Edible Mushrooms for Sustainable and Healthy Human Food: Nutritional and Medicinal Attributes
Abstract
1. Introduction
2. Methodology of the Review
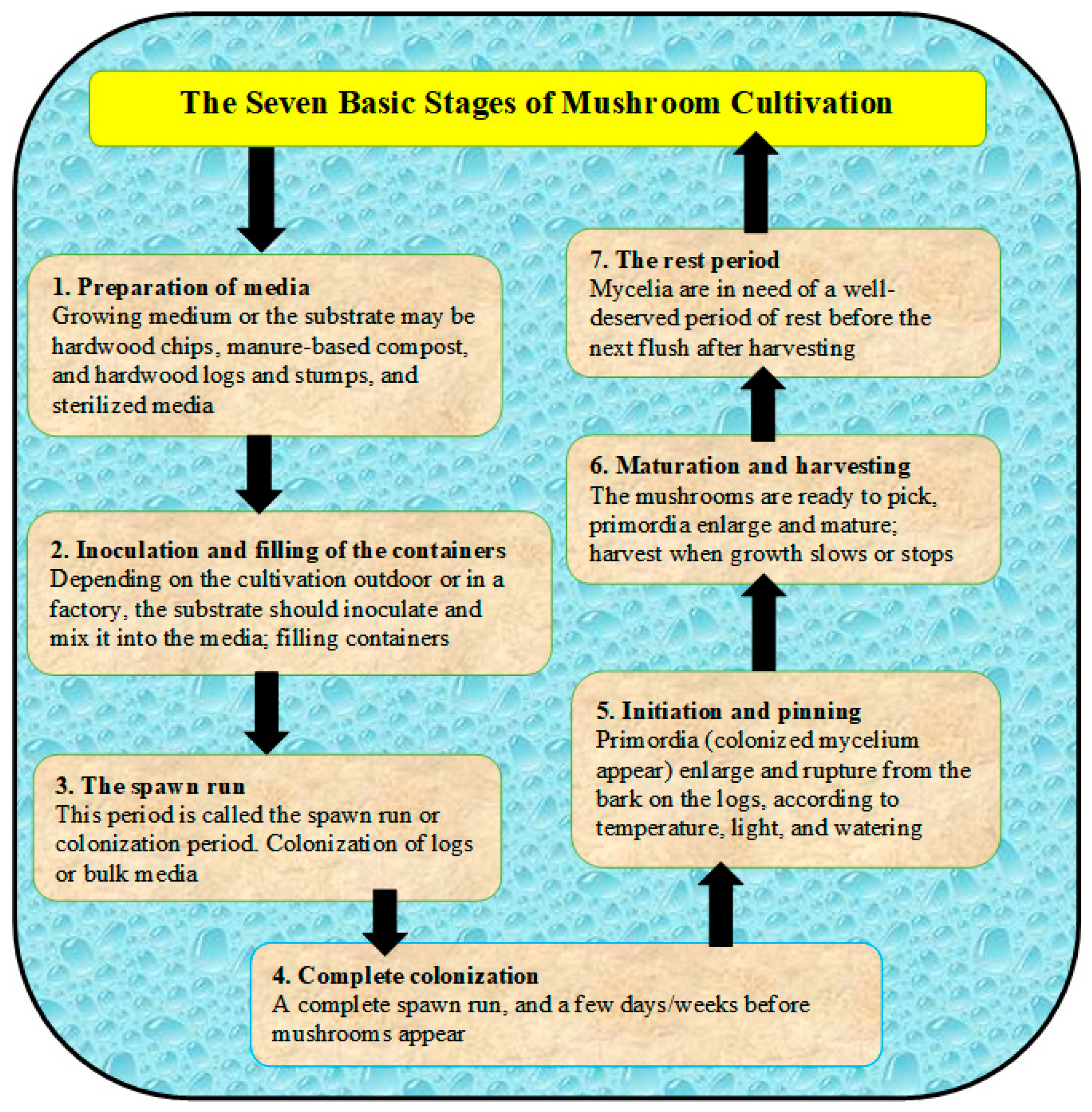
3. Sustainable Production of Edible Mushrooms
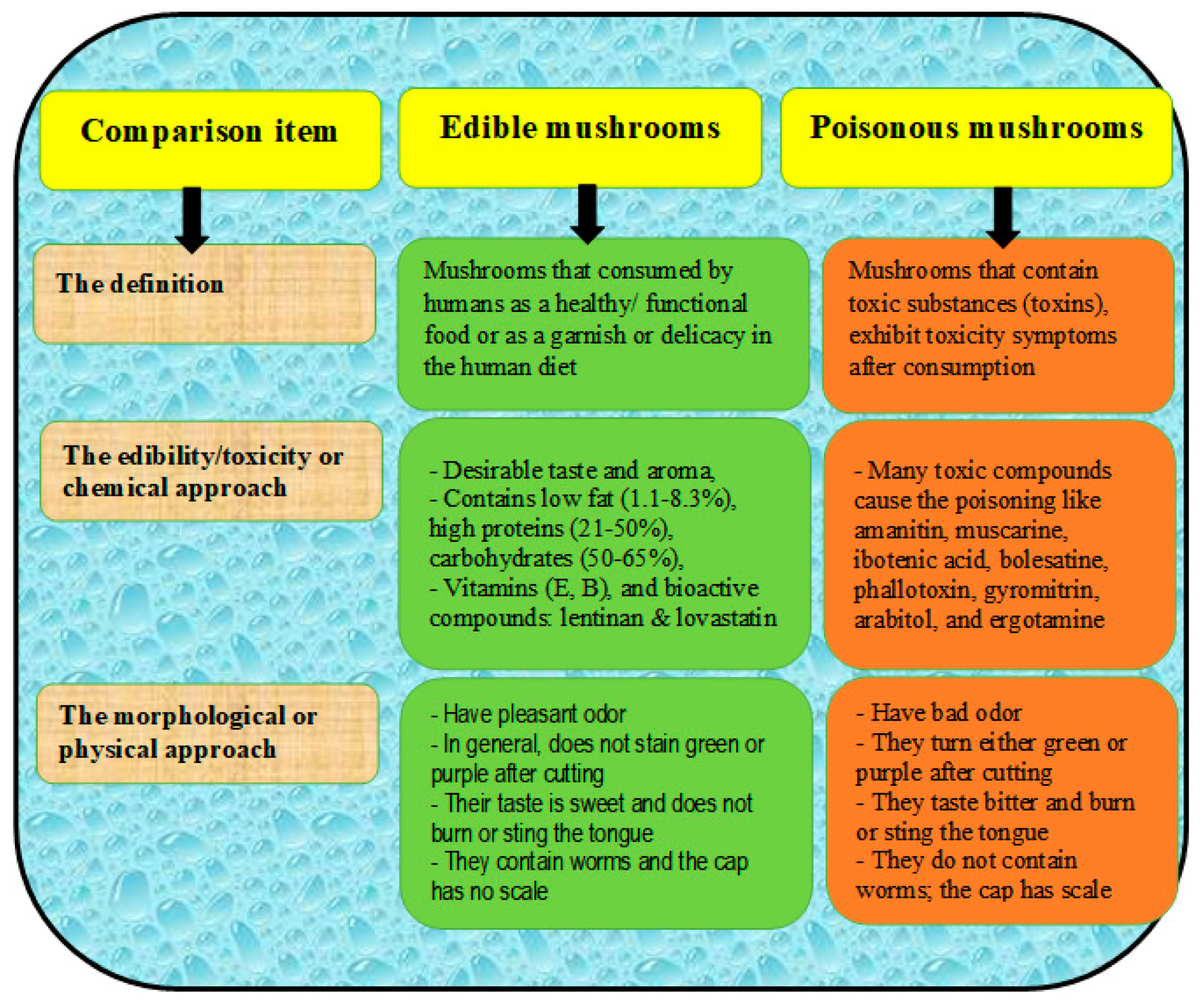
4. Edible vs. Poisonous Mushrooms
5. Common Species of Edible Mushrooms
- (1)
- Studies on edible mushrooms harvested from polluted areas with a focus on Africa [13], China [20,23,61,62], Greece [63], Iran [18,21], Poland [22,24,25,64,65,66], Turkey [27,28,67,68,69,70,71,72], and Spain [26] or on Europe as a whole, e.g., Świsłowski et al. [73], or on the global level, e.g., Dowlati et al. [18].
- (2)
- A study focusing on locals’ perspective concerning the changing of the abundance of wild edible mushrooms, which has decreased due to direct exploitation by 31% and land use change by 38% of all taxa [74].
- (3)
- Studies on the bioactive properties of different edible mushrooms, including total phenolic content, phenolic acid, antioxidant capacities, and antimicrobial activity in Pleurotus sajor-caju wild edible mushroom [75], in Agaricus bisporus, Cantharellus cibarius, Boletus edulis, Lactarius deliciosus, Lentinus edodes, Ganoderma lucidum, Hericium erinaceus, Morchella spp., and Pleurotus ostreatus [76], and in Pleurotus ostreatus [77]; polysaccharide in many edible mushrooms [78]; and bioactive components in several species of edible mushrooms [79].
- (4)
- Studies on determining the amount of potentially bioavailable compounds such as phenolic compounds in mycelia of Agaricus bisporus, Cantharellus cibarius, and Lentinula edodes [80].
