Stabilization of Waste Mercury with Sulfide through the Ball-Mill Method and Heat Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Apparatus
2.3. Analytical Methods
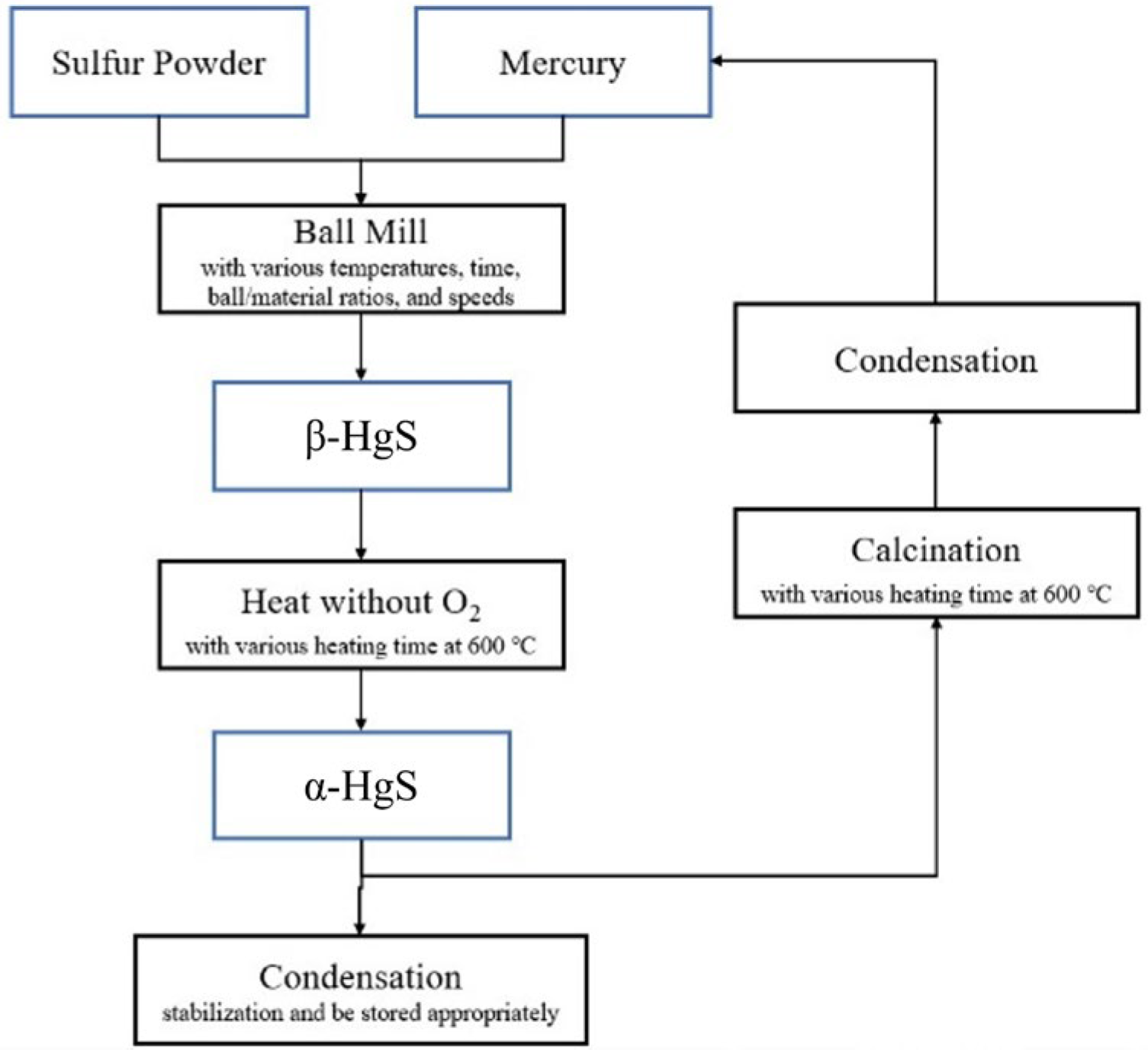
2.4. Ball Mill
2.5. Heat without O2 (β-HgS to α-HgS)
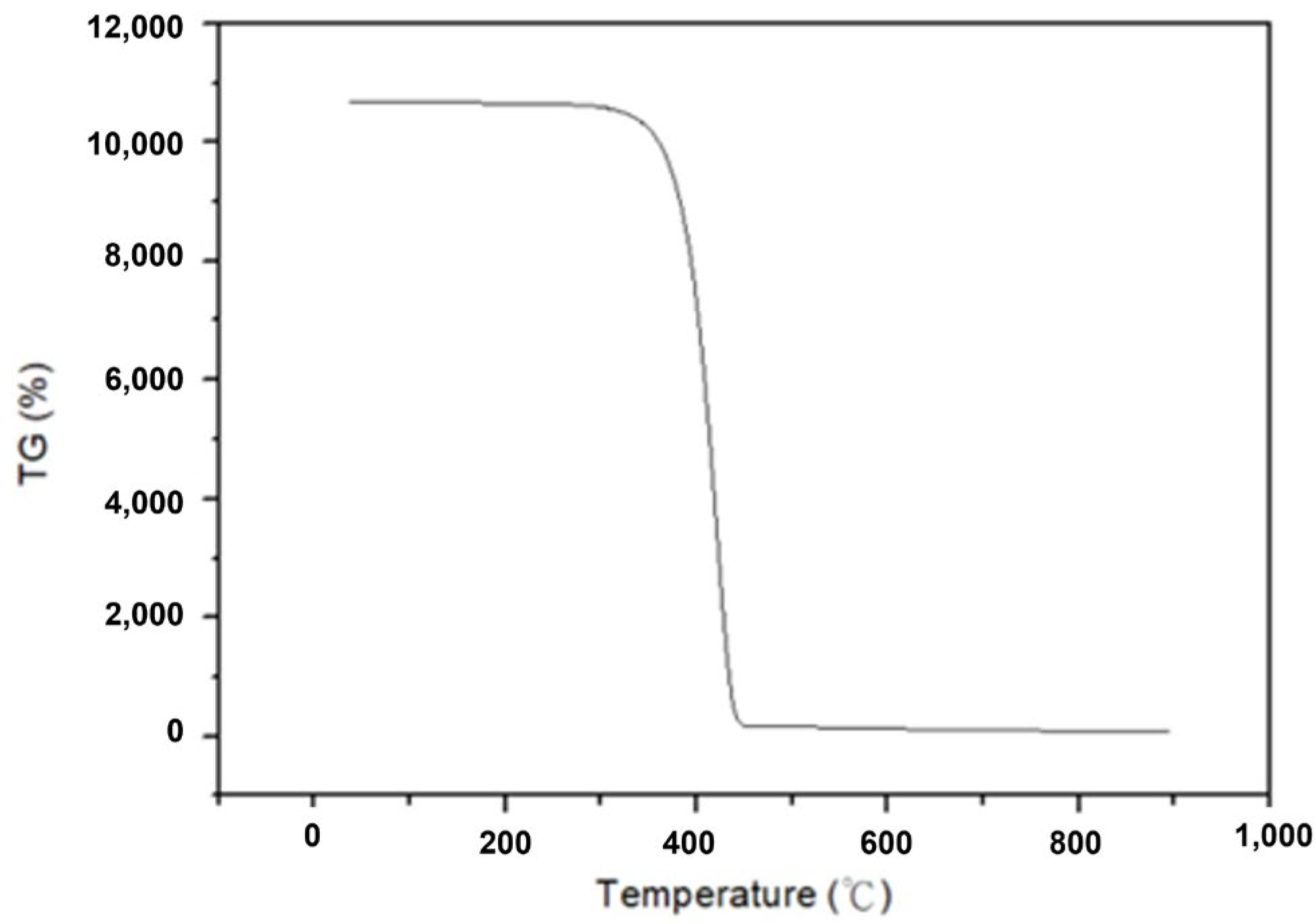
2.6. Calcination and Condensation (α-HgS to Mercury)
3. Results and Discussion
3.1. Ball Mill
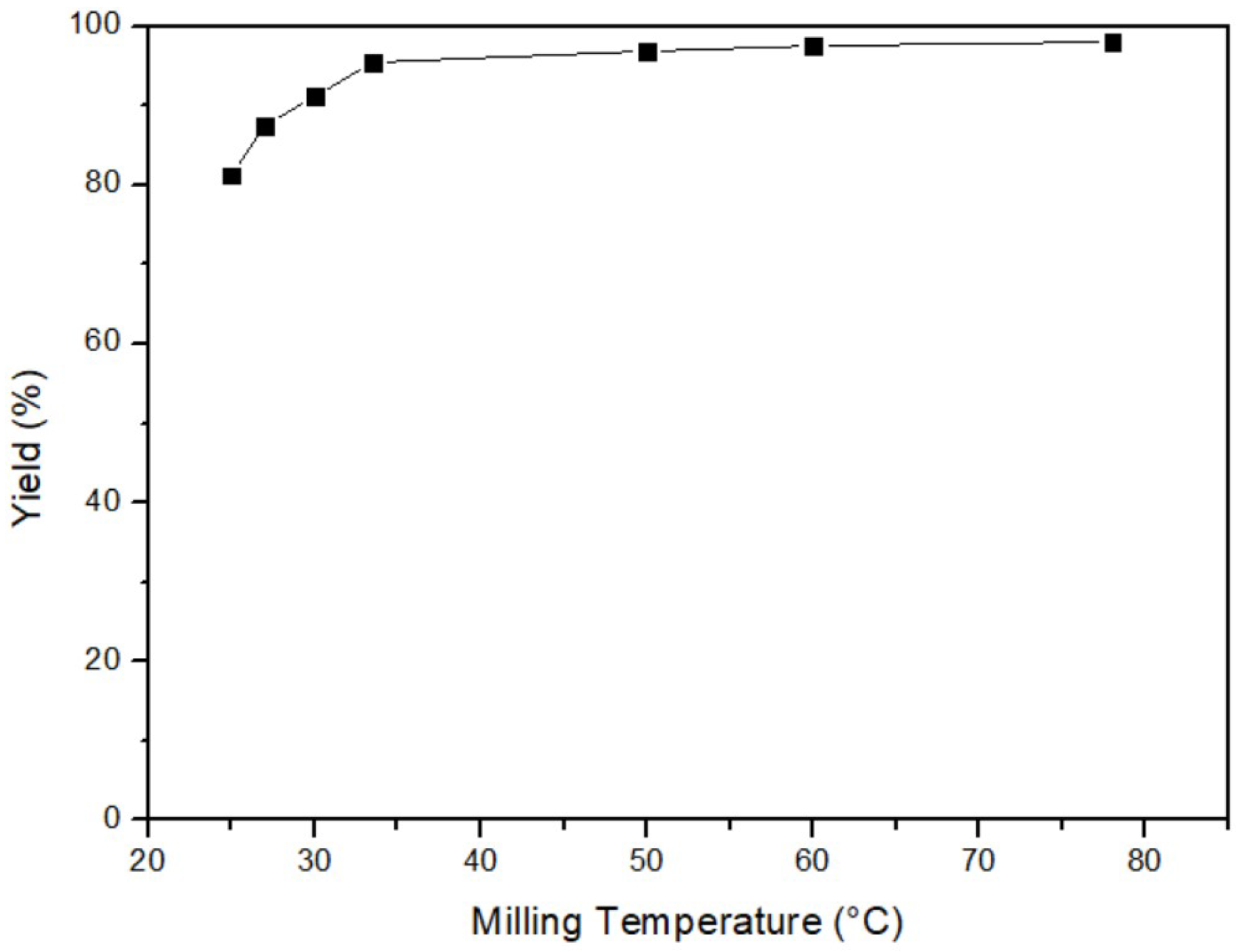
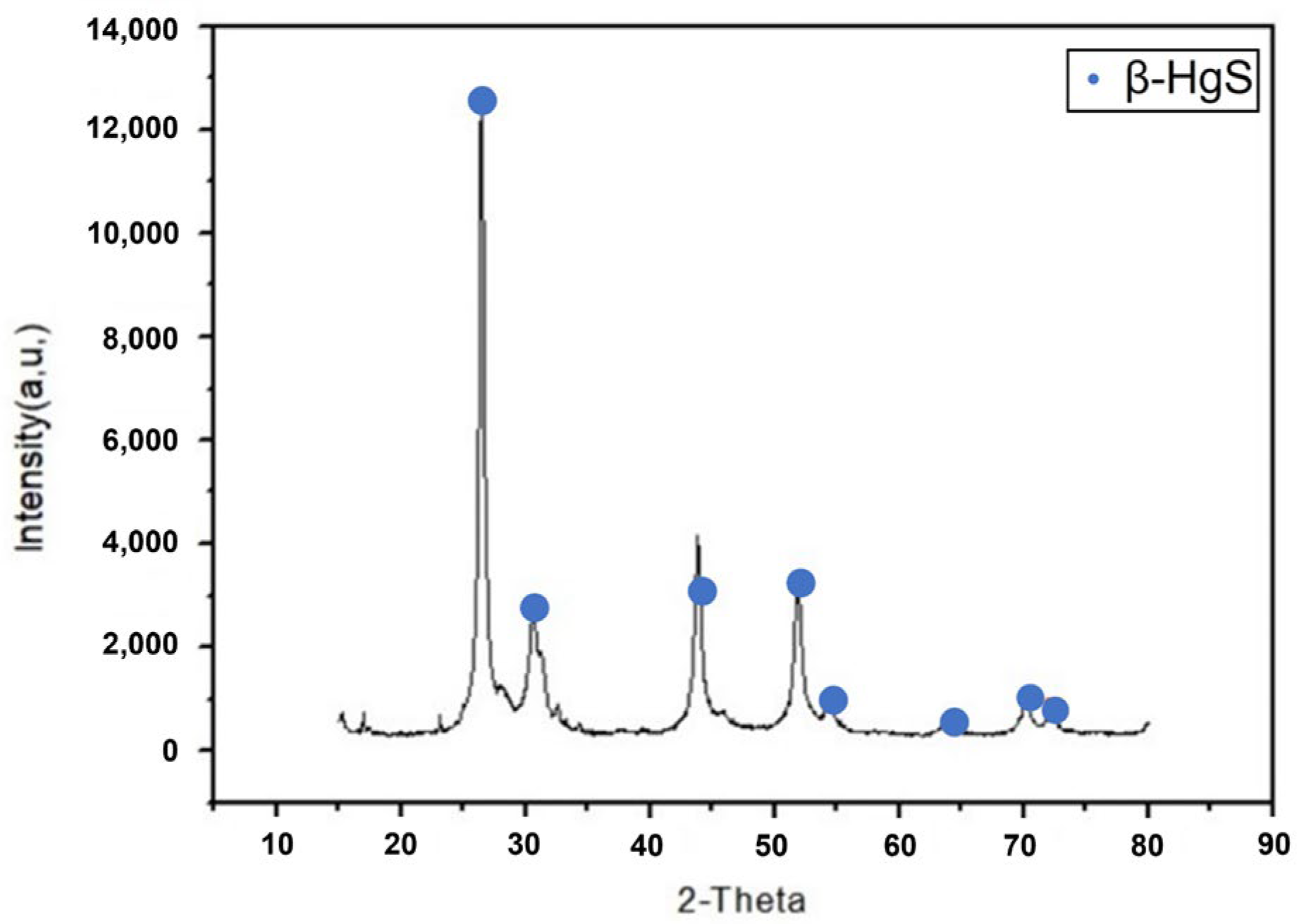
3.1.1. Milling Temperature of Mercury Transfers to β-HgS
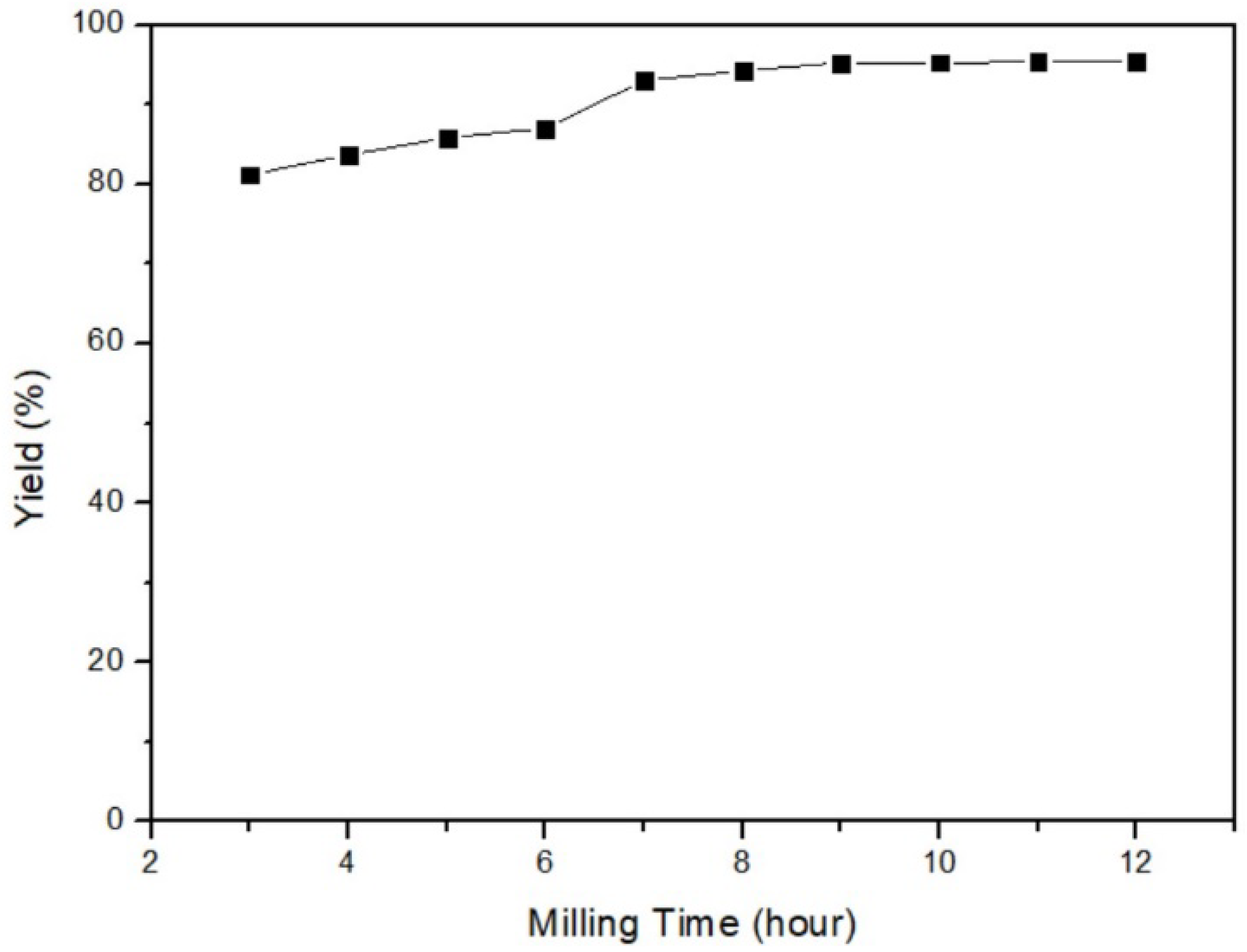
3.1.2. Milling Time of Mercury Transfers to β-HgS
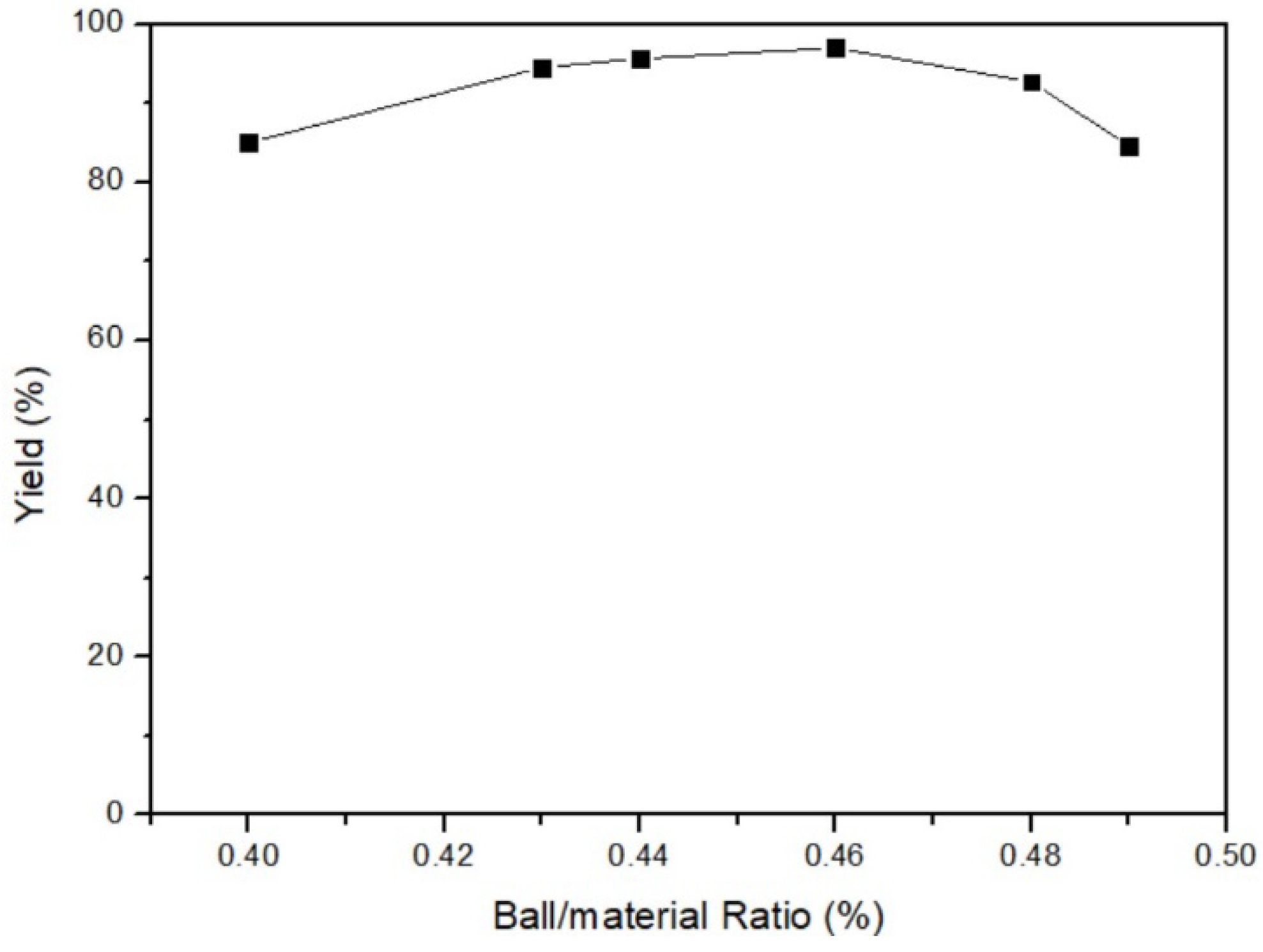
