Towards Balanced Fertilizer Management in South China: Enhancing Wax Gourd (Benincasa hispida) Yield and Produce Quality
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Experimental Design
2.3. Sampling and Measurements
2.4. Statistical Analyses
3. Results
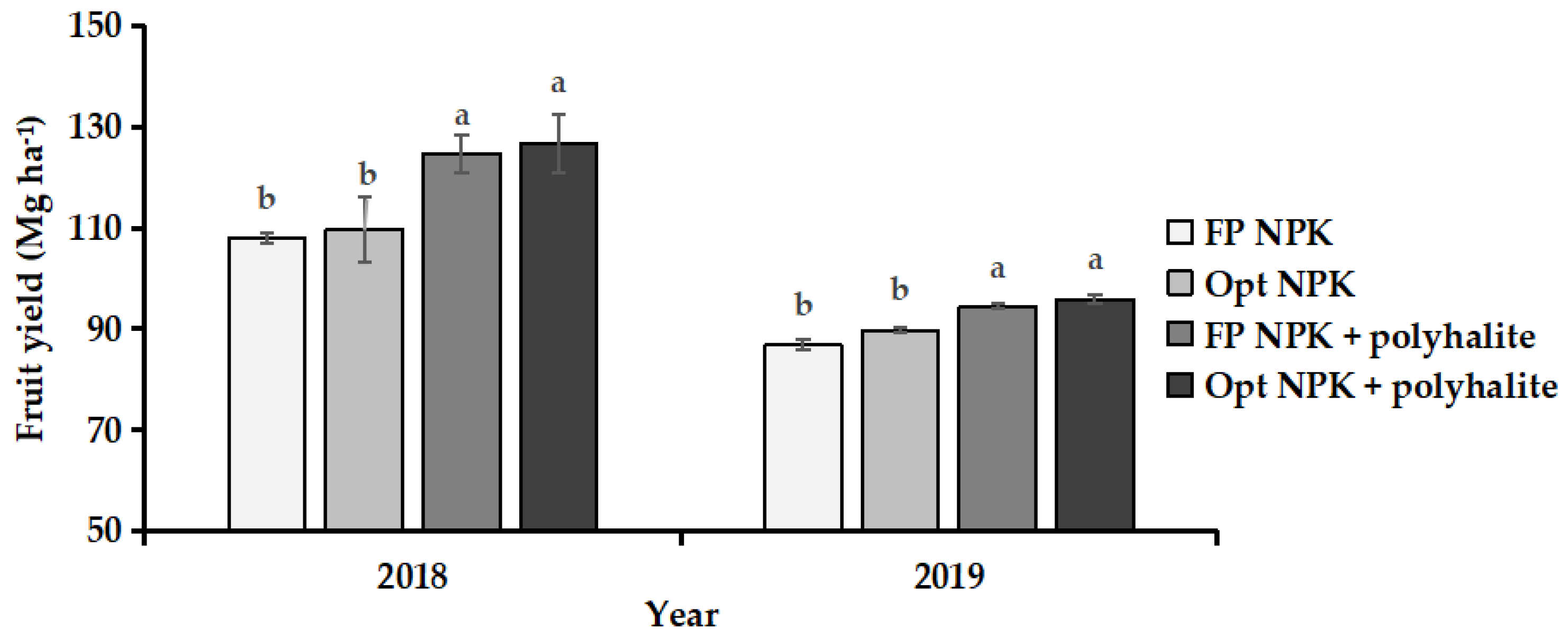
3.1. Fruit Yield
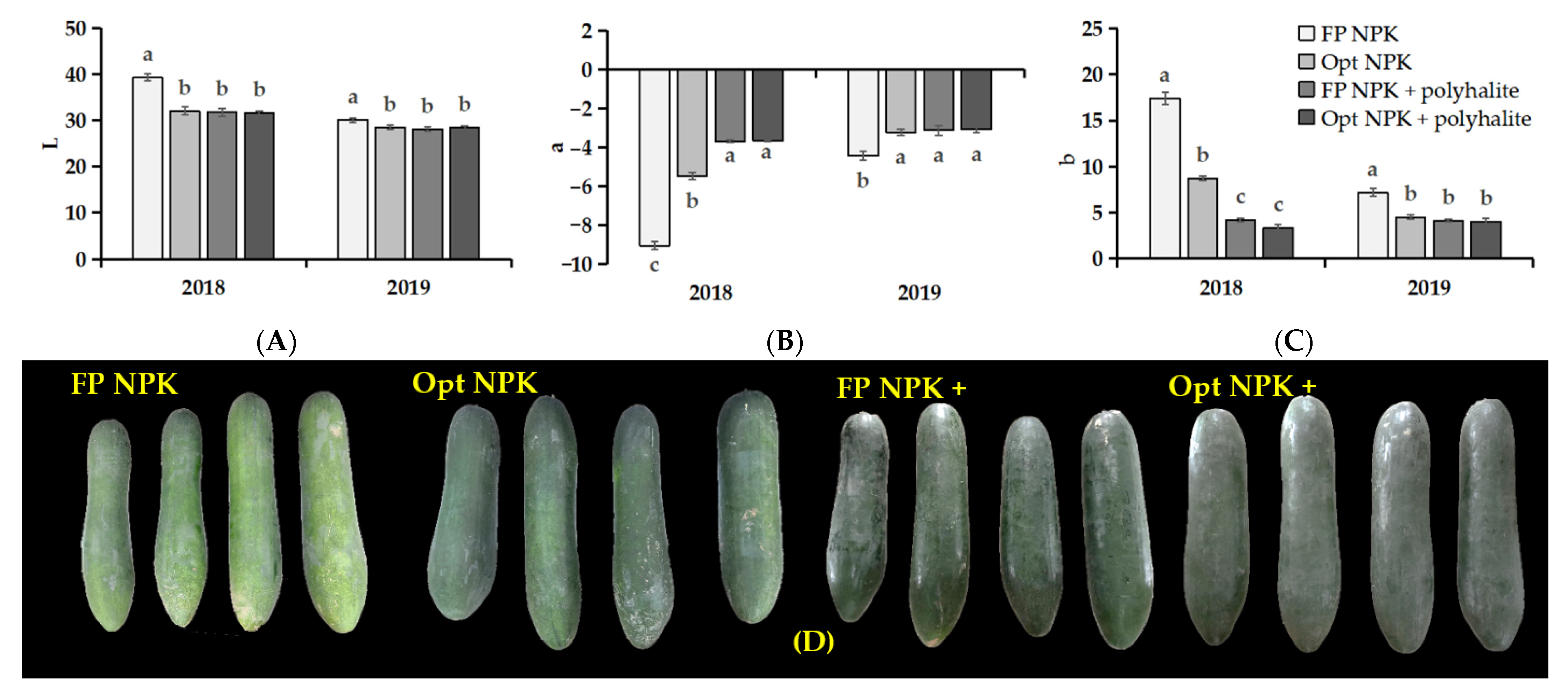
3.2. Fruit Quality
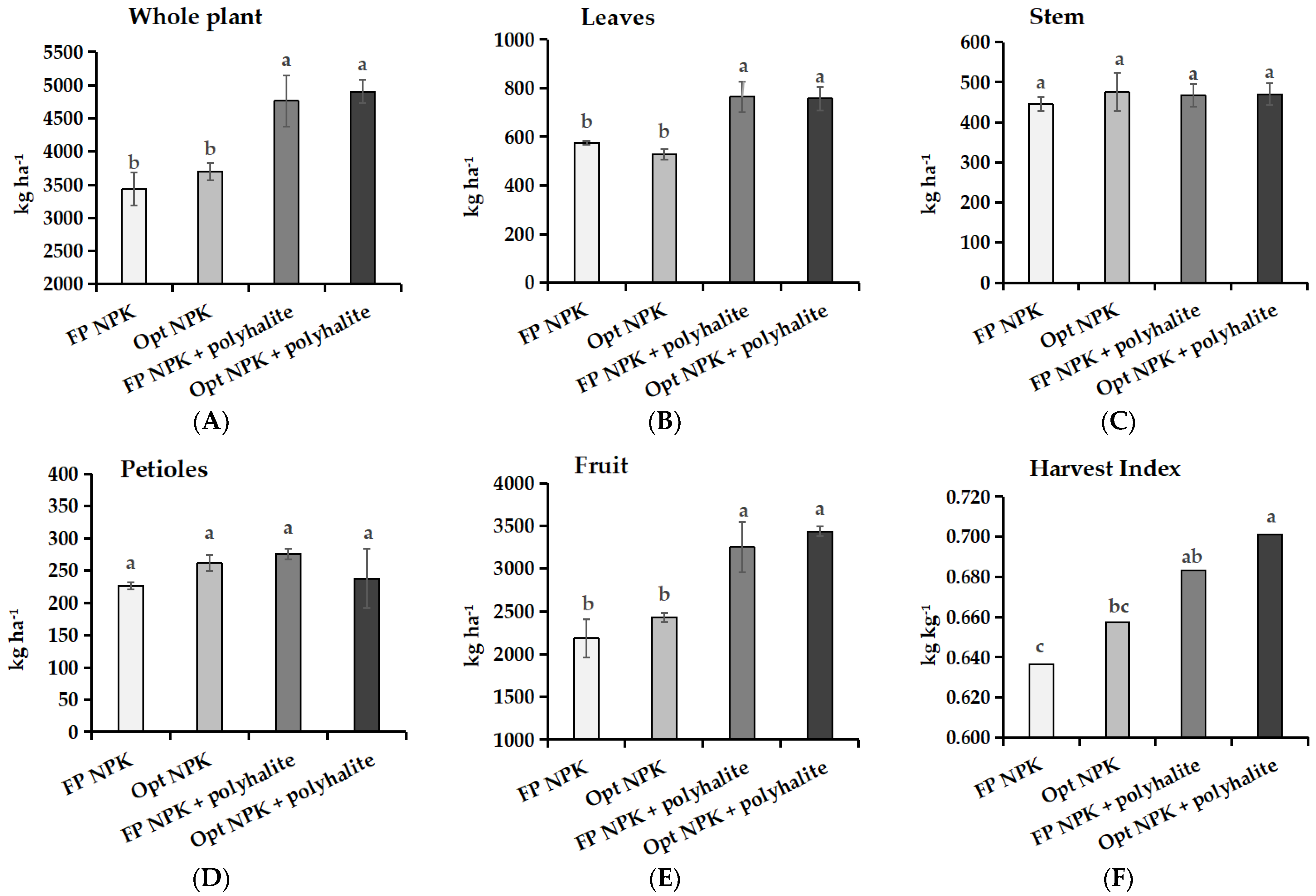
3.3. Crop Aboveground Biomass and Dry Matter Partitioning
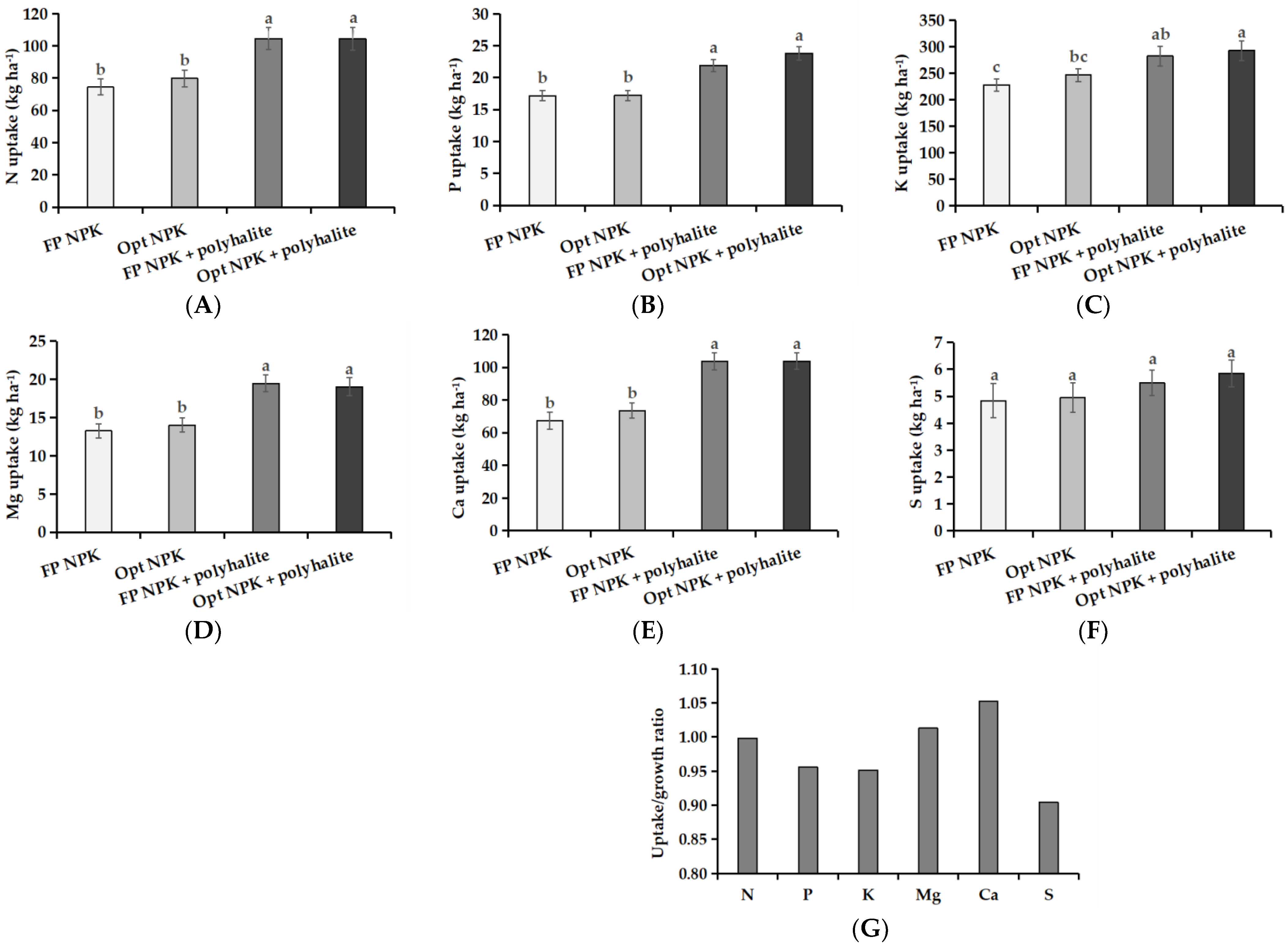
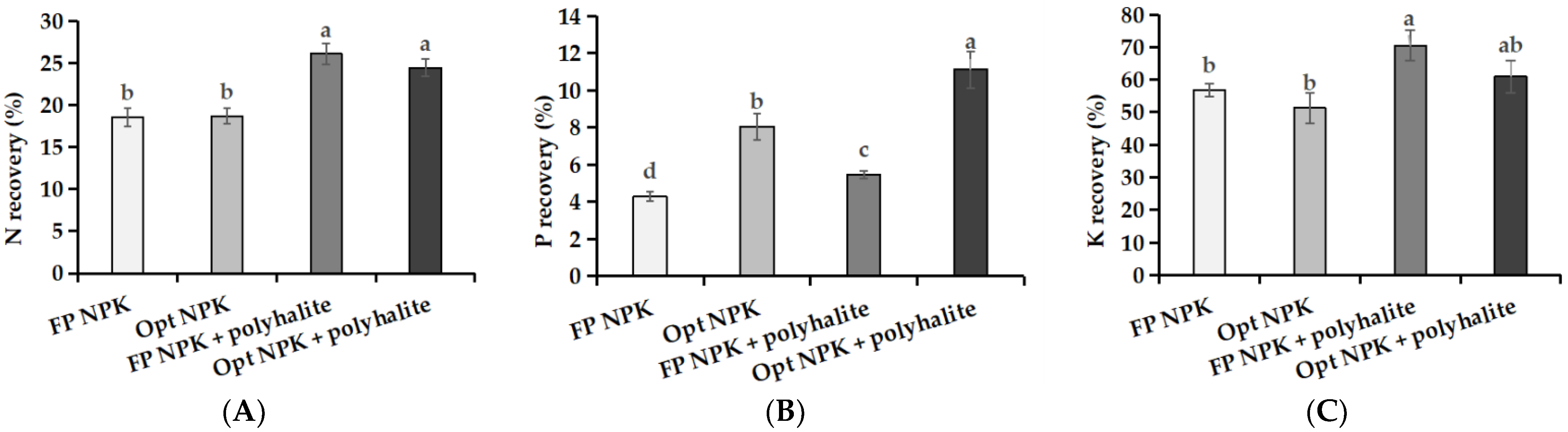
3.4. Nutrient Uptake and Recovery
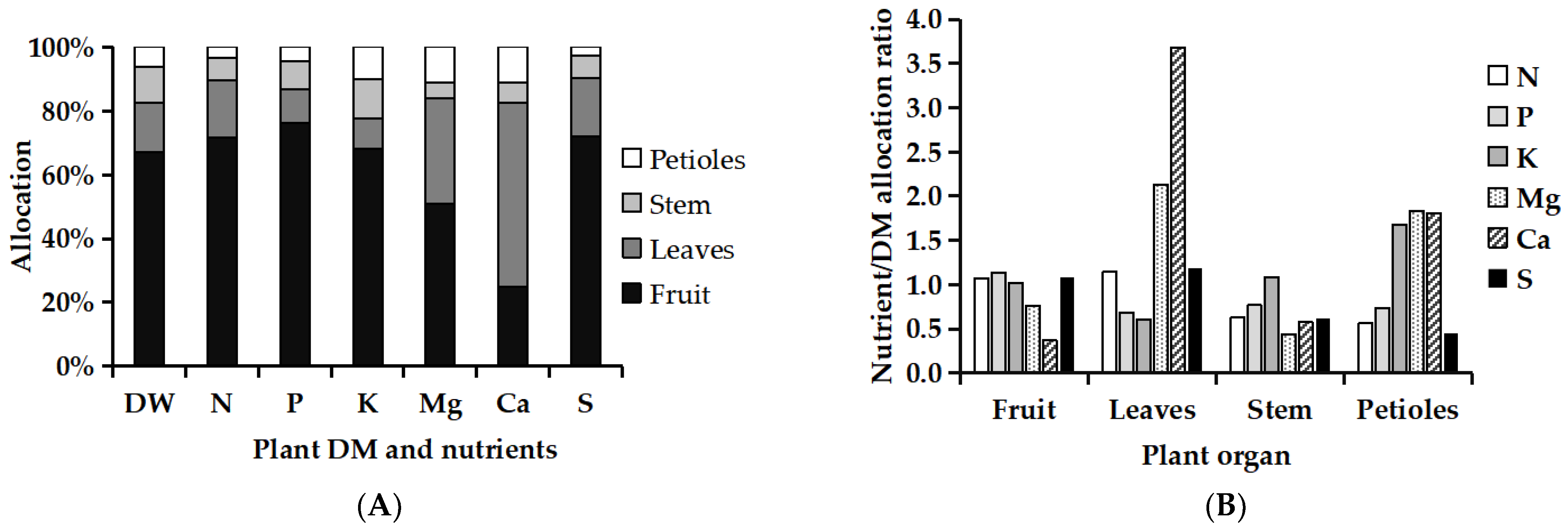
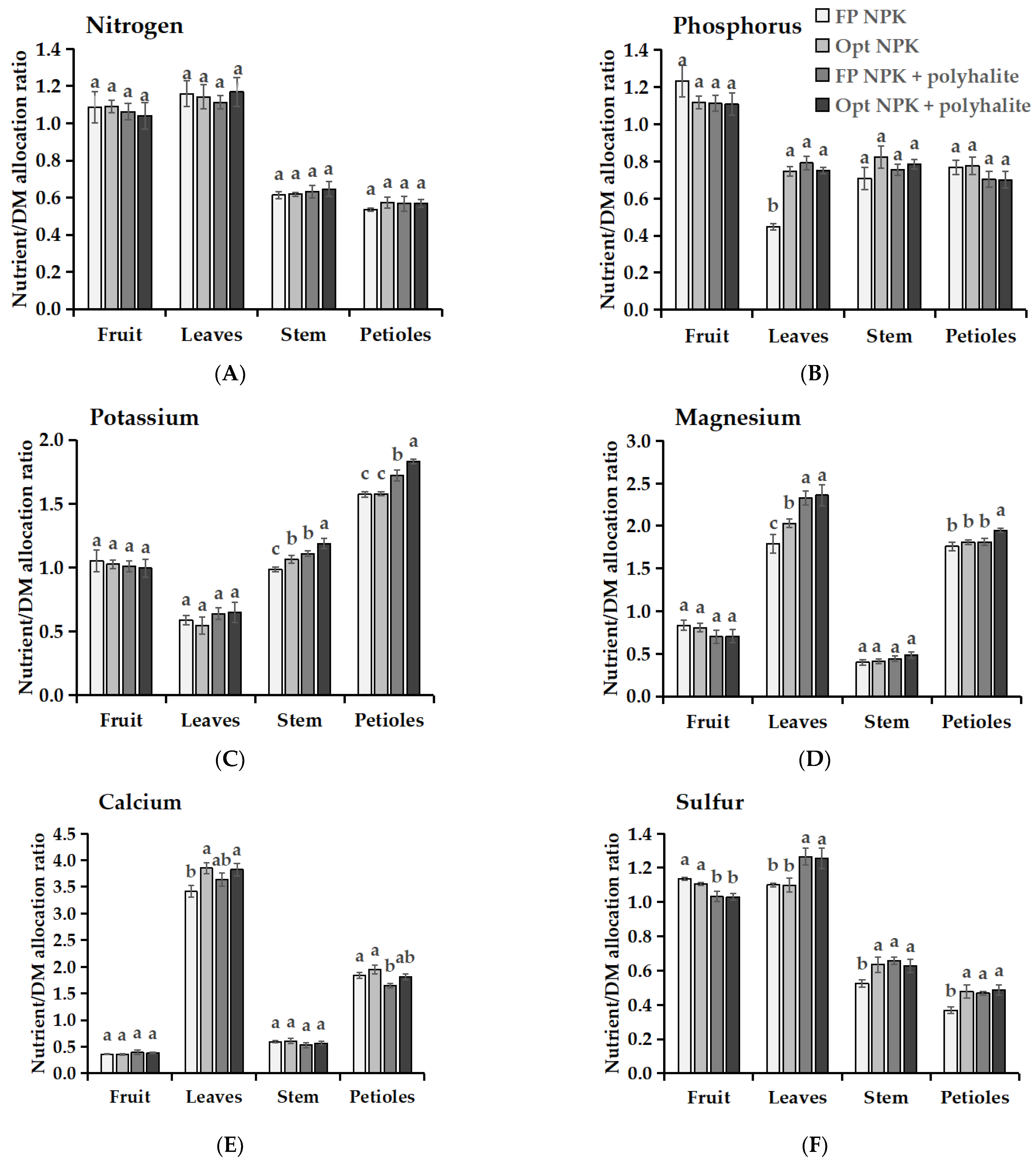
3.5. Nutrient Allocation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zaini, N.A.M.; Anwar, F.; Hamid, A.A.; Saari, N. Kundur [Benincasa hispida (Thunb.) Cogn.]: A potential source for valuable nutrients and functional foods. Food Res. Int. 2011, 44, 2368–2376. [Google Scholar] [CrossRef]
- Nakashima, M.; Shigekuni, Y.; Obi, T.; Shiraishi, M.; Miyamoto, A.; Yamasaki, H.; Etoh, T.; Iwai, S. Nitric oxide-dependent hypotensive effects of wax gourd juice. J. Ethnopharmacol. 2011, 138, 404–407. [Google Scholar] [CrossRef] [PubMed]
- Al-Snafi, A.E. The pharmacological importance of Benincasa hispida. A review. Int. J. Pharma Sci. Res. 2013, 4, 165–170. [Google Scholar]
- Dhillon, N.P.; Sanguansil, S.; Singh, S.P.; Masud, M.A.T.; Kumar, P.; Bharathi, L.K.; McCreight, J.D. Gourds: Bitter, bottle, wax, snake, sponge and ridge. In Genetics and Genomics of Cucurbitaceae; Springer: Cham, Switzerland, 2016; pp. 155–172. [Google Scholar]
- Wu, Z.F.; Sun, L.; Zhang, X.; Shen, X.Q.; Weng, P.F. Quantitative analysis of predominant yeasts and volatile compounds in the process of pickled wax gourd. CyTA-J. Food 2016, 14, 92–100. [Google Scholar] [CrossRef]
- Xie, D.; Xu, Y.; Wang, J.; Zhou, Q.; Luo, S.B.; Huang, W.; He, X.M.; Li, Q.; Peng, Q.; Yang, X.; et al. The wax gourd genomes offer insights into the genetic diversity and ancestral cucurbit karyotype. Nat. Commun. 2019, 10, 5158. [Google Scholar] [CrossRef]
- Palamthodi, S.; Lele, S.S. Nutraceutical applications of gourd family vegetables: Benincasa hispida, Lagenaria siceraria and Momordica charantia. Biomed. Prev. Nutr. 2014, 4, 15–21. [Google Scholar] [CrossRef]
