Coffee Consumption and Risk of Dementia and Alzheimer’s Disease: A Dose-Response Meta-Analysis of Prospective Studies
Abstract
:1. Introduction
2. Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Assessment of Study Quality
2.5. Statistical Analysis
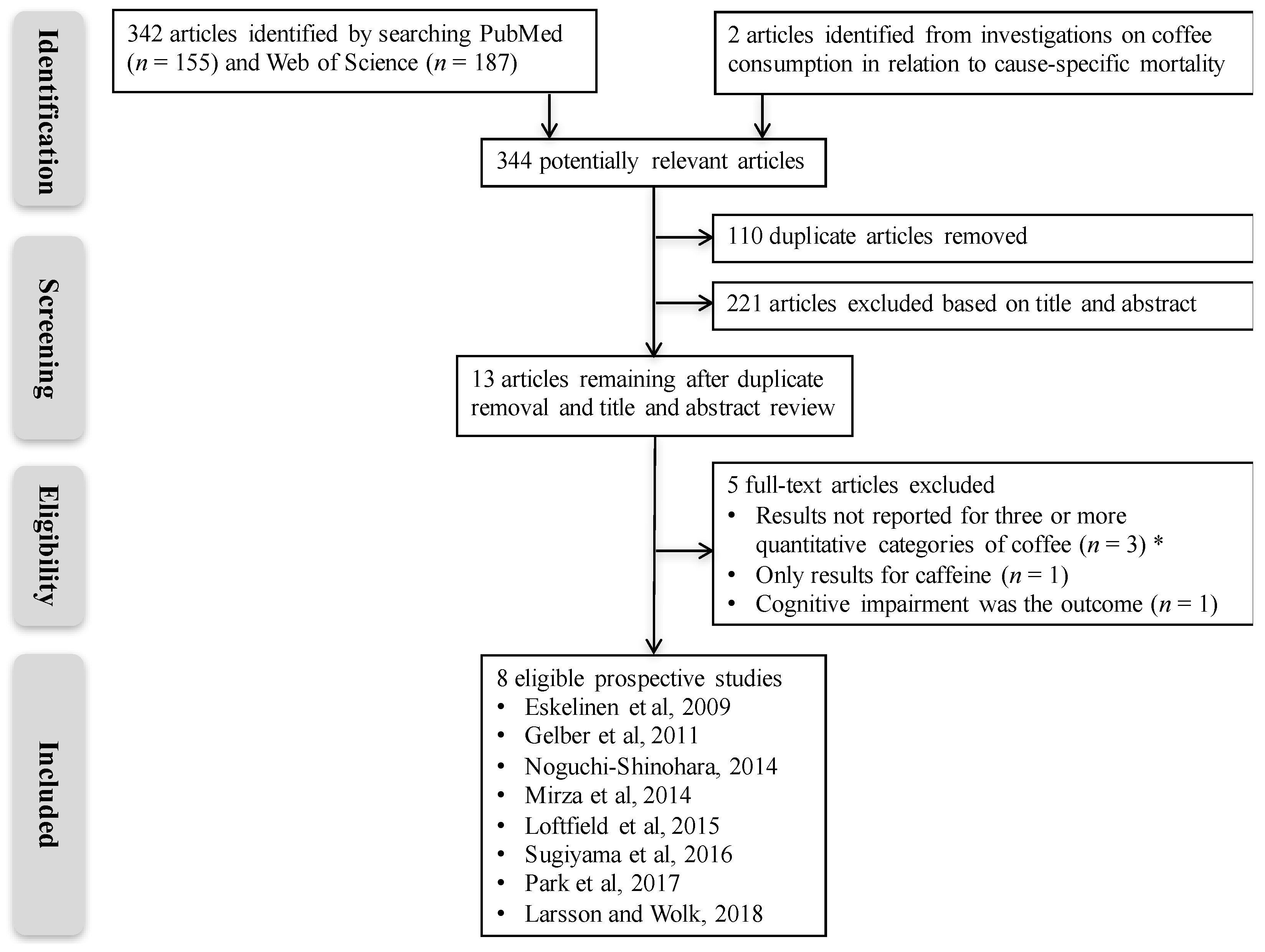
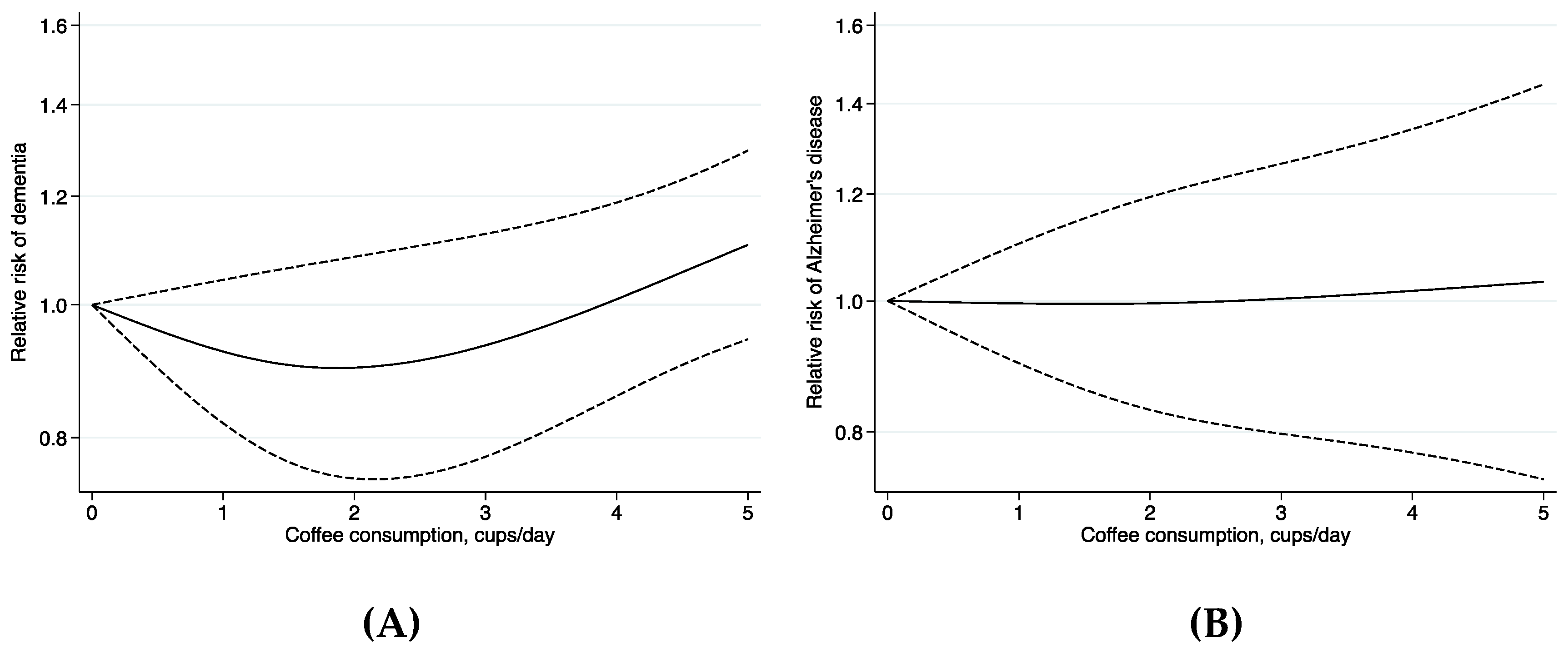
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Prince, M.; Wimo, A.; Guerchet, M.; Ali, G.C.; Wu, Y.T.; Prina, M. World Alzheimer Report 2015—The Global Impact of Dementia: An Analysis of Prevalence, Incidence, Cost and Trends; Alzheimer’s Disease International: London, UK, 2015. [Google Scholar]
- Alzheimer’s Association. Alzheimer’s association report: 2018 Alzheimer’s disease facts and figures. Alzheimers Dement. 2018, 14, 367–429. [Google Scholar] [CrossRef]
- Grosso, G.; Godos, J.; Galvano, F.; Giovannucci, E.L. Coffee, Caffeine, and Health Outcomes: An Umbrella Review. Annu. Rev. Nutr. 2017, 37, 131–156. [Google Scholar] [CrossRef] [PubMed]
- Poole, R.; Kennedy, O.J.; Roderick, P.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Coffee consumption and health: Umbrella review of meta-analyses of multiple health outcomes. BMJ 2017, 359, j5024. [Google Scholar] [CrossRef] [PubMed]
- Carlstrom, M.; Larsson, S.C. Coffee consumption and reduced risk of developing type 2 diabetes: A systematic review with meta-analysis. Nutr. Rev. 2018, 76, 395–417. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Carlstrom, M. Coffee consumption and gout: A Mendelian randomisation study. Ann. Rheum. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Orsini, N. Coffee consumption and risk of stroke: A dose-response meta-analysis of prospective studies. Am. J. Epidemiol. 2011, 174, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Fredholm, B.B.; Battig, K.; Holmen, J.; Nehlig, A.; Zvartau, E.E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar] [PubMed]
- Cao, C.; Cirrito, J.R.; Lin, X.; Wang, L.; Verges, D.K.; Dickson, A.; Mamcarz, M.; Zhang, C.; Mori, T.; Arendash, G.W.; et al. Caffeine suppresses amyloid–beta levels in plasma and brain of Alzheimer’s disease transgenic mice. J. Alzheimers Dis. 2009, 17, 681–697. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Wang, L.; Lin, X.; Mamcarz, M.; Zhang, C.; Bai, G.; Nong, J.; Sussman, S.; Arendash, G. Caffeine synergizes with another coffee component to increase plasma GCSF: Linkage to cognitive benefits in Alzheimer’s mice. J. Alzheimers Dis. 2011, 25, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Arendash, G.W.; Schleif, W.; Rezai-Zadeh, K.; Jackson, E.K.; Zacharia, L.C.; Cracchiolo, J.R.; Shippy, D.; Tan, J. Caffeine