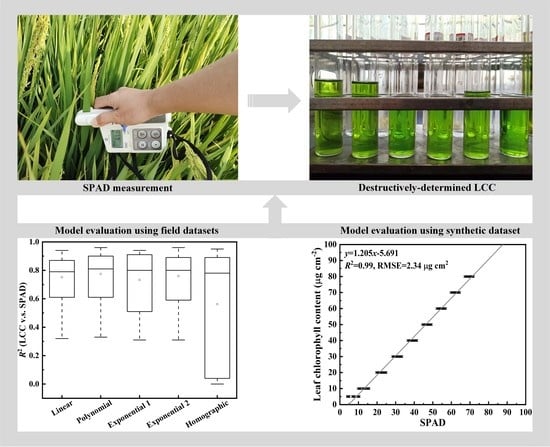
Evaluation of the Methods for Estimating Leaf Chlorophyll Content with SPAD Chlorophyll Meters
Abstract
1. Introduction
2. Materials and Methods
2.1. Datasets
2.1.1. Field Datasets
2.1.2. Simulated Dataset with the PROSPECT Model
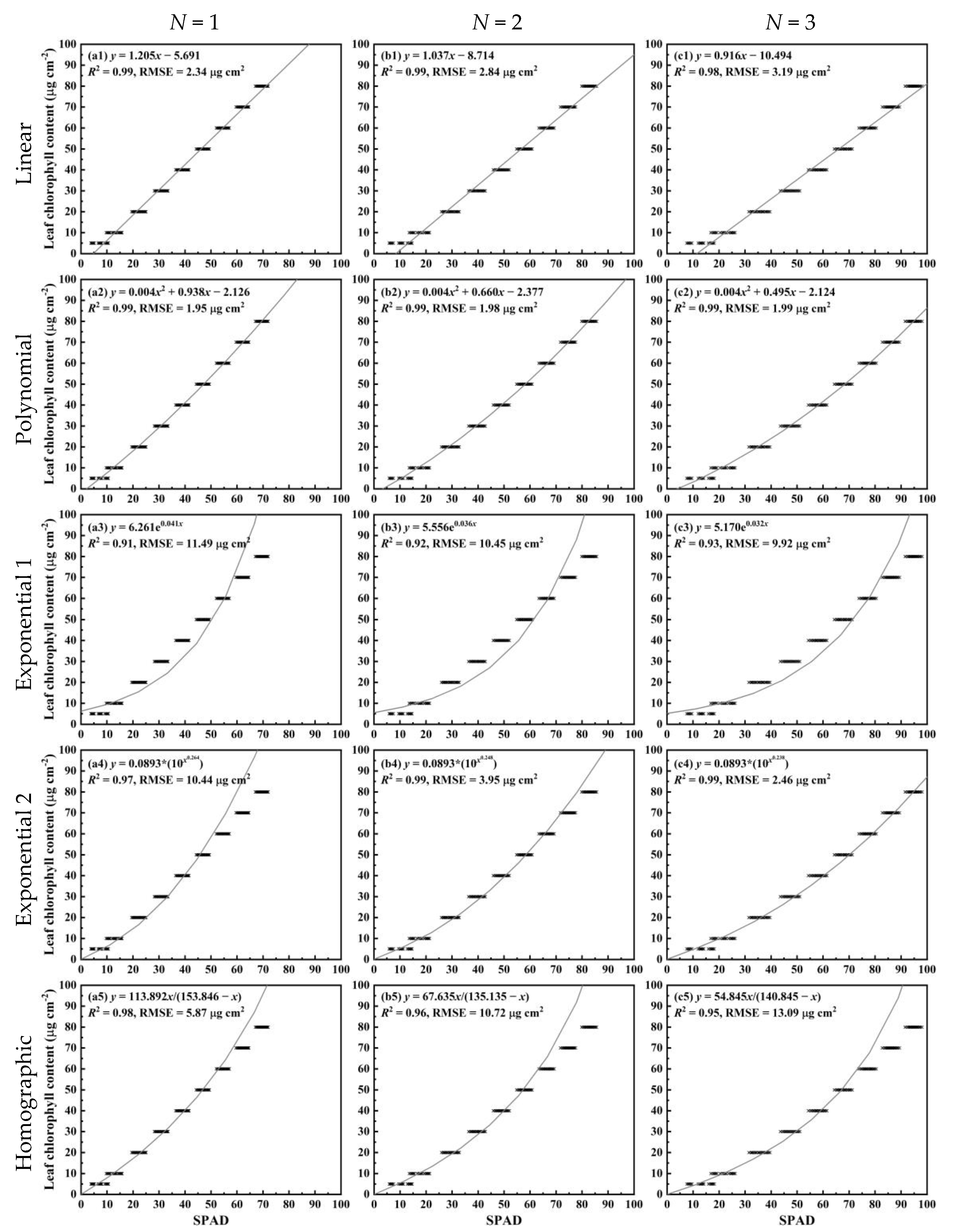
2.2. Mathematical Functions for Relationships between SPAD Readings and LCC
2.3. Accuracy Assessment
3. Results
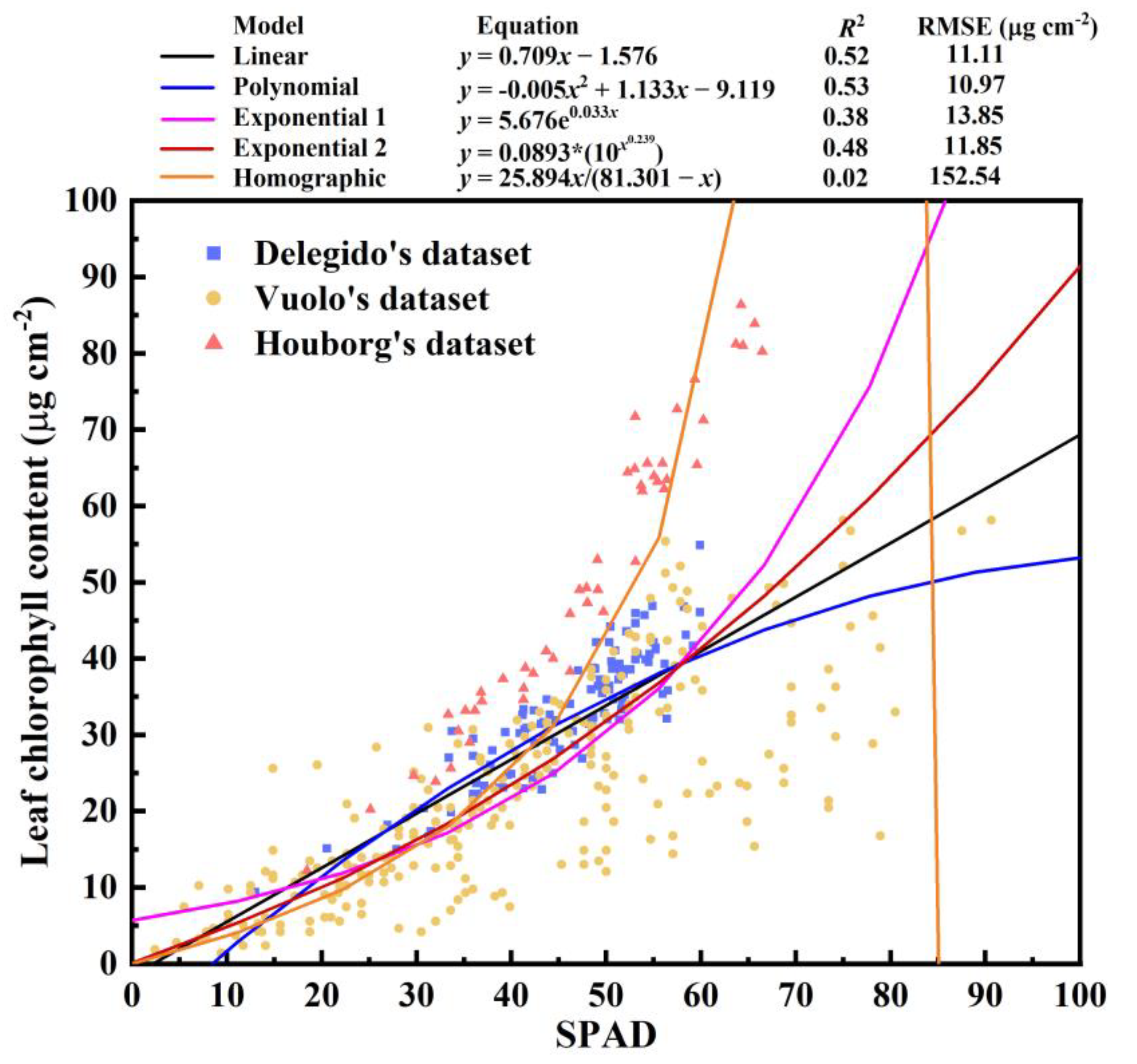
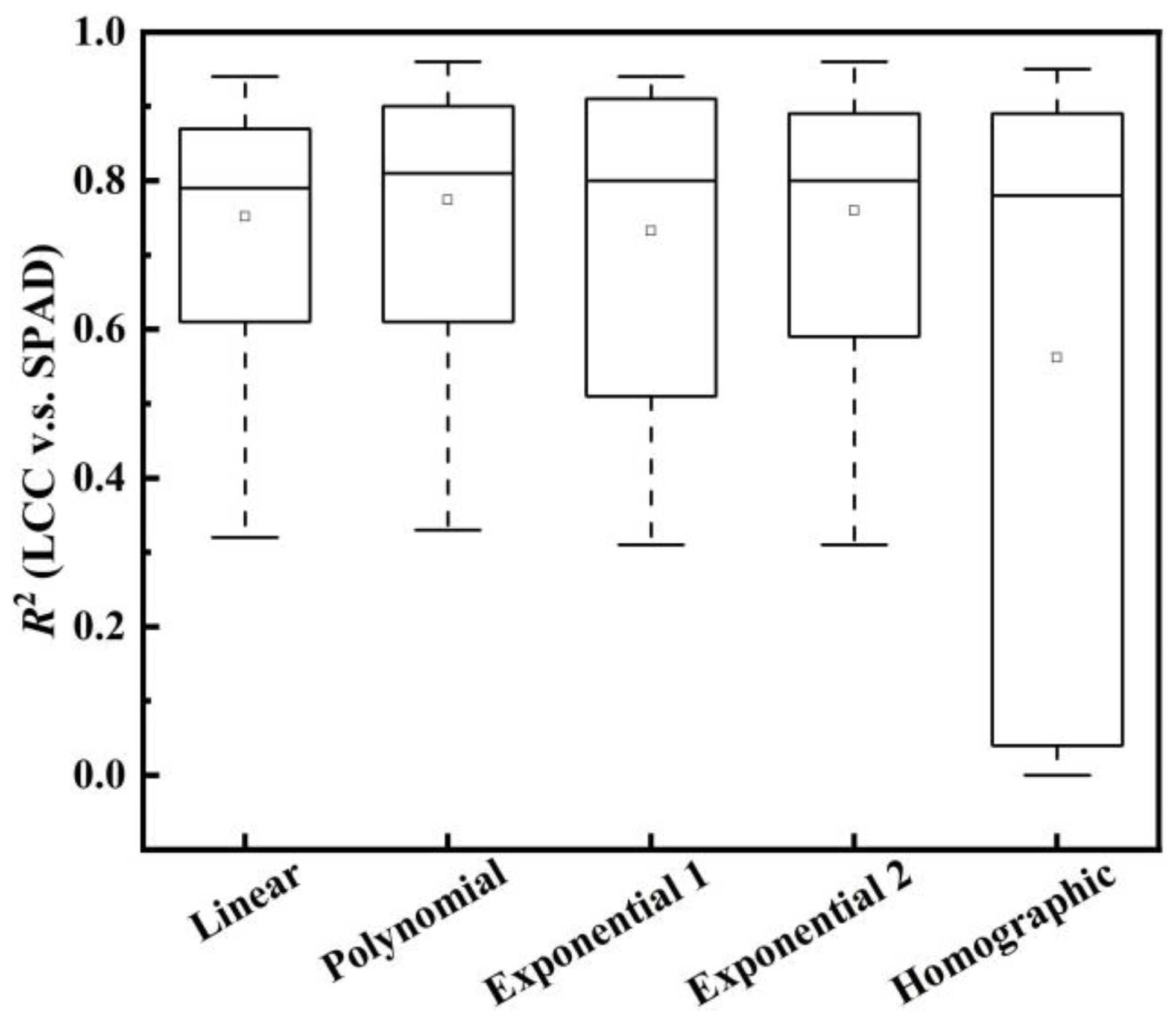
3.1. All the Field Datasets Together
3.2. For Each Field Dataset
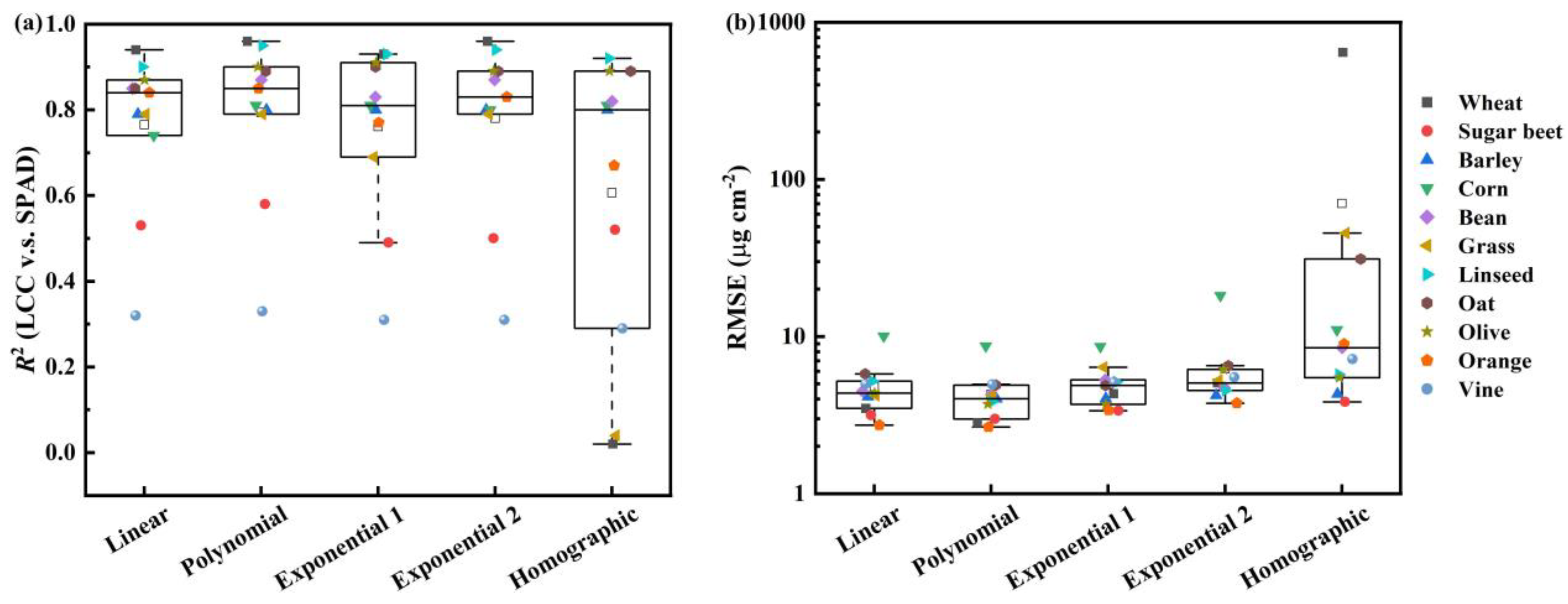
3.3. For Each Species
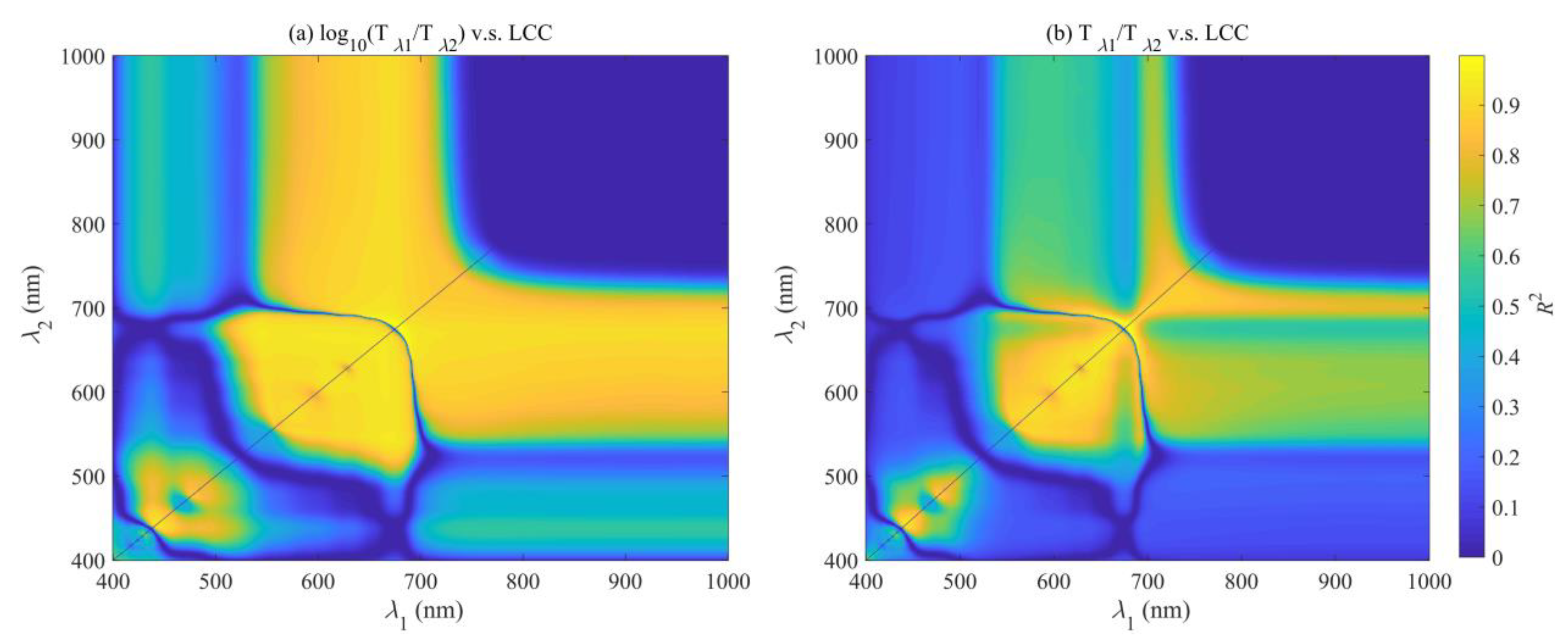
3.4. For the Simulated Dataset from the PROSPECT Model
4. Discussion
4.1. Relationship between LCC and SPAD Readings
4.2. Comparison of Different Functions
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Porcar-Castell, A.; Tyystjärvi, E.; Atherton, J.; Van Der Tol, C.; Flexas, J.; Pfündel, E.E.; Moreno, J.; Frankenberg, C.; Berry, J.A. Linking Chlorophyll a Fluorescence to Photosynthesis for Remote Sensing Applications: Mechanisms and Challenges. J. Exp. Bot. 2014, 65, 4065–4095. [Google Scholar] [CrossRef]
- Croft, H.; Chen, J.M.; Luo, X.; Bartlett, P.; Chen, B.; Staebler, R.M. Leaf Chlorophyll Content as a Proxy for Leaf Photosynthetic Capacity. Glob. Chang. Biol. 2017, 23, 3513–3524. [Google Scholar] [CrossRef] [PubMed]
- Palta, J.P. Leaf Chlorophyll Content. Remote Sens. Rev. 1990, 5, 207–213. [Google Scholar] [CrossRef]
- Wang, S.; Li, Y.; Ju, W.; Chen, B.; Chen, J.; Croft, H.; Mickler, R.A.; Yang, F. Estimation of Leaf Photosynthetic Capacity from Leaf Chlorophyll Content and Leaf Age in a Subtropical Evergreen Coniferous Plantation. J. Geophys. Res. Biogeosciences 2020, 125, e2019JG005020. [Google Scholar] [CrossRef]
- Zhang, J.; Blackmer, A.M.; Ellsworth, J.W.; Koehler, K.J. Sensitivity of Chlorophyll Meters for Diagnosing Nitrogen Deficiencies of Corn in Production Agriculture. Agron. J. 2008, 100, 543–550. [Google Scholar] [CrossRef]
