Global Trends in Exposure to Light Pollution in Natural Terrestrial Ecosystems
Abstract
:1. Introduction
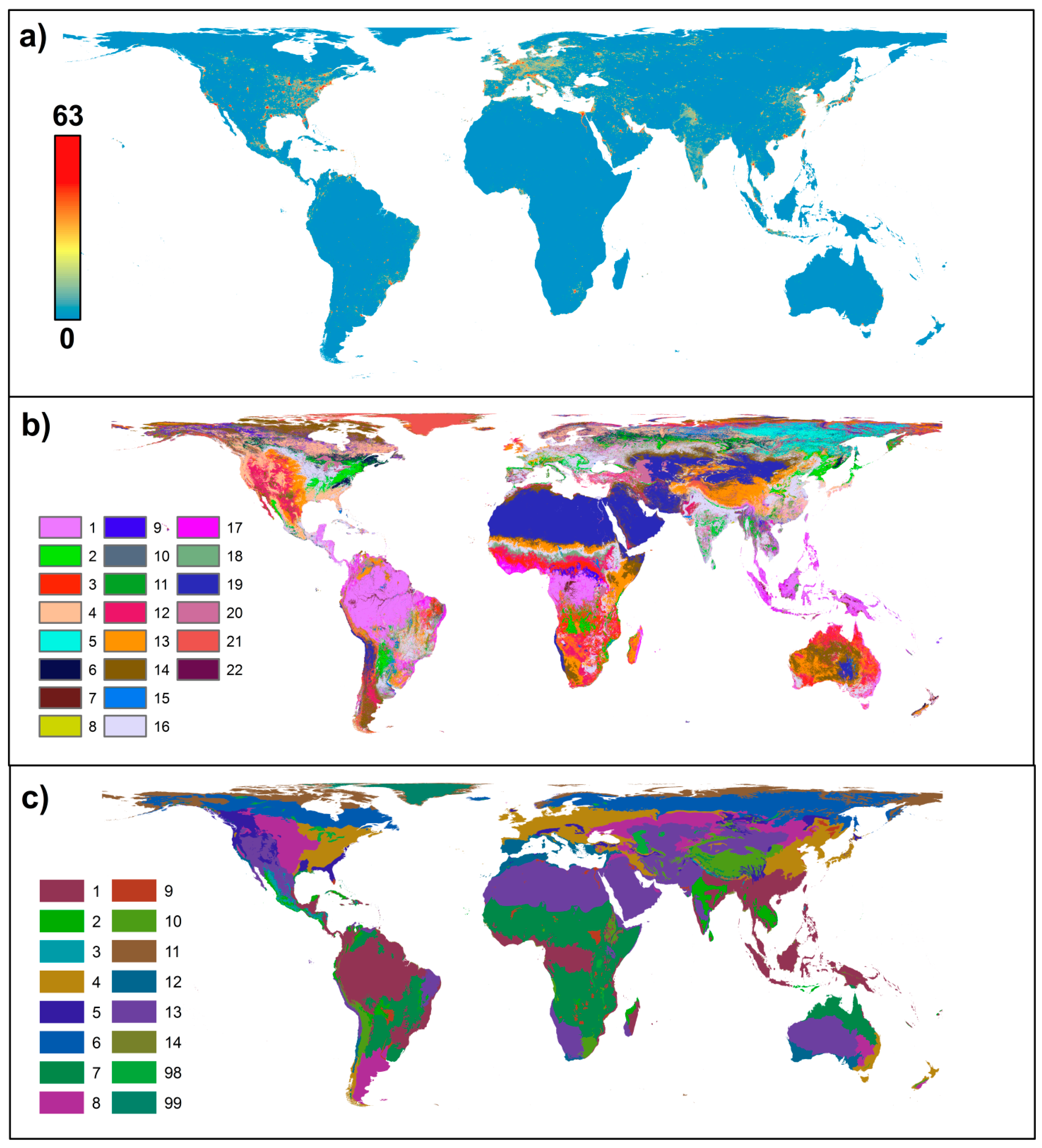
| GLC Global Landcover Class | WWF Terrestrial Ecoregion |
|---|---|
| 1. Tree Cover, broadleaved, evergreen | 1. Deserts and xeric landscapes |
| 2. Tree Cover, broadleaved, deciduous, closed | 2. Tropical/subtropical moist broadleaf forests |
| 3. Tree Cover, broadleaved, deciduous, open | 3. Tropical/subtropical dry broadleaf forests |
| 4. Tree Cover, needle-leaved, evergreen | 4. Tropical/subtropical coniferous forests |
| 5. Tree Cover, needle-leaved, deciduous | 5. Temperate broadleaf and mixed forests |
| 6. Tree Cover, mixed leaf type | 6. Temperate coniferous forest |
| 7. Tree Cover, regularly flooded, fresh water (& brackish) | 7. Boreal forests/Taiga |
| 8. Tree Cover, regularly flooded, saline water | 8. Tropical/subtropical grasslands, savannas and shrublands |
| 9. Mosaic: Tree cover/Other natural vegetation | 9.Flooded grasslands and shrublands |
| 10. Tree Cover, burnt | 10. Tundra |
| 11. Shrub Cover, closed-open, evergreen | 11. Mangroves |
| 12. Shrub Cover, closed-open, deciduous | |
| 13. Herbaceous Cover, closed-open | |
| 14. Sparse Herbaceous or sparse Shrub Cover | |
| 15. Regularly flooded Shrub and/or Herbaceous Cover | |
| 16. Cultivated and managed areas | |
| 17. Mosaic: Cropland/Tree Cover/Other natural vegetation | |
| 18. Mosaic: Cropland/Shrub or Grass Cover | |
| 19. Bare Areas | |
| 20. Water Bodies (natural & artificial) | |
| 21. Snow and Ice (natural & artificial) | |
| 22. Artificial surfaces |
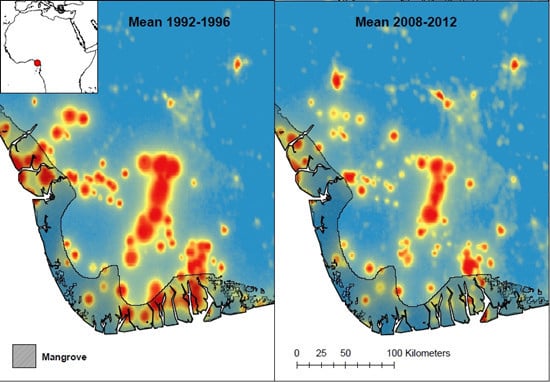
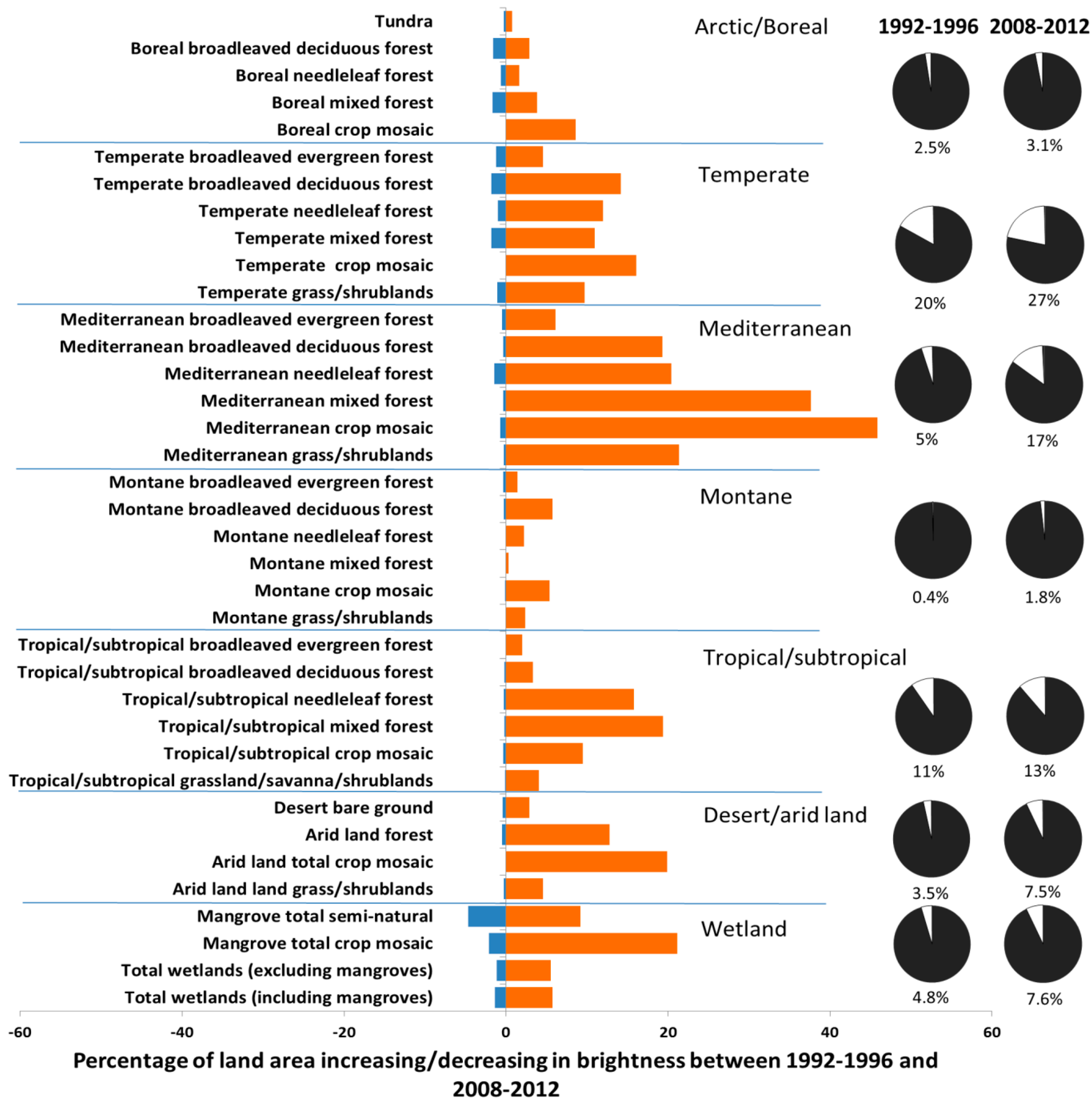
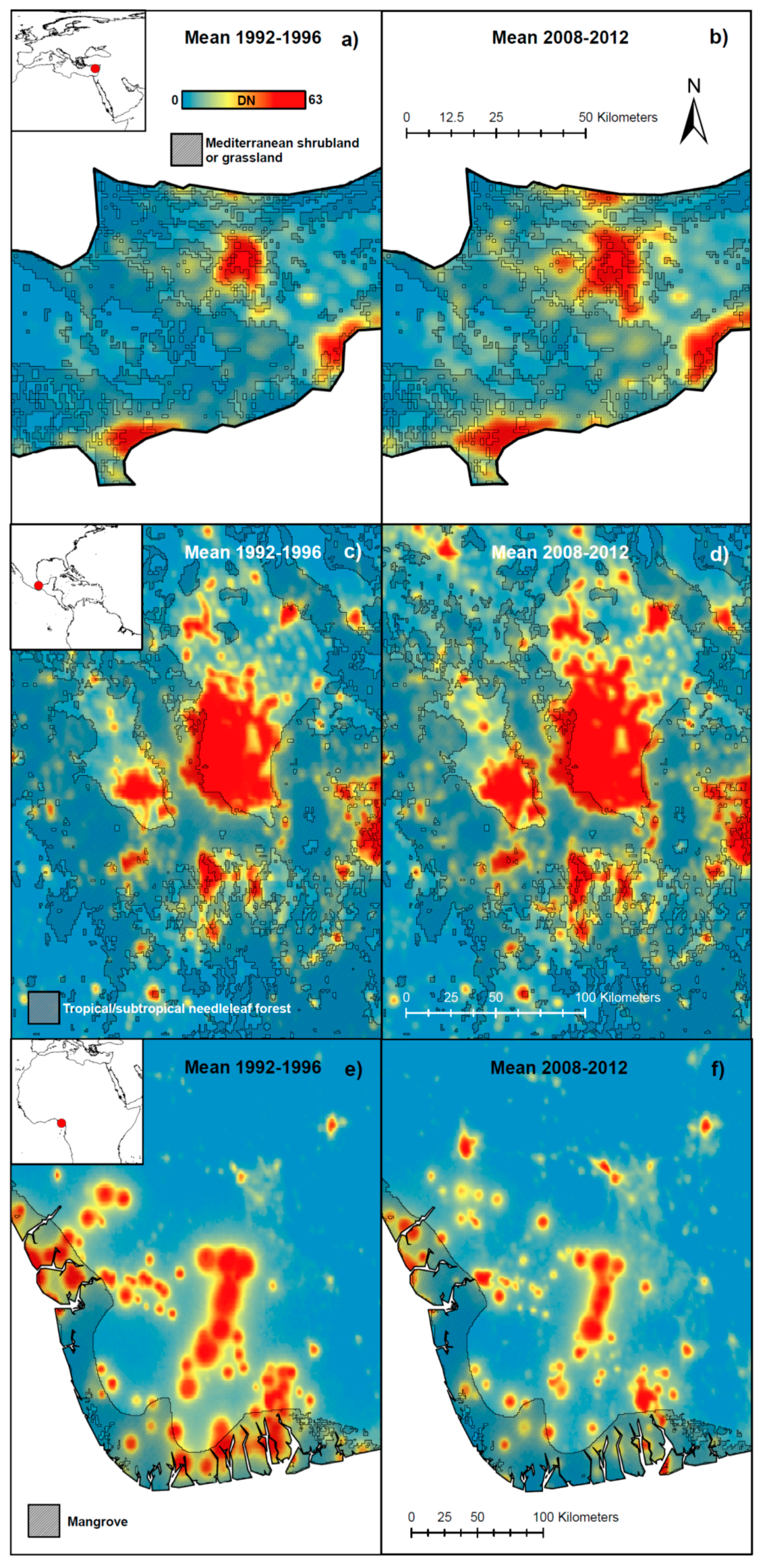
