G.P. Assessment of selected nutrient intake and adipocytokine profile among Saudi children and adults. Endocr. J. 2012, 59, 1057–1063. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goon, S.; Dey, S.R. A 24-h dietary recall for assessing the intake pattern of choline among Bangladeshi pregnant women at their third trimester of pregnancy. Cent. Asian J. Glob. Health 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Gossell-Williams, M.; Fletcher, H.; McFarlane-Anderson, N.; Jacob, A.; Patel, J.; Zeisel, S. Dietary intake of choline and plasma choline concentrations in pregnant women in Jamaica. West Indian Med. J. 2005, 54, 355–359. [Google Scholar] [CrossRef] [PubMed]
| Country | n | Betaine (µmol/L) | Choline (µmol/L) 1 | TC (mg/L) | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| FC | PCho | GPC | PC | SM | TC | |||||
| US | 10 | - | 85 | - | - | 180 | 206 | - | - | [127] |
| US | 16 | - | 116 | 570 | 362 | 82 | 124 | 1254 | 130 | [121] |
| US | 48 | 7 | 83 | 553 | 388 | 107 | 67 | 1198 | 125 | [119] |
| US | 60 | - | 158 | - | - | - | - | - | - | [128] |
| US | 28 | 3.8 | 84 | 500 | 403 | 63 | 175 | 1225 | 128 | [120] |
| Turkey | 12 | - | 286 | 438 | 465 | 155 | 97 | 1441 2 | 150 | [116] |
| Turkey 3 | 54 | - | 93 | 351 | 958 | - | - | 1532 | 159 | [129] |
| Japan | 62 | - | - | - | - | - | - | 950 | 99 | [117] |
| Korea | 36 | 31 | 283 | - | - | - | - | 1600 | 166 | [118] |
| Sweden | 1 | - | 188 | 704 | 672 | - | - | - | - | [130] |
| Canada | 301 | 4.8 | 155 | 535 | 416 | - | - | - | - | [131] |
| Cambodia | 67 | 5.1 | 143 | 562 | 390 | - | - | - | - | [131] |
| Food Item | Betaine | Choline | |||||
|---|---|---|---|---|---|---|---|
| FC | PCho | GPC | PC | SM | TC | ||
| Beef liver, cooked | 5.6 | 62.0 | 12.0 | 83.0 | 250.0 | 24.0 | 431.0 1 |
| Egg, hard boiled | 0.6 | 0.7 | 0.5 | 0.5 | 210.0 | 14.0 | 225.7 1 |
| Beef steak, cooked | 13.0 | 0.7 | 1.3 | 5.2 | 86.0 | 11.0 | 104.2 1 |
| Salmon, cooked | 1.8 | 7.8 | 1.2 | 41.0 | 37.0 | 3.4 | 90.4 1 |
| Pork chops, cooked | 2.8 | 1.1 | 0.6 | 12.0 | 57.0 | 7.5 | 78.2 |
| Chicken breast, cooked | 6.4 | 3.2 | 2.1 | 1.6 | 46.0 | 8.9 | 61.8 |
| Nuts, almonds | 0.5 | 9.4 | 1.9 | 1.2 | 40.0 | 0.0 | 52.5 1 |
| Broccoli, cooked | 0.1 | 8.5 | 9.3 | 1.3 | 21.0 | 0.0 | 40.1 |
| Beans, baked canned | 0.1 | 17.0 | 0.8 | 1.3 | 12.0 | 0.0 | 31.1 1 |
| Milk, 2% fat | 0.9 | 2.8 | 1.6 | 10.0 | 1.2 | 0.9 | 16.5 1 |
| Red potato, cooked | 0.2 | 8.5 | 1.2 | 3.8 | 5.3 | 0.0 | 18.8 |
| White rice, cooked | 0.3 | 0.7 | 0.0 | 1.0 | 0.4 | 0.0 | 2.1 |
| Stage | IOM—1998 1 | EFSA—2016 2 | ||||
|---|---|---|---|---|---|---|
| Age | AI (mg/day) | UL (mg/day) | Age | AI (mg/day) | ||
| Males | Females | |||||
| Infants | 0–6 month | 125 | 125 | - | 0–6 month | 120 |
| 7–12 month | 150 | 150 | - | 7–11 month | 160 | |
| Children | 1–3 year | 200 | 200 | 1000 | 1–3 year | 140 |
| 4–8 year | 250 | 250 | 1000 | 4–6 year | 170 | |
| 9–13 year | 375 | 375 | 2000 | 7–10 year | 250 | |
| 14–18 year | 550 | 400 | 3000 | 11–14 year | 340 | |
| 15–17 year | 400 | |||||
| Adults | ≥19 year | 550 | 425 | 3500 | ≥18 year | 400 |
| Pregnancy | - | - | 450 | 3000 | - | 480 |
| Lactation | - | - | 550 | 3500 | - | 520 |
| Country | Dietary Method | Total Choline Intake (mg/day) 1 | Reference | |||
|---|---|---|---|---|---|---|
| Men | Women | |||||
| n | Intake | n | Intake | |||
