Utilization of Spent Coffee Grounds as a Feed Additive for Enhancing the Nutritional Value of Tenebrio molitor Larvae
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Insects and Spent Coffee Grounds (SCG) Material
2.3. Feeding Trial
2.4. Larval Composition Analysis
2.4.1. Water Content
2.4.2. Crude Protein Content
2.4.3. Carbohydrates Content
2.4.4. Ash Content
2.4.5. Total Fat, Fatty Acids, and Calculated Oxidizability Value (COX)
2.4.6. Energy Content
2.4.7. Vitamin C Content
2.4.8. β-Carotene–Vitamin A Content
2.4.9. Ferric-Reducing Antioxidant Power (FRAP) Assay
2.4.10. Total Polyphenol Content (TPC) Determination
2.5. Statistical Analysis
3. Results and Discussion
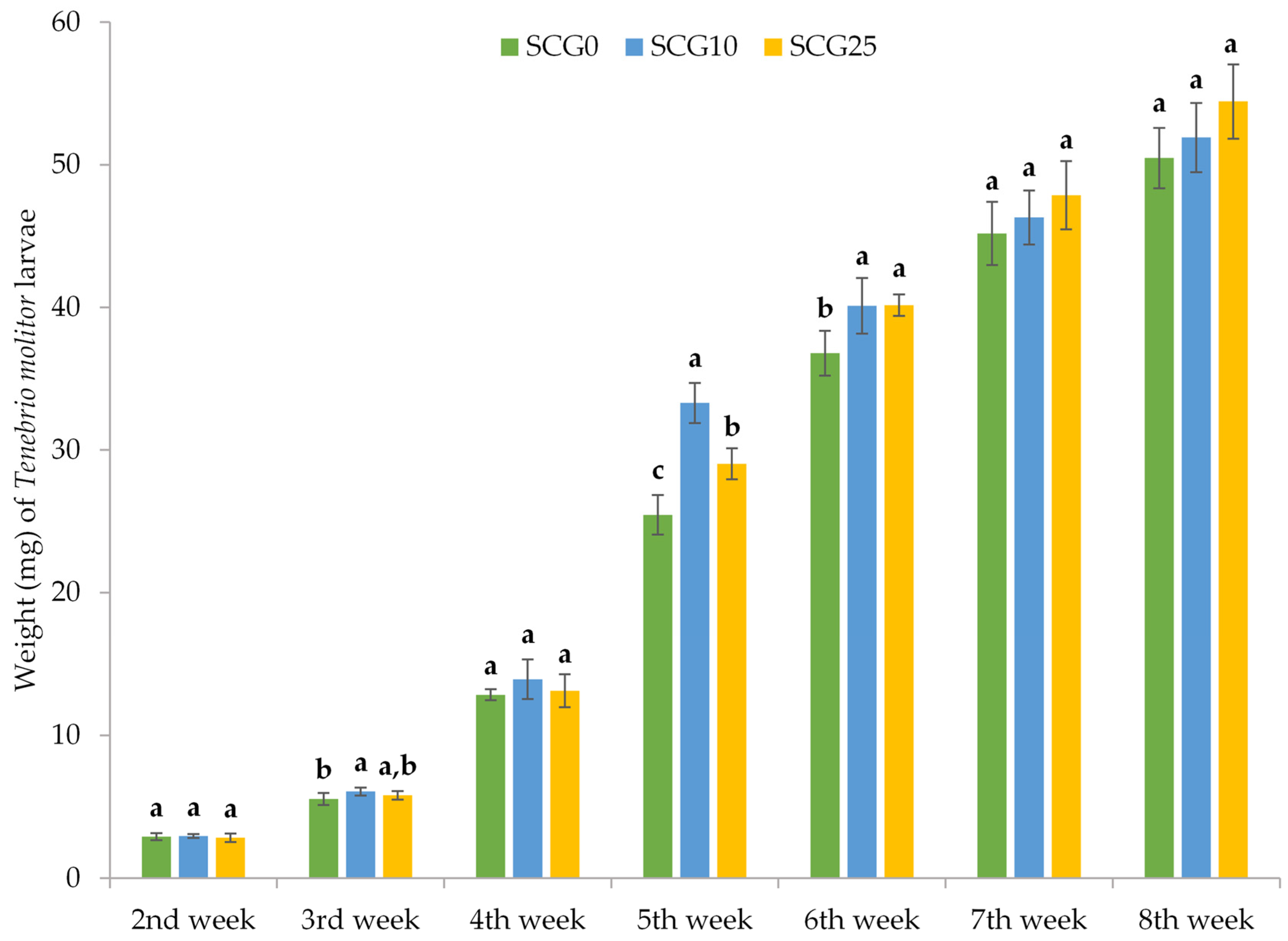
3.1. Survival and Growth of TM Larvae
3.2. Evaluation of the Nutritional Value of the Larvae
3.2.1. Proximate Composition
3.2.2. Vitamin C and A Content of TM Larvae
3.2.3. Antioxidant Properties of the Larvae’s Extracts
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| COX | Calculated Oxidizability Value |
| HH | Hypocholesterolemic/Hypercholesterolemic Ratio |
| HPI | Health-Promoting Index |
| IA | Index of Atherogenicity |
| IT | Index of Thrombogenicity |
| MUFA | Monounsaturated Fatty Acids |
| PUFA | Polyunsaturated Fatty Acids |
| SCG | Spent Coffee Grounds |
| SFA | Saturated Fatty Acids |
| TM | Tenebrio molitor |
| UFA | Unsaturated Fatty Acids |
References
- Kaza, S.; Yao, L.; Bhada-Tata, P.; Van Woerden, F. What a Waste 2.0 Introduction—“Snapshot of Solid Waste Management to 2050.” Overview Booklet; Urban Development Series; World Bank Group: Washington, DC, USA, 2018; pp. 1–38. [Google Scholar]
- FAO 2011. The Methodology of the FAO Study: “Global Food Losses and Food Waste—Extent, Causes and Prevention”; Vol. SIK Report; FAO: Rome, Italy, 2013; ISBN 9789172903234. [Google Scholar]
- Patwa, N.; Sivarajah, U.; Seetharaman, A.; Sarkar, S.; Maiti, K.; Hingorani, K. Towards a Circular Economy: An Emerging Economies Context. J. Bus. Res. 2021, 122, 725–735. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Carneiro, L.M.; Silva, J.P.A.; Roberto, I.C.; Teixeira, J.A. A Study on Chemical Constituents and Sugars Extraction from Spent Coffee Grounds. Carbohydr. Polym. 2011, 83, 368–374. [Google Scholar] [CrossRef]
- Chatzimitakos, T.; Athanasiadis, V.; Kotsou, K.; Palaiogiannis, D.; Bozinou, E.; Lalas, S.I. Optimized Isolation Procedure for the Extraction of Bioactive Compounds from Spent Coffee Grounds. Appl. Sci. 2023, 13, 2819. [Google Scholar] [CrossRef]
- Getachew, A.T.; Chun, B.S. Influence of Pretreatment and Modifiers on Subcritical Water Liquefaction of Spent Coffee Grounds: A Green Waste Valorization Approach. J. Clean. Prod. 2017, 142, 3719–3727. [Google Scholar] [CrossRef]
