A New Low-Cost Device Based on Thermal Infrared Sensors for Olive Tree Canopy Temperature Measurement and Water Status Monitoring
Abstract
1. Introduction
2. Materials and Methods
2.1. Sensor Performance Evaluation
- Sensor at ambient temperature (20 °C) measuring target at ambient temperature (20 °C).
- Sensor at ambient temperature (20 °C) measuring target at 30 °C.
- Sensor at 30 °C measuring target at ambient temperature (20 °C).
- Sensor at 30 °C measuring target at 30 °C.
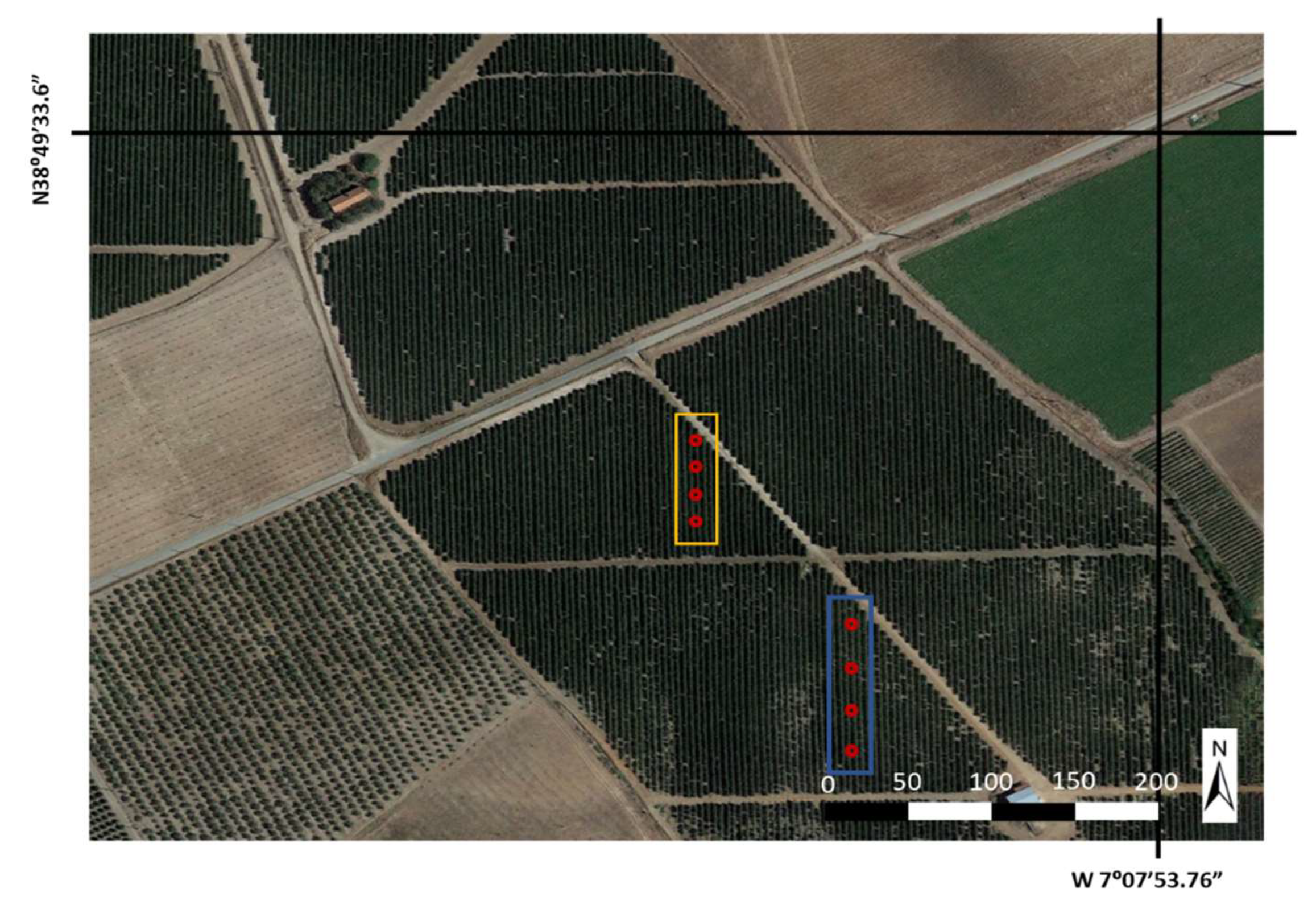
2.2. Experimental Case Study Design
2.3. Physiological Measurement
2.4. Crop Water Stress Index Calculation
2.5. Adaptative Temperature Thresholds
2.6. Statistical Analyses
3. Developed Device for Measuring Canopy Temperature and Water Status Monitoring
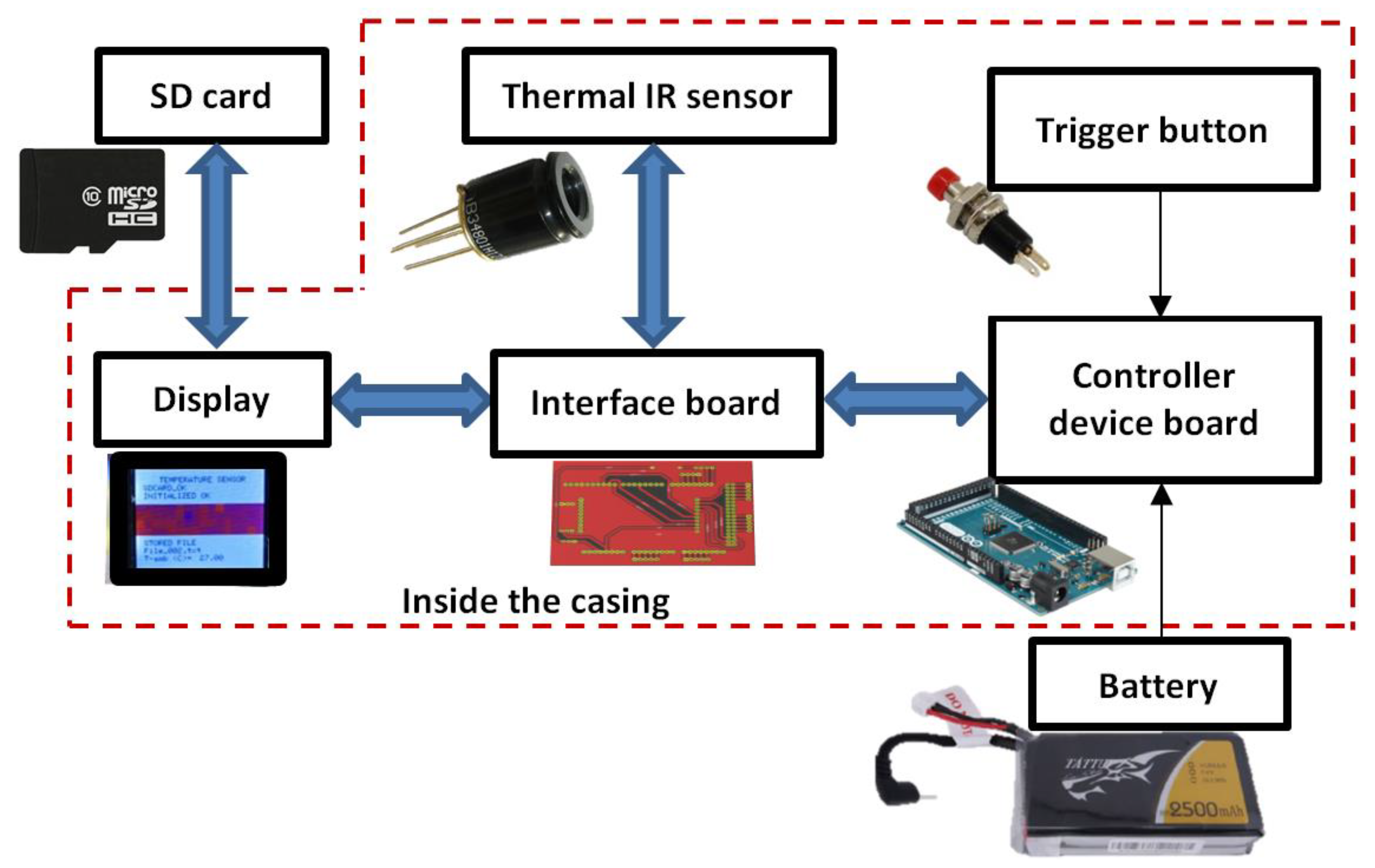
3.1. Thermal Infrared Sensor
3.2. Interface Board
3.3. Display
3.4. Device Casing
3.5. Device Controller Board
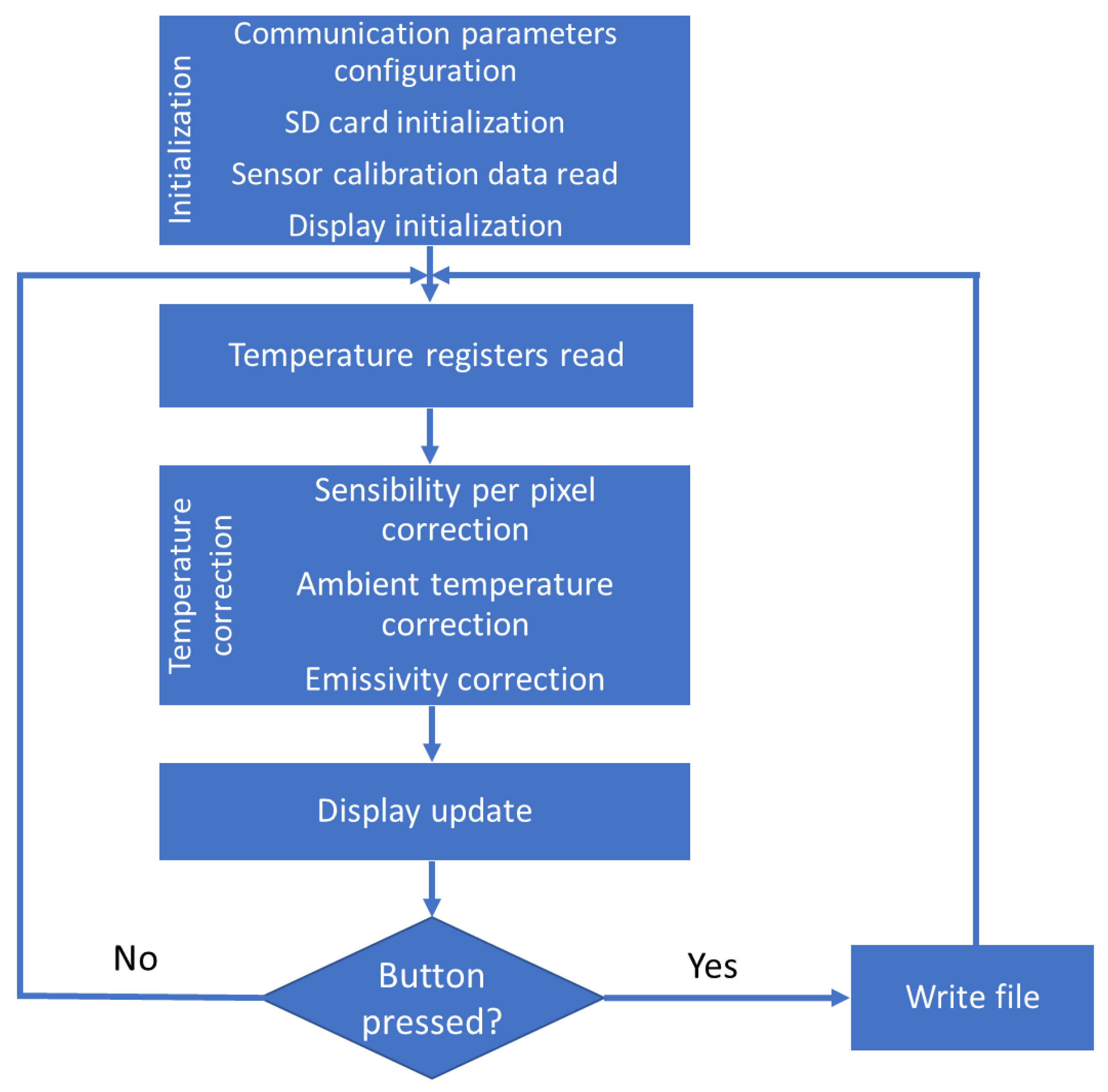
- Initialization. During this process, all the elements of the device are initialized:
- -
- Communication parameter configuration: The baud rates and port assignation for the communication between the different components of the developed device are set up.
- -
- SD card initialization: The SD slot is checked for card presence, after which the files present are examined to determine the next image file name.
- -
