Reduced Carotenoid and Retinoid Concentrations and Altered Lycopene Isomer Ratio in Plasma of Atopic Dermatitis Patients
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Characterisation of the Study Cohort
3.2. Carotenoid Concentrations and Lycopene Isomers
3.3. Retinoid Concentration and Ratios of Retinoic Acid Isomerization
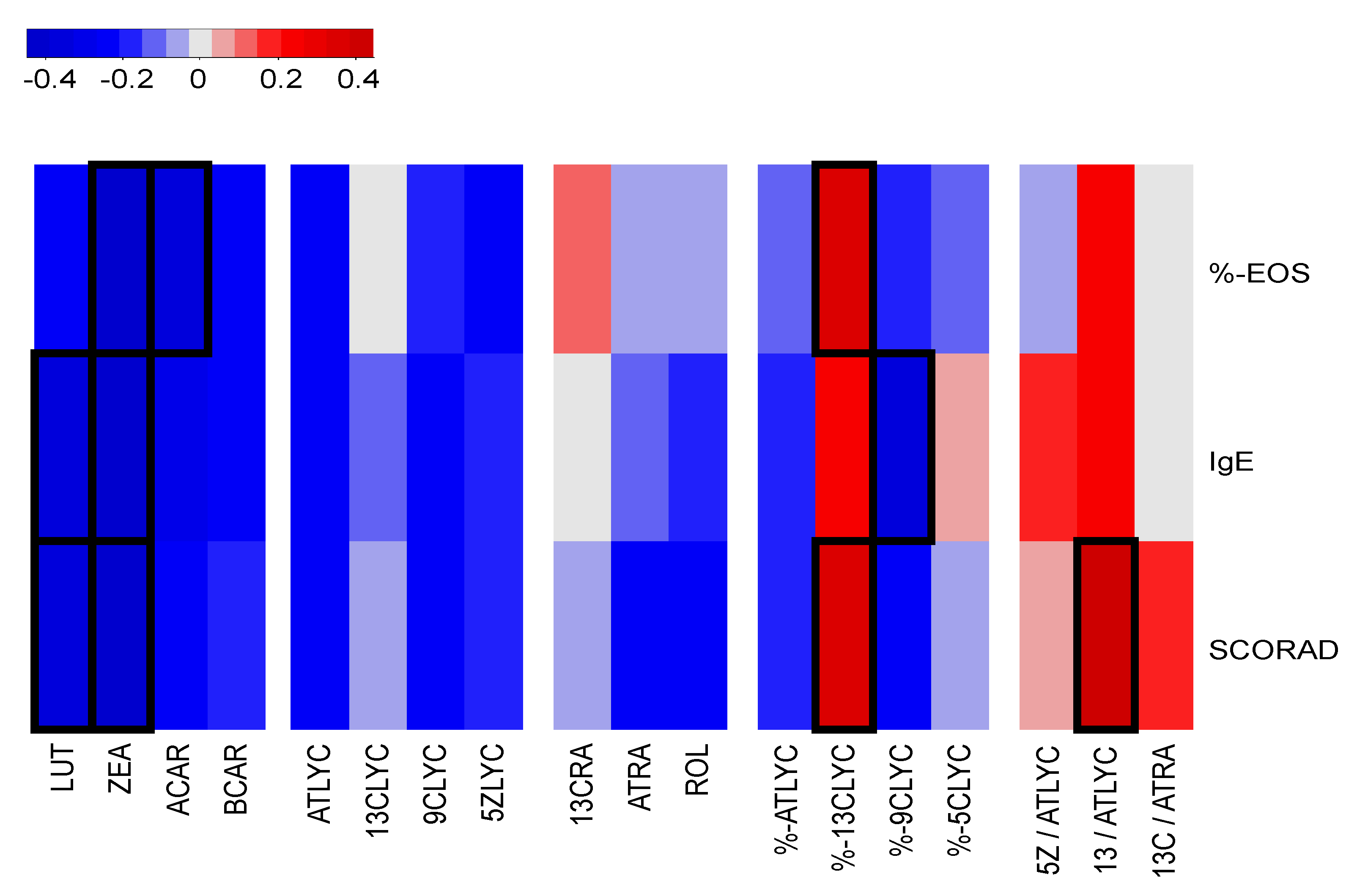
3.4. Correlation Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rühl, R.; Garcia, A.; Schweigert, F.J.; Worm, M. Modulation of cytokine production by low and high retinoid diets in ovalbumin-sensitized mice. Int. J. Vitam. Nutr. Res. 2004, 74, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Gericke, J.; Ittensohn, J.; Mihaly, J.; Alvarez, S.; Alvarez, R.; Töröcsik, D.; de Lera, A.R.; Rühl, R. Regulation of retinoid-mediated signaling involved in skin homeostasis by RAR and RXR agonists/antagonists in mouse skin. PLoS ONE 2013, 8, e642–e643. [Google Scholar] [CrossRef] [PubMed]
- Gericke, J.; Ittensohn, J.; Mihaly, J.; Dubrac, S.; Rühl, R. Allergen-induced dermatitis causes alterations in cutaneous retinoid-mediated signaling in mice. PLoS ONE 2013, 8, e71244. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Planchon, S.; Leclercq, C.C.; Renaut, J.; Mihaly, J.; Beke, G.; Rühl, R. Proteomic responses of carotenoid and retinol administration to Mongolian gerbils. Food Funct. 2018, 9, 3835–3844. [Google Scholar] [CrossRef] [PubMed]
- Rühl, R. Non-pro-vitamin A and pro-vitamin A carotenoids in atopy development. Int. Arch. Allergy Appl. Immunol. 2013, 161, 99–115. [Google Scholar] [CrossRef] [PubMed]
- Rühl, R.; Hanel, A.; Garcia, A.L.; Dahten, A.; Herz, U.; Schweigert, F.J.; Worm, M. Role of vitamin A elimination or supplementation diets during postnatal development on the allergic sensitisation in mice. Mol. Nutr. Food Res. 2007, 51, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Rühl, R.; Taner, C.; Schweigert, F.J.; Wahn, U.; Gruber, C. Serum carotenoids and atopy among children of different ethnic origin living in Germany. Pediatr. Allergy Immunol. 2010, 21, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Stephensen, C.B.; Jiang, X.; Freytag, T. Vitamin A deficiency increases the in vivo development of IL-10-positive Th2 cells and decreases development of Th1 cells in mice. J. Nutr. 2004, 134, 2660–2666. [Google Scholar] [CrossRef] [PubMed]
- Stephensen, C.B.; Rasooly, R.; Jiang, X.; Ceddia, M.A.; Weaver, C.T.; Chandraratna, R.A.; Bucy, R.P. Vitamin A enhances in vitro Th2 development via retinoid X receptor pathway. J. Immunol. 2002, 168, 4495–4503. [Google Scholar] [CrossRef] [PubMed]
