Can Nuts Mitigate Malnutrition in Older Adults? A Conceptual Framework
Abstract
:1. Introduction: Ageing and Risks of Malnutrition
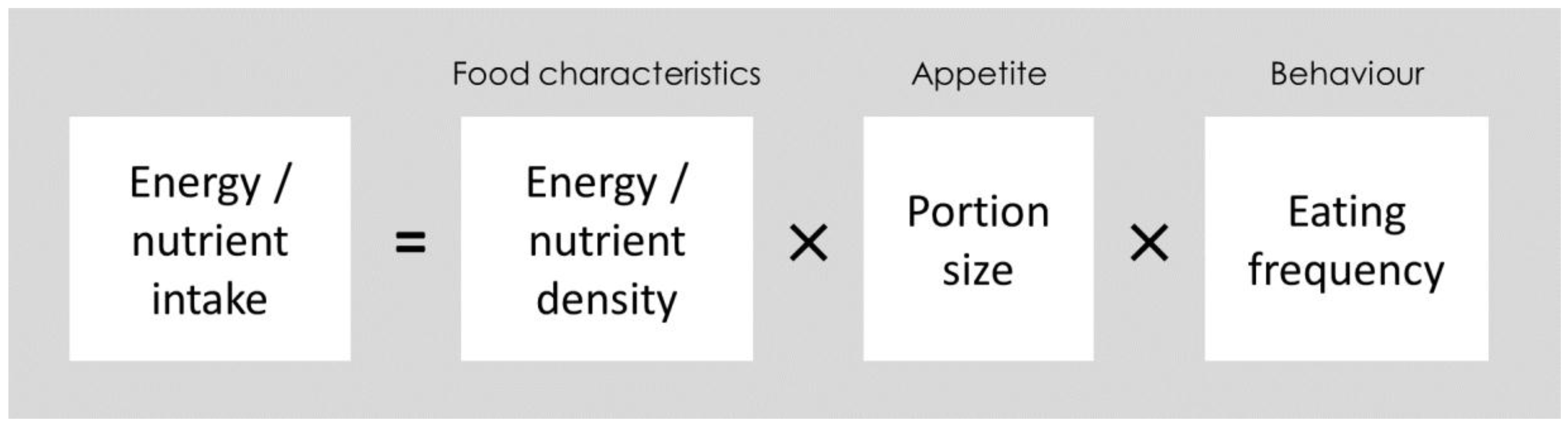
2. Nuts, Energy Balance and Undernutrition: Opportunities and Challenges
2.1. Food Characteristics: Energy Density
2.2. Appetite Regulation: Portion Size
3. Strategies to Incorporate Nuts into the Habitual Diets of Older Adults
3.1. Nut Forms
3.2. Variety
3.3. Timing
4. Improving Diet Quality with Nuts
5. Overcoming Potential Barriers and Side-Effects of Nuts
5.1. Phytate
5.2. Dental Issues
5.3. Allergies and Aflatoxins
6. Future Research Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United Nations. World population ageing 2017: Highlights. Available online: https://www.google.com.tw/url?sa=t&rct=j&q=&esrc=s&source=web&cd=4&ved=2ahUKEwjypbvvyeHdAhVGIIgKHQ51CnYQFjADegQICRAC&url=http%3A%2F%2Fwww.un.org%2Fen%2Fdevelopment%2Fdesa%2Fpopulation%2Fpublications%2Fpdf%2Fageing%2FWPA2017_Highlights.pdf&usg=AOvVaw29cbhXyZhSy_ipICc-g9vB (accessed on 2 September 2018).
- Wu, L.L.; Cheung, K.Y.; Lam, P.Y.P.; Gao, X. Oral health indicators for risk of malnutrition in elders. J. Nutr. Health Aging 2017. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E. Anorexia of aging: Physiologic and pathologic. Am. J. Clin. Nutr. 1997, 66, 760–773. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Calvani, R.; Tosato, M.; Martone, A.; Ortolani, E.; Savera, G.; Sisto, A.; Marzetti, E. Anorexia of aging: Risk factors, consequences, and potential treatments. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Abizanda, P.; Sinclair, A.; Barcons, N.; Lizán, L.; Rodríguez-Mañas, L. Costs of malnutrition in institutionalized and community-dwelling older adults: a systematic review. J. Am. Méd. Dir. Assoc. 2016, 17, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Freijer, K.; Tan, S.S.; Koopmanschap, M.A.; Meijers, J.M.M.; Halfens, R.J.G.; Nuijten, M.J.C. The economic costs of disease related malnutrition. Clin. Nutr. 2013, 32, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Muscaritoli, M.; Molfino, A. Malnutrition: The hidden killer in healthcare systems. BMJ 2013. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.; Stordalen, G.A.; Singh, S.; Wijesinha-Bettoni, R.; Shetty, P.; Lartey, A. Hunger and malnutrition in the 21st century. BMJ 2018, 361. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The double burden of malnutrition: Policy brief. Available online: http://www.who.int/nutrition/publications/doubleburdenmalnutrition-policybrief/en/ (accessed on 27 August 2018).
