Lead-Induced Changes in Fluorescence and Spectral Characteristics of Pea Leaves
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
- (1)
- Knop’s solution [1] containing (g·L−1) 0.8 CaNO3·4H2O, 0.2 KNO3, 0.2 KH2PO4, 0.2 MgSO4·7H2O, and 0.028 EDTA-Fe (EthyleneDiamineTetraacetic) enriched with A–Z microelement nutrients (Control);
- (2)
- Knop solution [1] with 5 mM Pb(NO3)2 (lead-treated plants; Knop + Pb);
- (3)
- Knop solution [1] without phosphorus with 5mM Pb(NO3)2 (described below as Knop (-P) +Pb);
- (4)
- distilled water with 5 mM Pb(NO3)2 (described below as H2O + Pb).
2.2. In Vivo Measurements of Chlorophyll a Fluorescence
2.3. Determination of Lead Content
- A is the Pb ion content in mg·g−1 dry weight,
- Cs is the corrected concentration Pb (mg·L−1),
- V is the volume of the sample for analysis (L), and
- m is the dry weight of a sample (g).
2.4. Laboratory Measurements
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mohr, H.; Schopfer, P. Physiology of Movement. In Plant Physiology; Springer: Berlin/Heidelberg, Germany, 1995; pp. 497–538. [Google Scholar] [CrossRef]
- Fryer, M.J.; Andrews, J.R.; Oxborough, K.; Blowers, D.A.; Baker, N.R. Relationship between CO2 Assimilation, Photosynthetic Electron Transport, and Active O2 Metabolism in Leaves of Maize in the Field during Periods of Low Temperature. Plant Physiol. 1998, 116, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, L.; Furbank, R.T.; Chow, W.S. A Simple Alternative Approach to Assessing the Fate of Absorbed Light Energy Using Chlorophyll Fluorescence. Photosynth. Res. 2004, 82, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Romanowska, E. Gas Exchange Functions in Heavy Metal Stressed Plants. In Physiology and Biochemistry of Metal Toxicity and Tolerance in Plants; Springer: Dordrecht, The Netherlands, 2002; pp. 257–285. [Google Scholar] [CrossRef]
- Rashid, A.; Camm, E.L.; Ekramoddoullah, A.K.M. Molecular mechanism of action of Pb2+ and Zn2+ on water oxidizing complex of photosystem II. FEBS Lett. 1994, 350, 296–298. [Google Scholar] [CrossRef]
- Yadav, S.K. Heavy metals toxicity in plants: An overview on the role of glutathione and phytochelatins in heavy metal stress tolerance of plants. S. Afr. J. Bot. 2010, 76, 167–179. [Google Scholar] [CrossRef]
- Schreiber, U. Pulse-Amplitude-Modulation (PAM) Fluorometry and Saturation Pulse Method: An Overview. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2007; pp. 279–319. [Google Scholar] [CrossRef]
- Burzyński, M.; Kłobus, G. Changes of photosynthetic parameters in cucumber leaves under Cu, Cd, and Pb stress. Photosynthetica 2004, 42, 505–510. [Google Scholar] [CrossRef]
- Woźniak, A.; Drzewiecka, K.; Kęsy, J.; Marczak, Ł.; Narożna, D.; Grobela, M.; Motała, R.; Bocianowski, J.; Morkunas, I. The Influence of Lead on Generation of Signalling Molecules and Accumulation of Flavonoids in Pea Seedlings in Response to Pea Aphid Infestation. Molecules 2017, 22, 1404. [Google Scholar] [CrossRef] [PubMed]
- Parys, E.; Romanowska, E.; Siedlecka, M.; Poskuta, J.W. The effect of lead on photosynthesis and respiration in detached leaves and in mesophyll protoplasts of Pisum sativum. Acta Physiol. Plant. 1998, 20, 313. [Google Scholar] [CrossRef]
