Using Sentinel-2-Based Metrics to Characterize the Spatial Heterogeneity of FLEX Sun-Induced Chlorophyll Fluorescence on Sub-Pixel Scale
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Airborne Data
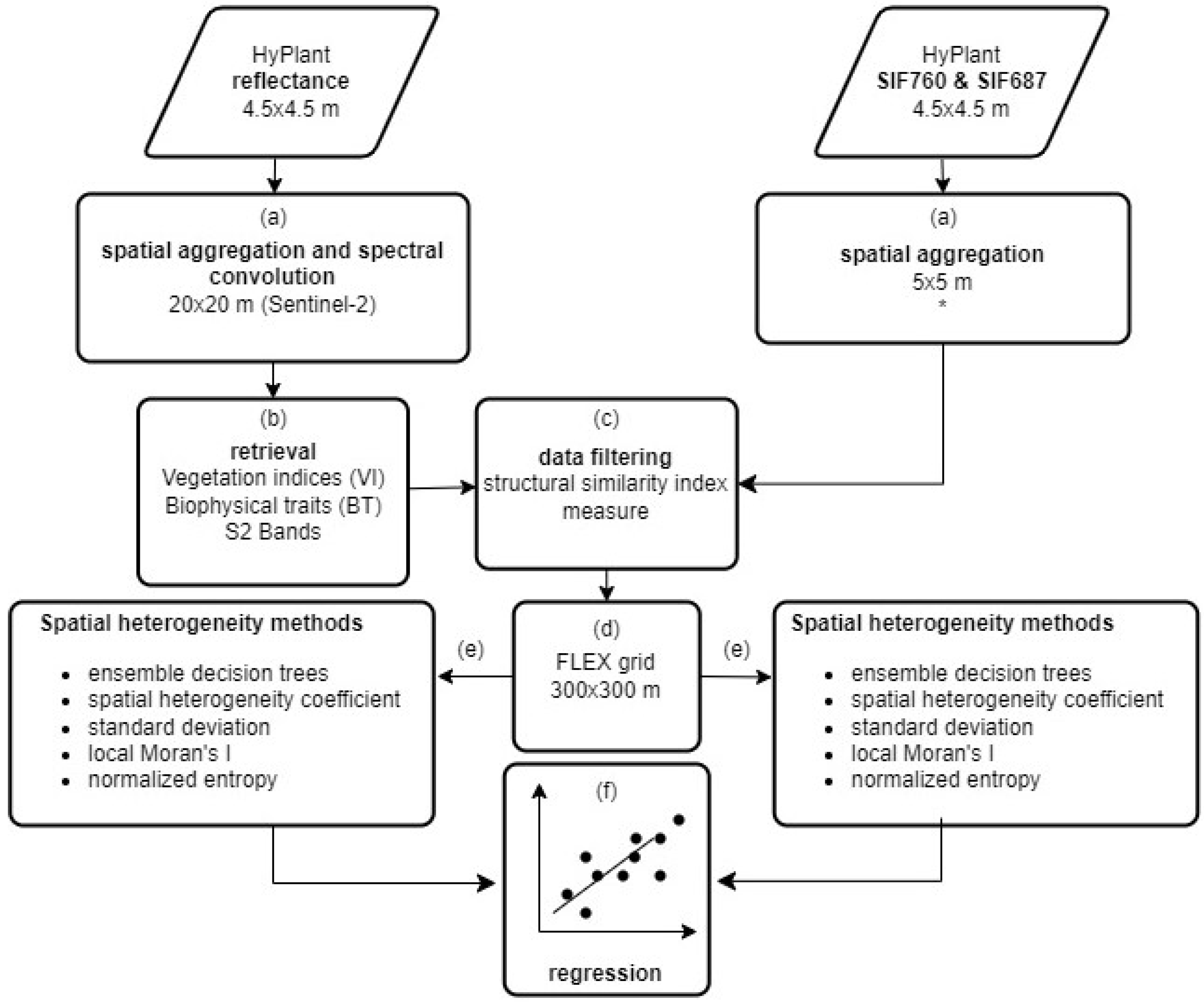
2.3. Data Processing Description and Heterogeneity Methods Evaluation
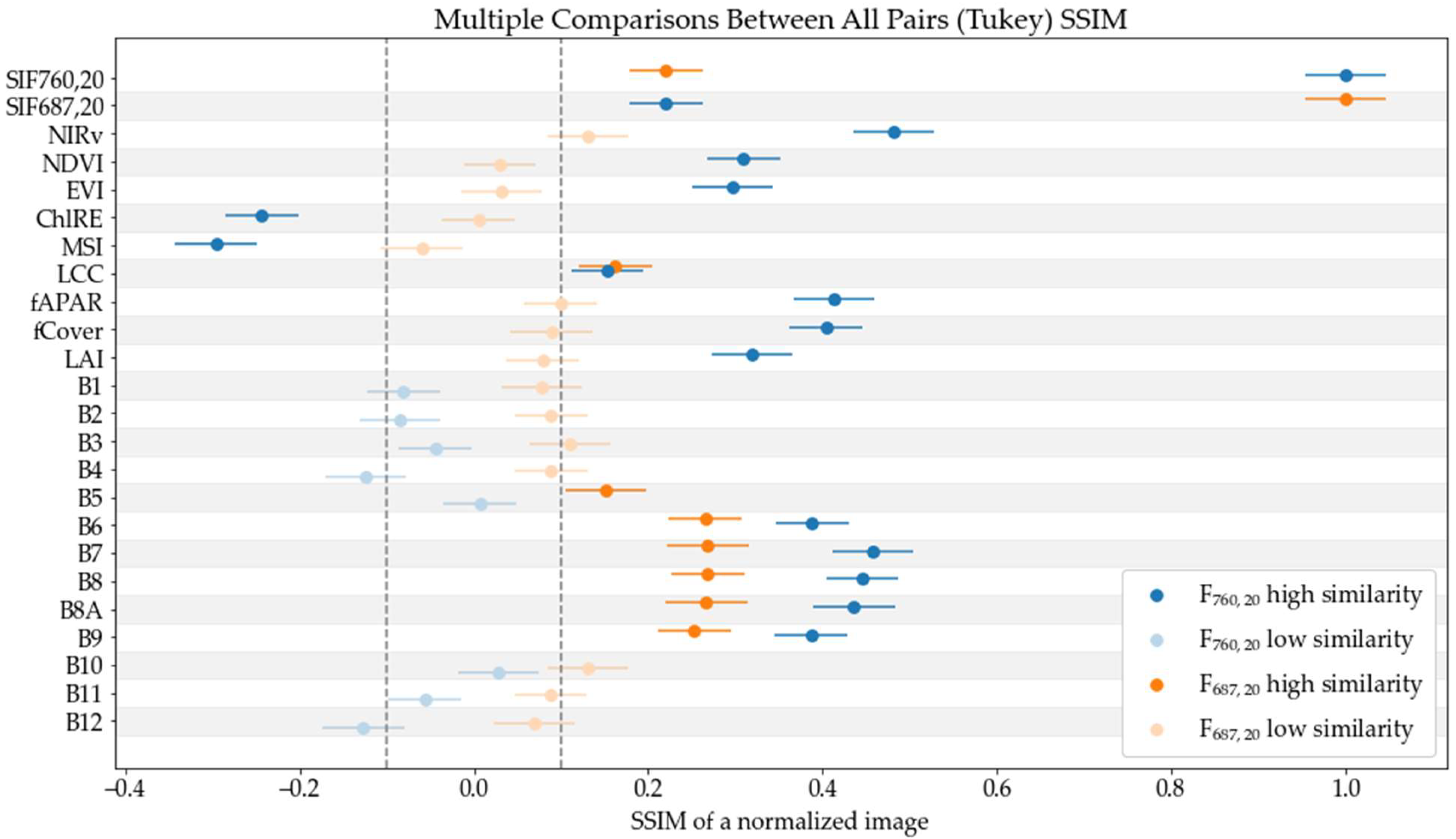
2.4. Data Preparation
- -
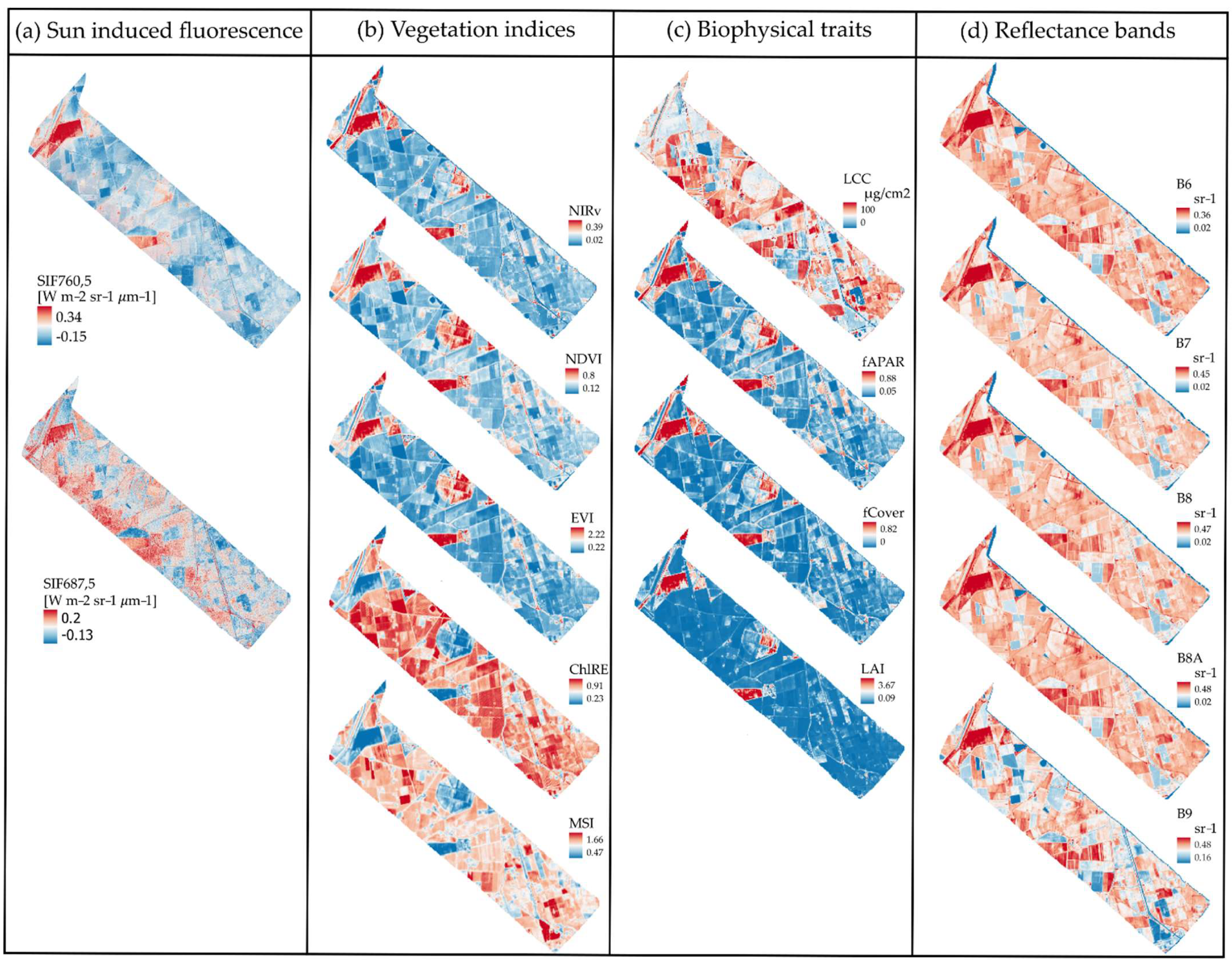
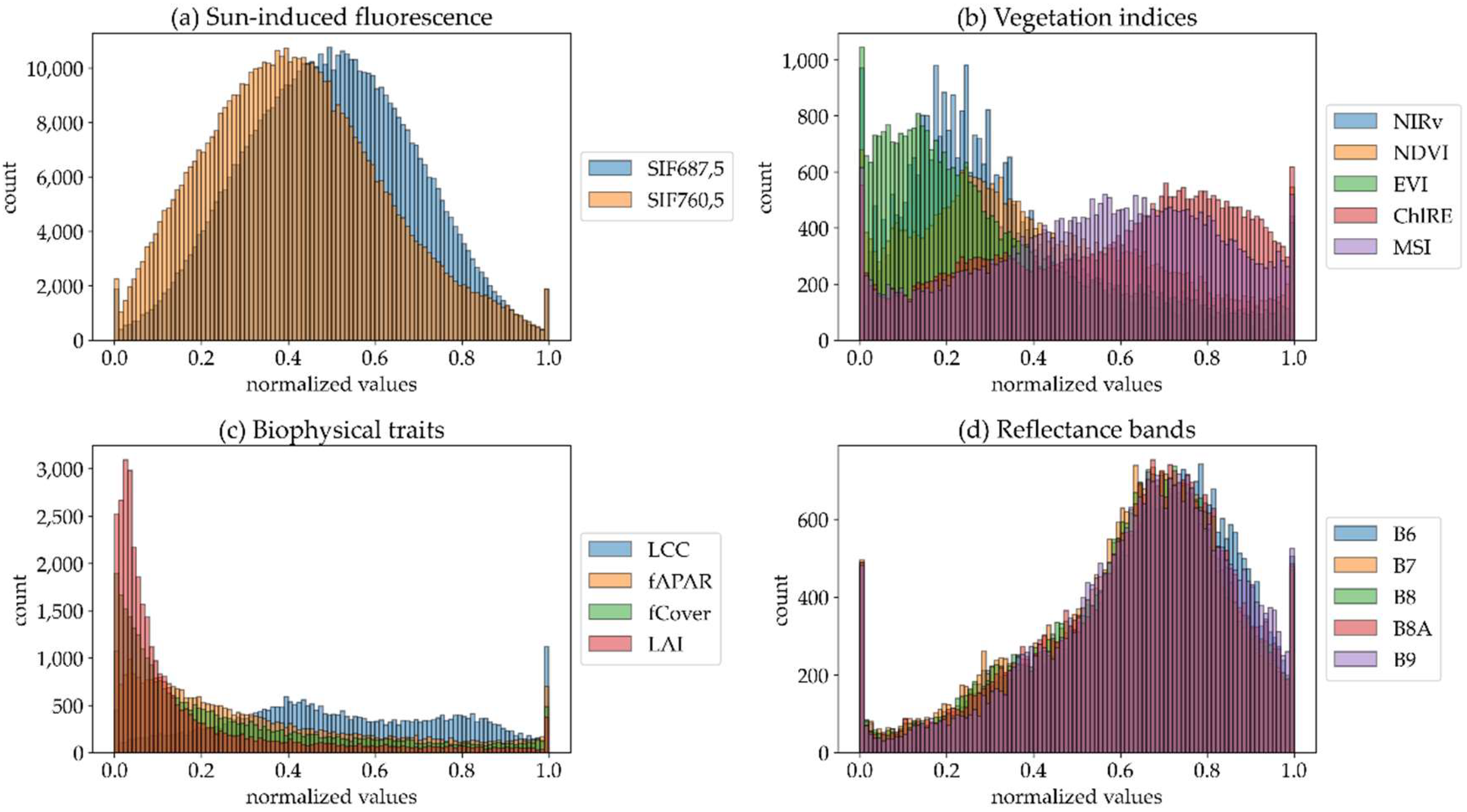
- From the FLUO sensor (4.5 × 4.5 m), SIF in the O2A (SIF760) and O2B (SIF687) bands was spatially aggregated in the software SAGA ([40], version 2.3.2) using the nearest neighbor algorithm to downscale it from 4.5 m to 5 m (SIF760,5 and SIF687,5). SIF was not aggregated to 20 m because we used the SIF image data as a reference, and therefore decreasing the resolution to 20 m would result in a loss of information needed for the characterization analysis. We did not exclude negative SIF values inherent to SIF retrieval uncertainty. Although they lack physical meaning (negative SIF is physically not possible), removing them would arbitrarily bias the resampled data. Therefore, retrieval uncertainty contributes unavoidably to the spatial heterogeneity of SIF.
- -
- Top-of-canopy reflectance data from the DUAL module of HyPlant (626 bands in total) were first spatially aggregated from 4.5 m to 20 m to mimic S2 pixels. We, again, used the software SAGA and information about the S2 grid to perform this task [40] (version 2.3.2). Spatial resampling was performed using the mean (cell area weighted) downscaling method. The output image was then processed in R using the hsdar package [41] for spectral convolution. This resulted in 13 synthetic S2 spectral reflectance bands at 20 m spatial resolution (S2-R20), which later were used to retrieve the biophysical traits (S2-BT20) and vegetation indices (S2-VI20) used to characterize SIF spatial heterogeneity (Table 1 and Table 2).
| Vegetation Index (VI) | General/Sentinel-2 Formula | Description |
|---|---|---|
| Normalized difference vegetation index (NDVI) | NDVI = (NIR − RED)/(NIR + RED) NDVI = ((B8A − B4)/(B8A + B4)) | Indicator of green vegetation [42]. |
| Near-infrared reflectance of terrestrial vegetation (NIRv) | NIRv = NIR×((NIR − RED)/(NIR + RED)) NIRv = B8A×((B8A − B4)/(B8A + B4)) | Proportion of pixel reflectance due to vegetation in the pixel; strongly correlated with SIF [21,43,44]. |
| Chlorophyll red-edge (ChlRE) | ChlRE = ([760:800]/[690:720]) – 1 ChlRE = B7/B5-1 | Estimates chlorophyll content in leaves [45]. |
| Enhanced vegetation Index (EVI) | EVI = 2.5×(NIR − RED)/((NIR + 6 × RED − 7.5 × BLUE) + 1) EVI = 2.5×(B8A − B4)/(B8A + 6 × B4 − 7.5 × B2) + 1) | Indicator of green vegetation similar to NDVI, but corrects for some atmospheric conditions and is more sensitive to dense vegetation [46,47]. |
| Moisture content (MSI) | MSI = SWIR/NIR MSI = B11/B08A | Indicator of leaf water content—higher values indicate high water stress with less water content and vice-versa [48,49]. |
| Biophysical Trait (BT) | Description |
|---|---|
| Fraction of Absorbed Photosynthetically Active Radiation (fAPAR) | Fraction of the down-welling photosynthetically active radiation that is absorbed by the canopy [51]. |
| Leaf Area Index (LAI) | Quantifies the amount of leaf material in a canopy. It is the ratio of one-sided leaf area per unit ground area [52,53]. |
| Fraction of green Vegetation Cover (fCover) | Quantifies the fraction of ground covered by green vegetation [54]. |
| Leaf Chlorophyll Content (LCC) | Leaf chlorophyll content (µg of chlorophyll per cm2 of leaf area) was computed from the retrieved canopy chlorophyll content (CCC), dividing it to the retrieved LAI [55]. |
2.5. Predictor Selection
2.6. Methods to Characterize Sun-Induced Chlorophyll Fluorescence Heterogeneity
2.7. Outliers’ Distribution
3. Results
3.1. Field Site Characterization
Spatial Analysis
3.2. Models’ Performances
3.2.1. Evaluation
3.2.2. Outliers’ Spatial Distribution
3.2.3. Best-Performing Models
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Method Name | Heterogeneity Values |
|---|---|
| Local Moran’s I | Range from 0 to 1, where 0 corresponds to low heterogeneity and 1 to high heterogeneity |
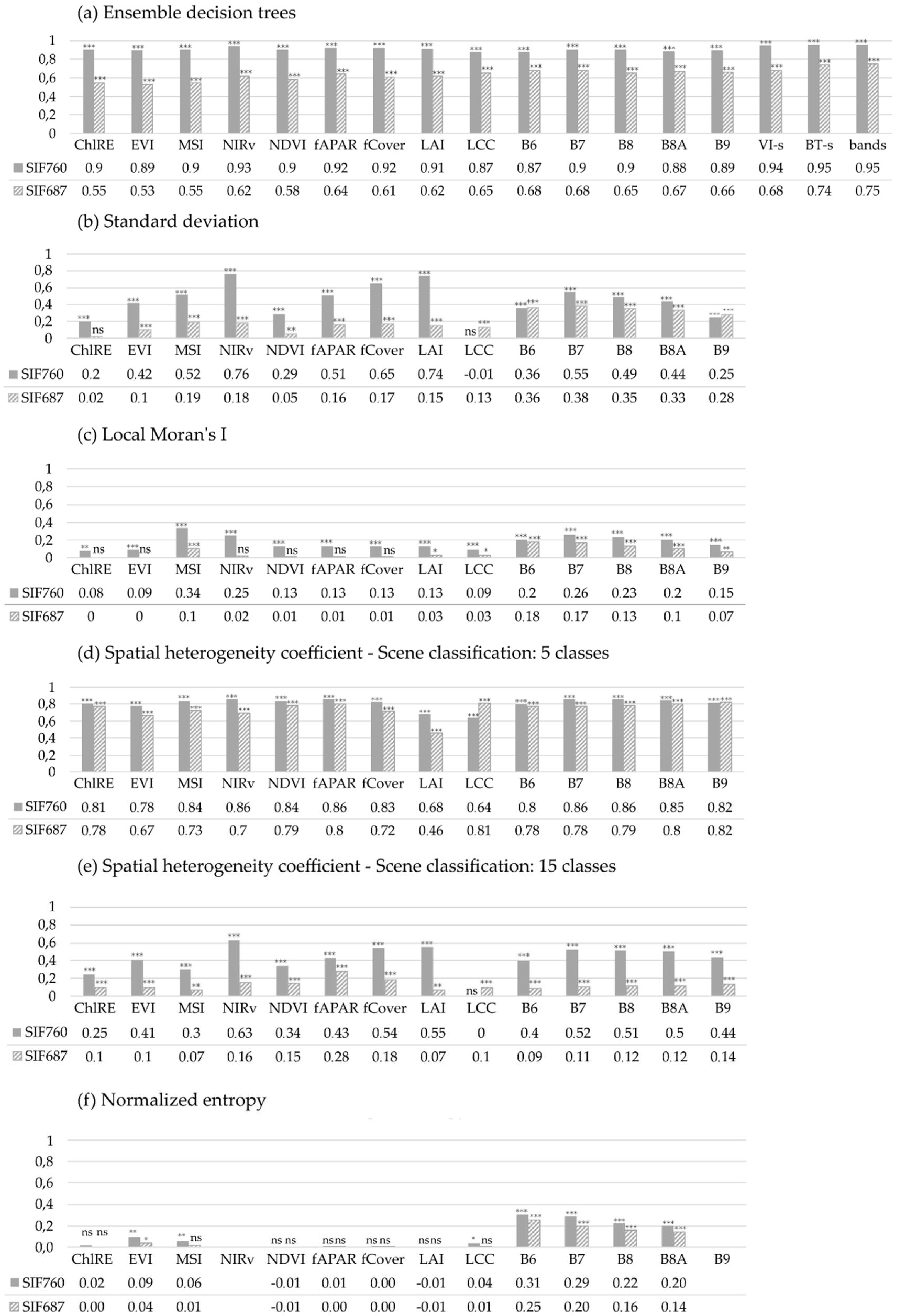
| We quantified the fluorescence heterogeneity at FLEX resolution as the fraction of pixels belonging to a single-pixel cluster or not assigned to any cluster. For each 300 × 300 m pixel, we independently clustered S2-R20 and SIFλ,5 using the Local Moran’s I method [57] implemented in the function Moran_Local() from the ESDA (Exploratory Spatial Data Analysis) package from the PySAL Python library [76]. The approach classifies pixels into four classes: diamond (a single high value among low values), doughnuts (a single low value among high values), hotspot (a high value among high values), and cold spot (a low value among low values); the first two are single-pixels classes. The classification is conducted based on the spatial autocorrelation Moran’s I metric whose statistical significance is defined using permutations (bootstrap). In this way, the pixels that were not significantly assigned to any of the former categories constituted a new “heterogeneous” class. FLEX pixel heterogeneity was then computed as the fraction of diamond, doughnut, and non-significant pixels within each 300 × 300 m FLEX pixel. | |