2. Results and Discussion
3. Methods
3.1. Night-Time Lights Data
3.2. Land Cover Data
3.3. Processing
| Land Cover | Biome (WWF) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| GLC Code | Land Cover Classification (GLC2000) | Boreal/Arctic (7,12) | Mangrove (14) | Mediterranean (13) | Montane (11) | Temperate (5,6,9) | Tropical/subtropical (2,3,4,8) | Desert (1) | Flooded (10) |
| 1 | Tree cover broadleaved evergreen | NA | Mangrove | Med. broadleaf evergreen forest | Mon. broadleaf evergreen forest | Temp. broadleaf evergreen forest | T/S broadleaf evergreen forest | Arid forest | Other wetland |
| 2 | Tree cover broadleaved deciduous closed | Boreal broadleaf forest | Mangrove | Med. broadleaf deciduous forest | Mon. broadleaf deciduous forest | Temp. broadleaf deciduous forest | T/S broadleaf deciduous forest | Arid forest | Other wetland |
| 3 | Tree cover broadleaved deciduous open | Boreal broadleaf forest | Mangrove | Med. broadleaf deciduous forest | Mon. broadleaf deciduous forest | Temp. broadleaf deciduous forest | T/S broadleaf deciduous forest | Arid forest | Other wetland |
| 4 | Tree cover needle-leaf evergreen | Boreal needleleaf forest | NA | Med. needleleaf forest | Mon. needleleaf forest | Temp. needleleaf forest | T/S needleleaf forest | Arid forest | Other wetland |
| 5 | Tree cover needle-leaved deciduous | Boreal needleleaf forest | NA | Med. needleleaf forest | Mon. needleleaf forest | Temp. needleleaf forest | T/S needleleaf forest | Arid forest | Other wetland |
| 6 | Tree cover mixed leaf type | Boreal mixed forest | Mangrove | Med. mixed forest | Mon. mixed forest | Temp. mixed forest | T/S mixed forest | Arid forest | Other wetland |
