Experiences of Mass Pig Carcass Disposal Related to Groundwater Quality Monitoring in Taiwan
Abstract
:1. Background of the Pig Industry in Taiwan
2. Foot and Mouth Disease Outbreak in 1997
2.1. Process of Outbreak
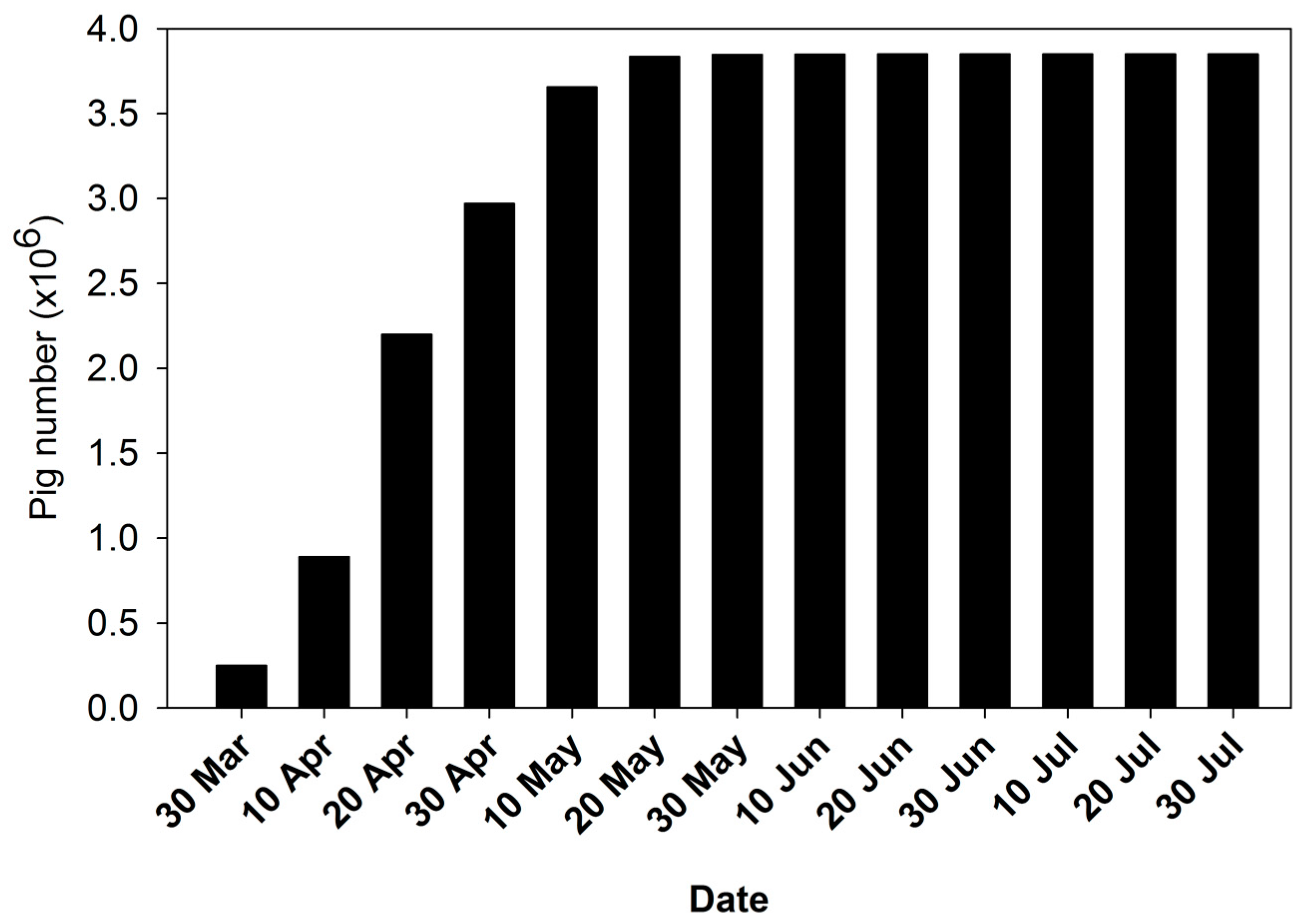
