1. Introduction
Under the current conditions of climate change, the early detection of crop and global vegetation stress is crucial to promote a sustainable agriculture and ecosystem management. With the upcoming European Space Agency’s Fluorescence Explorer (FLEX) and Sentinel 3 tandem mission [
1], vegetation fluorescence and the auxiliary parameters needed to interpret solar-induced vegetation fluorescence (SIF) will be available at a 300 × 300 m spatial resolution. SIF is primarily controlled by the amount of light reaching the photosynthetic surface (absorbed by chlorophyll a (
Chl a) molecules). At the photosystem level, given the same absorbed photosynthetically active radiation (APAR) and regardless of the species studied and the leaf structure, a relatively constant fluorescence emission is expected, driven by the change in fluorescence quenching [
2,
3]. At the leaf level, different studies have shown a similar fluorescence quantum efficiency (FQE) (i.e., the flux of fluorescence photons normalised by the flux of absorbed photons) when plants had a similar chlorophyll content and were grown under the same environmental conditions (i.e., light history and stress conditions) [
4,
5,
6]. However, at the canopy level, the vegetation structure distorts both, the light reaching the photosynthetic surface (affecting the photosynthetic reactions) and the fluorescence signal (emitted by the vegetation) measured at the top of the canopy (TOC). Therefore, before using remote measurements of SIF to determine the plant physiological status, three major challenges need to be overcome: (i) quantifying the amount of light incident on the photosynthetic surface, (ii) quantifying the amount of light absorbed by
Chl a molecules or the green/photosynthetically active surface, and (iii) correcting the measured top of the canopy fluorescence by canopy scattering and reabsorption [
7,
8,
9,
10,
11]. This study will focus on the first and second challenges (research question I—RQ I), with the ultimate goal of a remote estimation of the FQE (research question II—RQ II), which is going beyond fluorescence for early stress detection.
Regarding RQ I, fluorescence and photosynthesis are coupled by the actual amount of light reaching the photosynthetic surface. Under normal growing conditions, the quantity and quality of photosynthetically active radiation (PAR) varies within the canopy profile. Moreover, shade-adapted leaves often have higher Chl contents and tend to be larger and thinner than sun-adapted leaves [
12]. The biophysical and structural leaf heterogeneity results in a different light spectral response and leaf reflectance. Previous studies have shown that an inaccurate characterization of the at-target incoming radiance leads to an over- or underestimation of the retrieved vegetation properties when using reflectance indices due to a biased incoming radiance estimation [
13]. While inaccuracies in the estimation of the at-target PAR do not alter the relationship between SIF and photosynthesis, an accurate quantification of the flux of photons reaching the photosynthetic surface is essential for a mechanistic interpretation of SIF [
14].
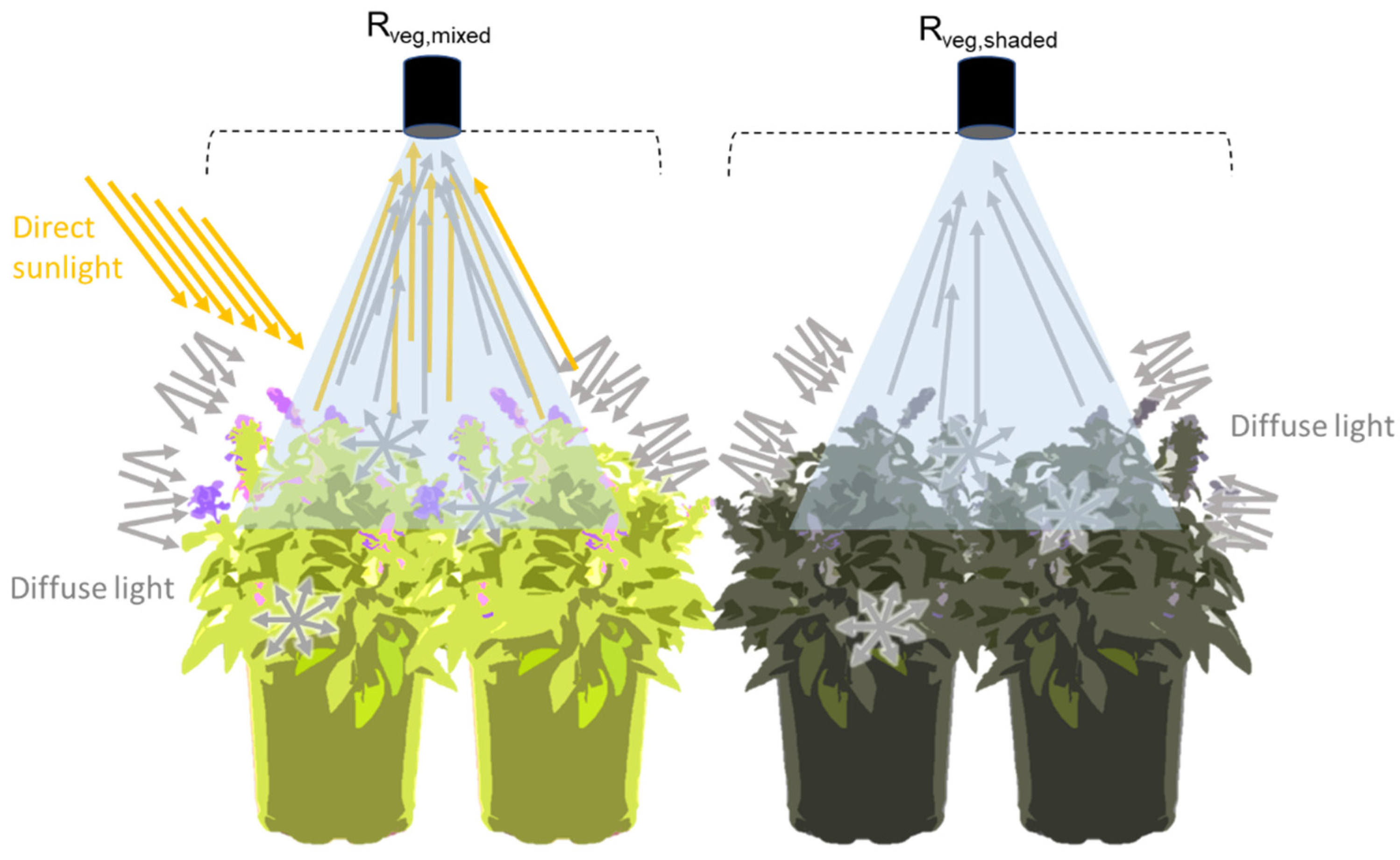
Hence, at the canopy level, the main factors influencing the light received by the photosynthetic surface are the vegetation’s structural properties such as the fraction of vegetation cover (FVC), leaf area index (LAI), and leaf angle distribution function (LADF) [
15]. A different vegetation structure leads to a different vertical distribution of light, resulting in different proportions of shaded and sunlit leaves observed within the canopy. The fluorescence flux measured from the top of the canopy integrates both the fluorescence emitted by the sunlit and shaded fractions of the canopy. Based on previous studies, we assume that when measuring from the nadir, sunlit leaves dominate the measured total fluorescence flux [
16,
17]. Therefore, we hypothesise that by accounting for the proportion of light reaching the sunlit and shaded leaves, as well as nonvegetated surfaces such as soil and woody material (RQ I), the remote estimation of the FQE estimates will be improved (RQ II).
To solve RQ I, the scientific community has developed several remote sensing methods to distinguish between the surface components described above [
18,
19,
20]. For instance, the methods based on spectral mixture analysis (SMA) define the different components of a scene as endmembers with a relatively constant spectral signature. The process of quantifying the abundance of each endmember in the total measured spectrum is known as unmixing. Unmixing methods range from the use of linear spectral combinations [
21] to machine learning approaches [
22], including the use of spectral indices [
23].