- Romanowska, E.; Igamberdiev, A.U.; Parys, E.; Gardestrom, P. Stimulation of respiration by Pb2+ in detached leaves and mitochondria of C3 and C4 plants. Physiol. Plant. 2002, 116, 148–154. [Google Scholar] [CrossRef]
- Nas, F.S.; Ali, M. The effect of lead on plants in terms of growing and biochemical parameters: A review. MOJ Ecol. Environ. Sci. 2018, 3, 265–268. [Google Scholar] [CrossRef]
- Romanowska, E.; Wasilewska, W.; Fristedt, R.; Vener, A.V.; Zienkiewicz, M. Phosphorylation of PSII proteins in maize thylakoids in the presence of Pb ions. J. Plant Physiol. 2012, 169, 345–352. [Google Scholar] [CrossRef]
- Romanowska, E.; Wróblewska, B.; Droƶak, A.; Siedlecka, M. High light intensity protects photosynthetic apparatus of pea plants against exposure to lead. Plant Physiol. Biochem. 2006, 44, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Aro, E.-M.; Suorsa, M.; Rokka, A.; Allahverdiyeva, Y.; Paakkarinen, V.; Saleem, A.; Battchikova, N.; Rintamäki, E. Dynamics of photosystem II: A proteomic approach to thylakoid protein complexes. J. Exp. Bot. 2005, 56, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.D.; Brandle, J.R.; Daniel, D.J.; Chu-Der, O.; Schnare, P.D.; Uhlik, D.J. Inhibition of Photosystem II in Isolated Chloroplasts by Lead. Plant Physiol. 1972, 49, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R. Chlorophyll Fluorescence: A Probe of Photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Ruban, A.V. Nonphotochemical Chlorophyll Fluorescence Quenching: Mechanism and Effectiveness in Protecting Plants from Photodamage. Plant Physiol. 2016, 170, 1903–1916. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R.; Oxborough, K. Chlorophyll Fluorescence as a Probe of Photosynthetic Productivity. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2007; pp. 65–82. [Google Scholar] [CrossRef]
- Clijsters, H.; Van Assche, F. Inhibition of photosynthesis by heavy metals. Photosynth. Res. 1985, 7, 31–40. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Loboda, T. Photosystem II of Barley seedlings under cadmium and lead stress. Plant Soil Environ. 2007, 53, 511–516. [Google Scholar] [CrossRef]
- Lazár, D.; Jablonsky, J. On the approaches applied in formulation of a kinetic model of photosystem II: Different approaches lead to different simulations of the chlorophyll a fluorescence transients. J. Theor. Biol. 2009, 257, 260–269. [Google Scholar] [CrossRef]
- Shamshad, S.; Shahid, M.; Rafiq, M.; Khalid, S.; Dumat, C.; Sabir, M.; Murtaza, B.; Farooq, A.B.U.; Shah, N.S. Effect of organic amendments on cadmium stress to pea: A multivariate comparison of germinating vs. young seedlings and younger vs. older leaves. Ecotoxicol. Environ. Saf. 2018, 151, 91–97. [Google Scholar] [CrossRef]
- Bazzaz, F.A.; Rolfe, G.L.; Carlson, R.W. Effect of cadmium on photosynthesis and transpiration of excised leaves of corn and sunflower. Physiol. Plant. 1974, 32, 373–376. [Google Scholar] [CrossRef]