2.2. Depopulation and Disposal of Culled Pigs
3. Groundwater Quality Monitoring
3.1. Potential Effect of Land Burial of Livestock Carcass on Water Quality
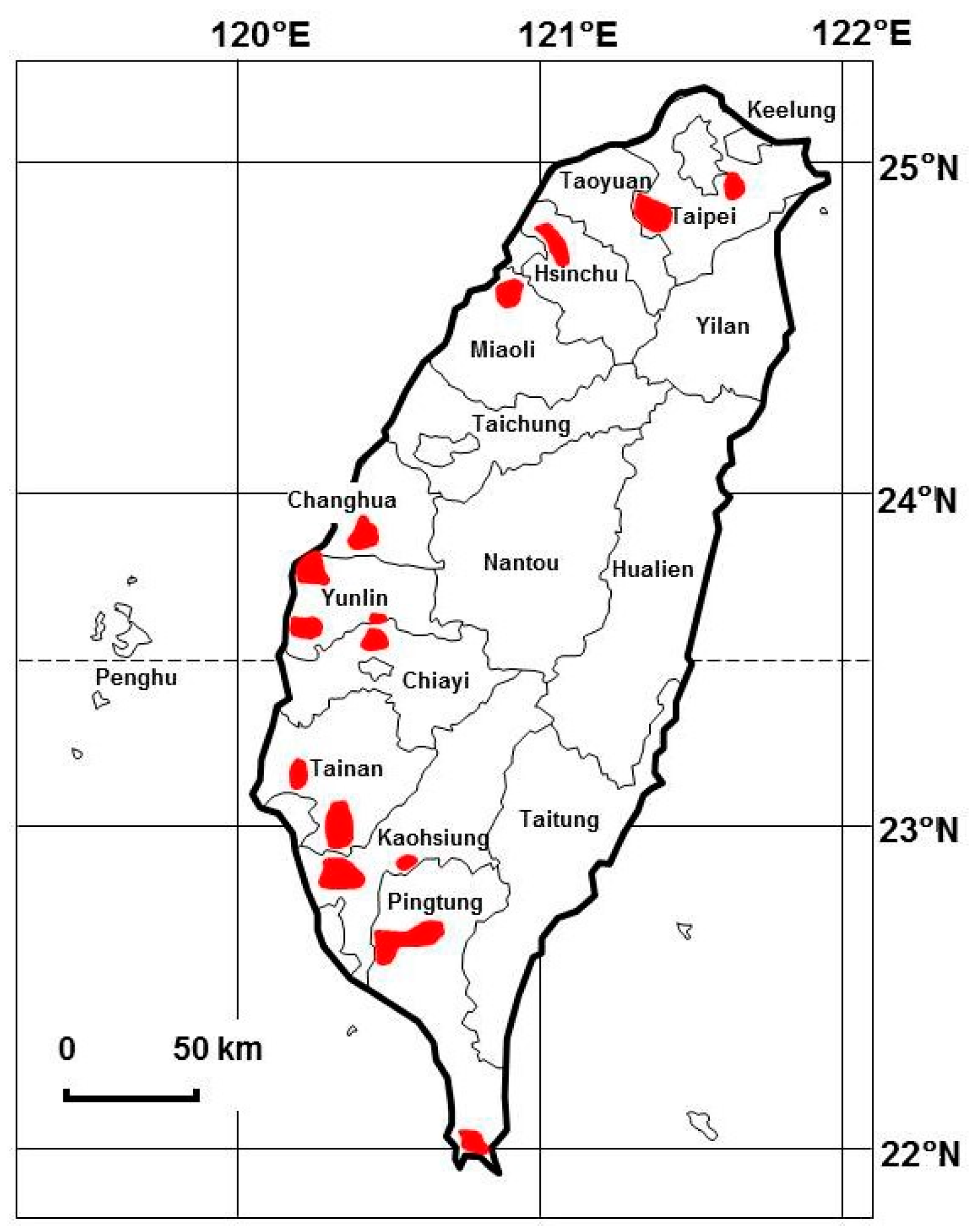