Linear unmixing consists of estimating the contribution of each endmember in a given spectrum based on a linear combination of them. Therefore, the measured TOC reflected radiance, if not strongly affected by multiple scattering due to complex vertical structure [
18,
24,
25], can be defined as a linear combination of four surface components: the vegetation sunlit and shaded fraction as well as the soil sunlit and shaded fraction [
26,
27,
28,
29]. SMA has long been used to analyse hyperspectral images for ecological studies [
25,
30]. Based on this approach, methods such as residual spectral unmixing [
31] have been developed to obtain soil properties from hyperspectral measurements. In addition, other authors have proposed the non-negative matrix factorization (NMF) to quantify soil salt content [
32] or wheat biomass [
33] as an example of other SMA techniques.
The spatial resolution of remote hyperspectral sensors implies mixed surface pixels (i.e., consisting of sunlit and shaded leaves, soil and woody material). External ancillary data could be used to determine the spatial heterogeneity of the canopy, but this involves differences in the values obtained due to different illumination and observation angles and temporal dynamics, especially relevant at the satellite level. To overcome these limitations, and in the context of the FLEX mission, we hypothesise that by taking advantage of the benchmark high-spectral resolution sensors and the linear spectral unmixing method, we will be able to quantify the sunlit canopy fraction (FVC
sunlit), which is necessary to calculate the effective APAR flux at the canopy level [
9,
34] (RQ I) to obtain the FQE from remote sensing techniques (RQ II).
With the aim to evaluate the proposed hypothesis, an experiment is designed with a wide range of vegetation fractional cover scenarios, using well-watered potted plants of two different species, characterized by different leaf size and canopy density. Regarding RQ II, as previously introduced, since both species will be measured under the same environmental conditions, we hypothesise that a similar FQE will be obtained at the leaf and canopy scales, regardless of the different FVC. As described in previous studies [
9,
14,
35], further processing and normalisation of initially derived stress proxies such as SIF can lead to a more detailed early stress detection directly related to photosynthetic light responses and a further global carbon assessment. These developments directly support the global monitoring of early vegetation stress using remote sensing techniques.
2. Materials and Methods
2.1. Plant Material and Experimental Setup
To create a plant canopy experiment with varying levels of vegetation cover, we designed a field experiment based on a transect monitoring of potted plants placed in different densities and patterns. The experiment was conducted at the Parc Científic of the University of Valencia (Paterna, Spain) between 25 and 29 July 2022. The average temperature during the experiment was 30 °C.
We used two species with different canopy architectures based on plant height, leaf size, and distribution. Mealy sage (
Salvia farinacea Benth.) was selected as the first plant, which had a dense canopy with a high number of small leaves, approximately 5 cm length. The mealy sage plants (n = 140) were in their first growth stage and had an average height of 30 cm, indicating the first flowering stage. The second plant species, thorn apple (
Datura stramonium L.), had a light canopy with few large leaves, measuring approximately 25 cm. The thorn apple plants (n = 15) were in the vegetative stage and had an average height of 50 cm. To avoid drought stress conditions, all plants were watered twice a day before and after the experiment (
Figure 1C,D).
Several designs were implemented to create vegetation transects with different levels of vegetation cover. The designs were focused on establishing different transects of either mealy sage or thorn apple, depending on the plant density, on a bare soil surface (
Figure 1A). The transect length was approximately 5 m, starting with full plant cover and gradually reducing the plant density towards the end of the transect. The last part of the transect always ended in bare soil. The covered plant area was two metres wide, covered by either 10 plants of mealy sage or 5 plants of thorn apple. Different planting patterns were implemented to mimic different natural layouts with a single species by placing plants in rows or dense blocks on one side of the transect. The main aim was to generate more variability in the proportion of sunlit and shaded canopy due to the shading effect between the plants.
2.2. Multisensor Setup and Transect Sampling Protocol
A multisensor cable-suspended motorized platform (UAV Works, Valencia, Spain) called FluoCat (
Figure 1B) was used to monitor the transects in the vegetation area. This platform was specifically designed to measure solar-induced fluorescence and provide additional data for the interpretation of fluorescence emission [
36]. It is equipped with the Piccolo Doppio dual spectrometer system (GeoSciences, University of Edinburgh, Edinburgh, UK), a MAIAS2 multispectral camera with the ILS irradiance sensor module (SAL Engineering, Russi, Italy), and a TeAx Thermal Capture Fusion camera (TeAx Technology, Wilnsdorf, Germany), as shown in
Figure 2. Two scissor lifts were used 20 m apart to suspend the system over the plant transect using ropes attached to the lifts. The lifts were positioned to ensure a minimum distance of 2.5 m between the sensors and the ground surface.
The Piccolo Doppio (hereafter referred to as Piccolo) is a dual-FOV hyperspectral point system [
37], consisting of two QEPro spectroradiometers (Ocean Optics, Dunedin, FL, USA) and a bifurcated fibre optic with shutters to alternately acquire the downwelling solar irradiance (FOV 180°) and the surface upwelling radiance (FOV ~23°) almost instantaneously. With the height setup mentioned above, a ground FOV of at least 1 m was covered. The fibre optics were connected to two spectroradiometers, one covering the entire VNIR spectral range from 400 to 1180 nm, with a spectral resolution of ~1.5 nm, and another (called FLUO) covering the spectral range from 640 to 790 nm, with a spectral resolution of ~0.3 nm. The TOC surface reflectance was calculated for both radiometers using the signal captured from the upwelling and downwelling channels as described in [
36]. The VNIR measurement was used to spectrally parameterise the vegetation, and the FLUO was used to retrieve the SIF. Each measurement consisted of 15 repetitions to compensate for instrumental and environmental variability.
The MAIAS2 multispectral camera provided reflectance imagery emulating the first 9 spectral bands of Sentinel-2. The camera had a horizontal field of view of 35° and a vertical field of view of 26°. The images were preprocessed by MultiCam Stitcher Pro v1.4, where the reflectance was calculated from the data measured by the irradiance sensor facing upwards and the images taken with the camera facing downwards. The purpose of MAIA was to extract the spatial distribution of the vegetation measured within the FOV of the Piccolo system as a reference for FVC and FVCsunlit.
A TeAx Thermal Capture Fusion camera was also installed. This is a dual system consisting of two snapshot cameras. The first camera has a thermal sensor covering the long wave infrared (LWIR) range between 7.5 and 13.5 μm with an image resolution of 640 × 512 pixels, a horizontal FOV of 25°, a vertical FOV of 20°, and a thermal resolution of 0.05 K. The second camera is an RGB camera with an image resolution of 1600 × 1200 pixels. This camera was not used in this experiment.
Piccolo’s upwelling channel optics and multispectral and thermal cameras were mounted on a gimbal so that they always pointed down. Piccolo’s downwelling channel optics, equipped with a cosine receptor, and MAIA’s irradiance sensor were mounted so that they always pointed upwards. The projected FOVs of Piccolo and MAIA were calculated to cover the vegetation transect surface with a 0.5 metre buffer zone around the projected FOV area of Piccolo.
The FluoCat was moved along the rope to obtain between 15 and 50 readings for each designed transect. Measurements were taken while the platform was stationary. The average time spent on each transect measurement was approximately 15 min, during which we moved the position of the system in small steps (~10 cm). All instruments on the platform were triggered simultaneously.
The sky conditions during the experiment were generally cloudless, and measurements took place avoiding moments of cloud overpasses.
2.3. Leaf Fluorescence
At the same time as the TOC measurements, reference leaf level measurements were performed in both plant species using a USB4000 radiometer (Ocean Optics, Dunedin, FL, USA) and a FluoWat leaf clip [
38]. The FluoWat leaf clip allows the measurement of leaf surface reflectance, transmittance, and up- and downwelling fluorescence. The full chlorophyll fluorescence emission spectrum (650–800 nm) can be obtained by following the protocol in [
39].
Sampling consisted of randomly selecting plants of both species from a set of extra pots. The number of leaves sampled each day varied according to the time spent on the TOC measurements. On day 25, 19 leaves were measured (12 from mealy sage, 7 from thorn apple), on day 26, 24 leaves (6 from mealy sage, 14 from thorn apple), on day 27, 6 leaves of thorn apple, and on day 28, 17 leaves (12 from mealy sage, 5 from thorn apple).