- Rühl, R. Effects of dietary retinoids and carotenoids on immune development. Proc. Nutr. Soc. 2007, 66, 458–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mihaly, J.; Gamlieli, A.; Worm, M.; Rühl, R. Decreased retinoid concentration and retinoid signalling pathways in human atopic dermatitis. Exp. Dermatol. 2011, 20, 326–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elias, P.M. The skin barrier as an innate immune element. Semin. Immunopathol. 2007, 29, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Rühl, R.; Bub, A.; Watzl, B. Modulation of plasma all-trans retinoic acid concentrations by the consumption of carotenoid-rich vegetables. Nutrition 2008, 24, 1224–1226. [Google Scholar] [CrossRef] [PubMed]
- Roos, T.C.; Jugert, F.K.; Merk, H.F.; Bickers, D.R. Retinoid metabolism in the skin. Pharmacol. Rev. 1998, 50, 315–333. [Google Scholar] [PubMed]
- Leung, D.Y. New insights into atopic dermatitis: Role of skin barrier and immune dysregulation. Allergol. Int. 2013, 62, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Heise, R.; Mey, J.; Neis, M.M.; Marquardt, Y.; Joussen, S.; Ott, H.; Wiederholt, T.; Kurschat, P.; Megahed, M.; Bickers, D.R.; et al. Skin retinoid concentrations are modulated by CYP26AI expression restricted to basal keratinocytes in normal human skin and differentiated 3D skin models. J. Investig. Dermatol. 2006, 126, 2473–2480. [Google Scholar] [CrossRef] [PubMed]
- Iwata, M.; Eshima, Y.; Kagechika, H. Retinoic acids exert direct effects on T cells to suppress Th1 development and enhance Th2 development via retinoic acid receptors. Int. Immunol. 2003, 15, 1017–1025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spilianakis, C.G.; Lee, G.R.; Flavell, R.A. Twisting the Th1/Th2 immune response via the retinoid X receptor: Lessons from a genetic approach. Eur. J. Immunol. 2005, 35, 3400–3404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mihaly, J.; Gericke, J.; Lucas, R.; de Lera, A.R.; Alvarez, S.; Torocsik, D.; Rühl, R. TSLP expression in the skin is mediated via RARgamma-RXR pathways. Immunobiology 2016, 221, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Mihaly, J.; Gericke, J.; Aydemir, G.; Weiss, K.; Carlsen, H.; Blomhoff, R.; Garcia, J.; Rühl, R. Reduced retinoid signaling in the skin after systemic retinoid-X receptor ligand treatment in mice with potential relevance for skin disorders. Dermatology 2013, 225, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Ruhl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1600685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krinsky, N.I. Possible biologic mechanisms for a protective role of xanthophylls. J. Nutr. 2002, 132, 540S–542S. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Desmarchelier, C.; El, S.N.; Keijer, J.; van Schothorst, E.M.; Rühl, R.; Borel, P. B-Carotene in the Human body—Metabolic acitivation pathways—From Digestion to Tissue Distribution. Proc. Nutr. Soc. 2018, in press. [Google Scholar]