- Eitel, J.U.H.; Long, D.S.; Gessler, P.E.; Hunt, E.R. Combined Spectral Index to Improve Ground-Based Estimates of Nitrogen Status in Dryland Wheat. Agron. J. 2008, 100, 1694–1702. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- Delegido, J.; Vergara, C.; Verrelst, J.; Gandía, S.; Moreno, J. Remote Estimation of Crop Chlorophyll Content by Means of High-Spectral-Resolution Reflectance Techniques. Agron. J. 2011, 103, 1834–1842. [Google Scholar] [CrossRef]
- Nishio, J.N. Why Are Higher Plants Green? Evolution of the Higher Plant Photosynthetic Pigment Complement. Plant Cell Environ. 2000, 23, 539–548. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Relationships between Leaf Pigment Content and Spectral Reflectance across a Wide Range of Species, Leaf Structures and Developmental Stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W. The Role of Xanthophyll Cycle Carotenoids in the Protection of Photosynthesis. Trends Plant Sci. 1996, 1, 21–26. [Google Scholar] [CrossRef]
- Barker, D.H.; Seaton, G.G.R.; Robinson, S.A. Internal and External Photoprotection in Developing Leaves of the CAM Plant Cotyledon Orbiculata. Plant Cell Environ. 1997, 20, 617–624. [Google Scholar] [CrossRef]
- Castelli, F.; Contillo, R.; Miceli, F. Non-Destructive Determination of Leaf Chlorophyll Content in Four Crop Species. J. Agron. Crop Sci. 1996, 177, 275–283. [Google Scholar] [CrossRef]
- Dong, T.; Shang, J.; Chen, J.M.; Liu, J.; Qian, B.; Ma, B.; Morrison, M.J.; Zhang, C.; Liu, Y.; Shi, Y.; et al. Assessment of Portable Chlorophyll Meters for Measuring Crop Leaf Chlorophyll Concentration. Remote Sens. 2019, 11, 2706. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of Total Carotenoids and Chlorophylls a and b of Leaf Extracts in Different Solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Shoaf, W.T.; Lium, B.W. Improved Extraction of Chlorophyll a and b from Algae Using Dimethyl Sulfoxide. Limnol. Oceanogr. 1976, 21, 926–928. [Google Scholar] [CrossRef]
