Assessing the Behavioural Responses of Small Cetaceans to Unmanned Aerial Vehicles
Abstract
1. Introduction
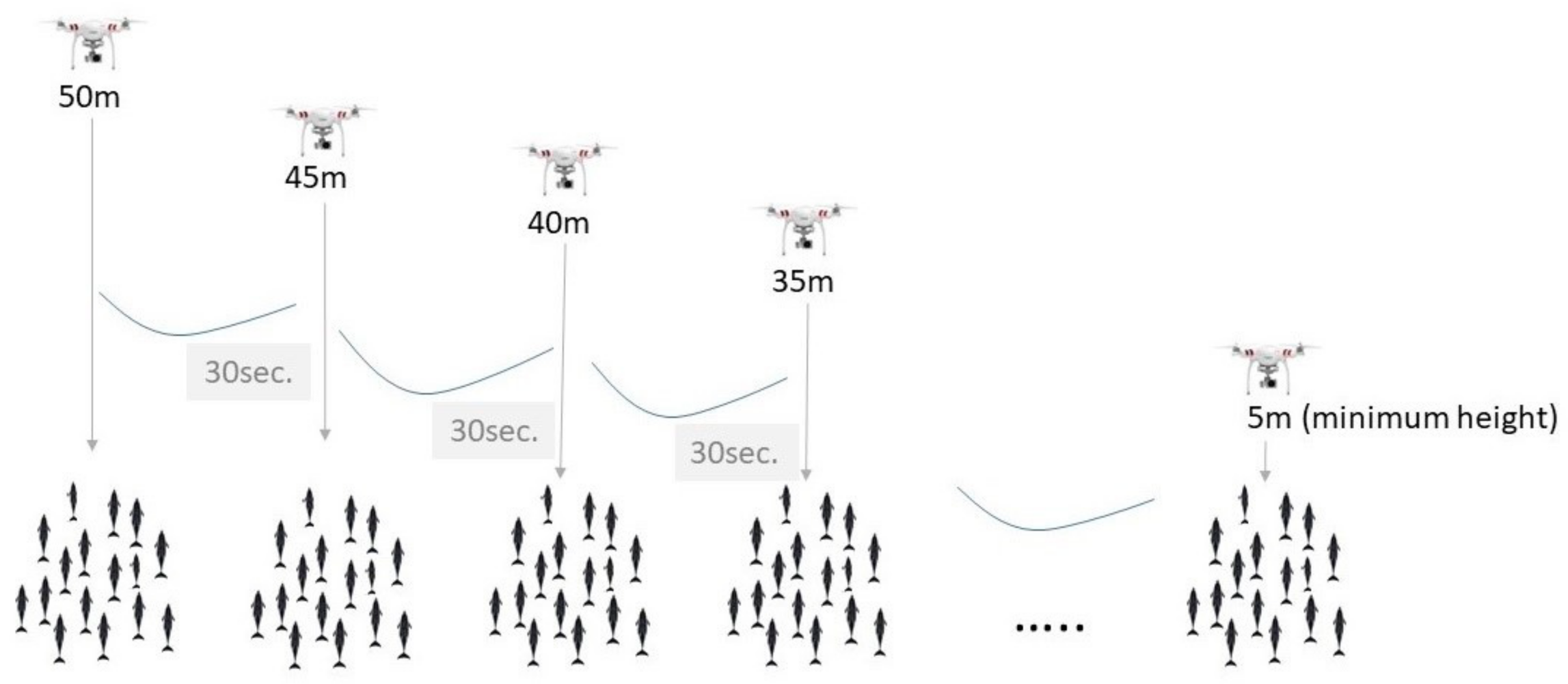
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fiori, L.; Doshi, A.; Martinez, E.; Orams, M.B.; Bollard-Breen, B. The Use of Unmanned Aerial Systems in Marine Mammal Research. Remote Sens. 2017, 9, 543. [Google Scholar] [CrossRef]
- Smith, C.E.; Sykora-Bodie, S.T.; Bloodworth, B.; Pack, S.M.; Spradlin, T.R.; LeBoeuf, N.R. Assessment of known impacts of unmanned aerial systems (UAS) on marine mammals: Data gaps and recommendations for researchers in the United States. J. Unmanned Veh. Syst. 2016, 4, 31–44. [Google Scholar] [CrossRef]
- Christiansen, F.; Rojano-Doñate, L.; Madsen, P.T.; Bejder, L. Noise Levels of Multi-Rotor Unmanned Aerial Vehicles with Implications for Potential Underwater Impacts on Marine Mammals. Front. Mar. Sci. 2016, 3, 277. [Google Scholar] [CrossRef]
- Koski, W.R.; Allen, T.; Ireland, D.; Buck, G.; Smith, P.R.; Macrander, A.M.; Halick, M.A.; Rushing, C.; Sliwa, D.J.; McDonald, T.L. Evaluation of an Unmanned Airborne System for Monitoring Marine Mammals. Aquat. Mamm. 2009, 35, 347–357. [Google Scholar] [CrossRef]
- Christie, K.S.; Gilbert, S.L.; Brown, C.L.; Hatfield, M.; Hanson, L. Unmanned aircraft systems in wildlife research: Current and future applications of a transformative technology. Front. Ecol. Environ. 2016, 14, 241–251. [Google Scholar] [CrossRef]
- Hodgson, A.; Kelly, N.; Peel, D. Unmanned Aerial Vehicles (UAVs) for Surveying Marine Fauna: A Dugong Case Study. PLoS ONE 2013, 8, e79556. [Google Scholar] [CrossRef]
- Weir, J.S.; Fiori, L.; Orbach, D.N.; Piwetz, S.; Protheroe, C.; Würsig, B. Dusky Dolphin (Lagenorhynchus obscurus) Mother–Calf Pairs: An Aerial Perspective. Aquat. Mamm. 2018, 44, 603–607. [Google Scholar] [CrossRef]
- Luksenburg, J.; Parsons, E.C.M. The effects of aircraft on cetaceans: Implications for aerial whalewatching. In Proceedings of the 61st Meeting of the International Whaling Commission, IWC, Madeira, Portugal, 31 May–12 June 2009. [Google Scholar]
- Patenaude, N.J.; Richardson, W.J.; Smultea, M.A.; Koski, W.R.; Miller, G.W.; Wursig, B.; Greene, C.R. Aircraft sound and disturbance to bowhead and beluga whales during spring migration in the Alaskan Beaufort Sea. Mar. Mammal Sci. 2002, 18, 309–335. [Google Scholar] [CrossRef]
- Moreland, E.E.; Cameron, M.F.; Angliss, R.P.; Boveng, P.L. Evaluation of a ship-based unoccupied aircraft system (UAS) for surveys of spotted and ribbon seals in the Bering Sea pack ice. J. Unmanned Veh. Syst. 2015, 3, 114–122. [Google Scholar] [CrossRef]