- Sensor calibration data read: The registers containing the calibration information (determined during fabrication), including the offset and slope calibration value for every pixel, are read during this phase. The data is stored in the RAM of the device controller board for faster correction calculation.
- -
- Display initialization: The display is initialized, showing the status of the system and the errors during the process (if present).
- Temperature registers read: The raw data of the temperature measured by the IR sensor is stored in the MLX90620 RAM. The value corresponding to every pixel and the temperature of the chip (through PTAT) are read by the device controller using the I2C bus.
- Temperature correction: The raw temperature data must be corrected to obtain canopy temperature values. This process is performed in three different steps:
- -
- Sensibility per pixel correction: The raw temperature data is operated in conjunction with the stored calibration values to standardize the sensor response and to obtain , the temperature corresponding to each pixel with the following equation:where α(i,j) corresponds to the individual pixel sensitivity coefficient calculated (as described in the MLX90620 datasheet) from data stored in EEPROM, Ta is the ambient temperature correction, calculated with Equation (5), and is the parasitic free IR compensated signal, obtained with Equation (6).
- -
- Ambient temperature correction: The temperature of the sensor (ambient temperature) affects the measurements. To correct this, MLX90620 includes a PTAT sensor to adjust the IR temperature values. The equation that calculates the ambient temperature is described below:where KT1, KT2, and VTH are constants (fixed during the in-factory calibration) stored in the MLX90620 EEPROM (are defined by the manufacturer), and PTAT_data refers to the value measured by the in-chip temperature sensor.
- -
- Emissivity correction: The device measures the thermal radiation of a target and estimates its temperature as corresponding to a black body. The emissivity corrects the measurement considering that the object under study is not a perfect black-body emitter. The emissivity was set to 0.98, as this value has been reported to induce errors of less than 1 °C when measuring the canopy of different horticultural crops [40].where is the emissivity and corresponds to the pixel thermal value after the pixel sensibility and offset compensation process, as described in MLX90620 datasheet.
- Display update: After all the corrections have taken place, the display is updated with the measured temperature. The temperatures are shown as a coloured matrix, with colour values reflecting the temperature associated with the corresponding pixel.
- File writing (in the event of the trigger button is pressed): When the trigger button is pressed, the temperature measurement (already corrected) is stored in a file and saved on the micro SD card for further processing. The name of the file is automatically generated.