3.1.3. Ball/Material Ratio of Mercury Transfers to β-HgS
3.1.4. Milling Speeds of Mercury Transfers to β-HgS
3.2. Soaking Time of β-HgS Transfers to α-HgS
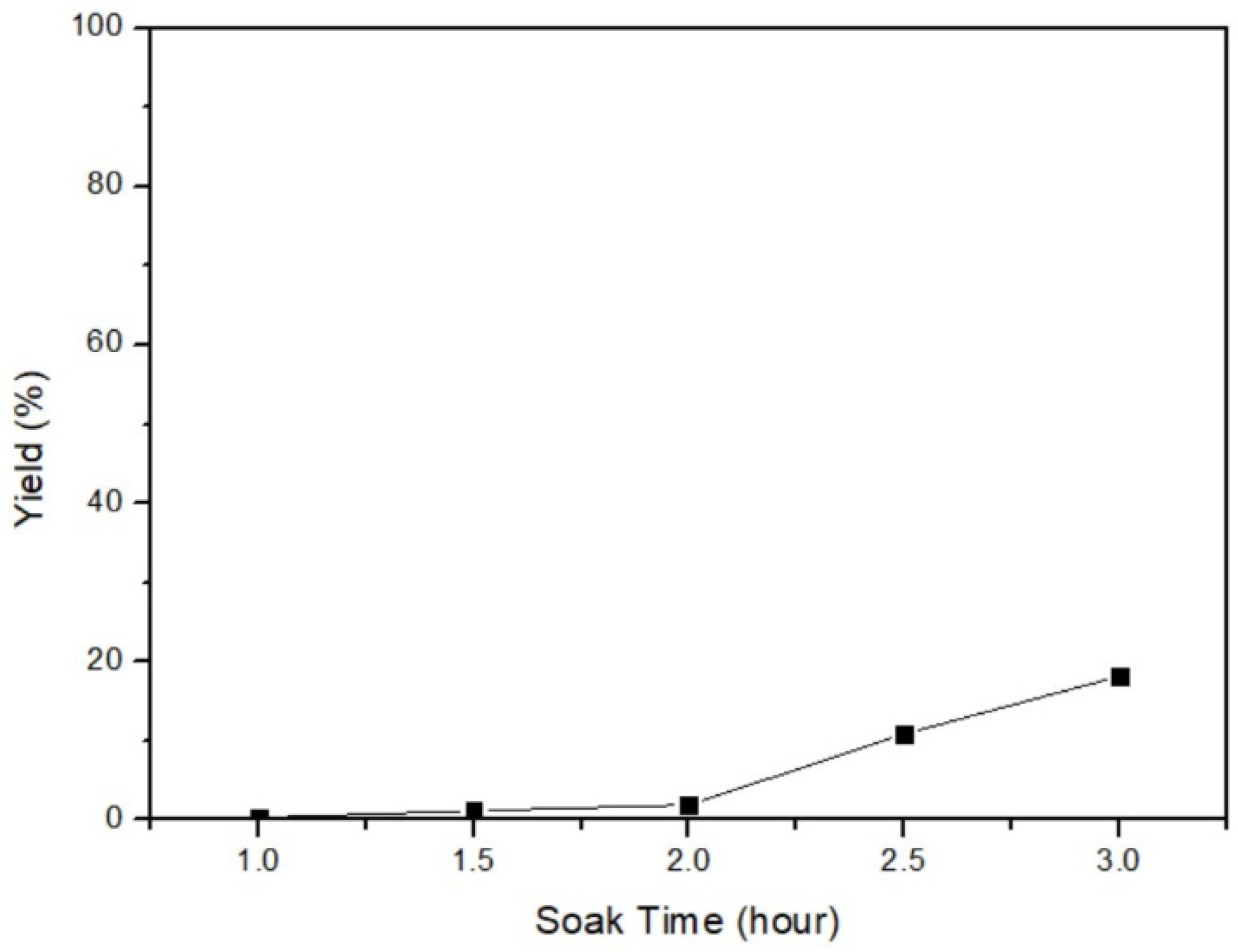
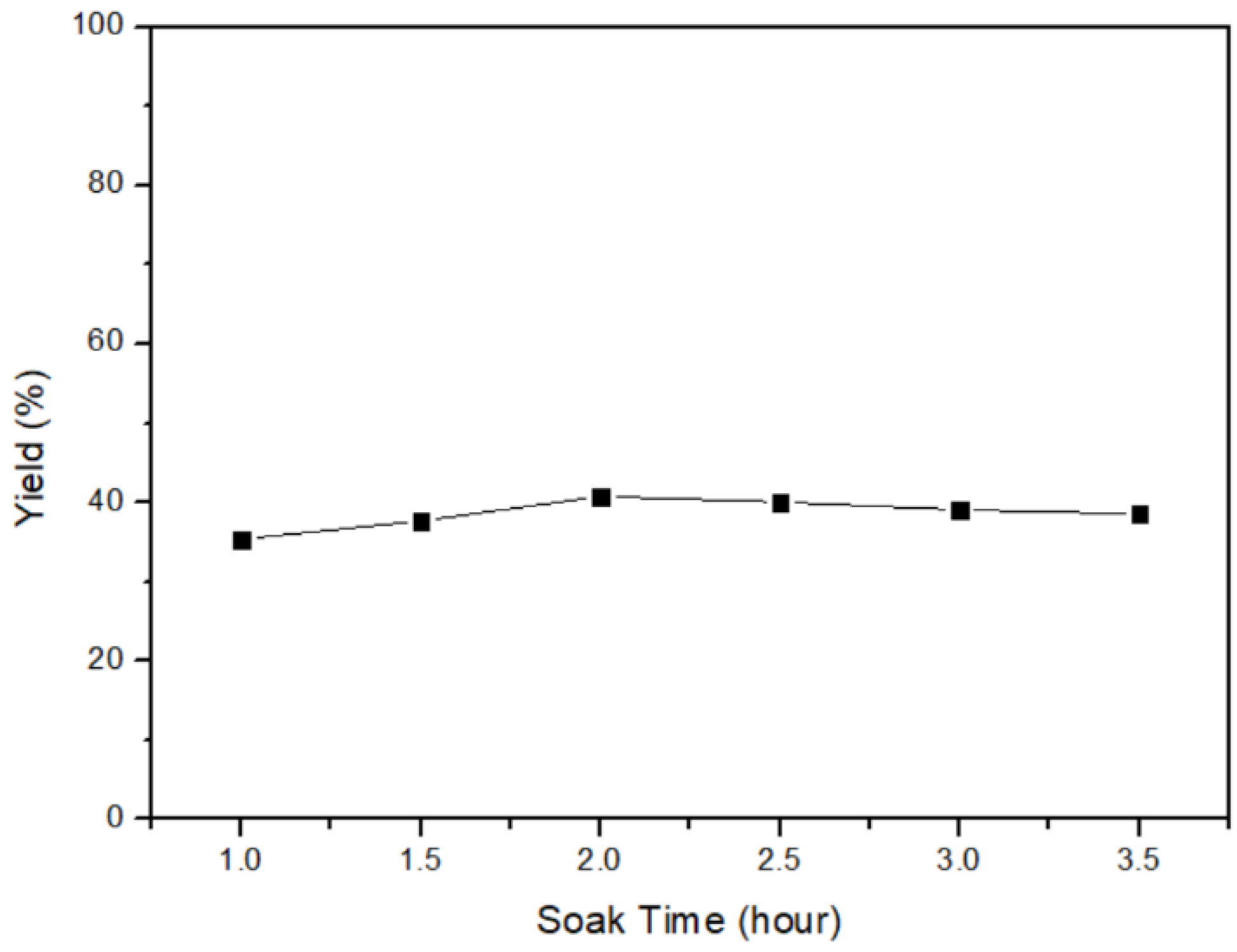
3.3. Soaking Time of α-HgS Transfers to Mercury
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Parsons, M.B.; Percival, J.B. A brief history of mercury and its environmental impact. In Mercury: Sources, Measurements, Cycles, and Effects; Mineralogical Association of Canada: Québec, QC, Canada, 2005; Volume 34, pp. 1–20. [Google Scholar]
