On-Site Soil Monitoring Using Photonics-Based Sensors and Historical Soil Spectral Libraries
Abstract
1. Introduction
2. Materials and Methods
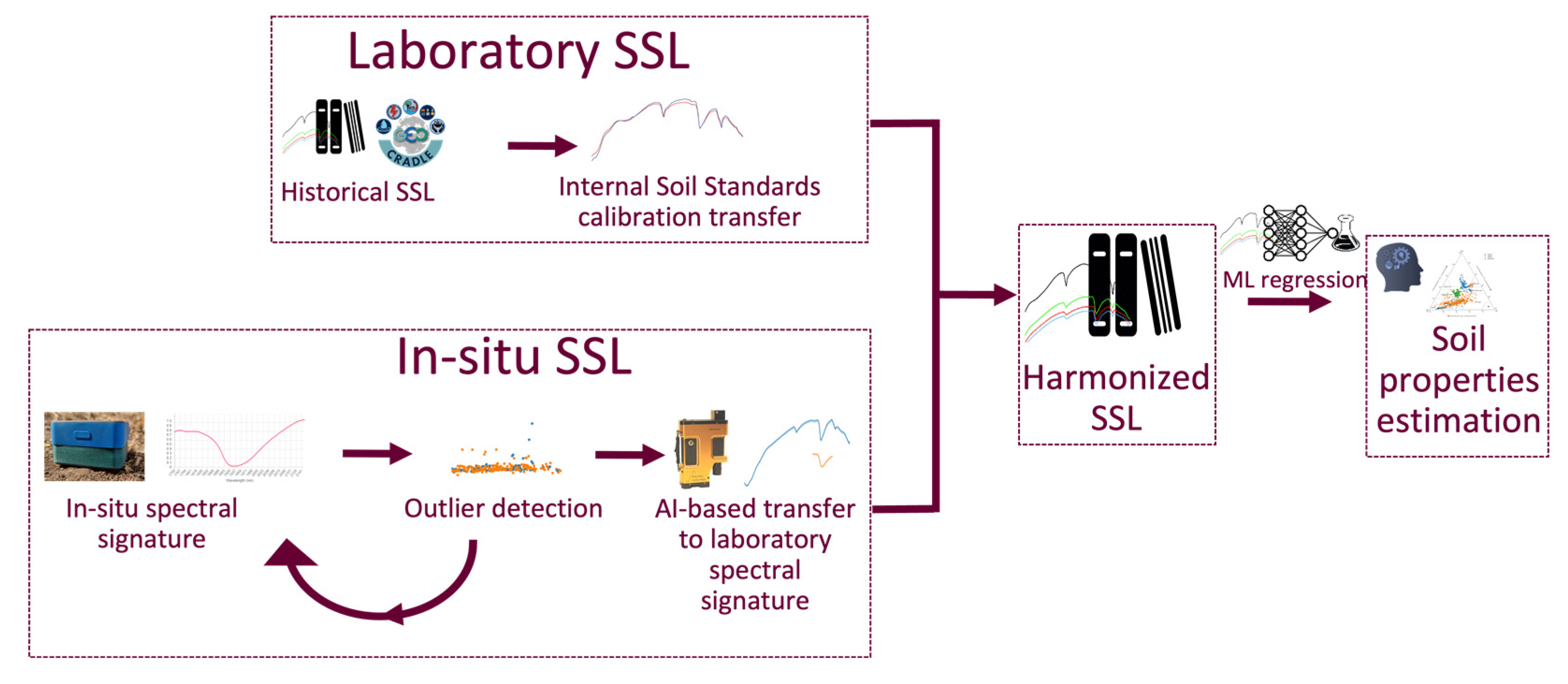
2.1. Overall Methodology
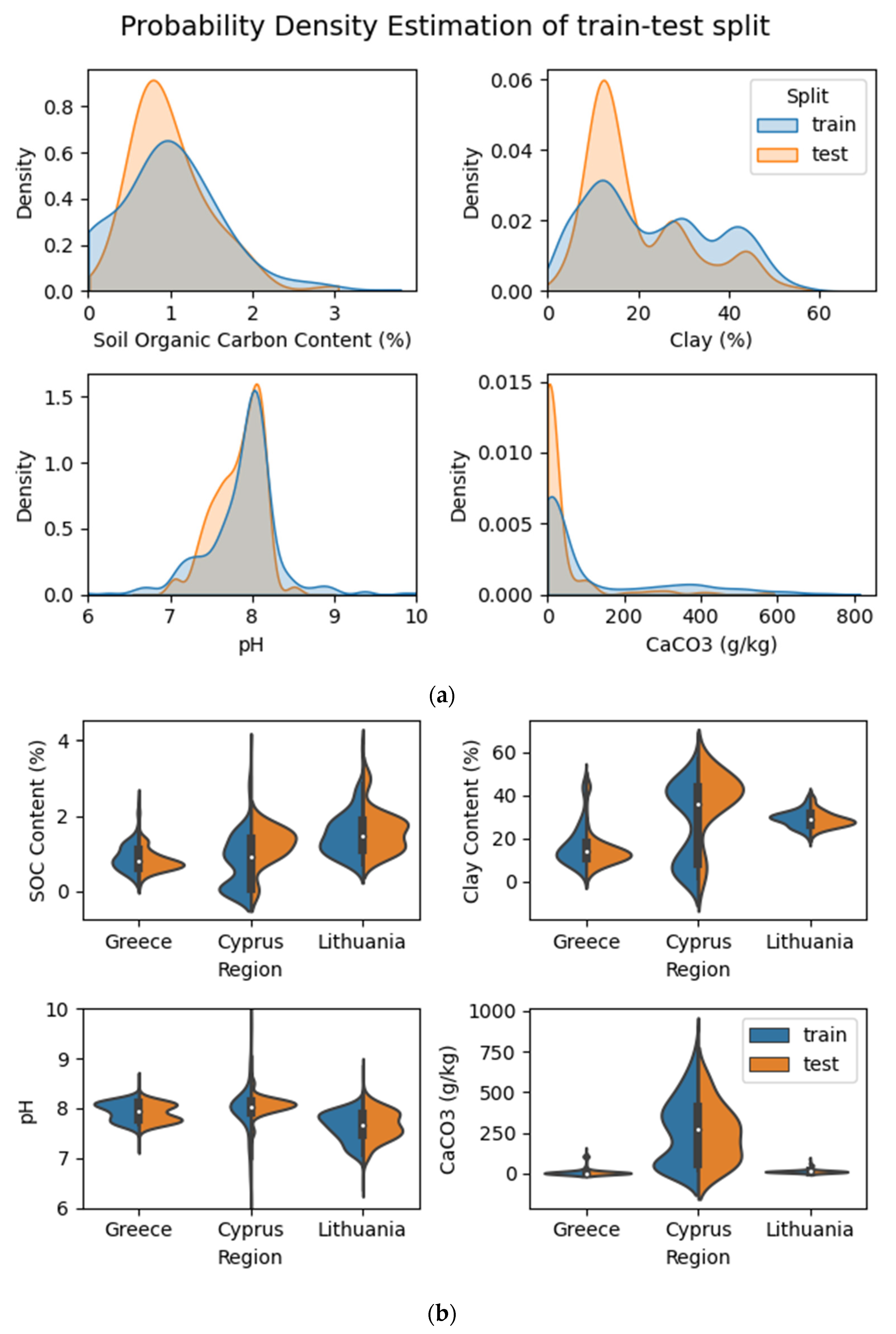
2.2. Data Collection
2.3. Chemical Analyses
- Texture (Sand, Silt, and Clay—%);
- Soil organic carbon content (SOC—%);
- pH;
- Calcium carbonates (CaCO3—g/kg).