3.6. Device Components and Cost
4. Results
4.1. Sensor Performance Evaluation
4.2. Plant-Based Variables Reference Values
4.3. Canopy Temperature
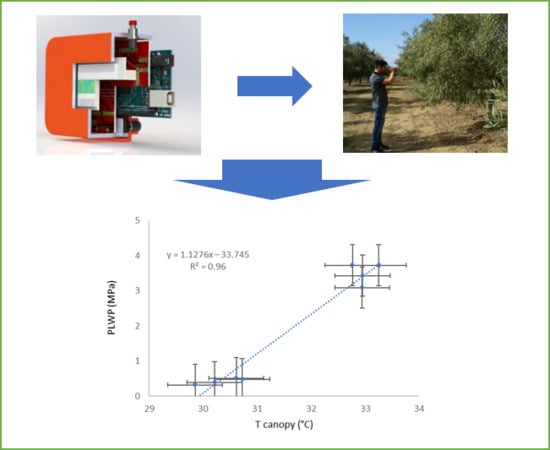
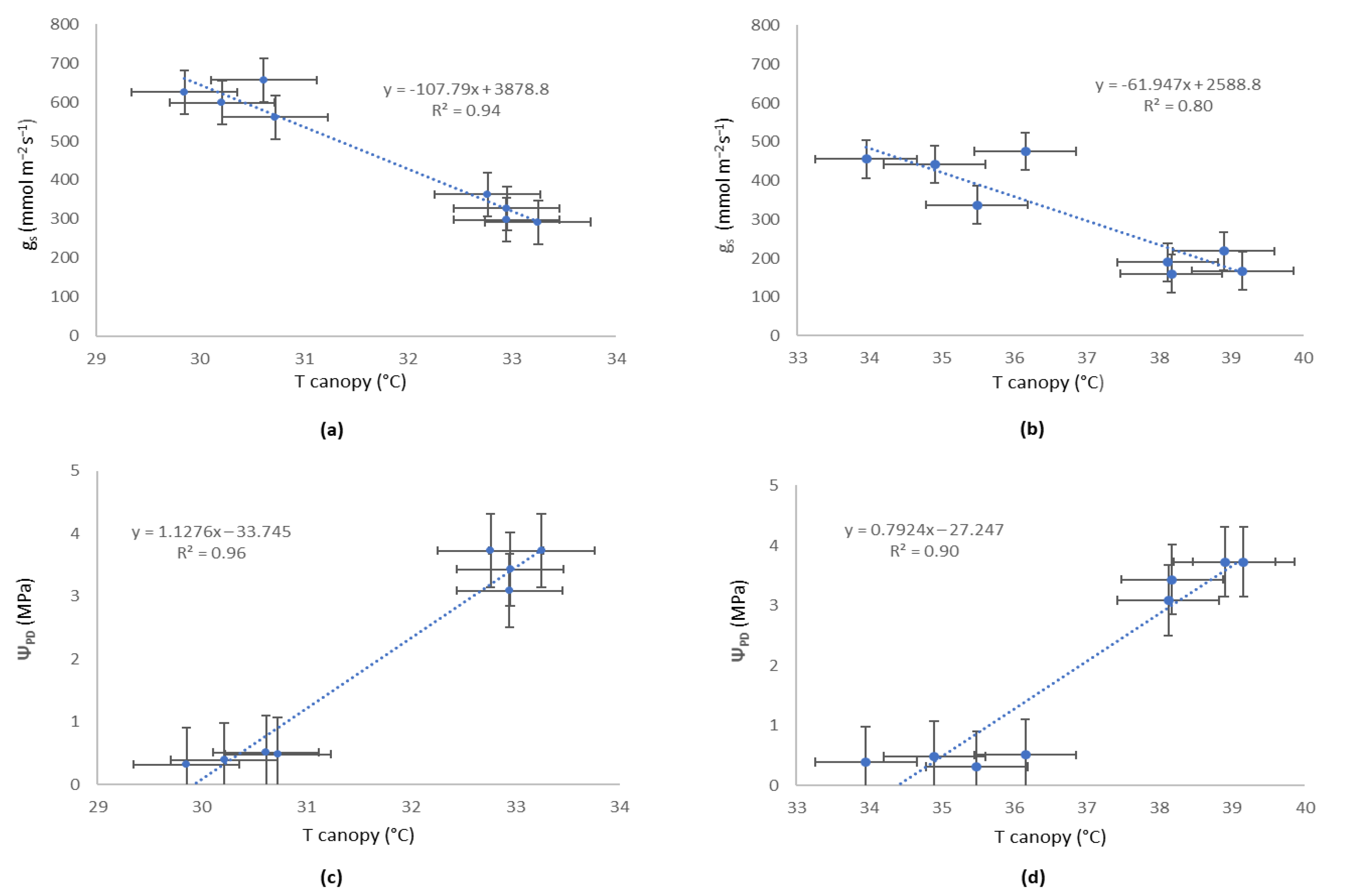
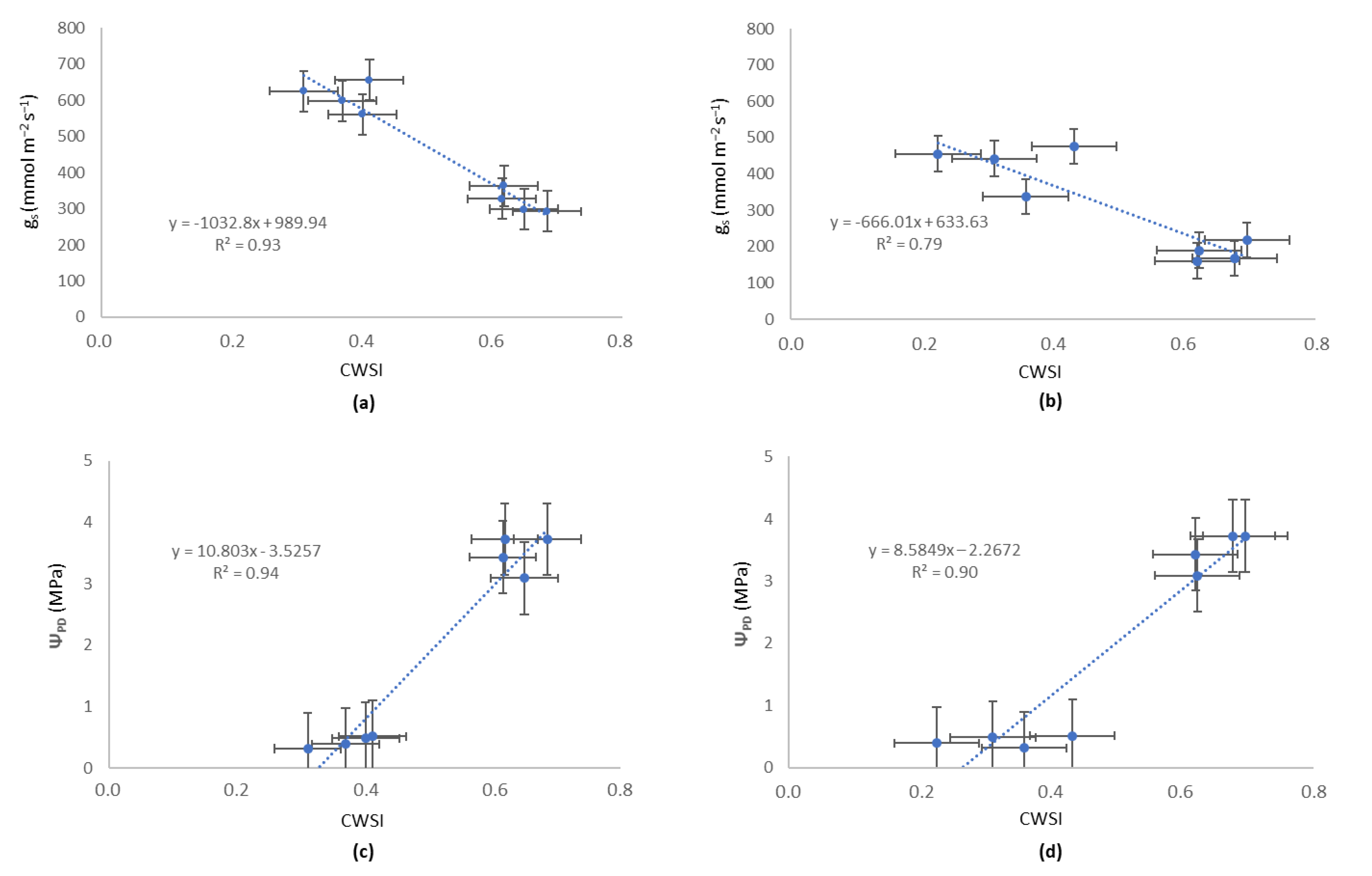
4.4. Relationship Between Canopy Temperature and Plant-Based Variable Reference Values
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | Analog-to-digital converter |
| AM | Ante meridian |
| ANOVA | Analysis of variance |
| ATV | All-terrain vehicle |
| CEa | Electrical conductivity |
| CWSI | Crop water stress index |
| RDC | Regulated deficit irrigated |
| ETc | Crop evapotranspiration |
| EEPROM | Electrically erasable programmable read-only memory |
| EP | Error probability |
| ERDF | European Regional Development Fund |
| FI | Fully irrigated |
| FOV | Field of view |
| I2C | Inter-integrated circuit protocol |
| IC | Integrated circuit |
| IR | Infrared |
| ISO | International Standarization Organization |
| LiPo | Lithium-ion Polymer |
| NETD | Noise equivalent temperature difference |
| PLA | Polylactic acid |
| ΨPD | Predawn leaf water potential |
| Ψ | Leaf water potential |
| PCB | Printed Circuit Board |
| PM | Post meridian |
| PTAT | Proportional to absolute temperature |
| RAM | Random access memory |
| RPAS | Remotely piloted aircraft systems |
| gs | Stomatal conductance |
| SD | Secure digital |
| SHD | Super-high density |
| SPI | Serial peripheral interface |
| Canopy temperature from the thermal images | |
| Temperature of a non-transpiring leaf | |
| Temperature of a fully transpiring leaf | |
| TFT-LCD | Thin film transistor- liquid crystal display |
| Data standard deviation |
References
- Iglesias, A.; Garrote, L. Local and collective actions for adaptation to use less water for agriculture in the mediterranean region. In Water Scarcity and Sustainable Agriculture in Semiarid Environment; Elsevier: Amsterdam, The Netherlands, 2018; pp. 73–84. [Google Scholar]
- Anuario de Estadística. Available online: https://www.mapa.gob.es/en/estadistica/temas/publicaciones/anuario-de-estadistica/default.aspx (accessed on 16 January 2020).