- Seo, H.S.; Park, B.H. Phenolic Compound Extraction from Spent Coffee Grounds for Antioxidant Recovery. Korean J. Chem. Eng. 2019, 36, 186–190. [Google Scholar] [CrossRef]
- Panusa, A.; Zuorro, A.; Lavecchia, R.; Marrosu, G.; Petrucci, R. Recovery of Natural Antioxidants from Spent Coffee Grounds. J. Agric. Food Chem. 2013, 61, 4162–4168. [Google Scholar] [CrossRef] [PubMed]
- Smyrnakis, G.; Stamoulis, G.; Palaiogiannis, D.; Chatzimitakos, T.; Athanasiadis, V.; Lalas, S.I.; Makris, D.P. Recovery of Polyphenolic Antioxidants from Coffee Silverskin Using Acid-Catalyzed Ethanol Organosolv Treatment. ChemEngineering 2023, 7, 72. [Google Scholar] [CrossRef]
- Santana-Gálvez, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Chlorogenic Acid: Recent Advances on Its Dual Role as a Food Additive and a Nutraceutical against Metabolic Syndrome. Molecules 2017, 22, 358. [Google Scholar] [CrossRef]
- Kang, N.J.; Lee, K.W.; Shin, B.J.; Jung, S.K.; Hwang, M.K.; Bode, A.M.; Heo, Y.S.; Lee, H.J.; Dong, Z. Caffeic Acid, a Phenolic Phytochemical in Coffee, Directly Inhibits Fyn Kinase Activity and UVB-Induced COX-2 Expression. Carcinogenesis 2009, 30, 321–330. [Google Scholar] [CrossRef]
- Haider, K.; Haider, M.R.; Neha, K.; Yar, M.S. Free Radical Scavengers: An Overview on Heterocyclic Advances and Medicinal Prospects. Eur. J. Med. Chem. 2020, 204, 112607. [Google Scholar] [CrossRef]
- Zarrinbakhsh, N.; Rodriguez, A.; Misra, M.; Mohanty, A. Characterization of Wastes and Coproducts from the Coffee Industry for Composite Material Production. BioRes 2016, 11, 7637–7653. [Google Scholar] [CrossRef]
- Bordiean, A.; Krzyżaniak, M.; Stolarski, M.J. Bioconversion Potential of Agro-Industrial Byproducts by Tenebrio molitor—Long-Term Results. Insects 2022, 13, 810. [Google Scholar] [CrossRef] [PubMed]
- Aidoo, O.F.; Osei-Owusu, J.; Asante, K.; Dofuor, A.K.; Boateng, B.O.; Debrah, S.K.; Ninsin, K.D.; Siddiqui, S.A.; Chia, S.Y. Insects as Food and Medicine: A Sustainable Solution for Global Health and Environmental Challenges. Front. Nutr. 2023, 10, 1113219. [Google Scholar] [CrossRef] [PubMed]
- Thrastardottir, R.; Olafsdottir, H.T.; Thorarinsdottir, R.I. Yellow Mealworm and Black Soldier Fly Larvae for Feed and Food Production in Europe, with Emphasis on Iceland. Foods 2021, 10, 2744. [Google Scholar] [CrossRef]
- Zielińska, E.; Baraniak, B.; Karaś, M.; Rybczyńska, K.; Jakubczyk, A. Selected Species of Edible Insects as a Source of Nutrient Composition. Food Res. Int. 2015, 77, 460–466. [Google Scholar] [CrossRef]