- Boileau, T.W.; Boileau, A.C.; Erdman, J.W.J. Bioavailability of all-trans and cis-isomers of lycopene. Exp. Biol. Med. 2002, 227, 914–919. [Google Scholar] [CrossRef]
- Boileau, T.W.; Clinton, S.K.; Erdman, J.W.J. Tissue lycopene concentrations and isomer patterns are affected by androgen status and dietary lycopene concentration in male F344 rats. J. Nutr. 2000, 130, 1613–1618. [Google Scholar] [CrossRef] [PubMed]
- Stahl, W.; Schwarz, W.; Sundquist, A.R.; Sies, H. Cis-trans isomers of lycopene and beta-carotene in human serum and tissues. Arch. Biochem. Biophys. 1992, 294, 173–177. [Google Scholar] [CrossRef]
- Erdman, J.W.J. How do nutritional and hormonal status modify the bioavailability, uptake, and distribution of different isomers of lycopene? J. Nutr. 2005, 135, 2046S–2047S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aydemir, G.; Kasiri, Y.; Birta, E.; Beke, G.; Garcia, A.L.; Bartok, E.M.; Rühl, R. Lycopene-derived bioactive retinoic acid receptors/retinoid-X receptors-activating metabolites may be relevant for lycopene’s anti-cancer potential. Mol. Nutr. Food Res. 2013, 57, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, G.; Carlsen, H.; Blomhoff, R.; Rühl, R. Lycopene induces Retinoic Acid Receptor transcriptional activation in mice. Mol. Nutr. Food Res. 2012, 56, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, G.; Kasiri, Y.; Bartok, E.M.; Birta, E.; Frohlich, K.; Bohm, V.; Mihaly, J.; Rühl, R. Lycopene supplementation restores vitamin A deficiency in mice and possesses thereby partial pro-vitamin A activity transmitted via RAR-signaling. Mol. Nutr. Food Res. 2016, 60, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Erdman, J.W.J.; Ford, N.A.; Lindshield, B.L. Are the health attributes of lycopene related to its antioxidant function? Arch. Biochem. Biophys. 2009, 483, 229–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biswas, R.; Chakraborti, G.; Mukherjee, K.; Bhattacharjee, D.; Mallick, S.; Biswas, T. Retinol Levels in Serum and Chronic Skin Lesions of Atopic Dermatitis. Indian J. Dermatol. 2018, 63, 251–254. [Google Scholar] [PubMed]