- Pomeroy, P.; O’Connor, L.; Davies, P. Assessing use of and reaction to unmanned aerial systems in gray and harbor seals during breeding and molt in the UK. J. Unmanned Veh. Syst. 2015, 3, 102–113. [Google Scholar] [CrossRef]
- Durban, J.W.; Fearnbach, H.; Barrett-Lennard, L.G.; Perryman, W.L.; Leroi, D.J. Photogrammetry of killer whales using a small hexacopter launched at sea. J. Unmanned Veh. Syst. 2015, 3, 131–135. [Google Scholar] [CrossRef]
- Fiori, L.; Martinez, E.; Bader, M.K.-F.; Orams, M.B.; Bollard, B. Insights into the use of an unmanned aerial vehicle (UAV) to investigate the behavior of humpback whales (Megaptera novaeangliae) in Vava’u, Kingdom of Tonga. Mar. Mammal Sci. 2019, 36, 209–223. [Google Scholar] [CrossRef]
- Hodgson, A.; Peel, D.; Kelly, N. Unmanned aerial vehicles for surveying marine fauna: Assessing detection probability. Ecol. Appl. 2017, 27, 1253–1267. [Google Scholar] [CrossRef] [PubMed]
- Torres, L.G.; Nieukirk, S.L.; Lemos, L.; Chandler, T.E. Drone Up! Quantifying Whale Behavior From a New Perspective Improves Observational Capacity. Front. Mar. Sci. 2018, 5, 319. [Google Scholar] [CrossRef]
- Nielsen, M.L.K.; Sprogis, K.R.; Bejder, L.; Madsen, P.T.; Christiansen, F. Behavioural development in southern right whale calves. Mar. Ecol. Prog. Ser. 2019, 629, 219–234. [Google Scholar] [CrossRef]
- Acevedo-Whitehouse, K.; Rocha-Gosselin, A.; Gendron, D. A novel non-invasive tool for disease surveillance of free-ranging whales and its relevance to conservation programs. Anim. Conserv. 2010, 13, 217–225. [Google Scholar] [CrossRef]
- Ramos, E.A.; Maloney, B.; Magnasco, M.O.; Reiss, D. Bottlenose Dolphins and Antillean Manatees Respond to Small Multi-Rotor Unmanned Aerial Systems. Front. Mar. Sci. 2018, 5, 316. [Google Scholar] [CrossRef]
- Morimura, N.; Mori, Y. Social responses of travelling finless porpoises to boat traffic risk in Misumi West Port, Ariake Sound, Japan. PLoS ONE 2019. [Google Scholar] [CrossRef]
- Fettermann, T.; Fiori, L.; Bader, M.; Doshi, A.; Breen, D.; Stockin, K.A.; Bollard, B. Behaviour reactions of bottlenose dolphins (Tursiops truncatus) to multirotor Unmanned Aerial Vehicles (UAVs). Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Sardà-Palomera, F.; Bota, G.; Viñolo, C.; Pallarés, O.; Sazatornil, V.; Brotons, L.; Gomáriz, S.; Sardà, F. Fine-scale bird monitoring from light unmanned aircraft systems. IBIS 2012, 154, 177–183. [Google Scholar]