- Jantzen da Silva Lucas, A.; Menegon de Oliveira, L.; da Rocha, M.; Prentice, C. Edible Insects: An Alternative of Nutritional, Functional and Bioactive Compounds. Food Chem. 2020, 311, 126022. [Google Scholar] [CrossRef]
- Andreadis, S.S.; Panteli, N.; Mastoraki, M.; Rizou, E.; Stefanou, V.; Tzentilasvili, S.; Sarrou, E.; Chatzifotis, S.; Krigas, N.; Antonopoulou, E. Towards Functional Insect Feeds: Agri-Food by-Products Enriched with Post-Distillation Residues of Medicinal Aromatic Plants in Tenebrio molitor (Coleoptera: Tenebrionidae) Breeding. Antioxidants 2022, 11, 68. [Google Scholar] [CrossRef]
- Bordiean, A.; Krzyżaniak, M.; Stolarski, M.J.; Peni, D. Growth Potential of Yellow Mealworm Reared on Industrial Residues. Agric. 2020, 10, 599. [Google Scholar] [CrossRef]
- Bordiean, A.; Krzyżaniak, M.; Aljewicz, M.; Stolarski, M.J. Influence of Different Diets on Growth and Nutritional Composition of Yellow Mealworm. Foods 2022, 11, 3075. [Google Scholar] [CrossRef]
- Biasato, I.; Gasco, L.; De Marco, M.; Renna, M.; Rotolo, L.; Dabbou, S.; Capucchio, M.T.; Biasibetti, E.; Tarantola, M.; Sterpone, L.; et al. Yellow Mealworm Larvae (Tenebrio molitor) Inclusion in Diets for Male Broiler Chickens: Effects on Growth Performance, Gut Morphology, and Histological Findings. Poult. Sci. 2018, 97, 540–548. [Google Scholar] [CrossRef]
- Rumpold, B.A.; Schlüter, O.K. Nutritional Composition and Safety Aspects of Edible Insects. Mol. Nutr. Food Res. 2013, 57, 802–823. [Google Scholar] [CrossRef] [PubMed]
- Truman, J.W. The Evolution of Insect Metamorphosis. Curr. Biol. 2019, 29, R1252–R1268. [Google Scholar] [CrossRef] [PubMed]
- May, J.C.; Wheeler, R.M.; Grim, E. The Gravimetric Method for the Determination of Residual Moisture in Freeze-Dried Biological Products. Cryobiology 1989, 26, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Kotsou, K.; Chatzimitakos, T.; Athanasiadis, V.; Bozinou, E.; Rumbos, C.I.; Athanassiou, C.G.; Lalas, S.I. Enhancing the Nutritional Profile of Tenebrio molitor Using the Leaves of Moringa oleifera. Foods 2023, 12, 2612. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.; Hwang, I.; Nho, C.; Kim, S.; Kim, S. Determination of Carbohydrate Composition in Mealworm. Foods 2021, 10, 640. [Google Scholar] [CrossRef] [PubMed]
- Al-Amiery, A.A.; Al-Majedy, Y.K.; Kadhum, A.A.H.; Mohamad, A.B. Hydrogen Peroxide Scavenging Activity of Novel Coumarins Synthesized Using Different Approaches. PLoS ONE 2015, 10, e0132175. [Google Scholar] [CrossRef]
- Ocampo, E.T.M.; Libron, J.A.M.A.; Guevarra, M.L.D.; Mateo, J.M.C. Phytochemical Screening, Phenolic Acid Profiling and Antioxidant Activity Analysis of Peels from Selected Mango (Mangifera spp.) Genotypes in the Philippines. Food Res. 2020, 4, 1116–1124. [Google Scholar] [CrossRef]