- Zhao, M.; Li, Y.; Wang, Z. Mercury and mercury-containing preparations: History of use, clinical applications, pharmacology, toxicology, and pharmacokinetics in traditional Chinese medicine. Front. Pharmacol. 2022, 13, 510. [Google Scholar] [CrossRef]
- Liu, J.; Shi, J.Z.; Yu, L.M.; Goyer, R.A.; Waalkes, M.P. Mercury in traditional medicines: Is cinnabar toxicologically similar to common mercurials? Exp. Biol. Med. 2008, 233, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Kosnett, M.J. The role of chelation in the treatment of arsenic and mercury poisoning. J. Med. Toxicol. 2013, 9, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Skaug, M.A.; Andersen, O.; Aaseth, J. Chelation therapy in intoxications with mercury, lead, and copper. J. Trace Elem. Med. Biol. 2015, 31, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C. Chronic neurological disease due to methylmercury poisoning. Can. J. Neurol. Sci. 2018, 45, 620–623. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.G.; Snider, T.E.; Montiel, M.M. Toxicity of a family from vacuumed mercury. Am. J. Emerg. Med. 1992, 10, 258–261. [Google Scholar] [CrossRef]
- Langford, N.J.; Ferner, R.E. Toxicity of mercury. J. Hum. Hypertens. 1999, 13, 651–656. [Google Scholar] [CrossRef]
