Potential of RGB-Derived Vegetation Indices as an Alternative to NIR-Based Vegetation Indices to Monitor Nitrogen Status in Maize
Highlights
- RGB-based vegetation indices showed a good capability in estimating nitrogen status in maize, with performance close to that of NIR-based vegetation indices.
- Indices derived from the blue and red channels (e.g., NDGBI and NDRBI) demonstrated clear sensitivity to nitrogen fertilization levels across different growth stages.
- RGB sensors can serve as an effective, low-cost alternative or complementary tool to NIR-based systems for nitrogen monitoring.
- The findings support more accurate and timely nitrogen management decisions in maize fields, especially in regions lacking advanced spectral equipment.
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Experimental Design
2.2. Data Collection
2.2.1. Image Acquisition and Processing
2.2.2. Vegetation Indices
2.2.3. Agronomic and Yield Variables
3. Results
3.1. Indices
3.1.1. NIR-Based Indices: NDVI and GNDVI


3.1.2. RGB-Based Indices: NDGBI and NDRBI


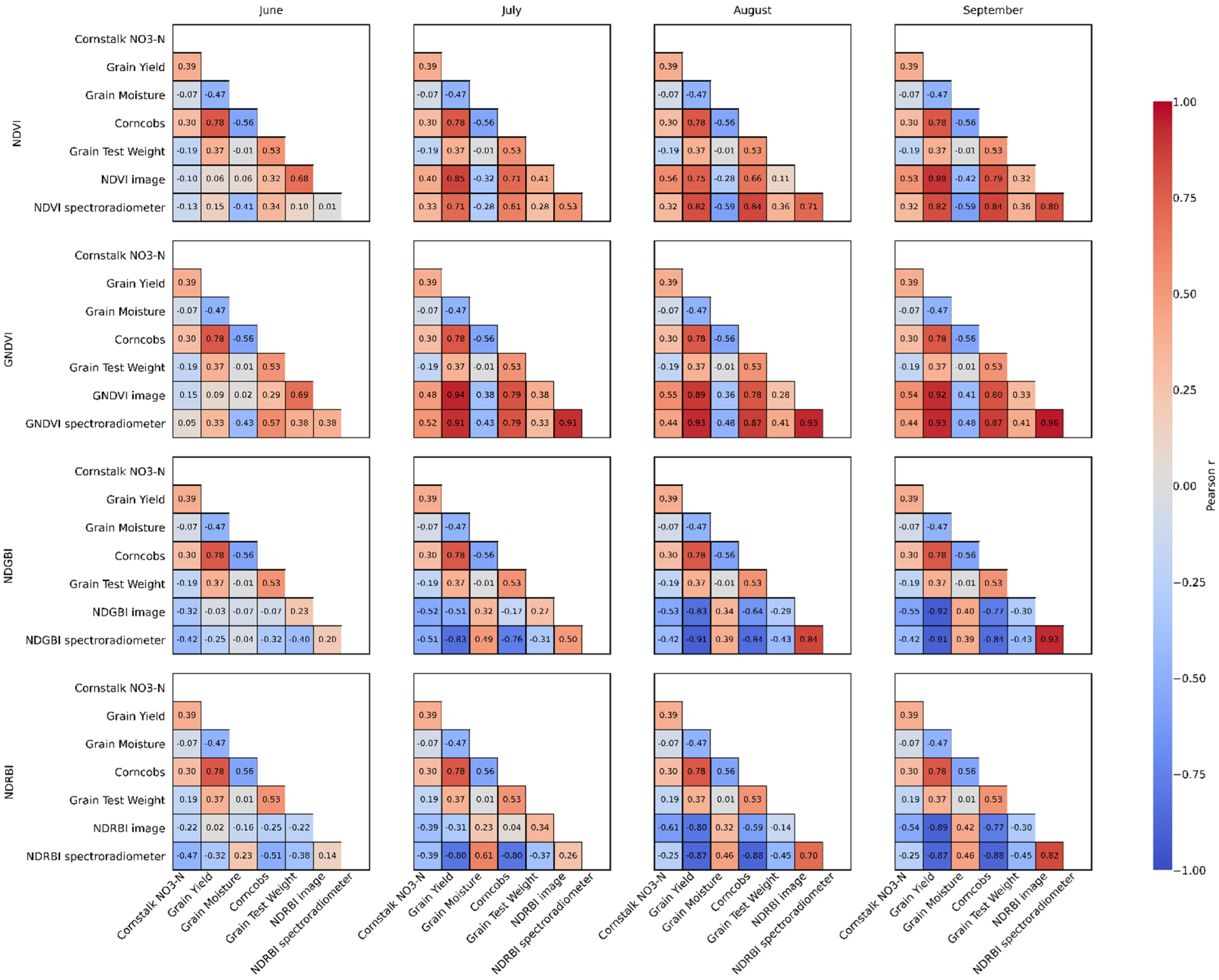
3.2. Relationship with Agronomic and Yield Variables
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| UAVs | Unmanned Aerial Vehicles |
| NIR | Near-Infrared |
| VIS | Visible |
| RGB | Red–Green–Blue |
| ExG | Excess Green |
| VIs | Vegetation Indices |
| NDVI | Normalized Difference Vegetation Index |
| GNDVI | Green Normalized Difference Vegetation Index |
| NDGBI | Normalized Difference Green–Blue Index |
| NDRBI | Normalized Difference Red–Blue Index |
| VTOL | Vertical Take-off and Landing |
| GSD | Ground Sample Distance |
| RTK | Real-Time Kinematic |
| RMSE | Root Mean Square Error |
| R2 | Determination Coefficient |
References
- Guo, Y.; Zhang, X.; Chen, S.; Wang, H.; Jayavelu, S.; Cammarano, D.; Fu, Y. Integrated UAV-Based Multi-Source Data for Predicting Maize Grain Yield Using Machine Learning Approaches. Remote Sens. 2022, 14, 6290. [Google Scholar] [CrossRef]
- Silva, D.; Madari, B.E.; Santana, C.; Costa, J.V.S.; Ferreira, M.E. Planning and Optimization of Nitrogen Fertilization in Corn Based on Multispectral Images and Leaf Nitrogen Content Using Unmanned Aerial Vehicle (UAV). Precis. Agric. 2025, 26, 30. [Google Scholar] [CrossRef]
- Arorra, A. Top-Corn Producing Countries in the World 2024. Adda247 2024. Available online: https://currentaffairs.adda247.com/top-10-corn-producing-countries-in-the-world (accessed on 16 May 2024).
- Cai, S.; Zhao, X.; Yan, X. Towards Precise Nitrogen Fertilizer Management for Sustainable Agriculture. Earth Crit. Zone 2025, 2, 100026. [Google Scholar] [CrossRef]
- Vries, W. Impacts of Nitrogen Emissions on Ecosystems and Human Health: A mini review. Curr. Opin. Env. Sci. Health 2021, 21, 100249. [Google Scholar] [CrossRef]