- Stephensen, C.B.; Borowsky, A.D.; Lloyd, K.C. Disruption of Rxra gene in thymocytes and T lymphocytes modestly alters lymphocyte frequencies, proliferation, survival and T helper type 1/type 2 balance. Immunology 2007, 121, 484–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanifin, J.; Rajka, G. Diagnostic features of atopic dermatitis. Acta. Derm. Venereol. 1980, 92, 44–47. [Google Scholar]
- Stalder, J.F.; Taïeb, A.; Atherton, D.J.; Bieber, P.; Bonifazi, E.; Broberg, A.; Calza, A.; Coleman, R.; De Prost, Y.; Stalder, J.F. Severity scoring of atopic dermatitis: The SCORAD index. Consensus Report of the European Task Force on Atopic Dermatitis. Dermatology 1993, 186, 23–31. [Google Scholar]
- Mihaly, J.; Sonntag, D.; Krebiehl, G.; Szegedi, A.; Torocsik, D.; Rühl, R. Steroid concentrations in patients with atopic dermatitis: Reduced plasma dehydroepiandrosterone sulfate and increased cortisone levels. Br. J. Dermatol. 2015, 172, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Mihaly, J.; Marosvolgyi, T.; Szegedi, A.; Koroskenyi, K.; Lucas, R.; Torocsik, D.; Garcia, A.L.; Decsi, T.; Rühl, R. Increased FADS2-Derived n-6 PUFAs and Reduced n-3 PUFAs in Plasma of Atopic Dermatitis Patients. Skin Pharmacol. Physiol. 2014, 27, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Mihaly, J.; Gericke, J.; Torocsik, D.; Gaspar, K.; Szegedi, A.; Rühl, R. Reduced lipoxygenase and cyclooxygenase mediated signaling in PBMC of atopic dermatitis patients. Prostaglandins Other Lipid Mediat. 2013, 107, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Lowe, G.M.; Bilton, R.F.; Davies, I.G.; Ford, T.C.; Billington, D.; Young, A.J. Carotenoid composition and antioxidant potential in subfractions of human low-density lipoprotein. Ann. Clin. Biochem. 1999, 36, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.L.; Carail, M.; Caris-Veyrat, C.; Lowe, G.M. (13Z)- and (9Z)-lycopene isomers are major intermediates in the oxidative degradation of lycopene by cigarette smoke and Sin-1. Free Radic. Res. 2012, 46, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.L.; Carail, M.; Caris-Veyrat, C.; Lowe, G.M. Cigarette smoke and human plasma lycopene depletion. Food Chem. Toxicol. 2004, 48, 2413–2420. [Google Scholar] [CrossRef] [PubMed]
- Rühl, R. Method to determine 4-oxo-retinoic acids, retinoic acids and retinol in serum and cell extracts by liquid chromatography/diode-array detection atmospheric pressure chemical ionisation tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 2497–2504. [Google Scholar] [CrossRef] [PubMed]
- The R Development Core Team. R: A Language and Environment for Statistical Computing. Foundation for Statistical Computing. Available online: http://softlibre.unizar.es/manuales/aplicaciones/r/fullrefman.pdf (accessed on 20 July 2018).