- Liu, W.; Jiang, B.; Peng, Q.; He, X.; Lin, Y.E.; Wang, M.; Liang, Z.; Xie, D.; Hu, K. Genetic analysis and QTL mapping of fruit-related traits in wax gourd (Benincasa hispida). Euphytica 2018, 214, 136. [Google Scholar] [CrossRef]
- Lampc, J.W. Health effects of vegetables and fruit: Assessing mechanisms of action in human esperimental studies. Am. J. Clin. Nutr. 1999, 70, 475–490. [Google Scholar] [CrossRef]
- Li, C. Status of Black Wax Gourd Production and Fertilization Technology for High Quality and High Yield in Sanshui District, Foshan, Guangdong Province. Master’s Thesis, China Agricultural University, Beijing, China, 2019. (In Chinese). [Google Scholar]
- Van Bueren, E.T.L.; Struik, P.C. Diverse concepts of breeding for nitrogen use efficiency. A review. Agron. Sustain. Dev. 2017, 37, 37–50. [Google Scholar]
- Hawkesford, M.; Horst, W.; Kichey, T.; Lambers, H.; Schjoerring, J.; Møller, I.S.; White, P. Functions of macronutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Marschner, P., Ed.; Academic Press: Cambridge, UK, 2012; pp. 135–189. [Google Scholar]
- Roussis, I.; Kakabouki, I.; Beslemes, D.; Tigka, E.; Kosma, C.; Triantafyllidis, V.; Mavroeidis, A.; Zotos, A.; Bilalis, D. Nitrogen Uptake, Use Efficiency, and Productivity of Nigella sativa L. in Response to Fertilization and Plant Density. Sustainability 2022, 14, 3842. [Google Scholar] [CrossRef]
- Cramer, M.D. Phosphate as a limiting resource: Introduction. Plant Soil 2010, 334, 1–10. [Google Scholar] [CrossRef]
- Martuscelli, M.; Di Mattia, C.; Stagnari, F.; Speca, S.; Pisante, M.; Mastrocola, D. Influence of phosphorus management on melon (Cucumis melo L.) fruit quality. J. Sci. Food Agric. 2016, 96, 2715–2722. [Google Scholar] [CrossRef] [PubMed]
- Zörb, C.; Senbayram, M.; Peiter, E. Potassium in agriculture–status and perspectives. Plant Physiol. 2014, 171, 656–669. [Google Scholar] [CrossRef] [PubMed]
- Mengel, K. Potassium. In Handbook of Plant Nutrition; CRC Press Inc.: Boca Raton, FL, USA, 2016; pp. 107–136. [Google Scholar]
- Lester, G.E.; Jifon, J.L.; Makus, D.J. Impact of potassium nutrition on postharvest fruit quality: Melon (Cucumis Melo L.) case study. Plant Soil 2010, 335, 117–331. [Google Scholar] [CrossRef]
- Deus, J.A.L.D.; Soares, I.; Neves, J.C.L.; Medeiros, J.F.D.; Miranda, F.R.D. Fertilizer recommendation system for melon based on nutritional balance. Rev. Bras. Ciênc. Solo 2015, 39, 498–511. [Google Scholar] [CrossRef][Green Version]