- Barnes, J.D.; Balaguer, L.; Manrique, E.; Elvira, S.; Davison, A.W. A Reappraisal of the Use of DMSO for the Extraction and Determination of Chlorophylls a and b in Lichens and Higher Plants. Environ. Exp. Bot. 1992, 32, 85–100. [Google Scholar] [CrossRef]
- Shinano, T.; Lei, T.T.; Kawamukai, T.; Inoue, M.T.; Koike, T.; Tadano, T. Dimethylsulfoxide Method for the Extraction of Chlorophylls a and b from the Leaves of Wheat, Field Bean, Dwarf Bamboo, and Oak. Photosynthetica 1996, 32, 409–415. [Google Scholar]
- Dugo, P.; Cacciola, F.; Kumm, T.; Dugo, G.; Mondello, L. Comprehensive Multidimensional Liquid Chromatography: Theory and Applications. J. Chromatogr. A 2008, 1184, 353–368. [Google Scholar] [CrossRef]
- Ritchie, R.J. Consistent Sets of Spectrophotometric Chlorophyll Equations for Acetone, Methanol and Ethanol Solvents. Photosynth. Res. 2006, 89, 27–41. [Google Scholar] [CrossRef]
- Richardson, A.D.; Duigan, S.P.; Berlyn, G.P. An Evaluation of Noninvasive Methods to Estimate Foliar Chlorophyll Content. New Phytol. 2002, 153, 185–194. [Google Scholar] [CrossRef]
- Cerovic, Z.G.; Masdoumier, G.; Ghozlen, N.B.; Latouche, G. A New Optical Leaf-Clip Meter for Simultaneous Non-Destructive Assessment of Leaf Chlorophyll and Epidermal Flavonoids. Physiol. Plant. 2012, 146, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, T.S.; Gardiner, E.S.; Comer, G.S. Modeling the Relationship between Extractable Chlorophyll and SPAD-502 Readings for Endangered Plant Species Research. J. Nat. Conserv. 2009, 17, 123–127. [Google Scholar] [CrossRef]
- Shibaeva, T.G.; Mamaev, A.V.; Sherudilo, E.G. Evaluation of a SPAD-502 Plus Chlorophyll Meter to Estimate Chlorophyll Content in Leaves with Interveinal Chlorosis. Russ. J. Plant Physiol. 2020, 67, 690–696. [Google Scholar] [CrossRef]
- Steele, M.R.; Gitelson, A.A.; Rundquist, D.C. A Comparison of Two Techniques for Nondestructive Measurement of Chlorophyll Content in Grapevine Leaves. Agron. J. 2008, 100, 779–782. [Google Scholar] [CrossRef]