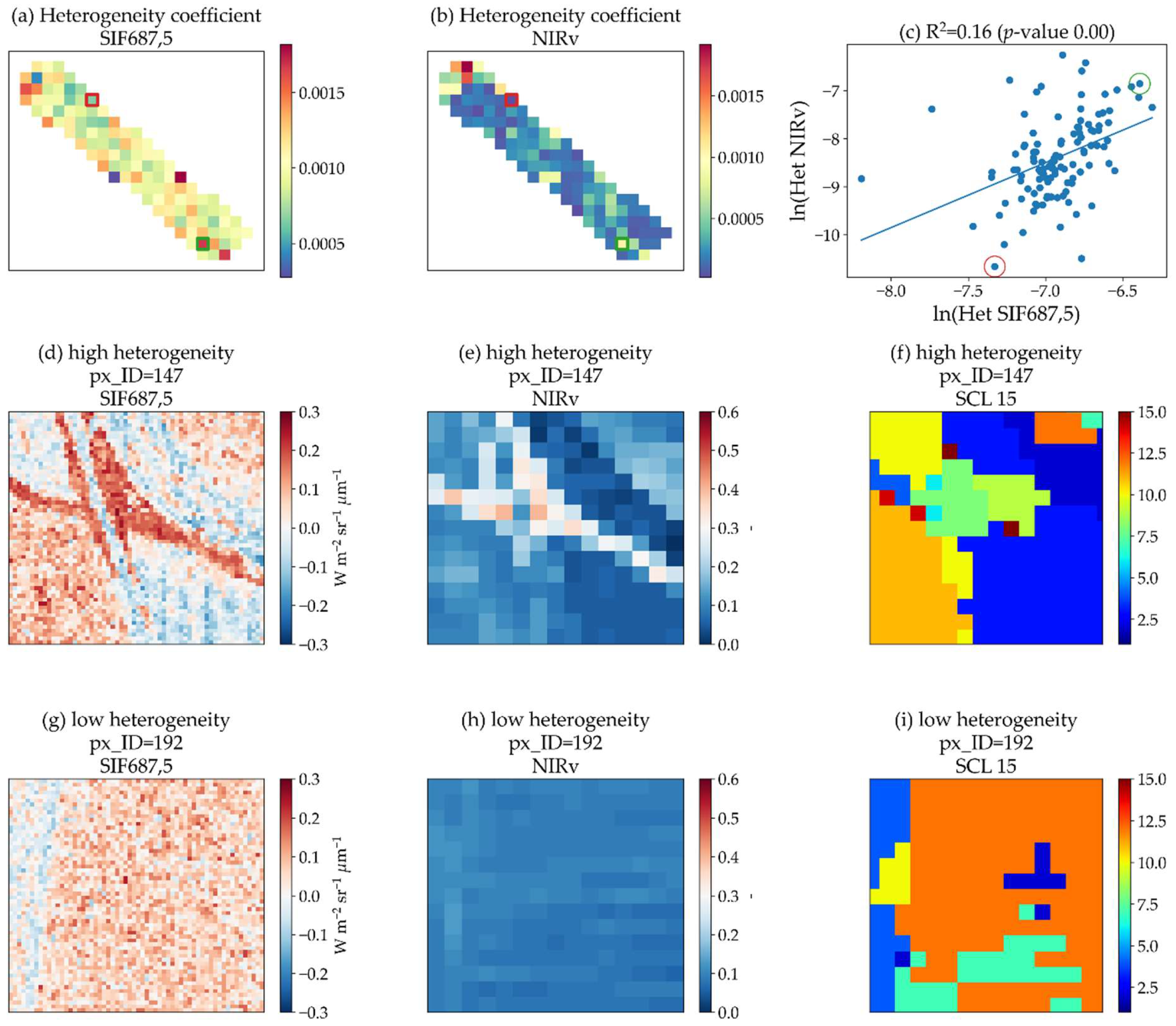
| Spatial heterogeneity coefficient | High values determine higher heterogeneity and lower values lower heterogeneity. The lowest possible value is 0, when all pixels belong to one class (homogeneous). The highest value is limited by the range of the values and the number of land cover types, 0.0053 W2 m−4 um−2 sr−2 for SIF760, 0.55 m4 m−4 for LAI |
| Heterogeneity was quantified as a function of land-use cover variability within each FLEX 300 m pixel, using the spatial heterogeneity coefficient described in [31]. Formula for spatial heterogeneity coefficient (Csh): | |
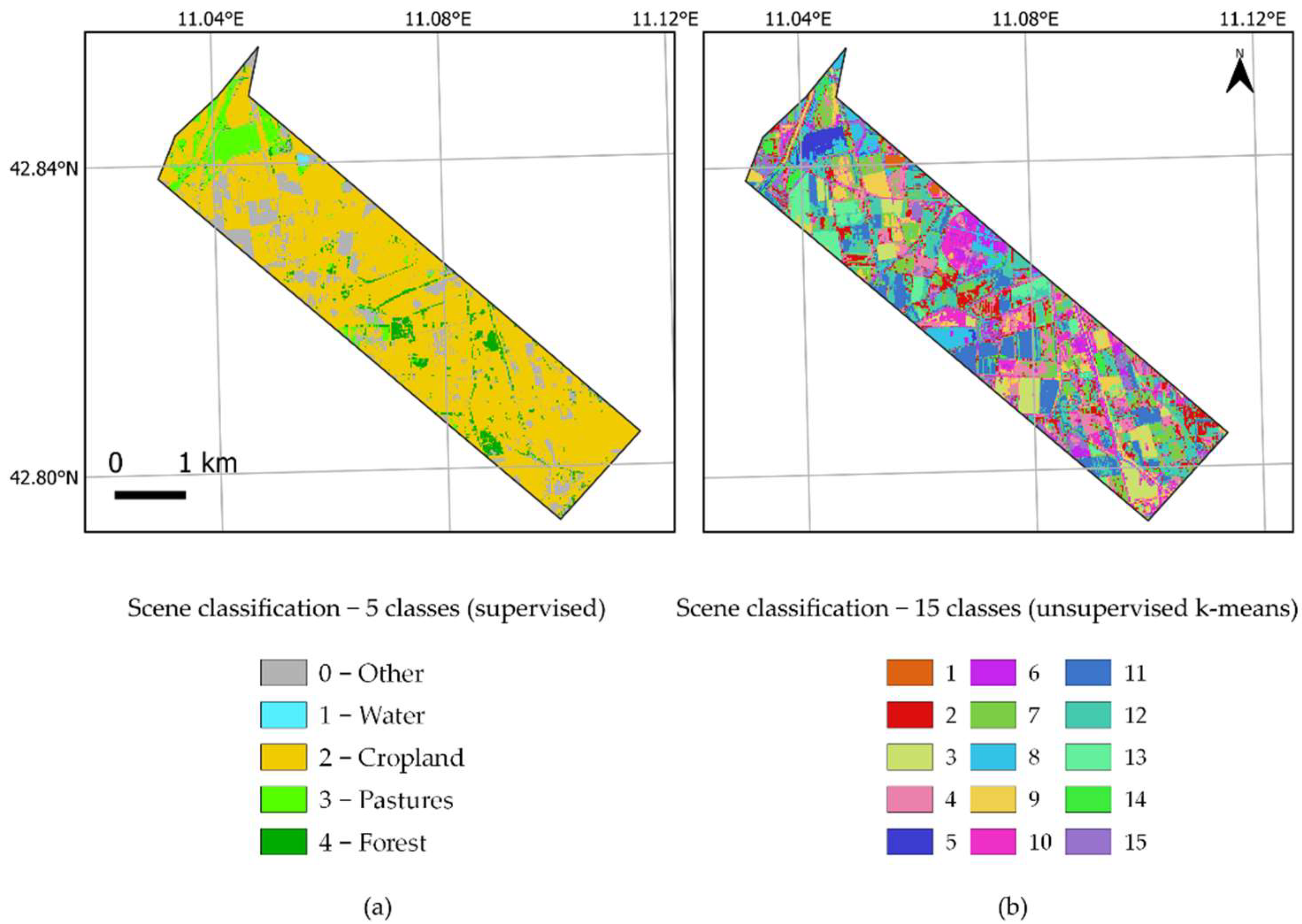
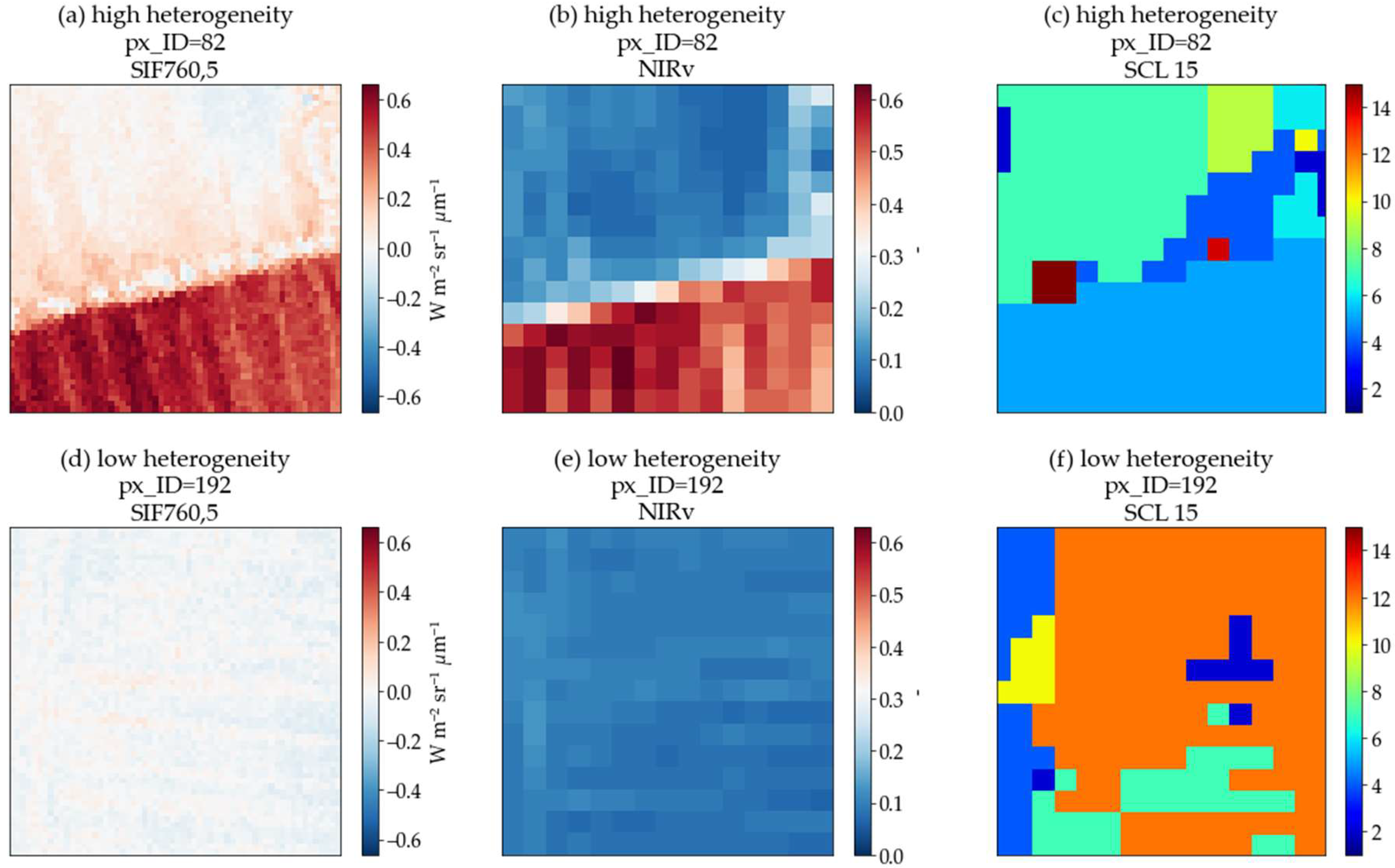
| N—total number of land cover classes of each sub-pixel included in a pixel; xm—m-th pixel value, which is included in the i-th land cover class; μ—mean value of the total sub-pixels that are included in one pixel; ni—total number of sub-pixels included in the i-th type; pi—fraction of the i-th land cover class in a pixel. One FLEX pixel could contain more than one land cover class; such a class is represented by sub-pixels (5 m for SIF and 20 m for S2 metrics). Heterogeneity combines class variance with information entropy. Class variance ( is the difference in sub-pixels reflecting intraclass (difference in growth conditions for the same vegetation type, i.e., different canopy densities) and interclass (i.e., land cover class patchiness) heterogeneity. Information entropy ( or class frequency explains how much each land cover class (pi) contributes to the pixels and is expressed as a fraction of a specific land cover class in a pixel multiplied by its natural logarithm. Two scene classification maps with 5 (SCL-5) and 15 classes (SCL-15) were produced using supervised and unsupervised approaches, respectively. Information from the Urban Atlas layer was used for creating a simpler SCL-5 (containing five classes defined as crops, pasture, water, forest, other) using semi-automatic classification plugin [60] on S2 bands in the QGIS environment (“QGIS Geographic Information System,” 2021) (Figure 4A)). Another SCL-15 map was produced using k-means clustering on the S2 dataset in SAGA with 15 clusters (the same number of land cover types for Braccagni image as in the Urban Atlas layer). Both maps were smoothed out with a 3 × 3 mode (majority) kernel. The spatial heterogeneity coefficient was calculated for every 300 m pixel using land cover frequencies and land cover class variances from scene classification maps. | |
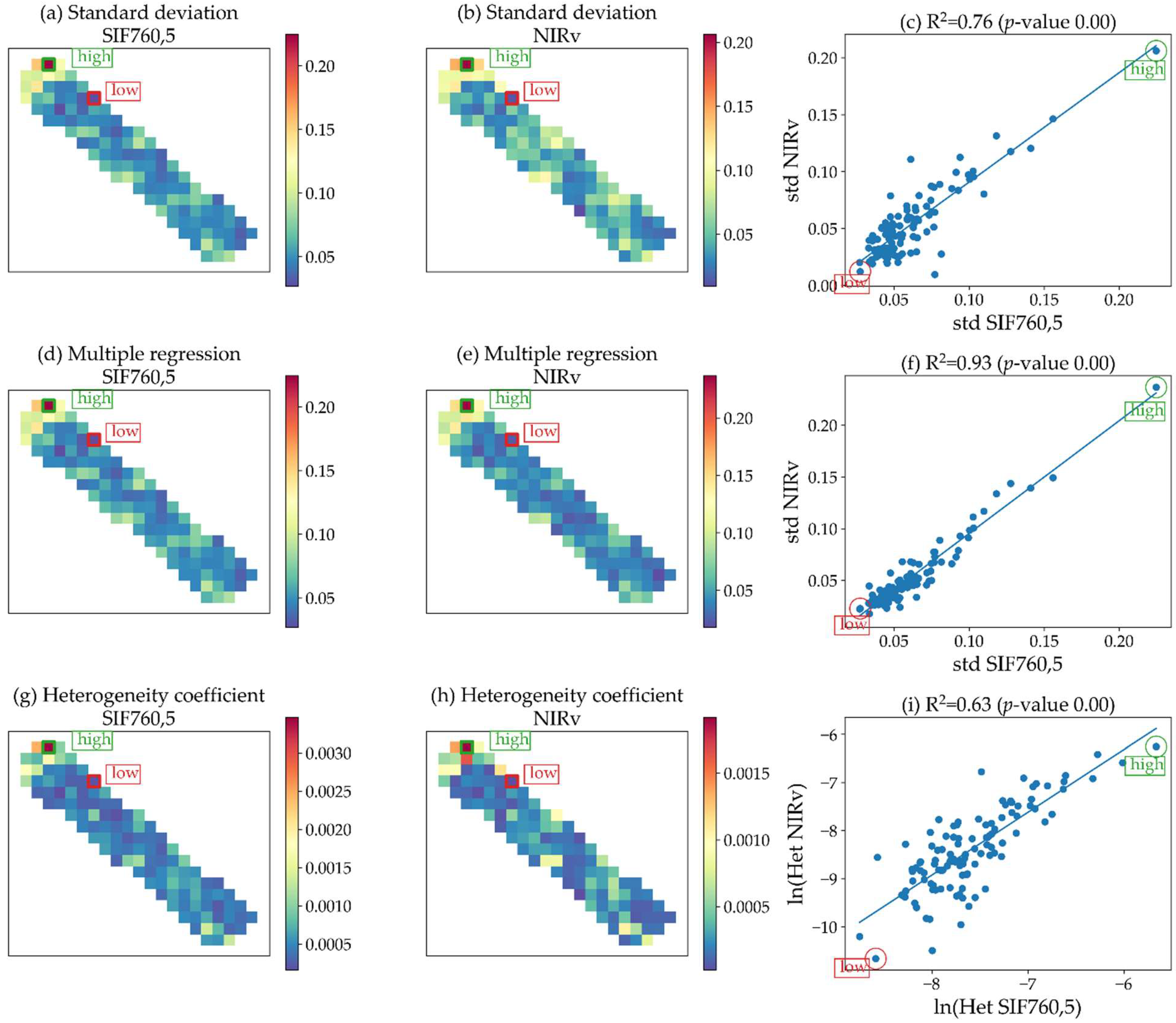
| Standard deviation | High values determine higher heterogeneity and lower values lower heterogeneity. The range depends on the range of the predictor, i.e., from 0.03 to 0.22 W m−2 um−1 sr−1 for SIF760, 0.05 to 2.52 m2 m−2 for LAI. |
| The standard deviation is a measure of how dispersed the data are in relation to their mean. Riera et al. [61] used the standard deviation of the NDVI as an expression of vegetation heterogeneity; moreover, Li and Rodell [62] used it as a measure of spatial variability of soil moisture. | |
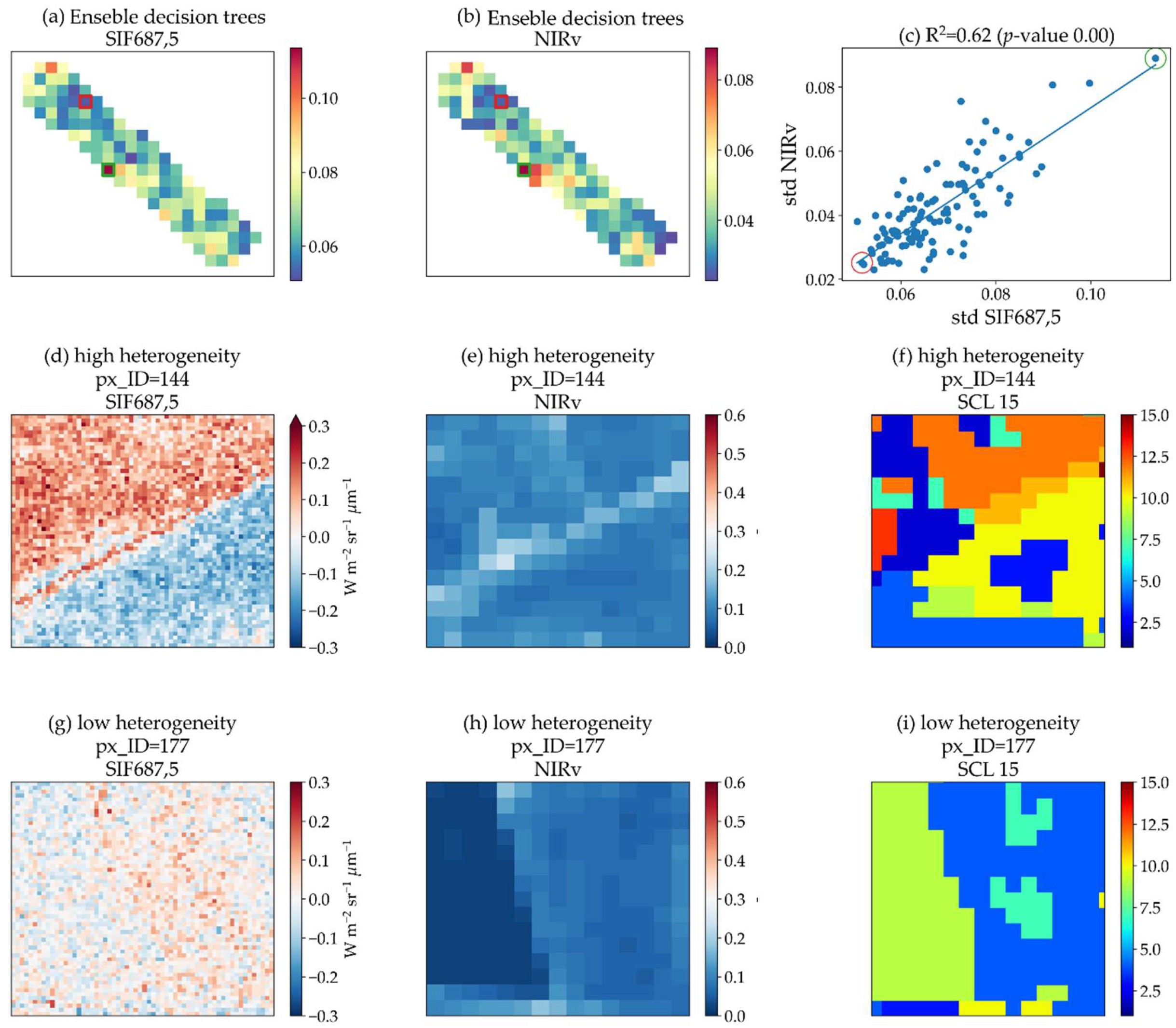
| Ensemble decision trees | High values determine higher heterogeneity and lower values lower heterogeneity. This method converted predictor values to SIF values; therefore, the range was always from 0.03 to 0.22 W m−2 um−1 sr−1 for SIF760 and from 0.05 to 0.11 W m−2 um−1 sr−1 for SIF687. |
| We assessed the capability of four different machine learning algorithms to predict SIFλ,20 as a function of SIFλ,300, and R20: eXtreme Gradient Boosting, Random Forests, support vector machines, and neural networks. The imagery was randomly split into training and validation subsets based on the finest resolution. For training models, 20% of the data (6800 samples) were used for computation economy. We used a k-fold (k = 5) cross-validation approach to assess each algorithm’s performance. Random Forests [58] was the most accurate algorithm. Thus, we made use of this approach to upscale SIF from the FLEX to the S2 spatial resolution. | |
| Normalized Entropy | Normalized entropy ranging from 0 to 1, where 1 corresponds to low heterogeneity and 0 to higher heterogeneity. Probability values pi are expressed for sub-pixel locations i inside the FLEX pixel, and their values correspond to the sub-pixel values in S2 or F |