- (5)
- (6)
- (7)
6. Bioactive Ingredients of Edible Mushrooms
7. Nutritional Values and Health Benefits of Edible Mushrooms
8. Biomedical Applications of Edible Mushrooms
- (1)
- Anticancer group
- (2)
- Antidiabetic group
- (3)
- Cardiovascular diseases group
- (4)
- Immune-function group
- (5)
- Rheumatoid arthritis group
- (6)
- Antiviral group
- (i)
- Antiviral mushrooms against HIV
- (ii)
- Antiviral mushrooms against HSV
- (iii)
- Antiviral mushrooms against influenza
- (iv)
- Antiviral mushrooms against hepatitis virus
- (v)
- Antiviral mushrooms against other viruses
- (7)
- Group of neurodegenerative diseases
- (8)
- DNA damage group
- (9)
- Antiaging group
- (10)
- Antiobesity group
9. Bioactive Compounds from Edible Mushrooms and Toxic Dose
10. Antinutritional Properties of Mushrooms
11. General Discussion
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Qing, Z.; Cheng, J.; Wang, X.; Tang, D.; Liu, X.; Zhu, M. The Effects of Four Edible Mushrooms (Volvariella volvacea, Hypsizygus marmoreus, Pleurotus ostreatus and Agaricus bisporus) on Physicochemical Properties of Beef Paste. LWT 2021, 135, 110063. [Google Scholar] [CrossRef]
- Li, M.; Yu, L.; Zhao, J.; Zhang, H.; Chen, W.; Zhai, Q.; Tian, F. Role of Dietary Edible Mushrooms in the Modulation of Gut Microbiota. J. Funct. Foods 2021, 83, 104538. [Google Scholar] [CrossRef]
- Balan, V.; Munafo, J.P.; Pattathil, S.; Merritt, B.B.; Venketachalam, S.; Ng, W.-O. Protocols to Evaluate the Nutritional and Potential Health Benefits of Edible Mushrooms. CBIOT 2018, 7, 34–58. [Google Scholar] [CrossRef]
- Cardwell, G.; Bornman, J.; James, A.; Black, L. A Review of Mushrooms as a Potential Source of Dietary Vitamin D. Nutrients 2018, 10, 1498. [Google Scholar] [CrossRef] [PubMed]
- Ślusarczyk, J.; Adamska, E.; Czerwik-Marcinkowska, J. Fungi and Algae as Sources of Medicinal and Other Biologically Active Compounds: A Review. Nutrients 2021, 13, 3178. [Google Scholar] [CrossRef] [PubMed]
- Taşkın, H.; Süfer, Ö.; Attar, Ş.H.; Bozok, F.; Baktemur, G.; Büyükalaca, S.; Kafkas, N.E. Total Phenolics, Antioxidant Activities and Fatty Acid Profiles of Six Morchella Species. J. Food Sci. Technol. 2021, 58, 692–700. [Google Scholar] [CrossRef]
- Dasgupta, A.; Acharya, K. Mushrooms: An Emerging Resource for Therapeutic Terpenoids. 3 Biotech 2019, 9, 369. [Google Scholar] [CrossRef]
- El-Maradny, Y.A.; El-Fakharany, E.M.; Abu-Serie, M.M.; Hashish, M.H.; Selim, H.S. Lectins Purified from Medicinal and Edible Mushrooms: Insights into Their Antiviral Activity against Pathogenic Viruses. Int. J. Biol. Macromol. 2021, 179, 239–258. [Google Scholar] [CrossRef] [PubMed]
- Acar, İ.; Blando, F.; Gul, B.; Greco, A.; Mukemre, M.; Uzun, Y.; Dalar, A. The Phenolic Profile and Biological Activities of the Wild-Edible Mushrooms Helvella leucopus and Morchella pulchella. Food Meas. 2021, 15, 555–566. [Google Scholar] [CrossRef]
- Cateni, F.; Gargano, M.L.; Procida, G.; Venturella, G.; Cirlincione, F.; Ferraro, V. Mycochemicals in Wild and Cultivated Mushrooms: Nutrition and Health. Phytochem. Rev. 2021, 1–45. [Google Scholar] [CrossRef]
- Saini, R.K.; Rauf, A.; Khalil, A.A.; Ko, E.-Y.; Keum, Y.-S.; Anwar, S.; Alamri, A.; Rengasamy, K.R.R. Edible Mushrooms Show Significant Differences in Sterols and Fatty Acid Compositions. S. Afr. J. Bot. 2021, 141, 344–356. [Google Scholar] [CrossRef]
- Nowak, R.; Nowacka-Jechalke, N.; Pietrzak, W.; Gawlik-Dziki, U. A New Look at Edible and Medicinal Mushrooms as a Source of Ergosterol and Ergosterol Peroxide-UHPLC-MS/MS Analysis. Food Chem. 2022, 369, 130927. [Google Scholar] [CrossRef] [PubMed]
- Gwenzi, W.; Tagwireyi, C.; Musiyiwa, K.; Chipurura, B.; Nyamangara, J.; Sanganyado, E.; Chaukura, N. Occurrence, Behavior, and Human Exposure and Health Risks of Potentially Toxic Elements in Edible Mushrooms with Focus on Africa. Environ. Monit. Assess. 2021, 193, 302. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.K.; Chaturvedi, V.K.; Mishra, D.; Bajpeyee, A.; Tiwari, A.; Singh, M.P. Role of Edible Mushroom as a Potent Therapeutics for the Diabetes and Obesity. 3 Biotech 2019, 9, 450. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, V.K.; Agarwal, S.; Gupta, K.K.; Ramteke, P.W.; Singh, M.P. Medicinal Mushroom: Boon for Therapeutic Applications. 3 Biotech 2018, 8, 334. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, K.; Xu, B. Anti-Obesity Effects of Medicinal and Edible Mushrooms. Molecules 2018, 23, 2880. [Google Scholar] [CrossRef]
- Rai, S.N.; Mishra, D.; Singh, P.; Vamanu, E.; Singh, M.P. Therapeutic Applications of Mushrooms and Their Biomolecules along with a Glimpse of in Silico Approach in Neurodegenerative Diseases. Biomed. Pharmacother. 2021, 137, 111377. [Google Scholar] [CrossRef]
- Dowlati, M.; Sobhi, H.R.; Esrafili, A.; FarzadKia, M.; Yeganeh, M. Heavy Metals Content in Edible Mushrooms: A Systematic Review, Meta-Analysis and Health Risk Assessment. Trends Food Sci. Technol. 2021, 109, 527–535. [Google Scholar] [CrossRef]
- El-Ramady, H.; Llanaj, X.; Prokisch, J. Edible Mushroom Cultivated in Polluted Soils and Its Potential Risks on Human Health: A Short Communication. Egypt. J. Soil Sci. 2021, 61, 1–10. [Google Scholar] [CrossRef]
- Ernst, A.-L.; Reiter, G.; Piepenbring, M.; Bässler, C. Spatial Risk Assessment of Radiocesium Contamination of Edible Mushrooms–Lessons from a Highly Frequented Recreational Area. Sci. Total Environ. 2022, 807, 150861. [Google Scholar] [CrossRef]
- Karami, H.; Shariatifar, N.; Nazmara, S.; Moazzen, M.; Mahmoodi, B.; Mousavi Khaneghah, A. The Concentration and Probabilistic Health Risk of Potentially Toxic Elements (PTEs) in Edible Mushrooms (Wild and Cultivated) Samples Collected from Different Cities of Iran. Biol. Trace Elem. Res. 2021, 199, 389–400. [Google Scholar] [CrossRef]
- Ronda, O.; Grządka, E.; Ostolska, I.; Orzeł, J.; Cieślik, B.M. Accumulation of Radioisotopes and Heavy Metals in Selected Species of Mushrooms. Food Chem. 2022, 367, 130670. [Google Scholar] [CrossRef]
- Wang, S.; Yang, B.; Zhou, Q.; Li, Z.; Li, W.; Zhang, J.; Tuo, F. Radionuclide Content and Risk Analysis of Edible Mushrooms in Northeast China. Radiat. Med. Prot. 2021, 2, 165–170. [Google Scholar] [CrossRef]
- Siwulski, M.; Budka, A.; Rzymski, P.; Gąsecka, M.; Kalač, P.; Budzyńska, S.; Magdziak, Z.; Niedzielski, P.; Mleczek, P.; Mleczek, M. Worldwide Basket Survey of Multielemental Composition of White Button Mushroom Agaricus Bisporus. Chemosphere 2020, 239, 124718. [Google Scholar] [CrossRef]
- Mleczek, M.; Budka, A.; Siwulski, M.; Mleczek, P.; Budzyńska, S.; Proch, J.; Gąsecka, M.; Niedzielski, P.; Rzymski, P. A Comparison of Toxic and Essential Elements in Edible Wild and Cultivated Mushroom Species. Eur. Food Res. Technol. 2021, 247, 1249–1262. [Google Scholar] [CrossRef]
- Melgar, M.J.; García, M.Á. Natural Radioactivity and Total K Content in Wild-Growing or Cultivated Edible Mushrooms and Soils from Galicia (NW, Spain). Environ. Sci. Pollut. Res. 2021, 28, 52925–52935. [Google Scholar] [CrossRef] [PubMed]
- Keskin, F.; Sarikurkcu, C.; Akata, I.; Tepe, B. Metal Concentrations of Wild Mushroom Species Collected from Belgrad Forest (Istanbul, Turkey) with Their Health Risk Assessments. Environ. Sci. Pollut. Res. 2021, 28, 36193–36204. [Google Scholar] [CrossRef] [PubMed]
- Keskin, F.; Sarikurkcu, C.; Akata, I.; Tepe, B. Element Concentration, Daily Intake of Elements, and Health Risk Indices of Wild Mushrooms Collected from Belgrad Forest and Ilgaz Mountain National Park (Turkey). Environ. Sci. Pollut. Res. 2021, 28, 51544–51555. [Google Scholar] [CrossRef] [PubMed]
- Shirur, M.; Barh, A.; Annepu, S.K. Sustainable Production of Edible and Medicinal Mushrooms: Implications on Mushroom Consumption. In Climate Change and Resilient Food Systems; Hebsale Mallappa, V.K., Shirur, M., Eds.; Springer: Singapore, 2021; pp. 315–346. ISBN 978-981-334-537-9. [Google Scholar]
- Korman, R. Growing Mushrooms: The Complete Grower’s Guide to Becoming a Mushroom Expert and Starting Cultivation at Home; Amazon Digital Services LLC: Seattle, WA, USA, 2020; ISBN 978-1-65911-727-1. [Google Scholar]
- Cotter, T. Organic Mushroom Farming and Mycoremediation: Simple to Advanced and Experimental Techniques for Indoor and Outdoor Cultivation; Chelsea Green Publishing: White River Junction, VT, USA, 2015. [Google Scholar]
- Shirur, M. Entrepreneurial Behaviour and Socio Economic Analysis of Mushroom Growers in Karnataka. Indian J. Agric. Sci. 2017, 6, 840–845. [Google Scholar]
- El-Ramady, H.; Abdalla, N.; Fawzy, Z.; Badgar, K.; Llanaj, X.; Törős, G.; Hajdú, P.; Eid, Y.; Prokisch, J. Green Biotechnology of Oyster Mushroom (Pleurotus ostreatus L.): A Sustainable Strategy for Myco-Remediation and Bio-Fermentation. Sustainability 2022, 14, 3667. [Google Scholar] [CrossRef]
- Arevalo, W. DIY Mushroom Cultivation: Growing Mushrooms at Home for Food, Medicine, and Soil. New Society Publishers, Gabriola Island, BC V0R 1X0, Canada. Available online: https://www.amazon.com/DIY-Mushroom-Cultivation-Mushrooms-Homesteader/dp/0865718954 (accessed on 17 March 2022).
- Antunes, F.; Marçal, S.; Taofiq, O.; Morais, A.M.M.B.; Freitas, A.C.; Ferreira, I.C.F.R.; Pintado, M. Valorization of Mushroom By-Products as a Source of Value-Added Compounds and Potential Applications. Molecules 2020, 25, 2672. [Google Scholar] [CrossRef]
- Wang, C.-Y. A Review on the Potential Reuse of Functional Polysaccharides Extracted from the By-Products of Mushroom Processing. Food Bioprocess Technol. 2020, 13, 217–228. [Google Scholar] [CrossRef]
- Kumar, H.; Bhardwaj, K.; Sharma, R.; Nepovimova, E.; Cruz-Martins, N.; Dhanjal, D.S.; Singh, R.; Chopra, C.; Verma, R.; Abd-Elsalam, K.A.; et al. Potential Usage of Edible Mushrooms and Their Residues to Retrieve Valuable Supplies for Industrial Applications. JoF 2021, 7, 427. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, D.; Chen, Y.; Liu, T.; Zhang, S.; Fan, H.; Liu, H.; Li, Y. Healthy Function and High Valued Utilization of Edible Fungi. Food Sci. Hum. Wellness 2021, 10, 408–420. [Google Scholar] [CrossRef]
- López-Hortas, L.; Flórez-Fernández, N.; Torres, M.D.; Domínguez, H. Update on Potential of Edible Mushrooms: High-value Compounds, Extraction Strategies and Bioactive Properties. Int. J. Food Sci. Technol. 2022, 57, 1378–1385. [Google Scholar] [CrossRef]
- Wu, F.; Zhou, L.-W.; Yang, Z.-L.; Bau, T.; Li, T.-H.; Dai, Y.-C. Resource Diversity of Chinese Macrofungi: Edible, Medicinal and Poisonous Species. Fungal Divers. 2019, 98, 1–76. [Google Scholar] [CrossRef]
- Boa, E. Wild Edible Fungi a Global Overview of Their Use and Importance to People. Available online: https://www.fao.org/3/y5489e/y5489e00.htm (accessed on 7 January 2022).
- Aoki, W.; Watanabe, M.; Watanabe, M.; Kobayashi, N.; Terajima, J.; Sugita-Konishi, Y.; Kondo, K.; Hara-kudo, Y. Discrimination between Edible and Poisonous Mushrooms among Japanese Entoloma sarcopum and Related Species Based on Phylogenetic Analysis and Insertion/Deletion Patterns of Nucleotide Sequences of the Cytochrome Oxidase 1 Gene. Genes Genet. Syst. 2020, 95, 133–139. [Google Scholar] [CrossRef]
- Wei, Y.; Li, L.; Liu, Y.; Xiang, S.; Zhang, H.; Yi, L.; Shang, Y.; Xu, W. Identification Techniques and Detection Methods of Edible Fungi Species. Food Chem. 2022, 374, 131803. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tian, Y.; Menolli, N.; Ye, L.; Karunarathna, S.C.; Perez-Moreno, J.; Rahman, M.M.; Rashid, M.H.; Phengsintham, P.; Rizal, L.; et al. Reviewing the World’s Edible Mushroom Species: A New Evidence-based Classification System. Compr. Rev. Food Sci. Food Saf. 2021, 20, 1982–2014. [Google Scholar] [CrossRef]
- Ramírez-Terrazo, A.; Adriana Montoya, E.; Garibay-Orijel, R.; Caballero-Nieto, J.; Kong-Luz, A.; Méndez-Espinoza, C. Breaking the Paradigms of Residual Categories and Neglectable Importance of Non-Used Resources: The “Vital” Traditional Knowledge of Non-Edible Mushrooms and Their Substantive Cultural Significance. J. Ethnobiol. Ethnomed. 2021, 17, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mo, M.; Yang, L.; Mi, F.; Cao, Y.; Liu, C.; Tang, X.; Wang, P.; Xu, J. Exploring the Species Diversity of Edible Mushrooms in Yunnan, Southwestern China, by DNA Barcoding. JoF 2021, 7, 310. [Google Scholar] [CrossRef] [PubMed]
- GBIF The Global Biodiversity Information Facility. SPECIES SYNONYM. Available online: https://www.gbif.org/search?q=SPECIES%20SYNONYM (accessed on 15 December 2021).
- BMS The British Mycological Society. Available online: https://www.britmycolsoc.org.uk (accessed on 7 January 2022).