| 7 | Tree cover regularly flooded, fresh water | Boreal/Arctic wetland | Mangrove | Med. wetland | Mon. wetland | Temp. wetland | T/S wetland | Aridland wetland | Other wetland |
| 8 | Tree cover regularly flooded, saline water | Boreal/Arctic wetland | Mangrove | Med. wetland | Mon. wetland | Temp. wetland | T/S wetland | Aridland wetland | Other wetland |
| 9 | Mosaic: tree cover/other natural vegetation | Tundra | NA | Med. shrub/grassland | Mon. shrub/grassland | Temp. shrub/grassland | T/S shrub/grassland/savanna | Arid shrub/grassland | Other wetland |
| 10 | Tree cover, burnt | NA | NA | NA | NA | NA | NA | NA | Other wetland |
| 11 | Shrub cover, closed-open, evergreen | Tundra | Mangrove | Med. shrub/grassland | Mon. shrub/grassland | Temp. shrub/grassland | T/S shrub/grassland/savanna | Arid shrub/grassland | Other wetland |
| 12 | Shrub cover, closed-open, deciduous | Tundra | Mangrove | Med. shrub/grassland | Mon. shrub/grassland | Temp. shrub/grassland | T/S shrub/grassland/savanna | Arid shrub/grassland | Other wetland |
| 13 | Herbaceous cover, closed-open | Tundra | NA | Med. shrub/grassland | Mon. shrub/grassland | Temp. shrub/grassland | T/S shrub/grassland/savanna | Arid shrub/grassland | Other wetland |
| 14 | Sparse herbaceous or sparse shrub cover | Tundra | NA | Med. shrub/grassland | Mon. shrub/grassland | Temp. shrub/grassland | T/S shrub/grassland/savanna | Desert/semidesert | Other wetland |
| 15 | Regularly flooded shrub and/or herbaceous cover | Boreal/Arctic wetland | Mangrove | Mediterranean wetland | Montane wetland | Temp. wetland | T/S shrub/grassland/savanna | Aridland wetland | Other wetland |
| 16 | cultivated and managed areas | NA | NA | NA | NA | NA | NA | NA | NA |