- Connor, D.J.; Gómez-del-Campo, M.; Rousseaux, M.C.; Searles, P.S. Structure, management and productivity of hedgerow olive orchards: A review. Sci. Hortic. 2014, 169, 71–93. [Google Scholar] [CrossRef]
- Fernández, J.E.; Perez-Martin, A.; Torres-Ruiz, J.M.; Cuevas, M.V.; Rodriguez-Dominguez, C.M.; Elsayed-Farag, S.; Morales-Sillero, A.; García, J.M.; Hernandez-Santana, V.; Diaz-Espejo, A. A regulated deficit irrigation strategy for hedgerow olive orchards with high plant density. Plant Soil 2013, 372, 279–295. [Google Scholar] [CrossRef]
- Sepúlveda-Reyes, D.; Ingram, B.; Bardeen, M.; Zúñiga, M.; Ortega-Farías, S.; Poblete-Echeverría, C. Selecting canopy zones and thresholding approaches to assess grapevine water status by using aerial and ground-based thermal imaging. Remote Sens. 2016, 8, 822. [Google Scholar] [CrossRef]
- Fernández, J.-E. Understanding olive adaptation to abiotic stresses as a tool to increase crop performance. Environ. Exp. Bot. 2014, 103, 158–179. [Google Scholar] [CrossRef]
- Poblete-Echeverría, C.; Sepulveda-Reyes, D.; Ortega-Farias, S.; Zuñiga, M.; Fuentes, S. Plant water stress detection based on aerial and terrestrial infrared thermography: A study case from vineyard and olive orchard. Acta Hortic. 2016, 1112, 141–146. [Google Scholar] [CrossRef]
- Scholander, P.F.; Bradstreet, E.D.; Hemmingsen, E.A.; Hammel, H.T. Sap pressure in vascular plants: Negative hydrostatic pressure can be measured in plants. Science 1965, 148, 339–346. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Diago, M.P.; Fernández-Novales, J.; Tardaguila, J. Vineyard water status assessment using on-the-go thermal imaging and machine learning. PLoS ONE 2018, 13, e0192037. [Google Scholar] [CrossRef]
- Brown, H.T.; Escombe, F. Researches on some of the physiological processes of green leaves, with special reference to the interchange of energy between the leaf and its surroundings. Proc. R. Soc. B Biol. Sci. 1905, 76, 29–111. [Google Scholar]
- Jones, H.G.; Stoll, M.; Santos, T.; de Sousa, C.; Chaves, M.M.; Grant, O.M. Use of infrared thermography for monitoring stomatal closure in the field: Application to grapevine. J. Exp. Bot. 2002, 53, 2249–2260. [Google Scholar] [CrossRef]
- Fuchs, M.; Tanner, C.B. Infrared thermometry of vegetation. Agron. J. 1966, 58, 597. [Google Scholar] [CrossRef]
- Idso, S.B.; Jackson, R.D.; Reginato, R.J. Extending the “degree day” concept of plant phenological development to include water stress effects. Ecology 1978, 59, 431–433. [Google Scholar] [CrossRef]
- Jones, H.G. Use of infrared thermometry for estimation of stomatal conductance as a possible aid to irrigation scheduling. Agric. For. Meteorol. 1999, 95, 139–149. [Google Scholar] [CrossRef]
- Idso, S.B.; Jackson, R.D.; Pinter, P.J.; Reginato, R.J.; Hatfield, J.L. Normalizing the stress-degree-day parameter for environmental variability. Agric. Meteorol. 1981, 24, 45–55. [Google Scholar] [CrossRef]
- Diaz-Espejo, A.; Nicolás, E.; Fernández, J.E. Seasonal evolution of diffusional limitations and photosynthetic capacity in olive under drought. Plant Cell Environ. 2007, 30, 922–933. [Google Scholar] [CrossRef]
- Maes, W.H.; Steppe, K. Estimating evapotranspiration and drought stress with ground-based thermal remote sensing in agriculture: A review. J. Exp. Bot. 2012, 63, 4671–4712. [Google Scholar] [CrossRef] [PubMed]
- Bian, J.; Zhang, Z.; Chen, J.; Chen, H.; Cui, C.; Li, X.; Chen, S.; Fu, Q. Simplified evaluation of cotton water stress using high resolution unmanned aerial vehicle thermal imagery. Remote Sens. 2019, 11, 267. [Google Scholar] [CrossRef]
- Pou, A.; Diago, M.P.; Medrano, H.; Baluja, J.; Tardaguila, J. Validation of thermal indices for water status identification in grapevine. Agric. Water Manag. 2014, 134, 60–72. [Google Scholar] [CrossRef]
- Poblete-Echeverría, C.; Espinace, D.; Sepúlveda-Reyes, D.; Zúñiga, M.; Sanchez, M. Analysis of crop water stress index (CWSI) for estimating stem water potential in grapevines: Comparison between natural reference and baseline approaches. Acta Hortic. 2017, 1150, 189–194. [Google Scholar] [CrossRef]
- Usamentiaga, R.; Venegas, P.; Guerediaga, J.; Vega, L.; Molleda, J.; Bulnes, F. Infrared thermography for temperature measurement and non-destructive testing. Sensors 2014, 14, 12305–12348. [Google Scholar] [CrossRef]