| Heterogeneity was quantified using the concept of entropy [64] that measures the average information content. Entropy is maximized when every sub-pixel within the 300 × 300 m FLEX pixel contains the same value (uniform probability distribution, no heterogeneity). Thus, within a 300 m pixel, the maximum possible entropy value for 225 20 m sub-pixels (S2 products) is 7.81 (all pi = 1/225) and for 3600 5 m sub-pixels it is 11.81 (all pi = 1/3600). For each pixel at FLEX resolution (SIFλ,300), we calculated the entropy using each dataset SIFλ,5, S2-VI20, S2-BT20, and S2-R20. We normalized the entropy by the entropy of the uniform distribution (Emax with N = sub-pixels in a FLEX pixel). This custom “normalized entropy” function was passed as an additional parameter to the python module rasterstats [77]. | |
Appendix B
| Method Name | Heterogeneity Definition | Predictors | Heterogeneity Values |
|---|---|---|---|
| Cluster entropy | Uncovered sub-pixel information by aggregating patterns with a similar distribution. | S2-VI20 S2-BT20 S2-R20 | Uncovered sub-pixel information by aggregating patterns with a similar distribution. |
| A set of spatial patterns were extracted from S2-VI20, S2-BT20 and S2-Rsingle,20 by means of a clustering approach, aggregating patterns with a similar distribution. Analogously, patterns from SIF at 5 m pixel resolution are also extracted. This allows us to compare the uncovered sub-pixel information for both co-registered datasets. For this method, 300 × 300 m patches from SIF and S2 data were extracted, and considering that the SIF spatial resolution is 5 m, each patch contains 60 × 60 sub-pixels or 3600 pixels, whereas for FLEX patches are represented by 15 × 15 or 225 pixels. All the patches containing missing values were discarded, leaving a total of 110 patches for SIF and 114 patches for S2. Nevertheless, only 104 patches of S2 and SIF overlapped and were used for the comparison. We used the SIF760 and SIF687 as reference data and the S2 reflectance bands and its derived indices as predictors. A Gaussian mixture model (GMM) clustering algorithm [78] grouped the 300 m patches of Fλ,5 into k = 3 clusters. S2-VI20, S2-BT20 and S2-Rsingle,20 were clustered into k = 4 groups as they showed higher heterogeneity than the patches of Fλ,5. Since cluster labeling was arbitrary, clusters were relabeled from 1 to 3, allowing for comparing Sentinel-2 and Fλ,5 groups. The capability of the different S2 predictors to capture Fλ,5 on FLEX scales (300 m) was evaluated using confusion matrices. | |||
| Fuzzy approach | Model fluorescence (SIF760,20 and SIF687,20) sub-pixels variance in a 300 × 300 FLEX pixel | S2-VI20 S2-BT20 S2-R20 | |
| Fuzzy modelling allows for building flexible weighted maps from several variables [79], which can be used as a predictor of a third variable once combined. It is a two-step method where the original variables (VI20 or BT20) are first transformed to the range [0, 1] using different “membership” functions. These are selected according to their expected or known relationship with the predicted variable (SIFλ in this case). Then, the transformed variables (membership values) can be combined through various operators [80]. The combined values can then be used as predictors of the variable of interest. In this work, we applied fuzzy modelling to VI20 and BT20 separately to eventually predict SIFλ. We selected the membership functions so that membership values positively correlated with SIF (Table A3). Membership values computed from VI20 or BT20 variables were combined using the fuzzy overlay operator GAMMA since it has been reported to offer a balance between over and underestimation of fluorescence radiance [80,81]. μ_γ (x) = 〖[μ_SUM (x)]〗^γ * 〖[μ_PRODUCT (x)]〗^(1 − γ) Then a linear model was fit using the integrated membership values as a predictor of SIFλ: (F_λ) ̂ = b_0 + b_1 μ_γ (x) Fuzzy modelling was applied to 5 and 20 m spatial resolution data, and then these maps were gridded to 300 m pixels. Predicted and observed SIFλ and their intrapixel variability were assessed. | |||
| HyPlant Derived VIs | Membership Functions | Equations | Justifications | Representing Traits | References |
|---|---|---|---|---|---|
| NDVI | Fuzzy MS Large | Positive correlation | Greenness Content | [82,83,84] | |
| Chl-Red edge | Red-edge position | [84] | |||
| EVI | Fuzzy Linear | Weak correlation | Biomass | [84,85] | |
| MSI | Fuzzy MS Small | Negative correlation | Canopy water stress | [84,86,87] |
Appendix C

References
- Rossini, M.; Celesti, M.; Bramati, G.; Migliavacca, M.; Cogliati, S.; Rascher, U.; Colombo, R. Evaluation of the Spatial Representativeness of In Situ SIF Observations for the Validation of Medium-Resolution Satellite SIF Products. Remote Sens. 2022, 14, 5107. [Google Scholar] [CrossRef]