| 17 | Mosaic: cropland/treecover/other natural vegetation | Boreal crop mosaic | Mangrove crop mosaic | Med. crop mosaic | Mod. crop mosaic | Temp. crop mosaic | T/S crop mosaic | Arid crop mosaic | Other wetland |
| 18 | Mosaic: cropland/shrub or grass cover | Boreal crop mosaic | Mangrove crop mosaic | Med. crop mosaic | Med. crop mosaic | Temp. crop mosaic | T/S crop mosaic | Arid crop mosaic | Other wetland |
| 19 | Bare areas | NA | NA | NA | Montane desert/semidesert | NA | NA | Desert/semidesert | Other wetland |
| 20 | Water bodies | NA | NA | NA | NA | NA | NA | NA | NA |
| 21 | Snow and ice | NA | NA | NA | NA | NA | NA | NA | NA |
| 22 | Artificial surfaces | NA | NA | NA | NA | NA | NA | NA | NA |
3.4. Assessment of Error and Bias
3.5. Change Detection
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cinzano, P.; Falchi, F.; Elvidge, C.D. The first World Atlas of the artificial sky brightness. Mon. Not. R. Astron. Soc. 2001, 328, 689–707. [Google Scholar] [CrossRef]
- Kyba, C.C.M.; Hölker, F. Do artificially illuminated skies affect biodiversity in nocturnal landscapes? Landsc. Ecol. 2013, 28, 1637–1640. [Google Scholar] [CrossRef]
- Longcore, T.; Rich, C. Ecological light pollution. Front. Ecol. Environ. 2004, 2, 191–198. [Google Scholar] [CrossRef]
- Rich, C.; Longcore, T. Ecological Consequences of Artificial Night Lighting; Rich, C., Longcore, T., Eds.; Island Press: Washington, DC, USA, 2006. [Google Scholar]
- Davies, T.W.; Duffy, J.P.; Bennie, J.; Gaston, K.J. The nature, extent and ecological significance of marine light pollution. Front. Ecol. Environ. 2014, 12, 347–355. [Google Scholar] [CrossRef]