2.4. Spectral Analyses
2.4.1. Spectral Measurements
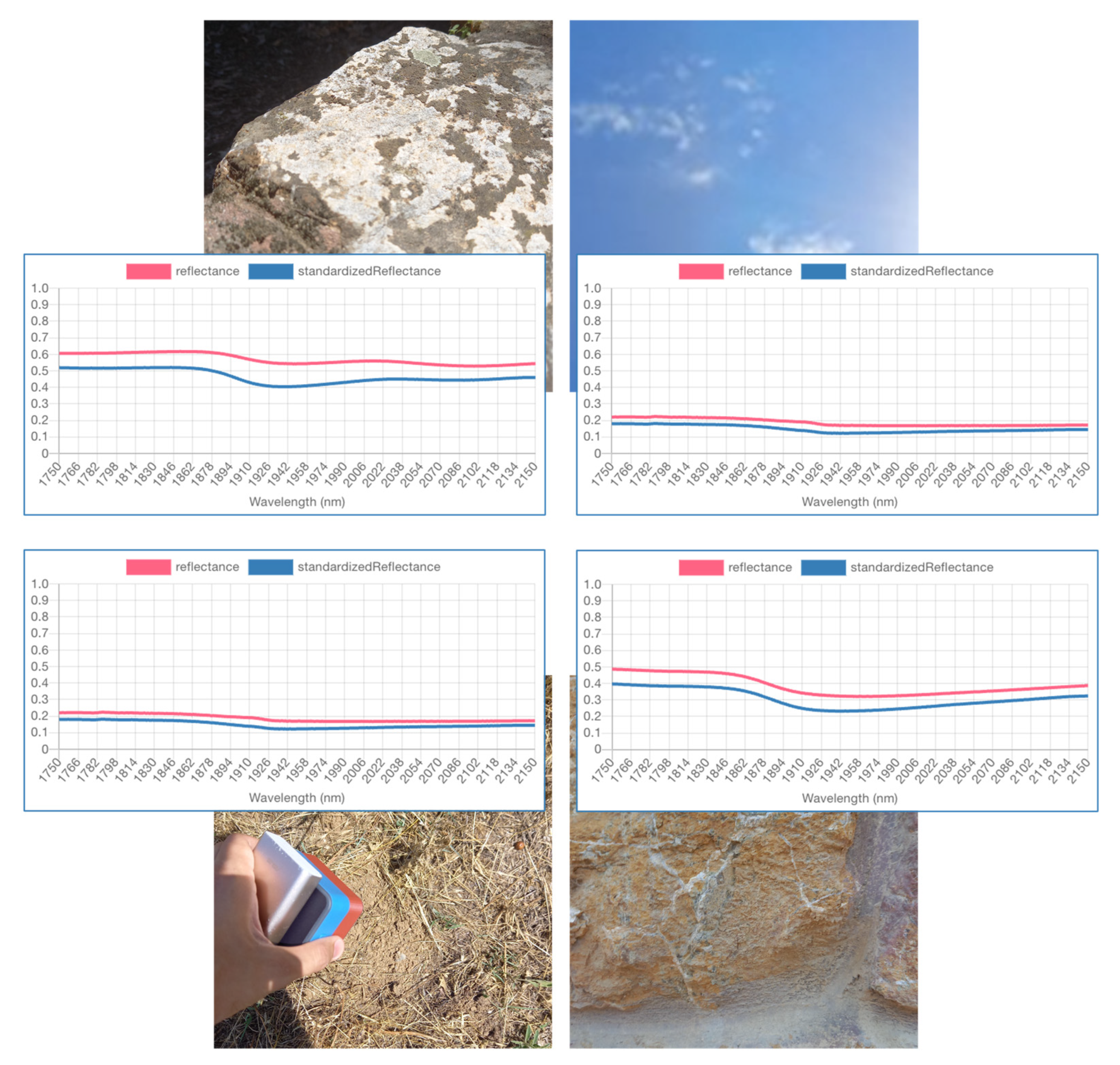
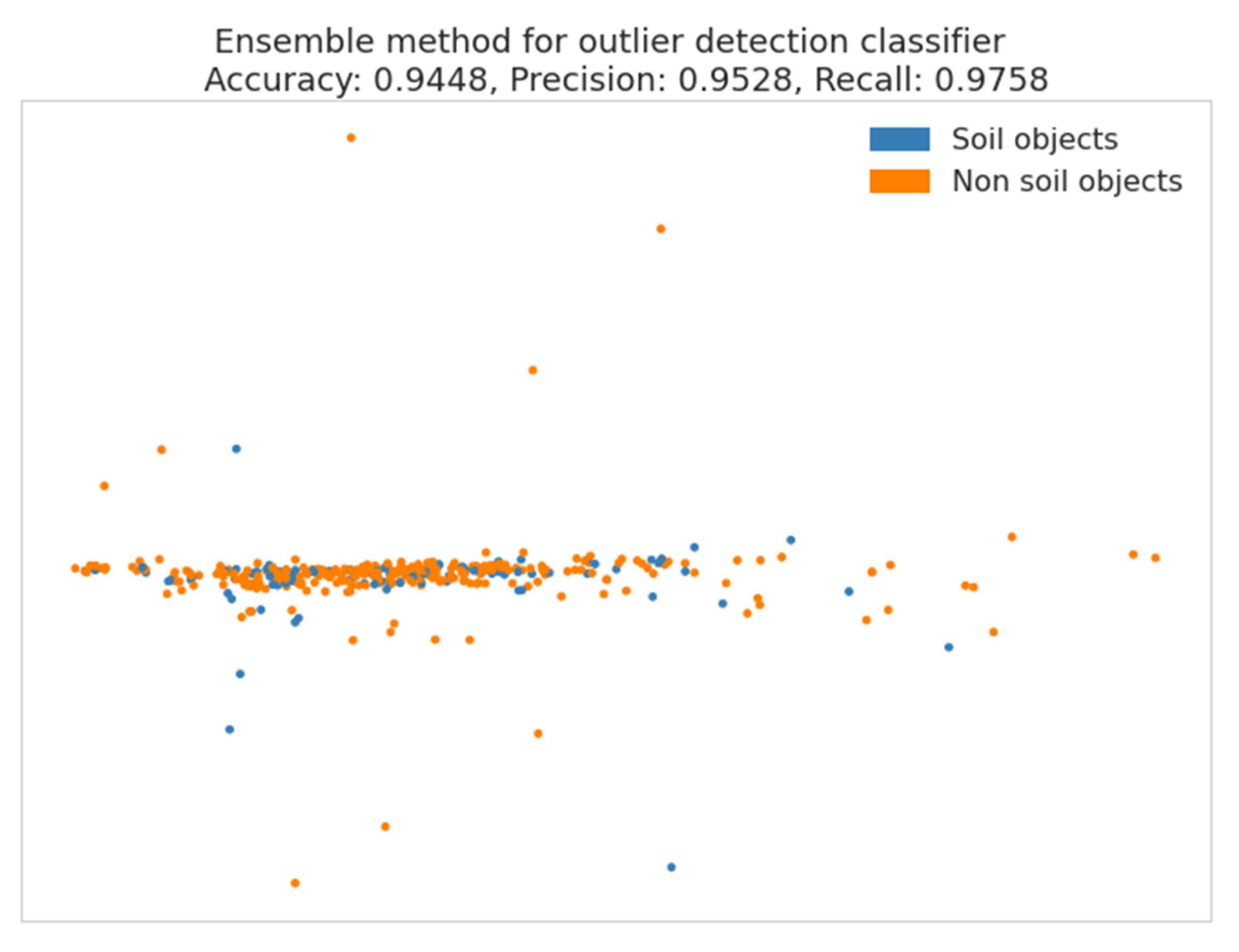
2.4.2. Outlier Detection
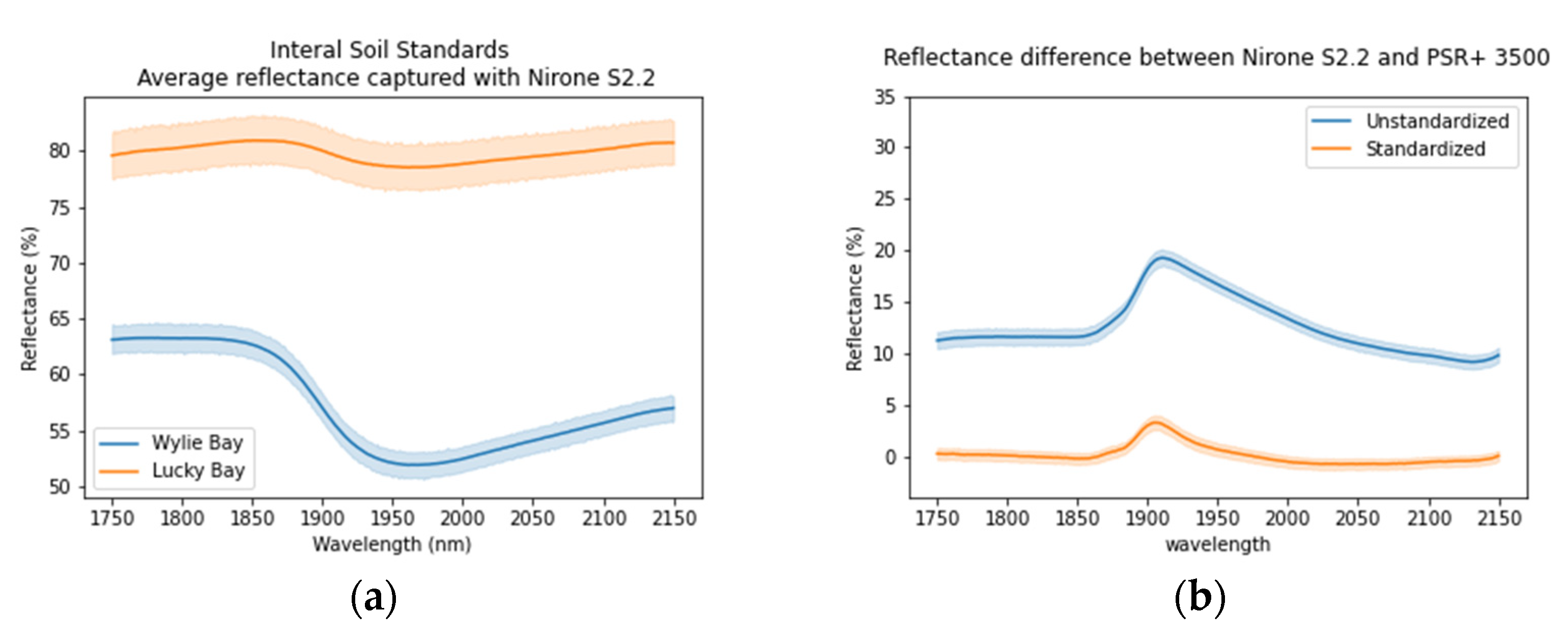
2.4.3. Ambient Factors Effect Elimination
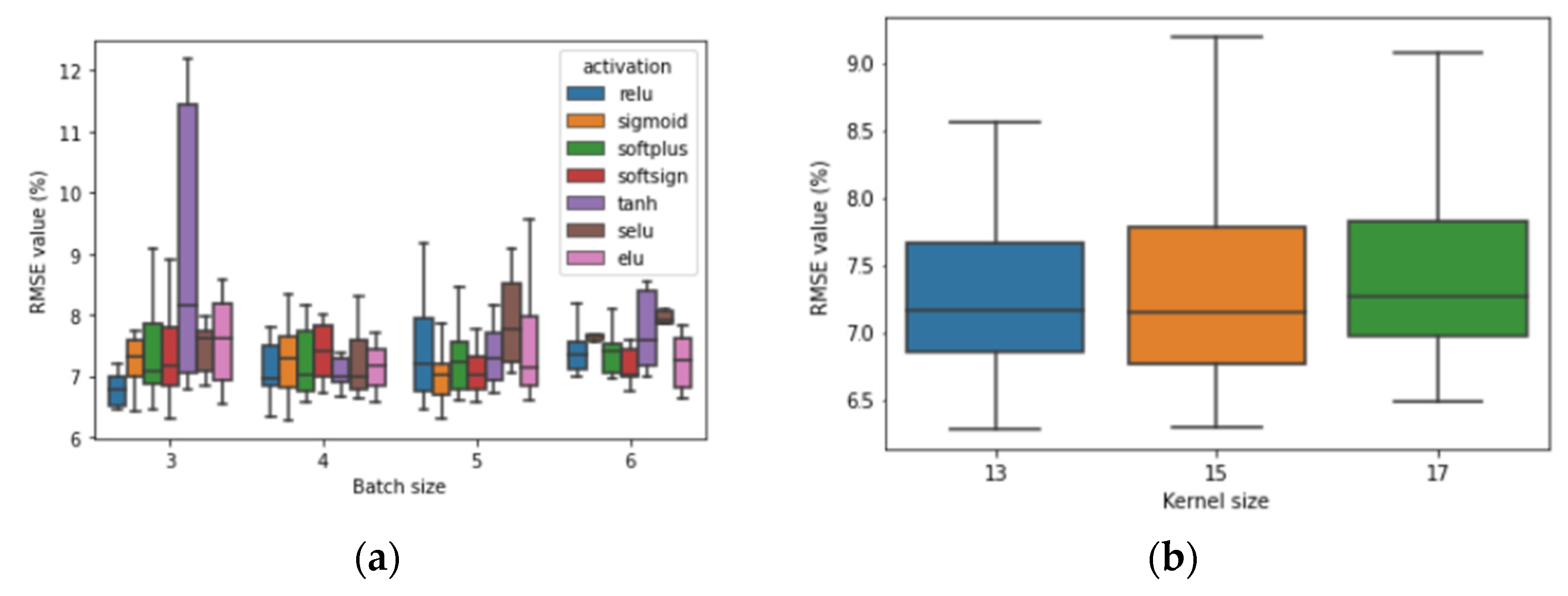
2.5. Modeling Soil Properties
3. Results and Discussion
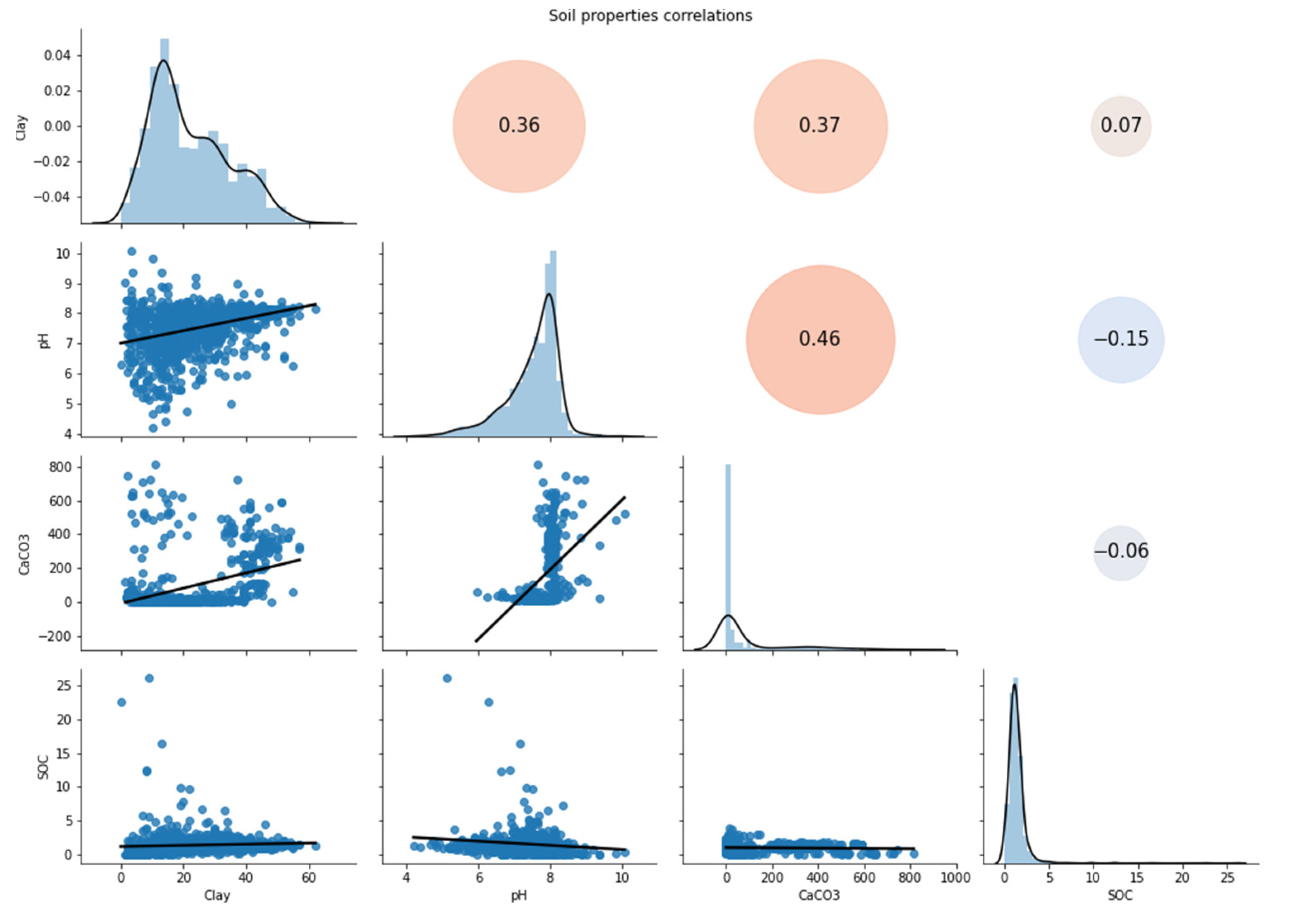
3.1. Chemical Results
3.2. Spectral Measurements
3.3. Outlier Detection
3.4. Ambient Factors Effect Elimination
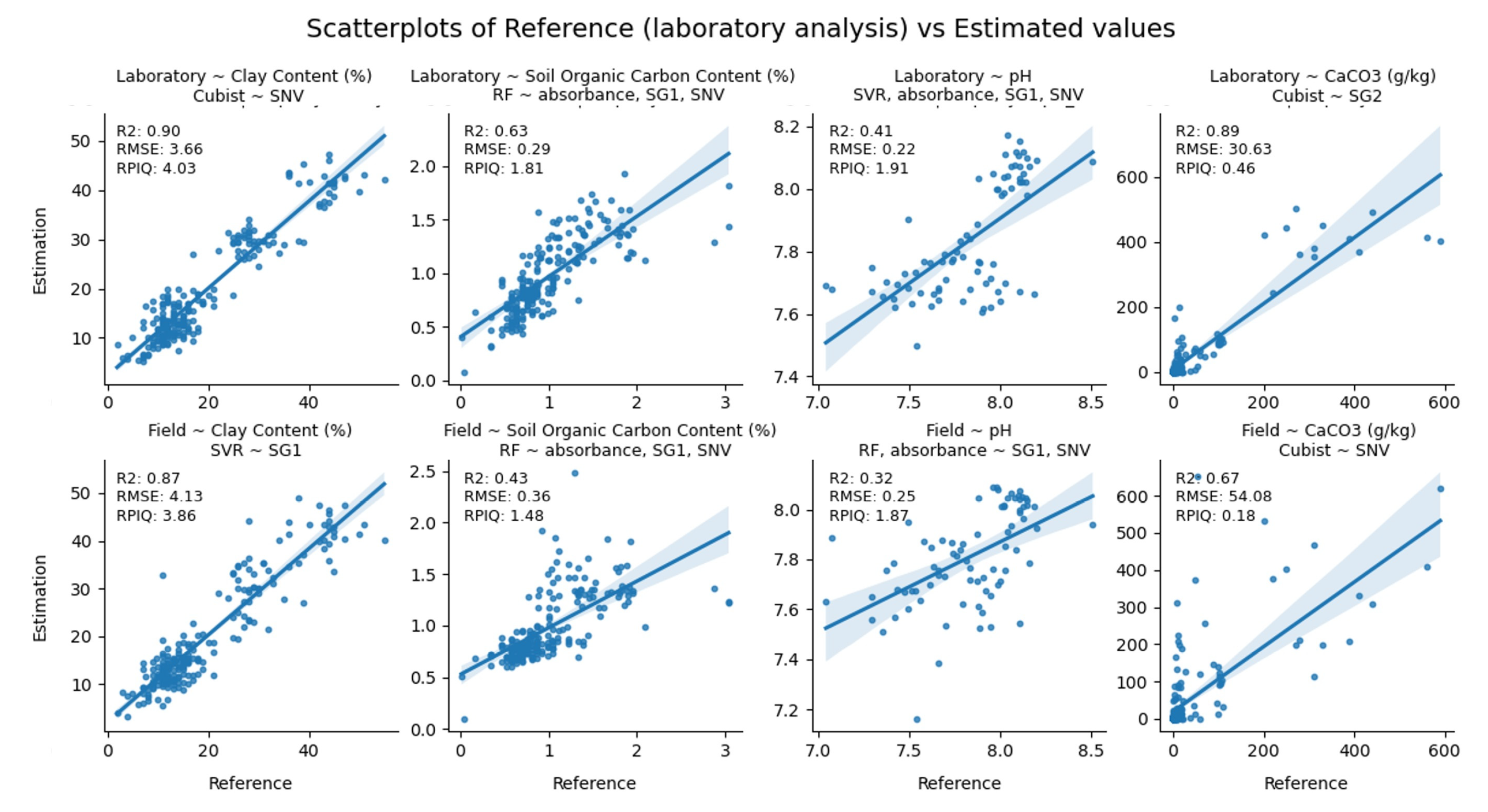
3.5. Modeling Assessment
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Silvero, N.E.; Demattê, J.A.; Minasny, B.; Rosin, N.A.; Nascimento, J.G.; Rodríguez Albarracín, H.S.; Sparks, D.L. Chapter Three-Sensing technologies for characterizing and monitoring soil functions: A review. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 2023; Volume 177, pp. 125–168. [Google Scholar] [CrossRef]
- Griffiths, P.R. Günter Gauglitz and David S. Moore (Eds.): Handbook of spectroscopy, 4 volume set, 2nd ed. Anal. Bioanal. Chem. 2014, 406, 7415–7416. [Google Scholar] [CrossRef]
- Di Raimo, L.A.; Couto, E.G.; de Mello, D.C.; Demattê, J.A.; Amorim, R.S.; Torres, G.N.; Fernandes-Filho, E.I. Characterizing and Modeling Tropical Sandy Soils through Vis-NIR-SWIR, MIR Spectroscopy, and X-ray Fluorescence. Remote Sens. 2022, 14, 4823. [Google Scholar] [CrossRef]
- Parson, W.W. Modern Optical Spectroscopy; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar] [CrossRef]