- Winter, J.; Flanagan, D.; McNaughton, S.A.; Nowson, C. Nutrition screening of older people in a community general practice, using the mna-sf. J. Nutr. Health Aging 2013, 17, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E.; Pedrolli, C.; Klersy, C.; Bonardi, C.; Quarleri, L.; Cappello, S.; Turri, A.; Rondanelli, M.; Caccialanza, R. Nutritional status in older persons according to healthcare setting: A systematic review and meta-analysis of prevalence data using mna. Clin. Nutr. 2016, 35, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- John, B.K.; Bullock, M.; Brenner, L.; McGaw, C.; Scolapio, J.S. Nutrition in the elderly. Frequently asked questions. Am. J. Gastroenterol. 2013, 108, 1252–1266. [Google Scholar] [CrossRef] [PubMed]
- Doets, E.L.; Kremer, S. The silver sensory experience—a review of senior consumers’ food perception, liking and intake. Food Qual. Prefer. 2016, 48, 316–332. [Google Scholar] [CrossRef]
- Rothenberg, E.; Wendin, K. 7-texture modification of food for elderly people. In Modifying Food Texture; Chen, J., Rosenthal, A., Eds.; Woodhead Publishing: Sawston, Cambridge, UK, 2015; Volume 2, pp. 163–185. [Google Scholar]
- Quandt, S.A.; Chen, H.; Bell, R.A.; Savoca, M.R.; Anderson, A.M.; Leng, X.; Kohrman, T.; Gilbert, G.H.; Arcury, T.A. Food avoidance and food modification practices of older rural adults: Association with oral health status and implications for service provision. Gerontologist 2010, 50, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, M.; Munoz, N. Position of the academy of nutrition and dietetics: Food and nutrition for older adults: Promoting health and wellness. J. Acad. Nutr. Diet. 2012, 112, 1255–1277. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-J.; Ya-Hui, C.; Wei, I.-L.; Kao, M.-D.; Yi-Chin, L.; Pan, W.-H. Intake levels and major food sources of energy and nutrients in the taiwanese elderly. Asia Pac. J. Clin. Nutr. 2005, 14, 211. [Google Scholar] [PubMed]
- World Health Organization. Integrated care for older people: Guidelines on community-level interventions to manage declines in intrinsic capacity. World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Lamuel-Raventos, R.M.; Onge, M.-P.S. Prebiotic nut compounds and human microbiota. Crit. Rev. Food Sci. Nutr. 2017, 57, 3154–3163. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Bolling, B.W. Review of nut phytochemicals, fat-soluble bioactives, antioxidant components and health effects. Br. J. Nutr. 2015, 113, S68–S78. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.-Y.; Dhillon, J.; Mattes, R.D. A review of the effects of nuts on appetite, food intake, metabolism, and body weight. Am. J. Clin. Nutr. 2014, 100, 412S–422S. [Google Scholar] [CrossRef] [PubMed]
- Ledikwe, J.H.; Blanck, H.M.; Kettel Khan, L.; Serdula, M.K.; Seymour, J.D.; Tohill, B.C.; Rolls, B.J. Dietary energy density is associated with energy intake and weight status in us adults. Am. J. Clin. Nutr. 2006, 83, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Vernarelli, J.A.; Mitchell, D.C.; Hartman, T.J.; Rolls, B.J. Dietary energy density is associated with body weight status and vegetable intake in U.S. Children. J. Nutr. 2011, 141, 2204–2210. [Google Scholar] [CrossRef] [PubMed]
- Wansink, B.; Painter, J.E.; North, J. Bottomless bowls: Why visual cues of portion size may influence intake. Obes. Res. 2005, 13, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Chia, E.M.E.; Forde, C.G. Impact of dose-response calorie reduction or supplementation of a covertly manipulated lunchtime meal on energy compensation. Physiol. Behav. 2016, 165, 15–21. [Google Scholar] [CrossRef] [PubMed]
- McCrickerd, K.; Salleh, N.; Forde, C. Removing energy from a beverage influences later food intake more than the same energy addition. Appetite 2016, 105, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Bell, E.A. Intake of fat and carbohydrate: Role of energy density. Eur. J. Clin. Nutr. 1999, 53, S166–S173. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.; Wingrove, K.; Naude, C.; Durao, S. Evidence synthesis and translation for nutrition interventions to combat micronutrient deficiencies with particular focus on food fortification. Nutrients 2016, 8, 555. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.; Wilcox, C.; Ibrahim, K.; Roberts, H. Can fortified foods and snacks increase the energy and protein intake of hospitalised older patients? A systematic review. J. Hum. Nutr. Diet. 2018, 31, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Trabal, J.; Farran-Codina, A. Effects of dietary enrichment with conventional foods on energy and protein intake in older adults: A systematic review. Nutr. Rev. 2015, 73, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Liu, X.; Malik, V.S.; Sun, Q.; Willett, W.C.; Manson, J.E.; Rexrode, K.M.; Li, Y.; Hu, F.B.; Bhupathiraju, S.N. Nut consumption and risk of cardiovascular disease. J. Am. Coll. Cardiol. 2017, 70, 2519–2532. [Google Scholar] [CrossRef] [PubMed]
- Barbour, J.A.; Howe, P.R.; Buckley, J.D.; Bryan, J.; Coates, A.M. Nut consumption for vascular health and cognitive function. Nutr. Res. Rev. 2014, 27, 131–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saaka, M.; Osman, S.M.; Amponsem, A.; Ziem, J.B.; Abdul-Mumin, A.; Akanbong, P.; Yirkyio, E.; Yakubu, E.; Ervin, S. Treatment outcome of severe acute malnutrition cases at the tamale teaching hospital. J. Nutr. Metab. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Defourny, I.; Minetti, A.; Harczi, G.; Doyon, S.; Shepherd, S.; Tectonidis, M.; Bradol, J.-H.; Golden, M. A large-scale distribution of milk-based fortified spreads: Evidence for a new approach in regions with high burden of acute malnutrition. PloS one 2009, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yebyo, H.G.; Kendall, C.; Nigusse, D.; Lemma, W. Outpatient therapeutic feeding program outcomes and determinants in treatment of severe acute malnutrition in tigray, northern ethiopia: A retrospective cohort study. Plos one 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Powley, T.L.; Phillips, R.J. Gastric satiation is volumetric, intestinal satiation is nutritive. Physiol. Behav. 2004, 82, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D.; Hollis, J.; Hayes, D.; Stunkard, A.J. Appetite: Measurement and manipulation misgivings. J. Am. Diet. Assoc. 2005, 105, S87–S97. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, J.; Running, C.A.; Tucker, R.M.; Mattes, R.D. Effects of food form on appetite and energy balance. Food Qual. Prefer. 2016, 48, 368–375. [Google Scholar] [CrossRef]
- Ello-Martin, J.A.; Ledikwe, J.H.; Rolls, B.J. The influence of food portion size and energy density on energy intake: Implications for weight management. Am. J. Clin. Nutr. 2005, 82, 236S–241S. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, J.; Craig, B.A.; Leidy, H.J.; Amankwaah, A.F.; Anguah, K.O.-B.; Jacobs, A.; Jones, B.L.; Jones, J.B.; Keeler, C.L.; Keller, C.E. The effects of increased protein intake on fullness: A meta-analysis and its limitations. J. Acad. Nutr. Diet. 2016, 116, 968–983. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.J.; Slavin, J.L. The effect of fiber on satiety and food intake: A systematic review. J. Am. Coll. Nutr. 2013, 32, 200–211. [Google Scholar] [CrossRef] [PubMed]