- Hölker, F.; Wolter, C.; Perkin, E.K.; Tockner, K. Light pollution as a biodiversity threat. Trends Ecol. Evol. 2010, 25, 681–682. [Google Scholar] [CrossRef] [PubMed]
- Gaston, K.J.; Bennie, J.; Davies, T.W.; Hopkins, J. The ecological effects of nighttime light pollution: A mechanistic appraisal. Biol. Rev. 2013, 88, 912–927. [Google Scholar] [PubMed]
- Gaston, K.J.; Bennie, J. Demographic effects of artificial nighttime lighting on animal populations. Environ. Rev. 2014, 22, 1–8. [Google Scholar] [CrossRef]
- Lewansik, D.; Voigt, C.C. Artificial light puts ecosystem services of frugivorous bats at risk. J. Appl. Ecol. 2014, 52, 388–394. [Google Scholar] [CrossRef]
- Bennie, J.; Davies, T.W.; Duffy, J.P.; Inger, R.; Gaston, K.J. Contrasting trends in light pollution across Europe based on satellite observed night time lights. Sci. Rep. 2014, 4, 1–6. [Google Scholar] [CrossRef]
- Elvidge, C.D.; Hsu, F.-C.; Baugh, K.; Ghosh, T. National trends in satellite observed lighting: 1992–2012. In Global Urban Monitoring and Assessment through Earth Observation; Weng, Q., Ed.; CRC Press: Boca Raton, FL, USA, 2014; pp. 97–120. [Google Scholar]
- Zhao, N.; Zhou, Y.; Samson, E.L. Correcting incompatible DN values and geometric errors in nighttime lights time-series images. IEEE Trans. Geosci. Remote Sens. 2014, 53, 2039–2049. [Google Scholar] [CrossRef]
- Butt, M.J. Estimation of light pollution using satellite remote sensing and geographic information system techniques. GISci. Remote Sens. 2012, 49, 609–621. [Google Scholar] [CrossRef]
- Sutton, P.C. A scale adjusted measure of “urban sprawl” using nighttime satellite imagery. Remote Sens. Environ. 2003, 86, 353–369. [Google Scholar] [CrossRef]