- Nummela, S.; Thewissen, J.G.M.; Bajpai, S.; Hussain, T.; Kumar, K. Sound transmission in archaic and modern whales: Anatomical adaptations for underwater hearing. Anat. Rec. 2007, 290, 716–733. [Google Scholar] [CrossRef] [PubMed]
- Weller, D.W. Predation on Marine Mammals. In Encyclopedia of Marine Mammals, 3rd ed.; Würsig, B.G., Thewissen, J.G.M., Kovacs, K.M., Eds.; Academic Press: London, UK; San Diego, CA, USA, 2018; pp. 772–780. ISBN 978-0-12-804327-1. [Google Scholar]
- Moura, A.E.; Sillero, N.; Rodrigues, A. Common dolphin (Delphinus delphis) habitat preferences using data from two platforms of opportunity. Acta oecologica 2012, 38, 24–32. [Google Scholar] [CrossRef]
- Castro, J.; Couto, A.; Borges, F.O.; Cid, A.; Laborde, M.I.; Pearson, H.C.; Rosa, R. Oceanographic Determinants of the Abundance of Common Dolphins (Delphinus delphis) in the South of Portugal. Oceans 2020, 1, 165–173. [Google Scholar] [CrossRef]
- Ely, C.P. Analysis of Bottlenose (Tursiops truncatus) and Common (Delphinus delphis) Dolphin Behaviour in the Presence of Whale-Watching Vessels in Albufeira, Portugal. Master’s Thesis, University of Southampton, Southampton, UK, 2018. [Google Scholar]
- Castro, J.M.C. Characterization of Cetaceans in the South Coast of Portugal between Lagos and Cape São Vicente. Master’s Thesis, Universidade de Lisboa, Lisboa, Portugal, 2010. [Google Scholar]
- Pearson, H.C. Influences on dusky dolphin (Lagenorhynchus obscurus) fission-fusion dynamics in Admiralty Bay, New Zealand. Behav. Ecol. Sociobiol. 2009, 63, 1437–1446. [Google Scholar] [CrossRef]
- Smolker, R.A.; Richards, A.F.; Connor, R.C.; Pepper, J.W. Sex Differences in Patterns of Association among Indian Ocean Bottlenose Dolphins. Behaviour 1992, 123, 38–69. [Google Scholar] [CrossRef]
- Frid, A.; Dill, L.M. Human-caused disturbance stimuli as a form of predation risk. Conserv. Ecol. 2020, 6, 11. [Google Scholar] [CrossRef]
- Howland, H.C. Optimal strategies for predator avoidance: The relative importance of speed and manoeuvrability. J. Theor. Biol. 1974, 47, 333–350. [Google Scholar] [CrossRef]
- Weihs, D.; Webb, P.W. Optimal avoidance and evasion tactics in predator-prey interactions. J. Theor. Biol. 1984, 106, 189–206. [Google Scholar] [CrossRef]
- Baker, C.S.; Herman, L.M. Behavioural Responses of Summering Humpback Whales to Vessel Traffic: Experimental and Opportunistic Observations; Final Report to the National Park Service; Alaska Regional Office: Anchorage, AK, USA, 1989. [Google Scholar]