- de Graaf, C. Texture and satiation: The role of oro-sensory exposure time. Physiol. Behav. 2012, 107, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Fraser, G.E.; Bennett, H.W.; Jaceldo, K.B.; Sabate, J. Effect on body weight of a free 76 kilojoule (320 calorie) daily supplement of almonds for six months. J. Am. Coll. Nutr. 2002, 21, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Kirkmeyer, S.V.; Mattes, R.D. Effects of food attributes on hunger and food intake. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1167–1175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hollis, J.; Mattes, R. Effect of chronic consumption of almonds on body weight in healthy humans. Br. J. Nutr. 2007, 98, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Alper, C.M.; Mattes, R.D. Effects of chronic peanut consumption on energy balance and hedonics. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 1129–1137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellis, P.R.; Kendall, C.W.C.; Ren, Y.L.; Parker, C.; Pacy, J.F.; Waldron, K.W.; Jenkins, D.J.A. Role of cell walls in the bioaccessibility of lipids in almond seeds. Am. J. Clin. Nutr. 2004, 80, 604–613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Novotny, J.A.; Gebauer, S.K.; Baer, D.J. Discrepancy between the atwater factor predicted and empirically measured energy values of almonds in human diets. Am. J. Clin. Nutr. 2012, 96, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Gebauer, S.K.; Novotny, J.A. Measured energy value of pistachios in the human diet. Br. J. Nutr. 2012, 107, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Gebauer, S.K.; Novotny, J.A. Walnuts consumed by healthy adults provide less available energy than predicted by the atwater factors. J. Nutr. 2015, 146, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Grundy, M.M.L.; Grassby, T.; Mandalari, G.; Waldron, K.W.; Butterworth, P.J.; Berry, S.E.E.; Ellis, P.R. Effect of mastication on lipid bioaccessibility of almonds in a randomized human study and its implications for digestion kinetics, metabolizable energy, and postprandial lipemia. Am. J. Clin. Nutr. 2015, 101, 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frecka, J.; Hollis, J.; Mattes, R. Effects of appetite, BMI, food form and flavor on mastication: Almonds as a test food. Eur. J. Clin. Nutr. 2008, 62, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, S.K.; Novotny, J.A.; Bornhorst, G.M.; Baer, D.J. Food processing and structure impact the metabolizable energy of almonds. Food Funct. 2016, 7, 4231–4238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKiernan, F.; Mattes, R.D. Effects of peanut processing on masticatory performance during variable appetitive states. J. Nutr. Metab. 2010, 2010. [Google Scholar] [CrossRef] [PubMed]
- Traoret, C.; Lokko, P.; Cruz, A.; Oliveira, C.; Costa, N.; Bressan, J.; Alfenas, R.; Mattes, R. Peanut digestion and energy balance. Int. J. Obes. 2008, 32, 322–328. [Google Scholar] [CrossRef] [PubMed]
- McKiernan, F.; Lokko, P.; Kuevi, A.; Sales, R.L.; Costa, N.M.; Bressan, J.; Alfenas, R.C.; Mattes, R.D. Effects of peanut processing on body weight and fasting plasma lipids. Br. J. Nutr. 2010, 104, 418–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasman, W.J.; Heimerikx, J.; Rubingh, C.M.; van den Berg, R.; O’Shea, M.; Gambelli, L.; Hendriks, H.F.J.; Einerhand, A.W.C.; Scott, C.; Keizer, H.G.; et al. The effect of korean pine nut oil on in vitro cck release, on appetite sensations and on gut hormones in post-menopausal overweight women. Lipids Health Dis. 2008, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Lauque, S.; Arnaud-Battandier, F.; Mansourian, R.; Guigoz, Y.; Paintin, M.; Nourhashemi, F.; Vellas, B. Protein-energy oral supplementation in malnourished nursing-home residents. A controlled trial. Age Ageing 2000, 29, 51–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nieuwenhuizen, W.F.; Weenen, H.; Rigby, P.; Hetherington, M.M. Older adults and patients in need of nutritional support: Review of current treatment options and factors influencing nutritional intake. Clin. Nutr. 2010, 29, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, G.P.; Elia, M.; Holdoway, A.; Stratton, R.J. A systematic review of compliance to oral nutritional supplements. Clin. Nutr. 2012, 31, 293–312. [Google Scholar] [CrossRef] [PubMed]
- Marcenes, W.; Steele, J.G.; Sheiham, A.; Walls, A.W.G. The relationship between dental status, food selection, nutrient intake, nutritional status, and body mass index in older people. Cad. Saude Publica 2003, 19, 809–815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolls, B.J. Sensory-specific satiety. Nutr. Rev. 1986, 44, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Delahunty, C.; Gray, A.; Chisholm, A.; Brown, R.C. Effects of regular consumption of different forms of almonds and hazelnuts on acceptance and blood lipids. Eur. J. Nutr. 2015, 54, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.B.; Provost, M.; Keaver, L.; Breen, C.; Ludy, M.-J.; Mattes, R.D. A randomized trial on the effects of flavorings on the health benefits of daily peanut consumption. Am. J. Clin. Nutr. 2014, 99, 490–496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tey, S.L.; Brown, R.; Chisholm, A.; Delahunty, C.; Gray, A.; Williams, S. Effects of different forms of hazelnuts on blood lipids and α-tocopherol concentrations in mildly hypercholesterolemic individuals. Eur. J. Clin. Nutr. 2011, 65, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Brown, R.; Chisholm, A.; Gray, A.; Williams, S.; Delahunty, C. Current guidelines for nut consumption are achievable and sustainable: A hazelnut intervention. Br. J. Nutr. 2011, 105, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Stroehla, B.C.; Malcoe, L.H.; Velie, E.M. Dietary sources of nutrients among rural native american and white children. J. Am. Diet. Assoc. 2005, 105, 1908–1916. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, T.A.; Demory-Luce, D.; Yang, S.J.; Baranowski, T.; Zakeri, I.; Berenson, G. Children’s food consumption patterns have changed over two decades (1973–1994): The bogalusa heart study. J. Am. Diet. Assoc. 2004, 104, 1127–1240. [Google Scholar] [CrossRef] [PubMed]
- Zizza, C.; Arsiwalla, D.D.; Ellison, K.J. Contribution of snacking to older adults’ vitamin, carotenoid, and mineral intakes. J. Am. Diet. Assoc. 2010, 10, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Ros, E. Health benefits of nut consumption. Nutrients 2010, 2, 652–682. [Google Scholar] [CrossRef] [PubMed]
- USDA National Nutrient Database for Standard Reference, Release 28. Available online: https://www.google.com.tw/url?sa=t&rct=j&q=&esrc=s&source=web&cd=16&ved=2ahUKEwiayNns6uTdAhXH7GEKHZYHBtw4ChAWMAV6BAgBEAI&url=https%3A%2F%2Fods.od.nih.gov%2Fpubs%2Fusdandb%2FCalcium-Content.pdf&usg=AOvVaw2csgADPanW96U7T_06Qu_G (accessed on 12 September 2018).