protects Alzheimer’s mice against cognitive impairment and reduces brain beta-amyloid production. Neuroscience 2006, 142, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Steger, R.; Kamal, A.; Lutchman, S.; Intrabartolo, L.; Sohail, R.; Brumberg, J.C. Chronic caffeine ingestion causes microglia activation, but not proliferation in the healthy brain. Brain Res. Bull. 2014, 106, 39–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Ghribi, O.; Geiger, J.D. Caffeine protects against disruptions of the blood-brain barrier in animal models of Alzheimer’s and Parkinson’s diseases. J. Alzheimers Dis. 2010, 20 (Suppl. 1), S127–S141. [Google Scholar] [CrossRef]
- Liu, Q.P.; Wu, Y.F.; Cheng, H.Y.; Xia, T.; Ding, H.; Wang, H.; Wang, Z.M.; Xu, Y. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta–analysis of prospective cohort studies. Nutrition 2016, 32, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Sun, D.; He, Y. Coffee intake and the incident risk of cognitive disorders: A dose-response meta-analysis of nine prospective cohort studies. Clin. Nutr. 2017, 36, 730–736. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Traylor, M.; Malik, R.; Dichgans, M.; Burgess, S.; Markus, H.S. Modifiable pathways in Alzheimer’s disease: Mendelian randomisation analysis. BMJ 2017, 359, j5375. [Google Scholar] [CrossRef] [PubMed]
- Loftfield, E.; Freedman, N.D.; Graubard, B.I.; Guertin, K.A.; Black, A.; Huang, W.Y.; Shebl, F.M.; Mayne, S.T.; Sinha, R. Association of Coffee Consumption With Overall and Cause-Specific Mortality in a Large US Prospective Cohort Study. Am. J. Epidemiol. 2015, 182, 1010–1022. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Freedman, N.D.; Haiman, C.A.; Le Marchand, L.; Wilkens, L.R.; Setiawan, V.W. Association of Coffee Consumption With Total and Cause-Specific Mortality Among Nonwhite Populations. Ann. Intern. Med. 2017, 167, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Wolk, A. The role of lifestyle factors and sleep duration for late-onset dementia: A cohort study. J. Alzheimers Dis. 2018, in press. [Google Scholar] [CrossRef]
- Tyas, S.L.; Manfreda, J.; Strain, L.A.; Montgomery, P.R. Risk factors for Alzheimer’s disease: A population-based, longitudinal study in Manitoba, Canada. Int. J. Epidemiol. 2001, 30, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.; Laurin, D.; Verreault, R.; Hebert, R.; Helliwell, B.; Hill, G.B.; McDowell, I. Risk factors for Alzheimer’s disease: A prospective analysis from the Canadian Study of Health and Aging. Am. J. Epidemiol. 