- Wei, J.; Tang, X.; Gu, Q.; Wang, M.; Ma, M.; Han, X. Using Solar-Induced Chlorophyll Fluorescence Observed by OCO-2 to Predict Autumn Crop Production in China. Remote Sens. 2019, 11, 1715. [Google Scholar] [CrossRef]
- Guanter, L.; Bacour, C.; Schneider, A.; Aben, I.; van Kempen, T.A.; Maignan, F.; Retscher, C.; Köhler, P.; Frankenberg, C.; Joiner, J.; et al. The TROPOSIF Global Sun-Induced Fluorescence Dataset from the Sentinel-5P TROPOMI Mission. Earth Syst. Sci. Data 2021, 13, 5423–5440. [Google Scholar] [CrossRef]
- Doughty, R.; Kurosu, T.P.; Parazoo, N.; Köhler, P.; Wang, Y.; Sun, Y.; Frankenberg, C. Global GOSAT, OCO-2, and OCO-3 Solar-Induced Chlorophyll Fluorescence Datasets. Earth Syst. Sci. Data 2022, 14, 1513–1529. [Google Scholar] [CrossRef]
- Coppo, P.; Taiti, A.; Pettinato, L.; Francois, M.; Taccola, M.; Drusch, M. Fluorescence Imaging Spectrometer (FLORIS) for ESA FLEX Mission. Remote Sens. 2017, 9, 649. [Google Scholar] [CrossRef]
- Drusch, M.; Moreno, J.; Del Bello, U.; Franco, R.; Goulas, Y.; Huth, A.; Kraft, S.; Middleton, E.M.; Miglietta, F.; Mohammed, G.; et al. The FLuorescence EXplorer Mission Concept—ESA’s Earth Explorer 8. IEEE Trans. Geosci. Remote Sens. 2017, 55, 1273–1284. [Google Scholar] [CrossRef]
- Mohammed, G.H.; Colombo, R.; Middleton, E.M.; Rascher, U.; van der Tol, C.; Nedbal, L.; Goulas, Y.; Pérez-Priego, O.; Damm, A.; Meroni, M.; et al. Remote Sens. of Solar-Induced Chlorophyll Fluorescence (SIF) in Vegetation: 50 years of Progress. Remote Sens. Environ. 2019, 231, 111177. [Google Scholar] [CrossRef]
- Damm, A.; Guanter, L.; Paul-Limoges, E.; van der Tol, C.; Hueni, A.; Buchmann, N.; Eugster, W.; Ammann, C.; Schaepman, M.E. Far-Red Sun-Induced Chlorophyll Fluorescence Shows Ecosystem-Specific Relationships to Gross Primary Production: An Assessment Based on Observational and Modeling Approaches. Remote Sens. Environ. 2015, 166, 91–105. [Google Scholar] [CrossRef]
- Lu, X.; Cheng, X.; Li, X.; Tang, J. Opportunities and Challenges of Applications of Satellite-Derived Sun-Induced Fluorescence at Relatively High Spatial Resolution. Sci. Total Environ. 2018, 619–620, 649–653. [Google Scholar] [CrossRef]
- Chen, A.; Mao, J.; Ricciuto, D.; Lu, D.; Xiao, J.; Li, X.; Thornton, P.E.; Knapp, A.K. Seasonal Changes in GPP/SIF Ratios and Their Climatic Determinants across the Northern Hemisphere. Global Change Biol. 2021, 27, 5186–5197. [Google Scholar] [CrossRef]
- Zhan, W.; Yang, X.; Ryu, Y.; Dechant, B.; Huang, Y.; Goulas, Y.; Kang, M.; Gentine, P. Two for One: Partitioning CO2 Fluxes and Understanding the Relationship between Solar-Induced Chlorophyll Fluorescence and Gross Primary Productivity Using Machine Learning. Agric. For. Meteorol. 2022, 321, 108980. [Google Scholar] [CrossRef]
- Perez-Priego, O.; Zarco-Tejada, P.J.; Miller, J.R.; Sepulcre-Canto, G.; Fereres, E. Detection of Water Stress in Orchard Trees with a High-Resolution Spectrometer through Chlorophyll Fluorescence in-Filling of the O/Sub 2/-A Band. IEEE Trans. Geosci. Remote Sens. 2005, 43, 2860–2869. [Google Scholar] [CrossRef]
- Lee, J.-E.; Frankenberg, C.; van der Tol, C.; Berry, J.A.; Guanter, L.; Boyce, C.K.; Fisher, J.B.; Morrow, E.; Worden, J.R.; Asefi, S.; et al. Forest Productivity and Water Stress in Amazonia: Observations from GOSAT Chlorophyll Fluorescence. Proc. R. Soc. B Biol. Sci. 2013, 280, 20130171. [Google Scholar] [CrossRef] [PubMed]
- Cendrero-Mateo, M.P.; Moran, M.S.; Papuga, S.A.; Thorp, K.R.; Alonso, L.; Moreno, J.; Ponce-Campos, G.; Rascher, U.; Wang, G. Plant Chlorophyll Fluorescence: Active and Passive Measurements at Canopy and Leaf Scales with Different Nitrogen Treatments. J. Exp. Bot. 2016, 67, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Clemente, R.; North, P.R.J.; Hornero, A.; Zarco-Tejada, P.J. Assessing the Effects of Forest Health on Sun-Induced Chlorophyll Fluorescence Using the FluorFLIGHT 3-D Radiative Transfer Model to Account for Forest Structure. Remote Sens. Environ. 2017, 193, 165–179. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Camino, C.; Beck, P.S.A.; Calderon, R.; Hornero, A.; Hernández-Clemente, R.; Kattenborn, T.; Montes-Borrego, M.; Susca, L.; Morelli, M.; et al. Previsual Symptoms of Xylella Fastidiosa Infection Revealed in Spectral Plant-Trait Alterations. Nat. Plants 2018, 4, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Gerhards, M.; Schlerf, M.; Rascher, U.; Udelhoven, T.; Juszczak, R.; Alberti, G.; Miglietta, F.; Inoue, Y. Analysis of Airborne Optical and Thermal Imagery for Detection of Water Stress Symptoms. Remote Sens. 2018, 10, 1139. [Google Scholar] [CrossRef]
- Xu, S.; Liu, Z.; Zhao, L.; Zhao, H.; Ren, S. Diurnal Response of Sun-Induced Fluorescence and PRI to Water Stress in Maize Using a Near-Surface Remote Sens. Platform. Remote Sens. 2018, 10, 1510. [Google Scholar] [CrossRef]