- Brown, R.C.; Tey, S.L.; Gray, A.R.; Chisholm, A.; Smith, C.; Fleming, E.; Parnell, W. Nut consumption is associated with better nutrient intakes: Results from the 2008/09 New Zealand adult nutrition survey. Br. J. Nutr. 2016, 115, 105–112. [Google Scholar] [CrossRef] [PubMed]
- King, J.C.; Blumberg, J.; Ingwersen, L.; Jenab, M.; Tucker, K.L. Tree nuts and peanuts as components of a healthy diet. J. Nutr. 2008, 138, S1736–S1740. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Tree nut consumption improves nutrient intake and diet quality in us adults: An analysis of national health and nutrition examination survey (nhanes) 1999–2004. Asia Pac. J. Clin. Nutr. 2010, 19, 142–150. [Google Scholar] [PubMed]
- O’Neil, C.E.; Keast, D.R.; Nicklas, T.A.; Fulgoni, V.L. Out-of-hand nut consumption is associated with improved nutrient intake and health risk markers in us children and adults: National health and nutrition examination survey 1999–2004. Nutr. Res. 2012, 32, 185–194. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, C.E.; Nicklas, T.A.; Fulgoni III, V.L. Tree nut consumption is associated with better nutrient adequacy and diet quality in adults: National health and nutrition examination survey 2005–2010. Nutrients 2015, 7, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Griel, A.E.; Eissenstat, B.; Juturu, V.; Hsieh, G.; Kris-Etherton, P.M. Improved diet quality with peanut consumption. J. Am. Coll. Nutr. 2004, 23, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Bitok, E.; Jaceldo-Siegl, K.; Rajaram, S.; Serra-Mir, M.; Roth, I.; Feitas-Simoes, T.; Ros, E.; Sabaté, J. Favourable nutrient intake and displacement with long-term walnut supplementation among elderly: Results of a randomised trial. Br. J. Nutr. 2017, 118, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Jaceldo-Siegl, K.; Joan, S.; Rajaram, S.; Fraser, G.E. Long-term almond supplementation without advice on food replacement induces favourable nutrient modifications to the habitual diets of free-living individuals. Br. J. Nutr. 2004, 92, 533–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tey, S.L.; Brown, R.; Gray, A.; Chisholm, A.; Delahunty, C. Nuts improve diet quality compared to other energy-dense snacks while maintaining body weight. J. Nutr. Metab. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Gray, A.R.; Chisholm, A.W.; Delahunty, C.M.; Brown, R.C. The dose of hazelnuts influences acceptance and diet quality but not inflammatory markers and body composition in overweight and obese individuals. J. Nutr. 2013, 143, 1254–1262. [Google Scholar] [CrossRef] [PubMed]
- Harland, B.F.; Smikle-Williams, S.; Oberleas, D. High performance liquid chromatography analysis of phytate (ip6) in selected foods. J. Food Compost. Anal. 2004, 17, 227–233. [Google Scholar] [CrossRef]
- Hurrell. Phytic acid degradation as a means of improving iron absorption. Int. J. Vitam. Nutr. Res. 2004, 74, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, M.; Sathe, S.K. Chemical composition of selected edible nut seeds. J. Agric. Food Chem. 2006, 54, 4705–4714. [Google Scholar] [CrossRef] [PubMed]
- International Zinc Nutrition Consultative Group. International zinc nutrition consultative group (izincg) technical document# 1. Assessment of the risk of zinc deficiency in populations and options for its control. Food Nutr. Bull. 2004, 25, S99–S203. [Google Scholar]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2015, 52, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Lestienne, I.; Mouquet-Rivier, C.; Icard-Vernière, C.; Rochette, I.; Trèche, S. The effects of soaking of whole, dehulled and ground millet and soybean seeds on phytate degradation and phy/fe and phy/zn molar ratios. Int. J. Food Sci. Tech. 2005, 40, 391–399. [Google Scholar] [CrossRef]
- Hotz, C.; Gibson, R.S. Assessment of home-based processing methods to reduce the phytate content and phytate/zinc molar ratio of white maize (zea mays). J. Agric. Food Chem. 2001, 49, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Giam, X.Y.; Foo, X.M.; Yeo, H.L.; Koh, J.L.; Sa’Aban, N.H.B.; Loke, W.M. Effects of pre-germination treatment on the phytate and phenolic contents of almond nuts. J. Nuts 2017, 8, 73–80. [Google Scholar]