- Poppiel, R.R.; Paiva, A.F.; Demattê, J.A. Bridging the gap between soil spectroscopy and traditional laboratory: Insights for routine implementation. Geoderma 2022, 425, 116029. [Google Scholar] [CrossRef]
- Tang, Y.; Jones, E.; Minasny, B. Evaluating low-cost portable near infrared sensors for rapid analysis of soils from South Eastern Australia. Geoderma Reg. 2020, 20, e00240. [Google Scholar] [CrossRef]
- Priori, S.; Mzid, N.; Pascucci, S.; Pignatti, S.; Casa, R. Performance of a Portable FT-NIR MEMS Spectrometer to Predict Soil Features. Soil Syst. 2022, 6, 66. [Google Scholar] [CrossRef]
- Shen, Z.; D’Agui, H.; Walden, L.; Zhang, M.; Yiu, T.M.; Dixon, K.; Viscarra Rossel, R.A. Miniaturised visible and near-infrared spectrometers for assessing soil health indicators in mine site rehabilitation. Soil 2022, 8, 467–486. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Banin, A. Near-infrared analysis as a rapid method to simultaneously evaluate several soil properties. Soil Sci. Soc. Am. J. 1995, 59, 364–372. [Google Scholar] [CrossRef]
- Summers, D.; Lewis, M.; Ostendorf, B.; Chittleborough, D. Visible near-infrared reflectance spectroscopy as a predictive indicator of soil properties. Ecol. Indic. 2011, 11, 123–131. [Google Scholar] [CrossRef]
- Tsakiridis, N.L.; Keramaris, K.D.; Theocharis, J.B.; Zalidis, G.C. Simultaneous prediction of soil properties from VNIR-SWIR spectra using a localized multi-channel 1-D convolutional neural network. Geoderma 2020, 367, 114208. [Google Scholar] [CrossRef]
- Francos, N.; Ben-Dor, E. A transfer function to predict soil surface reflectance from laboratory soil spectral libraries. Geoderma 2022, 405, 115432. [Google Scholar] [CrossRef]