- Nowacek, S.M.; Wells, R.S.; Solow, A.R. Short-term effects of boat traffic on bottlenose dolphins, Tursiops truncatus, in Sarasota Bay, Florida. Mar. Mammal Sci. 2001, 17, 673–688. [Google Scholar] [CrossRef]
- Williams, R.; Trites, A.W.; Bain, D.E. Behavioural responses of killer whales (Orcinus orca) to whale-watching boats: Opportunistic observations and experimental approaches. J. Zool. 2002, 256, 255–270. [Google Scholar] [CrossRef]
- Blane, J.; Jaakson, R. The Impact of Ecotourism Boats on the St Lawrence Beluga Whales. Environ. Cons. 1994, 21, 267–269. [Google Scholar] [CrossRef]
- Lusseau, D. Male and female bottlenose dolphins Tursiops spp. have different strategies to avoid interactions with tour boats in Doubtful Sound, New Zealand. Mar. Ecol. Prog. Ser. 2003, 257, 267–274. [Google Scholar] [CrossRef]
- Christiansen, F.; Dujon, A.M.; Sprogis, K.R.; Arnould, J.P.Y.; Bejder, L. Noninvasive unmanned aerial vehicle provides estimates of the energetic cost of reproduction in humpback whales. Ecosphere 2016, 7, e01468. [Google Scholar] [CrossRef]
- Würsig, B.; Lynn, S.K.; Jefferson, T.A.; Mullin, K.D. Behaviour of cetaceans in the northern Gulf of Mexico relative to survey ships and aircraft. Aquat. Mamm. 1998, 24, 41–50. [Google Scholar]
- Ditmer, M.A.; Vincent, J.B.; Werden, L.K.; Tanner, J.C.; Laske, T.G.; Iaizzo, P.A.; Garshelis, D.L.; Fieberg, J.R. Bears Show a Physiological but Limited Behavioral Response to Unmanned Aerial Vehicles. Curr. Biol. 2015, 25, 2278–2283. [Google Scholar] [CrossRef]
- Vermeulen, C.; Lejeune, P.; Lisein, J.; Sawadogo, P.; Bouché, P. Unmanned Aerial Survey of Elephants. PLoS ONE 2013, 8, e54700. [Google Scholar] [CrossRef] [PubMed]
- Koh, L.P.; Wich, S.A. Dawn of drone ecology: Low-cost autonomous aerial vehicles for conservation. Trop. Conserv. Sci. 2012, 5, 121–132. [Google Scholar] [CrossRef]
- Bejder, L.; Samuels, A.; Whitehead, H.; Finn, H.; Allen, S. Impact assessment research: Use and misuse of habituation, sensitisation and tolerance in describing wildlife responses to anthropogenic stimuli. Mar. Ecol. Prog. Ser. 2009, 395, 177–185. [Google Scholar] [CrossRef]
- Hodgson, J.C.; Koh, L.P. Best practice for minimising unmanned aerial vehicle disturbance to wildlife in biological field research. Curr. Biol. 2016, 26, R404–R405. [Google Scholar] [CrossRef]
| Behavioural State | Definition |
|---|---|