- Zagajewski, B.; Kycko, M.; Tømmervik, H.; Bochenek, Z.; Wojtuń, B.; Bjerke, J.W.; Kłos, A. Feasibility of hyperspectral vegetation indices for the detection of chlorophyll concentration in three high Arctic plants: Salix polaris, Bistorta vivipara, and Dryas octopetala. Acta Soc. Bot. Pol. 2018, 87, 3604. [Google Scholar] [CrossRef]
- Kycko, M.; Zagajewski, B.; Lavender, S.; Romanowska, E.; Zwijacz-Kozica, M. The Impact of Tourist Traffic on the Condition and Cell Structures of Alpine Swards. Remote Sens. 2018, 10, 220. [Google Scholar] [CrossRef]
- Clevers, J.G.P.W. Beyond NDVI: Extraction of Biophysical Variables from Remote Sensing Imagery. In Remote Sensing and Digital Image Processing; Springer: Dordrecht, The Netherlands, 2014; pp. 363–381. [Google Scholar] [CrossRef]
- Farquhar, G.D.; von Caemmerer, S.; Berry, J.A. A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 1980, 149, 78–90. [Google Scholar] [CrossRef]
- Guanter, L.; Zhang, Y.; Jung, M.; Joiner, J.; Voigt, M.; Berry, J.A.; Frankenberg, C.; Huete, A.R.; Zarco-Tejada, P.; Lee, J.-E.; et al. Global and time-resolved monitoring of crop photosynthesis with chlorophyll fluorescence. Proc. Natl. Acad. Sci. USA 2014, 111, E1327–E1333. [Google Scholar] [CrossRef]
- Norton, A.J.; Rayner, P.J.; Koffi, E.N.; Scholze, M. Assimilating solar-induced chlorophyll fluorescence into the terrestrial biosphere model BETHY-SCOPE v1.0: Model description and information content. Geosci. Model Dev. 2018, 11, 1517–1536. [Google Scholar] [CrossRef]
- Kancheva, R.; Georgiev, G. Spectrally-based quantification of plant heavy metal-induced stress. In Remote Sensing for Agriculture, Ecosystems, and Hydrology XIV; SPIE: Bellingham, WA, USA, 2012; Volume 8531, p. 85311D. [Google Scholar] [CrossRef]
- Rajewicz, P.A.; Atherton, J.; Alonso, L.; Porcar-Castell, A. Leaf-level spectral fluorescence measurements: Comparing methodologies for broadleaves and needles. Remote Sens. 2019, 11, 532. [Google Scholar] [CrossRef]
- Rathod, P.H.; Brackhage, C.; Van der Meer, F.D.; Müller, I.; Noomen, M.F.; Rossiter, D.G.; Dudel, G.E. Spectral changes in the leaves of barley plant due to phytoremediation of metals—Results from a pot study. Eur. J. Remote Sens. 2015, 48, 283–302. [Google Scholar] [CrossRef]
- Ren, H.-Y.; Zhuang, D.-F.; Pan, J.-J.; Shi, X.-Z.; Wang, H.-J. Hyper-spectral remote sensing to monitor vegetation stress. J. Soils Sediments 2008, 8, 323–326. [Google Scholar] [CrossRef]
- Dunagan, S.C.; Gilmore, M.S.; Varekamp, J.C. Effects of mercury on visible/nearinfrared reflectance spectra of mustard spinach plants (Brassica rapa P.). Environ. Pollut. 2007, 148, 301–311. [Google Scholar] [CrossRef]
- Su, Y.; Sridhar, B.B.M.; Han, F.X.; Monts, D.L.; Diehl, S.V. Effect of bioaccumulation of Cs and Sr natural isotopes on foliar structure and plant spectral reflectance of Indian mustard (Brassica juncea). Water Air Soil Pollut. 2007, 180, 65–74. [Google Scholar] [CrossRef]
- Bannari, A.; Morin, D.; Bonn, F.; Huete, A. A review of vegetation indices. Remote Sens. Rev. 1995, 13, 95–120. [Google Scholar] [CrossRef]
- Kycko, M.; Zagajewski, B.; Lavender, S.; Dabija, A. In Situ Hyperspectral Remote Sensing for Monitoring of Alpine Trampled and Recultivated Species. Remote Sens. 2019, 11, 1296. [Google Scholar] [CrossRef]