- Chen, Y.; Zhou, X.; Lin, Y.; Ma, L. Pumpkin yield affected by soil nutrients and the interactions of nitrogen, phosphorus, and potassium fertilizers. HortScience 2019, 54, 1831–1835. [Google Scholar] [CrossRef]
- Cakmak, I.; Hengeler, C.; Marschner, H. Changes in phloem export of sucrose in leaves in response to phosphorus, potassium and Mg deficiency in bean plants. J. Exp. Bot. 1994, 45, 1251–1257. [Google Scholar] [CrossRef]
- Ferrante, A.; Spinardi, A.; Maggiore, T.; Testoni, A.; Gallina, P.M. Effect of nitrogen fertilisation levels on melon fruit quality at the harvest time and during storage. J. Sci. Food Agric. 2008, 88, 707–713. [Google Scholar] [CrossRef]
- Shen, C.; Wang, J.; Shi, X.; Kang, Y.; Xie, C.; Peng, L.; Dong, C.; Shen, Q.; Xu, Y. Transcriptome analysis of differentially expressed genes induced by low and high potassium levels provides insight into fruit sugar metabolism of pear. Front. Plant Sci. 2017, 8, 938. [Google Scholar] [CrossRef]
- Tränkner, M.; Tavakol, E.; Jákli, B. Functioning of potassium and magnesium in photosynthesis, photosynthate translocation and photoprotection. Physiol. Plant. 2018, 163, 414–431. [Google Scholar] [CrossRef]
- Wang, R.; Min, J.; Kronzucker, H.J.; Li, Y.; Shi, W. N and P runoff losses in China’s vegetable production systems: Loss characteristics, impact and management practices. Sci. Total Environ. 2019, 663, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Song, X.Z.; Zhao, C.X.; Wang, X.L.; Li, J. Study of nitrate leaching and nitrogen fate under intensive vegetable production pattern in northern China. Comptes Rendus. Biol. 2009, 332, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Liu, P.; Li, Y.; Ma, L.; Alva, A.; Dou, Z.; Chen, Q.; Zhang, F. Phosphorus in China’s intensive vegetable production systems: Over-fertilization, soil enrichment, and environmental implications. J. Environ. Qual. 2013, 42, 982. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Zhou, Y.J.; Shao, Q.X.; Liu, H.B.; Lei, Q.L.; Zhai, X.Y.; Wang, X.L. Diffuse nutrient losses and the impact factors determining their regional differences in four catchments from North to South China. J. Hydrol. 2016, 543, 577–594. [Google Scholar] [CrossRef]