- Ackerson, J.P.; Demattê, J.A.; Morgan, C.L. Predicting clay content on field-moist intact tropical soils using a dried, ground VisNIR library with external parameter orthogonalization. Geoderma 2015, 259–260, 196–204. [Google Scholar] [CrossRef]
- Yang, M.; Chen, S.; Li, H.; Zhao, X.; Shi, Z. Effectiveness of different approaches for in situ measurements of organic carbon using visible and near infrared spectrometry in the Poyang Lake basin area. Land Degrad. Dev. 2021, 32, 1301–1311. [Google Scholar] [CrossRef]
- Knadel, M.; Castaldi, F.; Barbetti, R.; Ben-Dor, E.; Gholizadeh, A.; Lorenzetti, R. Mathematical techniques to remove moisture effects from visible–near-infrared–shortwave-infrared soil spectra—Review. Appl. Spectrosc. Rev. 2022, 1–34. [Google Scholar] [CrossRef]
- Ballabio, C.; Panagos, P.; Monatanarella, L. Mapping topsoil physical properties at European scale using the LUCAS database. Geoderma 2016, 261, 110–123. [Google Scholar] [CrossRef]
- Nocita, M.; Stevens, A.; van Wesemael, B.; Brown, D.J.; Shepherd, K.D.; Towett, E.; Montanarella, L. Soil spectroscopy: An opportunity to be seized. Glob. Chang. Biol. 2015, 21, 10–11. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, I.; Seidel, M.; Vohland, M.; Koch, H.-J.; Ludwig, B. Performance of in situ vs laboratory mid-infrared soil spectroscopy using local and regional calibration strategies. Geoderma 2022, 409, 115614. [Google Scholar] [CrossRef]