3.2. Framework of Monitoring Plans for Groundwater during the FMD Outbreak in Taiwan in 1997
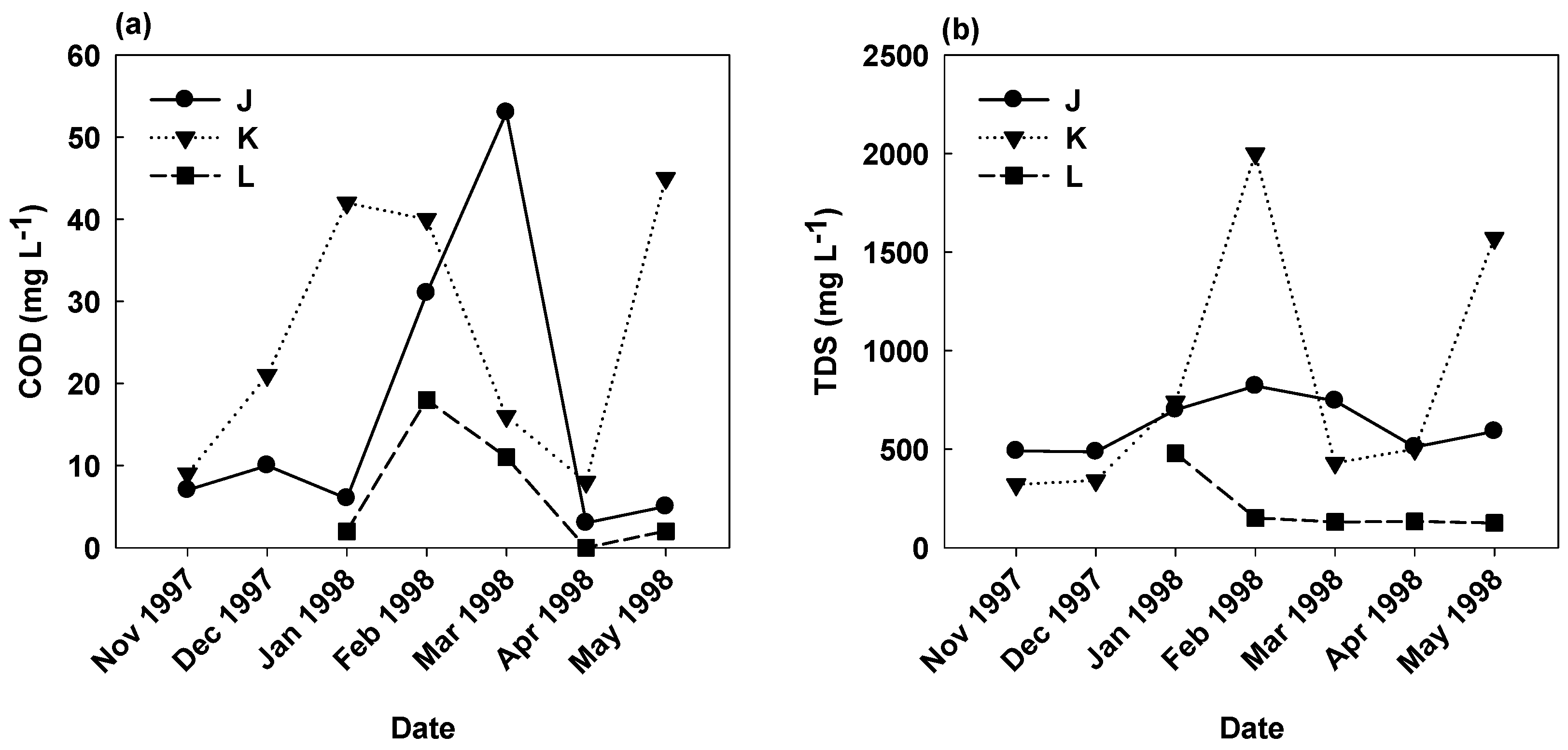
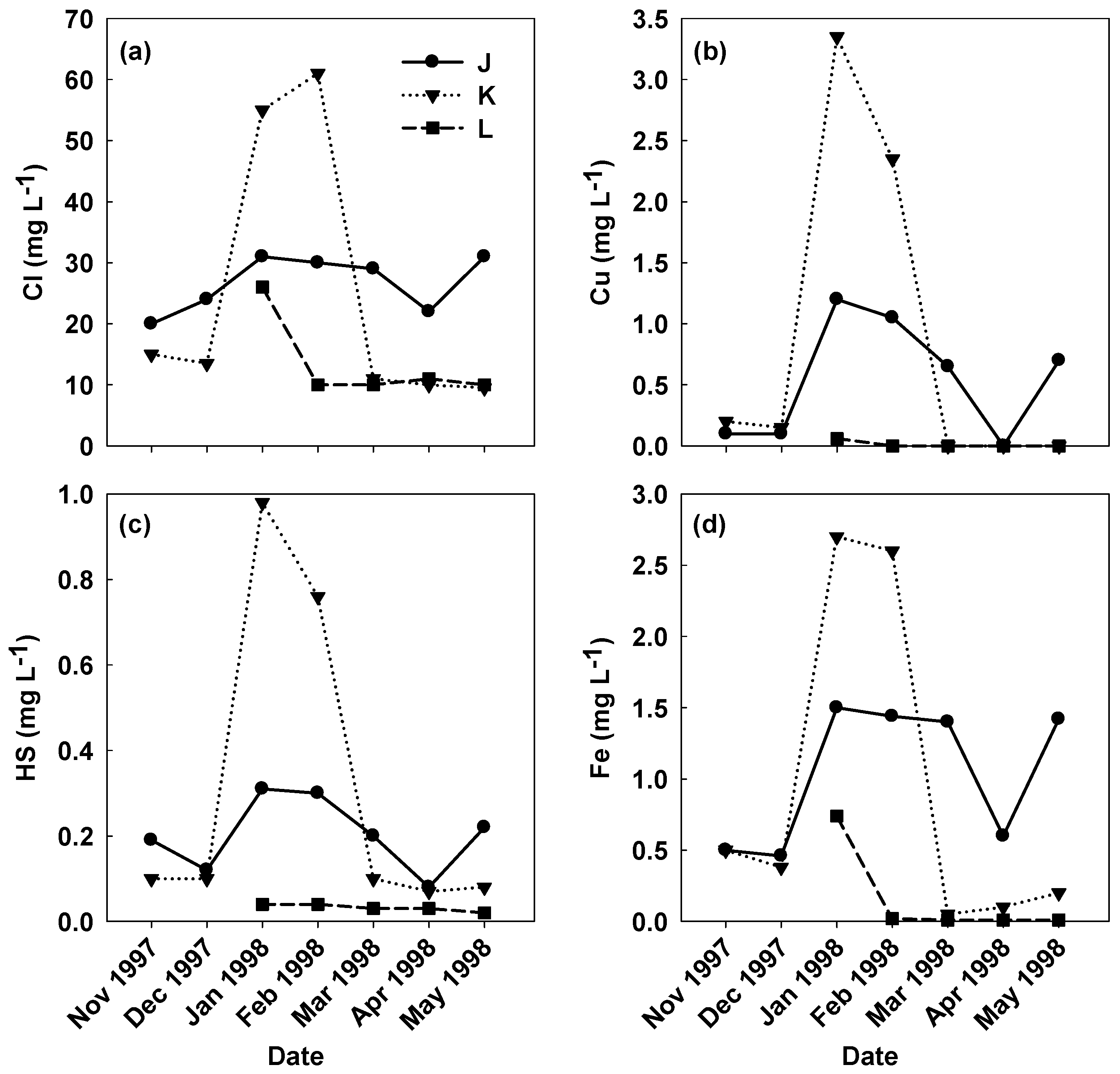
3.3. Effect of Pig Carcass Burial on Groundwater Quality
4. Future Directions
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- United State Department of Agriculture (USDA). Foreign Animal Disease Report. Animal and Plant Health Inspection Service; United State Department of Agriculture (USAD): Riverdale, MD, USA, 1998.