- Casa, R.; Castaldi, F.; Pascucci, S.; Pignatti, S. Chlorophyll Estimation in Field Crops: An Assessment of Handheld Leaf Meters and Spectral Reflectance Measurements. J. Agric. Sci. 2015, 153, 876–890. [Google Scholar] [CrossRef]
- Botha, E.J.; Leblon, B.; Zebarth, B.J.; Watmough, J. Non-Destructive Estimation of Wheat Leaf Chlorophyll Content from Hyperspectral Measurements through Analytical Model Inversion. Int. J. Remote Sens. 2010, 31, 1679–1697. [Google Scholar] [CrossRef]
- Schaper, H.; Chacko, E.K. Relation between Extractable Chlorophyll and Portable Chlorophyll Meter Readings in Leaves of Eight Tropical and Subtropical Fruit-Tree Species. J. Plant Physiol. 1991, 138, 674–677. [Google Scholar] [CrossRef]
- Monje, O.A.; Bugbee, B. Inherent Limitations of Nondestructive Chlorophyll Meters: A Comparison of Two Types of Meters. HortScience 1992, 27, 69–71. [Google Scholar] [CrossRef]
- Uddling, J.; Gelang-Alfredsson, J.; Piikki, K.; Pleijel, H. Evaluating the Relationship between Leaf Chlorophyll Concentration and SPAD-502 Chlorophyll Meter Readings. Photosynth. Res. 2007, 91, 37–46. [Google Scholar] [CrossRef]
- Markwell, J.; Osterman, J.C.; Mitchell, J.L. Calibration of the Minolta SPAD-502 Leaf Chlorophyll Meter. Photosynth. Res. 1995, 46, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Coste, S.; Baraloto, C.; Leroy, C.; Marcon, É.; Renaud, A.; Richardson, A.D.; Roggy, J.-C.; Schimann, H.; Uddling, J.; Hérault, B. Assessing Foliar Chlorophyll Contents with the SPAD-502 Chlorophyll Meter: A Calibration Test with Thirteen Tree Species of Tropical Rainforest in French Guiana. Ann. For. Sci. 2010, 67, 607. [Google Scholar] [CrossRef]
- Vuolo, F.; Dash, J.; Curran, P.J.; Lajas, D.; Kwiatkowska, E. Methodologies and Uncertainties in the Use of the Terrestrial Chlorophyll Index for the Sentinel-3 Mission. Remote Sens. 2012, 4, 1112–1133. [Google Scholar] [CrossRef]
- Houborg, R.; Anderson, M.; Daughtry, C. Utility of an Image-Based Canopy Reflectance Modeling Tool for Remote Estimation of LAI and Leaf Chlorophyll Content at the Field Scale. Remote Sens. Environ. 2009, 113, 259–274. [Google Scholar] [CrossRef]