2002, 156, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Fischer, K.; Melo van Lent, D.; Wolfsgruber, S.; Weinhold, L.; Kleineidam, L.; Bickel, H.; Scherer, M.; Eisele, M.; van den Bussche, H.; Wiese, B.; et al. Prospective Associations between Single Foods, Alzheimer’s Dementia and Memory Decline in the Elderly. Nutrients 2018, 10, 852. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Appl. Eng. Agric. 2014, 18, 727–734. [Google Scholar]
- Crippa, A.; Discacciati, A.; Bottai, M.; Spiegelman, D.; Orsini, N. One-stage dose-response meta-analysis for aggregated data. Stat. Methods Med. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Orsini, N.; Li, R.; Wolk, A.; Khudyakov, P.; Spiegelman, D. Meta-analysis for linear and nonlinear dose-response relations: Examples, an evaluation of approximations, and software. Am. J. Epidemiol. 2012, 175, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey, S.G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eskelinen, M.H.; Ngandu, T.; Tuomilehto, J.; Soininen, H.; Kivipelto, M. Midlife coffee and tea drinking and the risk of late-life dementia: A population-based CAIDE study. J. Alzheimers Dis. 2009, 16, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Gelber, R.P.; Petrovitch, H.; Masaki, K.H.; Ross, G.W.; White, L.R. Coffee intake in midlife and risk of dementia and its neuropathologic correlates. J. Alzheimers Dis. 2011, 23, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Noguchi-Shinohara, M.; Yuki, S.; Dohmoto, C.; Ikeda, Y.; Samuraki, M.; Iwasa, K.; Yokogawa, M.; Asai, K.; Komai, K.; Nakamura, H.; et al. Consumption of green tea, but not black tea or coffee, is associated with reduced risk of cognitive decline. PLoS ONE 2014, 9, e96013. [Google Scholar] [CrossRef] [PubMed]
- Mirza, S.S.; Tiemeier, H.; de Bruijn, R.F.; Hofman, A.; Franco, O.H.; Kiefte-de, J.J.; Koudstaal, P.J.; Ikram, M.A. Coffee consumption and incident dementia. Eur. J. Epidemiol. 2014, 29, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, K.; Tomata, Y.; Kaiho, Y.; Honkura, K.; Sugawara, Y.; Tsuji, I. Association between Coffee Consumption and Incident Risk of Disabling Dementia in Elderly Japanese: The Ohsaki Cohort 2006 Study. J. Alzheimers Dis. 2016, 50, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.; Taylor, A.E.; Karhunen, V.; Zhan, Y.; Rovio, S.P.; Lahti, J.; Sjogren, P.; Byberg, L.; Lyall, D.M.; Auvinen, J.; et al. Habitual coffee consumption and cognitive function: A Mendelian randomization meta-analysis in up to 415,530 participants. Sci. Rep. 2018, 8, 7526. [Google Scholar] [CrossRef] [PubMed]