- Kitchen, N.; Ransom, C.; Schepers, J.; Hatfield, J.; Massey, R.; Drummond, T. A New Perspective When Examining Maize Fertilizer Nitrogen Use Efficiency, incrementally. PLoS ONE 2022, 17, E0267215. [Google Scholar] [CrossRef]
- Yi, J.; Li, H.; Zhao, Y.; Shao, M.; Zhang, H.; Liu, M. Assessing Soil Water Balance to Optimize Irrigation Schedules of Flood-Irrigated Maize Fields with Different Cultivation Histories in the Arid Region. Agric. Water Manag. 2022, 265, 107543. [Google Scholar] [CrossRef]
- Lu, S.; Zhu, G.; Qiu, D.; Li, R.; Jiao, Y.; Meng, G.; Lin, X.; Wang, Q.; Zhang, W.; Chen, L. Optimizing Irrigation in Arid Irrigated Farmlands Based on Soil Water Movement Processes: Knowledge from Water Isotope Data. Geoderma 2025, 460, 117440. [Google Scholar] [CrossRef]
- Duan, J.; Rudnick, D.; Proctor, C.; Heeren, D.; Nakabuye, H.N.; Katimbo, A.; Shi, Y.; de Sousa Ferreira, V. Estimation of Corn Nitrogen Demand under Different Irrigation Conditions Based on UAV Multispectral Technology. Agric. Water Manag. 2024, 304, 109075. [Google Scholar] [CrossRef]
- Zheng, H.; Ma, J.; Zhou, M.; Li, D.; Yao, X.; Cao, W.; Zhu, Y.; Cheng, T. Enhancing the Nitrogen Signals of Rice Canopies across Critical Growth Stages through the Integration of Textural and Spectral Information from Unmanned Aerial Vehicle (UAV) Multispectral Imagery. Remote Sens. 2020, 12, 957. [Google Scholar] [CrossRef]
- Zahir, S.; Jamlos, M.F.; Omar, A.F.; Jamlos, M.A.; Mamat, R.; Muncan, J.; Tsenkova, R. Review-Plant Nutritional Status Analysis Employing the Visible and Near-Infrared Spectroscopy Spectral Sensor. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2024, 304, 123273. [Google Scholar] [CrossRef] [PubMed]
- Vergara, O.; Zaman-Allah, M.; Masuka, B.; Hornero, A.; Zarco-Tejada, P.; Prasanna, B.; Cairns, J.E.; Araus, J.L. A Novel Remote Sensing Approach for Prediction of Maize Yield under Different Conditions of Nitrogen Fertilization. Front. Plant Sci. 2016, 7, 666. [Google Scholar] [CrossRef]
- Parida, P.K.; Somasundaram, E.; Krishnan, R.; Radhamani, S.; Uthandi, S.E.P.; Parameswari, E.; Raja, R.; Shri Rangasami, S.R.; Sangeetha, S.P.; Gangai Selvi, R. Unmanned Aerial Vehicle-Measured Multispectral Vegetation Indices for Predicting LAI, SPAD Chlorophyll, and Yield of Maize. Agriculture 2024, 14, 1110. [Google Scholar] [CrossRef]
- Abdelmajeed, A.; Juszczak, R. Challenges and Limitations of Remote Sensing Applications in Northern Peatlands: Present and Future Prospects. Remote Sens. 2024, 16, 591. [Google Scholar] [CrossRef]
- Val, J.; González-Pérez, I.; Sanz-Ablanedo, E.; Maresma, Á.; Rodríguez-Pérez, J. Field Spectroscopy for Monitoring Nitrogen Fertilization and Estimating Cornstalk Nitrate Content in Maize. AgriEngineering 2025, 7, 264. [Google Scholar]