- De Michele, C.; Vuolo, F.; D’Urso, G.; Marotta, L.; Richter, K. The Irrigation Advisory Program of Campania Region: From Research to Operational Support for the Water Directive in Agriculture. In Proceedings of the 33rd International Symposium on Remote Sensing of Environment, Stresa, Italy, 4–8 May 2009; Volume II, pp. 965–968. [Google Scholar]
- Jacquemoud, S.; Baret, F. PROSPECT: A Model of Leaf Optical Properties Spectra. Remote Sens. Environ. 1990, 34, 75–91. [Google Scholar] [CrossRef]
- Feret, J.-B.; François, C.; Asner, G.P.; Gitelson, A.A.; Martin, R.E.; Bidel, L.P.R.; Ustin, S.L.; le Maire, G.; Jacquemoud, S. PROSPECT-4 and 5: Advances in the Leaf Optical Properties Model Separating Photosynthetic Pigments. Remote Sens. Environ. 2008, 112, 3030–3043. [Google Scholar] [CrossRef]
- Raymond Hunt, E.; Daughtry, C.S.T. Chlorophyll Meter Calibrations for Chlorophyll Content Using Measured and Simulated Leaf Transmittances. Agron. J. 2014, 106, 931–939. [Google Scholar] [CrossRef]
- Padilla, F.M.; Gallardo, M.; Peña-Fleitas, M.T.; De Souza, R.; Thompson, R.B. Proximal Optical Sensors for Nitrogen Management of Vegetable Crops: A Review. Sensors 2018, 18, 2083. [Google Scholar] [CrossRef]
- Fukshansky, L.; Remisowsky, A.M.V.; McClendon, J.; Ritterbusch, A.; Richter, T.; Mohr, H. Absorption Spectra of Leaves Corrected for Scattering and Distributional Error: A Radiative Transfer and Absorption Statistics Treatment. Photochem. Photobiol. 1993, 57, 538–555. [Google Scholar] [CrossRef]
- Manetas, Y.; Grammatikopoulos, G.; Kyparissis, A. The Use of the Portable, Non-Destructive, SPAD-502 (Minolta) Chlorophyll Meter with Leaves of Varying Trichome Density and Anthocyanin Content. J. Plant Physiol. 1998, 153, 513–516. [Google Scholar] [CrossRef]
- Swinehart, D.F. The Beer-Lambert Law. J. Chem. Educ. 1962, 39, 333. [Google Scholar] [CrossRef]