- Luus, K.A.; Commane, R.; Parazoo, N.C.; Benmergui, J.; Euskirchen, E.S.; Frankenberg, C.; Joiner, J.; Lindaas, J.; Miller, C.E.; Oechel, W.C.; et al. Tundra Photosynthesis Captured by Satellite-Observed Solar-Induced Chlorophyll Fluorescence. Geophys. Res. Lett. 2017, 44, 1564–1573. [Google Scholar] [CrossRef]
- Turner, A.J.; Köhler, P.; Magney, T.S.; Frankenberg, C.; Fung, I.; Cohen, R.C. A Double Peak in the Seasonality of California’s Photosynthesis as Observed from Space. Biogeosciences 2020, 17, 405–422. [Google Scholar] [CrossRef]
- Mengistu, A.G.; Mengistu Tsidu, G.; Koren, G.; Kooreman, M.L.; Boersma, K.F.; Tagesson, T.; Ardö, J.; Nouvellon, Y.; Peters, W. Sun-Induced Fluorescence and near-Infrared Reflectance of Vegetation Track the Seasonal Dynamics of Gross Primary Production over Africa. Biogeosciences 2021, 18, 2843–2857. [Google Scholar] [CrossRef]
- Porcar-Castell, A.; Tyystjärvi, E.; Atherton, J.; van der Tol, C.; Flexas, J.; Pfündel, E.E.; Moreno, J.; Frankenberg, C.; Berry, J.A. Linking Chlorophyll a Fluorescence to Photosynthesis for Remote Sens. Applications: Mechanisms and Challenges. J. Exp. Bot. 2014, 65, 4065–4095. [Google Scholar] [CrossRef] [PubMed]
- Van Wittenberghe, S.; Sabater, N.; Cendrero-Mateo, M.P.; Tenjo, C.; Moncholi, A.; Alonso, L.; Moreno, J. Towards the Quantitative and Physically-Based Interpretation of Solar-Induced Vegetation Fluorescence Retrieved from Global Imaging. Photosynthetica 2021, 59, 438–457. [Google Scholar] [CrossRef]
- Cogliati, S.; Celesti, M.; Cesana, I.; Miglietta, F.; Genesio, L.; Julitta, T.; Schuettemeyer, D.; Drusch, M.; Rascher, U.; Jurado, P.; et al. A Spectral Fitting Algorithm to Retrieve the Fluorescence Spectrum from Canopy Radiance. Remote Sens. 2019, 11, 1840. [Google Scholar] [CrossRef]
- Kováč, D.; Novotný, J.; Šigut, L.; Grace, J.; Urban, O. Dynamic of Fluorescence Emissions at O2A and O2B Telluric Absorption Bands in Forested Areas with Seasonal APAR and GPP Variations. Remote Sens. 2023, 15, 67. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Suárez, L.; González-Dugo, V. Spatial Resolution Effects on Chlorophyll Fluorescence Retrieval in a Heterogeneous Canopy Using Hyperspectral Imagery and Radiative Transfer Simulation. IEEE Geosci. Remote Sens. Lett. 2013, 10, 937–941. [Google Scholar] [CrossRef]
- Tagliabue, G.; Panigada, C.; Dechant, B.; Baret, F.; Cogliati, S.; Colombo, R.; Migliavacca, M.; Rademske, P.; Schickling, A.; Schuttemeyer, D.; et al. Exploring the Spatial Relationship between Airborne-Derived Red and Far-Red Sun-Induced Fluorescence and Process-Based GPP Estimates in a Forest Ecosystem. Remote Sens. Environ. 2019, 231, 111272. [Google Scholar] [CrossRef]
- Moncholi-Estornell, A.; Van Wittenberghe, S.; Pilar Cendrero-Mateo, M.; Alonso, L.; Jiménez, M.; Urrego, P.; Mac Arthur, A.; Moreno, J. FluoCat: A Cable-Suspended Multi-Sensor System for the Vegetation SIF Cal/Val Monitoring and Estimation of Effective Sunlit Surface Fluorescence. Int. J. Appl. Earth Obs. Geoinf. 2023, 116, 103147. [Google Scholar] [CrossRef]
- Li, H.; Reynolds, J.F. On Definition and Quantification of Heterogeneity. Oikos 1995, 73, 280–284. [Google Scholar] [CrossRef]
- Ding, Y.; Zhao, K.; Zheng, X. Multiscale Analysis of Landscape Spatial Heterogeneity Using Vegetation Indexes. In Proceedings of the 2013 the International Conference on Remote Sensing, Environment and Transportation Engineering (rsete 2013), Nanjing, China, 26–28 July 2013; Gahegan, M.N., Xiong, N., Eds.; Atlantis Press: Paris, France, 2013; Volume 31, pp. 609–612. [Google Scholar]
- Zhao, W.; Fan, W. Quantitative Representation of Spatial Heterogeneity in the LAI Scaling Transfer Process. IEEE Access 2021, 9, 83851–83862. [Google Scholar] [CrossRef]
- Quattrochi, D.A.; Goodchild, M.F. Scale in Remote Sens. and GIS; CRC Press: Boca Raton, FL, USA, 1997; ISBN 978-1-56670-104-4. [Google Scholar]
- Gustafson, E.J. Quantifying Landscape Spatial Pattern: What Is the State of the Art? Ecosystems 1998, 1, 143–156. [Google Scholar] [CrossRef]
- Murwira, A.; Skidmore, A.K. Characterising the Spatial Heterogeneity of a Landscape. In Scale Matters!: A New Approach to Quantify Spatial Heterogeneity for Predicting the Distribution of Wildlife; ITC Enschede/Wageningen University: Wageningen, The Netherlands, 2003; pp. 13–31. ISBN 978-90-5808-951-9. [Google Scholar]
- Buman, B.; Hueni, A.; Colombo, R.; Cogliati, S.; Celesti, M.; Julitta, T.; Burkart, A.; Siegmann, B.; Rascher, U.; Drusch, M.; et al. Towards Consistent Assessments of in Situ Radiometric Measurements for the Validation of Fluorescence Satellite Missions. Remote Sens. Environ. 2022, 274, 112984. [Google Scholar] [CrossRef]
- Rascher, U.; Alonso, L.; Burkart, A.; Cilia, C.; Cogliati, S.; Colombo, R.; Damm, A.; Drusch, M.; Guanter, L.; Hanus, J.; et al. Sun-Induced Fluorescence—A New Probe of Photosynthesis: First Maps from the Imaging Spectrometer HyPlant. Global Change Biol. 2015, 21, 4673–4684. [Google Scholar] [CrossRef] [PubMed]