| Author, Year (reference) | Study Name, Country | Ascertainment of AD and Dementia | Cases (Total Sample Size) | Age; % Men | Follow-up Time | Adjustment for Confounders | NOS Score * | Coffee Intake Category | RR (95% CI) of AD | RR (95% CI) of Dementia |
|---|---|---|---|---|---|---|---|---|---|---|
| Eskelinen et al., 2009 [28] | Cardiovascular Risk Factors, Aging and Dementia study, Finland | Neurological and clinical examination | 58 AD, 61 dementia (1409) | 65–79 years; 62 | 21 years (mean) | Age, sex, education, area of residence, smoking, BMI, systolic blood pressure, total cholesterol, ApoE ε4 status | 7 | 0–2 cups/day 3–5 cups/day >5 cups/day | Reference 0.42 (0.12–1.46) 1.01 (0.33–3.08) | Reference 0.30 (0.10–0.93) 0.83 (0.32–2.15) |
| Gelber et al., 2011 [29] | Honolulu-Asia Aging Study, United States (Hawaii) | Neurological and clinical examination | 118 AD, 226 dementia (3494) | 71–93 years; 100 | 25 years | Age, education, smoking, physical activity, elevated cholesterol, hypertension, ApoE ε4 status | 9 | 0 oz/day 4–8 oz/day 12–16 oz/day 20–24 oz/day ≥28 oz/day | Reference 0.89 (0.50–1.59) 1.09 (0.60–2.00) 0.95 (0.45–2.00) 0.59 (0.23–1.54) | Reference 0.93 (0.59–1.46) 1.24 (0.78–1.97) 1.14 (0.66–1.98) 1.09 (0.59–2.00) |
| Noguchi-Shinohara et al., 2014 [30] | Nakajima project, Japan | Neurological and clinical examination | 26 dementia (490) | >60 years; ~35 | 4.9 years (mean) | Age, sex, education, smoking, hypertension, diabetes, hyperlipidemia, ApoE ε4 status, physical activities and/or other hobbies, alcohol, tea | 7 | None 1–6 days/week Every day | NA | Reference 1.00 (0.34–2.99) 0.70 (0.22–2.17) |
| Mirza et al., 2014 [31] | Rotterdam Study, The Netherlands | Neurophysiological testing and linkage to medical records | 814 dementia (5408) | ≥55 years; 59 | 13.2 years (mean) | Age, sex, education, smoking, BMI, hypertension, diabetes, family history of dementia, working status, alcohol | 9 | 0–1 cup/day >1–3 cups/day >3 cups/day | NA | Reference 0.88 (0.67–1.16) 1.00 (0.76–1.30) |
| Loftfield et al., 2015 [17] | Prostate, Lung, Colorectal and Ovarian Screening Trial, United States | Linkage to the National Death Index | 93 AD deaths (90,317) | 55–74 years; NA | 12 years | Age, sex, education, marital status, race/ethnicity, smoking, BMI, diabetes, supplemental vitamin use, ibuprofen, aspirin, menopausal hormone therapy, and intake of alcohol, energy, red and processed meat, white meat, saturated fat, fruit, and vegetables | 7 | 0 cup/day <1 cup/day 1 cup/day 2–3 cups/day ≥4 cups/day | Reference 1.01 (0.53–1.95) 0.66 (0.32–1.36) 0.59 (0.31–1.11) 0.72 (0.33–1.58) | NA |