- Zagajewski, B.; Tømmervik, H.; Bjerke, J.; Raczko, E.; Bochenek, Z.; Kłos, A.; Jarocińska, A.; Lavender, S.; Ziółkowski, D. Intraspecific Differences in Spectral Reflectance Curves as Indicators of Reduced Vitality in High-Arctic Plants. Remote Sens. 2017, 9, 1289. [Google Scholar] [CrossRef]
- Kancheva, R.; Borisova, D.; Iliev, I. Chlorophyll fluorescence as a quantitative measure of plant stress. In New Developments and Challenges in Remote Sensing; Bochenek, Z., Ed.; Millpress: Rotterdam, The Netherlands, 2007; ISBN 978-90-5966-053-3. [Google Scholar]
- Holleboom, C.-P.; Walla, P.J. The back and forth of energy transfer between carotenoids and chlorophylls and its role in the regulation of light harvesting. Photosynth. Res. 2014, 119, 215–221. [Google Scholar] [CrossRef]
- Rossini, M.; Fava, F.; Cogliati, S.; Meroni, M.; Marchesi, A.; Panigada, C.; Giardino, C.; Busetto, L.; Migliavacca, M.; Amaducci, S.; et al. Assessing canopy PRI from airborne imagery to map water stress in maize. ISPRS J. Photogramm. Remote Sens. 2013, 86, 168–177. [Google Scholar] [CrossRef]
- Cierniewski, J.; Kazmierowski, C.; Krolewicz, S.; Piekarczyk, J.; Wrobel, M.; Zagajewski, B. Effects of different illumination and observation techniques of cultivated soils on their hyperspectral bidirectional measurements under field and laboratory conditions. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2014, 7, 2525–2530. [Google Scholar] [CrossRef]
- Pinto, F.; Damm, A.; Schickling, A.; Panigada, C.; Cogliati, S.; Müller-Linow, M.; Balvora, A.; Rascher, U. Sun-induced chlorophyll fluorescence from high-resolution imaging spectroscopy data to quantify spatio-temporal patterns of photosynthetic function in crop canopies. Plant. Cell Environ. 2016, 39, 1500–1512. [Google Scholar] [CrossRef]
- Colombo, R.; Celesti, M.; Bianchi, R.; Campbell, P.K.E.; Cogliati, S.; Cook, B.D.; Corp, L.A.; Damm, A.; Domec, J.-C.; Guanter, L.; et al. Variability of sun-induced chlorophyll fluorescence according to stand age-related processes in a managed loblolly pine forest. Glob. Change Biol. 2018, 24, 2980–2996. [Google Scholar] [CrossRef]
- Drusch, M.; Moreno, J.; Del Bello, U.; Franco, R.; Goulas, Y.; Huth, A.; Kraft, S.; Middleton, E.M.; Miglietta, F.; Mohammed, G.; et al. The Fluorescence Explorer Mission Concept—ESA’s Earth Explorer 8. IEEE Trans. Geosci. Remote Sens. 2017, 55, 1273–1284. [Google Scholar] [CrossRef]
- Rascher, U.; Alonso, L.; Burkart, A.; Cilia, C.; Cogliati, S.; Colombo, R.; Damm, A.; Drusch, M.; Guanter, L.; Hanus, J.; et al. Sun-induced fluorescence—A new probe of photosynthesis: First maps from the imaging spectrometer HyPlant. Glob. Change Biol. 2015, 21, 4673–4684. [Google Scholar] [CrossRef]
- Genty, B.; Briantais, J.-M.; Baker, N.R. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta Gen. Subj. 1989, 990, 87–92. [Google Scholar] [CrossRef]
- Arnon, D.I. Cooper enzymes in isolated chloroplasts. Polypenyloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Dawson, T.P.; Curran, P.J. Technical note A new technique for interpolating the reflectance red edge position. Int. J. Remote Sens. 1998, 19, 2133–2139. [Google Scholar] [CrossRef]