- Guerrero, C.; Stenberg, B.; Wetterlind, J.; Viscarra Rossel, R.A.; Maestre, F.T.; Mouazen, A.M.; Kuang, B. Assessment of soil organic carbon at local scale with spiked NIR calibrations: Effects of selection and extra-weighting on the spiking subset. Eur. J. Sci. 2014, 65, 248–263. [Google Scholar] [CrossRef]
- Pimstein, A.; Notesco, G.; Ben-Dor, E. Performance of Three Identical Spectrometers in Retrieving Soil Reflectance under Laboratory Conditions. Soil Sci. Soc. Am. J. 2011, 75, 746–759. [Google Scholar] [CrossRef]
- Tsakiridis, N.L.; Tziolas, N.; Theocharis, J.; Zalidis, G. A genetic algorithm-based stacking algorithm for predicting soil organic matter from vis–NIR spectral data. Eur. J. Soil Sci. 2019, 70, 578–590. [Google Scholar] [CrossRef]
- Minasny, B.; McBratney, A. A conditioned Latin hypercube method for sampling in the presence of ancillary information. Comput. Geosci. 2006, 13, 1378–1388. [Google Scholar] [CrossRef]
- Demattê, J.A.; Huete, A., Jr.; Ferreira, L.; Nanni, M.; Alves, M.; Fiorio, P. Methodology for Bare Soil Detection and Discrimination by Landsat TM Image. Open Remote Sens. J. 2009, 2, 24–35. [Google Scholar] [CrossRef]
- Castaldi, F.; Chabrillat, S.; Don, A.; van Wesemael, B. Soil Organic Carbon Mapping Using LUCAS Topsoil Database and Sentinel-2 Data: An Approach to Reduce Soil Moisture and Crop Residue Effects. Remote Sens. 2019, 11, 2121. [Google Scholar] [CrossRef]
- Farr, T.G.; Rosen, P.A.; Caro, E.; Crippen, R.; Duren, R.; Hensley, S.; Kobrick, M.; Paller, M.; Rodriguez, E.; Roth, L.; et al. The Shuttle Radar Topography Mission. Rev. Geophys. 2007, 45, 1–33. [Google Scholar] [CrossRef]