- Zhang, B.; Li, Q.; Cao, J.; Zhang, C.; Song, Z.; Zhang, F.; Chen, X. Reducing nitrogen leaching in a subtropical vegetable system. Agric. Ecosyst. Environ. 2017, 241, 133–141. [Google Scholar] [CrossRef]
- Yuan, Z.W.; Jiang, S.Y.; Sheng, H.; Liu, X.; Hua, H.; Liu, X.W.; Zhang, Y. Human Perturbation of the Global Phosphorus Cycle:Changes and Consequences. Environ. Sci. Technol. 2018, 52, 2438–2450. [Google Scholar] [CrossRef]
- Liu, X.; Sheng, H.; Jiang, S.Y. IIntensification of phosphorus cycling in China since the 1600s. Proc. Natl. Acad. Sci. USA 2016, 113, 2609–2614. [Google Scholar] [CrossRef]
- West, J.; Zhang, Y.; Smith, S.; Silva, R.; Lamarque, J.F. Co-benefits of global and domestic greenhouse gas emissions for air quality and human health. Lancet 2017, 389, S23. [Google Scholar] [CrossRef]
- Hermans, C.; Bourgis, F.; Faucher, M.; Strasser, R.J.; Delrot, S.; Verbruggen, N. Magnesium deficiency in sugar beets alters sugar partitioning and phloem loading in young mature leaves. Planta 2005, 220, 541–549. [Google Scholar] [CrossRef]
- Römheld, V.; Kirkby, E.A. Magnesium functions in crop nutrition and yield. Nawozy Nawożenie Fertil. Fertil. 2009, 34, 163–182. [Google Scholar]
- Senbayram, M.; Gransee, A.; Wahle, V.; Thiel, H. Role of magnesium fertilisers in agriculture: Plant–soil continuum. Crop Pasture Sci. 2009, 66, 1219–1229. [Google Scholar] [CrossRef]
- Moss, G.I.; Higgins, M.L. Magnesium influences on the fruit quality of sweet orange (Citrus sinensis L. osbeck). Plant Soil 1974, 41, 103–112. [Google Scholar] [CrossRef]
- Ding, Y.; Luo, W.; Xu, G. Characterization of magnesium nutrition and interaction of magnesium and potassium in rice. Ann. Appl. Biol. 2006, 149, 111–123. [Google Scholar] [CrossRef]
- Cakmak, I. Magnesium in crop production, food quality and human health. Plant Soil 2013, 368, 1–4. [Google Scholar] [CrossRef]
- Gerendás, J.; Führs, H. The significance of magnesium for crop quality. Plant Soil 2013, 368, 101–128. [Google Scholar] [CrossRef]
- Stael, S.; Wurzinger, B.; Mair, A.; Mehlmer, N.; Vothknecht, U.C.; Teige, M. Plant organellar calcium signalling: An emerging field. J. Exp. Bot. 2012, 63, 1525–1542. [Google Scholar] [CrossRef]
- Hocking, B.; Tyerman, S.D.; Burton, R.A.; Gilliham, M. Fruit calcium: Transport and physiology. Front. Plant Sci. 2016, 7, 569. [Google Scholar] [CrossRef]