- Tawatsin, A.; Parnmen, S.; Thavara, U.; Siriyasatien, P.; Kongtip, P. Mushroom Poisoning in Thailand: Incidence and Intoxication to Human Health. Med. Res. Arch. 2018, 6, 1–12. [Google Scholar] [CrossRef]
- Parnmen, S.; Nooron, N.; Leudang, S.; Sikaphan, S.; Polputpisatkul, D.; Rangsiruji, A. Phylogenetic Evidence Revealed Cantharocybe virosa (Agaricales, Hygrophoraceae) as a New Clinical Record for Gastrointestinal Mushroom Poisoning in Thailand. Toxicol. Res. 2020, 36, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Parnmen, S.; Nooron, N.; Leudang, S.; Sikaphan, S.; Polputpisatkul, D.; Pringsulaka, O.; Binchai, S.; Rangsiruji, A. Foodborne Illness Caused by Muscarine-Containing Mushrooms and Identification of Mushroom Remnants Using Phylogenetics and LC-MS/MS. Food Control 2021, 128, 108182. [Google Scholar] [CrossRef]
- White, J.; Weinstein, S.A.; De Haro, L.; Bédry, R.; Schaper, A.; Rumack, B.H.; Zilker, T. Mushroom Poisoning: A Proposed New Clinical Classification. Toxicon 2019, 157, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Arici, M.A.; Tuncok, Y. Mushroom-Related Toxins, Alpha Amanitin, and Usage of Antioxidants: Directions toward Antioxidant Capacity. In Toxicology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 447–456. ISBN 978-0-12-819092-0. [Google Scholar]
- Beuhler, M.C. Overview of Mushroom Poisoning. In Critical Care Toxicology; Brent, J., Burkhart, K., Dargan, P., Hatten, B., Megarbane, B., Palmer, R., White, J., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 2103–2128. ISBN 978-3-319-17899-8. [Google Scholar]
- Hedman, H.; Holmdahl, J.; Mölne, J.; Ebefors, K.; Haraldsson, B.; Nyström, J. Long-Term Clinical Outcome for Patients Poisoned by the Fungal Nephrotoxin Orellanine. BMC Nephrol. 2017, 18, 121. [Google Scholar] [CrossRef]
- Flores-Holguín, N.; Frau, J.; Glossman-Mitnik, D. Chemical Reactivity and Bioactivity Properties of the Phallotoxin Family of Fungal Peptides Based on Conceptual Peptidology and DFT Study. Heliyon 2019, 5, e02335. [Google Scholar] [CrossRef] [PubMed]
- Govorushko, S.; Rezaee, R.; Dumanov, J.; Tsatsakis, A. Poisoning Associated with the Use of Mushrooms: A Review of the Global Pattern and Main Characteristics. Food Chem. Toxicol. 2019, 128, 267–279. [Google Scholar] [CrossRef]
- Ukwuru, M.; Muritala, A.; Lu, E. Edible and Non-Edible Wild Mushrooms: Nutrition, Toxicity and Strategies for Recognition. J. Clin. Nutr. Metab. 2018, 2, 9. [Google Scholar]
- Karimi, G.; Razavi, B.M. Poisonous Mushrooms. In Clinical Toxinology; Gopalakrishnakone, P., Faiz, S.M.A., Gnanathasan, C.A., Habib, A.G., Fernando, R., Yang, C.-C., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 1–18. ISBN 978-94-007-6288-6. [Google Scholar]
- Kumar, S.; Jain, T.; Banerjee, D. Fungal Diseases and Their Treatment: A Holistic Approach. In Pathogenicity and Drug Resistance of Human Pathogens; Hameed, S., Fatima, Z., Eds.; Springer: Singapore, 2019; pp. 111–134. ISBN 978-981-329-448-6. [Google Scholar]
- Falandysz, J.; Zhang, J.; Saniewski, M. 137Cs, 40K, and K in Raw and Stir-Fried Mushrooms from the Boletaceae Family from the Midu Region in Yunnan, Southwest China. Environ. Sci. Pollut. Res. 2020, 27, 32509–32517. [Google Scholar] [CrossRef]
- Fu, Z.; Liu, G.; Wang, L. Assessment of Potential Human Health Risk of Trace Element in Wild Edible Mushroom Species Collected from Yunnan Province, China. Environ. Sci. Pollut. Res. 2020, 27, 29218–29227. [Google Scholar] [CrossRef]
- Kokkoris, V.; Massas, I.; Polemis, E.; Koutrotsios, G.; Zervakis, G.I. Accumulation of Heavy Metals by Wild Edible Mushrooms with Respect to Soil Substrates in the Athens Metropolitan Area (Greece). Sci. Total Environ. 2019, 685, 280–296. [Google Scholar] [CrossRef]
- Gałgowska, M.; Pietrzak-Fiećko, R. The Level of Selected Organochlorine Compounds Residues in Popular Edible Mushrooms from North-Eastern Poland. Food Chem. 2021, 353, 129441. [Google Scholar] [CrossRef] [PubMed]
- Mleczek, M.; Budka, A.; Siwulski, M.; Budzyńska, S.; Kalač, P.; Karolewski, Z.; Lisiak-Zielińska, M.; Kuczyńska-Kippen, N.; Niedzielski, P. Anthropogenic Contamination Leads to Changes in Mineral Composition of Soil- and Tree-Growing Mushroom Species: A Case Study of Urban vs. Rural Environments and Dietary Implications. Sci. Total Environ. 2021, 809, 151162. [Google Scholar] [CrossRef]
- Mleczek, M.; Siwulski, M.; Budka, A.; Mleczek, P.; Budzyńska, S.; Szostek, M.; Kuczyńska-Kippen, N.; Kalač, P.; Niedzielski, P.; Gąsecka, M.; et al. Toxicological Risks and Nutritional Value of Wild Edible Mushroom Species—A Half-Century Monitoring Study. Chemosphere 2021, 263, 128095. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Akata, I.; Guven, G.; Tepe, B. Metal Concentration and Health Risk Assessment of Wild Mushrooms Collected from the Black Sea Region of Turkey. Environ. Sci. Pollut. Res. 2020, 27, 26419–26441. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Akata, I.; Tepe, B. Metal Concentration and Health Risk Assessment of Eight Russula Mushrooms Collected from Kizilcahamam-Ankara, Turkey. Environ. Sci. Pollut. Res. 2021, 28, 15743–15754. [Google Scholar] [CrossRef] [PubMed]
- Sarikurkcu, C.; Popović-Djordjević, J.; Solak, M.H. Wild Edible Mushrooms from Mediterranean Region: Metal Concentrations and Health Risk Assessment. Ecotoxicol. Environ. Saf. 2020, 190, 110058. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Sarikurkcu, R.T.; Akata, I.; Tepe, B. Metal Concentration and Health Risk Assessment of Fifteen Wild Mushrooms Collected from the Ankara University Campus (Turkey). Environ. Sci. Pollut. Res. 2020, 27, 32474–32480. [Google Scholar] [CrossRef] [PubMed]
- Sarikurkcu, C.; Yildiz, D.; Akata, I.; Tepe, B. Evaluation of the Metal Concentrations of Wild Mushroom Species with Their Health Risk Assessments. Environ. Sci. Pollut. Res. 2021, 28, 21437–21454. [Google Scholar] [CrossRef] [PubMed]
- Türkmen, M.; Budur, D. Heavy Metal Contaminations in Edible Wild Mushroom Species from Turkey’s Black Sea Region. Food Chem. 2018, 254, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Świsłowski, P.; Dołhańczuk-Śródka, A.; Rajfur, M. Bibliometric Analysis of European Publications between 2001 and 2016 on Concentrations of Selected Elements in Mushrooms. Environ. Sci. Pollut. Res. 2020, 27, 22235–22250. [Google Scholar] [CrossRef] [PubMed]
- Schunko, C.; Li, X.; Klappoth, B.; Lesi, F.; Porcher, V.; Porcuna-Ferrer, A.; Reyes-García, V. Local Communities’ Perceptions of Wild Edible Plant and Mushroom Change: A Systematic Review. Glob. Food Secur. 2022, 32, 100601. [Google Scholar] [CrossRef]
- Kandasamy, S.; Chinnappan, S.; Thangaswamy, S.; Balakrishnan, S.; Khalifa, A.Y.Z. Assessment of Antioxidant, Antibacterial Activities and Bioactive Compounds of the Wild Edible Mushroom Pleurotus Sajor-Caju. Int. J. Pept. Res. Ther. 2020, 26, 1575–1581. [Google Scholar] [CrossRef]
- Alkin, M.; Söğüt, E.; Seydim, A.C. Determination of Bioactive Properties of Different Edible Mushrooms from Turkey. Food Meas. 2021, 15, 3608–3617. [Google Scholar] [CrossRef]
- Goswami, B.; Majumdar, S.; Das, A.; Barui, A.; Bhowal, J. Evaluation of Bioactive Properties of Pleurotus Ostreatus Mushroom Protein Hydrolysate of Different Degree of Hydrolysis. LWT 2021, 149, 111768. [Google Scholar] [CrossRef]
- Maity, P.; Sen, I.K.; Chakraborty, I.; Mondal, S.; Bar, H.; Bhanja, S.K.; Mandal, S.; Maity, G.N. Biologically Active Polysaccharide from Edible Mushrooms: A Review. Int. J. Biol. Macromol. 2021, 172, 408–417. [Google Scholar] [CrossRef]
- Yadav, D.; Negi, P.S. Bioactive Components of Mushrooms: Processing Effects and Health Benefits. Food Res. Int. 2021, 148, 110599. [Google Scholar] [CrossRef]
- Kała, K.; Krakowska, A.; Szewczyk, A.; Ostachowicz, B.; Szczurek, K.; Fijałkowska, A.; Muszyńska, B. Determining the Amount of Potentially Bioavailable Phenolic Compounds and Bioelements in Edible Mushroom Mycelia of Agaricus bisporus, Cantharellus cibarius, and Lentinula edodes. Food Chem. 2021, 352, 129456. [Google Scholar] [CrossRef] [PubMed]
- Mešić, A.; Šamec, D.; Jadan, M.; Bahun, V.; Tkalčec, Z. Integrated Morphological with Molecular Identification and Bioactive Compounds of 23 Croatian Wild Mushrooms Samples. Food Biosci. 2020, 37, 100720. [Google Scholar] [CrossRef]
- Ao, T.; Deb, C.R.; Rao, S.R. Molecular Strategies for Identification and Characterization of Some Wild Edible Mushrooms of Nagaland, India. Mol. Biol. Rep. 2020, 47, 621–630. [Google Scholar] [CrossRef]
- Altaf, U.; Lalotra, P.; Sharma, Y.P. Nutritional and Mineral Composition of Four Wild Edible Mushrooms from Jammu and Kashmir, India. Indian Phytopathol. 2020, 73, 313–320. [Google Scholar] [CrossRef]
- Atri, N.S.; Sharma, Y.P.; Kumar, S. Mridu Wild Edible Mushrooms of North West Himalaya: Their Nutritional, Nutraceutical, and Sociobiological Aspects. In Microbial Diversity in Ecosystem Sustainability and Biotechnological Applications; Satyanarayana, T., Das, S.K., Johri, B.N., Eds.; Springer: Singapore, 2019; pp. 533–563. ISBN 9789811384868. [Google Scholar]
- Sifat, N.; Lovely, F.; Zihad, S.M.N.K.; Hossain, M.G.; Shilpi, J.A.; Grice, I.D.; Mubarak, M.S.; Uddin, S.J. Investigation of the Nutritional Value and Antioxidant Activities of Common Bangladeshi Edible Mushrooms. Clin. Phytosci. 2020, 6, 88. [Google Scholar] [CrossRef]
- Jacinto-Azevedo, B.; Valderrama, N.; Henríquez, K.; Aranda, M.; Aqueveque, P. Nutritional Value and Biological Properties of Chilean Wild and Commercial Edible Mushrooms. Food Chem. 2021, 356, 129651. [Google Scholar] [CrossRef]
- Barbosa, J.R.; de Carvalho Junior, R.N. Polysaccharides Obtained from Natural Edible Sources and Their Role in Modulating the Immune System: Biologically Active Potential That Can Be Exploited against COVID-19. Trends Food Sci. Technol. 2021, 108, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Rangsinth, P.; Sillapachaiyaporn, C.; Nilkhet, S.; Tencomnao, T.; Ung, A.T.; Chuchawankul, S. Mushroom-Derived Bioactive Compounds Potentially Serve as the Inhibitors of SARS-CoV-2 Main Protease: An in Silico Approach. J. Tradit. Complementary Med. 2021, 11, 158–172. [Google Scholar] [CrossRef]
- Gurbuz, I.B. Nongreen Revolution: A Case Study of Wild-Grown Edible Mushroom. Environ. Sci. Pollut. Res. 2019, 26, 7954–7959. [Google Scholar] [CrossRef] [PubMed]