- Di Gennaro, S.F.; Toscano, P.; Gatti, M.; Poni, S.; Berton, A.; Matese, A. Spectral Comparison of UAV-Based Hyper and Multispectral Cameras for Precision Viticulture. Remote Sens. 2022, 14, 449. [Google Scholar] [CrossRef]
- Yu, K.; Belwalkar, A.; Wang, W.; Hu, Y.; Hunegnaw, A.; Nurunnabi, A.; Ruf, T.; Li, F.; Jia, L.; Kooistra, L.; et al. UAV Hyperspectral Remote Sensing for Crop Nitrogen Monitoring: Progress, Challenges, and Perspectives. Smart Agric. Technol. 2025, 12, 101507. [Google Scholar] [CrossRef]
- Feng, H.; Tao, H.; Li, Z.; Yang, G.; Zhao, C. Comparison of UAV RGB Imagery and Hyperspectral Remote-Sensing Data for Monitoring Winter Wheat Growth. Remote Sens. 2022, 14, 3811. [Google Scholar] [CrossRef]
- Guan, S.; Fukami, K.; Matsunaka, H.; Okami, M.; Tanaka, R.; Nakano, H.; Sakai, T.; Nakano, K.; Ohdan, H.; Takahashi, K. Assessing Correlation of High-Resolution NDVI with Fertilizer Application Level and Yield of Rice and Wheat Crops Using Small UAVs. Remote Sens. 2019, 11, 112. [Google Scholar] [CrossRef]
- Naik, B.; Nayak, H.; Govinda, S. Prediction of Wheat Yield by Using UAV RGB Drone Imagery and Advanced Machine Learning Techniques. Int. J. Stat. Appl. Math. 2023, 8, 961–969. [Google Scholar]
- Anzar, S.M.; Sherin, K.; Panthakkan, A.; Al Mansoori, S.; Al-Ahmad, H. Evaluation of UAV-Based RGB and Multispectral Vegetation Indices for Precision Agriculture in Palm Tree Cultivation. Int. Arch. Photogramm. Remote Sens. Spat. Inf. Sci. 2025, XLVIII-G-2, 163–170. Available online: https://isprs-archives.copernicus.org/articles/XLVIII-G-2025/163/2025/ (accessed on 2 February 2026). [CrossRef]
- Radocz, L.; Juhász, C.; Tamás, A.; Illés, Á.; Ragán, P. Multispectral UAV-Based Disease Identification Using Vegetation Indices for Maize Hybrids. Agriculture 2024, 14, 2002. [Google Scholar] [CrossRef]
- Santana, D.; Cotrim, M.; Flores, M.; Shiratsuchi, L.; Silva, C.; Ribeiro, L.; Teodoro, P.E. UAV-Based Multispectral Sensor to Measure Variations in Corn as a Function of Nitrogen Topdressing. Remote Sens. Appl. Soc. Environ. 2021, 22, 100534. [Google Scholar] [CrossRef]
- García-Fernández, M.; Sanz-Ablanedo, E.; Rodríguez-Pérez, J.R. High-Resolution Drone-Acquired RGB Imagery to Estimate Spatial Grape Quality Variability. Agronomy 2021, 11, 655. [Google Scholar] [CrossRef]
- Colovic, M.; Stellacci, A.; Mzid, N.; Di Venosa, M.; Todorovic, M.; Cantore, V.; Albrizio, R. Comparative Performance of Aerial RGB vs. Ground Hyperspectral Indices for Evaluating Water and Nitrogen Status in Sweet Maize. Agronomy 2024, 14, 562. [Google Scholar] [CrossRef]
- Cvetković, N.; Đoković, A.; Dobrota, M.; Radojicic, M. New Methodology for Corn Stress Detection Using Remote Sensing and Vegetation Indices. Sustainability 2023, 15, 5487. [Google Scholar] [CrossRef]
