Vitamin E (α- and γ-Tocopherol) Levels in the Community: Distribution, Clinical and Biochemical Correlates, and Association with Dietary Patterns
Abstract
1. Introduction
2. Methods
2.1. Study Sample and Design
2.2. Clinical Examination and Definitions
2.3. Assessment of Dietary Variables
2.4. Laboratory Analyses
2.5. Statistical Analyses
2.6. Correlates of Circulating Vitamin E Biomarkers
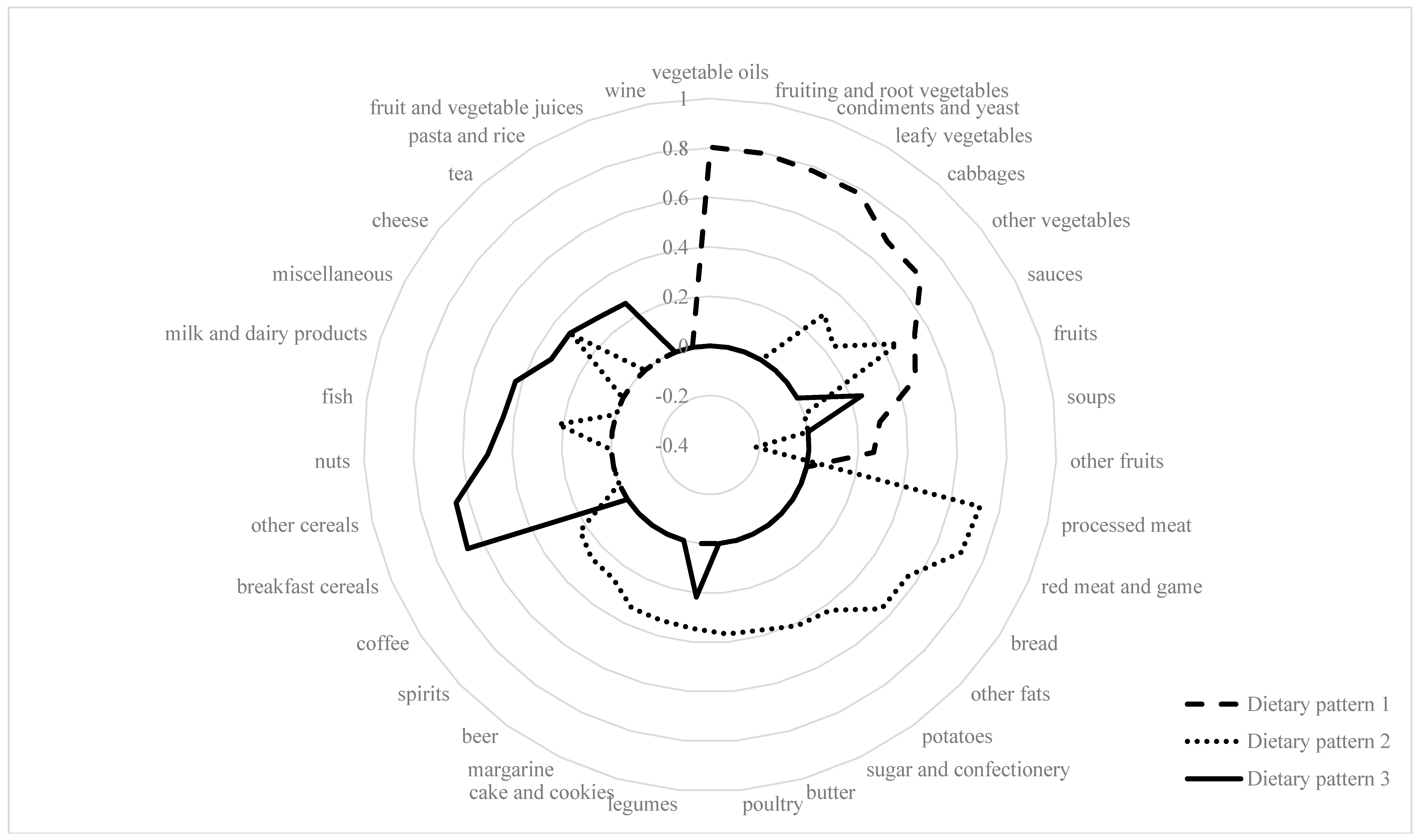
2.7. Dietary Pattern Analyses
3. Results
3.1. Correlates of Vitamin E Biomarkers
3.2. Dietary Pattern Analyses
4. Discussion
4.1. Principal Findings
4.2. In the Context of the Published Literature
Dietary Vitamin E Intake and Distribution of Circulating Vitamin E Levels in the Population
4.3. Correlates of Vitamin E Biomarkers
4.4. Lack of Association between Estimated Dietary α-Tocopherol Intake and Circulating Vitamin E Levels
4.5. Association of Dietary Patterns with Dietary Vitamin E Intake and Circulating Vitamin E Levels
4.6. Strength and Limitations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflict of Interest
References
- Sauberlich, H.E. Vitamin E (Tocopherols). In Laboratory Tests for the Assessment of Nutritional Status, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1999; pp. 249–266. [Google Scholar]
- Jiang, Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US). Panel on Dietary Antioxidants and Related Compounds. In Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; Hardcopy Version at National Academies Press: Washington, DC, USA, 2000; pp. 186–283. [Google Scholar]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Ohrvall, M.; Sundlof, G.; Vessby, B. Gamma, but not alpha, tocopherol levels in serum are reduced in coronary heart disease patients. J. Intern. Med. 1996, 239, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Wright, M.E.; Lawson, K.A.; Weinstein, S.J.; Pietinen, P.; Taylor, P.R.; Virtamo, J.; Albanes, D. Higher baseline serum concentrations of vitamin E are associated with lower total and cause-specific mortality in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Am. J. Clin. Nutr. 2006, 84, 1200–1207. [Google Scholar] [PubMed]
- Stampfer, M.J.; Hennekens, C.H.; Manson, J.E.; Colditz, G.A.; Rosner, B.; Willett, W.C. Vitamin E consumption and the risk of coronary disease in women. N. Engl. J. Med. 1993, 328, 1444–1449. [Google Scholar] [CrossRef] [PubMed]
- Rimm, E.B.; Stampfer, M.J.; Ascherio, A.; Giovannucci, E.; Colditz, G.A.; Willett, W.C. Vitamin E consumption and the risk of coronary heart disease in men. N. Engl. J. Med. 1993, 328, 1450–1456. [Google Scholar] [CrossRef] [PubMed]
- Riemersma, R.A.; Wood, D.A.; Macintyre, C.C.; Elton, R.A.; Gey, K.F.; Oliver, M.F. Risk of angina pectoris and plasma concentrations of vitamins A, C, and E and carotene. Lancet 1991, 337, 1–5. [Google Scholar] [CrossRef]
- Dong, Y.; Liu, Y.; Shu, Y.; Chen, X.; Hu, J.; Zheng, R.; Ma, D.; Yang, C.; Guan, X. Link between risk of colorectal cancer and serum vitamin E levels: A meta-analysis of case-control studies. Medicine 2017, 96, e7470. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Appel, L.J. Supplementation of diets with alpha-tocopherol reduces serum concentrations of gamma- and delta-tocopherol in humans. J. Nutr. 2003, 133, 3137–3140. [Google Scholar] [PubMed]
- Raederstorff, D.; Wyss, A.; Calder, P.C.; Weber, P.; Eggersdorfer, M. Vitamin E function and requirements in relation to PUFA. Br. J. Nutr. 2015, 114, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- German Nutrition Society (2017). Vitamin E (Tocopherole). Available online: https://www.dge.de/wissenschaft/referenzwerte/vitamin-e/ (accessed on 18 June 2017).