- Isik, H. Fatty Acid Contents of Three Wild Edible Mushroom Species. Chem. Nat. Compd. 2020, 56, 1114–1116. [Google Scholar] [CrossRef]
- Koutrotsios, G.; Tagkouli, D.; Bekiaris, G.; Kaliora, A.; Tsiaka, T.; Tsiantas, K.; Chatzipavlidis, I.; Zoumpoulakis, P.; Kalogeropoulos, N.; Zervakis, G.I. Enhancing the Nutritional and Functional Properties of Pleurotus citrinopileatus Mushrooms through the Exploitation of Winery and Olive Mill Wastes. Food Chem. 2022, 370, 131022. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, P.; Cheng, J.; Yang, H.; Zou, J.; Liu, X. The Role of Endogenous Enzyme from Straw Mushroom (Volvariella volvacea) in Improving Taste and Volatile Flavor Characteristics of Cantonese Sausage. LWT 2022, 154, 112627. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, F.; Ng, T.B. Interrelationship among Paraptosis, Apoptosis and Autophagy in Lung Cancer A549 Cells Induced by BEAP, an Antitumor Protein Isolated from the Edible Porcini Mushroom Boletus edulis. Int. J. Biol. Macromol. 2021, 188, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Pohleven, J.; Brzin, J.; Vrabec, L.; Leonardi, A.; Čokl, A.; Štrukelj, B.; Kos, J.; Sabotič, J. Basidiomycete Clitocybe nebularis Is Rich in Lectins with Insecticidal Activities. Appl. Microbiol. Biotechnol. 2011, 91, 1141–1148. [Google Scholar] [CrossRef] [PubMed]
- Ganeshpurkar, A.; Pardhi, P.; Bhadoriya, S.S.; Jain, N.; Rai, G.; Jain, A.P. Antioxidant Potential of White Oyster Culinary-Medicinal Mushroom, Pleurotus florida (Higher Basidiomycetes). Int. J. Med. Mushrooms 2015, 17, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.J.; Rezoagli, E.; Pogue, R.; Simonassi-Paiva, B.; Abidin, I.I.Z.; Fehrenbach, G.W.; O’Neil, E.; Major, I.; Laffey, J.G.; Rowan, N. Immunomodulatory Activity of β-Glucan Polysaccharides Isolated from Different Species of Mushroom—A Potential Treatment for Inflammatory Lung Conditions. Sci. Total Environ. 2022, 809, 152177. [Google Scholar] [CrossRef]
- Lavi, I.; Nimri, L.; Levinson, D.; Peri, I.; Hadar, Y.; Schwartz, B. Glucans from the Edible Mushroom Pleurotus pulmonarius Inhibit Colitis-Associated Colon Carcinogenesis in Mice. J. Gastroenterol. 2012, 47, 504–518. [Google Scholar] [CrossRef] [PubMed]
- Fogarasi, M.; Socaci, S.; Dulf, F.; Diaconeasa, Z.; Fărcaș, A.; Tofană, M.; Semeniuc, C. Bioactive Compounds and Volatile Profiles of Five Transylvanian Wild Edible Mushrooms. Molecules 2018, 23, 3272. [Google Scholar] [CrossRef]
- Alshammaa, D.A.S. Phytochemical Investigation and Quantitative Comparison of Ergosterol Between Agaricus bisporus and Pleurotus ostreatus by HPLC and GC-MS Methods. Int. J. Pharm. Sci. Rev. Res. 2017, 44, 215–220. [Google Scholar]
- Verma, N.K.; Singh, A.P.; Singh, V.K. Agaricus bisporus (Fungi) Chemical Constituents and Pharmacological Activities—A Review. Asian J. Phytomedicine Clin. Res. 2019, 7, 82–87. [Google Scholar]
- Liu, G.; Ye, J.; Li, W.; Zhang, J.; Wang, Q.; Zhu, X.; Miao, J.; Huang, Y.; Chen, Y.; Cao, Y. Extraction, Structural Characterization, and Immunobiological Activity of ABP Ia Polysaccharide from Agaricus bisporus. Int. J. Biol. Macromol. 2020, 162, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Feng, T.; Li, Z.-H.; Dong, Z.-J.; Su, J.; Li, Y.; Liu, J.-K. Non-Isoprenoid Botryane Sesquiterpenoids from Basidiomycete Boletus edulis and Their Cytotoxic Activity. Nat. Prod. Bioprospect. 2011, 1, 29–32. [Google Scholar] [CrossRef]
- Žurga, S.; Nanut, M.P.; Kos, J.; Sabotič, J. Fungal Lectin MpL Enables Entry of Protein Drugs into Cancer Cells and Their Subcellular Targeting. Oncotarget 2017, 8, 26896–26910. [Google Scholar] [CrossRef][Green Version]
- Luo, A.; Luo, A.; Huang, J.; Fan, Y. Purification, Characterization and Antioxidant Activities in Vitro and in Vivo of the Polysaccharides from Boletus edulis Bull. Molecules 2012, 17, 8079–8090. [Google Scholar] [CrossRef] [PubMed]
- Gąsecka, M.; Mleczek, M.; Siwulski, M.; Niedzielski, P.; Kozak, L. Phenolic and Flavonoid Content in Hericium erinaceus, Ganoderma lucidum, and Agrocybe aegerita under Selenium Addition. Acta Aliment. 2016, 45, 300–308. [Google Scholar] [CrossRef]
- Surup, F.; Hennicke, F.; Sella, N.; Stroot, M.; Bernecker, S.; Pfütze, S.; Stadler, M.; Rühl, M. New Terpenoids from the Fermentation Broth of the Edible Mushroom Cyclocybe aegerita. Beilstein J. Org. Chem. 2019, 15, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Citores, L.; Ragucci, S.; Ferreras, J.M.; Maro, A.D.; Iglesias, R.; Citores, L.; Ferreras, J.M.; Iglesias, R. Ageritin, a Ribotoxin from Poplar Mushroom (Agrocybe aegerita) with Defensive and Antiproliferative Activities. ACS Chem. Biol. 2019, 14, 1319–1327. [Google Scholar] [CrossRef] [PubMed]
- Motoshima, R.A.; Rosa, T.D.F.; Mendes, L.D.C.; da Silva, E.V.; Viana, S.R.; do Amaral, B.S.; de Souza, D.H.; Lião, L.M.; da Silva, M.D.L.C.; de Sousa, L.R.; et al. Inhibition of Leishmania Amazonensis Arginase by Fucogalactan Isolated from Agrocybe aegerita Mushroom. Carbohydr. Polym. 2018, 201, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Nowakowski, P.; Naliwajko, S.K.; Markiewicz-Żukowska, R.; Borawska, M.H.; Socha, K. The Two Faces of Coprinus comatus—Functional Properties and Potential Hazards. Phytother. Res. 2020, 34, 2932–2944. [Google Scholar] [CrossRef] [PubMed]
- Dulay, M.R.; Sanguesa, K.B.; Ablaza, J.; Joson, A.J.M.; Peria, J.N.T.; Quejada, C.S.; Basa, J.O.; Castro, M. Bioactive Myco-Nutrients of Aseptically Cultured Fruiting Bodies of coprinus comatus (o.f. Müll.) Pers. On Rice Bran-Enriched Ruminants’ Dung. Available online: https://www.semanticscholar.org/paper/BIOACTIVE-MYCO-NUTRIENTS-OF-ASEPTICALLY-CULTURED-OF-Dulay-Sanguesa/6ab75659b7e5c608804664d0bc0d3618a33798ad (accessed on 7 January 2022).
- Zhao, H.; Zhang, J.; Liu, X.; Yang, Q.; Dong, Y.; Jia, L. The Antioxidant Activities of Alkalic-Extractable Polysaccharides from Coprinus comatus on Alcohol-Induced Liver Injury in Mice. Sci. Rep. 2019, 8, 11695. [Google Scholar] [CrossRef] [PubMed]
- Kalogeropoulos, N.; Yanni, A.E.; Koutrotsios, G.; Aloupi, M. Bioactive Microconstituents and Antioxidant Properties of Wild Edible Mushrooms from the Island of Lesvos, Greece. Food Chem. Toxicol. 2013, 55, 378–385. [Google Scholar] [CrossRef]
- Feussi Tala, M.; Qin, J.; Ndongo, J.T.; Laatsch, H. New Azulene-Type Sesquiterpenoids from the Fruiting Bodies of Lactarius deliciosus. Nat. Prod. Bioprospect. 2017, 7, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Khaund, P.; Joshi, S.R. Enzymatic Profiling of Wild Edible Mushrooms Consumed by the Ethnic Tribes of India. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 263–271. [Google Scholar] [CrossRef]
- Su, S.; Ding, X.; Fu, L.; Hou, Y. Structural Characterization and Immune Regulation of a Novel Polysaccharide from Maerkang Lactarius deliciosus Gray. Int. J. Mol. Med. 2019, 44, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Sarma, D.; Saha, A.K.; Datta, B.K. Bioactive Compounds with Special References to Anticancer Property of Oyster Mushroom. J. Pharmacogn. Phytochem. 2018, 7, 2694–2698. [Google Scholar]
- Souilem, F.; Fernandes, Â.; Calhelha, R.C.; Barreira, J.C.M.; Barros, L.; Skhiri, F.; Martins, A.; Ferreira, I.C.F.R. Wild Mushrooms and Their Mycelia as Sources of Bioactive Compounds: Antioxidant, Anti-Inflammatory and Cytotoxic Properties. Food Chem. 2017, 230, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Liu, Y.; Qiang, Z. A Potent Pharmacological Mushroom: Pleurotus eryngii. Fungal Genom. Biol. 2016, 6, 1–5. [Google Scholar] [CrossRef]
- Alam, N.; Yoon, K.; Shin, P.; Cheong, J.-C.; Yoo, Y.; Lee, T.-S. Antioxidant, Phenolic Compounds Concentration, Xanthine Oxidase and Tyrosinase Inhibitory Activities of Pleurotus cornucopiae. Aust. J. Basic Appl. Sci. 2011, 5, 229–239. [Google Scholar]
- Lee, S.R.; Lee, D.; Lee, H.-J.; Noh, H.J.; Jung, K.; Kang, K.S.; Kim, K.H. Renoprotective Chemical Constituents from an Edible Mushroom, Pleurotus cornucopiae in Cisplatin-Induced Nephrotoxicity. Bioorg. Chem. 2017, 71, 67–73. [Google Scholar] [CrossRef]
- Golak-Siwulska, I.; Kałużewicz, A.; Spiżewski, T.; Siwulski, M.; Sobieralski, K. Bioactive Compounds and Medicinal Properties of Oyster Mushrooms (Pleurotus sp.). Folia Hortic. 2018, 30, 191–201. [Google Scholar] [CrossRef]
- Minato, K.; Ohara, A.; Mizuno, M. A Proinflammatory Effect of the β -Glucan from Pleurotus cornucopiae Mushroom on Macrophage Action. Mediat. Inflamm. 2017, 2017, 8402405. [Google Scholar] [CrossRef]
- Li, X.; Zhang, X.; Ye, L.; Kang, Z.; Jia, D.; Yang, L.; Zhang, B. LC-MS-Based Metabolomic Approach Revealed the Significantly Different Metabolic Profiles of Five Commercial Truffle Species. Front. Microbiol. 2019, 10, 2227. [Google Scholar] [CrossRef]
- Chen, H.-P.; Zhao, Z.-Z.; Li, Z.-H.; Huang, Y.; Zhang, S.-B.; Tang, Y.; Yao, J.-N.; Chen, L.; Isaka, M.; Feng, T.; et al. Anti-Proliferative and Anti-Inflammatory Lanostane Triterpenoids from the Polish Edible Mushroom Macrolepiota procera. J. Agric. Food Chem. 2018, 66, 3146–3154. [Google Scholar] [CrossRef] [PubMed]
- Nowak, R.; Nowacka-Jechalke, N.; Juda, M.; Malm, A. The Preliminary Study of Prebiotic Potential of Polish Wild Mushroom Polysaccharides: The Stimulation Effect on Lactobacillus Strains Growth. Eur. J. Nutr. 2018, 57, 1511–1521. [Google Scholar] [CrossRef] [PubMed]
- Butkhup, L.; Samappito, W.; Jorjong, S. Evaluation of Bioactivities and Phenolic Contents of Wild Edible Mushrooms from Northeastern Thailand. Food Sci. Biotechnol. 2018, 27, 193–202. [Google Scholar] [CrossRef]
- Zhu, M.-J.; Du, F.; Zhang, G.-Q.; Wang, H.-X.; Ng, T.-B. Purification a Laccase Exhibiting Dye Decolorizing Ability from an Edible Mushroom Russula virescens. Int. Biodeterior. Biodegrad. 2013, 82, 33–39. [Google Scholar] [CrossRef]
- Li, H.; Wang, X.; Xiong, Q.; Yu, Y.; Peng, L. Sulfated Modification, Characterization, and Potential Bioactivities of Polysaccharide from the Fruiting Bodies of Russula virescens. Int. J. Biol. Macromol. 2020, 154, 1438–1447. [Google Scholar] [CrossRef]