- Keeling, M.J.; Woolhouse, M.E.J.; Shaw, D.J.; Matthews, L.; Chase-Topping, M.; Haydon, D.T.; Cornell, S.J.; Kappey, J.; Wilesmith, J.; Grenfell, B.T. Dynamics of the 2001 UK foot and mouth epidemic: Stochastic dispersal in a heterogeneous landscape. Science 2001, 294, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.; Muriel, P.; Russell, D.; Osborne, P.; Bromley, A.; Rowland, M.; Creigh-Tyte, S.; Brown, C. Economic costs of the foot and mouth disease outbreak in the United Kingdom in 2001. Rev. Sci. Tech. 2002, 21, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Freedman, R.; Fleming, R. Water Quality Impacts of Burying Livestock Mortalities; Report Presented to the Livestock Mortality Recycling Project Steering Committee; Ridgetown College: Ridgetown, ON, Canada, 2003. [Google Scholar]
- Kim, H.S.; Kim, K.J. Microbial and chemical contamination of groundwater around livestock mortality burial sites in Korea—A review. Geosci. J. 2012, 16, 479–489. [Google Scholar] [CrossRef]
- Gwyther, C.L.; Williams, A.P.; Golyshin, P.N.; Edwards-Jones, G.; Jones, D.L. The environmental and biosecurity characteristics of livestock carcass disposal methods: A review. Waste Manag. 2011, 31, 767–778. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (U.S. EPA). Disposal of Domestic Birds Infected by Avian Influenza—An Overview of Considerations and Options; EPA530-R-06-009; U.S. Environmental Protection Agency: Washington, DC, USA, 2006.
- Chen, S.J.; Hsieh, L.T.; Chiu, S.C. Emission of polycyclic aromatic hydrocarbons from animal carcass incinerators. Sci. Total Environ. 2003, 313, 61–76. [Google Scholar] [CrossRef]
- Chen, S.J.; Hung, M.C.; Huang, K.L.; Hwang, W.I. Emission of heavy metals from animal carcass incinerators in Taiwan. Chemosphere 2004, 55, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Fonstad, T.A.; Meier, D.E.; Ingram, L.J.; Leonard, J. Evaluation and demonstration of composting as an option for dead animal management in Saskatchewan. Can. Biosyst. Eng. 2003, 45, 619–625. [Google Scholar]
- Ritter, W.F.; Chirnside, A.E.M. Impact of dead bird disposal pits on ground-water quality on the Delmarva Peninsula. Bioresour. Technol. 1995, 53, 105–111. [Google Scholar] [CrossRef]
- Yuan, Q.; Snow, D.D.; Bartelt-Hunt, S.L. Potential water quality impacts originating from land burial of cattle carcasses. Sci. Total Environ. 2013, 456–457, 246–253. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, A.J.; Milne, J.C. Leachate characteristics and management requirements arising from the foot & mouth operations in Scotland. In Proceedings of the Waste 2002, Integrated Waste Management and Pollution Control: Research, Policy, and Practice, Stratford upon Avon, UK, 24–26 September 2002; pp. 305–314.
- Pratt, D.L.; Fonstad, T.A. Livestock mortalities burial leachate chemistry after two years of decomposition. In Proceedings of the 3rd International Symposium on Management of Animal Carcasses, Tissue, and Related by-Products, Davis, CA, USA, 21–23 July 2009.