- Max Rubner-Institut. Nationale Verzehrsstudie II. Ergebnisbericht Teil 2; MRI: Karlsruhe, Germany, 2008. [Google Scholar]
- Borel, P.; Preveraud, D.; Desmarchelier, C. Bioavailability of vitamin E in humans: An update. Nutr. Rev. 2013, 71, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Wagner, K.H.; Kamal-Eldin, A.; Elmadfa, I. Gamma-tocopherol—An underestimated vitamin? Ann. Nutr. Metab. 2004, 48, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Martin, A.; Lin, H.; Bermudez, O.I.; Tucker, K.L. alpha-Tocopherol intake and plasma concentration of Hispanic and non-Hispanic white elders is associated with dietary intake pattern. J. Nutr. 2006, 136, 2574–2579. [Google Scholar] [PubMed]
- Talegawkar, S.A.; Johnson, E.J.; Carithers, T.C.; Taylor, H.A., Jr.; Bogle, M.L.; Tucker, K.L. Serum carotenoid and tocopherol concentrations vary by dietary pattern among African Americans. J. Am. Diet. Assoc. 2008, 108, 2013–2020. [Google Scholar] [CrossRef] [PubMed]
- Nothlings, U.; Krawczak, M. PopGen. A population-based biobank with prospective follow-up of a control group. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2012, 55, 831–835. [Google Scholar] [PubMed]
- Nothlings, U.; Hoffmann, K.; Bergmann, M.M.; Boeing, H. Fitting portion sizes in a self-administered food frequency questionnaire. J. Nutr. 2007, 137, 2781–2786. [Google Scholar] [PubMed]
- Barbaresko, J.; Siegert, S.; Koch, M.; Aits, I.; Lieb, W.; Nikolaus, S.; Laudes, M.; Jacobs, G.; Nothlings, U. Comparison of two exploratory dietary patterns in association with the metabolic syndrome in a Northern German population. Br. J. Nutr. 2014, 112, 1364–1372. [Google Scholar] [CrossRef] [PubMed]
- Haftenberger, M.; Schuit, A.J.; Tormo, M.J.; Boeing, H.; Wareham, N.; Bueno-de-Mesquita, H.B.; Kumle, M.; Hjartaker, A.; Chirlaque, M.D.; Ardanaz, E.; et al. Physical activity of subjects aged 50–64 years involved in the European Prospective Investigation into Cancer and Nutrition (EPIC). Public Health Nutr. 2002, 5, 1163–1176. [Google Scholar] [CrossRef] [PubMed]
- Friedenreich, C.; Norat, T.; Steindorf, K.; Boutron-Ruault, M.C.; Pischon, T.; Mazuir, M.; Clavel-Chapelon, F.; Linseisen, J.; Boeing, H.; Bergman, M.; et al. Physical activity and risk of colon and rectal cancers: The European prospective investigation into cancer and nutrition. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 2398–2407. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 Compendium of Physical Activities: A second update of codes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef] [PubMed]
- Dehne, L.I.; Klemm, C.; Henseler, G.; Hermann-Kunz, E. The German Food Code and Nutrient Data Base (BLS II.2). Eur. J. Epidemiol. 1999, 15, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Kroger, J.; Ferrari, P.; Jenab, M.; Bamia, C.; Touvier, M.; Bueno-de-Mesquita, H.B.; Fahey, M.T.; Benetou, V.; Schulz, M.; Wirfalt, E.; et al. Specific food group combinations explaining the variation in intakes of nutrients and other important food components in the European Prospective Investigation into Cancer and Nutrition: An application of the reduced rank regression method. Eur. J. Clin. Nutr. 2009, 63 (Suppl. S4), S263–S274. [Google Scholar] [CrossRef] [PubMed]