- Tejedor-Calvo, E.; Morales, D.; Marco, P.; Sánchez, S.; Garcia-Barreda, S.; Smiderle, F.R.; Iacomini, M.; Villalva, M.; Santoyo, S.; Soler-Rivas, C. Screening of Bioactive Compounds in Truffles and Evaluation of Pressurized Liquid Extractions (PLE) to Obtain Fractions with Biological Activities. Food Res. Int. 2020, 132, 109054. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Sarikurkcu, C.; Yalcin, O.U.; Cengiz, M.; Gungor, H. Metal Concentration, Phenolics Profiling, and Antioxidant Activity of Two Wild Edible Melanoleuca Mushrooms (M. cognata and M. stridula). Microchem. J. 2019, 150, 104172. [Google Scholar] [CrossRef]
- Taofiq, O.; Calhelha, R.C.; Heleno, S.; Barros, L.; Martins, A.; Santos-Buelga, C.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. The Contribution of Phenolic Acids to the Anti-Inflammatory Activity of Mushrooms: Screening in Phenolic Extracts, Individual Parent Molecules and Synthesized Glucuronated and Methylated Derivatives. Food Res. Int. 2015, 76, 821–827. [Google Scholar] [CrossRef]
- Saltarelli, R.; Palma, F.; Gioacchini, A.M.; Calcabrini, C.; Mancini, U.; De Bellis, R.; Stocchi, V.; Potenza, L. Phytochemical Composition, Antioxidant and Antiproliferative Activities and Effects on Nuclear DNA of Ethanolic Extract from an Italian Mycelial Isolate of Ganoderma lucidum. J. Ethnopharmacol. 2019, 231, 464–473. [Google Scholar] [CrossRef]
- Papoutsis, K.; Grasso, S.; Menon, A.; Brunton, N.P.; Lyng, J.G.; Jacquier, J.-C.; Bhuyan, D.J. Recovery of Ergosterol and Vitamin D2 from Mushroom Waste-Potential Valorization by Food and Pharmaceutical Industries. Trends Food Sci. Technol. 2020, 99, 351–366. [Google Scholar] [CrossRef]
- Lu, H.; Lou, H.; Hu, J.; Liu, Z.; Chen, Q. Macrofungi: A Review of Cultivation Strategies, Bioactivity, and Application of Mushrooms. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2333–2356. [Google Scholar] [CrossRef] [PubMed]
- Das, A.K.; Nanda, P.K.; Dandapat, P.; Bandyopadhyay, S.; Gullón, P.; Sivaraman, G.K.; McClements, D.J.; Gullón, B.; Lorenzo, J.M. Edible Mushrooms as Functional Ingredients for Development of Healthier and More Sustainable Muscle Foods: A Flexitarian Approach. Molecules 2021, 26, 2463. [Google Scholar] [CrossRef] [PubMed]
- Di Anibal, C.; Farenzena, S.; Rodríguez, M.S.; Albertengo, L. Chemical Composition and Nutritional Value of Argentine Commercial Edible Mushrooms. J. Verbr. Lebensm. 2015, 10, 155–164. [Google Scholar] [CrossRef]
- Yu, Q.; Guo, M.; Zhang, B.; Wu, H.; Zhang, Y.; Zhang, L. Analysis of Nutritional Composition in 23 Kinds of Edible Fungi. J. Food Qual. 2020, 2020, 8821315. [Google Scholar] [CrossRef]
- Ao, T.; Deb, C.R. Nutritional and Antioxidant Potential of Some Wild Edible Mushrooms of Nagaland, India. J. Food Sci. Technol. 2019, 56, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Usman, M.; Murtaza, G.; Ditta, A. Nutritional, Medicinal, and Cosmetic Value of Bioactive Compounds in Button Mushroom (Agaricus bisporus): A Review. Appl. Sci. 2021, 11, 5943. [Google Scholar] [CrossRef]
- Xu, Z.; Fu, L.; Feng, S.; Yuan, M.; Huang, Y.; Liao, J.; Zhou, L.; Yang, H.; Ding, C. Chemical Composition, Antioxidant and Antihyperglycemic Activities of the Wild Lactarius deliciosus from China. Molecules 2019, 24, 1357. [Google Scholar] [CrossRef]
- Das, P.; Sikdar, S.R.; Samanta, A. Nutritional Analysis and Molecular Characterization of Hybrid Mushrooms Developed through Intergeneric Protoplast Fusion between Pleurotus Sajor-Caju and Calocybe indica with the Purpose to Achieve Improved Strains. World J. Microbiol. Biotechnol. 2021, 37, 69. [Google Scholar] [CrossRef]
- Wunjuntuk, K.; Ahmad, M.; Techakriengkrai, T.; Chunhom, R.; Jaraspermsuk, E.; Chaisri, A.; Kiwwongngam, R.; Wuttimongkolkul, S.; Charoenkiatkul, S. Proximate Composition, Dietary Fibre, Beta-Glucan Content, and Inhibition of Key Enzymes Linked to Diabetes and Obesity in Cultivated and Wild Mushrooms. J. Food Compos. Anal. 2022, 105, 104226. [Google Scholar] [CrossRef]
- Nayak, P.C.; Raju, C.V.; Lakshmisha, I.P.; Singh, R.R.; Sofi, F.R. Influence of Button Mushroom (Agaricus bisporus) on Quality and Refrigerated Storage Stability of Patties Prepared from Sutchi Catfish (Pangasius hypophthalmus). J. Food Sci. Technol. 2014. [Google Scholar] [CrossRef]
- Jo, K.; Lee, J.; Jung, S. Quality Characteristics of Low-Salt Chicken Sausage Supplemented with a Winter Mushroom Powder. Korean J. Food Sci. Anim. Resour. 2018, 38, 768–779. [Google Scholar] [CrossRef]
- Mau, J.-L.; Lin, H.-C.; Ma, J.-T.; Song, S.-F. Non-Volatile Taste Components of Several Speciality Mushrooms. Food Chem. 2001, 73, 461–466. [Google Scholar] [CrossRef]
- Teklit, G.A. Chemical Composition and Nutritional Value of the Most Widely Used Mushrooms Cultivated in Mekelle Tigray Ethiopia. J. Nutr. Food Sci. 2015, 5, 1. [Google Scholar] [CrossRef]
- Michael, H.W.; Bultosa, G.; Pant, L.M. Nutritional Contents of Three Edible Oyster Mushrooms Grown on Two Substrates at Haramaya, Ethiopia, and Sensory Properties of Boiled Mushroom and Mushroom Sauce: Nutrient of Edible Oyster Mushrooms. Int. J. Food Sci. Technol. 2011, 46, 732–738. [Google Scholar] [CrossRef]
- Crisan, E.V.; Sands, A. Nutritional Value. In Biology and Cultivation of Edible Mushrooms; Chang, S.T., Hayer, W.A., Eds.; Academic Press: New York, NY, USA, 1978; pp. 138–168. [Google Scholar]
- Ghosh, K. A Review: Edible Mushrooms as Source of Dietary Fiber and Its Health Effects. J. Phys. Sci. 2016, 21, 129–137. [Google Scholar]
- Koç, E.; Karayiğit, B. Assessment of Biofortification Approaches Used to Improve Micronutrient-Dense Plants that Are a Sustainable Solution to Combat Hidden Hunger. J. Soil Sci. Plant Nutr. 2021, 4, 1–26. [Google Scholar] [CrossRef]
- Kora, A.J. Nutritional and Antioxidant Significance of Selenium-Enriched Mushrooms. Bull. Natl. Res. Cent. 2020, 44, 34. [Google Scholar] [CrossRef]
- Siwulski, M.; Budzyńska, S.; Rzymski, P.; Gąsecka, M.; Niedzielski, P.; Kalač, P.; Mleczek, M. The Effects of Germanium and Selenium on Growth, Metalloid Accumulation and Ergosterol Content in Mushrooms: Experimental Study in Pleurotus ostreatus and Ganoderma lucidum. Eur. Food Res. Technol. 2019, 245, 1799–1810. [Google Scholar] [CrossRef]
- Hu, T.; Liang, Y.; Zhao, G.; Wu, W.; Li, H.; Guo, Y. Selenium Biofortification and Antioxidant Activity in Cordyceps militaris Supplied with Selenate, Selenite, or Selenomethionine. Biol. Trace Elem. Res. 2019, 187, 553–561. [Google Scholar] [CrossRef]
- Hu, T.; Hui, G.; Li, H.; Guo, Y. Selenium Biofortification in Hericium erinaceus (Lion’s Mane Mushroom) and Its in Vitro Bioaccessibility. Food Chem. 2020, 331, 127287. [Google Scholar] [CrossRef]
- Xu, M.; Zhu, S.; Wang, L.; Wei, Z.; Zhao, L.; Shi, G.; Ding, Z. Influence of Selenium Biofortification on the Growth and Bioactive Metabolites of Ganoderma lucidum. Foods 2021, 10, 1860. [Google Scholar] [CrossRef] [PubMed]
- El-Ramady, H.; Abdalla, N.; Elbasiouny, H.; Elbehiry, F.; Elsakhawy, T.; Omara, A.E.-D.; Amer, M.; Bayoumi, Y.; Shalaby, T.A.; Eid, Y.; et al. Nano-Biofortification of Different Crops to Immune against COVID-19: A Review. Ecotoxicol. Environ. Saf. 2021, 222, 112500. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Zhao, J.; Wang, L.; Liu, Q.; Fan, Y.; Li, B.; Yu, Y.-L.; Chen, C.; Li, Y.-F. Using Nano-Selenium to Combat Coronavirus Disease 2019 (COVID-19)? Nano Today 2021, 36, 101037. [Google Scholar] [CrossRef]
- Hyde, K.D.; Xu, J.; Rapior, S.; Jeewon, R.; Lumyong, S.; Niego, A.G.T.; Abeywickrama, P.D.; Aluthmuhandiram, J.V.S.; Brahamanage, R.S.; Brooks, S.; et al. The Amazing Potential of Fungi: 50 Ways We Can Exploit Fungi Industrially. Fungal Divers. 2019, 97, 1–136. [Google Scholar] [CrossRef]
- Panda, S.K.; Luyten, W. Medicinal Mushrooms: Clinical Perspective and Challenges. Drug Discov. Today 2021, 27, 636–651. [Google Scholar] [CrossRef] [PubMed]
- Bertollo, A.G.; Mingoti, M.E.D.; Plissari, M.E.; Betti, G.; Roman Junior, W.A.; Luzardo, A.R.; Ignácio, Z.M. Agaricus blazei Murrill Mushroom: A Review on the Prevention and Treatment of Cancer. Pharmacol. Res. Mod. Chin. Med. 2022, 2, 100032. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, H.; Cai, M.; Liang, X.; Wu, X.; Wang, A.; Chen, X.; Li, X.; Xiao, C.; Huang, L.; et al. Whole Genome Sequencing of an Edible and Medicinal Mushroom, Russula griseocarnosa, and Its Association with Mycorrhizal Characteristics. Gene 2022, 808, 145996. [Google Scholar] [CrossRef]
- He, P.-Y.; Hou, Y.-H.; Yang, Y.; Li, N. The Anticancer Effect of Extract of Medicinal Mushroom Sanghuangprous vaninii against Human Cervical Cancer Cell via Endoplasmic Reticulum Stress-Mitochondrial Apoptotic Pathway. J. Ethnopharmacol. 2021, 279, 114345. [Google Scholar] [CrossRef]
- Li, Y.-M.; Zhong, R.-F.; Chen, J.; Luo, Z.-G. Structural Characterization, Anticancer, Hypoglycemia and Immune Activities of Polysaccharides from Russula virescens. Int. J. Biol. Macromol. 2021, 184, 380–392. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.; Zhang, J.; Li, Z.; Liu, H.; Wang, Y. Traditional Uses, Chemical Components and Pharmacological Activities of the Genus Ganoderma P. Karst.: A Review. RSC Adv. 2020, 10, 42084–42097. [Google Scholar] [CrossRef]
- Ren, L.; Zhang, J.; Zhang, T. Immunomodulatory Activities of Polysaccharides from Ganoderma on Immune Effector Cells. Food Chem. 2021, 340, 127933. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-J.; Chen, B.-S.; Dai, H.-Q.; Ren, J.-W.; Zhou, L.-W.; Wu, S.-H.; Liu, H.-W. Sesquiterpenes and Polyphenols with Glucose-Uptake Stimulatory and Antioxidant Activities from the Medicinal Mushroom Sanghuangporus sanghuang. Chin. J. Nat. Med. 2021, 19, 693–699. [Google Scholar] [CrossRef]
- Kurd-Anjaraki, S.; Ramezan, D.; Ramezani, S.; Samzadeh-Kermani, A.; Pirnia, M.; Shahi, B.Y. Potential of Waste Reduction of Agro-Biomasses through Reishi Medicinal Mushroom (Ganoderma lucidum) Production Using Different Substrates and Techniques. Acta Ecol. Sin. 2021, 42, 90–101. [Google Scholar] [CrossRef]
- Hyder, M.S.; Dutta, S.D. Mushroom-Derived Polysaccharides as Antitumor and Anticancer Agent: A Concise Review. Biocatal. Agric. Biotechnol. 2021, 35, 102085. [Google Scholar] [CrossRef]
- Mirmazloum, I.; Ladányi, M.; Omran, M.; Papp, V.; Ronkainen, V.-P.; Pónya, Z.; Papp, I.; Némedi, E.; Kiss, A. Co-Encapsulation of Probiotic Lactobacillus acidophilus and Reishi Medicinal Mushroom (Ganoderma lingzhi) Extract in Moist Calcium Alginate Beads. Int. J. Biol. Macromol. 2021, 192, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Krakowska, A.; Zięba, P.; Włodarczyk, A.; Kała, K.; Sułkowska-Ziaja, K.; Bernaś, E.; Sękara, A.; Ostachowicz, B.; Muszyńska, B. Selected Edible Medicinal Mushrooms from Pleurotus Genus as an Answer for Human Civilization Diseases. Food Chem. 2020, 327, 127084. [Google Scholar] [CrossRef] [PubMed]