- Makris, D.P.; Kefalas, P. Characterization of Polyphenolic Phytochemicals in Red Grape Pomace. Int. J. Waste Resour. 2013, 3, 2–5. [Google Scholar] [CrossRef]
- Cicco, N.; Lanorte, M.T.; Paraggio, M.; Viggiano, M.; Lattanzio, V. A Reproducible, Rapid and Inexpensive Folin-Ciocalteu Micro-Method in Determining Phenolics of Plant Methanol Extracts. Microchem. J. 2009, 91, 107–110. [Google Scholar] [CrossRef]
- Pinotti, L.; Ottoboni, M. Substrate as Insect Feed for Bio-Mass Production. J. Insects Food Feed 2021, 7, 585–596. [Google Scholar] [CrossRef]
- LeCato, G.; Flaherty, B. Description of Eggs of Selected Species of Stored-Product Insects (Coleoptera and Lepidoptera). J. Kansas Entomol. Soc. 1974, 39, 308–317. [Google Scholar]
- Pakshir, K.; Dehghani, A.; Nouraei, H.; Zareshahrabadi, Z.; Zomorodian, K. Evaluation of Fungal Contamination and Ochratoxin A Detection in Different Types of Coffee by HPLC-Based Method. J. Clin. Lab. Anal. 2021, 35, e24001. [Google Scholar] [CrossRef] [PubMed]
- Kröncke, N.; Benning, R. Self-Selection of Feeding Substrates by Tenebrio molitor Larvae of Different Ages to Determine Optimal Macronutrient Intake and the Influence on Larval Growth and Protein Content. Insects 2022, 13, 657. [Google Scholar] [CrossRef] [PubMed]
- Hussein, H.; Abouamer, W.; Ali, H.; Elkhadragy, M.; Yehia, H.; Farouk, A. The Valorization of Spent Coffee Ground Extract as a Prospective Insecticidal Agent against Some Main Key Pests of Phaseolus Vulgaris in the Laboratory and Field. Plants 2022, 11, 1124. [Google Scholar] [CrossRef]
- Thanasoponkul, W.; Changbunjong, T.; Sukkurd, R.; Saiwichai, T. Spent Coffee Grounds and Novaluron Are Toxic to Aedes Aegypti (Diptera: Culicidae) Larvae. Insects 2023, 14, 564. [Google Scholar] [CrossRef] [PubMed]
- Kröncke, N.; Wittke, S.; Steinmann, N.; Benning, R. Analysis of the Composition of Different Instars of Tenebrio molitor Larvae Using Near-Infrared Reflectance Spectroscopy for Prediction of Amino and Fatty Acid Content. Insects 2023, 14, 310. [Google Scholar] [CrossRef]
- Kröncke, N.; Benning, R. Determination of Moisture and Protein Content in Living Mealworm Larvae (Tenebrio molitor L.) Using Near-Infrared Reflectance Spectroscopy (NIRS). Insects 2022, 13, 560. [Google Scholar] [CrossRef]
- Costa, S.; Pedro, S.; Lourenço, H.; Batista, I.; Teixeira, B.; Bandarra, N.M.; Murta, D.; Nunes, R.; Pires, C. Evaluation of Tenebrio molitor Larvae as an Alternative Food Source. NFS J. 2020, 21, 57–64. [Google Scholar] [CrossRef]