- Siegmann, B.; Alonso, L.; Celesti, M.; Cogliati, S.; Colombo, R.; Damm, A.; Douglas, S.; Guanter, L.; Hanuš, J.; Kataja, K.; et al. The High-Performance Airborne Imaging Spectrometer HyPlant—From Raw Images to Top-of-Canopy Reflectance and Fluorescence Products: Introduction of an Automatized Processing Chain. Remote Sens. 2019, 11, 2760. [Google Scholar] [CrossRef]
- Cogliati, S.; Verhoef, W.; Kraft, S.; Sabater, N.; Alonso, L.; Vicent, J.; Moreno, J.; Drusch, M.; Colombo, R. Retrieval of Sun-Induced Fluorescence Using Advanced Spectral Fitting Methods. Remote Sens. Environ. 2015, 169, 344–357. [Google Scholar] [CrossRef]
- Siegmann, B.; Cendrero-Mateo, M.P.; Cogliati, S.; Damm, A.; Gamon, J.; Herrera, D.; Jedmowski, C.; Junker-Frohn, L.V.; Kraska, T.; Muller, O.; et al. Downscaling of Far-Red Solar-Induced Chlorophyll Fluorescence of Different Crops from Canopy to Leaf Level Using a Diurnal Data Set Acquired by the Airborne Imaging Spectrometer HyPlant. Remote Sens. Environ. 2021, 264, 112609. [Google Scholar] [CrossRef] [PubMed]
- Conrad, O.; Bechtel, B.; Bock, M.; Dietrich, H.; Fischer, E.; Gerlitz, L.; Wehberg, J.; Wichmann, V.; Böhner, J. System for Automated Geoscientific Analyses (SAGA) v. 2.1.4. Geosci. Model Dev. 2015, 8, 1991–2007. [Google Scholar] [CrossRef]
- Lehnert, L.W.; Meyer, H.; Obermeier, W.A.; Silva, B.; Regeling, B.; Bendix, J. Hyperspectral Data Analysis in R: The Hsdar Package. J. Stat. Soft. 2019, 89, 1–23. [Google Scholar] [CrossRef]
- Rouse, J.; Rh, H.; Ja, S.; Dw, D. Monitoring Vegetation Systems in the Great Plains with ERTS. NASA Spec. Publ. 1974, 351, 309. [Google Scholar]
- Badgley, G.; Field, C.B.; Berry, J.A. Canopy Near-Infrared Reflectance and Terrestrial Photosynthesis. Sci. Adv. 2017, 3, e1602244. [Google Scholar] [CrossRef]
- Zhang, J.; Xiao, J.; Tong, X.; Zhang, J.; Meng, P.; Li, J.; Liu, P.; Yu, P. NIRv and SIF Better Estimate Phenology than NDVI and EVI: Effects of Spring and Autumn Phenology on Ecosystem Production of Planted Forests. Agric. For. Meteorol. 2022, 315, 108819. [Google Scholar] [CrossRef]
- Vogelmann, J.E.; Rock, B.N.; Moss, D.M. Red Edge Spectral Measurements from Sugar Maple Leaves. Int. J. Remote Sens. 1993, 14, 1563–1575. [Google Scholar] [CrossRef]
- Huete, A.; Justice, C.; Liu, H. Development of Vegetation and Soil Indices for MODIS-EOS. Remote Sens. Environ. 1994, 49, 224–234. [Google Scholar] [CrossRef]
- Huete, A.R.; Liu, H.Q.; Batchily, K.; van Leeuwen, W. A Comparison of Vegetation Indices over a Global Set of TM Images for EOS-MODIS. Remote Sens. Environ. 1997, 59, 440–451. [Google Scholar] [CrossRef]
- Hunt, E.R.; Rock, B.N. Detection of Changes in Leaf Water Content Using Near- and Middle-Infrared Reflectances. Remote Sens. Environ. 1989, 30, 43–54. [Google Scholar] [CrossRef]
- Ceccato, P.; Flasse, S.; Tarantola, S.; Jacquemoud, S.; Grégoire, J.-M. Detecting Vegetation Leaf Water Content Using Reflectance in the Optical Domain. Remote Sens. Environ. 2001, 77, 22–33. [Google Scholar] [CrossRef]
- Weiss, M.; Jay, S.; Baret, F. Sentinel2 ToolBox Level2 Products: LAI, FAPAR, FCOVER, Version 2.0.; INRAE: Paris, France, 2020. [Google Scholar]
- Prince, S.D. A Model of Regional Primary Production for Use with Coarse Resolution Satellite Data. Int. J. Remote Sens. 1991, 12, 1313–1330. [Google Scholar] [CrossRef]
- Nathalie, B. Leaf Area Index. In Encyclopedia of Ecology; Academic Press: Cambridge, MA, USA, 2008; pp. 2148–2154. ISBN 978-0-08-045405-4. [Google Scholar]
- Weiss, M.; Baret, F. S2ToolBox Level 2 Products: LAI, FAPAR, FCOVER. Version 1.1; INRA Avignon: Paris, France, 2016. [Google Scholar]
- Liang, S.; Wang, J. (Eds.) Chapter 12—Fractional Vegetation Cover. In Advanced Remote Sensing, 2nd ed.; Liang, S.; Wang, J. (Eds.) Academic Press: Cambridge, MA, USA, 2020; pp. 477–510. ISBN 978-0-12-815826-5. [Google Scholar]
- Croft, H.; Chen, J.M.; Luo, X.; Bartlett, P.; Chen, B.; Staebler, R.M. Leaf Chlorophyll Content as a Proxy for Leaf Photosynthetic Capacity. Global Change Biol. 2017, 23, 3513–3524. [Google Scholar] [CrossRef]
- Wang, Z.; Bovik, A.C.; Sheikh, H.R.; Simoncelli, E.P. Image Quality Assessment: From Error Visibility to Structural Similarity. IEEE Trans. Image Process. 2004, 13, 600–612. [Google Scholar] [CrossRef]
- Anselin, L. Local Indicators of Spatial Association—LISA. Geogr. Anal. 1995, 27, 93–115. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Shannon, C.E. A Mathematical Theory of Communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Moncholi-Estornell, A.; Cendrero-Mateo, M.P.; Antala, M.; Cogliati, S.; Moreno, J.; Van Wittenberghe, S. Enhancing Solar-Induced Fluorescence Interpretation: Quantifying Fractional Sunlit Vegetation Cover Using Linear Spectral Unmixing. Remote Sens. 2023, 15, 4274. [Google Scholar] [CrossRef]