- Poggio, L.; de Sousa, L.M.; Batjes, N.H.; Heuvelink, G.B.M.; Kempen, B.; Ribeiro, E.; Rossiter, D. SoilGrids 2.0: Producing soil information for the globe with quantified spatial uncertainty. Soil 2021, 7, 217–240. [Google Scholar] [CrossRef]
- Bouyoucos, G.J. Hydrometer Method Improved for Making Particle Size Analysis of Soils. Agron. J. 1962, 54, 464–465. [Google Scholar] [CrossRef]
- Walkley, A.; Black, A. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Rhoades, J.D.; Manteghi, N.A.; Shouse, P.J.; Alves, W.J. Estimating soil salinity from saturated soil-paste electrical conductivity. Soil Sci. Soc. Am. J. 1989, 53, 428–433. [Google Scholar] [CrossRef]
- Ben Dor, E.; Ong, C.; Lau, I.C. Reflectance measurements of soils in the laboratory: Standards and protocols. Geoderma 2015, 245–246, 112–124. [Google Scholar] [CrossRef]
- Karyotis, K.; Angelopoulou, T.; Tziolas, N.; Palaiologou, E.; Samarinas, N.; Zalidis, G. Evaluation of a Micro-Electro Mechanical Systems Spectral Sensor for Soil Properties Estimation. Land 2021, 10, 63. [Google Scholar] [CrossRef]
- Ganaie, M.A.; Hu, M.; Malik, A.K.; Tanveer, M.; Suganthan, P.N. Ensemble deep learning: A review. Eng. Appl. Artif. Intell. 2022, 115, 105151. [Google Scholar] [CrossRef]
- Brieman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Max, K.; Weston, S.; Keefer, C.; Coulter, N.; Quinlan, R. Cubist: Rule-and Instance-Based Regression Modeling. R Package Version 0.0 13 (2014). 2014. Available online: https://cran.r-project.org/web/packages/Cubist/Cubist.pdf (accessed on 30 January 2023).