- Parry, C.; Blonquist, J.M.; Bugbee, B. In Situ Measurement of Leaf Chlorophyll Concentration: Analysis of the Optical/Absolute Relationship. Plant Cell Environ. 2014, 37, 2508–2520. [Google Scholar] [CrossRef] [PubMed]
- Nauš, J.; Prokopová, J.; Řebíček, J.; Špundová, M. SPAD Chlorophyll Meter Reading Can Be Pronouncedly Affected by Chloroplast Movement. Photosynth. Res. 2010, 105, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.A.; Williams, O.; Dash, J. Calibration and Characterisation of Four Chlorophyll Meters and Transmittance Spectroscopy for Non-Destructive Estimation of Forest Leaf Chlorophyll Concentration. Agric. For. Meteorol. 2022, 323, 109059. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Gritz, Y.; Merzlyak, M.N. Relationships between Leaf Chlorophyll Content and Spectral Reflectance and Algorithms for Non-Destructive Chlorophyll Assessment in Higher Plant Leaves. J. Plant Physiol. 2003, 160, 271–282. [Google Scholar] [CrossRef]
- Ustin, S.L.; Gitelson, A.A.; Jacquemoud, S.; Schaepman, M.; Asner, G.P.; Gamon, J.A.; Zarco-Tejada, P. Retrieval of Foliar Information about Plant Pigment Systems from High Resolution Spectroscopy. Remote Sens. Environ. 2009, 113, S67–S77. [Google Scholar] [CrossRef]
- Clevers, J.G.P.W.; De Jong, S.M.; Epema, G.F.; Van Der Meer, F.; Bakker, W.H.; Skidmore, A.K.; Addink, E.A. MERIS and the Red-Edge Position. Int. J. Appl. Earth Obs. Geoinf. 2001, 3, 313–320. [Google Scholar] [CrossRef]
- Jiang, C.; Johkan, M.; Hohjo, M.; Tsukagoshi, S.; Maruo, T. A Correlation Analysis on Chlorophyll Content and SPAD Value in Tomato Leaves. HortResearch 2017, 71, 37–42. [Google Scholar] [CrossRef]
- Marenco, R.A.; Antezana-Vera, S.A.; Nascimento, H.C.S. Relationship between Specific Leaf Area, Leaf Thickness, Leaf Water Content and SPAD-502 Readings in Six Amazonian Tree Species. Photosynthetica 2009, 47, 184–190. [Google Scholar] [CrossRef]
- Stuckens, J.; Verstraeten, W.W.; Delalieux, S.; Swennen, R.; Coppin, P. A Dorsiventral Leaf Radiative Transfer Model: Development, Validation and Improved Model Inversion Techniques. Remote Sens. Environ. 2009, 113, 2560–2573. [Google Scholar] [CrossRef]