2.4. Sunlit Fractional Vegetation Cover Based on MAIA Multispectral Reflectance Images
The MAIA reflectance images were used to obtain the reference total fractional vegetation cover (MAIA FVC
total) and the reference sunlit fractional vegetation cover (MAIA FVC
sunlit) to validate the FVC
total and FVC
sunlit products derived from the Piccolo hyperspectral point spectroradiometer (
Section 2.5).
Based on the MAIA’s reflectance images and following the protocol described in [
36], we classified each MAIA’s pixel as soil, sunlit, or shaded vegetation fraction. To define the area where the Maia and Piccolo measurements overlapped, the Piccolo FOV was first projected onto the MAIA surface images. To correctly define the coincidence area, the relative position of the optics of the two instruments in the platform gimbal was taken into account (
Figure 3). Later, the normalised difference vegetation index (NDVI) was calculated from the reflectance bands 4 and 7 (NDVI = R783 − R665/R783 + R665) [
40]. Finally, an NDVI threshold was used to discriminate between soil and vegetation, resulting in the reference MAIA FVC
total. Based on the MAIA FVC
total and a reflectance band 7 threshold, a second mask was created to discriminate between sunlit and shaded leaves, resulting in the reference MAIA FVC
sunlit and MAIA FVC
shaded. For further details see [
36].
2.5. Development of Piccolo FVCsunlit Obtained from the TOC Surface Reflectance
2.5.1. Surface Reflectance Unmixing Concept
The spectral response of both vegetation and soil surfaces is mixed in the measured Piccolo VNIR TOC reflectance signal (400–1100 nm). To decompose the fractions of the mixed reflectance signals, the contribution of each element (
) was assumed to add linearly to the total surface reflectance,
. In order to linearly unmix the surface components, the pure reflectance factors of the sunlit soil and the sunlit and shaded vegetation fractions are required, hereafter referred to as the endmember weights (
). The total TOC surface reflectance is then defined as the sum of
effective reflectance surface components (
), given as the endmember weight (
times the pure endmember reflectance spectrum (
The proposed assumption is based on the decomposition of the surface reflectance (
) into three components associated with the soil, the sunlit vegetation, and the shaded vegetation fractions,
By knowing the measured surface endmembers and combining them with a spectral fitting algorithm, it is possible to unravel the abundance of each element. The weights used to combine the endmembers to fit the surface reflectance are understood as the abundance of each endmember. This methodology does not consider the distinction between sunlit and shaded soils, as the spectral feature differences obtained between both types of soils and the white reference with both conditions were lower than 6% across the entire spectrum. This means that the features of the soil spectral signature do not change significantly when the fraction of direct and diffuse light exposition change.
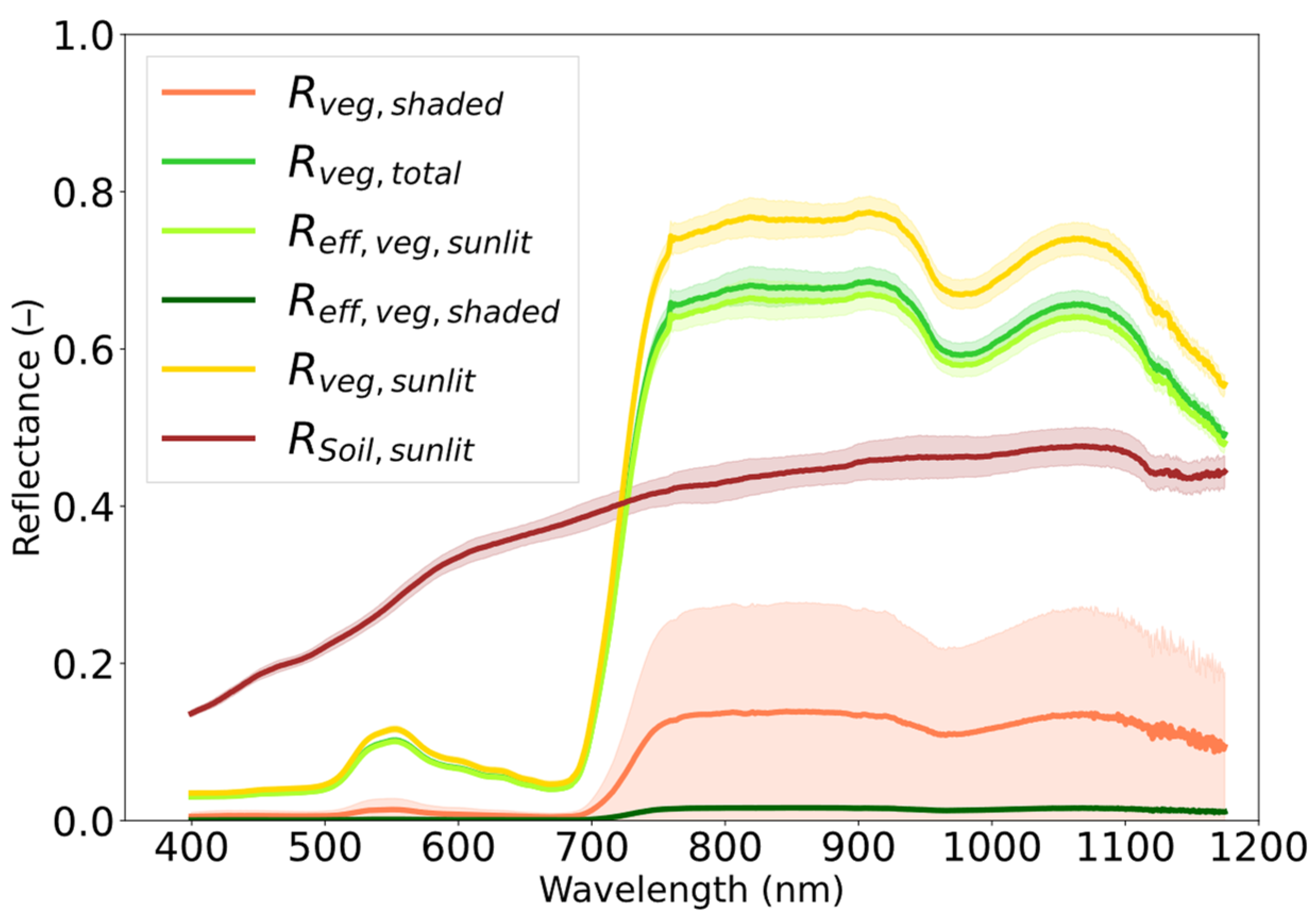
2.5.2. Extraction of Surface Reflectance Endmembers
The proposed unmixing method requires defining three unknown endmembers, these are
(Equation (2)). Soil, vegetation-sunlit, and vegetation-shaded surfaces were obtained based on MAIA FVC characterization. Reflectance endmembers were defined using ground measurements taken during the experiment with the Piccolo at a 2.5 m height. The
endmember was obtained from a soil surface where MAIA FVC
total = 0% (n = 26) (
Figure 4,
). The pure
endmember was obtained by shielding a vegetation area, MAIA FVC
total = 100% (n = 1), from direct sunlight with a panel at the side. Regarding
, since it is not possible to measure a spectrum of 100% sunlit vegetation, the strategy was to obtain it indirectly from a TOC total (mixed) vegetation canopy (i.e., sunlit + shaded) and to correct it for the shaded contribution based on the previously obtained
endmember. Furthermore, it must be considered that both the sunlit and total vegetation endmembers are composed of a direct and a diffuse illumination component, while the shaded endmember is composed only of the diffuse component [
41]. For simplicity, the potted plants were assumed to have a constant vertical structure (i.e., canopy height, leaf density, and leaf area index). A constant ratio between diffuse and direct components was assumed for the sunlit canopies, and a single diffuse component for the shaded canopies.
TOC mixed vegetation (
measurements (assuming that the measured surface is completely covered by vegetation) are defined by the addition of the sunlit and shaded components (Equation (3)),
Moreover,
is defined as,
where
is the reflectance spectrum of all the Piccolo measurements where MAIA FVCtotal is higher than 95% (n = 16);
and
are the weight values of sunlit and shaded fractions obtained from the corresponding MAIA reference images, MAIA FVC
sunlit, and MAIA FVC
shaded, respectively;
Section 2.4 (n = 16);
is the shaded endmember reflectance spectrum previously defined (n = 1).