- Taylor, H.; Webster, K.; Gray, A.R.; Tey, S.L.; Chisholm, A.; Bailey, K.; Kumari, S.; Brown, R.C. The effects of ‘activating’ almonds on consumer acceptance and gastrointestinal tolerance. Eur. J. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Yong, L.C.; Gray, A.R.; Chisholm, A.; Leong, S.L.; Tey, S.L.; Brown, R.C. Barriers to and facilitators and perceptions of nut consumption among the general population in New Zealand. Public Health Nutr. 2017, 20, 3166–3182. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.C.; Yong, L.C.; Gray, A.R.; Tey, S.L.; Chisholm, A.; Leong, S.L. Perceptions and knowledge of nuts amongst health professionals in New Zealand. Nutrients 2017, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- Brough, H.; Turner, P.; Wright, T.; Fox, A.; Taylor, S.; Warner, J.; Lack, G. Dietary management of peanut and tree nut allergy: What exactly should patients avoid? Clin. Exp. Allergy 2015, 45, 859–871. [Google Scholar] [CrossRef] [PubMed]
- Rona, R.J.; Keil, T.; Summers, C.; Gislason, D.; Zuidmeer, L.; Sodergren, E.; Sigurdardottir, S.T.; Lindner, T.; Goldhahn, K.; Dahlstrom, J. The prevalence of food allergy: A meta-analysis. J. Allergy Clin. Immunol. 2007, 120, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Davis, P.A.; Jenab, M.; Vanden Heuvel, J.P.; Furlong, T.; Taylor, S. Tree nut and peanut consumption in relation to chronic and metabolic diseases including allergy. J. Nutr. 2008, 138, S1757–S1762. [Google Scholar] [CrossRef] [PubMed]
- van Egmond, H.P.; Schothorst, R.C.; Jonker, M.A. Regulations relating to mycotoxins in food: Perspectives in a global and european context. Anal. Bioanal. Chem. 2007, 389, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Edlayne, G.; Simone, A.; Felicio, J.D. Chemical and biological approaches for mycotoxin control: A review. Recent Pat. Food Nutr. Agric. 2009, 1, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Shahidi, F. Tree nuts: Composition, phytochemicals, and health effects: An overview. In Tree nuts: Composition, phytochemicals, and health effects; Alasalvar, C., Shahidi, F., Eds.; Taylor & Francis Group: Boca Raton, Florida, USA, 2008; pp. 1–10. [Google Scholar]
- Eneroth, H.; Wallin, S.; Leander, K.; Nilsson Sommar, J.; Åkesson, A. Risks and benefits of increased nut consumption: Cardiovascular health benefits outweigh the burden of carcinogenic effects attributed to aflatoxin b1 exposure. Nutrients 2017, 9, 1355. [Google Scholar] [CrossRef] [PubMed]
| NZ/Australia RDI/AI | USA RDA/AI | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Males | Females | Males | Females | ||||||
| 51–70 years | >70 years | 51–70 years | >70 years | 51–70 years | >70 years | 51–70 years | >70 years | ||
| Protein (g) | Amount per 30 g | % Daily Value | |||||||
| 64 * | 81 * | 46 * | 57 * | 56 * | 56 * | 46 * | 46 * | ||
| Almonds | 6.35 | 9.9 | 7.8 | 13.8 | 11.1 | 11.3 | 11.3 | 13.8 | 13.8 |
| Brazil nuts | 4.30 | 6.7 | 5.3 | 9.3 | 7.5 | 7.7 | 7.7 | 9.3 | 9.3 |
| Cashew nuts | 5.30 | 8.3 | 6.5 | 11.5 | 9.3 | 9.5 | 9.5 | 11.5 | 11.5 |
| Hazelnuts | 4.49 | 7.0 | 5.5 | 9.8 | 7.9 | 8.0 | 8.0 | 9.8 | 9.8 |
| Macadamia nuts | 2.37 | 3.7 | 2.9 | 5.2 | 4.2 | 4.2 | 4.2 | 5.2 | 5.2 |
| Peanuts | 7.74 | 12.1 | 9.6 | 16.8 | 13.6 | 13.8 | 13.8 | 16.8 | 16.8 |
| Pecans | 2.75 | 4.3 | 3.4 | 6.0 | 4.8 | 4.9 | 4.9 | 6.0 | 6.0 |
| Pine nuts | 4.11 | 6.4 | 5.1 | 8.9 | 7.2 | 7.3 | 7.3 | 8.9 | 8.9 |
| Pistachios | 6.05 | 9.5 | 7.5 | 13.2 | 10.6 | 10.8 | 10.8 | 13.2 | 13.2 |
| Walnuts | 4.57 | 7.1 | 5.6 | 9.9 | 8.0 | 8.2 | 8.2 | 9.9 | 9.9 |
| Range | 2.37–7.74 | 3.7–12.1 | 2.9–9.6 | 5.2–16.8 | 4.2–13.6 | 4.2–13.8 | 4.2–13.8 | 5.2–16.8 | 5.2–16.8 |
| Dietary fibre (g) | Amount per 30 g | % Daily Value | |||||||
| 30 † | 30 † | 25 † | 25 † | 30 † | 30 † | 21 † | 21 † | ||
| Almonds | 3.75 | 12.5 | 12.5 | 15.0 | 15.0 | 12.5 | 12.5 | 17.9 | 17.9 |
| Brazil nuts | 2.25 | 7.5 | 7.5 | 9.0 | 9.0 | 7.5 | 7.5 | 10.7 | 10.7 |