- Fallahi, E.; Conway, W.S.; Hickey, K.D.; Sams, C.E. The role of calcium and nitrogen in postharvest quality and disease resistance of apples. HortScience 1997, 32, 831–835. [Google Scholar] [CrossRef]
- Manganaris, G.A.; Vasilakakis, M.; Mignani, I.; Diamantidis, G.; Tzavella-Klonari, K. The effect of preharvest calcium sprays on quality attributes, physicochemical aspects of cell wall components, and susceptibility to brown rot of peach fruits (Prunus persica L. cv. Andross). Sci. Hortic. 2005, 107, 43–50. [Google Scholar] [CrossRef]
- Bouzo, C.A.; Céccoli, G.; Muñoz, F. Effect of potassium and calcium upon the yield and fruit quality of Cucumis melo. AgriScientia 2018, 35, 25–33. [Google Scholar] [CrossRef]
- Brosnan, J.T.; Brosnan, M.E. The sulfur-containing amino acids: An overview. J. Nutr. 2006, 136, 16365–16405. [Google Scholar] [CrossRef] [PubMed]
- Nira, M.; Powers, S.J.; Stephen, E.J.; Mottram, D.S.; Halford, N.G. Effects of nitrogen and sulfur fertilization on free amino acids, sugars, and acrylamide-forming potential in potato. J. Agric. Food Chem. 2013, 61, 6734–6742. [Google Scholar]
- Dai, Z.; Plessis, A.; Vincent, J.; Duchateau, N.; Besson, A.; Dardevet, M.; Prodhomme, D.; Gibon, Y.; Hilbert, G.; Pailloux, M.; et al. Transcriptional and metabolic alternations rebalance wheat grain storage protein accumulation under variable nitrogen and sulfur supply. Plant J. 2015, 83, 326–343. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Pan, X.; Li, J. Current agricultural practices threaten future global food production. J. Agric. Environ. Ethics 2015, 28, 203–216. [Google Scholar] [CrossRef]
- Chen, X.; Cui, Z.; Fan, M.; Vitousek, P.; Zhao, M.; Ma, W.; Wang, Z.; Zhang, W.; Yan, X.; Yang, J.; et al. Producing more grain with lower environmental costs. Nature 2014, 514, 486–489. [Google Scholar] [CrossRef]
- Khan, M.A.; Kim, K.W.; Wang, M.; Lim, B.K.; Lee, W.H.; Lee, J.Y. Nutrient-impregnated charcoal: An environmentally friendly slow-release fertilizer. Environmentalist 2008, 28, 231–235. [Google Scholar] [CrossRef]
- Bar-Yosef, B. Advances in fertigation. Adv. Agron. 1999, 65, 1–77. [Google Scholar]
- Vale, F. Ca and Mg movement in soil profile with polysulphate as potassium fertilizer for soybean crop. In Proceedings of the FERTBIO, Goiana, Brazil, 16–20 October 2016; pp. 16–20. [Google Scholar]