- Hurault, G.; Schram, M.E.; Roekevisch, E.; Spuls, P.I.; Tanaka, R.J. Relationship and probabilistic stratification of EASI and oSCORAD severity scores for atopic dermatitis. Br. J. Dermatol. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Vahlquist, A. What are natural retinoids? Dermatology 1999, 199, 3–11. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, D.N.; Clugston, R.D.; Blaner, W.S. Vitamin A metabolism: An update. Nutrients 2011, 3, 63–103. [Google Scholar] [CrossRef] [PubMed]
- Blomhoff, R.; Blomhoff, H.K. Overview of retinoid metabolism and function. J. Neurobiol. 2006, 66, 606–630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rühl, R.; Krzyzosiak, A.; Niewiadomska-Cimicka, A.; Rochel, N.; Szeles, L.; Vaz, B.; Wietrzych-Schindler, M.; Alvarez, S.; Szklenar, M.; Nagy, L.; et al. 9-cis-13,14-dihydroretinoic acid is an endogenous retinoid acting as RXR ligand in mice. PLoS Genet. 2015, 11, e1005213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Lera, A.R.; Krezel, W.; Rühl, R. An Endogenous Mammalian Retinoid X Receptor Ligand, At Last! Chem. Med. Chem. 2016, 11, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Idres, N.; Marill, J.; Flexor, M.A.; Chabot, G.G. Activation of retinoic acid receptor-dependent transcription by all-trans-retinoic acid metabolites and isomers. J. Biol. Chem. 2002, 277, 31491–31498. [Google Scholar] [CrossRef] [PubMed]
- Kane, M.A.; Folias, A.E.; Wang, C.; Napoli, J.L. Quantitative profiling of endogenous retinoic acid in vivo and in vitro by tandem mass spectrometry. Anal. Chem. 2008, 80, 1702–1708. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Moiseyev, G.; Nikolaeva, O.; Ma, J.X. Identification of the key residues determining the product specificity of isomerohydrolase. Biochemistry 2012, 51, 4217–4225. [Google Scholar] [CrossRef] [PubMed]
- McBee, J.K.; Van Hooser, J.P.; Jang, G.F.; Palczewski, K. Isomerization of 11-cis-retinoids to all-trans-retinoids in vitro and in vivo. J. Biol. Chem. 2001, 276, 48483–48493. [Google Scholar] [CrossRef] [PubMed]
- Redmond, T.M.; Poliakov, E.; Kuo, S.; Chander, P.; Gentleman, S. RPE65, visual cycle retinol isomerase, is not inherently 11-cis-specific: Support for a carbocation mechanism of retinol isomerization. J. Biol. Chem. 2010, 285, 1919–1927. [Google Scholar] [CrossRef] [PubMed]
- Muller, H.; Bub, A.; Watzl, B.; Rechkemmer, G. Plasma concentrations of carotenoids in healthy volunteers after intervention with carotenoid-rich foods. Eur. J. Nutr. 1999, 38, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Brevik, A.; Andersen, L.F.; Karlsen, A.; Trygg, K.U.; Blomhoff, R.; Drevon, C.A. Six carotenoids in plasma used to assess recommended intake of fruits and vegetables in a controlled feeding study. Eur. J. Clin. Nutr. 2004, 58, 1166–1173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conrady, C.D.; Bell, J.P.; Besch, B.M.; Gorusupudi, A.; Farnsworth, K.; Ermakov, I.; Sharifzadeh, M.; Ermakova, M.; Gellermann, W.; Bernstein, P.S. Correlations Between Macular, Skin, and Serum Carotenoids. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3616–3627. [Google Scholar] [CrossRef] [PubMed]