- Yu, J.; Wang, J.; Leblon, B. Evaluation of Soil Properties, Topographic Metrics, Plant Height, and Unmanned Aerial Vehicle Multispectral Imagery Using Machine Learning Methods to Estimate Canopy Nitrogen Weight in Corn. Remote Sens. 2021, 13, 3105. [Google Scholar] [CrossRef]
- Murtrey, J.; Middleton, E.; Campbell, L.; Daughtry, C. Optical Reflectance and Fluorescence for Detecting Nitrogen Needs in Zea mays L. In Proceedings of the 2003 IEEE International Geoscience and Remote Sensing Symposium Proceedings (IEEE Cat No03CH37477); IEEE: Piscataway, NJ, USA, 2003; pp. 4602–4604. [Google Scholar] [CrossRef]
- Pereira, L.; Rodrigues, C.; Marques, C.; Santos, T. Applying RGB-Based Vegetation Indices Obtained from UAS Imagery for Monitoring the Rice Crop at the Field Scale: A Case Study in Portugal. Agriculture 2023, 13, 1916. [Google Scholar] [CrossRef]
- Saberioon, M.M.; Amin, M.S.M.; Aimrun, W.; Gholizadeh, A.; Anuar, A.R. Assessment of colour indices derived from conventional digital camera for determining nitrogen status in rice plants. J. Food Agric. Environ. 2013, 11, 655–662. [Google Scholar]
- Sastre, L.F.; Alte da Veiga, N.M.S.; Ruiz, N.M.; Carrión-Prieto, P.; Marcos-Robles, J.L.; Navas-Gracia, L.M.; Martín-Ramos, P. Assessment of RGB Vegetation Indices to Estimate Chlorophyll Content in Sugar Beet Leaves in the Final Cultivation Stage. AgriEngineering 2020, 2, 128–149. [Google Scholar] [CrossRef]
- Bendig, J.; Bolten, A.; Bennertz, S.; Broscheit, J.; Eichfuss, S.; Bareth, G. Estimating Biomass of Barley Using Crop Surface Models (CSMs) Derived from UAV-Based RGB Imaging. Remote Sens. 2014, 6, 10395–10412. [Google Scholar] [CrossRef]
- Szechyńska-Hebda, M.; Hołownicki, R.; Doruchowski, G.; Sas, K.; Puławska, J.; Jarecka-Boncela, A.; Ptaszek, M.; Włodarek, A. Application of Hyperspectral Imaging for Early Detection of Pathogen-Induced Stress in Cabbage as Case Study. Agronomy 2025, 15, 1516. [Google Scholar] [CrossRef]
- Zheng, H.; Cheng, T.; Li, D.; Yao, X.; Tian, Y.; Cao, W.; Zhu, Y. Combining Unmanned Aerial Vehicle (UAV)-Based Multispectral Imagery and Ground-Based Hyperspectral Data for Plant Nitrogen Concentration Estimation in Rice. Front. Plant Sci. 2018, 9, 936. [Google Scholar] [CrossRef]
- Lee, K.J.; Lee, B.W. Estimating Canopy Cover from Color Digital Camera Image of Rice Field. J. Crop. Sci. Biotechnol. 2011, 14, 151–155. [Google Scholar] [CrossRef]
- Bagnall, G.; Thomasson, J.; Yang, C.; Wang, T.; Han, X.; Sima, C.; Chang, A. Uncrewed Aerial Vehicle Radiometric Calibration: A Comparison of Autoexposure and Fixed Exposure. Plant Phenome J. 2023, 6, e20082. [Google Scholar] [CrossRef]
- Xue, J.; Su, B. Significant Remote Sensing Vegetation Indices: A Review of Developments and Applications. J. Sens. 2017, 2017, 1353691. [Google Scholar] [CrossRef]
- Dutta, R. Review of Vegetation Indices for Vegetation Monitoring. In Proceedings of the 35th Asian Conference on Remote Sensing (ACRS 2014); Asian Conference on Remote Sensing Society: Pathumthani, Thailand, 2014. [Google Scholar]