- Tsai, W.T. Multimedia Pollution Prevention of Mercury-Containing Waste and Articles: Case Study in Taiwan. Sustainability 2022, 14, 1557. [Google Scholar] [CrossRef]
- Jain, A.; Sarsaiya, S.; Wu, Q.; Shi, J.; Lu, Y. New insights and rethinking of cinnabar for chemical and its pharmacological dynamics. Bioengineered 2019, 10, 353–364. [Google Scholar] [CrossRef]
- Eto, K. Minamata disease. Neuropathology 2000, 20, 14–19. [Google Scholar] [CrossRef]
- Dufault, R.J.; Wolle, M.M.; Kingston, H.S.; Gilbert, S.G.; Murray, J.A. Connecting inorganic mercury and lead measurements in blood to dietary sources of exposure that may impact child development. World J. Methodol. 2021, 11, 144. [Google Scholar] [CrossRef] [PubMed]
- Shah-Kulkarni, S.; Lee, S.; Jeong, K.S.; Hong, Y.-C.; Park, H.; Ha, M.; Kim, Y.; Ha, E.-H. Prenatal exposure to mixtures of heavy metals and neurodevelopment in infants at 6 months. Environ. Res. 2020, 182, 109122. [Google Scholar] [CrossRef] [PubMed]
- Saghazadeh, A.; Rezaei, N. Systematic review and meta-analysis links autism and toxic metals and highlight the impact of country development status: Higher blood and erythrocyte levels for mercury and lead, and higher hair antimony, cadmium, lead, and mercury. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 79, 340–368. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Xun, P.; Liu, K.; Morris, S.; Reis, J.; Guallar, E. Mercury exposure in young adulthood and incidence of diabetes later in life: The CARDIA Trace Element Study. Diabetes Care 2013, 36, 1584–1589. [Google Scholar] [CrossRef]
- Tsai, T.L.; Kuo, C.C.; Pan, W.H.; Wu, T.N.; Lin, P.; Wang, S.L. Type 2 diabetes occurrence and mercury exposure–From the National Nutrition and Health Survey in Taiwan. Environ. Int. 2019, 126, 260–267. [Google Scholar] [CrossRef] [PubMed]
- US EPA. Quality Assurance Guidance Document-Model Quality Assurance Project Plan for the PM Ambient Air; United States Environmental Protection Agency: Washington, DC, USA, 2001; Volume 2, p. 12.