- Augustin, K.; Blank, R.; Boesch-Saadatmandi, C.; Frank, J.; Wolffram, S.; Rimbach, G. Dietary green tea polyphenols do not affect vitamin E status, antioxidant capacity and meat quality of growing pigs. J. Anim. Physiol. Anim. Nutr. 2008, 92, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Thurnham, D.I.; Davies, J.A.; Crump, B.J.; Situnayake, R.D.; Davis, M. The use of different lipids to express serum tocopherol: Lipid ratios for the measurement of vitamin E status. Ann. Clin. Biochem. 1986, 23 Pt 5, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K.; Schulze, M.B.; Schienkiewitz, A.; Nothlings, U.; Boeing, H. Application of a new statistical method to derive dietary patterns in nutritional epidemiology. Am. J. Epidemiol. 2004, 159, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, Z.C. Vitamin E Intake and Cardiovascular Diseases in the EPIC-Potsdam Study. Ph.D. Thesis, Technical University of Berlin, Berlin, Germany, 2011. [Google Scholar]
- Jenab, M.; Salvini, S.; van Gils, C.H.; Brustad, M.; Shakya-Shrestha, S.; Buijsse, B.; Verhagen, H.; Touvier, M.; Biessy, C.; Wallstrom, P.; et al. Dietary intakes of retinol, beta-carotene, vitamin D and vitamin E in the European Prospective Investigation into Cancer and Nutrition cohort. Eur. J. Clin. Nutr. 2009, 63 (Suppl. S4), S150–S178. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wilde, P.E.; Lichtenstein, A.H.; Bermudez, O.I.; Tucker, K.L. The maximal amount of dietary alpha-tocopherol intake in U.S. adults (NHANES 2001–2002). J. Nutr. 2006, 136, 1021–1026. [Google Scholar] [PubMed]
- Max Rubner-Institut. Nationale Verzehrsstudie II. Lebensmittelverzehr und Nährstoffzufuhr auf Basis von 24h-Recalls; MRI: Karlsruhe, Germany, 2013. [Google Scholar]
- Wolters, M.; Hermann, S.; Golf, S.; Katz, N.; Hahn, A. Selenium and antioxidant vitamin status of elderly German women. Eur. J. Clin. Nutr. 2006, 60, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Kompauer, I.; Heinrich, J.; Wolfram, G.; Linseisen, J. Association of carotenoids, tocopherols and vitamin C in plasma with allergic rhinitis and allergic sensitisation in adults. Public Health Nutr. 2006, 9, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Schleicher, R.L.; Mokdad, A.H.; Ajani, U.A.; Liu, S. Distribution of serum concentrations of alpha-tocopherol and gamma-tocopherol in the US population. Am. J. Clin. Nutr. 2006, 84, 375–383. [Google Scholar] [PubMed]
- Péter, S.; Friedel, A.; Roos, F.F.; Wyss, A.; Eggersdorfer, M.; Hoffmann, K.; Weber, P. A Systematic Review of Global Alpha-Tocopherol Status as Assessed by Nutritional Intake Levels and Blood Serum Concentrations. Int. J. Vitam. Nutr. Res. 2016, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Stuetz, W.; Weber, D.; Dolle, M.E.; Jansen, E.; Grubeck-Loebenstein, B.; Fiegl, S.; Toussaint, O.; Bernhardt, J.; Gonos, E.S.; Franceschi, C.; et al. Plasma Carotenoids, Tocopherols, and Retinol in the Age-Stratified (35–74 Years) General Population: A Cross-Sectional Study in Six European Countries. Nutrients 2016, 8, 614. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Christen, S.; Shigenaga, M.K.; Ames, B.N. gamma-tocopherol, the major form of vitamin E in the US diet, deserves more attention. Am. J. Clin. Nutr. 2001, 74, 714–722. [Google Scholar] [PubMed]