As the experiment was carried out in summer under high solar illumination angles, the solar sensor target geometry was assumed to have only a minor effect on the distribution and scattering of direct and diffuse light within the canopy. All the endmember spectra used are shown in
Figure 4.
2.5.3. Unmixing Strategy
Based on the Piccolo measurements and the reflectance endmembers defined in the previous section, the different fractions of signal components’ contributions (i.e., soil, sunlit vegetation, and shaded vegetation) were unmixed from the surface apparent reflectance using a non-negative least squares (NNLS) fitting (i.e., ‖
aX −
Y‖
2,
a is the endmember weight and could vary from 0 to 1,
X is an
m ×
n matrix (wavelengths × number of endmembers) where
X ∈ R
n, and
Y ∈ R
m, and
Y is the apparent reflectance for each wavelength (
m)). Two different approaches were implemented, i.e., approach A, where only soil and mixed vegetation endmembers were considered, and approach B, where three endmembers corresponding to sunlit vegetation, shaded vegetation, and soil were considered. In both cases, the implemented Lawson–Hanson NNLS package in R [
42] was used to solve for the contribution (weight and fitted value) of the different signal components. The sum of the estimated weights (
or
should be equal to 1. However, in some scenarios, the sum of the weights resulted in values greater than 1. This was proposed to consider other elements present in the scene and the multiple scattering of vegetation, which mainly influences the shaded fraction of vegetation, in the unmixing process [
25].
Since the weights represent the vegetation cover fraction, in the next section of the manuscript, we refer to Piccolo FVCtotal, Piccolo FVCsunlit, and Piccolo FVCshaded when referring to the total (i.e., mixed sunlit and shaded), sunlit, and shaded vegetation fractions, respectively. Finally, to validate the unmixing results, for both approaches A and B, the spectral-fitting-based FVC from the Piccolo measurements was compared with the image-based reference FVC of the MAIA measurements by means of the root-mean-square error (RMSE) between the two data pairs (i.e., for approach A: Piccolo FVCtotal vs. Maia FVCtotal and Piccolo FVCtotal vs. Maia FVCsunlit; for approach B: Piccolo FVCtotal vs. Maia FVCtotal, and Piccolo FVCsunlit vs. Maia FVCsunlit).
2.6. TOC Fluorescence Retrieval
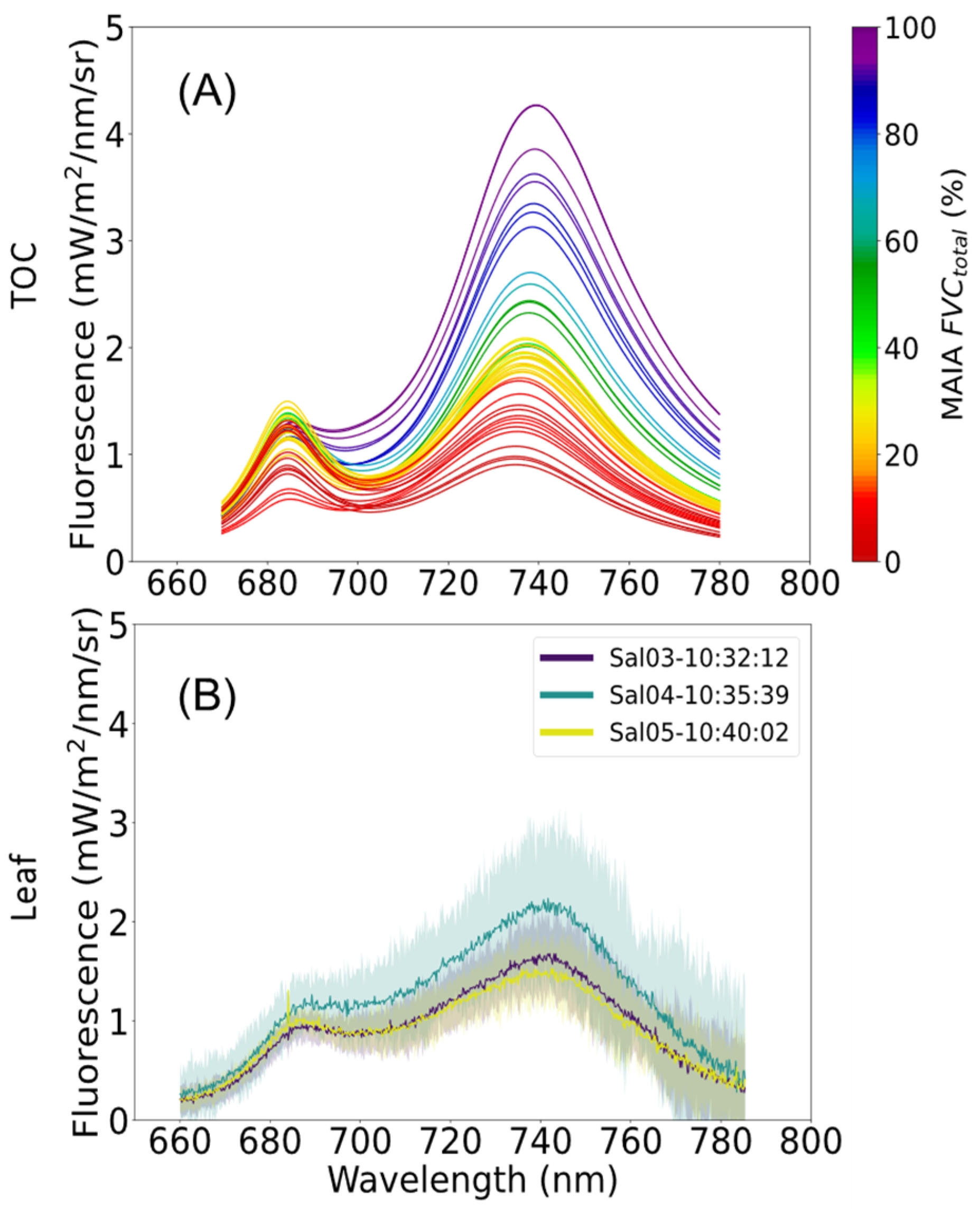
Spectrally resolved TOC fluorescence was retrieved using the SpecFit method [
43], generating the chlorophyll fluorescence spectrum from 670 to 870 nm. The SpecFit method is based on a weighted spectral fitting method which uses the incoming and surface’s reflected radiance to model the actual reflectance and fluorescence spectra by means of an iterative optimization technique.
From the fluorescence spectrum obtained, the total fluorescence flux
was calculated based on photon flux units, integrated for the hemisphere, assuming an ideal diffuse scattering behaviour [
9] as:
2.7. Retrieval of Piccolo-Based Green APAR Flux of Sunlit Canopy Fraction and Fluorescence Quantum Efficiency
To obtain the sunlit green fluorescence (Equation (6)), also defined as the effective surface fluorescence emission, the green APAR flux of the sunlit canopy fraction (
) was calculated based on the Piccolo FVC
sunlit previously retrieved:
where the leaf level absorbance factor (A
leaf) was assumed to be constant at 0.84 [
9,
44,
45], and the PAR was spectrally integrated based on photon flux units obtained from the Piccolo’s downwelling radiance channel. The proposed method assumed that the sunlit fraction was the dominant surface triggering fluorescence emission, and that chlorophyll absorption was constant at the leaf level.
Both the total fluorescence and green sunlit absorbed PAR photon fluxes were then used to calculate the fluorescence quantum efficiency (FQE) as shown by Equation (7).
Further we compared the FQE obtained at the TOC with the FQE values obtained in situ at the leaf level with the FluoWat leaf clip, where the FQE was estimated according to the same formula, considering a FVCsunlit of 1 (i.e., 100% FVCsunlit). The values obtained for each transect were compared with the results at the leaf level made within a time span of 20 min before and after each transect. The transects without leaf measurements in that time range were discarded for the comparison. Two tests were used to compare the pairwise variance of the data from the different scales. Fligner’s test was used to discriminate between different types of data variance. Either Student’s t-test (equal variances) or Welch’s t-test (unequal variances) was used to compare data pairs. All calculations were performed using the Python library Scipy version 1.1 (“PyPI ⋅ The Python Package Index”, 2021).