- Kushairi, N.; Tarmizi, N.A.K.A.; Phan, C.W.; Macreadie, I.; Sabaratnam, V.; Naidu, M.; David, P. Modulation of Neuroinflammatory Pathways by Medicinal Mushrooms, with Particular Relevance to Alzheimer’s Disease. Trends Food Sci. Technol. 2020, 104, 153–162. [Google Scholar] [CrossRef]
- Gründemann, C.; Reinhardt, J.K.; Lindequist, U. European Medicinal Mushrooms: Do They Have Potential for Modern Medicine?—An Update. Phytomedicine 2020, 66, 153131. [Google Scholar] [CrossRef]
- Neergheen, V.S.; Hip Kam, A.; Pem, Y.; Ramsaha, S.; Bahorun, T. Regulation of Cancer Cell Signaling Pathways as Key Events for Therapeutic Relevance of Edible and Medicinal Mushrooms. Semin. Cancer Biol. 2020, 80, 145–156. [Google Scholar] [CrossRef]
- Karuppaiya, P.; Akbar, A.K. Mycotherapy of Antrodia Salmonea: A Taiwanese Medicinal Mushroom. In Medicinal Mushrooms; Agrawal, D.C., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 409–419. ISBN 9789811363818. [Google Scholar]
- Fung, S.-Y.; Tan, C.-S. Tiger Milk Mushroom (The Lignosus trinity) in Malaysia: A Medicinal Treasure Trove. In Medicinal Mushrooms; Agrawal, D.C., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 349–369. ISBN 9789811363818. [Google Scholar]
- Yongabi, K.A. African Medicinal Mushrooms: Source of Biopharmaceuticals for the Treatment of Noncommunicable Diseases—A Review. In Medicinal Mushrooms; Agrawal, D.C., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 335–347. ISBN 9789811363818. [Google Scholar]
- Venturella, G.; Saporita, P.; Gargano, M.L. Current Research on Medicinal Mushrooms in Italy. In Medicinal Mushrooms; Agrawal, D.C., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 317–333. ISBN 9789811363818. [Google Scholar]
- Chan, X.H.; Sabaratnam, V.; Abdullah, N.; Phan, C.-W. A 53-Year Bibliometric and Scientometric Analysis of Research in Culinary and Medicinal Mushrooms. Int. J. Med. Mushrooms 2020, 22, 521–534. [Google Scholar] [CrossRef]
- Buranrat, B.; Sangdee, K.; Thammawat, S.; Sangdee, A. Mechanisms of Crude Protein from Medicinal Mushroom Ophiocordyceps sobolifera against Human Breast MCF-7 Cancer Cells. Biologia 2020, 75, 1759–1768. [Google Scholar] [CrossRef]
- Rizzo, G.; Goggi, S.; Giampieri, F.; Baroni, L. A Review of Mushrooms in Human Nutrition and Health. Trends Food Sci. Technol. 2021, 117, 60–73. [Google Scholar] [CrossRef]
- Khursheed, R.; Singh, S.K.; Wadhwa, S.; Gulati, M.; Awasthi, A. Therapeutic Potential of Mushrooms in Diabetes Mellitus: Role of Polysaccharides. Int. J. Biol. Macromol. 2020, 164, 1194–1205. [Google Scholar] [CrossRef] [PubMed]
- Deveci, E.; Çayan, F.; Tel-Çayan, G.; Duru, M.E. Inhibitory Activities of Medicinal Mushrooms on α-Amylase and α-Glucosidase-Enzymes Related to Type 2 Diabetes. S. Afr. J. Bot. 2021, 137, 19–23. [Google Scholar] [CrossRef]
- Krittanawong, C.; Isath, A.; Hahn, J.; Wang, Z.; Fogg, S.E.; Bandyopadhyay, D.; Jneid, H.; Virani, S.S.; Tang, W.H.W. Mushroom Consumption and Cardiovascular Health: A Systematic Review. Am. J. Med. 2021, 134, 637–642.e2. [Google Scholar] [CrossRef]
- Lin, D.S. Case Report: Resolution of Rheumatoid Arthritis in a Patient Consuming Psilocybin Mushrooms. J. Healthc. 2020, 3, 25–32. [Google Scholar] [CrossRef]
- Ellan, K.; Thayan, R.; Raman, J.; Hidari, K.I.P.J.; Ismail, N.; Sabaratnam, V. Anti-Viral Activity of Culinary and Medicinal Mushroom Extracts against Dengue Virus Serotype 2: An in-Vitro Study. BMC Complementary Altern. Med. 2019, 19, 260. [Google Scholar] [CrossRef]
- Pradeep, P.; Manju, V.; Ahsan, M.F. Antiviral Potency of Mushroom Constituents. In Medicinal Mushrooms; Agrawal, D.C., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 275–297. ISBN 9789811363818. [Google Scholar]
- Shahzad, F.; Anderson, D.; Najafzadeh, M. The Antiviral, Anti-Inflammatory Effects of Natural Medicinal Herbs and Mushrooms and SARS-CoV-2 Infection. Nutrients 2020, 12, 2573. [Google Scholar] [CrossRef]
- Lee, W.; Fujihashi, A.; Govindarajulu, M.; Ramesh, S.; Deruiter, J.; Majrashi, M.; Almaghrabi, M.; Nadar, R.M.; Moore, T.; Agrawal, D.C.; et al. Role of Mushrooms in Neurodegenerative Diseases. In Medicinal Mushrooms; Agrawal, D.C., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 223–249. ISBN 9789811363818. [Google Scholar]
- Živković, L.; Bajić, V.; Bruić, M.; Borozan, S.; Popić, K.; Topalović, D.; Santibanez, J.; Spremo-Potparević, B. Antigenotoxic and Antioxidant Potential of Medicinal Mushrooms (Immune assist) against DNA Damage Induced by Free Radicals—An in Vitro Study. Mutat. Res./Genet. Toxicol. Environ. Mutagenesis 2019, 845, 403078. [Google Scholar] [CrossRef]
- Khatun, M.A.; Sato, S.; Konishi, T. Obesity Preventive Function of Novel Edible Mushroom, Basidiomycetes-X (Echigoshirayukidake): Manipulations of Insulin Resistance and Lipid Metabolism. J. Tradit. Complementary Med. 2020, 10, 245–251. [Google Scholar] [CrossRef]
- Niego, A.G.; Rapior, S.; Thongklang, N.; Raspé, O.; Jaidee, W.; Lumyong, S.; Hyde, K.D. Macrofungi as a Nutraceutical Source: Promising Bioactive Compounds and Market Value. JoF 2021, 7, 397. [Google Scholar] [CrossRef] [PubMed]
- Venturella, G.; Ferraro, V.; Cirlincione, F.; Gargano, M.L. Medicinal Mushrooms: Bioactive Compounds, Use, and Clinical Trials. IJMS 2021, 22, 634. [Google Scholar] [CrossRef]
- Shah, A.; ul Ashraf, Z.; Gani, A.; Masoodi, F.A.; Gani, A. β-Glucan from Mushrooms and Dates as a Wall Material for Targeted Delivery of Model Bioactive Compound: Nutraceutical Profiling and Bioavailability. Ultrason. Sonochem. 2022, 82, 105884. [Google Scholar] [CrossRef] [PubMed]
- Moumita, S.; Das, B. Assessment of the Prebiotic Potential and Bioactive Components of Common Edible Mushrooms in India and Formulation of Synbiotic Microcapsules. LWT 2022, 156, 113050. [Google Scholar] [CrossRef]
- Gąsecka, M.; Siwulski, M.; Magdziak, Z.; Budzyńska, S.; Stuper-Szablewska, K.; Niedzielski, P.; Mleczek, M. The Effect of Drying Temperature on Bioactive Compounds and Antioxidant Activity of Leccinum scabrum (Bull.) Gray and Hericium erinaceus (Bull.) Pers. J. Food Sci. Technol. 2020, 57, 513–525. [Google Scholar] [CrossRef]
- Saetang, N.; Rattanapot, T.; Manmai, N.; Amornlerdpison, D.; Ramaraj, R.; Unpaprom, Y. Effect of Hot Water Extraction Process on Schizophyllan from Split Gill Mushroom. Biomass Convers. Biorefin. 2022. [Google Scholar] [CrossRef]
- Khovpachev, A.A.; Basharin, V.A.; Chepur, S.V.; Volobuev, S.V.; Yudin, M.A.; Gogolevsky, A.S.; Nikiforov, A.S.; Kalinina, L.B.; Tyunin, M.A. Actual Concepts of Higher Fungi’s Toxins: Simple Nitrogen-Containing Compounds. Biol. Bull. Rev. 2021, 11, 198–212. [Google Scholar] [CrossRef]
- Nieminen, P.; Mustonen, A.-M. Toxic Potential of Traditionally Consumed Mushroom Species—A Controversial Continuum with Many Unanswered Questions. Toxins 2020, 12, 639. [Google Scholar] [CrossRef]
- Golovko, O.; Kaczmarek, M.; Asp, H.; Bergstrand, K.-J.; Ahrens, L.; Hultberg, M. Uptake of Perfluoroalkyl Substances, Pharmaceuticals, and Parabens by Oyster Mushrooms (Pleurotus ostreatus) and Exposure Risk in Human Consumption. Chemosphere 2022, 291, 132898. [Google Scholar] [CrossRef]
- Ohta, H.; Watanabe, D.; Nomura, C.; Saito, D.; Inoue, K.; Miyaguchi, H.; Harada, S.; Aita, Y. Toxicological Analysis of Satratoxins, the Main Toxins in the Mushroom Trichoderma Cornu-Damae, in Human Serum and Mushroom Samples by Liquid Chromatography–Tandem Mass Spectrometry. Forensic Toxicol. 2021, 39, 101–113. [Google Scholar] [CrossRef]
- Barea-Sepúlveda, M.; Espada-Bellido, E.; Ferreiro-González, M.; Bouziane, H.; López-Castillo, J.G.; Palma, M.; Barbero, G.F. Toxic Elements and Trace Elements in Macrolepiota procera Mushrooms from Southern Spain and Northern Morocco. J. Food Compos. Anal. 2022, 108, 104419. [Google Scholar] [CrossRef]
- Wong, J.H.; Ng, T.B.; Chan, H.H.L.; Liu, Q.; Man, G.C.W.; Zhang, C.Z.; Guan, S.; Ng, C.C.W.; Fang, E.F.; Wang, H.; et al. Mushroom Extracts and Compounds with Suppressive Action on Breast Cancer: Evidence from Studies Using Cultured Cancer Cells, Tumor-Bearing Animals, and Clinical Trials. Appl. Microbiol. Biotechnol. 2020, 104, 4675–4703. [Google Scholar] [CrossRef] [PubMed]
- Oly-Alawuba, N.; Obiakor-Okeke, P.N. Antinutrient Profile of Three Mushroom Varieties Consumed in Amaifeke, Orlu, Imo State. Food Sci. Qual. Manag. 2014, 32, 1. [Google Scholar]
- Kim, O.-H.; Booth, C.J.; Choi, H.S.; Lee, J.; Kang, J.; Hur, J.; Jung, W.J.; Jung, Y.-S.; Choi, H.J.; Kim, H.; et al. High-Phytate/Low-Calcium Diet Is a Risk Factor for Crystal Nephropathies, Renal Phosphate Wasting, and Bone Loss. eLife 2020, 9, e52709. [Google Scholar] [CrossRef] [PubMed]
- López-Moreno, M.; Garcés-Rimón, M.; Miguel, M. Antinutrients: Lectins, Goitrogens, Phytates and Oxalates, Friends or Foe? J. Funct. Foods 2022, 89, 104938. [Google Scholar] [CrossRef]
- Petroski, W.; Minich, D.M. Is There Such a Thing as “Anti-Nutrients”? A Narrative Review of Perceived Problematic Plant Compounds. Nutrients 2020, 12, 2929. [Google Scholar] [CrossRef]
- Elsakhawy, T.; Omara, A.E.-D.; Abowaly, M.; El-Ramady, H.; Badgar, K.; Llanaj, X.; Törős, G.; Hajdú, P.; Prokisch, J. Green Synthesis of Nanoparticles by Mushrooms: A Crucial Dimension for Sustainable Soil Management. Sustainability 2022, 14, 4328. [Google Scholar] [CrossRef]
- Vetter, J. Chitin Content of Cultivated Mushrooms Agaricus bisporus, Pleurotus ostreatus and Lentinula edodes. Food Chem. 2007, 102, 6–9. [Google Scholar] [CrossRef]
- Ba, D.M.; Gao, X.; Al-Shaar, L.; Muscat, J.; Chinchilli, V.M.; Ssentongo, P.; Zhang, X.; Liu, G.; Beelman, R.B.; Richie, J.P. Prospective Study of Dietary Mushroom Intake and Risk of Mortality: Results from Continuous National Health and Nutrition Examination Survey (NHANES) 2003-2014 and a Meta-Analysis. Nutr. J. 2021, 20, 80. [Google Scholar] [CrossRef]
- Urbina-Salazar, A. del R.; Inca-Torres, A.R.; Falcón-García, G.; Carbonero-Aguilar, P.; Rodríguez-Morgado, B.; del Campo, J.A.; Parrado, J.; Bautista, J. Chitinase Production by Trichoderma Harzianum Grown on a Chitin-Rich Mushroom Byproduct Formulated Medium. Waste Biomass Valorization 2019, 10, 2915–2923. [Google Scholar] [CrossRef]