- Millen, A.E.; Dodd, K.W.; Subar, A.F. Use of vitamin, mineral, nonvitamin, and nonmineral supplements in the United States: The 1987, 1992, and 2000 National Health Interview Survey results. J. Am. Diet. Assoc. 2004, 104, 942–950. [Google Scholar] [CrossRef] [PubMed]
- White, E.; Kristal, A.R.; Shikany, J.M.; Wilson, A.C.; Chen, C.; Mares-Perlman, J.A.; Masaki, K.H.; Caan, B.J. Correlates of serum alpha- and gamma-tocopherol in the Women’s Health Initiative. Ann. Epidemiol. 2001, 11, 136–144. [Google Scholar] [CrossRef]
- McBurney, M.I.; Yu, E.A.; Ciappio, E.D.; Bird, J.K.; Eggersdorfer, M.; Mehta, S. Suboptimal Serum alpha-Tocopherol Concentrations Observed among Younger Adults and Those Depending Exclusively upon Food Sources, NHANES 2003-20061-3. PLoS ONE 2015, 10, e0135510. [Google Scholar] [CrossRef] [PubMed]
- Handelman, G.J.; Machlin, L.J.; Fitch, K.; Weiter, J.J.; Dratz, E.A. Oral alpha-tocopherol supplements decrease plasma gamma-tocopherol levels in humans. J. Nutr. 1985, 115, 807–813. [Google Scholar] [PubMed]
- Gaedicke, S.; Zhang, X.; Huebbe, P.; Boesch-Saadatmandi, C.; Lou, Y.; Wiswedel, I.; Gardemann, A.; Frank, J.; Rimbach, G. Dietary vitamin E, brain redox status and expression of Alzheimer’s disease-relevant genes in rats. Br. J. Nutr. 2009, 102, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G. Mechanisms for the prevention of vitamin E excess. J. Lipid Res. 2013, 54, 2295–2306. [Google Scholar] [CrossRef] [PubMed]
- Hosomi, A.; Arita, M.; Sato, Y.; Kiyose, C.; Ueda, T.; Igarashi, O.; Arai, H.; Inoue, K. Affinity for alpha-tocopherol transfer protein as a determinant of the biological activities of vitamin E analogs. FEBS Lett. 1997, 409, 105–108. [Google Scholar] [CrossRef]
- Traber, M.G. Vitamin E regulatory mechanisms. Annu. Rev. Nutr. 2007, 27, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Sontag, T.J.; Parker, R.S. Cytochrome P450 omega-hydroxylase pathway of tocopherol catabolism. Novel mechanism of regulation of vitamin E status. J. Biol. Chem. 2002, 277, 25290–25296. [Google Scholar] [CrossRef] [PubMed]
- Boeing, H.; Bohlscheid-Thomas, S.; Voss, S.; Schneeweiss, S.; Wahrendorf, J. The relative validity of vitamin intakes derived from a food frequency questionnaire compared to 24-h recalls and biological measurements: Results from the EPIC pilot study in Germany. European Prospective Investigation into Cancer and Nutrition. Int. J. Epidemiol. 1997, 26 (Suppl. S1), S82–S90. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.A.; Marks, G.C.; Gaffney, P.; Williams, G.; Green, A. Validation of a food-frequency questionnaire assessment of carotenoid and vitamin E intake using weighed food records and plasma biomarkers: The method of triads model. Eur. J. Clin. Nutr. 2005, 59, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Stampfer, M.J.; Colditz, G.A.; Rimm, E.B.; Litin, L.; Willett, W.C. Correlations of vitamin A and E intakes with the plasma concentrations of carotenoids and tocopherols among American men and women. J. Nutr. 1992, 122, 1792–1801. [Google Scholar] [PubMed]