- Chappelle, E.W.; Kim, M.S.; McMurtrey, J.E. Ratio analysis of reflectance spectra (RARS): An algorithm for the remote estimation of the concentrations of chlorophyll A, chlorophyll B, and carotenoids in soybean leaves. Remote Sens. Environ. 1992, 39, 239–247. [Google Scholar] [CrossRef]
- Peñuelas, J.; Baret, F.; Filella, I. Semi-Empirical Indices to Assess Carotenoids/Chlorophyll-a Ratio from Leaf Spectral Reflectance. Photosynthetica 1995, 31, 221–230. [Google Scholar]
- Fourty, T.; Baret, F. On spectral estimates of fresh leaf biochemistry. Int. J. Remote Sens. 1998, 19, 1283–1297. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Gitelson, A.A.; Chivkunova, O.B.; Rakitin, V.Y. Non-destructive optical detection of pigment changes during leaf senescence and fruit ripening. Physiol. Plant. 1999, 106, 135–141. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Zur, Y.; Chivkunova, O.B.; Merzlyak, M.N. Assessing Carotenoid Content in Plant Leaves with Reflectance Spectroscopy. Photochem. Photobiol. 2002, 75, 272–281. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N.; Chivkunova, O.B. Optical Properties and Nondestructive Estimation of Anthocyanin Content in Plant Leaves. Photochem. Photobiol. 2001, 74, 38. [Google Scholar] [CrossRef]
- Fuentes, D.A.; Gamon, J.A.; Qiu, H.; Sims, D.A.; Roberts, D.A. Mapping Canadian boreal forest vegetation using pigment and water absorption features derived from the AVIRIS sensor. J. Geophys. Res. Atmos. 2001, 106, 33565–33577. [Google Scholar] [CrossRef]
- Gao, B. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Babar, M.A.; Reynolds, M.P.; van Ginkel, M.; Klatt, A.R.; Raun, W.R.; Stone, M.L. Spectral Reflectance Indices as a Potential Indirect Selection Criteria for Wheat Yield under Irrigation. Crop Sci. 2006, 46, 578. [Google Scholar] [CrossRef]
- Galvão, L.S.; Epiphanio, J.C.N.; Breunig, F.M.; Formaggio, A.R. Biophysical and Biochemical Characterization and Plant Species Studies; Thenkabail, P.S., Lyon, J.G., Huete, A., Eds.; CRC Press: Boca Raton, FL, USA, 2018; ISBN 9780429431180. [Google Scholar] [CrossRef]
- Evans, E.H.; Brown, R.G. New trends in photobiology. J. Photochem. Photobiol. B Biol. 1994, 22, 95–104. [Google Scholar] [CrossRef]
- Fisher, R.A. Statistical Methods for Research Workers; Landmark Writings in Western Mathematics: Case Studies, 11th ed.; Grattan-Guinness, I., Ed.; Elsevier: Amsterdam, The Netherlands, 1925; pp. 1640–1940. [Google Scholar]
- StatSoft. StatSoft Manual, Internetowy Podręcznik Statystyki; Statistica: Krakow, Poland, 2012. [Google Scholar]
- Sharma, P.; Dubey, R.S. Lead Toxicity in Plants. Braz. J. Plant Physiol. 2005, 17, 1–19. [Google Scholar] [CrossRef]
- Myśliwa-Kurdziel, B.; Prasad, M.N.V.; Strzałtka, K. Photosynthesis in heavy metal stressed plants. In Heavy Metal Stress in Plants; Springer: Berlin/Heidelberg, Germany, 2004; pp. 146–181. [Google Scholar] [CrossRef]
- Bavi, K.; Kholdebarin, B.; Moradshahi, A. Effect of cadmium on growth, protein content and peroxidase activity in pea plants. Pak. J. Bot. 2011, 43, 1467–1470. [Google Scholar]