| Cashew nuts | 0.99 | 3.3 | 3.3 | 4.0 | 4.0 | 3.3 | 3.3 | 4.7 | 4.7 |
| Hazelnuts | 2.91 | 9.7 | 9.7 | 11.6 | 11.6 | 9.7 | 9.7 | 13.9 | 13.9 |
| Macadamia nuts | 2.58 | 8.6 | 8.6 | 10.3 | 10.3 | 8.6 | 8.6 | 12.3 | 12.3 |
| Peanuts | 2.55 | 8.5 | 8.5 | 10.2 | 10.2 | 8.5 | 8.5 | 12.1 | 12.1 |
| Pecans | 2.88 | 9.6 | 9.6 | 11.5 | 11.5 | 9.6 | 9.6 | 13.7 | 13.7 |
| Pine nuts | 1.11 | 3.7 | 3.7 | 4.4 | 4.4 | 3.7 | 3.7 | 5.3 | 5.3 |
| Pistachios | 3.18 | 10.6 | 10.6 | 12.7 | 12.7 | 10.6 | 10.6 | 15.1 | 15.1 |
| Walnuts | 2.01 | 6.7 | 6.7 | 8.0 | 8.0 | 6.7 | 6.7 | 9.6 | 9.6 |
| Range | 0.99–3.75 | 3.3–12.5 | 3.3–12.5 | 4.0–15.0 | 4.0–15.0 | 3.3–12.5 | 3.3–12.5 | 4.7–17.9 | 4.7–17.9 |
| α-Tocopherol (mg) | Amount per 30 g | % Daily Value | |||||||
| 10 † | 10 † | 7 † | 7 † | 15 * | 15 * | 15 * | 15 * | ||
| Almonds | 7.69 | 76.9 | 76.9 | 109.9 | 109.9 | 51.3 | 51.3 | 51.3 | 51.3 |
| Brazil nuts | 1.70 | 17.0 | 17.0 | 24.3 | 24.3 | 11.3 | 11.3 | 11.3 | 11.3 |
| Cashew nuts | 0.90 | 9.0 | 9.0 | 12.9 | 12.9 | 6.0 | 6.0 | 6.0 | 6.0 |
| Hazelnuts | 5.51 | 55.1 | 55.1 | 78.7 | 78.7 | 36.7 | 36.7 | 36.7 | 36.7 |
| Macadamia nuts | 0.16 | 1.6 | 1.6 | 2.3 | 2.3 | 1.1 | 1.1 | 1.1 | 1.1 |
| Peanuts | 2.50 | 25.0 | 25.0 | 35.7 | 35.7 | 16.7 | 16.7 | 16.7 | 16.7 |
| Pecans | 0.42 | 4.2 | 4.2 | 6.0 | 6.0 | 2.8 | 2.8 | 2.8 | 2.8 |
| Pine nuts | 2.80 | 28.0 | 28.0 | 40.0 | 40.0 | 18.7 | 18.7 | 18.7 | 18.7 |
| Pistachios | 0.86 | 8.6 | 8.6 | 12.3 | 12.3 | 5.7 | 5.7 | 5.7 | 5.7 |
| Walnuts | 0.21 | 2.1 | 2.1 | 3.0 | 3.0 | 1.4 | 1.4 | 1.4 | 1.4 |
| Range | 0.16–7.69 | 1.6–76.9 | 1.6–76.9 | 2.3–109.9 | 2.3–109.9 | 1.1–51.3 | 1.1–51.3 | 1.1–51.3 | 1.1–51.3 |
| Folate (µg) | Amount per 30 g | % Daily Value | |||||||
| 400 * | 400 * | 400 * | 400 * | 400 * | 400 * | 400 * | 400 * | ||
| Almonds | 13.2 | 3.3 | 3.3 | 3.3 | 3.3 | 3.3 | 3.3 | 3.3 | 3.3 |
| Brazil nuts | 6.6 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 |
| Cashew nuts | 7.5 | 1.9 | 1.9 | 1.9 | 1.9 | 1.9 | 1.9 | 1.9 | 1.9 |
| Hazelnuts | 33.9 | 8.5 | 8.5 | 8.5 | 8.5 | 8.5 | 8.5 | 8.5 | 8.5 |
| Macadamia nuts | 3.3 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Peanuts | 72.0 | 18.0 | 18.0 | 18.0 | 18.0 | 18.0 | 18.0 | 18.0 | 18.0 |
| Pecans | 6.6 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 | 1.7 |
| Pine nuts | 10.2 | 2.6 | 2.6 | 2.6 | 2.6 | 2.6 | 2.6 | 2.6 | 2.6 |
| Pistachios | 15.3 | 3.8 | 3.8 | 3.8 | 3.8 | 3.8 | 3.8 | 3.8 | 3.8 |
| Walnuts | 29.4 | 7.4 | 7.4 | 7.4 | 7.4 | 7.4 | 7.4 | 7.4 | 7.4 |
| Range | 3.3–72.0 | 0.8–18.0 | 0.8–18.0 | 0.8–18.0 | 0.8–18.0 | 0.8–18.0 | 0.8–18.0 | 0.8–18.0 | 0.8–18.0 |
| Calcium (mg) | Amount per 30 g | % Daily Value | |||||||
| 1000 * | 1300 * | 1300 * | 1300 * | 1000 * | 1200 * | 1200 * | 1200 * | ||
| Almonds | 80.7 | 8.1 | 6.2 | 6.2 | 6.2 | 8.1 | 6.7 | 6.7 | 6.7 |
| Brazil nuts | 48.0 | 4.8 | 3.7 | 3.7 | 3.7 | 4.8 | 4.0 | 4.0 | 4.0 |
| Cashew nuts | 11.1 | 1.1 | 0.9 | 0.9 | 0.9 | 1.1 | 0.9 | 0.9 | 0.9 |
| Hazelnuts | 34.2 | 3.4 | 2.6 | 2.6 | 2.6 | 3.4 | 2.9 | 2.9 | 2.9 |
| Macadamia nuts | 25.5 | 2.6 | 2.0 | 2.0 | 2.0 | 2.6 | 2.1 | 2.1 | 2.1 |
| Peanuts | 27.7 | 2.8 | 2.1 | 2.1 | 2.1 | 2.8 | 2.3 | 2.3 | 2.3 |
| Pecans | 21.0 | 2.1 | 1.6 | 1.6 | 1.6 | 2.1 | 1.8 | 1.8 | 1.8 |
| Pine nuts | 4.8 | 0.5 | 0.4 | 0.4 | 0.4 | 0.5 | 0.4 | 0.4 | 0.4 |
| Pistachios | 31.5 | 3.2 | 2.4 | 2.4 | 2.4 | 3.2 | 2.6 | 2.6 | 2.6 |
| Walnuts | 29.0 | 2.9 | 2.2 | 2.2 | 2.2 | 2.9 | 2.4 | 2.4 | 2.4 |
| Range | 4.8–80.7 | 0.5–8.1 | 0.4–6.2 | 0.4–6.2 | 0.4–6.2 | 0.5–8.1 | 0.4–6.7 | 0.4–6.7 | 0.4–6.7 |
| Copper (µg) | Amount per 30 g | % Daily Value | |||||||
| 1700 † | 1700 † | 1200 † | 1200 † | 900 * | 900 * | 900 * | 900 * | ||
| Almonds | 309 | 18.2 | 18.2 | 25.8 | 25.8 | 34.3 | 34.3 | 34.3 | 34.3 |
| Brazil nuts | 523 | 30.8 | 30.8 | 43.6 | 43.6 | 58.1 | 58.1 | 58.1 | 58.1 |
| Cashew nuts | 659 | 38.8 | 38.8 | 54.9 | 54.9 | 73.2 | 73.2 | 73.2 | 73.2 |
| Hazelnuts | 518 | 30.5 | 30.5 | 43.2 | 43.2 | 57.6 | 57.6 | 57.6 | 57.6 |
| Macadamia nuts | 227 | 13.4 | 13.4 | 18.9 | 18.9 | 25.2 | 25.2 | 25.2 | 25.2 |
| Peanuts | 343 | 20.2 | 20.2 | 28.6 | 28.6 | 38.1 | 38.1 | 38.1 | 38.1 |