- Yermiyahu, U.; Zipori, I.; Faingold, I.; Yusopov, L.; Faust, N.; Bar-Tal, A. Polyhalite as a multi nutrient fertilizer–potassium, magnesium, calcium and sulfate. Isr. J. Plant Sci. 2017, 64, 145–157. [Google Scholar]
- Yermiyahu, U.; Zipori, I.; Omer, C.; Beer, Y. Solubility of granular polyhalite under laboratory and field conditions. Int. Potash Inst. 2019, 58, 3–9. [Google Scholar]
- Bernardi, A.C.C.; de Souza, G.B.; Vale, F. Polyhalite compared to KCl and gypsum in alfalfa fertilization. Int. Potash Inst. 2018, 52, 3–9. [Google Scholar]
- Satisha, G.C.; Ganeshamurthy, A.N. Bioefficacy of polysulphate application on yield and quality of cabbage and cauliflower. Int. Potash Inst. 2016, 44, 21–31. [Google Scholar]
- Zhou, Z.; Chen, K.; Yu, H.; Chen, Q.; Wu, F.; Zeng, X.; Fan, X. Changes in tea performance and soil properties after three years of polyhalite application. Agron. J. 2019, 111, 1967–1976. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, J.; Wang, J.; Zou, Z.; Zhao, Z. Effects of ventilation, nitrogen and potassium on growth and quality of melon in solar greenhouse. J. Northwest A F Univ.-Nat. Sci. Ed. 2010, 38, 117–122. [Google Scholar]
- Sadler, G.D.; Murphy, P.A. pH and titratable acidity. In Food Analysis; Springer: Boston, MA, USA, 2010; pp. 219–238. [Google Scholar]
- Hansen, J.; Møller, I.B. Percolation of starch and soluble carbohydrates from plant tissue for quantitative determination with anthrone. Anal. Biochem. 1975, 68, 87–94. [Google Scholar] [CrossRef]
- McGuire, R.G. Reporting of objective color measurements. HortScience 1992, 27, 1254–1255. [Google Scholar] [CrossRef]
- Khoshnevisan, B.; Rafiee, S.; Omid, M.; Mousadeh, H.; Clark, S. Environmental impact assessment of tomato and cucumber cultivation in greenhouses using life cycle assessment and adaptive neuro-fuzzy inference system. J. Clean. Prod. 2014, 73, 183–192. [Google Scholar] [CrossRef]
- Nachshon, U. Cropland soil salinization and associated hydrology: Trends, processes and examples. Water 2018, 10, 1030. [Google Scholar]
- McGrath, S.P.; Zhao, F.J. Sulphur uptake, yield response and the interactions between N and S in winter oilseed rape (Brassica napus). J. Agric. Sci. 1996, 126, 53–62. [Google Scholar] [CrossRef]