- Quinlan, R. Combining Instance-Based and Model-Based Learning. In Proceedings of the Machine Learning Proceedings 1993, Amherst, MA, USA, 27–29 June 1993; pp. 236–243. Available online: https://dl.acm.org/doi/10.5555/3091529.3091560 (accessed on 30 January 2023).
- Drucker, H.; Burges, C.J.; Kaufman, L.; Smola, A.; Vapnik, V. Support vector regression machines. Adv. Neural Inf. Process. Syst. 1996, 9, 155–161. [Google Scholar]
- Savitzky, A.; Golay, M.J. Smoothing and Differentiation of Data by Simplified Least Squares Procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Rinnan, A.; van den Berg, F.; Engelsen, S.B. Review of the most common pre-processing techniques for near-infrared spectra. TrAC Trends Anal. Chem. 2009, 28, 1201–1222. [Google Scholar] [CrossRef]
- Kennard, R.W.; Stone, L.A. Computer Aided Design of Experiments. Technometrics 1969, 11, 137–148. [Google Scholar] [CrossRef]
- Camera, C.; Zomeni, Z.; Noller, J.; Zissimos, A.; Christoforou, I.; Bruggeman, A. A high resolution map of soil types and physical properties for Cyprus: A digital soil mapping optimization. Geoderma 2017, 285, 35–49. [Google Scholar] [CrossRef]
- Buivydaitė, V.V. Soil Survey and Available Soil Date in Lihuania; Research Report; Europian Soil Bureau: Akademija-Kaunas, Lithuana, 2001; Volume 9, pp. 211–223. Available online: https://esdac.jrc.ec.europa.eu/ESDB_Archive/eusoils_docs/esb_rr/n09_soilresources_of_europe/Lithuania.pdf (accessed on 15 March 2023).
- Tziolas, N.; Tsakiridis, N.; Ben-Dor, E.; Theocharis, J.; Zalidis, G. A memory-based learning approach utilizing combined spectral sources and geographical proximity for improved VIS-NIR-SWIR soil properties estimation. Geoderma 2019, 340, 11–24. [Google Scholar] [CrossRef]
- Oshunsanya, S. Introductory Chapter: Relevance of Soil pH to Agriculture. In Soil pH for Nutrient Availability and Crop Performance; Oshunsanya, S., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Hong, S.; Gan, P.; Chen, A. Environmental controls on soil pH in planted forest and its response to nitrogen deposition. Environ. Res. 2019, 172, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Hassink, J. The capacity of soils to preserve organic C and N by their association with clay and silt particles. Plant Soil 1997, 191, 77–87. [Google Scholar] [CrossRef]
- Six, J.; Conant, R.T.; Paul, E.A.; Paustian, K. Stabilization mechanisms of soil organic matter: Implications for C-saturation of soils. Plant Soil 2002, 241, 155–176. [Google Scholar] [CrossRef]
- FAO. Global Soil Organic Carbon Sequestration Potential Map–SOCseq v.1.1; Technical Report; FAO: Rome, Italy, 2022. [Google Scholar] [CrossRef]
- You, J.; Xing, L.; Liang, L.; Pan, J.; Meng, T. Application of short-wave infrared (SWIR) spectroscopy in quantitative estimation of clay mineral contents. IOP Conf. Ser. Earth Environ. Sci. 2014, 17, 012256. [Google Scholar] [CrossRef]
- Stenberg, B.; Viscarra Rossel, R.A.; Mouazen, A.M.; Wetterlind, J. Chapter Five-Visible and Near Infrared Spectroscopy in Soil Science. Adv. Agron. 2010, 107, 163–215. [Google Scholar] [CrossRef]
- Gras, J.-P.; Barthès, B.G.; Mahaut, B.; Trupin, S. Best practices for obtaining and processing field visible and near infrared (VNIR) spectra of topsoils. Geoderma 2014, 214–215, 126–134. [Google Scholar] [CrossRef]
- Emery, L.P.; Powell, J.W. The hydration dependence of CaCO3 absorption lines in the Far IR. In American Astronomical Society Meeting Abstracts; American Astronomical Society: Washington, DC, USA, 2014; Volume 224, Available online: https://ui.adsabs.harvard.edu/abs/2014AAS...22422011P (accessed on 30 January 2023).