- Romanowska, E.; Wróblewska, B.; Drożak, A.; Zienkiewicz, M.; Siedlecka, M. Effect of Pb ions on superoxide dismutase and catalase activities in leaves of pea plants grown in high and low irradiance. Biol. Plant. 2008, 52, 80–86. [Google Scholar] [CrossRef]
- Sengar, R.S.; Gautam, M.; Sengar, R.S.; Sengar, R.S.; Garg, S.K.; Sengar, K.; Chaudhary, R. Lead Stress Effects on Physiobiochemical Activities of Higher Plants. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2008; pp. 73–93. ISBN 9780387784434. [Google Scholar] [CrossRef]
- Pourrut, B.; Shahid, M.; Dumat, C.; Winterton, P.; Pinelli, E. Lead Uptake, Toxicity, and Detoxification in Plants. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2011; pp. 113–136. ISBN 9781441998590. [Google Scholar] [CrossRef]
- Łukaszek, M.; Poskuta, J.W. Development of photosynthetic apparatus and respiration in pea seedlings during as influenced by toxic concentration of lead. Acta Physiol. Plant. 1998, 20, 35. [Google Scholar] [CrossRef]
- Joshi, M.K.; Mohanty, P. Chlorophyll a Fluorescence as a Probe of Heavy Metal Ion Toxicity in Plants. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2007; pp. 637–661. [Google Scholar] [CrossRef]
- Moustakas, M.; Lanaras, T.; Symeonidis, L.; Karataglis, S. Growth and some photosynthesis characteristics of field grown Avena sativa under copper and lead stress. Photosynthetica 1994, 30, 389–396. [Google Scholar]
- Ernst, W.H.O.; Verkleij, J.A.C.; Schat, H. Metal tolerance in plants. Acta Bot. Neerl. 1992, 41, 229–248. [Google Scholar] [CrossRef]
- Gallego, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznik, M.S.; Groppa, M.D.; Benavides, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Bot. 2012, 83, 33–46. [Google Scholar] [CrossRef]
- Kłos, A.; Ziembik, Z.; Rajfur, M.; Dołhańczuk-Śródka, A.; Bochenek, Z.; Bjerke, J.W.; Tømmervik, H.; Zagajewski, B.; Ziółkowski, D.; Jerz, D.; et al. Using moss and lichens in biomonitoring of heavy-metal contamination of forest areas in southern and north-eastern Poland. Sci. Total Environ. 2018, 627, 438–449. [Google Scholar] [CrossRef]
- Usman, K.; Al-Ghouti, M.A.; Abu-Dieyeh, M.H. The assessment of cadmium, chromium, copper, and nickel tolerance and bioaccumulation by shrub plant Tetraena qataranse. Sci. Rep. 2019, 9, 5658. [Google Scholar] [CrossRef]
- Cannata, M.G.; Carvalho, R.; Bertoli, A.C.; Augusto, A.S.; Bastos, A.R.R.; Carvalho, J.G.; Freitas, M.P. Effects of Cadmium and Lead on Plant Growth and Content of Heavy Metals in Arugula Cultivated in Nutritive Solution. Commun. Soil Sci. Plant Anal. 2013, 44, 952–961. [Google Scholar] [CrossRef]
- Prasad, M.N.V.; Strzałka, K. Impact of heavy metals on photosynthesis. In Heavy Metal Stress in Plants; Springer: Berlin/Heidelberg, Germany, 1999; pp. 117–138. [Google Scholar] [CrossRef]
- Külheim, C.; Ågren, J.; Jansson, S. Rapid regulation of light harvesting and plant fitness in the field. Science 2002, 297, 91–93. [Google Scholar] [CrossRef]
- Pandey, N.; Pathak, G.C. Nickel alters antioxidative defense and water status in green gram. Ind. J. Plant Physiol. 2006, 11, 113–118. [Google Scholar]
- Zagajewski, B. Remote Sensing Measurements of Lead Concentration in Plants. Misc. Geogr. 2000, 9, 267–282. [Google Scholar] [CrossRef]