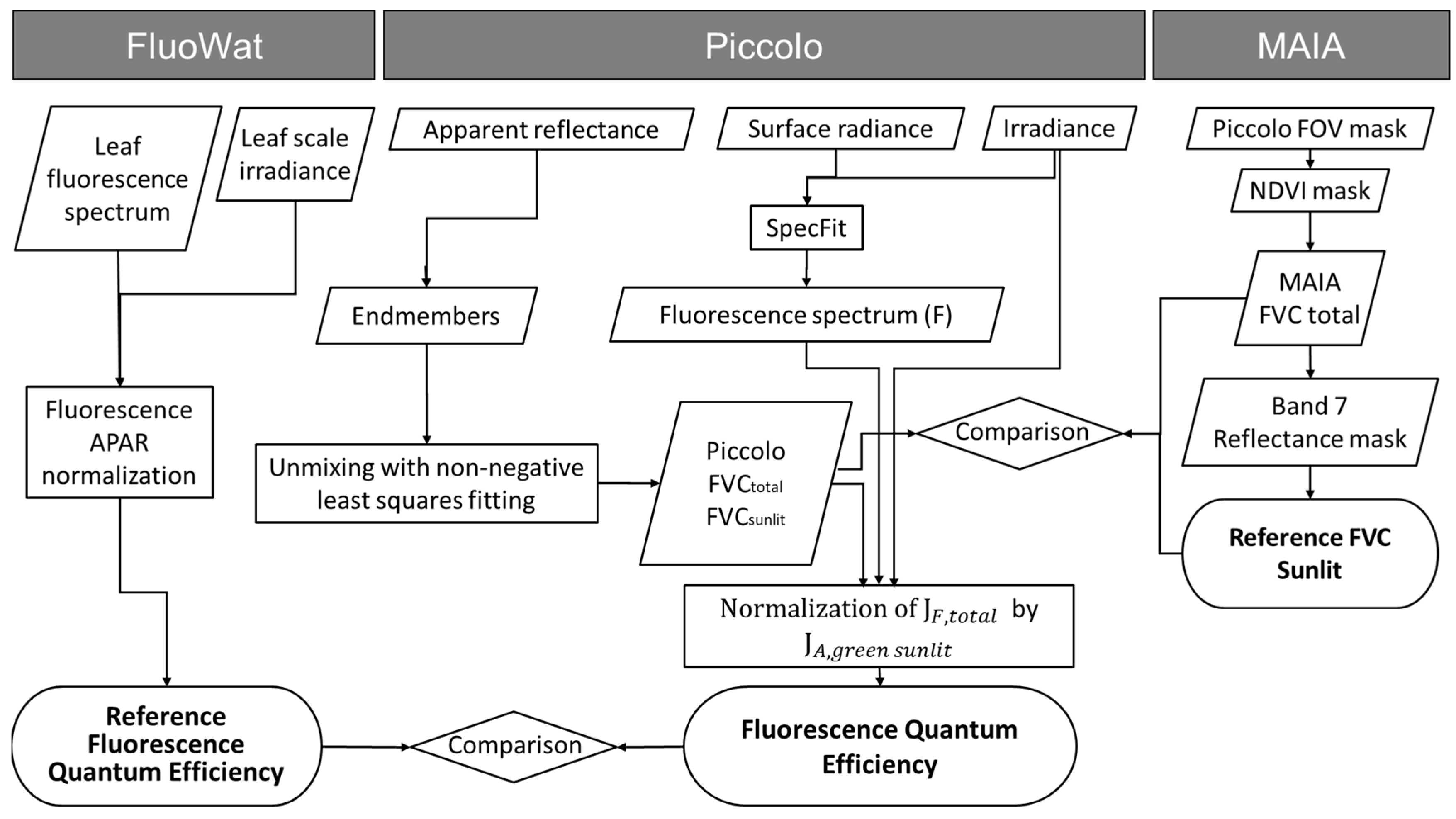
Figure 5 summarises the processing chain applied to obtain the FQE, considering the necessary input parameters and the advanced products obtained due to the sensor’s synergies.
4. Discussion
The accurate quantification of the vegetation fluorescence quantum efficiency using remote sensing techniques requires the prior structural characterization of the canopy object acquired in the sensor’s field of view (i.e., point sensor’s field of view and/or imaging sensor’s pixel spatial resolution) [
8,
10,
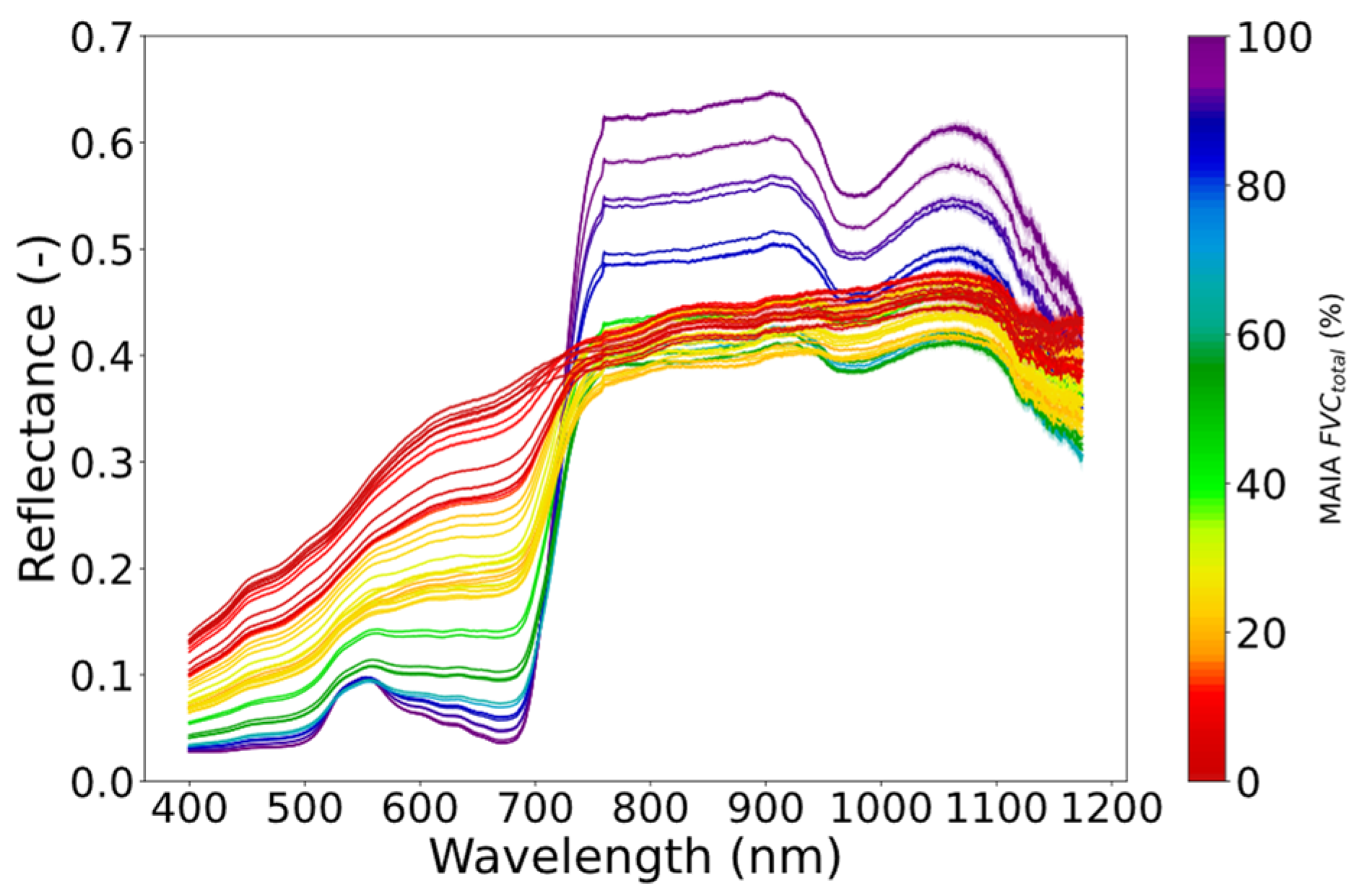
11]. As previously mentioned, this requires the quantification of the amount of incident PAR and APAR, and a correction for the fluorescence scattering and reabsorption within the canopy. In this study, in order to address RQ I, which focuses on disentangling the proportion of sunlit and shaded absorbed PAR, an ad hoc experimental setup was designed to provide a wide range of FVC scenarios by varying the plant density within the sensor FOV, but keeping the vertical vegetation structure constant, thus reducing the variable effect of the canopy scattering and reabsorption. In addition, the use of two plant types with different architectures was used to explore RQ II, assuming that regardless of the FVC and the proportion of sunlit and shaded vegetation, a similar FQE is obtained at the leaf and canopy scales when plants grow under the same environmental conditions. The resulting dataset represented the full range of FVC
total from 0 to 100% (
Figure 6 and
Figure 7), having a peak at 70 measurements for 0–10% FVC
sunlit and decreasing to 16 measurements for 80% FVC
sunlit (
Figure 6).