- Zeng, Y.; Badgley, G.; Dechant, B.; Ryu, Y.; Chen, M.; Berry, J.A. A Practical Approach for Estimating the Escape Ratio of Near-Infrared Solar-Induced Chlorophyll Fluorescence. Remote Sens. Environ. 2019, 232, 111209. [Google Scholar] [CrossRef]
- Dechant, B.; Ryu, Y.; Badgley, G.; Köhler, P.; Rascher, U.; Migliavacca, M.; Zhang, Y.; Tagliabue, G.; Guan, K.; Rossini, M.; et al. NIRVP: A Robust Structural Proxy for Sun-Induced Chlorophyll Fluorescence and Photosynthesis across Scales. Remote Sens. Environ. 2022, 268, 112763. [Google Scholar] [CrossRef]
- Sarrias, M. Random Parameters and Spatial Heterogeneity Using Rchoice in R. REGION 2020, 7, 1–19. [Google Scholar] [CrossRef][Green Version]
- Li, B.; Rodell, M. Spatial Variability and Its Scale Dependency of Observed and Modeled Soil Moisture over Different Climate Regions. Hydrol. Earth Syst. Sci. 2013, 17, 1177–1188. [Google Scholar] [CrossRef]
- Riera, J.L.; Magnuson, J.J.; Vande Castle, J.R.; MacKenzie, M.D. Analysis of Large-Scale Spatial Heterogeneity in Vegetation Indices among North American Landscapes. Ecosystems 1998, 1, 268–282. [Google Scholar] [CrossRef]
- Doxa, A.; Prastacos, P. Using Rao’s Quadratic Entropy to Define Environmental Heterogeneity Priority Areas in the European Mediterranean Biome. Biol. Conserv. 2020, 241, 108366. [Google Scholar] [CrossRef]
- Rao, C.R. Diversity and Dissimilarity Coefficients: A Unified Approach. Theor. Popul. Biol. 1982, 21, 24–43. [Google Scholar] [CrossRef]
- Tagliabue, G.; Panigada, C.; Celesti, M.; Cogliati, S.; Colombo, R.; Migliavacca, M.; Rascher, U.; Rocchini, D.; Schuttemeyer, D.; Rossini, M. Sun-Induced Fluorescence Heterogeneity as a Measure of Functional Diversity. Remote Sens. Environ. 2020, 247, 111934. [Google Scholar] [CrossRef]
- Rossini, M.; Panigada, C.; Cilia, C.; Meroni, M.; Busetto, L.; Cogliati, S.; Amaducci, S.; Colombo, R. Discriminating Irrigated and Rainfed Maize with Diurnal Fluorescence and Canopy Temperature Airborne Maps. ISPRS Int. J. Geo-Inf. 2015, 4, 626–646. [Google Scholar] [CrossRef]
- Wieneke, S.; Ahrends, H.; Damm, A.; Pinto, F.; Stadler, A.; Rossini, M.; Rascher, U. Airborne Based Spectroscopy of Red and Far-Red Sun-Induced Chlorophyll Fluorescence: Implications for Improved Estimates of Gross Primary Productivity. Remote Sens. Environ. 2016, 184, 654–667. [Google Scholar] [CrossRef]
- Van Wittenberghe, S.; Alonso, L.; Verrelst, J.; Moreno, J.; Samson, R. Bidirectional Sun-Induced Chlorophyll Fluorescence Emission Is Influenced by Leaf Structure and Light Scattering Properties—A Bottom-up Approach. Remote Sens. Environ. 2015, 158, 169–179. [Google Scholar] [CrossRef]
- van der Tol, C.; Vilfan, N.; Dauwe, D.; Cendrero-Mateo, M.P.; Yang, P. The Scattering and Re-Absorption of Red and near-Infrared Chlorophyll Fluorescence in the Models Fluspect and SCOPE. Remote Sens. Environ. 2019, 232, 111292. [Google Scholar] [CrossRef]
- Biriukova, K.; Celesti, M.; Evdokimov, A.; Pacheco-Labrador, J.; Julitta, T.; Migliavacca, M.; Giardino, C.; Miglietta, F.; Colombo, R.; Panigada, C.; et al. Effects of Varying Solar-View Geometry and Canopy Structure on Solar-Induced Chlorophyll Fluorescence and PRI. Int. J. Appl. Earth Obs. Geoinf. 2020, 89, 102069. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll Fluorescence: A Probe of Photosynthesis in Vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Porcar-Castell, A.; Malenovský, Z.; Magney, T.; Van Wittenberghe, S.; Fernández-Marín, B.; Maignan, F.; Zhang, Y.; Maseyk, K.; Atherton, J.; Albert, L.P.; et al. Chlorophyll a Fluorescence Illuminates a Path Connecting Plant Molecular Biology to Earth-System Science. Nat. Plants 2021, 7, 998–1009. [Google Scholar] [CrossRef]
- Rey, S.J.; Anselin, L. PySAL: A Python Library of Spatial Analytical Methods. In Handbook of Applied Spatial Analysis: Software Tools, Methods and Applications; Fischer, M.M., Getis, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 175–193. ISBN 978-3-642-03647-7. [Google Scholar]
- Perry, M.T. Rasterstats. 2015. Available online: https://pythonhosted.org/rasterstats/ (accessed on 18 October 2021).
- Figueiredo, M.A.T.; Jain, A.K. Unsupervised Learning of Finite Mixture Models. IEEE Trans. Pattern Anal. Mach. Intell. 2002, 24, 381–396. [Google Scholar] [CrossRef]
- Zadeh, L.A. Fuzzy Sets. Inf. Control. 1965, 8, 338–353. [Google Scholar] [CrossRef]
- Vakhshoori, V.; Zare, M. Landslide Susceptibility Mapping by Comparing Weight of Evidence, Fuzzy Logic, and Frequency Ratio Methods. Geomat. Nat. Hazards Risk 2016, 7, 1731–1752. [Google Scholar] [CrossRef]
- Bandopadhyay, S.; Rastogi, A.; Cogliati, S.; Rascher, U.; Gąbka, M.; Juszczak, R. Can Vegetation Indices Serve as Proxies for Potential Sun-Induced Fluorescence (SIF)? A Fuzzy Simulation Approach on Airborne Imaging Spectroscopy Data. Remote Sens. 2021, 13, 2545. [Google Scholar] [CrossRef]