| Data Sources | Species | n | Minimum (μg cm−2) | Maximum (μg cm−2) | Mean (μg cm−2) | S.D. (μg cm−2) | C.V. (%) |
|---|---|---|---|---|---|---|---|
| Delegido et al. (2011) | Wheat | 20 | 9.40 | 46.79 | 38.17 | 7.38 | 19.33 |
| Sugar beet | 29 | 15.12 | 35.48 | 28.74 | 4.63 | 16.11 | |
| Barley | 20 | 23.10 | 54.88 | 35.46 | 9.05 | 25.53 | |
| Corn | 36 | 15.00 | 43.57 | 32.27 | 8.02 | 24.85 | |
| Vuolo et al. (2012) | Bean | 32 | 1.86 | 43.26 | 25.54 | 11.68 | 46.00 |
| Grass | 23 | 2.38 | 37.62 | 16.69 | 9.25 | 55.43 | |
| Wheat | 30 | 1.40 | 47.44 | 21.29 | 13.98 | 65.66 | |
| Linseed | 28 | 2.79 | 58.14 | 29.09 | 16.77 | 57.64 | |
| Corn | 28 | 3.26 | 34.42 | 16.73 | 10.36 | 61.94 | |
| Oat | 30 | 3.26 | 55.35 | 28.67 | 15.05 | 52.48 | |
| Olive | 26 | 9.30 | 58.14 | 30.25 | 11.95 | 39.49 | |
| Orange | 24 | 4.19 | 28.84 | 14.52 | 6.76 | 46.59 | |
| Vine | 25 | 9.77 | 28.84 | 18.40 | 6.07 | 32.96 | |
| Houborg et al. (2009) | Corn | 48 | 12.20 | 86.34 | 50.82 | 18.92 | 37.23 |
| Parameter | Interpretation | Unit | Input Values |
|---|---|---|---|
| N | Leaf structure parameter | — | 1.0, 2.0 or 3.0 |
| Chlorophyll a + b content | μg cm−2 | 5, 10, 20, 30, 40, 50, 60, 70 or 80 | |
| Carotenoid content | μg cm−2 | 10, 20 or 30 | |
| Brown pigments content | — | 0.0, 0.5 or 1.0 | |
| Equivalent water thickness | cm | 0.02, 0.04, 0.08 or 0.10 | |
| Dry matter content | g cm−2 | 0.005, 0.010 or 0.020 |
| Model Forms | Equations | References |
|---|---|---|
| Linear | Schaper and Chacko (1991) | |
| Polynomial | Monje and Bugbee (1992) | |
| Exponential 1 | Uddling et al. (2007) | |
| Exponential 2 | Markwell et al. (1995) | |
| Homographic | Coste et al. (2010); Cerovic et al., (2012) |
| Data Sources | Species | a | b | R2 | RMSE (μg cm−2) |
|---|---|---|---|---|---|
| All | All | 0.709 | −1.576 | 0.52 | 11.11 |
| Delegido et al. (2011) | Wheat | 0.788 | −1.053 | 0.88 | 2.56 |