- Li, X.; Chen, X.; Zhao, Y.; Xu, J.; Chen, F.; Li, H. Automatic calibration of night-time light imagery using robust regression. Remote Sens. Lett. 2013, 4, 46–55. [Google Scholar]
- Amaral, S.; Monteiro, A.M.V.; Camara, G.; Quintanilha, J.A. DMSP/OLS night-time light imagery for urban population estimates in the Brazilian Amazon. Int. J. Remote Sens. 2006, 27, 855–870. [Google Scholar] [CrossRef]
- Chen, X.; Nordhaus, W.D. Using luminosity as a proxy for economic statistics. Proc. Natl. Acad. Sci. USA 2011, 108, 8589–8594. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Yang, X.; Gao, B.; Yang, Y.; Zhao, Y. Application of DMSP/OLS nighttime light images: A meta-analysis and a systematic literature review. Remote Sens. 2014, 6, 6844–6866. [Google Scholar] [CrossRef]
- Miller, S.D.; Straka, W.; Mills, S.P.; Elvidge, C.D.; Lee, T.F.; Solbrig, J.; Walther, A.; Heidinger, A.K.; Weiss, S.C. Illuminating the capabilities of the Suomi National Polar-Orbiting Partnership (NPP) Visible Infrared Imaging Radiometer Suite (VIIRS) day/night band. Remote Sens. 2013, 5, 6717–6766. [Google Scholar] [CrossRef]
- Elvidge, C.D.; Baugh, K.E.; Safran, J.; Tuttle, B.T.; Howard, A.T.; Hayes, A.T.; Jantzen, J.; Erwin, E.H. Preliminary results from nighttime lights detection. Int. Arch. Photogamm. Remote Sens. Spat. Inf. Sci. 2005, 36, 8–18. [Google Scholar]
- De Miguel, A.S.; Zamorano, J.; Castaño, J.G.; Pascual, S. Evolution of the energy consumed by street lighting in Spain estimated with DMSP-OLS data. J. Quant. Spectrosc. Radiat. Transf. 2014, 139, 109–117. [Google Scholar] [CrossRef]
- Médail, F.; Quézel, P. Hot-spots analysis for conservation of plant biodiversity in the Mediterranean basin. Ann. Mo. Bot. Gard. 1997, 84, 112–127. [Google Scholar] [CrossRef]