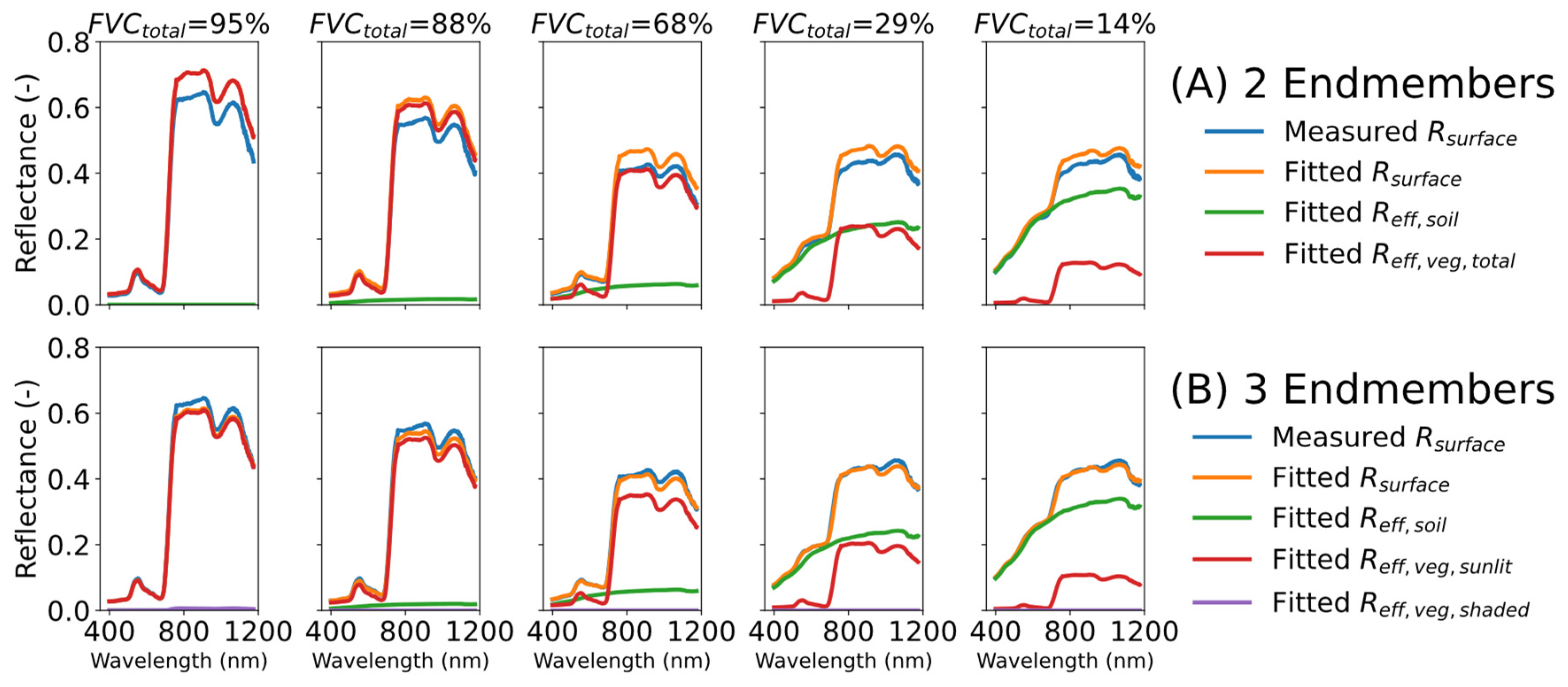
In this study the sunlit and shaded green areas (RQ I) were quantified using a linear unmixing algorithm based on the surface reflectance fitting with specific endmember components. Two different approaches were compared, a two-endmember (R
eff,soil, R
eff,veg,total, approach A) and a three-endmember (R
eff,soil, R
eff,veg,sunlit, and R
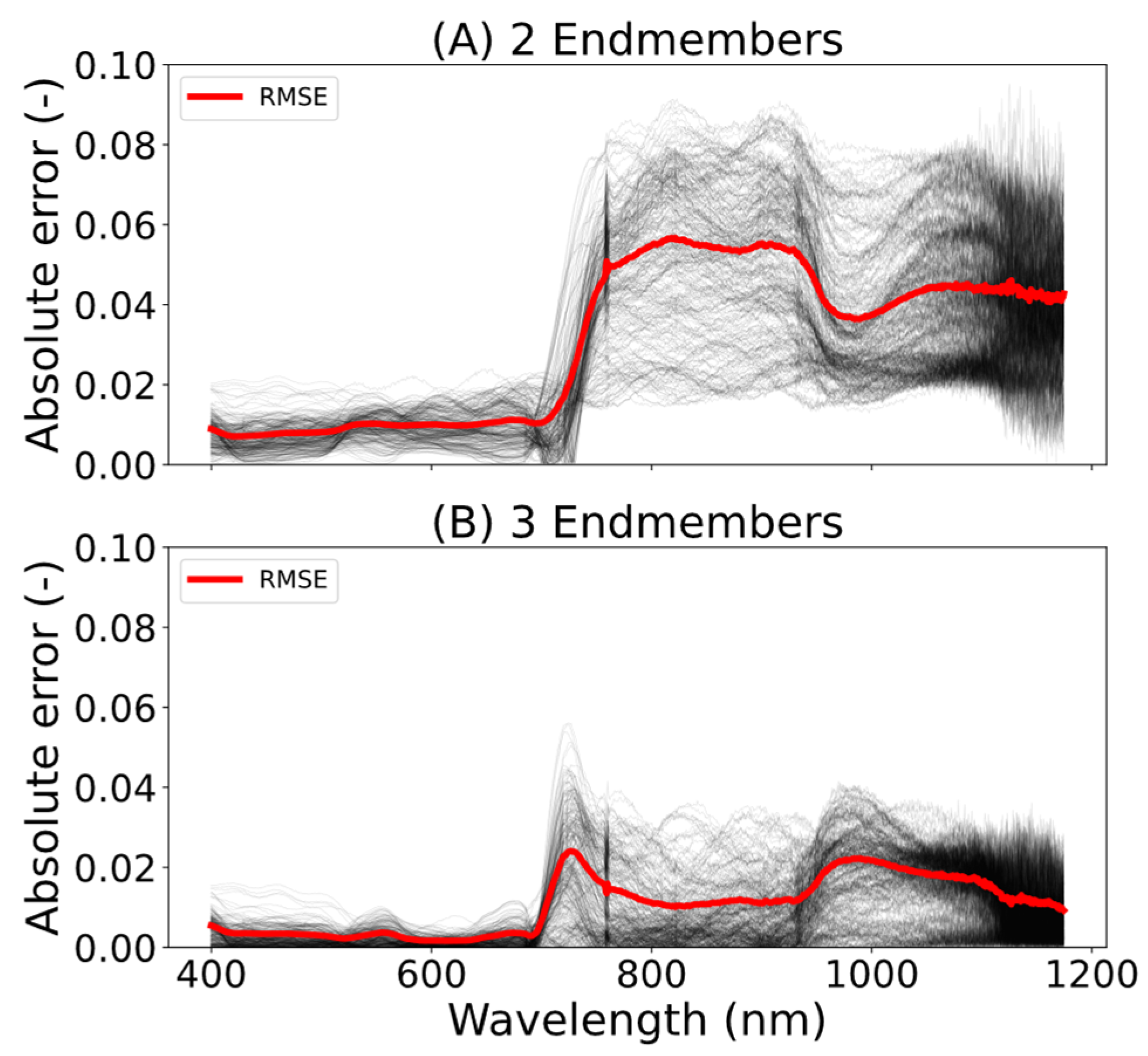
eff,veg,shade, approach B) approach. When comparing the reference and fitted reflectance spectra, the results showed that decomposing the signal using three endmembers provided better results than with two endmembers, reducing the averaged spectral RMSE between the actual reflectance spectrum and the fitted spectrum from 0.03 to 0.01 (
Figure 10). The largest difference in terms of RMSE was observed after the red edge, in the NIR range. This can be explained by the nonlinear behaviour of the canopy’s multiple scattering caused by the interaction of the light within the canopy [
18]. Moreover, this is the reason why the three-endmembers provided better results than the two-endmember approach. The addition of a shaded vegetation endmember introduced a diffuse component into the fit. This improved the results of the spectral vegetation unmixing in the NIR range in [
25].