- Devereux, G.; Seaton, A. Diet as a risk factor for atopy and asthma. J. Allergy Clin. Immunol. 2005, 115, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Shegokar, R.; Mitri, K. Carotenoid lutein: A promising candidate for pharmaceutical and nutraceutical applications. J. Diet. Suppl. 2012, 9, 183–210. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.; Frank, E.; Gierhart, D.; Simpson, P.; Frumento, R. Zeaxanthin-based dietary supplement and topical serum improve hydration and reduce wrinkle count in female subjects. J. Cosmet. Dermatol. 2016, 15, e13–e20. [Google Scholar] [CrossRef] [PubMed]
- Grüber, C.; Taner, C.; Mihaly, J.; Matricardi, P.M.; Wahn, U.; Rühl, R. Serum retinoic acid and atopy among children of different ethnic origin living in Germany. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 558–560. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G.; Garg, M.L.; Blake, R.J.; Garcia-Caraballo, S.; Gibson, P.G. Airway and circulating levels of carotenoids in asthma and healthy controls. J. Am. Coll. Nutr. 2005, 24, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Canfield, L.M.; Clandinin, M.T.; Davies, D.P.; Fernandez, M.C.; Jackson, J.; Hawkes, J.; Goldman, W.J.; Pramuk, K.; Reyes, H.; Sablan, B.; et al. Multinational study of major breast milk carotenoids of healthy mothers. Eur. J. Nutr. 2003, 42, 133–141. [Google Scholar] [PubMed]
- Borel, P. Genetic variations involved in interindividual variability in carotenoid status. Mol. Nutr. Food Res. 2011, 56, 228–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Vliet, T.; van Vlissingen, M.F.; van Schaik, F.; van den Berg, H. beta-Carotene absorption and cleavage in rats is affected by the vitamin A concentration of the diet. J. Nutr. 1996, 126, 499–508. [Google Scholar] [CrossRef] [PubMed]
- van Het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Grolier, P.; Duszka, C.; Borel, P.; Alexandre-Gouabau, M.C.; Azais-Braesco, V. In vitro and in vivo inhibition of beta-carotene dioxygenase activity by canthaxanthin in rat intestine. Arch. Biochem. Biophys. 1997, 348, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Shaw, N.; Elholm, M.; Noy, N. Retinoic acid is a high affinity selective ligand for the peroxisome proliferator-activated receptor beta/delta. J. Biol. Chem. 2003, 278, 41589–41592. [Google Scholar] [CrossRef] [PubMed]
- Romanowska, M.; al Yacoub, N.; Seidel, H.; Donandt, S.; Gerken, H.; Phillip, S.; Haritonova, N.; Artuc, M.; Schweiger, S.; Sterry, W.; et al. PPARdelta enhances keratinocyte proliferation in psoriasis and induces heparin-binding EGF-like growth factor. J. Investig. Dermatol. 2008, 128, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Romanowska, M.; Reilly, L.; Palmer, C.N.; Gustafsson, M.C.; Foerster, J. Activation of PPARbeta/delta causes a psoriasis-like skin disease in vivo. PLoS ONE 2010, 5, e9701. [Google Scholar] [CrossRef] [PubMed]
| Healthy Volunteers | AD-Patients | p-Value | |
|---|---|---|---|