- Yang, H.; Yang, X.; Zhang, Y.; Heskel, M.A.; Lu, X.; Munger, J.W.; Sun, S.; Tang, J. Chlorophyll Fluorescence Tracks Seasonal Variations of Photosynthesis from Leaf to Canopy in a Temperate Forest. Glob. Change Biol. 2017, 23, 2874–2886. [Google Scholar] [CrossRef]
- Du, S.; Liu, L.; Liu, X.; Zhang, X.; Zhang, X.; Bi, Y.; Zhang, L. Retrieval of Global Terrestrial Solar-Induced Chlorophyll Fluorescence from TanSat Satellite. Sci. Bull. 2018, 63, 1502–1512. [Google Scholar] [CrossRef]
- Bandopadhyay, S.; Rastogi, A.; Rademske, P.; Schickling, A.; Cogliati, S.; Julitta, T.; Mac Arthur, A.; Hueni, A.; Tomelleri, E.; Celesti, M.; et al. Hyplant-Derived Sun-Induced Fluorescence-A New Opportunity to Disentangle Complex Vegetation Signals from Diverse Vegetation Types. Remote Sens. 2019, 11, 1691. [Google Scholar] [CrossRef]
- Guo, M.; Li, J.; Huang, S.; Wen, L. Feasibility of Using MODIS Products to Simulate Sun-Induced Chlorophyll Fluorescence (SIF) in Boreal Forests. Remote Sens. 2020, 12, 680. [Google Scholar] [CrossRef]
- Wieneke, S.; Burkart, A.; Cendrero-Mateo, M.P.; Julitta, T.; Rossini, M.; Schickling, A.; Schmidt, M.; Rascher, U. Linking Photosynthesis and Sun-Induced Fluorescence at Sub-Daily to Seasonal Scales. Remote Sens. Environ. 2018, 219, 247–258. [Google Scholar] [CrossRef]
- Hazrati, S.; Tahmasebi-Sarvestani, Z.; Modarres-Sanavy, S.A.M.; Mokhtassi-Bidgoli, A.; Nicola, S. Effects of Water Stress and Light Intensity on Chlorophyll Fluorescence Parameters and Pigments of Aloe Vera L. Plant Physiol. Biochem. 2016, 106, 141–148. [Google Scholar] [CrossRef]



| Method Name | Heterogeneity Definition | Predictors | Reference |
|---|---|---|---|
| Local Moran’s I | The classification of sub-pixels is based on the spatial autocorrelation metric Moran’s I, whose statistical significance is defined by permutations (bootstrap). Heterogeneity is expressed as the fraction of sub-pixels belonging to the “no class” or “single pixel cluster class” over the total number of sub-pixels in a 300 × 300 FLEX pixel. | S2-VI20 S2-BT20 S2-R20 | [57] |
| Spatial heterogeneity coefficient | Interclass and intraclass differences combined with their spatial distribution. Classes are generated using supervised and unsupervised approaches in the form of Scene Classification Maps (SCLs). | S2-VI20 S2-BT20 S2-R20 | [31] |
| Standard deviation | Standard deviation over the total number of sub-pixels in a 300 × 300 FLEX pixel. | S2-VI20 S2-BT20 S2-R20 | / |
| Ensemble decision trees | Four different machine learning algorithms to predict SIFλ,20 as a function of SIFλ,300, and S2-VI20, S2-BT20, S2-R20, S2-Rmulti20, S2-VImulti20, S2-BTmulti20: eXtreme Gradient Boosting, Random Forests, Support Vector Machines, and Neural Networks. The most accurate algorithm (Random Forest) was used to upscale SIF from FLEX to S2 spatial resolution. | S2-VI20 S2-BT20 S2-R20 S2-Rmulti20 S2-VImulti20 S2-BTmulti20 | [58] |
| Normalized Entropy | Heterogeneity was quantified using the concept of entropy that measures the average information content. The entropy was normalized by the entropy of the uniform distribution (Emax with N = sub-pixels in a FLEX pixel). | S2-VI20 S2-BT20 S2-R20 | [59] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jantol, N.; Prikaziuk, E.; Celesti, M.; Hernandez-Sequeira, I.; Tomelleri, E.; Pacheco-Labrador, J.; Van Wittenberghe, S.; Pla, F.; Bandopadhyay, S.; Koren, G.; et al. Using Sentinel-2-Based Metrics to Characterize the Spatial Heterogeneity of FLEX Sun-Induced Chlorophyll Fluorescence on Sub-Pixel Scale. Remote Sens. 2023, 15, 4835. https://doi.org/10.3390/rs15194835
Jantol N, Prikaziuk E, Celesti M, Hernandez-Sequeira I, Tomelleri E, Pacheco-Labrador J, Van Wittenberghe S, Pla F, Bandopadhyay S, Koren G, et al. Using Sentinel-2-Based Metrics to Characterize the Spatial Heterogeneity of FLEX Sun-Induced Chlorophyll Fluorescence on Sub-Pixel Scale. Remote Sensing. 2023; 15(19):4835. https://doi.org/10.3390/rs15194835
Chicago/Turabian StyleJantol, Nela, Egor Prikaziuk, Marco Celesti, Itza Hernandez-Sequeira, Enrico Tomelleri, Javier Pacheco-Labrador, Shari Van Wittenberghe, Filiberto Pla, Subhajit Bandopadhyay, Gerbrand Koren, and et al. 2023. "Using Sentinel-2-Based Metrics to Characterize the Spatial Heterogeneity of FLEX Sun-Induced Chlorophyll Fluorescence on Sub-Pixel Scale" Remote Sensing 15, no. 19: 4835. https://doi.org/10.3390/rs15194835
APA StyleJantol, N., Prikaziuk, E., Celesti, M., Hernandez-Sequeira, I., Tomelleri, E., Pacheco-Labrador, J., Van Wittenberghe, S., Pla, F., Bandopadhyay, S., Koren, G., Siegmann, B., Legović, T., Kutnjak, H., & Cendrero-Mateo, M. P. (2023). Using Sentinel-2-Based Metrics to Characterize the Spatial Heterogeneity of FLEX Sun-Induced Chlorophyll Fluorescence on Sub-Pixel Scale. Remote Sensing, 15(19), 4835. https://doi.org/10.3390/rs15194835