- Kopriva, S.; Suter, M.; Ballmoos, P.V.; Hesse, H.; Krahenbuhl, U.; Rennenberg, H.; Brunold, C. Interaction of sulphate assimilation with carbon and nitrogen metabolism in Lemna minor. Plant Physiol. 2002, 130, 1406–1413. [Google Scholar] [CrossRef]
- Hu, C.; Ham, B.K.; El-shabrawi, H.M.; Alexander, D.; Zhang, D.; Ryals, J.; Lucas, W.J. Proteomics and metabolomics analyses reveal the cucurbit sieve tube system as a complex metabolic space. Plant J. 2016, 87, 442–454. [Google Scholar] [CrossRef]
- Willumsen, J.; Petersen, K.K.; Kaack, K. Yield and blossom-end rot of tomato as affected by salinity and cation activity ratios in the root zone. J. Hortic. Sci. 1996, 71, 81–98. [Google Scholar] [CrossRef]
- Dilmaghani, M.R.; Malakouti, M.J.; Neilsen, G.H.; Fallahi, E. Interactive effects of potassium and calcium on K/Ca Ratio and its consequences on apple fruit quality in calcareous soils of Iran. J. Plant Nutr. 2005, 27, 1149–1162. [Google Scholar] [CrossRef]
- Sharples, R.O. The influence of preharvest conditions on the quality of stored fruits. Acta Hortic. 1985, 157, 93–104. [Google Scholar] [CrossRef]
- Kondo, T.; Higuchi, H. Acidity of passion fruit as affected by potassium fertilizer. Acta Hortic. 2013, 984, 385–391. [Google Scholar] [CrossRef]
- Tanoi, K.; Kobayashi, N. Leaf senescence by magnesium deficiency. Plants 2015, 4, 756–772. [Google Scholar] [CrossRef]
| Soil Property | 2018 | 2019 |
|---|---|---|
| pH | 6.3 | 6.2 |
| Ammonium N (mg kg−1) | 0.01 | 0.01 |
| Nitrate N (mg kg−1) | 32.3 | 31.2 |
| P (Olsen) (mg kg−1) | 22.3 | 8.07 |
| Available K (mg kg−1) | 204.8 | 105.9 |
| Exchangeable Ca (mg kg−1) | 1792 | 1711 |
| Exchangeable Mg (mg kg−1) | 191.8 | 186.0 |
| Treatment | Nutrient Input (kg ha−1) | |||||
|---|---|---|---|---|---|---|
| N | P2O5 | K2O | CaO | MgO | SO3 | |
| FP NPK | 401 | 401 | 401 | 0 | 0 | 0 |
| Opt NPK | 427 | 214 | 481 | 0 | 0 | 0 |
| FP NPK + polyhalite | 401 | 401 | 401 | 404 | 143 | 1140 |
| Opt NPK + polyhalite | 427 | 214 | 481 | 404 | 143 | 1140 |
| Treatment | Firmness (kg) | Titratable Acid (%) | Total Soluble Sugars (%) | Sugar/Acid Ratio | |
|---|---|---|---|---|---|
| External | Internal | ||||
| FP NPK | 2.62 ± 0.04 b | 1.11 ± 0.05 c | 0.43 ± 0.06 a | 4.70 ± 0.05 b | 10.93 b |
| Opt NPK | 2.63 ± 0.02 b | 1.17 ± 0.03 c | 0.37 ± 0.06 ab | 4.96 ± 0.32 b | 13.40 b |
| FP NPK + polyhalite | 3.05 ± 0.07 a | 1.38 ± 0.03 b | 0.29 ± 0.02 b | 5.86 ± 0.31 a | 20.20 a |
