From Organic Wastes and Hydrocarbons Pollutants to Polyhydroxyalkanoates: Bioconversion by Terrestrial and Marine Bacteria
Abstract
:1. Introduction
2. Bioconversion of Organic Wastes to PHA
2.1. Bioprocess for PHA Production from Terrestrial Bacterial Strains
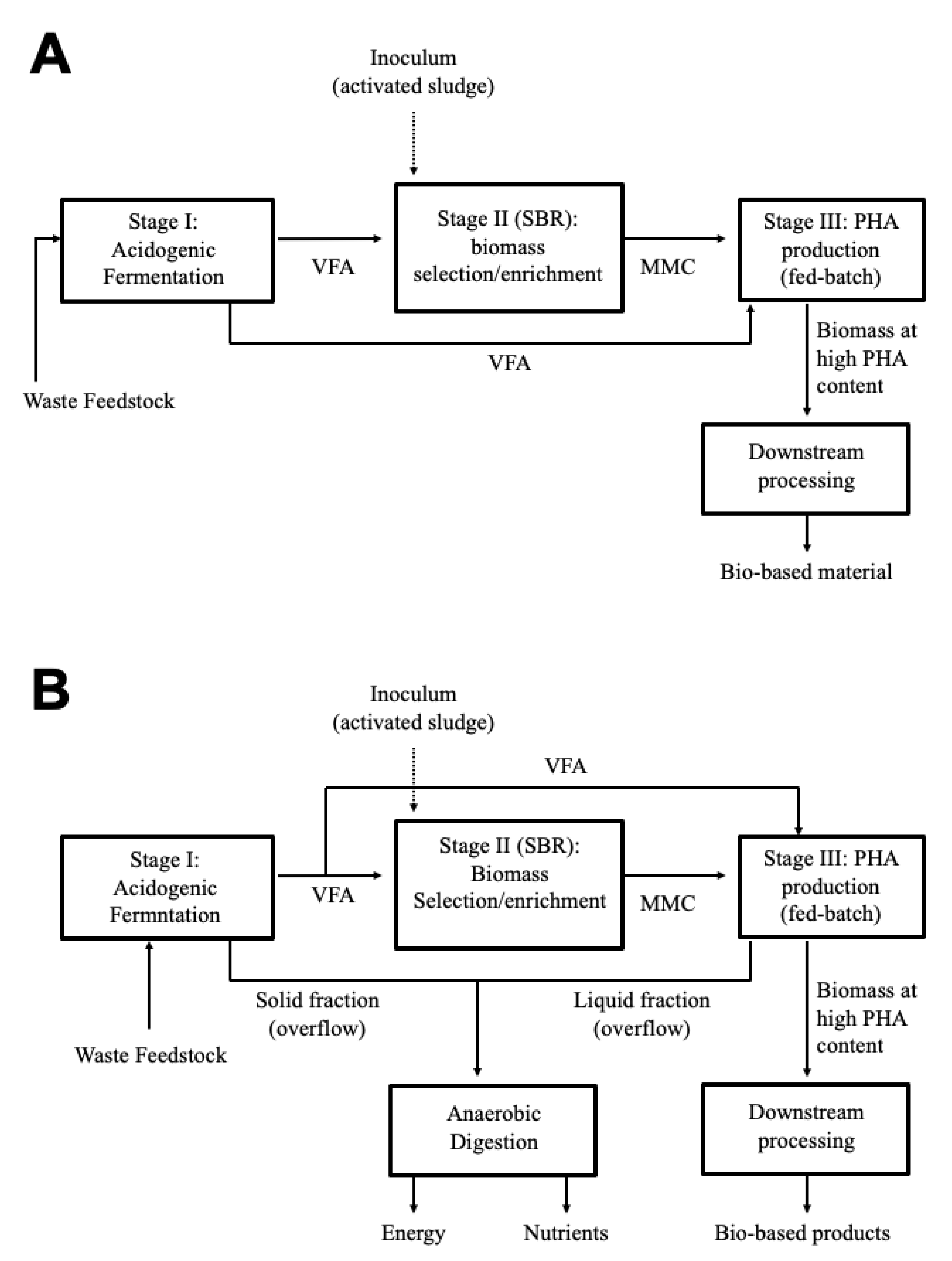
2.1.1. Role of the “Feast and Famine Regime” in MMC-PHA Production from Waste Feedstock
2.1.2. Terrestrial PHA-Accumulating Bacteria in Open Mixed Culture
| Main Microorganisms | Feedstock | Operating Parameters | Biomass PHA Content (wt%) and PHA Composition (%) | Reference |
|---|---|---|---|---|
| Thauera 48.9%, Hypomonas 3.9%, Aquimonas 1.8% | HAc rich wastewater (synthetic) | HRT = 2 d SRT = 10 d OLR ≈ 1.5 gCOD/(L d) | - 3HB/3HV: 100 wt% | [70] |
| Bdellovibrio bacteriovorus (3.5%) Thauera (84%) | fermented sewage sludge/wet air oxidation of sewage sludge: HAc 2734 mg/L, HPr 460 mg/L | HRT = 1 d SRT = 4 d | 41% gPHA/gVSS; 3HB/3HV: 77/23 wt% | [71] |
| Rhodobacter and Roseobacter 20–50%, Amaricoccus 20–50%, Paracoccus 5–20%, Zoogloea 20–50%, Plasticicumulans 5–20% | sewage sludge and fermented fruit waste: HLac 0.05 g/gCODSOL, HAc 0.95, HPr 0.07, EtOH 0.48, HBut 2.25, HVal 0.17, HCap 9.97. | HRT = 1 d SRT = 4 d OLR = 3 gCOD/(L d) | - 3HB/3HV/3HH: 33/1/66 wt% | [42] |
| Paracoccus up to 87.2%, Lampopredia up to 33.0%, Rhodobacteraceae up to 21.7%, Rhizobiales Incertae Sedis up to 18.5%, Amaricoccus up to 9.6%, Thiothrix up to 14.9%, Shinella up to 10%, Leucobacter up to 10.5%, Paracoccus up to 9.6%, Gemmobacter up to 10.7% | fermented fruit waste: HLac 2%, HAc 31%, HPr 13%, EtOH 9%, HBut 68%, HVal 12%. | HRT = 2 d SRT = 2–4 d OLR = 2–14 gCOD/(L d) | 55–75% gPHA/gVSS 3HB/3HV: 84–87/13–16 wt% | [52] |
| Paracoccus and Rhodobacter (up to 43%) | hardwood sulfite spent liquor | HRT = 1 d SRT = 5 d OLR = 17 gCOD/(L d) | 10% PHA/VSS (molar) - | [72] |
| Acidovorax 16.9%, Alcaligenes 13.0%, Paracoccus, Rhodobacter, Rhizobium 16.3%, Comamonas (up to 43.3%) | fermented hardwood spent sulfite liquor: HAc/HPr/HBut/HLac/HVal = 62:18:13:10:1 | HRT = 1–2 d SRT = 5 d OLR = 2–7 gCOD/(L d) | - 3HB/3HV: 62–83/17–38 wt% | [73] |