| Pecans | 360 | 21.2 | 21.2 | 30.0 | 30.0 | 40.0 | 40.0 | 40.0 | 40.0 |
| Pine nuts | 397 | 23.4 | 23.4 | 33.1 | 33.1 | 44.1 | 44.1 | 44.1 | 44.1 |
| Pistachios | 390 | 22.9 | 22.9 | 32.5 | 32.5 | 43.3 | 43.3 | 43.3 | 43.3 |
| Walnuts | 476 | 28.0 | 28.0 | 39.7 | 39.7 | 52.9 | 52.9 | 52.9 | 52.9 |
| Range | 277–659 | 13.4–38.8 | 13.4–38.8 | 18.9–54.9 | 18.9–54.9 | 25.2–73.2 | 25.2–73.2 | 25.2–73.2 | 25.2–73.2 |
| Iron (mg) | Amount per 30 g | % Daily Value | |||||||
| 8 * | 8 * | 8 * | 8 * | 8 * | 8 * | 8 * | 8 * | ||
| Almonds | 1.11 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 |
| Brazil nuts | 0.73 | 9.1 | 9.1 | 9.1 | 9.1 | 9.1 | 9.1 | 9.1 | 9.1 |
| Cashew nuts | 2.00 | 25.0 | 25.0 | 25.0 | 25.0 | 25.0 | 25.0 | 25.0 | 25.0 |
| Hazelnuts | 1.41 | 17.6 | 17.6 | 17.6 | 17.6 | 17.6 | 17.6 | 17.6 | 17.6 |
| Macadamia nuts | 1.11 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 | 13.9 |
| Peanuts | 1.37 | 17.1 | 17.1 | 17.1 | 17.1 | 17.1 | 17.1 | 17.1 | 17.1 |
| Pecans | 0.76 | 9.5 | 9.5 | 9.5 | 9.5 | 9.5 | 9.5 | 9.5 | 9.5 |
| Pine nuts | 1.55 | 19.4 | 19.4 | 19.4 | 19.4 | 19.4 | 19.4 | 19.4 | 19.4 |
| Pistachios | 1.18 | 14.8 | 14.8 | 14.8 | 14.8 | 14.8 | 14.8 | 14.8 | 14.8 |
| Walnuts | 0.87 | 10.9 | 10.9 | 10.9 | 10.9 | 10.9 | 10.9 | 10.9 | 10.9 |
| Range | 0.73–2.00 | 9.1–25.0 | 9.1–25.0 | 9.1–25.0 | 9.1–25.0 | 9.1–25.0 | 9.1–25.0 | 9.1–25.0 | 9.1–25.0 |
| Magnesium (mg) | Amount per 30 g | % Daily Value | |||||||
| 420 * | 420 * | 320 * | 320 * | 420 * | 420 * | 320 * | 320 * | ||
| Almonds | 81 | 19.3 | 19.3 | 25.3 | 25.3 | 19.3 | 19.3 | 25.3 | 25.3 |
| Brazil nuts | 113 | 26.9 | 26.9 | 35.3 | 35.3 | 26.9 | 26.9 | 35.3 | 35.3 |
| Cashew nuts | 88 | 21.0 | 21.0 | 27.5 | 27.5 | 21.0 | 21.0 | 27.5 | 27.5 |
| Hazelnuts | 49 | 11.7 | 11.7 | 15.3 | 15.3 | 11.7 | 11.7 | 15.3 | 15.3 |
| Macadamia nuts | 39 | 9.3 | 9.3 | 12.2 | 12.2 | 9.3 | 9.3 | 12.2 | 12.2 |
| Peanuts | 50 | 11.9 | 11.9 | 15.6 | 15.6 | 11.9 | 11.9 | 15.6 | 15.6 |
| Pecans | 36 | 8.6 | 8.6 | 11.3 | 11.3 | 8.6 | 8.6 | 11.3 | 11.3 |
| Pine nuts | 75 | 17.9 | 17.9 | 23.4 | 23.4 | 17.9 | 17.9 | 23.4 | 23.4 |
| Pistachios | 36 | 8.6 | 8.6 | 11.3 | 11.3 | 8.6 | 8.6 | 11.3 | 11.3 |
| Walnuts | 47 | 11.2 | 11.2 | 14.7 | 14.7 | 11.2 | 11.2 | 14.7 | 14.7 |
| Range | 36–113 | 8.6–26.9 | 8.6–26.9 | 11.3–35.3 | 11.3–35.3 | 8.6–26.9 | 8.6–26.9 | 11.3–35.3 | 11.3–35.3 |
| Phosphorous (mg) | Amount per 30 g | % Daily Value | |||||||
| 1000 * | 1000 * | 1000 * | 1000 * | 700 * | 700 * | 700 * | 700 * | ||
| Almonds | 144 | 14.4 | 14.4 | 14.4 | 14.4 | 20.6 | 20.6 | 20.6 | 20.6 |
| Brazil nuts | 218 | 21.8 | 21.8 | 21.8 | 21.8 | 31.1 | 31.1 | 31.1 | 31.1 |
| Cashew nuts | 178 | 17.8 | 17.8 | 17.8 | 17.8 | 25.4 | 25.4 | 25.4 | 25.4 |
| Hazelnuts | 87 | 8.7 | 8.7 | 8.7 | 8.7 | 12.4 | 12.4 | 12.4 | 12.4 |
| Macadamia nuts | 56 | 5.6 | 5.6 | 5.6 | 5.6 | 8.0 | 8.0 | 8.0 | 8.0 |
| Peanuts | 113 | 11.3 | 11.3 | 11.3 | 11.3 | 16.1 | 16.1 | 16.1 | 16.1 |
| Pecans | 83 | 8.3 | 8.3 | 8.3 | 8.3 | 11.9 | 11.9 | 11.9 | 11.9 |
| Pine nuts | 173 | 17.3 | 17.3 | 17.3 | 17.3 | 24.7 | 24.7 | 24.7 | 24.7 |
| Pistachios | 147 | 14.7 | 14.7 | 14.7 | 14.7 | 21.0 | 21.0 | 21.0 | 21.0 |
| Walnuts | 104 | 10.4 | 10.4 | 10.4 | 10.4 | 14.9 | 14.9 | 14.9 | 14.9 |
| Range | 56–218 | 5.6–21.8 | 5.6–21.8 | 5.6–21.8 | 5.6–21.8 | 8.0–31.1 | 8.0–31.1 | 8.0–31.1 | 8.0–31.1 |
| Potassium (mg) | Amount per 30 g | % Daily Value | |||||||
| 3800 † | 3800 † | 2800 † | 2800 † | 4700 † | 4700 † | 4700 † | 4700 † | ||
| Almonds | 220 | 5.8 | 5.8 | 7.9 | 7.9 | 4.7 | 4.7 | 4.7 | 4.7 |
| Brazil nuts | 198 | 5.2 | 5.2 | 7.1 | 7.1 | 4.2 | 4.2 | 4.2 | 4.2 |
| Cashew nuts | 198 | 5.2 | 5.2 | 7.1 | 7.1 | 4.2 | 4.2 | 4.2 | 4.2 |
| Hazelnuts | 204 | 5.4 | 5.4 | 7.3 | 7.3 | 4.3 | 4.3 | 4.3 | 4.3 |
| Macadamia nuts | 110 | 2.9 | 2.9 | 3.9 | 3.9 | 2.3 | 2.3 | 2.3 | 2.3 |
| Peanuts | 212 | 5.6 | 5.6 | 7.6 | 7.6 | 4.5 | 4.5 | 4.5 | 4.5 |
| Pecans | 123 | 3.2 | 3.2 | 4.4 | 4.4 | 2.6 | 2.6 | 2.6 | 2.6 |
| Pine nuts | 179 | 4.7 | 4.7 | 6.4 | 6.4 | 3.8 | 3.8 | 3.8 | 3.8 |