| Opt NPK + polyhalite | 3.13 ± 0.03 a | 1.48 ± 0.02 a | 0.27 ± 0.02 b | 5.73 ± 0.27 a | 21.22 a |
| Organ | Treatment | Cation Ratio | ||
|---|---|---|---|---|
| K/Ca | K/Mg | (K+Mg)/Ca | ||
| Fruit | FP NPK | 9.86 a | 21.7 a | 10.3 a |
| Opt NPK | 9.70 a | 22.4 a | 10.1 a | |
| FP NPK + polyhalite | 7.00 b | 20.9 a | 7.3 b | |
| Opt NPK + polyhalite | 7.34 b | 21.7 a | 7.7 b | |
| Leaves | FP NPK | 0.58 a | 5.65 a | 0.69 a |
| Opt NPK | 0.48 b | 4.75 b | 0.58 b | |
| FP NPK + polyhalite | 0.48 b | 3.98 c | 0.60 b | |
| Opt NPK + polyhalite | 0.48 b | 4.24 bc | 0.59 b | |
| Stem | FP NPK | 5.65 b | 42.4 a | 5.78 b |
| Opt NPK | 5.87 a | 45.5 a | 6.00 a | |
| FP NPK + polyhalite | 5.69 b | 36.4 b | 5.84 b | |
| Opt NPK + polyhalite | 5.90 a | 37.7 b | 6.06 a | |
| Petioles | FP NPK | 2.90 a | 15.4 a | 3.09 a |
| Opt NPK | 2.72 a | 15.4 a | 2.89 b | |
| FP NPK + polyhalite | 2.84 a | 13.8 b | 3.05 a | |
| Opt NPK + polyhalite | 2.86 a | 14.5 a | 3.06 a | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Chen, X.; Jiao, J.; Zhang, F.; Chen, X.; Li, G.; Song, Z.; Sokolowski, E.; Imas, P.; Magen, H.; et al. Towards Balanced Fertilizer Management in South China: Enhancing Wax Gourd (Benincasa hispida) Yield and Produce Quality. Sustainability 2022, 14, 5646. https://doi.org/10.3390/su14095646
Chen X, Chen X, Jiao J, Zhang F, Chen X, Li G, Song Z, Sokolowski E, Imas P, Magen H, et al. Towards Balanced Fertilizer Management in South China: Enhancing Wax Gourd (Benincasa hispida) Yield and Produce Quality. Sustainability. 2022; 14(9):5646. https://doi.org/10.3390/su14095646
Chicago/Turabian StyleChen, Xiao, Xiaodong Chen, Jiabin Jiao, Fusuo Zhang, Xinping Chen, Guohua Li, Zhao Song, Eldad Sokolowski, Patricia Imas, Hillel Magen, and et al. 2022. "Towards Balanced Fertilizer Management in South China: Enhancing Wax Gourd (Benincasa hispida) Yield and Produce Quality" Sustainability 14, no. 9: 5646. https://doi.org/10.3390/su14095646
APA StyleChen, X., Chen, X., Jiao, J., Zhang, F., Chen, X., Li, G., Song, Z., Sokolowski, E., Imas, P., Magen, H., Bustan, A., He, Y., Xie, D., & Zhang, B. (2022). Towards Balanced Fertilizer Management in South China: Enhancing Wax Gourd (Benincasa hispida) Yield and Produce Quality. Sustainability, 14(9), 5646. https://doi.org/10.3390/su14095646