- Nowak, V.; Persijn, D.; Rittenschober, D.; Charrondiere, U.R. Review of Food Composition Data for Edible Insects. Food Chem. 2016, 193, 39–46. [Google Scholar] [CrossRef]
- Iriondo-Dehond, A.; Uranga, J.A.; Del Castillo, M.D.; Abalo, R. Effects of Coffee and Its Components on the Gastrointestinal Tract and the Brain–Gut Axis. Nutrients 2021, 13, 88. [Google Scholar] [CrossRef]
- Siulapwa, N.; Mwambungu, A.; Lungu, E.; Sichilima, W. Nutritional Value of Four Common Edible Insects in Zambia. Int. J. Sci. Res. 2014, 3, 876–884. [Google Scholar]
- Melo, V.; Garcia, M.; Sandoval, H.; Jiménez, H.D.; Calvo, C. Quality Proteins from Edible Indigenous Insect Food of Latin America and Asia. Emirates J. Food Agric. 2011, 23, 283–289. [Google Scholar]
- Daniel, J.R.; Vidovic, N. Carbohydrates, Role in Human Nutrition; Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar] [CrossRef]
- Abidin, N.A.Z.; Kormin, F.; Abidin, N.A.Z.; Anuar, N.A.F.M.; Bakar, M.F.A. The Potential of Insects as Alternative Sources of Chitin: An Overview on the Chemical Method of Extraction from Various Sources. Int. J. Mol. Sci. 2020, 21, 4978. [Google Scholar] [CrossRef] [PubMed]
- Dong, T.; Guo, M.; Zhang, P.; Sun, G.; Chen, B. The Effects of Low-Carbohydrate Diets on Cardiovascular Risk Factors: A Meta-Analysis. PLoS ONE 2020, 15, e0225348. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.L.; Esteves, S.S.; da Costa Pereira, A.; Yancy, W.S.J.; Nunes, J.P.L. Systematic Review and Meta-Analysis of Clinical Trials of the Effects of Low Carbohydrate Diets on Cardiovascular Risk Factors. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2012, 13, 1048–1066. [Google Scholar] [CrossRef]
- Ghaly, A.E.; Alkoaik, F.N. The Yellow Mealworm as a Novel Source of Protein. Am. J. Agric. Biol. Sci. 2009, 4, 319–331. [Google Scholar] [CrossRef]
- Dreassi, E.; Cito, A.; Zanfini, A.; Materozzi, L.; Botta, M.; Francardi, V. Dietary Fatty Acids Influence the Growth and Fatty Acid Composition of the Yellow Mealworm Tenebrio molitor (Coleoptera: Tenebrionidae). Lipids 2017, 52, 285–294. [Google Scholar] [CrossRef]
- Ghosh, S.; Lee, S.M.; Jung, C.; Meyer-Rochow, V.B. Nutritional Composition of Five Commercial Edible Insects in South Korea. J. Asia. Pac. Entomol. 2017, 20, 686–694. [Google Scholar] [CrossRef]
- Jajic, I.; Popovic, A.; Urosevic, M.; Krstovic, S.; Petrovic, M.; Guljas, D.; Samardzic, M. Fatty and Amino Acid Profile of Mealworm Larvae (Tenebrio molitor L.). Biotechnol. Anim. Husb. 2020, 36, 167–180. [Google Scholar] [CrossRef]
- Pipoyan, D.; Stepanyan, S.; Stepanyan, S.; Beglaryan, M.; Costantini, L.; Molinari, R.; Merendino, N. The Effect of Trans Fatty Acids on Human Health: Regulation and Consumption Patterns. Foods 2021, 10, 2452. [Google Scholar] [CrossRef]