- Jones, H.G. Application of thermal imaging and infrared sensing in plant physiology and ecophysiology. In Advances in Botanical Research; Academic Press: Cambridge, MA, USA, 2004; Volume 41, pp. 107–163. ISBN 9780120059416. [Google Scholar]
- Rashid, A.; Stark, J.C.; Tanveer, A.; Mustafa, T. Use of canopy temperature measurements as a screening tool for drought tolerance in spring wheat. J. Agron. Crop Sci. 1999, 182, 231–238. [Google Scholar] [CrossRef]
- Fuentes, S.; de Bei, R.; Pech, J.; Tyerman, S. Computational water stress indices obtained from thermal image analysis of grapevine canopies. Irrig. Sci. 2012, 30, 523–536. [Google Scholar] [CrossRef]
- Petrie, P.R.; Wang, Y.; Liu, S.; Lam, S.; Whitty, M.A.; Skewes, M.A. The accuracy and utility of a low cost thermal camera and smartphone-based system to assess grapevine water status. Biosyst. Eng. 2019, 179, 126–139. [Google Scholar] [CrossRef]
- Bellvert, J.; Zarco-Tejada, P.J.; Marsal, J.; Girona, J.; González-Dugo, V.; Fereres, E. Vineyard irrigation scheduling based on airborne thermal imagery and water potential thresholds. Aust. J. Grape Wine Res. 2016, 22, 307–315. [Google Scholar] [CrossRef]
- Anderson, M.C.; Allen, R.G.; Morse, A.; Kustas, W.P. Use of landsat thermal imagery in monitoring evapotranspiration and managing water resources. Remote Sens. Environ. 2012, 122, 50–65. [Google Scholar] [CrossRef]
- Egea, G.; Padilla-Díaz, C.M.; Martinez-Guanter, J.; Fernández, J.E.; Pérez-Ruiz, M. Assessing a crop water stress index derived from aerial thermal imaging and infrared thermometry in super-high density olive orchards. Agric. Water Manag. 2017, 187, 210–221. [Google Scholar] [CrossRef]
- Crawford, K.E. Remote Sensing of Almond and Walnut Tree Canopy Temperatures Using An Inexpensive Infrared Sensor on A Small Unmanned Aerial Vehicle; University of California Davis: Davis, CA, USA, 2012. [Google Scholar]
- Gago, J.; Douthe, C.; Coopman, R.E.; Gallego, P.P.; Ribas-Carbo, M.; Flexas, J.; Escalona, J.; Medrano, H. UAVs challenge to assess water stress for sustainable agriculture. Agric. Water Manag. 2015, 153, 9–19. [Google Scholar] [CrossRef]
- Sepulcre-Cantó, G.; Zarco-Tejada, P.J.; Jiménez-Muñoz, J.C.; Sobrino, J.A.; de Miguel, E.; Villalobos, F.J. Detection of water stress in an olive orchard with thermal remote sensing imagery. Agric. For. Meteorol. 2006, 136, 31–44. [Google Scholar] [CrossRef]
- Martínez, J.; Egea, G.; Agüera, J.; Pérez-Ruiz, M. A cost-effective canopy temperature measurement system for precision agriculture: A case study on sugar beet. Precis. Agric. 2017, 18, 95–110. [Google Scholar] [CrossRef]
- Parker, J.S.; DeNiro, J.; Ivey, M.L.; Doohan, D. Are small and medium scale produce farms inherent food safety risks? J. Rural Stud. 2016, 44, 250–260. [Google Scholar] [CrossRef]
- Marinetti, S.; Cesaratto, P.G. Emissivity estimation for accurate quantitative thermography. NDT E Int. 2012, 51, 127–134. [Google Scholar] [CrossRef]
- Köppen, W.; Geiger, R. Handbuch der Klimatologie: Das Geographische System der Klimate, 1st ed.; Borntraeger: Berlin, Germany, 1936; ISBN 0936-577X. [Google Scholar]
- Gucci, R.; Lombardini, L.; Tattini, M. Analysis of leaf water relations in leaves of two olive (Olea europaea) cultivars differing in tolerance to salinity. Tree Physiol. 1997, 17, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.G. Plants and Microclimate: A Quantitative Approach to Environmental Plant Physiology, 3rd ed.; Cambridge University Press: New York, NY, USA, 2013. [Google Scholar]
- Park, S.; Ryu, D.; Fuentes, S.; Chung, H.; Hernández-Montes, E.; O’Connell, M. Adaptive estimation of crop water stress in nectarine and peach orchards using high-resolution imagery from an unmanned aerial vehicle (UAV). Remote Sens. 2017, 9, 828. [Google Scholar] [CrossRef]
- García-Tejero, I.F.; Hernández, A.; Padilla-Díaz, C.M.; Diaz-Espejo, A.; Fernández, J.E. Assessing plant water status in a hedgerow olive orchard from thermography at plant level. Agric. Water Manag. 2017, 188, 50–60. [Google Scholar] [CrossRef]
- López, A.; Molina-Aiz, F.D.; Valera, D.L.; Peña, A. Determining the emissivity of the leaves of nine horticultural crops by means of infrared thermography. Sci. Hortic. 2012, 137, 49–58. [Google Scholar] [CrossRef]