- Viscarra Rossel, R.A.; Cattle, S.R.; Ortega, A.; Fouad, Y. In situ measurements of soil colour, mineral composition and clay content by vis–NIR spectroscopy. Geoderma 2009, 150, 253–266. [Google Scholar] [CrossRef]
- Ji, W.; Shi, Z.; Huang, J.; Li, S. In Situ Measurement of Some Soil Properties in Paddy Soil Using Visible and Near-Infrared Spectroscopy. PLoS ONE 2016, 11, e0159785. [Google Scholar] [CrossRef]
- Hutengs, C.; Seidel, M.; Oertel, F.; Ludwig, B.; Vohland, M. In situ and laboratory soil spectroscopy with portable visible-to-near-infrared and mid-infrared instruments for the assessment of organic carbon in soils. Geoderma 2019, 355, 113900. [Google Scholar] [CrossRef]
- Greenberg, I.; Seidel, M.; Vohland, M.; Ludwig, B. Performance of field-scale lab vs in situ visible/near- and mid-infrared spectroscopy for estimation of soil properties. Eur. J. Soil Sci. 2021, 73, e13180. [Google Scholar] [CrossRef]
- Ballabio, C.; Lugato, E.; Fernández-Ugalde, O.; Orgiazzi, A.; Jones, A.; Borrelli, P.; Montanarella, L.; Panagos, P. Mapping LUCAS topsoil chemical properties at European scale using Gaussian process regression. Geoderma 2019, 355, 113912. [Google Scholar] [CrossRef] [PubMed]





| Soil Attribute | Sand (%) | Clay (%) | Silt (%) | pH | CaCO3 (g/kg) | SOC (%) |
|---|---|---|---|---|---|---|
| Augmented dataset (GEO-Cradle and collected samples) | ||||||
| Min | 9 | 1.5 | 6 | 5.95 | 0 | 0 |
| 1st quartile | 31 | 11 | 26 | 7.67 | 0 | 0.63 |
| Median | 45 | 18 | 31 | 7.95 | 10 | 0.91 |
| Mean | 45.87 | 22.24 | 31.89 | 7.87 | 91.32 | 0.98 |
| 3rd quartile | 59 | 32 | 39 | 8.09 | 80 | 1.32 |
| Max | 89 | 57 | 62 | 10.07 | 815 | 3.8 |
| Standard Deviation | 17.85 | 13.48 | 10.04 | 0.43 | 165.18 | 0.59 |
| Collected samples | ||||||
| Min | 12 | 10 | 13 | 7.04 | 3.00 | 0.70 |
| 1st quartile | 24 | 28.25 | 26 | 7.70 | 12.00 | 0.95 |
| Median | 32 | 35 | 29 | 7.95 | 36.50 | 1.29 |
| Mean | 34.68 | 34.87 | 30.46 | 7.86 | 142.46 | 1.35 |
| 3rd quartile | 45 | 43 | 34 | 8.07 | 280.00 | 1.59 |
| Max | 75 | 57 | 48 | 8.27 | 590.00 | 3.80 |
| Standard Deviation | 14.43 | 10.12 | 7.12 | 0.28 | 168.16 | 0.49 |
| GEO-Cradle | ||||||
| Min | 9 | 1.50 | 6 | 5.95 | 0.00 | 0.00 |
| 1st quartile | 43 | 9 | 24 | 7.46 | 0.00 | 0.47 |
| Median | 54 | 13 | 33 | 7.97 | 2.00 | 0.70 |
| Mean | 53.66 | 13.47 | 32.89 | 7.91 | 55.98 | 0.74 |
| 3rd quartile | 63.80 | 17 | 41 | 8.35 | 20.00 | 0.99 |
| Max | 89 | 48 | 62 | 10.07 | 815.00 | 3.66 |
| Standard Deviation | 15.74 | 7.02 | 11.54 | 0.72 | 153.44 | 0.54 |
| Actual Value | |||
|---|---|---|---|
| True | False | ||
| Predicted value | True | 33 | 6 |
| False | 3 | 121 | |
| Set-Up | Selected Model | Property | R2 | RMSE | RPIQ |
|---|---|---|---|---|---|
| Laboratory | Cubist | Clay | 0.90 | 3.66% | 4.03 |
| RF | SOC | 0.63 | 0.29% | 1.81 | |
| SVR | pH | 0.41 | 0.22 | 1.91 | |
| Cubist | CaCO3 | 0.89 | 30.63 (g/kg) | 0.46 | |
| In-situ | SVR | Clay | 0.87 | 4.13% | 3.86 |
| RF | SOC | 0.43 | 0.36% | 1.48 | |
| RF | pH | 0.32 | 0.25 | 1.87 | |
| Cubist | CaCO3 | 0.67 | 54.08 (g/kg) | 0.18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karyotis, K.; Tsakiridis, N.L.; Tziolas, N.; Samarinas, N.; Kalopesa, E.; Chatzimisios, P.; Zalidis, G. On-Site Soil Monitoring Using Photonics-Based Sensors and Historical Soil Spectral Libraries. Remote Sens. 2023, 15, 1624. https://doi.org/10.3390/rs15061624
Karyotis K, Tsakiridis NL, Tziolas N, Samarinas N, Kalopesa E, Chatzimisios P, Zalidis G. On-Site Soil Monitoring Using Photonics-Based Sensors and Historical Soil Spectral Libraries. Remote Sensing. 2023; 15(6):1624. https://doi.org/10.3390/rs15061624
Chicago/Turabian StyleKaryotis, Konstantinos, Nikolaos L. Tsakiridis, Nikolaos Tziolas, Nikiforos Samarinas, Eleni Kalopesa, Periklis Chatzimisios, and George Zalidis. 2023. "On-Site Soil Monitoring Using Photonics-Based Sensors and Historical Soil Spectral Libraries" Remote Sensing 15, no. 6: 1624. https://doi.org/10.3390/rs15061624
APA StyleKaryotis, K., Tsakiridis, N. L., Tziolas, N., Samarinas, N., Kalopesa, E., Chatzimisios, P., & Zalidis, G. (2023). On-Site Soil Monitoring Using Photonics-Based Sensors and Historical Soil Spectral Libraries. Remote Sensing, 15(6), 1624. https://doi.org/10.3390/rs15061624