- Yun, J.M.; Surh, J. Fatty Acid Composition as a Predictor for the Oxidation Stability of Korean Vegetable Oils with or without Induced Oxidative Stress. Prev. Nutr. Food Sci. 2012, 17, 158–165. [Google Scholar] [CrossRef]
- Baum, S.J.; Kris-Etherton, P.M.; Willett, W.C.; Lichtenstein, A.H.; Rudel, L.L.; Maki, K.C.; Whelan, J.; Ramsden, C.E.; Block, R.C. Fatty Acids in Cardiovascular Health and Disease: A Comprehensive Update. J. Clin. Lipidol. 2012, 6, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Paszczyk, B.; Polak-śliwińska, M.; Łuczyńska, J. Fatty Acids Profile, Trans Isomers, and Lipid Quality Indices in Smoked and Unsmoked Cheeses and Cheese-like Products. Int. J. Environ. Res. Public Health 2020, 17, 71. [Google Scholar] [CrossRef] [PubMed]
- Atik, I.; Karasu, S.; Sevik, R. Physicochemical and Bioactive Properties of Cold Press Wild Plum (Prunus spinosa) and Sour Cherry (Prunus cerasus) Kernel Oils: Fatty Acid, Sterol and Phenolic Profile. Riv. Ital. Sostanze Grasse 2022, 99, 13–20. [Google Scholar]
- Wali, J.A.; Jarzebska, N.; Raubenheimer, D.; Simpson, S.J.; Rodionov, R.N.; O’Sullivan, J.F. Cardio-Metabolic Effects of High-Fat Diets and Their Underlying Mechanisms-A Narrative Review. Nutrients 2020, 12, 1505. [Google Scholar] [CrossRef] [PubMed]
- Al-Amrousi, E.F.; Badr, A.N.; Abdel-Razek, A.G.; Gromadzka, K.; Drzewiecka, K.; Hassanein, M.M.M. A Comprehensive Study of Lupin Seed Oils and the Roasting Effect on Their Chemical and Biological Activity. Plants 2022, 11, 2301. [Google Scholar] [CrossRef]
- Nantapo, C.T.W.; Muchenje, V.; Hugo, A. Atherogenicity Index and Health-Related Fatty Acids in Different Stages of Lactation from Friesian, Jersey and Friesian × Jersey Cross Cow Milk under a Pasture-Based Dairy System. Food Chem. 2014, 146, 127–133. [Google Scholar] [CrossRef]
- Kumar, M.; Kumari, P.; Trivedi, N.; Shukla, M.K.; Gupta, V.; Reddy, C.R.K.; Jha, B. Minerals, PUFAs and Antioxidant Properties of Some Tropical Seaweeds from Saurashtra Coast of India. J. Appl. Phycol. 2011, 23, 797–810. [Google Scholar] [CrossRef]
- Chen, J.; Liu, H. Nutritional Indices for Assessing Fatty Acids: A Mini-Review. Int. J. Mol. Sci. 2020, 21, 5695. [Google Scholar] [CrossRef]
- Makhutova, O.N.; Nokhsorov, V.V.; Stoyanov, K.N.; Dudareva, L.V.; Petrov, K.A. Preliminary Estimation of Nutritional Quality of the Meat, Liver, and Fat of the Indigenous Yakutian Cattle Based on Their Fatty Acid Profiles. Foods 2023, 12, 3226. [Google Scholar] [CrossRef]