- Stryker, W.S.; Kaplan, L.A.; Stein, E.A.; Stampfer, M.J.; Sober, A.; Willett, W.C. The relation of diet, cigarette smoking, and alcohol consumption to plasma beta-carotene and alpha-tocopherol levels. Am. J. Epidemiol. 1988, 127, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Kardinaal, A.F.; van ’t Veer, P.; Brants, H.A.; van den Berg, H.; van Schoonhoven, J.; Hermus, R.J. Relations between antioxidant vitamins in adipose tissue, plasma, and diet. Am. J. Epidemiol. 1995, 141, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.F.; Solvoll, K.; Johansson, L.R.; Salminen, I.; Aro, A.; Drevon, C.A. Evaluation of a food frequency questionnaire with weighed records, fatty acids, and alpha-tocopherol in adipose tissue and serum. Am. J. Epidemiol. 1999, 150, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Schmolz, L.; Birringer, M.; Lorkowski, S.; Wallert, M. Complexity of vitamin E metabolism. World J. Biol. Chem. 2016, 7, 14–43. [Google Scholar] [CrossRef] [PubMed]
- Galli, F.; Azzi, A.; Birringer, M.; Cook-Mills, J.M.; Eggersdorfer, M.; Frank, J.; Cruciani, G.; Lorkowski, S.; Ozer, N.K. Vitamin E: Emerging aspects and new directions. Free Radic. Biol. Med. 2017, 102, 16–36. [Google Scholar] [CrossRef] [PubMed]
- Mah, E.; Sapper, T.N.; Chitchumroonchokchai, C.; Failla, M.L.; Schill, K.E.; Clinton, S.K.; Bobe, G.; Traber, M.G.; Bruno, R.S. alpha-Tocopherol bioavailability is lower in adults with metabolic syndrome regardless of dairy fat co-ingestion: A randomized, double-blind, crossover trial. Am. J. Clin. Nutr. 2015, 102, 1070–1080. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Mah, E.; Leonard, S.W.; Bobe, G.; Bruno, R.S. Metabolic syndrome increases dietary alpha-tocopherol requirements as assessed using urinary and plasma vitamin E catabolites: A double-blind, crossover clinical trial. Am. J. Clin. Nutr. 2017, 105, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Chuang, J.C.; Matel, H.D.; Nambiar, K.P.; Kim, S.H.; Fadel, J.G.; Holstege, D.M.; Clifford, A.J. Quantitation of [5-14CH3]-(2R, 4′R, 8′R)-alpha-tocopherol in humans. J. Nutr. 2011, 141, 1482–1488. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | |
| Men, % | 59.4 |
| Age, years | 61.2 (11.6) |
| Body mass index, kg/m2 | 27.2 (4.5) |
| Weight, kg | 80.5 (15.7) |
| Hip circumference, cm | 102.3 (8.8) |
| Waist circumference, cm | 96.2 (13.0) |
| Systolic blood pressure, mmHg | 139.6 (18.2) |
| Diastolic blood pressure, mmHg | 85.0 (8.9) |
| Prevalent hypertension, % | 69.0 |
| Current smokers, % | 10.9 |
| High education (≥11 years), % | 35.9 |
| Prevalent diabetes, % | 10.8 |
| Vitamin E supplementation, % | 7.5 |
| Physical activity, MET-hour/week | 90.0 (58.3, 131.6) |
| Alcohol consumption, g/day | 9.6 (3.7, 18.7) |
| Dietary α-tocopherol intake (FFQ), mg/day | 11.6 (9.7, 13.9) |
| Biochemical features | |
| α-tocopherol, μmol/L | 31.5 (27.27, 37.03) |
| α-tocopherol > 30 μmol/L, % * | 57.6 |
| α-tocopherol/cholesterol ratio, μmol/mmol | 5.53 (4.88, 6.33) |
| γ-tocopherol, μmol/L | 1.35 (0.99, 1.79) |
| γ-tocopherol/cholesterol ratio, μmol/mmol | 0.24 (0.18, 0.31) |
| HbA1c, % | 5.60 (5.40, 5.90) |
| C-reactive protein, mg/dL | 1.20 (0.45, 2.50) |
| HDL-cholesterol, mg/dL | 65.81 (18.61) |