- Rouget, M.; Richardson, D.M.; Cowling, R.M.; Lloyd, J.W.; Lombard, A.T. Current patterns of habitat transformation and future threats to Biodiversity in terrestrial ecosystems of the Cape Floristic Region. Biol. Conserv. 2003, 112, 63–85. [Google Scholar] [CrossRef]
- Goldblatt, P.; Manning, J.C. Plant diversity of the Cape Region of South Africa. Ann. Mo. Bot. Gard. 2002, 89, 281–302. [Google Scholar] [CrossRef]
- Beard, J.S.; Chapman, A.R.; Gioia, P. Species richness and endemism in the western Australian flora. J. Biogeogr. 2000, 27, 1257–1268. [Google Scholar] [CrossRef]
- Farjon, A. A Handbook of the World’s Conifers; Brill Academic Publishers: Boston, MA, USA, 2010. [Google Scholar]
- Challenger, A.; Soberon, J. Los ecosistemas terrestres. In Capital Natural de México, Volume 1: Conocimiento Actual de la Diversidad; CONABIO: Mexico, Mexico, 2008. [Google Scholar]
- Valiela, I.; Bowen, J.L.; York, J.K. Mangrove forests: One of the world’s threatened major tropical environments. BioScience 2010, 51, 807–815. [Google Scholar] [CrossRef]
- Barbier, E.B.; Hacker, S.D.; Kennedy, C.; Koch, E.W.; Stier, A.C.; Silliman, B.R. The value of estuarine and coastal ecosystem services. Ecol. Monogr. 2011, 81, 169–193. [Google Scholar] [CrossRef]
- Donato, D.C.; Kauffman, J.B.; Murdiyarso, D.; Kurnianto, S.; Stidham, M.; Kanninen, M. Mangroves among the most carbon-rich forests in the tropics. Nat. Geosci. 2011, 4, 293–297. [Google Scholar] [CrossRef]
- National Oceanic and Atmospheric Administration/National Geophysical Data Center (NOAA/NGDC) Earth Observation Group. Available online: http://ngdc.noaa.gov/eog/ (accessed on 6 July 2014).