- Doley, J. Vitamins and Minerals in Older Adults: Causes, Diagnosis, and Treatment of Deficiency; Elsevier Inc.: Amsterdam, The Netherlands, 2017; ISBN 9780128092996. [Google Scholar]
- Oonincx, D.G.A.B.; Finke, M.D. Insects as a Complete Nutritional Source. J. Insects Food Feed 2023, 9, 541–543. [Google Scholar] [CrossRef]
- Wu, C.T.; Agrawal, D.C.; Huang, W.Y.; Hsu, H.C.; Yang, S.J.; Huang, S.L.; Lin, Y.S. Functionality Analysis of Spent Coffee Ground Extracts Obtained by the Hydrothermal Method. J. Chem. 2019, 2019, 4671438. [Google Scholar] [CrossRef]
- Kapała, A.; Szlendak, M.; Motacka, E. The Anti-Cancer Activity of Lycopene: A Systematic Review of Human and Animal Studies. Nutrients 2022, 14, 5152. [Google Scholar] [CrossRef] [PubMed]
- Anand, R.; Mohan, L.; Bharadvaja, N. Disease Prevention and Treatment Using β-Carotene: The Ultimate Provitamin A. Rev. Bras. Farmacogn. 2022, 32, 491–501. [Google Scholar] [CrossRef]
- Payne, C.L.R.; Scarborough, P.; Rayner, M.; Nonaka, K. A Systematic Review of Nutrient Composition Data Available for Twelve Commercially Available Edible Insects, and Comparison with Reference Values. Trends Food Sci. Technol. 2016, 47, 69–77. [Google Scholar] [CrossRef]
- Andrade, C.; Perestrelo, R.; Câmara, J.S. Bioactive Compounds and Antioxidant Activity from Spent Coffee Grounds as a Powerful Approach for Its Valorization. Molecules 2022, 27, 7504. [Google Scholar] [CrossRef]
- Turck, D.; Bohn, T.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; Pelaez, C.; et al. Safety of Frozen and Dried Formulations from Whole Yellow Mealworm (Tenebrio molitor Larva) as a Novel Food Pursuant to Regulation (EU) 2015/2283. EFSA J. 2021, 19, e06778. [Google Scholar] [CrossRef]
| Diets | 2nd Week | 3rd Week | 4th Week | 5th Week | 6th Week | 7th Week | 8th Week |
|---|---|---|---|---|---|---|---|
| SCG0 | 97 ± 2 | 95 ± 3 | 94 ± 3 | 93 ± 4 | 91 ± 3 | 90 ± 2 a | 89 ± 2 a |
| SCG10 | 96 ± 4 | 95 ± 4 | 93 ± 4 | 92 ± 3 | 92 ± 3 | 87 ± 5 a,b | 86 ± 5 a |
| SCG25 | 96 ± 2 | 95 ± 2 | 93 ± 3 | 91 ± 4 | 90 ± 3 | 83 ± 3 b | 80 ± 3 b |
| % Composition of Dry Weight | SCG0 | SCG10 | SCG25 |
|---|---|---|---|
| Crude protein | 32.59 ± 0.43 c,* | 47.34 ± 0.17 a | 42.65 ± 0.26 b |
| Crude fat | 22.77 ± 1.29 a | 20.2 ± 1.07 a | 21.93 ± 1.55 a |
| Carbohydrates | 25.72 ± 0.04 a | 13.41 ± 0.77 c | 19.42 ± 0.21 b |
| Crude ash | 1.89 ± 0.07 c | 3.51 ± 0.04 a | 2.44 ± 0.03 b |
| Energy (kcal/100 g) | 438.17 ± 13.49 a | 424.8 ± 13.39 a | 445.65 ± 15.83 a |
| Fatty Acid (%) | Diets | ||
|---|---|---|---|
| SCG0 | SCG10 | SCG25 | |
| C12:0 | 0.08 ± 0.00 b,* | 0.10 ± 0.00 a | nd ** |
| C14:0 | 2.06 ± 0.08 b | 2.77 ± 0.19 a | 2.36 ± 0.06 b |
| C16:0 | 19.3 ± 1.37 a | 18.94 ± 0.53 a | 20.99 ± 1.26 a |