- Maresma, Á.; Ariza, M.; Martínez, E.; Lloveras, J.; Martínez-Casasnovas, J.A. Analysis of Vegetation Indices to Determine Nitrogen Application and Yield Prediction in Maize (Zea mays L.) from a Standard UAV Service. Remote Sens. 2016, 8, 973. [Google Scholar] [CrossRef]
- Kavak, M.; Karadogan, S.; Özdemir, G. A Long Term NDVI Investigation of Hevsel Gardens Using Remote Sensing Techniques. In Remote Sensing; Dicle University: Diyarbakır, Turkey, 2007. [Google Scholar]
- Schowengerdt, R.A. Remote Sensing: Models and Methods for Image Processing, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Hunt, E.; Hively, W.; Fujikawa, S.; Linden, D.; Daughtry, C.; McCarty, G. Acquisition of NIR-Green-Blue Digital Photographs from Unmanned Aircraft for Crop Monitoring. Remote Sens. 2010, 2, 290–305. [Google Scholar] [CrossRef]
- Woebbecke, D.; Meyer, E.; Von Bargen, K. Color Indices for Weed Identification Under Various Soil, Residue, and Lighting Conditions. Trans. ASAE 1995, 38, 259–269. [Google Scholar] [CrossRef]
- Xue, B.; Ming, B.; Xin, J.; Yang, H.; Gao, S.; Guo, H.; Feng, D.; Nie, C.; Wang, K.; Li, S. Radiometric Images Captured under Changing Ambient Light Conditions and Applications in Crop Monitoring. Drones 2023, 7, 223. [Google Scholar] [CrossRef]
- Rouse, J.W.; Haas, R.H.; Schell, J.A.; Deering, D.W. Monitoring Vegetation Systems in the Great Plains with ERTS. In Proceedings of the Third Earth Resources Technology Satellite-1 Symposium; NASA Special Publication SP-351; NASA: Washington, DC, USA, 1974; Volume 1, pp. 309–317. [Google Scholar]
- Gitelson, A.A.; Kaufman, Y.J.; Merzlyak, M.N. Use of a Green Channel in Remote Sensing of Global Vegetation from EOS-MODIS. Remote Sens. Environ. 1996, 58, 289–298. [Google Scholar] [CrossRef]
- Gitelson, A.; Kaufman, Y.; Stark, R.; Rundquist, D. Novel Algorithms for Remote Estimation of Vegetation Fraction. Remote Sens. Environ. 2002, 80, 76–87. [Google Scholar] [CrossRef]
- Marcial, M.D.J.; González-Sanchez, A.; Jiménez-Jiménez, S.; Ontiveros-Capurata, R.E.; Ojeda, W. Estimation of Vegetation Fraction Using RGB and Multispectral Images from UAV. Int. J. Remote Sens. 2018, 40, 420–438. [Google Scholar] [CrossRef]
- Tilling, A.K.; O’Leary, G.J.; Ferwerda, J.G.; Jones, S.D.; Fitzgerald, G.J.; Rodriguez, D.; Belford, R. Remote sensing of nitrogen and water stress in wheat. Field Crops Res. 2007, 104, 77–85. [Google Scholar] [CrossRef]
- Miller, E.; Bushong, J.; Raun, W.; Abit, J.; Arnall, B. Predicting Early Season Nitrogen Rates of Corn Using Indicator Crops. Agron. J. 2017, 109, 2863–2870. [Google Scholar] [CrossRef]