- Koenker, R. Quantreg: Quantile Regression. Available online: http://cran.fyxm.net/web/packages/quantreg/quantreg.pdf (accessed on 30 September 2014).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Olson, D.M.; Dinerstein, E.; Wikramanayake, E.D.; Burgess, N.D.; Powell, G.V.N.; Underwood, E.C.; D’Amico, J.A.; Itoua, I.; Strand, H.E.; Morrison, J.C.; et al. Terrestrial ecoregions of the world: A new map of life on Earth. Bioscience 2001, 51, 933–938. [Google Scholar] [CrossRef]
- Ecoregions of the World. Available online: http://www.worldwildlife.org/publications/terrestrial-ecoregions-of-the-world (accessed on 1 August 2014).
- Global Land Cover 2000 Database. Available online: http://bioval.jrc.ec.europa.eu/products/glc2000/products.php (accessed on 1 August 2014).
- Bivand, R.; Keitt, T.; Rowlingson, B. Rgdal: Bindings for the Geospatial Data Abstraction Library. Available online: http://cran.univ-lyon1.fr/web/packages/rgdal/rgdal.pdf (accessed on 30 September 2014).
- Hijmans, R.J.; van Etten, J. Raster: Geographic Data Analysis and Modeling. Available online: http://cran.stat.unipd.it/web/packages/raster/raster.pdf (accessed on 30 September 2014).
- Wade, T.G.; Riitters, K.H.; Wickham, J.D.; Jones, K.B. Distribution and Causes of Global Forest Fragmentation. Available online: http://www.ecologyandsociety.org/vol7/iss2/art7/ (accessed on 30 September 2014).
- Moreno-Sanchez, R.; Buxton-Torres, T.; Sinbernagel, K.; Moreno, S.F. Fragmentation of the forest in Mexico: National level assessments for 1993, 2002 and 2008. Int. J. Statist. Geogr. 2014, 5, 4–17. [Google Scholar]
- De Miguel, A.S.; Castaño, J.G.; Zamorano, J.; Kyba, C.M.; Ángeles, M.; Cayuela, L.; Martinez, G.M.; Callupner, P. Atlas of astronaut photos of Earth at night. Astron. Geophys. 2014, 55, 4–36. [Google Scholar] [CrossRef]
- Rydell, J. Exploitation of insects around streetlights by bats in Sweden. Funct. Ecol. 1992, 6, 744–750. [Google Scholar] [CrossRef]
- Stone, E.L.; Jones, G.; Harris, S. Street lighting disturbs commuting bats. Curr. Biol. 2009, 19, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Frank, K.D. Impact of outdoor lighting on moths: An assessment. J. Lepid. Soc. 1988, 42, 63–93. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bennie, J.; Duffy, J.P.; Davies, T.W.; Correa-Cano, M.E.; Gaston, K.J. Global Trends in Exposure to Light Pollution in Natural Terrestrial Ecosystems. Remote Sens. 2015, 7, 2715-2730. https://doi.org/10.3390/rs70302715
Bennie J, Duffy JP, Davies TW, Correa-Cano ME, Gaston KJ. Global Trends in Exposure to Light Pollution in Natural Terrestrial Ecosystems. Remote Sensing. 2015; 7(3):2715-2730. https://doi.org/10.3390/rs70302715
Chicago/Turabian StyleBennie, Jonathan, James P. Duffy, Thomas W. Davies, Maria Eugenia Correa-Cano, and Kevin J. Gaston. 2015. "Global Trends in Exposure to Light Pollution in Natural Terrestrial Ecosystems" Remote Sensing 7, no. 3: 2715-2730. https://doi.org/10.3390/rs70302715
APA StyleBennie, J., Duffy, J. P., Davies, T. W., Correa-Cano, M. E., & Gaston, K. J. (2015). Global Trends in Exposure to Light Pollution in Natural Terrestrial Ecosystems. Remote Sensing, 7(3), 2715-2730. https://doi.org/10.3390/rs70302715