| C18:0 | 0.21 ± 0.01 b | 0.23 ± 0.01 a | nd |
| C18:1 | 23.98 ± 1.39 b | 24.83 ± 1.86 b | 34.98 ± 1.64 a |
| C18:2 (ω-6) | 53.87 ± 4.04 a | 52.67 ± 3.53 a | 40.88 ± 1.92 b |
| C20:0 | 0.50 ± 0.03 b | 0.45 ± 0.03 b | 0.78 ± 0.02 a |
| ∑ SFA 1 | 22.15 ± 1.49 a | 22.5 ± 0.76 a | 24.13 ± 1.34 a |
| ∑ MUFA 2 | 23.98 ± 1.39 b | 24.83 ± 1.86 b | 34.98 ± 1.64 a |
| ∑ PUFA 3 | 53.87 ± 4.04 a | 52.67 ± 3.53 a | 40.88 ± 1.92 b |
| ∑ UFA 4 | 77.85 ± 5.43 a | 77.5 ± 5.39 a | 75.87 ± 3.57 a |
| PUFA:SFA ratio | 2.43 ± 0.02 a | 2.34 ± 0.08 a | 1.69 ± 0.01 b |
| MUFA:PUFA ratio | 0.45 ± 0.01 c | 0.47 ± 0.00 b | 0.86 ± 0.00 a |
| COX 5 | 5.79 ± 0.43 a | 5.67 ± 0.38 a | 4.56 ± 0.21 b |
| IA 6 | 0.35 ± 0.00 b | 0.39 ± 0.01 a | 0.4 ± 0.00 a |
| IT 7 | 0.55 ± 0.00 b | 0.57 ± 0.02 b | 0.62 ± 0.01 a |
| HH 8 | 3.63 ± 0.01 a | 3.55 ± 0.13 a | 3.25 ± 0.03 b |
| HPI 9 | 2.82 ± 0.02 a | 2.57 ± 0.07 b | 2.49 ± 0.01 b |
| Diets | Vitamin C (μg/g) | β-Carotene (μg/g) | Vitamin A (μg RAE/100 g) |
|---|---|---|---|
| SCG0 | 218.09 ± 2.09 c,* | 0.91 ± 0.06 c | 2.72 ± 0.19 c |
| SCG10 | 313.25 ± 3.24 b | 4.36 ± 0.56 b | 13.02 ± 1.68 b |
| SCG25 | 395.35 ± 1.98 a | 8.42 ± 0.34 a | 25.1 ± 1 a |
| Diets | FRAP (μmol AAE/g) | TPC (mg GAE/g) |
|---|---|---|
| SCG0 | 130.8 ± 2.48 c,* | 43.22 ± 1.8 b |
| SCG10 | 143.23 ± 1.87 b | 53.31 ± 1.48 a |
| SCG25 | 250.95 ± 3.53 a | 55.76 ± 0.24 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotsou, K.; Chatzimitakos, T.; Athanasiadis, V.; Bozinou, E.; Athanassiou, C.G.; Lalas, S.I. Utilization of Spent Coffee Grounds as a Feed Additive for Enhancing the Nutritional Value of Tenebrio molitor Larvae. Sustainability 2023, 15, 16224. https://doi.org/10.3390/su152316224
Kotsou K, Chatzimitakos T, Athanasiadis V, Bozinou E, Athanassiou CG, Lalas SI. Utilization of Spent Coffee Grounds as a Feed Additive for Enhancing the Nutritional Value of Tenebrio molitor Larvae. Sustainability. 2023; 15(23):16224. https://doi.org/10.3390/su152316224
Chicago/Turabian StyleKotsou, Konstantina, Theodoros Chatzimitakos, Vassilis Athanasiadis, Eleni Bozinou, Christos G. Athanassiou, and Stavros I. Lalas. 2023. "Utilization of Spent Coffee Grounds as a Feed Additive for Enhancing the Nutritional Value of Tenebrio molitor Larvae" Sustainability 15, no. 23: 16224. https://doi.org/10.3390/su152316224
APA StyleKotsou, K., Chatzimitakos, T., Athanasiadis, V., Bozinou, E., Athanassiou, C. G., & Lalas, S. I. (2023). Utilization of Spent Coffee Grounds as a Feed Additive for Enhancing the Nutritional Value of Tenebrio molitor Larvae. Sustainability, 15(23), 16224. https://doi.org/10.3390/su152316224