- Anderson, I. Foot and Mouth Disease 2001: Lessons to Be Learned Inquiry; The Stationary Office Limited: London, UK, 2002. [Google Scholar]
- Lim, H.H.; Shin, H.S.; Jeon, T.W.; Shin, S.K.; Jeung, Y.W. Arachidonic acid and fatty acid profiles as indicators of contamination from the leachates of animal carcasses. Water Sci. Technol. 2016. [Google Scholar] [CrossRef]
- Beal, C.D.; Gardner, E.A.; Menzies, N.W. Process, performance, and pollution potential: A review of septic tank-soil absorption systems. Aust. J. Soil Res. 2005, 43, 781–802. [Google Scholar] [CrossRef]
- Glanville, T.D.; Ahn, H.K.; Richard, T.L.; Shiers, L.E.; Harmon, J.D. Soil contamination caused by emergency bio-reduction of catastrophic livestock mortalities. Water Air Soil Pollut. 2009, 198, 285–295. [Google Scholar] [CrossRef]
- Engel, B.A.; Lim, K.J.; Choi, J.Y.; Theller, L. Chapter 14: Evaluating Environmental Impacts. In Carcass Disposal: A Comprehensive Review; National Agricultural Biosecurity Center, Kansas State University: Manhattan, KS, USA, 2004. [Google Scholar]


| Burial Site | Carcass Number | Impermeable Liner | Monitoring Well | Distance of Well from Burial Site (m) |
|---|---|---|---|---|
| Linlo | 14,000 | some | A 1 | 250 |
| B 1 | 200 | |||
| C 2 | 250 | |||
| D 2 | 400 | |||
| E 3 | 2000 | |||
| Chawchou | 10,000 | with | F 1 | 100 |
| G 2 | 10 | |||
| Sinbei | 110,000 | with | H 1 | 100 |
| I 2 | 500 | |||
| Pousan | 1000 | without | J 1 | 7 |
| K 2 | 20 | |||
| L 3 | 2000 |
| Burial Site | Well | TC 1 | FC 2 | COD 3 | NPOC 4 | Total Oil | TDS 5 |
|---|---|---|---|---|---|---|---|
| Linlo | A | 1.5 × 102 | 2.9 × 102 | 2.8 | 1.434 | 1.7 | 360 |
| ±180 | ±540 | ±4.0 | ±0.764 | ±1.4 | ±85 | ||
| B | 2.1 × 103 | 2.4 × 104 | 61.4 | 2.552 | 3.5 | 507 | |
| ±2300 | ±61,000 | ±117.3 | ±2.046 | ±2.2 | ±72 | ||
| C | 1.8 × 101 | 5.0 × 100 | 15.7 | 1.411 | 4.0 | 296 | |
| ±28 | ±10 | ±33.9 | ±1.045 | ±5.9 | ±112 | ||
| D | 2.5 × 103 | 4.7 × 103 | 5.7 | 1.265 | 3.7 | 444 | |
| ±6100 | ±13,000 | ±5.9 | ±1.356 | ±3.1 | ±152 | ||
| E | 8.0 × 103 | 2.7 × 103 | 5.3 | 1.101 | 1.6 | 410 | |
| ±14,000 | ±4400 | ±3.1 | ±0.648 | ±1.0 | ±109 | ||
| Chawchou | F | 1.3 × 103 | 1.5 × 104 | 7.3 | 1.316 | 1.9 | 619 |
| ±1100 | ±36,000 | ±8.5 | ±1.246 | ±1.3 | ±146 | ||
| G | 1.9 × 102 | 2.1 × 104 | 4.2 | 0.895 | 1.9 | 546 | |
| ±250 | ±50,000 | ±4.5 | ±0.512 | ±1.3 | ±211 | ||
| Sinbei | H | 1.2 × 103 | 1.4 × 103 | 1.7 | 1.010 | 2.7 | 159 |
| ±2500 | ±2400 | ±2.1 | ±0.829 | ±3.8 | ±52 | ||
| I | 4.4 × 102 | 2.8 × 103 | 1.7 | 0.640 | 1.4 | 319 | |
| ±520 | ±5800 | ±2.8 | ±0.417 | ±1.1 | ±186 | ||
| Pousan | J | 7.9 × 103 | 3.6 × 102 | 16.1 | 1.873 | 3.3 | 633 |
| ±7400 | ±470 | ±18.7 | ±1.016 | ±3.0 | ±176 | ||