- Furukawa, F.; Maruyama, K.; Saito, Y.; Kaneko, M. Corn Height Estimation Using UAV for Yield Prediction and Crop Monitoring. In Unmanned Aerial Vehicle: Applications in Agriculture and Environment; Springer International Publishing: Cham, Switzerland, 2020; pp. 51–69. [Google Scholar]
- Jjagwe, P.; Chandel, A.; Langston, D. Pre-Harvest Corn Grain Moisture Estimation Using Aerial Multispectral Imagery and Machine Learning Techniques. Land 2023, 12, 2188. [Google Scholar] [CrossRef]
- Killeen, P.; Kiringa, I.; Yeap, T.; Branco, P. Corn Grain Yield Prediction Using UAV-Based High Spatiotemporal Resolution Imagery, Machine Learning, and Spatial Cross-Validation. Remote Sens. 2024, 16, 683. [Google Scholar] [CrossRef]
- García-Martínez, H.; Flores-Magdaleno, H.; Ascencio-Hernández, R.; Khalil-Gardezi, A.; Tijerina-Chávez, L.; Mancilla-Villa, O.R.; Vázquez-Peña, M.A. Corn Grain Yield Estimation from Vegetation Indices, Canopy Cover, Plant Density, and a Neural Network Using Multispectral and RGB Images Acquired with Unmanned Aerial Vehicles. Agriculture 2020, 10, 277. [Google Scholar] [CrossRef]
- Wang, N.; Wu, Q.; Gui, Y.; Hu, Q.; Li, W. Cross-Modal Segmentation Network for Winter Wheat Mapping in Complex Terrain Using Remote-Sensing Multi-Temporal Images and DEM Data. Remote Sens. 2024, 16, 1775. [Google Scholar] [CrossRef]
- Bao, L.; Yu, L.; Yu, E.; Li, R.; Cai, Z.; Yu, J.; Li, X. Improving the Simulation of Maize Growth Using WRF-Crop Model Based on Data Assimilation and Local Maize Characteristics. Agric. For. Meteorol. 2025, 365, 110478. [Google Scholar] [CrossRef]

| Treatment | Pre-Sowing (8 May 2024) | V2—Second Leaf (12 June 2024) | V6—Sixth Leaf (3 July 2024) | N Rate (kg N·ha−1) |
|---|---|---|---|---|
| T01 | 0 | 0 | 0 | 0 |
| T02 | 91 | 0 | 229 | 320 |
| T03 | 117 | 0 | 203 | 320 |
| T04 | 140 | 0 | 180 | 320 |
| T05 | 0 | 320 | 0 | 320 |
| T06 | 220 | 0 | 162 | 382 |
| Date of Spectroradiometer Measurement | Date of UAV Flight |
|---|---|
| 24 June 2024 | 24 June 2024 |
| 22 July 2024 | 22 July 2024 |
| 28 August 2024 | 5 August 2024 |
| 10 September 2024 |
| Index | Expression | References |
|---|---|---|
| Normalized Difference Vegetation Index (NDVI) | (NIR − R)/(NIR + R) | [45] |
| Green Normalized Difference Vegetation Index (GNDVI) | (NIR − G)/(NIR + G) | [46] |
| Normalized Difference Green–Blue Index (NDGBI) | (G − B)/(G + B) | [47] |
| Normalized Difference Red–Blue Index (NDRBI) | (R − B)/(R + B) | [47] |
| Treatment | Cornstalk NO3–N Content (ppm) | Grain Yield (kg/Plot) | Grain Yield (t/ha) | Grain Moisture (%) | Grain Test Weight (kg/hl) | Corncobs (n) |
|---|---|---|---|---|---|---|
| T01R01 | 67 | 9.81 | 10.283 | 21.0 | 72.3 | 63 |
| T01R02 | 364 | 7.07 | 7.411 | 21.7 | 70.4 | 53 |
| T01R03 | 1656 | 6.54 | 6.855 | 22.1 | 70.0 | 51 |
| T01R04 | 678 | 6.61 | 6.929 | 20.0 | 73.6 | 53 |
| T02R01 | 2003 | 17.78 | 18.637 | 20.8 | 72.1 | 71 |
| T02R02 | 4467 | 17.27 | 18.102 | 20.4 | 72.1 | 72 |