- Chen, H. The Possibility of Assessing Heavy Metal Concentrations in Reed along le an River (China) Using Hyperspectral Data; International Institute for Geo-information Science and Earth Observation ITC: Enschede, The Netherlands, 2008; pp. 1–54. [Google Scholar]
- Götze, C.; Jung, A.; Henrich, V.; Merbach, I.; Gläßer, C. Spectrometric analyses in comparison to the physiological condition of heavy metal stressed floodplain vegetation in a standardised experiment. In Proceedings of the 6th EARSeL Workshop on Imaging Spectroscopy SIG, Tel Aviv, Israel, 16–19 March 2009. [Google Scholar]
- Hawari, A.H.; Mulligan, C.N. Effect of the presence of lead on the biosorption of copper, cadmium and nickel by anaerobic biomass. Process Biochem. 2007, 42, 1546–1552. [Google Scholar] [CrossRef]
- Lamhamdi, M.; El Galiou, O.; Bakrim, A.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Aarab, A.; Lafont, R. Effect of lead stress on mineral content and growth of wheat (Triticum aestivum) and spinach (Spinacia oleracea) seedlings. Saudi J. Biol. Sci. 2013, 20, 29–36. [Google Scholar] [CrossRef]
- Haider, S.; Kanwal, S.; Uddin, F.; Azmat, R. Phytotoxicity of Pb: II. Changes in Chlorophyll Absorption Spectrum due to Toxic Metal Pb Stress on Phaseolus mungo and Lens culinaris. Pak. J. Biol. Sci. 2006, 9, 2062–2068. [Google Scholar] [CrossRef]
- Akinci, I.E.; Akinci, S.; Yilmaz, K. Response of tomato (Solanum lycopersicum L.) to lead toxicity: Growth, element uptake, chlorophyll and water content. Afr. J. Agric. Res. 2010, 5, 416–423. [Google Scholar]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Estimation of chlorophyll fluorescence under natural illumination from hyperspectral data. Int. J. Appl. Earth Obs. Geoinf. 2001, 3, 321–327. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Vegetation Stress Detection through Chlorophyll + Estimation and Fluorescence Effects on Hyperspectral Imagery. J. Environ. Qual. 2002, 31, 1433. [Google Scholar] [CrossRef]
- Zagajewski, B.; Lechnio, J.; Sobczak, M. Wykorzystanie Teledetekcji Hiperspektralnej w Analizie Roślinności Zanieczyszczonej Metalami Ciężkimi; Teledetekcja Środowiska: Warszawa, Poland, 2007; Volume 37, pp. 82–100. [Google Scholar]
- Grossmann, K.; Frankenberg, C.; Magney, T.S.; Hurlock, S.C.; Seibt, U.; Stutz, J. PhotoSpec: A new instrument to measure spatially distributed red and far-red solar-induced chlorophyll fluorescence. Remote Sens. Environ. 2018, 216, 311–327. [Google Scholar] [CrossRef]




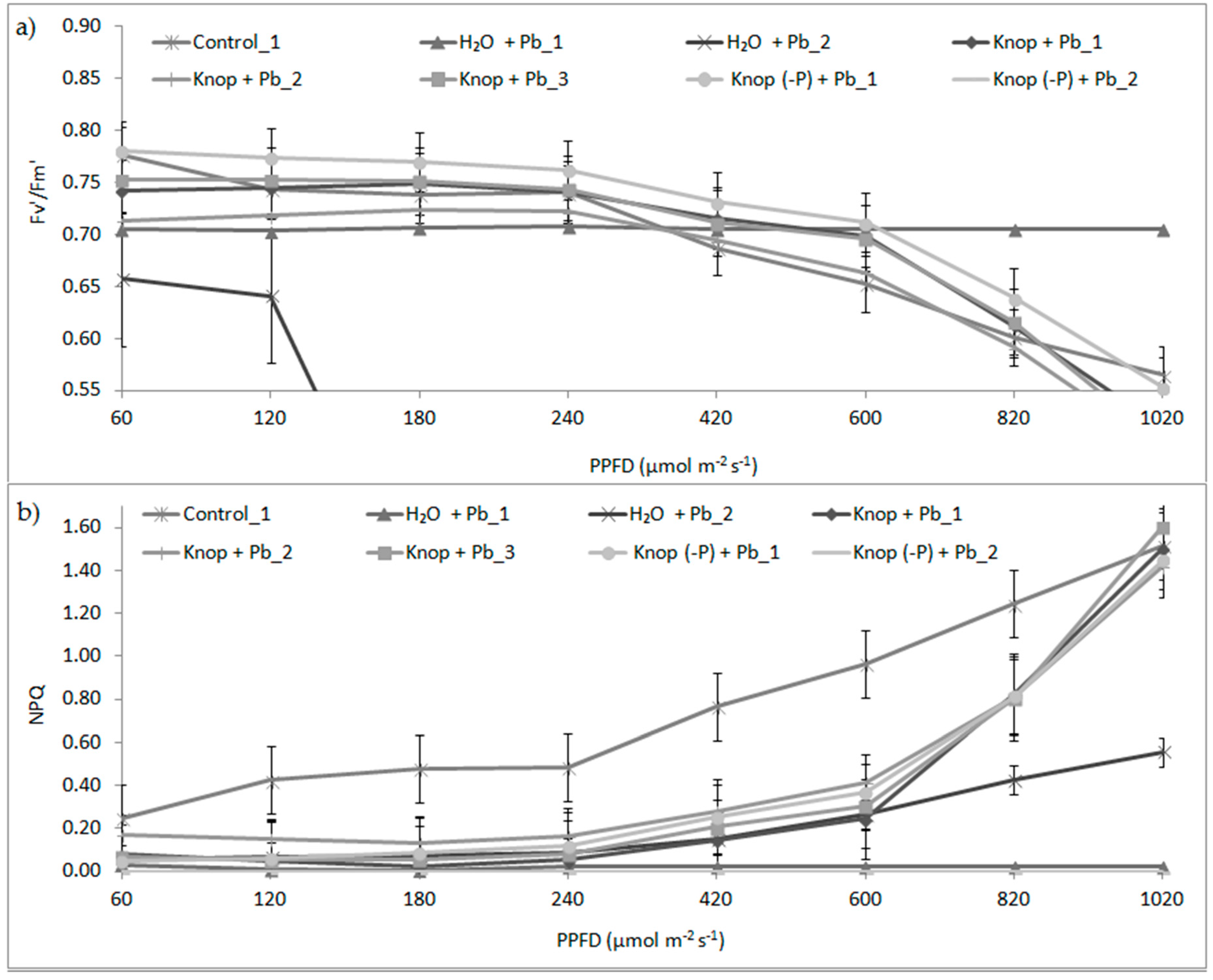

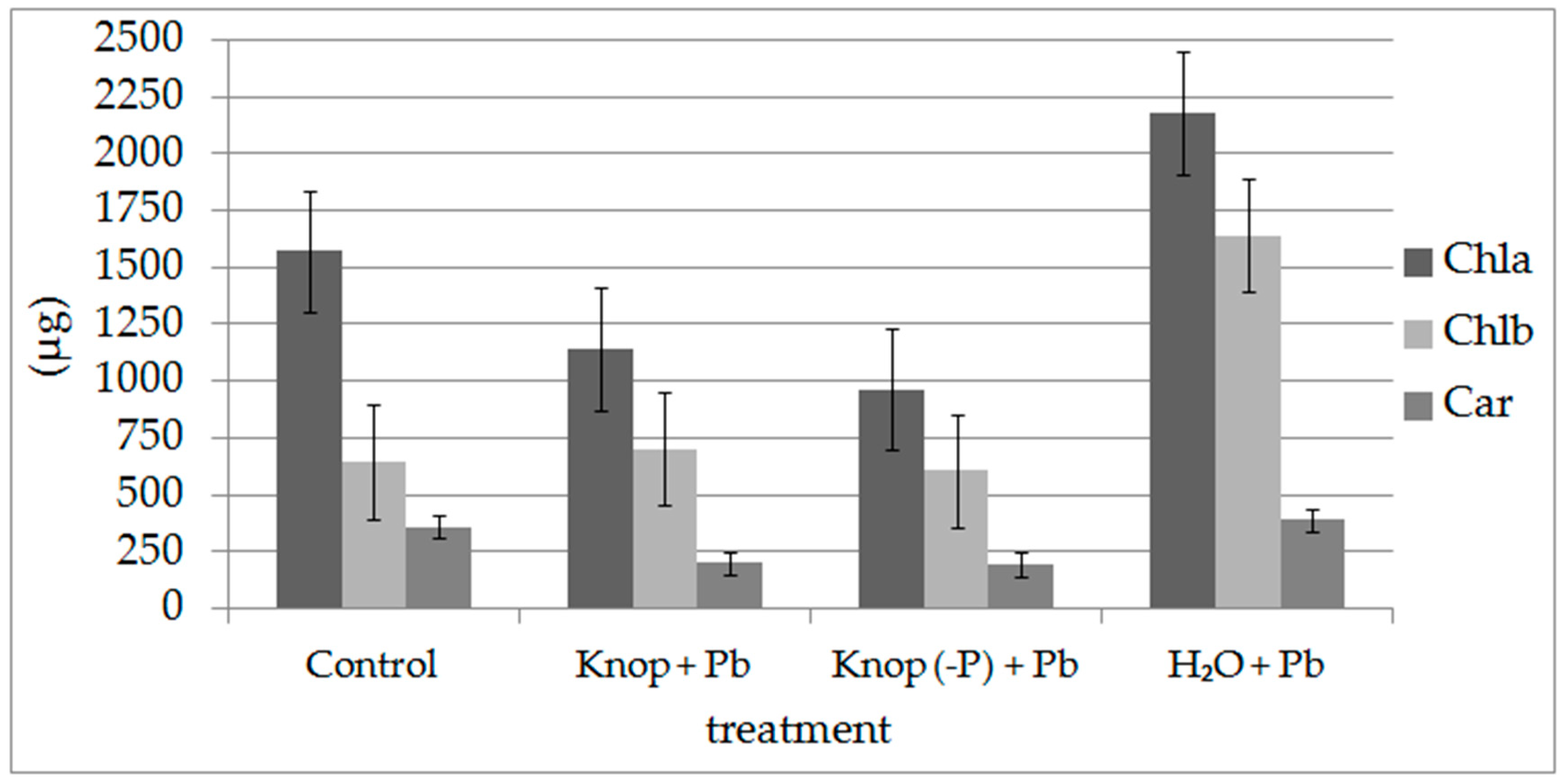
| Control | Knop + 5 mM Pb(NO3)2 | Knop (-P) + 5 mM Pb(NO3)2 | H2O + 5 mM Pb(NO3)2 | |
|---|---|---|---|---|
| Number of spectrometric measurements | 60 | 40 | 40 | 40 |
| Plant Characteristic | Index | H2O + Pb | Knop (-P) + Pb | Knop + Pb | Control |
|---|---|---|---|---|---|
| Chlorophyll content | mNDVI 705 | 0.41 ± 0.11 * | 0.46 ± 0.07 * | 0.57 ± 0.04 | 0.57 ± 0.09 |
| REPI1 | 715.15 ± 1.48 * | 715.91 ± 1.79 * | 719.94 ± 1.9 | 719.23 ± 1.94 | |
| RARSc | 5.52 ± 0.9 | 8.79 ± 0.46 | 8.38 ± 0.89 | 7.83 ± 0.45 | |
| RARSa | 5.98 ± 0.91 * | 9.49 ± 0.78 | 9.99 ± 0.39 | 9.18 ± 0.42 | |