- Jones, M.; Kujundzic, M.; John, S.; Bismarck, A. Crab vs. Mushroom: A Review of Crustacean and Fungal Chitin in Wound Treatment. Mar. Drugs 2020, 18, 64. [Google Scholar] [CrossRef] [PubMed]
- Boureghda, Y.; Satha, H.; Bendebane, F. Chitin–Glucan Complex from Pleurotus ostreatus Mushroom: Physicochemical Characterization and Comparison of Extraction Methods. Waste Biomass Valorization 2021, 12, 6139–6153. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, L.; Qu, H.; Zhou, H.; Yang, H.; Chen, H. Physicochemical Characterization, Adsorption Function and Prebiotic Effect of Chitin-Glucan Complex from Mushroom Coprinus comatus. Int. J. Biol. Macromol. 2022, 206, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Yoshida, S.; Mitani, N.; Egusa, M.; Takagi, M.; Izawa, H.; Matsumoto, T.; Kaminaka, H.; Ifuku, S. Disease Resistance and Growth Promotion Activities of Chitin/Cellulose Nanofiber from Spent Mushroom Substrate to Plant. Carbohydr. Polym. 2022, 284, 119233. [Google Scholar] [CrossRef]
- Mat Zin, M.I.; Jimat, D.N.; Wan Nawawi, W.M.F. Physicochemical Properties of Fungal Chitin Nanopaper from Shiitake (L. edodes), Enoki (F. velutipes) and Oyster Mushrooms (P. ostreatus). Carbohydr. Polym. 2022, 281, 119038. [Google Scholar] [CrossRef]
- Khoo, S.C.; Ma, N.L.; Peng, W.X.; Ng, K.K.; Goh, M.S.; Chen, H.L.; Tan, S.H.; Lee, C.H.; Luang-In, V.; Sonne, C. Valorisation of Biomass and Diaper Waste into a Sustainable Production of the Medical Mushroom Lingzhi Ganoderma Lucidum. Chemosphere 2022, 286, 131477. [Google Scholar] [CrossRef]
- Patocka, J.; Wu, R.; Nepovimova, E.; Valis, M.; Wu, W.; Kuca, K. Chemistry and Toxicology of Major Bioactive Substances in Inocybe Mushrooms. IJMS 2021, 22, 2218. [Google Scholar] [CrossRef]





| Environmental Conditions | Spawn Run | Primordia Formation | Fruitbody Development |
|---|---|---|---|
| Temperature | 21–27 °C | 10–21 °C | 10–24 °C |
| Relative Humidity | 85–95% | 95–100% | 80–90% |
| Light | 50–100 lux | 1000–1500 lux | 1000–1500 lux |
| CO2 level | 5000–20,000 ppm; 1 FAE/h | 500–2000 ppm; 4–8 FAE/h | <1000–2000 ppm; 4–8 FAE/h |
| Duration | 2–8 weeks | 3–12 days | 5–8 days |
| Mushroom Species Name | Common English Name | Family |
|---|---|---|
| Agaricus pilatianus (Bohus) Bohus | Basionym | Agaricaceae |
| Agaricus phaeolepidotus F.H. Møller | Agaric | Agaricaceae |
| Agaricus praeclaresquamosus A.E. Freeman | Ink-smelling mushroom | Agaricaceae |
| Agaricus xanthoderma var. xanthoderma | Carbolic mushroom | Agaricaceae |
| Amanita gemmate (Fr.) Gillet | Yellow agaric | Amanitaceae |
| Amanita muscarina | Fly agaric | Amanitaceae |
| Amanita pantherine (DC ex Fr.) Secr. | Panther | Amanitaceae |
| Amanita phalloides | Killer agaric | Amanitaceae |
| Boletus calopus Fr. | Farkino | Boletaceae |
| Boletus radicans Pers. | Rooting bolete | Boletaceae |
| Boletus satanas Lenz | Devil’s bolete | Boletaceae |
| Clitocybe dealbata (Sowerby) P. Kumm | Field or ivory funnel | Tricholomataceae |
| Clitocybe cerussata (Fr.) P Kumm | Wax white funnel | Tricholomataceae |
| Cortinarius orellanus Fr. | Fool’s webcap | Cortinariaceae |
| Entoloma sinuatum | Livid pinkgill | Entolomataceae |
| Entoloma rhodopolium (Fr.) P. Kumm. | Wood pinkgill | Entolomataceae |
| Gyromitra fastigiata (Krombh.) Rehm | Red-brown priest mushrooms | Discinaceae |
| Gyromitra esculenta (Pers.) Fr. | False Morel | Discinaceae |
| Galerina marginata (Batsch) Kühner | Funeral Bell | Hymenogastraceae |
| Helvella crispa Scop. | Curly paprika mushrooms | Helvellaceae |
| Helvella leucopus Pers. | Pale foot saddle | Helvellaceae |
| Hygrocybe conica (Schaeff.) P. Kumm | Blackening waxcap | Hygrophoraceae |
| Hypholoma fasciculare (Huds.) P. Kumm | Basionym | Strophariaceae |
| Hebeloma synapizans (Paulet) Gillet | Bitter poisonpie | Hymenogastraceae |
| Inocybe asterospora Quelet | Star spore susula | Inocybaceae |
| Inocybe fastigiate (Schaeff.) Quél. | Kerti susulyula | Inocybaceae |
| Inocybe patouillardii Bres. | Brick red susula | Inocybaceae |
| Lepiota cristata (Bolton) P. Kumm | Stinky dapperling | Agaricaceae |
| Lepiota brunneoincarnata | Meat brown venison | Agaricaceae |
| Lepiota pseudohelveola Kühner ex Hora | Roe deer mushrooms | Agaricaceae |
| Mycena pura (Pers.) P. Kumm | Lilac Bonnet | Mycenaceae |
| Omphalotus olearius (DC.) Singer | Illuminated funnel (Basionym) | Omphalotaceae |
| Psilocybe semilanceata (Fr.) P. Kumm | Magic mushroom | Hymenogastraceae |
| Paxillus involutus (Fr.) Fr. | Brown rollrim | Paxillaceae |
| Tricholoma sulphureum (Bull.) P. Kumm | Stinky Knight | Tricholomataceae |
| Ramaria Formosa (Pers.) Quél | Salmon coral | Gomphaceae |
| Russula emetica (Schaeff.) Pers. | Sickener | Russulaceae |
| Scleroderma citrinum Pers. | Common Earthball | Sclerodermataceae |
| Sarcosphaera eximia var. nivea M.M. Moser | Tulip cup mushrooms | Pezizaceae |
| Tricholoma pardinum (Pers.) Quél. | Panther (Leopard Knight) | Tricholomataceae |
| Tricholoma saponaceum (Fr.) P. Kumm | Soap scent | Tricholomataceae |
| Toxins | Poisonous Mushrooms | Clinical Features |
|---|---|---|
| Group 1: Cytotoxic mushroom poisoning | ||
| α-amanitin | Amanita phalloides, A. virosa, A. verna, A. exitialis | Liver damage 1–3 days after ingestion due to hepatotoxic and nephrotoxic effects |
| Amanitin | Amanita verna, A. virosa, A. phalloides, Lepiota helveola, Galerina marginata | Gastrointestinal disturbances; liver and renal failure |
| Orellanine | Amanita smithiana, A. pseudoporphyria | Kidney failure (early primary nephrotoxicity) |
| Cortinarius orellanus, C. rubellus | Delayed primary nephrotoxicity | |
| Group 2: Neurotoxic mushroom poisoning | ||
| Muscarine | Omphalotus, Clitocybe cerussata, C. dealbata, Inocybe fibrosa, I. hystrix, I. geophylla, I. rimosa, I. sambucina | Respiratory failure, bradycardia, miosis, salivation, lacrimation, diarrhea and bronchospasm perspiration, lachrymation |
| Psilocybin and psilocin | Psilocybe caerulescens, P. cubensis, P. semilanceata, Panaeolus cinctulus | Euphoria, hallucinations, tachycardia and blood pressure, mydriasis, tremors, and fever |
| Ibotenic acid | Amanita muscaria, A. pantherina, A. gemmate, A. ibotengutake, | Neurotoxicity, continuous periods of excitation and inhibition in the nervous system |
| Muscinol | Amanita muscaria, A. pantherina, A. gemmate, A. cothurnata, A. regalis | Central nervous system depression and hallucinations |
| Group 3: Myotoxic mushroom poisoning | ||
| Bolesatine | Rubroboletus satanas (Lenz), R. lupinus, R. legaliae | Non-lethal; causes gastrointestinal irritation, vomiting, nausea |
| Russuphelins | Russula subnigricans | Gastrointestinal disturbances, and renal failure |
| Saponaceolide | Tricholoma equestre, T. terreum | Delayed onset myotoxicity |
| Group 4: Metabolic, endocrine and related toxicity mushroom poisoning | ||
| Coprine | Coprinopsis alopecia, C. atramentaria, C. romagnesiana | Flushing, headache, dyspnea, sweating, arrhythmia, hypotension, and confusion |
| Gyromitrin | Gyromitra esculenta, G. californica, G. ambigua, G. gigas, G. infula | Neurotoxicity, gastrointestinal upset, and destruction of blood cells |
| Polyporic acid | Hapalopilus rutilans | Abdominal pain, nausea, vomiting, diarrhea |
| Trichothecenes | Podostroma cornu-damae | A multi-organ illness, often fatal |
| Group 5: Gastrointestinal irritant mushroom poisoning | ||
| --------- | Chlorophyllum molybdites, Entoloma sinuatum, Tricholoma pardalotum | Gastrointestinal effects without causing other significant effects |
| Group 6: Miscellaneous adverse reactions | ||
| Ergotamine | Claviceps spp. | Deadly can affect the vascular system and lead to loss of limbs and/or cardiac arrest |
| Acromelic acid | Paralepistopsis amoenolens, Clitocybe acromelalga | An erythromelalgia-like syndrome; reddening, swelling, and burning pain in the extremities |
| Hydro-Cyanic Acid | Pleurocybella porrigens, Grifola frondose, Pleurotus eringii | Delayed onset with cramps and coma; re-existing chronic renal failure |
| Lentinan (Shiitake) | Lentinola edodes, Auricularia auricular-judae | Dermatitis, linear rash/whiplike lesions, photosensitivity |
| Phallotoxin | Amanita phalloides | Gastrointestinal upset |
| Paxillus syndrome | Paxillus involutus | Abdominal pain, nausea, vomiting, and acute respiratory failure |
| Mushroom Species Name | Common English Name | Family |
|---|---|---|
| Agaricus arvensis Schaeff. | Horse mushroom | Agaricaceae |
| Agaricus bitorquis (Quél.) Sacc. | Pavement mushroom | Agaricaceae |
| Agaricus bisporus (J.E. Lange) Imbach, white | White cultivated mushroom | Agaricaceae |
| Agaricus bisporus (J.E. Lange) Imbach, brown | Brown cultivated mushroom | Agaricaceae |
| Agaricus sylvaticus Schaeff. | Blushing wood mushroom | Agaricaceae |
| Agrocybe cylindracea (Brig.) Fayod | Poplar mushroom | Strophariaceae |
| Amanita ovoidea (Bull.) Link | Bearded amanita | Amanitaceae |
| Amanita vaginata (Bull.) Lam. | Grisette | Amanitaceae |
| Armillaria mellea (Vahl) P. Kumm. | Honey fungus | Physalacriaceae |
| Auricularia auricula-judae (Fr.) Quél | Black wood ear | Auriculariaceae |
| Cantharellus cibarius Fr. | Chanterelle | Cantharellaceae |
| Clavariadelphus pistillaris (L.) Donk | Giant club | Clavariadelphaceae |
| Clitocybe nebularis (Batsch) P. Kumm. | Clouded funnel | Tricholomataceae |
| Clitopilus prunulus (Scop.) P. Kumm. | The Miller | Entolomataceae |
| Cordyceps militaris (L.) Fr. | Scarlet caterpillar club | Cordycipitaceae |
| Coprinus comatus (O.F. Müll.) Gray | Shaggy inkcap/lawyer’s wig | Agaricaceae |
| Gyroporus castaneus (Bull.) Quél. | Chestnut bolete | Gyroporaceae |
| Helvella acetabulum (L.) Quél. | Vinegar cup | Helvellaceae |
| Hygrophorus marzuolus (Fr.) Bres. | March mushroom | Hygrophoraceae |
| Hygrophorus russula (Schaeff. ex Fr.) Kauffman | Pinkmottle woodwax | Hygrophoraceae |
| Lactarius pubescens Fr. | Bearded milkcap | Russulaceae |
| Lactarius repraesentaneus Britzelm. | Yellow bearded milkcap | Russulaceae |
| Lactarius rufus (Scop.) Fr. | Rufous milkcap | Russulaceae |
| Lactarius scrobiculatus (Scop.) Fr. | Spotted milkcap | Russulaceae |