| Canada | FFQ | 822 | 372 ± 287 | 2232 | 292 ± 213 | [195] |
| China | FFQ | 18,763 | 318 ± 92 | 37,432 | 289 ± 85 | [56] |
| Finland 4 | 48HR ×2 | 585 | 450 (425) 2 | 710 | 344 (327) 2 | [160] |
| France 4 | DR ×7 | 936 | 370 (362) 2 | 1340 | 291 (283) 2 | [160] |
| Greece | FFQ | 1514 | 291 ± 79 | 1528 | 285 ± 75 | [50] |
| Japan | FFQ | 13,355 | 445–513 3 | 15,724 | 388–442 3 | [199] |
| Italy 4 | DR ×3 | 1068 | 357 (341) 2 | 1245 | 293 (282) 2 | [160] |
| Ireland 4 | DR ×4 | 634 | 461 (443) 2 | 640 | 318 (314) 2 | [160] |
| Mexico | FFQ | - | - | 1027 | 263 ± 105 | [196] |
| New Zealand | WFR ×3 | - | - | 125 | 316 ± 65 | [200] |
| Sweden 4 | DR ×4 | 623 | 468 (442) 2 | 807 | 374 (356) 2 | [160] |
| Taiwan | FFQ | 321 | 284 ± 145 | 227 | 230 ± 120 | [197] |
| The Netherlands 4 | 24HR ×2 | 1023 | 448 (425) 2 | 1034 | 334 (317) 2 | [160] |
| UK 4 | DR ×4 | 560 | 407 (385) 2 | 706 | 294 (282) 2 | [160] |
| US 4 | 24HR ×2 | 2563 | 421 (408) 2 | 2704 | 279 (271) 2 | [194] |
| Age Group and Country | Dietary Method | Subgroup | Total Choline Intake (mg/day) 1 | Reference | |
|---|---|---|---|---|---|
| n | Intake | ||||
| Toddlers (1–3 year) | |||||
| Canada | DR ×3 | 1 year | 110 | 174 ± 56 | [74] |
| Finland 5 | DR ×3 | 1–3 year | 500 | 176 (172) 2,4 | [160] |
| US 5 | 24HR ×2 | 2–3 year | 1316 | 224 (217) 2 | [194] |
| Children (4–9 year) | |||||
| Canada | FFQ | 6 year | 193 | 302 ± 100 | [213] |
| Germany 5 | DR ×3 | 6–10 year | 835 | 288 (276) 2,4 | [160] |
| Romania | DR ×3 | 4–6 year | 71 | 215 ± 32 | [214] |
| US 5 | 24HR ×2 | 4–8 year | 2774 | 243 (235) 2 | [194] |
| Adolescents (10–18 year) | |||||
| Bangladesh | FFQ | Boys and girls | 47 | 128 ± 3.2 3 | [215] |
| The Netherlands | 24HR ×2 | 10–18 year, boys | 566 | 353 (338) 2 | [160] |
| 10–18 year, girls | 576 | 291 (279) 2 | |||
| US 5 | 24HR ×2 | 14–18 year, boys | 1207 | 295 (288) 2 | [194] |
| 14–18 year, girls | 1147 | 244 (237) 2 | |||
| Pregnancy | |||||
| Bangladesh | 24HR ×1 | T3 | 103 | 190 ± 98 | [216] |
| Belgium | FFQ | T2 | 85 | 268 ± 7.4 3 | [210] |
| Canada | FFQ | T3 | 290 | 302 ± 122 | [208] |
| Jamaica | FFQ | T1 | 16 | 279 ± 116 | [217] |
| Latvia | 24HR ×2 | T1–T3 | 990 | 356 (330) 2 | [160] |
| US 5 | 24HR ×2 | T1–T3 | 593 | 319 (309) 2 | [194] |
| Lactation | |||||
| Belgium | FFQ | 6 mpp | 60 | 268 ± 7.8 3 | [210] |
| Canada | 24HR ×1 | 3 mpp | 488 | 346 ± 151 | [206] |
| US | DR ×3 | 1.5 mpp | 98 | 356 ± 109 4 | [72] |
| Elderly (> 65 year) | |||||
| Italy 5 | DR ×3 | Men | 69 | 335 (320) 2 | [160] |
| Women | 159 | 269 (269) 2 | |||
| Poland | FFQ | Women | 122 | 392 ± 26 3 | [209] |
| US 5 | 24HR ×2 | Men | 1099 | 363 (351) 2 | [194] |
| Women | 1145 | 266 (259) 2 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary Choline Intake: Current State of Knowledge Across the Life Cycle. Nutrients 2018, 10, 1513. https://doi.org/10.3390/nu10101513
Wiedeman AM, Barr SI, Green TJ, Xu Z, Innis SM, Kitts DD. Dietary Choline Intake: Current State of Knowledge Across the Life Cycle. Nutrients. 2018; 10(10):1513. https://doi.org/10.3390/nu10101513
Chicago/Turabian StyleWiedeman, Alejandra M., Susan I. Barr, Timothy J. Green, Zhaoming Xu, Sheila M. Innis, and David D. Kitts. 2018. "Dietary Choline Intake: Current State of Knowledge Across the Life Cycle" Nutrients 10, no. 10: 1513. https://doi.org/10.3390/nu10101513