| RARSb | 5.31 ± 0.4 * | 7.91 ± 0.45 | 8.62 ± 0.1 | 8.21 ± 0.54 | |
| Amount of light used in photosynthesis | SIPI | 1.06 ± 0.07 * | 1.025 ± 0.02 * | 0.99 ± 0.005 | 0.99 ± 0.01 |
| Amount of nitrogen | NDNI | 0.14 ± 0.05 * | 0.22 ± 0.02 | 0.22 ± 0.02 | 0.21 ± 0.04 |
| Amount of carbon | NDLI | 0.05 ± 0.01 | 0.06 ± 0.01 * | 0.06 ± 0.01 * | 0.05 ± 0.01 |
| PSRI | 0.001 ± 0.03 | 0.0004 ± 0.01 * | −0.0145 ± 0.01 | −0.0133 ± 0.01 | |
| Amount of carotenoids and other pigments | CRI 1 | 2.94 ± 0.07 * | 6.065 ± 0.05 * | 5.46 ± 0.26 | 5.54 ± 0.27 |
| ARI 1 | 1.02 ± 0.06 * | 0.44 ± 0.06 * | −0.66 ± 0.04 | -0.78 ± 0.02 | |
| RGR | 1.32 ± 0.05 * | 1.05 ± 0.08 | 0.98 ± 0.04 | 1.07 ± 0.07 | |
| Water content in the plant | NDWI | −0.003 ± 0.02 * | 0.04 ± 0.03 * | 0.066 ± 0.01 * | 0.06 ± 0.01 |
| WI | 0.99 ± 0.02 * | 1.03 ± 0.02 | 1.05 ± 0.01 * | 1.04 ± 0.01 | |
| NWI-2 | 0.015 ± 0.01 * | −0.006 ± 0.02 * | −0.027 ± 0.003 * | −0.02 ± 0.004 | |
| DSWI | 1.27 ± 0.22 * | 1.66 ± 0.22 | 1.91 ± 0.1 * | 1.74 ± 0.1 |
| Treatment | Root | Stalk | Leaf |
|---|---|---|---|
| Control | 6 | 133 ± 10 | 0 |
| Knop + 5 mM Pb(NO3)2 | 15,260 ± 330 | 6430 ± 142 | 68 ± 8 |
| H2O + 5 mM Pb(NO3)2 | 13,260 ± 245 | ||
| Knop -P (without phosphate) + 5 mM Pb(NO3)2 | 1730 ± 75 |
| Treatment | Stalk | Root | ||
|---|---|---|---|---|
| Control | 14.58 ± 1.76 | 100% | 18.35 ± 1.18 | 100% |
| Knop + 5 mM Pb(NO3)2 | 10.08 ± 1.15 | 70% | 13.05 ± 1.65 | 71% |
| Knop -P (without phosphate) + 5 mM Pb(NO3)2 | 8.98 ± 0.94 | 61% | 12.45 ± 1.42 | 68% |
| H2O + 5 mM Pb(NO3)2 | 9.08 ± 1.94 | 62% | 11.53 ± 1.50 | 63% |
| Treatment | Stalk | Root | Leaf | |||
|---|---|---|---|---|---|---|
| Control | 0.54 ± 0.17 | 100% | 1.19 ± 0.43 | 100% | 1.27 ± 0.31 | 100% |
| Knop + 5 mM Pb(NO3)2 | 0.36 ± 0.05 | 67% | 0.63 ± 0.10 | 53% | 0.82 ± 0.10 | 64% |
| Knop -P (without phosphate) + 5 mM Pb(NO3)2 | 0.24 ± 0.05 | 44% | 0.50 ± 0.07 | 42% | 0.69 ± 0.09 | 54% |
| H2O + 5 mM Pb(NO3)2 | 0.14 ± 0.03 | 26% | 0.31 ± 0.16 | 26% | 0.29 ± 0.08 | 23% |
| Treatment | Chl a/b | Chl/Car | RARSa/RARSb | (RARSa + RARSb)/RARSc |
|---|---|---|---|---|
| Control | 2.7 | 5.9 | 1.12 | 2.22 |
| Knop + 5 mM Pb(NO3)2 | 1.8 | 7.4 | 1.16 | 2.22 |
| Knop -P (without phosphate) + 5 mM Pb(NO3)2 | 1.6 | 6.7 | 1.20 | 1.98 |
| H2O + 5 mM Pb(NO3)2 | 1.3 | 10.0 | 1.13 | 2.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kycko, M.; Romanowska, E.; Zagajewski, B. Lead-Induced Changes in Fluorescence and Spectral Characteristics of Pea Leaves. Remote Sens. 2019, 11, 1885. https://doi.org/10.3390/rs11161885
Kycko M, Romanowska E, Zagajewski B. Lead-Induced Changes in Fluorescence and Spectral Characteristics of Pea Leaves. Remote Sensing. 2019; 11(16):1885. https://doi.org/10.3390/rs11161885
Chicago/Turabian StyleKycko, Marlena, Elżbieta Romanowska, and Bogdan Zagajewski. 2019. "Lead-Induced Changes in Fluorescence and Spectral Characteristics of Pea Leaves" Remote Sensing 11, no. 16: 1885. https://doi.org/10.3390/rs11161885
APA StyleKycko, M., Romanowska, E., & Zagajewski, B. (2019). Lead-Induced Changes in Fluorescence and Spectral Characteristics of Pea Leaves. Remote Sensing, 11(16), 1885. https://doi.org/10.3390/rs11161885