| Foraging | Searching for or consuming prey, as indicated by long, deep dives followed by loud forceful exhalations (“chuffs”) and directionless movement; may include coordinated “burst swims” (rapid bursts of speed), “clean” noiseless headfirst re-entry leaps, coordinated clean leaps and tail slaps |
| Resting | Slow directionless movement at speeds of < 3 knots close to the surface with low activity level; often includes slow surfacing and floating near the surface |
| Socialising | Interacting with each other or inanimate objects; usually directionless movement and may include body and pectoral fin rubbing, rolling, belly-up swimming, spyhops, splashing at the surface, chasing, leaping, mating and playing with seaweed |
| Travelling slow | Steady movement in one direction at speeds of < 3 knots |
| Travelling average | Steady movement in one direction at speeds of 3 to 5 knots |
| Travelling fast | Steady movement in one direction at speeds of > 5 knots |
| Species | Travelling Slow (%) | Travelling Average (%) | Travelling Fast (%) | Socialising (%) | Feeding (%) |
|---|---|---|---|---|---|
| Delphinus delphis | 53.3 | 13.3 | 20.0 | 6.7 | 6.7 |
| Tursiops truncatus | 20.0 | 40.0 | 0 | 40.0 | 0 |
| Species | Behavioural Response | N | Cochran’s Q | df | p-Value |
|---|---|---|---|---|---|
| Delphinus delphis | Diving | 15 | 14.27 | 9 | 0.113 |
| Direction change | 15 | 18.00 | 9 | 0.035 | |
| Swimming speed | 15 | 13.09 | 9 | 0.159 | |
| Tursiops truncatus | Diving | 10 | 13.05 | 9 | 0.161 |
| Swimming speed | 10 | 9.00 | 9 | 0.437 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro, J.; Borges, F.O.; Cid, A.; Laborde, M.I.; Rosa, R.; Pearson, H.C. Assessing the Behavioural Responses of Small Cetaceans to Unmanned Aerial Vehicles. Remote Sens. 2021, 13, 156. https://doi.org/10.3390/rs13010156
Castro J, Borges FO, Cid A, Laborde MI, Rosa R, Pearson HC. Assessing the Behavioural Responses of Small Cetaceans to Unmanned Aerial Vehicles. Remote Sensing. 2021; 13(1):156. https://doi.org/10.3390/rs13010156
Chicago/Turabian StyleCastro, Joana, Francisco O. Borges, André Cid, Marina I. Laborde, Rui Rosa, and Heidi C. Pearson. 2021. "Assessing the Behavioural Responses of Small Cetaceans to Unmanned Aerial Vehicles" Remote Sensing 13, no. 1: 156. https://doi.org/10.3390/rs13010156
APA StyleCastro, J., Borges, F. O., Cid, A., Laborde, M. I., Rosa, R., & Pearson, H. C. (2021). Assessing the Behavioural Responses of Small Cetaceans to Unmanned Aerial Vehicles. Remote Sensing, 13(1), 156. https://doi.org/10.3390/rs13010156