The fitting accuracy of the presented study is in the same order of magnitude as the results presented in [
46,
47,
48]. These studies, however, reported a higher number of endmembers (e.g., 4–5). For instance, Ref. [
47] added to the set of four endmembers (sunlit soil, shaded soil, sunlit vegetation, and shaded vegetation) a fifth endmember with zero reflectance, called photometric shade, which included the multiple scattered signals from the shaded surfaces in the fitting. Their study achieved a maximum RMSE of 0.01 across the optical range. We hypothesise that the reason why our results are consistent with previous studies, despite the use of only three endmembers, is due to the simplicity of the proposed measurement setup, where a reduced number of elements, i.e., flat terrain, homogenous soil surface, and two vegetation structures, define the measured transect. Furthermore, in this study it was not necessary to add a shaded soil endmember because, as assumed by [
49], the surface true reflectance was the same, and the differences between sunlit and shaded soil reflectance were mostly due to scattered light from the canopy. Therefore, in the present work, the contribution of the shaded soil was mostly fitted to the shaded vegetation signal, which explains why the addition of FVC
sunlit and FVC
shaded provided values greater than one.
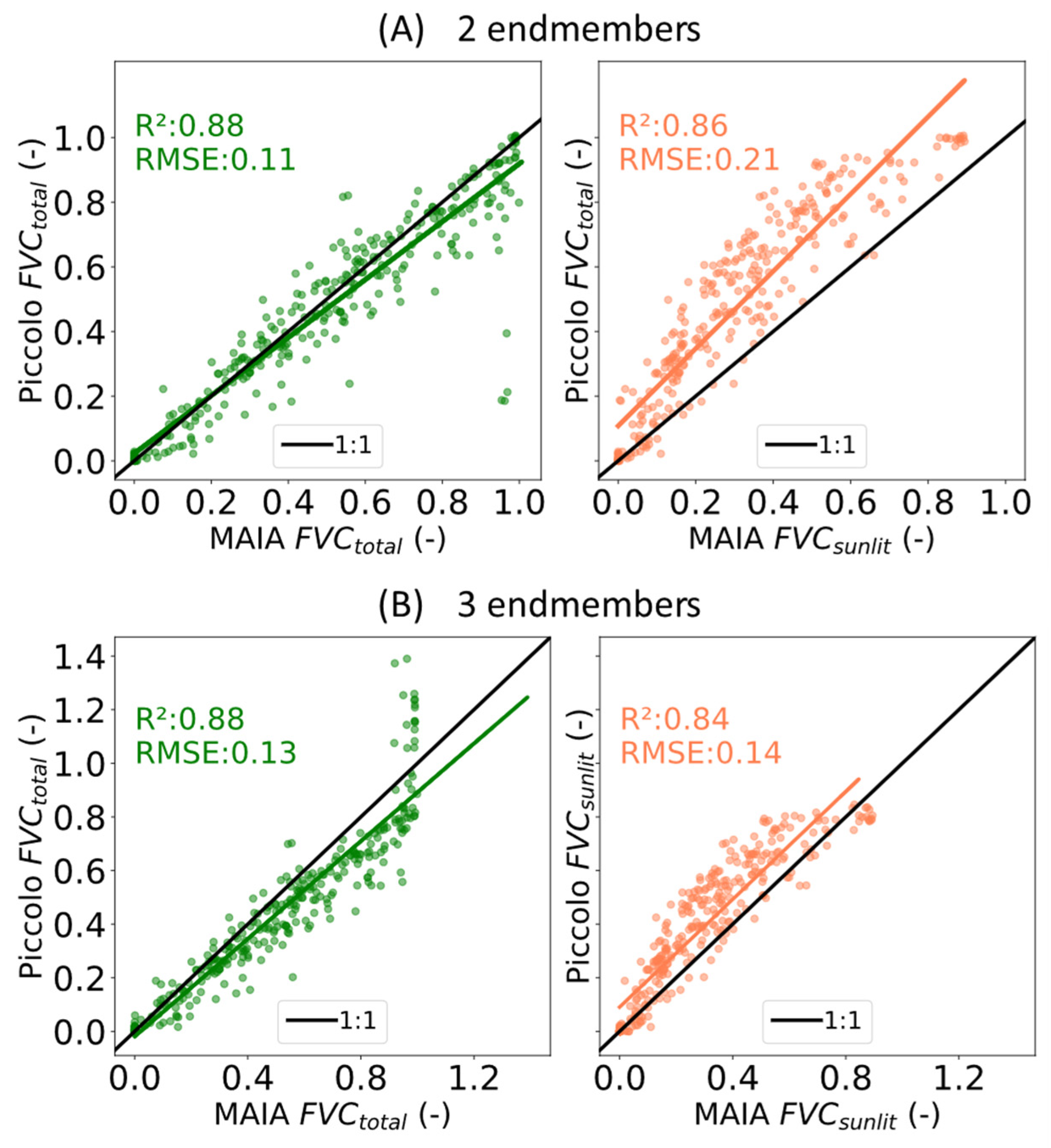
Regarding the discrimination between FVC
total and FVC
sunlit, both the two-endmember and three-endmember approaches provided accurate results when comparing Piccolo FVC
total and MAIA FVC
total (R
2 = 0.88 and RMSE = 0.11–0.13). However, only the three-endmember approach was able to discriminate between FVC
total and FVC
sunlit, improving the estimation of FVC
sunlit (RMSE = 0.14) compared to using the two-endmember approach’s FVC
total used as an estimation of the sunlit fraction (RMSE = 0.21) (
Figure 11). Nevertheless, the results obtained in this study are in the same order of magnitude as other works where more complex hyperspectral unmixing methods have been proposed [
50,
51,
52].