| T02R03 | 16,377 | 14.54 | 15.241 | 21.1 | 70.7 | 60 |
| T02R04 | 4495 | 16.51 | 17.306 | 19.7 | 74.1 | 73 |
| T03R01 | 3734 | 18.93 | 19.842 | 20.5 | 73.1 | 72 |
| T03R02 | 8121 | 17.27 | 18.103 | 21.5 | 72.2 | 72 |
| T03R03 | 7831 | 15.49 | 16.236 | 20.7 | 72.6 | 64 |
| T03R04 | 5952 | 17.32 | 18.155 | 20.0 | 74.2 | 73 |
| T04R01 | 12,717 | 19.83 | 20.786 | 21.3 | 73.2 | 82 |
| T04R02 | 10,018 | 17.29 | 18.123 | 21.8 | 70.3 | 74 |
| T04R03 | 5769 | 17.61 | 18.459 | 21.3 | 71.9 | 72 |
| T04R04 | 4040 | 18.16 | 19.035 | 20.0 | 75.8 | 81 |
| T05R01 | 555 | 21.49 | 22.526 | 20.3 | 73.7 | 83 |
| T05R02 | 7815 | 18.13 | 19.004 | 21.3 | 72.5 | 67 |
| T05R03 | 9110 | 18.66 | 19.559 | 21.5 | 72.7 | 80 |
| T05R04 | 8142 | 17.97 | 18.836 | 20.0 | 73.4 | 72 |
| T06R01 | 1412 | 17.06 | 17.882 | 20.2 | 73.5 | 74 |
| T06R02 | 18,702 | 15.86 | 16.624 | 21.4 | 71.3 | 66 |
| T06R03 | 6002 | 17.92 | 18.784 | 20.9 | 71.7 | 75 |
| T06R04 | 14,679 | 18.50 | 19.392 | 20.9 | 72.1 | 79 |
| Vegetation Index | Date | R2 | RMSE |
|---|---|---|---|
| NDVI | June | 0.0005 | 0.039 |
| July | 0.284 | 0.023 | |
| August | 0.508 | 0.024 | |
| September | 0.647 | 0.037 | |
| GNDVI | June | 0.145 | 0.032 |
| July | 0.831 | 0.021 | |
| August | 0.868 | 0.024 | |
| September | 0.919 | 0.028 |
| Index | Date | R2 | RMSE |
|---|---|---|---|
| NDGBI | June | 0.041 | 0.038 |
| July | 0.246 | 0.052 | |
| August | 0.707 | 0.043 | |
| September | 0.861 | 0.035 | |
| NDRBI | June | 0.021 | 0.037 |
| July | 0.068 | 0.047 | |
| August | 0.484 | 0.055 | |
| September | 0.675 | 0.069 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mhaidat, M.; González-Pérez, I.; Rodríguez-Pérez, J.R.; Val-Aguasca, J.P.; Sanz-Ablanedo, E. Potential of RGB-Derived Vegetation Indices as an Alternative to NIR-Based Vegetation Indices to Monitor Nitrogen Status in Maize. Remote Sens. 2026, 18, 528. https://doi.org/10.3390/rs18030528
Mhaidat M, González-Pérez I, Rodríguez-Pérez JR, Val-Aguasca JP, Sanz-Ablanedo E. Potential of RGB-Derived Vegetation Indices as an Alternative to NIR-Based Vegetation Indices to Monitor Nitrogen Status in Maize. Remote Sensing. 2026; 18(3):528. https://doi.org/10.3390/rs18030528
Chicago/Turabian StyleMhaidat, Mohammad, Iván González-Pérez, José Ramón Rodríguez-Pérez, Jesús P. Val-Aguasca, and Enoc Sanz-Ablanedo. 2026. "Potential of RGB-Derived Vegetation Indices as an Alternative to NIR-Based Vegetation Indices to Monitor Nitrogen Status in Maize" Remote Sensing 18, no. 3: 528. https://doi.org/10.3390/rs18030528
APA StyleMhaidat, M., González-Pérez, I., Rodríguez-Pérez, J. R., Val-Aguasca, J. P., & Sanz-Ablanedo, E. (2026). Potential of RGB-Derived Vegetation Indices as an Alternative to NIR-Based Vegetation Indices to Monitor Nitrogen Status in Maize. Remote Sensing, 18(3), 528. https://doi.org/10.3390/rs18030528