| Pistachios | 308 | 8.1 | 8.1 | 11.0 | 11.0 | 6.6 | 6.6 | 6.6 | 6.6 |
| Walnuts | 132 | 3.5 | 3.5 | 4.7 | 4.7 | 2.8 | 2.8 | 2.8 | 2.8 |
| Range | 110–308 | 2.9–8.1 | 2.9–8.1 | 3.9–11.0 | 3.9–11.0 | 2.3–6.6 | 2.3–6.6 | 2.3–6.6 | 2.3–6.6 |
| Selenium (µg) | Amount per 30 g | % Daily Value | |||||||
| 70 * | 70 * | 60 * | 60 * | 55 * | 55 * | 55 * | 55 * | ||
| Almonds | 1.23 | 1.8 | 1.8 | 2.1 | 2.1 | 2.2 | 2.2 | 2.2 | 2.2 |
| Brazil nuts | 575 | 821 | 821 | 958 | 958 | 1045 | 1045 | 1045 | 1045 |
| Cashew nuts | 5.97 | 8.5 | 8.5 | 10.0 | 10.0 | 10.9 | 10.9 | 10.9 | 10.9 |
| Hazelnuts | 0.72 | 1.0 | 1.0 | 1.2 | 1.2 | 1.3 | 1.3 | 1.3 | 1.3 |
| Macadamia nuts | 1.08 | 1.5 | 1.5 | 1.8 | 1.8 | 2.0 | 2.0 | 2.0 | 2.0 |
| Peanuts | 2.16 | 3.1 | 3.1 | 3.6 | 3.6 | 3.9 | 3.9 | 3.9 | 3.9 |
| Pecans | 1.14 | 1.6 | 1.6 | 1.9 | 1.9 | 2.1 | 2.1 | 2.1 | 2.1 |
| Pine nuts | 0.21 | 0.3 | 0.3 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 | 0.4 |
| Pistachios | 2.1 | 3.0 | 3.0 | 3.5 | 3.5 | 3.8 | 3.8 | 3.8 | 3.8 |
| Walnuts | 1.47 | 2.1 | 2.1 | 2.5 | 2.5 | 2.7 | 2.7 | 2.7 | 2.7 |
| Range | 0.21–575 | 0.3–821 | 0.3–821 | 0.4–958 | 0.4–958 | 0.4–1045 | 0.4–1045 | 0.4–1045 | 0.4–1045 |
| Sodium (mg) | Amount per 30 g | % Daily Value | |||||||
| 460–920 † | 460–920 † | 460–920 † | 460–920 † | 1300 † | 1200 † | 1300 † | 1200 † | ||
| Almonds | 0.3 | 0.03–0.07 | 0.03–0.07 | 0.03–0.07 | 0.03–0.07 | 0.02 | 0.03 | 0.02 | 0.03 |
| Brazil nuts | 0.9 | 0.10–0.20 | 0.10–0.20 | 0.10–0.20 | 0.10–0.20 | 0.07 | 0.08 | 0.07 | 0.08 |
| Cashew nuts | 3.6 | 0.39–0.78 | 0.39–0.78 | 0.39–0.78 | 0.39–0.78 | 0.28 | 0.30 | 0.28 | 0.30 |
| Hazelnuts | 0.0 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Macadamia nuts | 1.5 | 0.16–0.33 | 0.16–0.33 | 0.16–0.33 | 0.16–0.33 | 0.12 | 0.13 | 0.12 | 0.13 |
| Peanuts | 5.4 | 0.59–1.17 | 0.59–1.17 | 0.59–1.17 | 0.59–1.17 | 0.42 | 0.45 | 0.42 | 0.45 |
| Pecans | 0.0 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pine nuts | 0.6 | 0.07–0.13 | 0.07–0.13 | 0.07–0.13 | 0.07–0.13 | 0.05 | 0.05 | 0.05 | 0.05 |
| Pistachios | 0.3 | 0.03–0.07 | 0.03–0.07 | 0.03–0.07 | 0.03–0.07 | 0.02 | 0.03 | 0.02 | 0.03 |
| Walnuts | 0.6 | 0.07–0.13 | 0.07–0.13 | 0.07–0.13 | 0.07–0.13 | 0.05 | 0.05 | 0.05 | 0.05 |
| Range | 0.0–5.4 | 0.00–1.17 | 0.00–1.17 | 0.00–1.17 | 0.00–1.17 | 0.00–0.42 | 0.00–0.45 | 0.00–0.42 | 0.00–0.45 |
| Zinc (mg) | Amount per 30 g | % Daily Value | |||||||
| 12 * | 12 * | 6.5 * | 6.5 * | 11 * | 11 * | 8 * | 8 * | ||
| Almonds | 0.94 | 7.8 | 7.8 | 14.5 | 14.5 | 8.5 | 8.5 | 11.8 | 11.8 |
| Brazil nuts | 1.28 | 10.7 | 10.7 | 19.7 | 19.7 | 11.6 | 11.6 | 16.0 | 16.0 |
| Cashew nuts | 1.73 | 14.4 | 14.4 | 26.6 | 26.6 | 15.7 | 15.7 | 21.6 | 21.6 |
| Hazelnuts | 0.74 | 6.2 | 6.2 | 11.4 | 11.4 | 6.7 | 6.7 | 9.3 | 9.3 |
| Macadamia nuts | 0.39 | 3.3 | 3.3 | 6.0 | 6.0 | 3.5 | 3.5 | 4.9 | 4.9 |
| Peanuts | 0.98 | 8.2 | 8.2 | 15.1 | 15.1 | 8.9 | 8.9 | 12.3 | 12.3 |
| Pecans | 1.36 | 11.3 | 11.3 | 20.9 | 20.9 | 12.4 | 12.4 | 17.0 | 17.0 |
| Pine nuts | 1.94 | 16.2 | 16.2 | 29.8 | 29.8 | 17.6 | 17.6 | 24.3 | 24.3 |
| Pistachios | 0.66 | 5.5 | 5.5 | 10.2 | 10.2 | 6.0 | 6.0 | 8.3 | 8.3 |
| Walnuts | 0.93 | 7.8 | 7.8 | 14.3 | 14.3 | 8.5 | 8.5 | 11.6 | 11.6 |
| Range | 0.39–1.94 | 3.3–16.2 | 3.3–16.2 | 6.0–29.8 | 6.0–29.8 | 3.5–17.6 | 3.5–17.6 | 4.9–24.3 | 4.9–24.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, S.-Y.; Tey, S.L.; Brown, R. Can Nuts Mitigate Malnutrition in Older Adults? A Conceptual Framework. Nutrients 2018, 10, 1448. https://doi.org/10.3390/nu10101448
Tan S-Y, Tey SL, Brown R. Can Nuts Mitigate Malnutrition in Older Adults? A Conceptual Framework. Nutrients. 2018; 10(10):1448. https://doi.org/10.3390/nu10101448
Chicago/Turabian StyleTan, Sze-Yen, Siew Ling Tey, and Rachel Brown. 2018. "Can Nuts Mitigate Malnutrition in Older Adults? A Conceptual Framework" Nutrients 10, no. 10: 1448. https://doi.org/10.3390/nu10101448
APA StyleTan, S.-Y., Tey, S. L., & Brown, R. (2018). Can Nuts Mitigate Malnutrition in Older Adults? A Conceptual Framework. Nutrients, 10(10), 1448. https://doi.org/10.3390/nu10101448