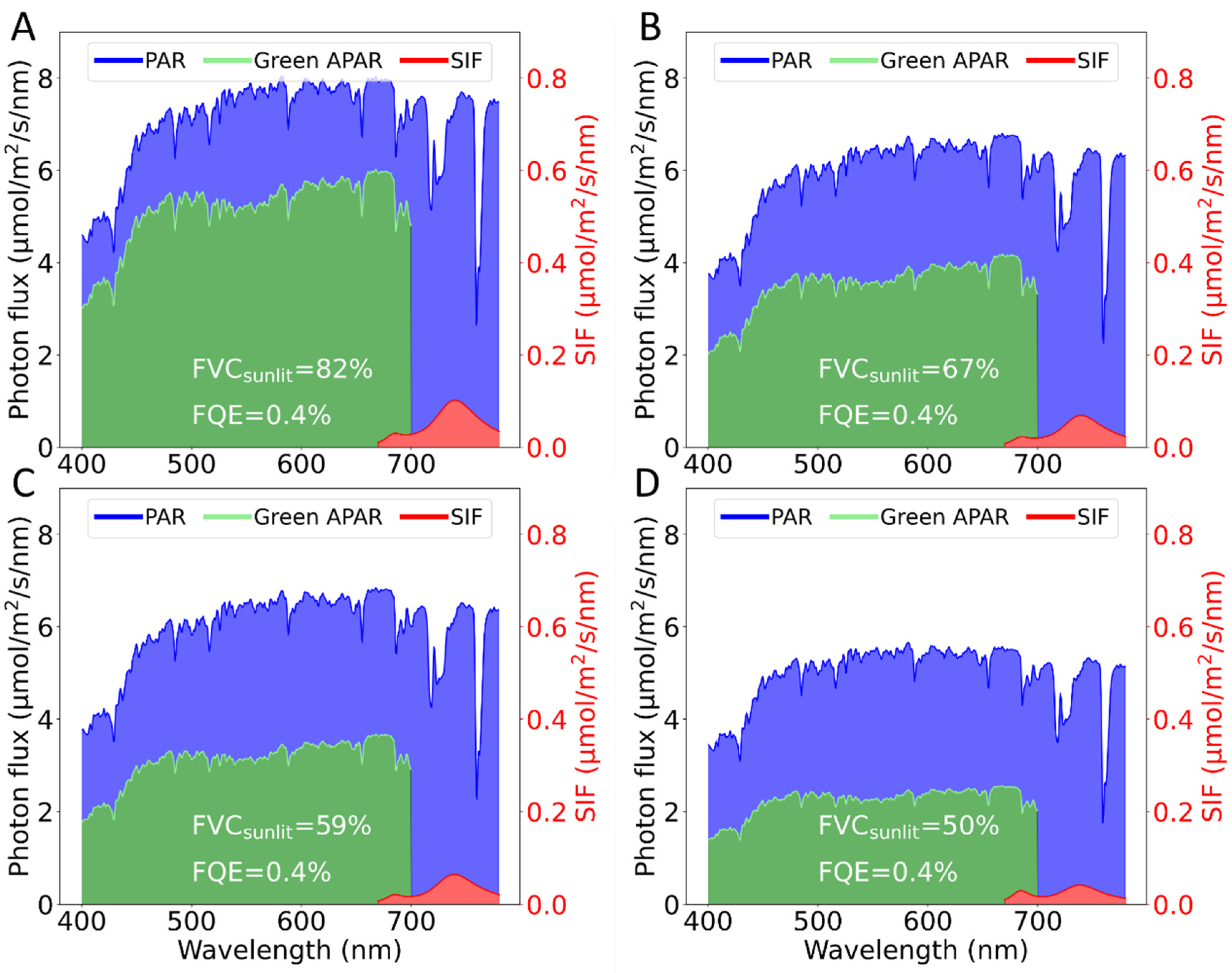
Compared to previous studies where the total (i.e., sunlit + shaded) incoming radiation was estimated using vegetation indices [
53,
54], the work presented in this study, provides the opportunity to calculate in physical units the actual energy flux arriving at the sunlit photosynthetic surface (
) (
Figure 12A). Furthermore, the results presented in
Figure 13B, where the correlation between SIF and MAIA FVC
sunlit (R
2 = 0.76) is higher than when correlated with FVC
total (R
2 = 0.58), corroborate the hypothesis that the measured TOC SIF is mainly emitted by sunlit leaves [
16,
17]. Hence, both
and the total calculated fluorescence flux (
) show a strong relationship with the FVC
sunlit. The combination of these two terms allows us to decouple the changes in
dynamics driven only by the physiological component [
9].
Regarding RQ II, the quantification of the incident APAR and emitted fluorescence photons flux allows the remote quantification of the real fluorescence quantum efficiency (FQE) (
Figure 12C). Interestingly, except for FVC
sunlit below 20%, similar FQE values were obtained throughout the experiment regardless of the vegetation fractional cover (
Figure 12C). These results support the hypothesis that under equal environmental conditions, and without any applied stress such as drought or nitrogen excess/deficit, similar FQE values should be obtained (
Figure 13). An accurate estimation of FQE is essential for a quantitative interpretation of SIF and its role in early plant stress detection. Regarding the high FQE values obtained when FVC
sunlit < 20%, these results could be explained by the positive (and overestimated) SIF values retrieved over samples where bare soil predominates in the measurement transect (
Figure 8, red lines). In this study, unfortunately, these observations indicate an overestimation of the retrieved values obtained by the Specfit method in cases with a low FVC. These results are consistent with the findings in [
43], where a decrease in performance is observed with a low LAI. These results do not alter the findings of the present study but suggest a revision of the proposed retrieval method when low FVC values are investigated. Consequently, measurements with FVC
total lower than 20% were discarded for the comparison between TOC and leaf level measurements (
Figure 14).
In general terms, the FQE-TOC underestimated the retrieved FQE at the leaf level (
Figure 14). In 4 of the 11 transects analysed, the TOC measurements were statistically different from the leaf level measurements. However, despite the slight differences, the FQE obtained at the canopy and leaf levels showed values in the same range and the variability along the experiment was similar. A possible explanation for the differences between them is a combined effect of an overestimation of the fluorescence retrieved, and the omission of the canopy scattering and reabsorption effects when measuring at the TOC due to the light interactions with the canopy [
55].
This experiment tested a methodology capable of retrieving the FQE from hyperspectral TOC data, with a protocol to validate the spectral-based FQE values with image-based measurements. However, before extrapolating the proposed methodologies to a larger and more complex ecosystems, further studies are needed to properly characterize and correct for the effects of the canopy’s vertical structure in more heterogeneous surfaces, and to improve the fluorescence retrieval to downscale TOC measurements to the leaf—photosynthetic surface—level. In addition, a further characterization of green (i.e., specific pigment absorption) and nongreen (i.e., woody material and soil absorption libraries) endmembers as well as the atmospheric correction should be incorporated into the proposed methodology. Given the proposed improvements, it is expected that the next studies will apply this methodology to larger scales, such as airborne or satellite measurements, allowing the remote estimation of FQE and consequently the early detection of crop and global vegetation stress.
5. Conclusions
Combining a state-of-the-art set of instruments mounted on a cable-driven platform, this work exploited the synergies between a single-point high-spectral resolution spectroradiometer and a VNIR multispectral imaging camera for reflectance and fluorescence interpretation. This setup allowed the development of an unmixing retrieval scheme to disentangle the fraction of sunlit vegetation cover from the total TOC reflectance, validated by the VNIR imaging camera. By normalising the retrieved TOC fluorescence by the green APAR flux (i.e., sunlit FVC × PAR) the fluorescence quantum yield was quantified. The proposed methodology allows the comparison and validation of the FQE at different scales, from leaf to canopy, with proximal and remote sensors. Furthermore, the methodology presented here, together with the FluoCat system, provides the ability to operate and test similar protocols over large fields, and thus characterize the heterogeneity of the surface, which brings the opportunity to explain/understand the signal measured by the sensor over such an area. In this study, the spectral signature of the sunlit vegetation fraction was used to describe the vegetated surface, which is crucial for Cal/Val activities. From the point of view of the remote sensing methodologies, the quantification of the components present inside the measured area overcomes the limitation of the structural effects on the fluorescence signal, demonstrating that the proposed spectral approach has the potential to unravel the intricate complexities of underlying vegetated surfaces with hyperspectral sensors. The development of more robust methods to obtain products beyond fluorescence, such as the surface effective PAR and APAR, and most importantly the FQE, will allow the remote quantification of the vegetation energy balance, closing the gap between the flux of photons absorbed, dissipated, and used for photochemistry, which is critical for a remote quantification of photosynthesis. Future research lines are needed to investigate the extrapolation of this method to larger scales, with the aim of studying the FQE’s temporal dynamics and variable stress conditions for a global monitoring of vegetation using orbiting satellites.