| K | 2.7 × 104 | 7.8 × 102 | 26.0 | 4.092 | 7.8 | 823 | |
| ±20,000 | ±810 | ±16.4 | ±2.454 | ±4.3 | ±727 | ||
| L | 2.0 × 103 | 1.3 × 102 | 6.6 | 1.143 | 1.8 | 204 | |
| ±4100 | ±180 | ±7.8 | ±0.859 | ±0.4 | ±120 |
| Burial Site | Well | NO3−N | NO2−N | NH4-N | SO42− | Cl− | Cu | HS− | Fe |
|---|---|---|---|---|---|---|---|---|---|
| Linlo | A | 1.21 | 0.035 | ND 1 | 91.7 | 12.1 | ND 1 | 0.034 | ND |
| ±1.06 | ±0.030 | ±2.9 | ±1.2 | ±0.010 | |||||
| B | 6.57 | 0.081 | 0.546 | 112.9 | 21.9 | 0.21 | 0.104 | 2.75 | |
| ±17.55 | ±0.102 | ±0.465 | ±50.8 | ±10.6 | ±0.24 | ±0.061 | ±1.62 | ||
| C | 4.13 | 0.024 | ND | 67.3 | 9.1 | ND | 0.034 | ND | |
| ±0.26 | ±0.029 | ±12.6 | ±0.3 | ±0.010 | |||||
| D | 5.67 | 0.035 | 0.004 | 80.8 | 10.5 | 0 | 0.051 | ND | |
| ±0.58 | ±0.028 | ±0.133 | ±27.1 | ±1.0 | ±0.1 | ±0.028 | |||
| E | 7.09 | 0.035 | 0.031 | 89.8 | 8.8 | ND | 0.078 | 0.06 | |
| ±0.78 | ±0.032 | ±0.081 | ±32.9 | ±3.2 | ±0.049 | ±0.15 | |||
| Chawchou | F | 5.65 | 0.026 | ND | 54.0 | 8.7 | ND | 0.048 | ND |
| ±2.92 | ±0.035 | ±24.0 | ±0.9 | ±0.025 | |||||
| G | 6.95 | 0.023 | 0.024 | 184.2 | 23.7 | ND | 0.041 | ND | |
| ±6.90 | ±0.024 | ±0.044 | ±85.9 | ±3.1 | ±0.019 | ||||
| Sinbei | H | 2.35 | 0.023 | ND | 33.5 | 6.2 | ND | 0.052 | 0.03 |
| ±3.26 | ±0.025 | ±22.8 | ±3.2 | ±0.019 | ±0.09 | ||||
| I | 9.96 | 0.823 | 0.356 | 43.9 | 10.9 | ND | 0.037 | 0.02 | |
| ±2.73 | ±2.254 | ±0.941 | ±17.3 | ±2.1 | ±0.029 | ±0.07 | |||
| Pousan | J | 0.09 | 0.078 | 0.312 | 71.2 | 27.1 | 0.53 | 0.200 | 1.01 |
| ±0.14 | ±0.046 | ±0.115 | ±26.2 | ±4.7 | ±0.46 | ±0.092 | ±0.45 | ||
| K | 1.2 | 0.095 | 0.510 | 28.3 | 26.4 | 0.83 | 0.295 | 0.91 | |
| ±1.99 | ±0.107 | ±0.431 | ±22.2 | ±22.1 | ±1.36 | ±0.394 | ±1.19 | ||
| L | 0.09 | 0.010 | 0.078 | 21.3 | 13.6 | 0.02 | 0.033 | 0.15 | |
| ±0.17 | ±0.021 | ±0.047 | ±44.6 | ±7.0 | ±0.04 | ±0.006 | ±0.33 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hseu, Z.-Y.; Chen, Z.-S. Experiences of Mass Pig Carcass Disposal Related to Groundwater Quality Monitoring in Taiwan. Sustainability 2017, 9, 46. https://doi.org/10.3390/su9010046
Hseu Z-Y, Chen Z-S. Experiences of Mass Pig Carcass Disposal Related to Groundwater Quality Monitoring in Taiwan. Sustainability. 2017; 9(1):46. https://doi.org/10.3390/su9010046
Chicago/Turabian StyleHseu, Zeng-Yei, and Zueng-Sang Chen. 2017. "Experiences of Mass Pig Carcass Disposal Related to Groundwater Quality Monitoring in Taiwan" Sustainability 9, no. 1: 46. https://doi.org/10.3390/su9010046
APA StyleHseu, Z.-Y., & Chen, Z.-S. (2017). Experiences of Mass Pig Carcass Disposal Related to Groundwater Quality Monitoring in Taiwan. Sustainability, 9(1), 46. https://doi.org/10.3390/su9010046