| Sugiyama et al., 2016 [32] | Ohsaki Cohort, Japan | Linkage to the Long-term Care Insurance database | 1107 dementia (13,137) | ≥65 years; ~45 | 5.7 years | Age, sex, education, smoking, BMI, walking duration, history of stroke, hypertension, myocardial infarction, diabetes, arthritis, osteoporosis, and fracture, psychological distress, participating in any community activities, alcohol, green tea | 7 | Never Occasionally 1–2 cups/day ≥3 cups/day | NA | Reference 0.73 (0.62–0.86) 0.72 (0.61–0.84) 0.82 (0.65–1.02) |
| Park et al., 2017 [18] | Multiethnic Cohort, United States | Linkage to the death register | 1404 AD deaths (185,855) | 45–75 years; ~45 | 16.2 years (mean) | Age, sex, ethnicity, education, smoking, preexisting illness, BMI, physical activity, and intake of alcohol, total energy, and energy from fat | 7 | None 1–3 cups/month 1–6 cups/week 1 cup/day 2–3 cups/day ≥4 cups/day | Reference 1.01 (0.72–1.41) 0.92 (0.69–1.24) 0.90 (0.71–1.14) 1.16 (0.90–1.49) 1.33 (0.86–2.04) | NA |
| Larsson and Wolk, 2018 [19] | Swedish Mammography Cohort and Cohort of Swedish Men, Sweden | Linkage to national patient register | 1299 AD, 3755 dementia (28,775) | 65–83 years; 53 | 12.6 years (mean) | Age, sex, education, smoking, BMI, exercise, walking or bicycling, history of hypertension, hypercholesterolemia, or diabetes, sleep duration, alcohol, DASH diet | 8 | <1 cup/day 1–2.9 cups/day 3–4.9 cups/day ≥5 cups/day | Reference 0.90 (0.70–1.17) 1.01 (0.78–1.30) 0.93 (0.70–1.24) | Reference 0.99 (0.85–1.16) 1.03 (0.88–1.21) 1.07 (0.90–1.28) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larsson, S.C.; Orsini, N. Coffee Consumption and Risk of Dementia and Alzheimer’s Disease: A Dose-Response Meta-Analysis of Prospective Studies. Nutrients 2018, 10, 1501. https://doi.org/10.3390/nu10101501
Larsson SC, Orsini N. Coffee Consumption and Risk of Dementia and Alzheimer’s Disease: A Dose-Response Meta-Analysis of Prospective Studies. Nutrients. 2018; 10(10):1501. https://doi.org/10.3390/nu10101501
Chicago/Turabian StyleLarsson, Susanna C., and Nicola Orsini. 2018. "Coffee Consumption and Risk of Dementia and Alzheimer’s Disease: A Dose-Response Meta-Analysis of Prospective Studies" Nutrients 10, no. 10: 1501. https://doi.org/10.3390/nu10101501
APA StyleLarsson, S. C., & Orsini, N. (2018). Coffee Consumption and Risk of Dementia and Alzheimer’s Disease: A Dose-Response Meta-Analysis of Prospective Studies. Nutrients, 10(10), 1501. https://doi.org/10.3390/nu10101501