| LDL-cholesterol, mg/dL | 131.36 (34.07) |
| Total cholesterol, mg/dL | 223.42 (41.38) |
| Triglycerides, mg/dL | 106.0 (76.0, 139.0) |
| (a) α-Tocopherol * | β Estimate | SE | p Value |
| Age | −0.0006 | 0.0007 | 0.358 |
| Sex | −0.0108 | 0.0184 | 0.556 |
| Triglycerides | 0.0015 | 0.0001 | <0.0001 |
| HDL-cholesterol | 0.0033 | 0.0005 | <0.0001 |
| LDL-cholesterol | 0.0027 | 0.0002 | <0.0001 |
| Vitamin E supplementation | 0.0863 | 0.0307 | 0.005 |
| R2 = 0.359 | |||
| (b) α-Tocopherol/Cholesterol Ratio * | |||
| Age | −0.0014 | 0.0007 | 0.040 |
| Sex | −0.0096 | 0.0187 | 0.606 |
| Triglycerides | 0.0009 | 0.0001 | <0.0001 |
| HDL-cholesterol | −0.0013 | 0.0005 | 0.015 |
| LDL-cholesterol | −0.0021 | 0.0002 | <0.0001 |
| Vitamin E supplementation | 0.0777 | 0.0311 | 0.013 |
| R2 = 0.197 | |||
| (c) γ-Tocopherol * | |||
| Age | −0.0011 | 0.1562 | 0.474 |
| Sex | −0.0681 | 0.0016 | 0.108 |
| Triglycerides | 0.0019 | 0.0423 | <0.0001 |
| HDL-cholesterol | 0.0031 | 0.0003 | 0.011 |
| LDL-cholesterol | 0.0018 | 0.0012 | 0.001 |
| Vitamin E supplementation | −0.3170 | 0.0006 | <0.0001 |
| R2 = 0.127 | |||
| (d) γ-Tocopherol/Cholesterol Ratio * | |||
| Age | −0.0020 | 0.0016 | 0.201 |
| Sex | −0.0454 | 0.0381 | 0.235 |
| Triglycerides | 0.0015 | 0.0003 | <0.0001 |
| LDL-cholesterol | −0.0030 | 0.0006 | <0.0001 |
| Vitamin E supplementation | −0.3225 | 0.0707 | <0.0001 |
| R2 = 0.106 |
| α-Tocopherol | γ-Tocopherol | |||
|---|---|---|---|---|
| rho | 95% CI | rho | 95% CI | |
| Leafy vegetables | 0.03 | −0.05, 0.10 | 0.01 | −0.07, 0.08 |
| Fruiting and root vegetables | 0.01 | −0.06, 0.09 | 0.04 | −0.04, 0.12 |
| Cabbages | −0.04 | −0.12, 0.04 | 0.05 | −0.02, 0.13 |
| Other vegetables | −0.04 | −0.12, 0.04 | 0.03 | −0.04, 0.11 |
| Legumes | 0.02 | −0.06, 0.10 | 0.05 | −0.03, 0.12 |
| Nuts | 0.08 | −0.002, 0.15 | 0.07 | −0.01, 0.15 |
| Other fruits | 0.11 | 0.04, 0.20 | 0.12 | 0.04, 0.19 |
| Breakfast cereals | 0.06 | −0.001, 0.14 | 0.03 | −0.05, 0.11 |
| Other cereals | 0.03 | −0.04, 0.11 | 0.01 | −0.07, 0.09 |
| Margarine | 0.01 | −0.07, 0.08 | 0.02 | −0.06, 0.10 |
| Vegetables oils | 0.06 | −0.07, 0.14 | 0.02 | −0.06, 0.10 |
| Dietary α-tocopherol intake (FFQ) | 0.01 | −0.07, 0.09 | 0.005 | −0.07, 0.08 |
| Dietary α-tocopherol intake (FFQ) * | 0.01 | −0.07, 0.09 | 0.01 | −0.07, 0.09 |
| α-Tocopherol | |||||||||
| Overall (n = 641) | Dietary Pattern 1 | Dietary Pattern 2 | Dietary Pattern 3 | ||||||
| β Estimate | SE | p Value | β Estimate | SE | p Value | β Estimate | SE | p Value | |
| α-tocopherol, µmol/L *,† | −0.007 | 0.008 | 0.415 | −0.026 | 0.015 | 0.089 | 0.015 | 0.008 | 0.077 |
| α-tocopherol/cholesterol ratio, µmol/mmol *,† | −0.006 | 0.009 | 0.508 | −0.020 | 0.016 | 0.207 | 0.015 | 0.009 | 0.087 |
| Dietary α-tocopherol intake (FFQ), mg/day † | 0.72 ‡ | 0.68, 0.75 | <0.0001 | −0.18 ‡ | −0.25, −0.10 | 0.002 | 0.32 ‡ | 0.24, 0.39 | <0.0001 |
| Non vitamin E supplement users (n = 593) | |||||||||
| α-tocopherol, µmol/L *,§ | −0.003 | 0.009 | 0.712 | −0.032 | 0.016 | 0.047 | 0.015 | 0.009 | 0.097 |
| α-tocopherol/cholesterol ratio, µmol/mmol *,§ | −0.002 | 0.009 | 0.790 | −0.025 | 0.016 | 0.135 | 0.014 | 0.009 | 0.112 |