- E.U. Directive. Restriction of the use of certain hazardous substances in electrical and electronic equipment (RoHS). Off. J. Eur. Commun. 2013, 46, 19–23. [Google Scholar]
- Jones, H. EU Bans Mercury in Barometers, and Thermometers. Reuters, 10 July 2007. [Google Scholar]
- Kessler, R. The Minamata Convention on Mercury: A first step toward protecting future generations. Environ. Health Perspect. 2013, 121, 10. [Google Scholar] [CrossRef]
- Chalkidis, A.; Jampaiah, D.; Aryana, A.; Wood, C.; Hartley, P.G.; Sabri, Y.M.; Bhargava, S.K. Mercury-bearing wastes: Sources, policies and treatment technologies for mercury recovery and safe disposal. J. Environ. Manag. 2020, 270, 110945. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, B.J.; Szuszkiewicz, W.; Orlowski, B.A.; He, Z.Q.; Ilver, L.; Kanski, J.; Nilsson, P.O. Photoemission study of β-HgS. J. Electron Spectrosc. Relat. Phenom. 1997, 85, 17–22. [Google Scholar] [CrossRef]
- Fukuda, N.; Takaoka, M.; Oshita, K.; Mizuno, T. Stabilizing conditions of metal mercury in mercury sulfurization using a planetary ball mill. J. Hazard. Mater. 2014, 276, 433–441. [Google Scholar] [CrossRef]
- Cardona, M.; Kremer, R.K.; Lauck, R.; Siegle, G.; Muñoz, A.; Romero, A.H. Electronic, vibrational, and thermodynamic properties of metacinnabar β-HgS, HgSe, and HgTe. Phys. Rev. B 2009, 80, 195204. [Google Scholar] [CrossRef]
- Lynch, A.J.; Rowland, C.A. The History of Grinding; SME: Littleton, CO, USA, 2005. [Google Scholar]
- Gaudin, A.M. Principles of Mineral Dressing (No. 622.7 GAU); McGraw Hill: New York, NY, USA, 1939. [Google Scholar]
- Menczel, J.D.; Prime, R.B. (Eds.) Thermal Analysis of Polymers: Fundamentals and Applications; John Wiley and Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Chen, W.S.; Chen, Y.C.; Lee, C.H. Hydrometallurgical Recovery of Iron, Nickel, and Chromium from Stainless Steel Sludge with Emphasis on Solvent Extraction and Chemical Precipitation. Processes 2022, 10, 748. [Google Scholar] [CrossRef]
- Rayner-Canham, G.W.; Rayner-Canham, G.; Overton, T. Descriptive Inorganic Chemistry; Macmillan: New York, NY, USA, 2003. [Google Scholar]
- Ballirano, P.; Botticelli, M.; Maras, A. Thermal behavior of cinnabar, α-HgS, and the kinetics of the β-HgS (metacinnabar)→ α-HgS conversion at room temperature. Eur. J. Mineral. 2013, 25, 957–965. [Google Scholar] [CrossRef]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Laugier, A.; Garai, J. Derivation of the ideal gas law. J. Chem. Educ. 2007, 84, 1832. [Google Scholar] [CrossRef]
- Hagemann, S. Technologies for the Stabilization of Elemental Mercury and Mercury-Containing Wastes; Gesellschaft für Anlagen-und Reaktorsicherheit (GRS): Köln, Germany, 2009. [Google Scholar]
- Randall, P.; Chattopadhyay, S. Advances in encapsulation technologies for the management of mercury-contaminated hazardous wastes. J. Hazard. Mater. 2004, 114, 211–223. [Google Scholar] [CrossRef] [PubMed]



| Method | Narrative |
|---|---|
| Complexation with sulfur through ball mill | This method can stabilize waste mercury under simple, safe, and low energy consumption situations. However, the yields of mercury products are not high. |
| Hydrometallurgical routes | Hydrometallurgical routes can obtain high yield and purity of mercury products from waste mercury. Nevertheless, chemicals and organic solvents applied during the process may cause secondary pollution. |
| Vacuum distillation and retorting | Vacuum distillation and retorting is a simple method as well. However, the purities of mercury products through this method are not high. |
| Amalgamation | Amalgamation is a simple method that can stabilize mercury with other metals. However, the risks of volatilization or leaching are not eliminated, so it still needs to combine with other methods. |
| Solidification [33,34] | The process of solidification involves immobilizing mercury physically by trapping it within an impermeable substance like Portland-based cements. To achieve this, a stabilizing pretreatment step, such as using HgS, is typically necessary because there is no chemical interaction between the encasing material and mercury. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.-S.; Chi, C.-C.; Lee, C.-H. Stabilization of Waste Mercury with Sulfide through the Ball-Mill Method and Heat Treatment. Sustainability 2023, 15, 10333. https://doi.org/10.3390/su151310333
Chen W-S, Chi C-C, Lee C-H. Stabilization of Waste Mercury with Sulfide through the Ball-Mill Method and Heat Treatment. Sustainability. 2023; 15(13):10333. https://doi.org/10.3390/su151310333
Chicago/Turabian StyleChen, Wei-Sheng, Chien-Ching Chi, and Cheng-Han Lee. 2023. "Stabilization of Waste Mercury with Sulfide through the Ball-Mill Method and Heat Treatment" Sustainability 15, no. 13: 10333. https://doi.org/10.3390/su151310333
APA StyleChen, W.-S., Chi, C.-C., & Lee, C.-H. (2023). Stabilization of Waste Mercury with Sulfide through the Ball-Mill Method and Heat Treatment. Sustainability, 15(13), 10333. https://doi.org/10.3390/su151310333