| Lactarius sanguifluus (Poulet) Fr | Bloody milkcap | Russulaceae |
| Lactarius volemus (Fr.) Fr. | Fishy milkca | Russulaceae |
| Lentinus edodes (Berk.) Pegler | Shiitake | Marasmiaceae |
| Lentinus tigrinus (Bull.) Fr | Tiger sawgill | Hygrophoraceae |
| Lepista irina (Fr.) H. E. Bigelow | Flowery blewit | Tricholomataceae |
| Lepista nuda (Bull.) Cooke | Wood blewit | Tricholomataceae |
| Leucoagaricus leucothites (Vittad.) Wasser | White dapperling | Agaricaceae |
| Lycoperdon molle Pers. | Soft puffball | Lycoperdaceae |
| Lyophyllum decastes (Fr.) Singer | Clustered domecap | Tricholomataceae |
| Pleurotus citrinopileatus Singer | Golden oyster mushroom | Pleurotaceae |
| Pleurotus ostreatus (Jacq. ex Fr.) P. Kumm | Oyster mushroom | Pleurotaceae |
| Polyporus squamosus (Huds.) Fr. | Dryad’s saddle | Hygrophoraceae |
| Macrolepiota mastoidea (Fr.) Singer | Slender parasol | Agaricaceae |
| Neoboletus luridiformis (Rostk.) | Basionym | Boletaceae |
| Neolentinus lepideus (Fr.) Redhead & Ginns | Scaly sawgill | Polyporaceae |
| Otidea cochleata (L.) Fuckel | Brown ear | Pyronemataceae |
| Pluteus petasatus (Fr.) Gillet | Scaly shield | Pluteaceae |
| Pluteus salicinus (Pers.) P. Kumm | Willow shield | Pluteaceae |
| Russula albonigra (Krombh.) Fr. | Menthol brittlegill | Russulaceae |
| Russula delica Fr. | Milk White brittlegill | Russulaceae |
| Russula fellea (Fr.) Fr. | Geranium brittlegill | Russulaceae |
| Russula viscida Kudřna | Viscid brittlegill | Russulaceae |
| Strobilomyces strobilaceus (Scop.) Berk. | Old man of the woods | Boletaceae |
| Suillus bellinii (Inzenga) Kuntze | Champagne bolete | Suillaceae |
| Suillus bovinus (L.) Roussel | Bovine bolete | Suillaceae |
| Suillus luteus (L.) Roussel | Slippery jack | Suillaceae |
| Tricholoma focale (Fr.) Ricken | Booted knight | Tricholomataceae |
| Tricholoma terreum (Schaeff.: Fr.) Kumm | Grey knight | Tricholomataceae |
| Volvopluteus gloiocephalus (DC.) | Basionym | Pluteaceae |
| Volvariella volvacea (Bull.) Singer | Straw mushroom | Pluteaceae |
| Xerocomus subtomentosus (L.) Fr. | Homotypic synonym | Boletaceae |
| Edible Mushrooms | Phenolics | Polysaccharides | Proteins | Triterpenoids | Refs. |
|---|---|---|---|---|---|
| Agaricus bisporus | Gallocatechin | Heteropoly-saccharide ABP | Protein type FIIb-1 | Ergosterol | [98,99,100,101] |
| Boletus edulis | Gallic acid | Polysaccharides (BEBP-1) | β-Trefoil lectin | Boledulins A-C | [98,102,103,104] |
| Cordyceps aegerita | Proto-catechuic acid | Fucogalactan | Ageritin | Bovistols A-C | [105,106,107,108] |
| Coprinus comatus | Flavones, and flavonols | Modified polysaccharide | Laccases | Terpenoids | [109,110,111] |
| Lactarius deliciosus | Syringic acid, vanillic acid | Polysaccharide (LDG-M) | Laccase | Azulene-type sesquiterpene | [112,113,114,115] |
| Pleurotus ostreatus | Caffeic acid, and ferulic acid | Mycelium polysaccharides | Concanavalin A | Ergosterol | [116] |
| Pleurotus eryngii | Cinnamic acid | PEPE-A1 and PEPE-A2 | Laccase | Ergosterol | [117,118] |
| Pleurotus cornucopiae | Gallic acid | β-glucan | Oligopeptides | Ergostane-type sterols | [119,120,121,122] |
| Macrolepiota procera | Proto-catechuic acid | Polysaccharides | β-Trefoil lectin | Lanostane triterpenoids | [103,123,124,125] |
| Russula virescens | Quercetin | SRVPs | Laccase | ---------- | [126,127,128] |
| Tuber melanosporum | Flavonoids, phenols | Exo-poly-saccharides (TP1) | ------------- | Ergosterol | [101,120,121,122,123,124,125,126,127,128,129] |
| Mushroom Species Used in the Study | Moisture (%) | Total Protein (%) | Total Carbo-Hydrate (%) | Crude Fiber (%) | Ash (%) | Total Phenols (%) | Refs. |
|---|---|---|---|---|---|---|---|
| I. Studied many species | |||||||
| Agaricus bisporus, Agrocybe cylindracea, Boletus loyo, Cortinarius lebre, Cyttaria espinosae | 86–96 | 8.56–23.88 | 62.97–83.65 | 7–15 | 5–13 | 0.75–4.72 | [86] |
| Apioperdon pyriforme, Helvella elastica, Morchella conica and Rhizopogon luteolus) | - | 11.5–24.5 | - | 2.6–4.8 | 9.5–14.7 | 5.0–12.3 | [83] |
| Auricularia auricula-judae, A. polytricha, Lactifluus piperatus, Laetiporus sulphureus | 49–88 | 19–56 | 7–18 | 5–11 | 3–8 | 7.3–10.2 | [138] |
| Auricularia auricular, Ganoderma lucidum, Pleurotus citrinopleatus, P. djamor, P. eryngii, P. ostreatus, and P. ostreatus | 4–11 | 20–45 | 11–61 | 5–40 | 6–10 | 1–8 | [85] |
| Agaricus bisporus, Pleurotus ostreatus, Lentinula edodes | 88–92 | 1.7–2.11 | 3–9 | 20–37 | 0.8–1.15 | 1.1–1.5 | [136] |
| Tricholoma, Shiitake mushroom, Pleurotus eryngii, Dictyophora indusiata | 6.9–15.5 | 8.5–36.9 | 0.5–37.3 | 14.4–70.2 | 1.3–10.1 | - | [137] |
| Agaricus bisporus, Boletus edulis, Cantharellus cibarius, Lactarius deliciosus, Leccinum rufom | 82.6–91 | 1.5–3.6 | 3.2–8.3 | - | - | - | [5] |
| Astraeus odoratus, Craterellus aureus, Lentinus edodes, Phaeogyroporus portentosus | - | 13.1–32.8 | 2.79–44.3 | 77.1 | - | - | [142] |
| Agaricus bisporus, Boletus edulis, Calocybe indica, C. gambosa, Grifola frondosa, Flammulina velutipes | - | 18.1–62.8 | 31.1–70.6 | 7.81–32.3 | 3.5–19.7 | - | [135] |
| Pleurotus sajor-caju; Calocybe indica | 87–89 | 1.74–3.4 | 3.37–3.33 | - | 1.2–1.3 | - | [141] |
| II. Studied only one species | |||||||
| Agaricus bisporus | 81.79 | 29.29 | 20.57 | 24.56 | 7.12 | - | [143] |
| Flammulina velutipes | - | 18.42 | 56.37 | 7.81 | 6.33 | - | [144] |
| Grifola frondosa | 83.06 | 21.1 | 58.8 | 10.1 | 7.0 | - | [145] |
| Hericium erinaceus | 95.69 | 23.3 | 57.0 | 7.8 | 9.4 | - | [145] |
| Lactarius deliciosus | 92 | 17.19 | 66.61 | 31.81 | 8.62 | 4.5–13.6 | [140] |
| Lyophyllum decastes | - | 18.31 | 34.36 | 29.02 | 14.20 | - | [146] |
| Pleurotus florida | 87.05 | 34.56 | 31.59 | 11.41 | 7.40 | - | [147] |
| Pleurotus ostreatus | 85.55 | 30.92 | 31.40 | 12.10 | 7.05 | - | [147] |
| Russula delica | - | 26.25 | 34.88 | 15.42 | 17.92 | - | [146] |
| Tremella fuciformis | 91.73 | 4.6 | 94.8 | 1.4 | 0.4 | - | [148] |
| Tricholoma giganteum | - | 16.1 | 70.1 | 4.5 | 5.0 | - | [149] |
| Volvariella volvacea | - | 30.1 | 50.9 | 11.9 | 12.6 | - | [149] |
| Mushroom Species | Study Model | Pharmacological or Medicinal Activities (Disease) | Place | Refs. |
|---|---|---|---|---|
| Agaricus blazei | Animal/cell line | Tumoricidal, anticarcinogenic activity and antimetastatic activity (cancer) | Brazil | [160] |
| Russula griseocarnosa | Not available | Antioxidation, immune regulation, hypoglycemic/hypolipidemic activities (diabetes) | China | [161] |
| Macrolepiota procera, Gymnopus dryophilus | Not available | Anticancer, anti-inflammatory, and reducing cardiovascular diseases (cancer) | Poland | [12] |
| Lentinus edodes and Hypsizygus tessellatus | Cell line | Immunomodulatory activity (inflammatory lung conditions) | Ireland | [96] |
| Sanghuangprous vaninii | Cell line | Anticancer particularly gynecological cancers (cervical cancer) | China | [162] |
| Russula virescens | In vitro | Anticancer, hypoglycemia and immune activities (cancer) | China | [163] |
| Ganoderma lucidum, G. artum and G. tsugae | Cell line | Anti-inflammatory, antitumor, antioxidation, anti-hyperglycemic or hyperlipidemic effects (diabetes) | China | [164,165] |
| Sanghuangporus sanghuang | Cell line | Antioxidant activities and anti-coronavirus (SARS-CoV-2) activity | China | [166] |
| Ganoderma lucidum | Not available | Arthritis, bronchitis, hypertension, gastric ulcer, and nephritis (tumorigenic diseases) | China/Iran | [167] |
| Agaricus spp., Pleurotus spp., Lentinus spp. | Cell line | Antitumor and anticancer agents (cancer) | India | [168] |
| Ganoderma lingzhi, G. applanatum, G. lucidum, and G. tsugae | Simulated gastrointestinal condition | For immunological diseases (gastrointestinal diseases) | Hungary | [169] |
| Pleurotus citrinopileatus, P. djamor, P. eryngii, and P. ostreatus | In vitro | Anti-atherosclerotic properties (high lovastatin content) (cardiovascular diseases and cancers) | Poland | [170] |
| Armillaria mellea, Antrodia camphorata, Hericium erinaceus, Ganoderma spp. | In vitro/in vivo | Anti-neuroinflammatory activities (Alzheimer’s disease) | Malaysia | [171] |
| Calvatia gigantea, Fomes fomentarius, Phallus impudicus | Cell culture | Anti -bacterial, antitumor, antioxidative and anti-inflammatory (cancer) | European Union | [172] |
| Agaricus bisporus, Ganoderma frondosa, Lentinula edodes | Cell line | Anticancer (cancer) | Mauritius | [173] |
| Antrodia salmonea | Cell line | Antioxidant, anti-inflammatory, antitumor, and antiatherosclerosis activity (atherosclerosis) | Taiwan | [174] |
| Lignosus rhinocerus, L. tigris, L. cameronensis | Cell line | Anticancer, anti-inflammatory, and antioxidative activity (cancer) | Malaysia | [175] |
| Species of Agaricus, Ganoderma, Pleurotus | Animal | Anticancer, alleviation of osteoarthritis (cancer and osteoarthritis) | Cameroon/Nigeria | [176] |
| Ganoderma applanatum, Pleurotus eryngii, Lentinula edodes | Clinical trials on human and animals | Antioxidant, antibacterial activity, antitumor, and antiproliferative activity (Alzheimer’s disease, and human colon cancer) | Italy | [177] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Ramady, H.; Abdalla, N.; Badgar, K.; Llanaj, X.; Törős, G.; Hajdú, P.; Eid, Y.; Prokisch, J. Edible Mushrooms for Sustainable and Healthy Human Food: Nutritional and Medicinal Attributes. Sustainability 2022, 14, 4941. https://doi.org/10.3390/su14094941
El-Ramady H, Abdalla N, Badgar K, Llanaj X, Törős G, Hajdú P, Eid Y, Prokisch J. Edible Mushrooms for Sustainable and Healthy Human Food: Nutritional and Medicinal Attributes. Sustainability. 2022; 14(9):4941. https://doi.org/10.3390/su14094941
Chicago/Turabian StyleEl-Ramady, Hassan, Neama Abdalla, Khandsuren Badgar, Xhensila Llanaj, Gréta Törős, Peter Hajdú, Yahya Eid, and József Prokisch. 2022. "Edible Mushrooms for Sustainable and Healthy Human Food: Nutritional and Medicinal Attributes" Sustainability 14, no. 9: 4941. https://doi.org/10.3390/su14094941
APA StyleEl-Ramady, H., Abdalla, N., Badgar, K., Llanaj, X., Törős, G., Hajdú, P., Eid, Y., & Prokisch, J. (2022). Edible Mushrooms for Sustainable and Healthy Human Food: Nutritional and Medicinal Attributes. Sustainability, 14(9), 4941. https://doi.org/10.3390/su14094941