| Dietary α-tocopherol intake (FFQ), mg/day § | 0.71 ‡ | 0.67, 0.75 | <0.0001 | −0.16 ‡ | −0.24, −0.08 | 0.0001 | 0.34 ‡ | 0.27, 0.41 | <0.0001 |
| γ-Tocopherol | |||||||||
| Overall (n = 641) | |||||||||
| γ-tocopherol, µmol/L *,† | 0.006 | 0.019 | 0.759 | −0.013 | 0.036 | 0.715 | 0.027 | 0.019 | 0.165 |
| γ-tocopherol/cholesterol ratio, µmol/mmol *,|| | 0.007 | 0.019 | 0.715 | −0.004 | 0.036 | 0.920 | 0.024 | 0.019 | 0.216 |
| Non vitamin E supplement users (n = 593) | |||||||||
| γ-tocopherol, µmol/L *,§ | 0.004 | 0.020 | 0.844 | 0.004 | 0.036 | 0.906 | 0.022 | 0.019 | 0.262 |
| γ-tocopherol/cholesterol ratio, µmol/mmol *,¶ | 0.005 | 0.020 | 0.790 | 0.014 | 0.036 | 0.695 | 0.018 | 0.019 | 0.352 |
| α-Tocopherol | ||||
| Overall (n = 641) | β Estimate | SE | p Value | R2 |
| α-tocopherol, µmol/L *,† | −0.002 | 0.003 | 0.475 | 0.359 |
| α-tocopherol/cholesterol ratio, µmol/mmol *,† | −0.001 | 0.004 | 0.683 | 0.198 |
| Dietary α-tocopherol intake (FFQ), mg/day † | 0.51 ‡ | 0.45, 0.56 | <0.0001 | |
| Non vitamin E supplement users (n = 593) | ||||
| α-tocopherol, µmol/L *,§ | −0.001 | 0.004 | 0.749 | 0.355 |
| α-tocopherol/cholesterol ratio, µmol/mmol *,§ | −0.0002 | 0.004 | 0.947 | 0.179 |
| Dietary α-tocopherol intake (FFQ), mg/day § | 0.49 ‡ | 0.42, 0.55 | <0.0001 | |
| γ-Tocopherol | ||||
| Overall (n = 641) | ||||
| γ-tocopherol, µmol/L *,† | 0.006 | 0.008 | 0.431 | 0.100 |
| γ-tocopherol/cholesterol ratio, µmol/mmol *,|| | 0.008 | 0.008 | 0.346 | 0.080 |
| Non vitamin E supplement users (n = 593) | ||||
| γ-tocopherol, µmol/L *,§ | 0.005 | 0.008 | 0.549 | 0.107 |
| γ-tocopherol/cholesterol ratio, µmol/mmol *,¶ | 0.007 | 0.008 | 0.412 | 0.084 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waniek, S.; Di Giuseppe, R.; Esatbeyoglu, T.; Plachta-Danielzik, S.; Ratjen, I.; Jacobs, G.; Nöthlings, U.; Koch, M.; Schlesinger, S.; Rimbach, G.; et al. Vitamin E (α- and γ-Tocopherol) Levels in the Community: Distribution, Clinical and Biochemical Correlates, and Association with Dietary Patterns. Nutrients 2018, 10, 3. https://doi.org/10.3390/nu10010003
Waniek S, Di Giuseppe R, Esatbeyoglu T, Plachta-Danielzik S, Ratjen I, Jacobs G, Nöthlings U, Koch M, Schlesinger S, Rimbach G, et al. Vitamin E (α- and γ-Tocopherol) Levels in the Community: Distribution, Clinical and Biochemical Correlates, and Association with Dietary Patterns. Nutrients. 2018; 10(1):3. https://doi.org/10.3390/nu10010003
Chicago/Turabian StyleWaniek, Sabina, Romina Di Giuseppe, Tuba Esatbeyoglu, Sandra Plachta-Danielzik, Ilka Ratjen, Gunnar Jacobs, Ute Nöthlings, Manja Koch, Sabrina Schlesinger, Gerald Rimbach, and et al. 2018. "Vitamin E (α- and γ-Tocopherol) Levels in the Community: Distribution, Clinical and Biochemical Correlates, and Association with Dietary Patterns" Nutrients 10, no. 1: 3. https://doi.org/10.3390/nu10010003
APA StyleWaniek, S., Di Giuseppe, R., Esatbeyoglu, T., Plachta-Danielzik, S., Ratjen, I., Jacobs, G., Nöthlings, U., Koch, M., Schlesinger, S., Rimbach, G., & Lieb, W. (2018). Vitamin E (α- and γ-Tocopherol) Levels in the Community: Distribution, Clinical and Biochemical Correlates, and Association with Dietary Patterns. Nutrients, 10(1), 3. https://doi.org/10.3390/nu10010003