- Moriana, A.; Villalobos, F.J.; Fereres, E. Stomatal and photosynthetic responses of olive (Olea europaea L.) leaves to water deficits. Plant Cell Environ. 2002, 25, 395–405. [Google Scholar] [CrossRef]
- Torres-Ruiz, J.M.; Diaz-Espejo, A.; Morales-Sillero, A.; Martín-Palomo, M.J.; Mayr, S.; Beikircher, B.; Fernández, J.E. Shoot hydraulic characteristics, plant water status and stomatal response in olive trees under different soil water conditions. Plant Soil 2013, 373, 77–87. [Google Scholar] [CrossRef]
- Gonzalez-Dugo, V.; Zarco-Tejada, P.J.; Fereres, E. Applicability and limitations of using the crop water stress index as an indicator of water deficits in citrus orchards. Agric. For. Meteorol. 2014, 198–199, 94–104. [Google Scholar] [CrossRef]
- Blonquist, J.M.; Norman, J.M.; Bugbee, B. Automated measurement of canopy stomatal conductance based on infrared temperature. Agric. For. Meteorol. 2009, 149, 2183–2197. [Google Scholar] [CrossRef]
- Ben-Gal, A.; Agam, N.; Alchanatis, V.; Cohen, Y.; Yermiyahu, U.; Zipori, I.; Presnov, E.; Sprintsin, M.; Dag, A. Evaluating water stress in irrigated olives: Correlation of soil water status, tree water status, and thermal imagery. Irrig. Sci. 2009, 27, 367–376. [Google Scholar] [CrossRef]

| Description | Part Number | Manufacturer | Cost (€) |
|---|---|---|---|
| IR sensor | MLX90620 | MelexisTM | 40 |
| Arduino MEGA 2560 | A000067 | ArduinoTM | 35 |
| Arduino display and micro SD card reader | A000096 | ArduinoTM | 35 |
| LiPo Battery 7.4v (2s) | TA-2500-2S1P | TattuTM | 25 |
| Other components (Interface board, button, PLA for device casing, etc.) | - | - | 10 |
| Total | 145 |
| Sensor | Sensor Temperature (°C) | Target Temperature (°C) | Average Measured Temperature (°C) | Measured Temperature Standard Deviation (°C) |
|---|---|---|---|---|
| TestoTM 875-1i | 20 | 20 | 18.94 | 0.14 |
| 20 | 30 | 30.50 | 0.62 | |
| 30 | 20 | 17.27 | 0.75 | |
| 30 | 30 | 33.73 | 1.78 | |
| Low-cost IR sensor device | 20 | 20 | 22.78 | 0.77 |
| 20 | 30 | 30.62 | 0.67 | |
| 30 | 20 | 22.33 | 0.77 | |
| 30 | 30 | 31.78 | 1.09 |
| Treatments | Tree | gs at 10:00 AM (mol m−2 s−1) | gs at 15:00 PM (mol m−2 s−1) | ΨPD (MPa) |
|---|---|---|---|---|
| FI | 1 | 561.13 | 441.83 | 0.49 |
| 2 | 598.77 | 455.17 | 0.39 | |
| 3 | 656.47 | 475.60 | 0.51 | |
| 4 | 625.43 | 337.37 | 0.32 | |
| Average | 610.45 a | 427.49 c | 0.43 a | |
| RDC | 1 | 363.40 | 166.97 | 3.73 |
| 2 | 292.30 | 217.97 | 3.73 | |
| 3 | 327.33 | 160.33 | 3.43 | |
| 4 | 298.47 | 189.83 | 3.09 | |
| Average | 320.38 b | 183.78 d | 3.49 b |
| Treatments | Tree | Sunlit (°C) | Shaded (°C) | ||
|---|---|---|---|---|---|
| 10:00 AM | 15:00 PM | 10:00 AM | 15:00 PM | ||
| FI | 1 | 30.69 | 34.93 | 27.02 | 33.10 |
| 2 | 30.43 | 34.07 | 25.64 | 32.32 | |
| 3 | 30.79 | 36.15 | 26.23 | 32.01 | |
| 4 | 29.91 | 35.41 | 26.58 | 33.11 | |
| Average | 30.45 a | 35.14 c | 26.37 e | 32.63 g | |
| RDC | 1 | 32.58 | 38.62 | 27.48 | 33.23 |
| 2 | 33.15 | 38.81 | 28.04 | 33.59 | |
| 3 | 32.55 | 38.04 | 28.35 | 33.60 | |
| 4 | 32.85 | 38.07 | 29.01 | 33.71 | |
| Average | 32.78 b | 38.38 d | 28.22 f | 33.53 h | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noguera, M.; Millán, B.; Pérez-Paredes, J.J.; Ponce, J.M.; Aquino, A.; Andújar, J.M. A New Low-Cost Device Based on Thermal Infrared Sensors for Olive Tree Canopy Temperature Measurement and Water Status Monitoring. Remote Sens. 2020, 12, 723. https://doi.org/10.3390/rs12040723
Noguera M, Millán B, Pérez-Paredes JJ, Ponce JM, Aquino A, Andújar JM. A New Low-Cost Device Based on Thermal Infrared Sensors for Olive Tree Canopy Temperature Measurement and Water Status Monitoring. Remote Sensing. 2020; 12(4):723. https://doi.org/10.3390/rs12040723
Chicago/Turabian StyleNoguera, Miguel, Borja Millán, Juan José Pérez-Paredes, Juan Manuel Ponce, Arturo Aquino, and José Manuel Andújar. 2020. "A New Low-Cost Device Based on Thermal Infrared Sensors for Olive Tree Canopy Temperature Measurement and Water Status Monitoring" Remote Sensing 12, no. 4: 723. https://doi.org/10.3390/rs12040723
APA StyleNoguera, M., Millán, B., Pérez-Paredes, J. J., Ponce, J. M., Aquino, A., & Andújar, J. M. (2020). A New Low-Cost Device Based on Thermal Infrared Sensors for Olive Tree Canopy Temperature Measurement and Water Status Monitoring. Remote Sensing, 12(4), 723. https://doi.org/10.3390/rs12040723