| Sugar beet | 0.486 | 8.664 | 0.53 | 3.16 | |
| Barley | 1.174 | −22.248 | 0.79 | 4.13 | |
| Corn | 0.879 | −8.602 | 0.84 | 3.17 | |
| All | 0.840 | −5.783 | 0.78 | 3.83 | |
| Vuolo et al. (2012) | Bean | 0.770 | −6.765 | 0.85 | 4.48 |
| Grass | 0.797 | −7.232 | 0.79 | 4.22 | |
| Wheat | 0.879 | −9.350 | 0.94 | 3.28 | |
| Linseed | 0.776 | −7.157 | 0.90 | 5.20 | |
| Maize | 0.664 | −1.761 | 0.88 | 3.62 | |
| Oat | 0.828 | −1.520 | 0.85 | 5.78 | |
| Olive | 0.875 | −27.494 | 0.87 | 4.36 | |
| Orange | 0.388 | −5.950 | 0.84 | 2.73 | |
| Vine | 0.495 | 4.376 | 0.32 | 5.00 | |
| All | 0.550 | 0.443 | 0.61 | 8.33 | |
| Houborg et al. (2009) | Corn/All | 1.639 | −26.955 | 0.94 | 4.60 |
| Models | Deficiencies |
|---|---|
| Relatively lower accuracy compared to a polynomial model | |
| Unsuitable for limited data | |
| Moderate dependence on dataset and species Slightly lower accuracy than the linear and polynomial models | |
| Slightly lower accuracy than the linear and polynomial models | |
| Strong dependence on dataset and species Significant variability Numerical singularity |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, R.; Yang, P.; Liu, S.; Wang, C.; Liu, J. Evaluation of the Methods for Estimating Leaf Chlorophyll Content with SPAD Chlorophyll Meters. Remote Sens. 2022, 14, 5144. https://doi.org/10.3390/rs14205144
Zhang R, Yang P, Liu S, Wang C, Liu J. Evaluation of the Methods for Estimating Leaf Chlorophyll Content with SPAD Chlorophyll Meters. Remote Sensing. 2022; 14(20):5144. https://doi.org/10.3390/rs14205144
Chicago/Turabian StyleZhang, Runfei, Peiqi Yang, Shouyang Liu, Caihong Wang, and Jing Liu. 2022. "Evaluation of the Methods for Estimating Leaf Chlorophyll Content with SPAD Chlorophyll Meters" Remote Sensing 14, no. 20: 5144. https://doi.org/10.3390/rs14205144
APA StyleZhang, R., Yang, P., Liu, S., Wang, C., & Liu, J. (2022). Evaluation of the Methods for Estimating Leaf Chlorophyll Content with SPAD Chlorophyll Meters. Remote Sensing, 14(20), 5144. https://doi.org/10.3390/rs14205144