| n = 20 | n = 20 | n = 20 | |
| Age in years | 21 ± 0.3 | 20 ± 1.2 | 0.48 |
| Gender | 70% female | 60% female | - |
| SCORAD | 0 ± 0 | 35.2 ± 4.3 | <0.01 |
| Total IgE (KU/L) | 32 ± 0.0 | 2941 ± 1134 | 0.01 |
| EOS % | 2.5 ± 0 | 7.3 ± 1.1 | <0.01 |
| (A) Carotenoid Concentrations | |||
|---|---|---|---|
| in Plasma of Healthy Volunteers and AD-Patients. | |||
| Healthy Volunteers | AD-Patients | p-Value | |
| n = 20 (ng/mL) | n = 20 (ng/mL) | ||
| lutein | 198 ± 14 | 158 ± 12 | 0.02 |
| zeaxanthin | 349 ± 30 | 236 ± 18 | <0.01 |
| α-carotene | 171 ± 21 | 149 ± 24 | 0.24 |
| β-carotene | 492 ± 77 | 394 ± 65 | 0.17 |
| (B) Total Sums and Concentration of Lycopene Isomers | |||
| in Plasma of Healthy Volunteers and AD-Patients. | |||
| lycopene (sum) | 281 ± 30 | 248 ± 35 | 0.24 |
| lycopene (all-trans-) | 126 ± 15 | 107 ± 17 | 0.20 |
| lycopene (13-cis-) | 36 ± 3 | 40 ± 5 | 0.29 |
| lycopene (9-cis-) | 25 ± 3 | 21 ± 3 | 0.17 |
| lycopene (5-cis-) | 94 ± 10 | 80 ± 12 | 0.19 |
| (C) Calculated %-Amounts of Lycopene Isomers | |||
| in Plasma of Healthy Volunteers and AD-Patients. | |||
| n = 20 (in%) | n = 20 (in%) | ||
| lycopene (all-trans-) | 44.1 ± 0.9 | 41.8 ± 1.3 | 0.08 |
| lycopene (13-cis-) | 13.3 ± 0.4 | 17.2 ± 1.7 | 0.01 |
| lycopene (9-cis-) | 8.8 ± 0.2 | 8.6 ± 0.3 | 0.24 |
| lycopene (5-cis-) | 33.8 ± 0.8 | 32.4 ± 1.2 | 0.16 |
| (D) Calculated Ratios of Selected Lycopene Isomers in Plasma of Healthy Volunteers and AD-Patients. | |||
| n = 20 (Ratio) | n = 20 (Ratio) | ||
| 13-cis-/all-trans-lycopene | 0.31 ± 0.01 | 0.45 ± 0.07 | 0.03 |
| 5-cis-/all-trans-lycopene | 0.78 ± 0.03 | 0.79 ± 0.04 | 0.40 |
| (A) Retinoic Acid and Retinol Concentrations in Human Plasma from Healthy Volunteers as well as AD-Patients. | |||
|---|---|---|---|
| Healthy Volunteers | AD-Patients | p-Value | |
| n = 20 (ng/mL) | n = 20 (ng/mL) | ||
| ATRA | 1.1 ± 0.1 | 0.7 ± 0.1 | 0.04 |
| 13CRA | 1.2 ± 0.1 | 1.0 ± 0.1 | 0.17 |
| ROL | 216 ± 20 | 167 ± 17 | 0.04 |
| (B) Ratio of the Plasma Levels of Retinoic Acid Isomers 13CRA/ATRA. | |||
| n = 20 (Ratio) | n = 20 (Ratio) | ||
| 13CRA/ATRA | 1.4 ± 0.2 | 2.6 ± 2.6 | 0.03 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucas, R.; Mihály, J.; Lowe, G.M.; Graham, D.L.; Szklenar, M.; Szegedi, A.; Töröcsik, D.; Rühl, R. Reduced Carotenoid and Retinoid Concentrations and Altered Lycopene Isomer Ratio in Plasma of Atopic Dermatitis Patients. Nutrients 2018, 10, 1390. https://doi.org/10.3390/nu10101390
Lucas R, Mihály J, Lowe GM, Graham DL, Szklenar M, Szegedi A, Töröcsik D, Rühl R. Reduced Carotenoid and Retinoid Concentrations and Altered Lycopene Isomer Ratio in Plasma of Atopic Dermatitis Patients. Nutrients. 2018; 10(10):1390. https://doi.org/10.3390/nu10101390
Chicago/Turabian StyleLucas, Renata, Johanna Mihály, Gordon M. Lowe, Daniel L. Graham, Monika Szklenar, Andrea Szegedi, Daniel Töröcsik, and Ralph Rühl. 2018. "Reduced Carotenoid and Retinoid Concentrations and Altered Lycopene Isomer Ratio in Plasma of Atopic Dermatitis Patients" Nutrients 10, no. 10: 1390. https://doi.org/10.3390/nu10101390