| Hydrogenophaga, Thauera, Pseudoxanthomonas, Flavobacterium, Paracoccus, Leifsonia, Exiguobacterium, Rhodobacter | fermented food waste and sewage sludge | HRT = 1 d SRT = 1 d OLR = 3–6 gCOD/(L d) | 45–55% gPHA/gVSS 3HB/3HV: 90/10 wt% | [49] |
| Acidovorax and Hydrogenophaga (52–79%), Thauera and Azoarcus (12%) | fermented food waste: HAc 21.5%, HBut 38.0%, HPr 12.7%, HVal 11.6%, HCap 10.0% | HRT = 1 d SRT = 1 d OLR = 2.5 gCOD/(L d) | 40–45% gPHA/gVSS 3HB/3HV: 88/12 wt% | [48] |
| Allorhizobium, Neorhizobium, Pararhizobium, Rhizobium (up to 38.3%); Acidivorax, Aquimonas, Comamonas, Hydrogenophaga, Ramlibacter, Zooglea (up to 35.3%) | fermented food waste: HAc 23%, HPr 19%, HBut 46% (COD basin) | HRT = 1 d SRT = 1 d OLR = 3.5 gCOD/(L d) | 40–45% gPHA/gVSS 3HB/3HV: 90/10 wt% | [74] |
| Clostridium (29%), Pseudomonaas (8%), Rhodopseudomonas (5%) | synthetic VFA: 3 g/L HAc and 0.5 g/L HBut | OLR = 2 gCOD/(L d) | - 3HB/3HV: 87/13 wt% | [75] |
| Enterobacter and Pseudomonas (66.6%) | synthetic wastewater and glucose | - 3HB/3HV: 76–88/8–21 wt% | [76] | |
| Amaricoccus and Thauera from 56.3% to 72.4% | crude glycerol fermentation (90 Cmmol/L): HAc 2.38 Cmmol/L, HPr 12.10 Cmmol/L, HBut 30.52 Cmmol/L. | HRT = 1 d SRT = 1 d OLR = 3.7 gCOD/(L d) | - 3HB/3HV: 75/25 wt% | [77] |
| Zoogloea (10.1%) Zoogloea resiniphila, Dechloromonas (4.45%), Azospira (2.82%) | sodium acetate | HRT = 2 d | 68% gPHA/gVSS | [78] |
| Uncultured Rhodocyclaceae | fermented food waste | HRT = 0.7 d SRT = 1 d OLR ≈ 8 gCOD/(L d) | - 3HB/3HV: 50/50 wt% | [79] |
| Plasticicumulans Acidivorans | fermented paper mill wastewater: VFA/CODSOL 0.72; HAc 37%, HPr 21%, HBut 29%, HVal 16% | HRT = 1 d SRT = 1 d OLR ≈ 3 gCOD/(L d) | - 3HB/3HV: 75/25 wt% | [47] |
| Corynebacterium, Xantomonadaceae, Bosea, Amaricoccus, Paracoccus | fermented cheese whey: EtOH 41 mg/L, HAc 52 mg/L, HBu 14.8 mg/L, TOA 294 mg/L | HRT = 1 d SRT = 4–5 d OLR = 2 gCOD/(L d) | - 3HB/3HV: 87/13% (molar) | [67] |
| Proteobacteria (77.6%), Bacteroidetes (77.6%), Nitrospira (1.75%), Armatimonadetes (1.3%) | fermented paperboard mill wastewater: CODSOL 0.92 g/L; 0.34 g/L VFA | HRT = 1 d SRT = 10 d OLR = 3 gCOD/(L d) | - 3HB/3HV: 84–92/8–16 wt% | [80] |
| On HAc: Moraxellaceae (12%), Rhodobacteraceae (11.7%), Bacillaceae (11.6%) Flavobacteriaceae (7%), Comamonadaceae (6.7%); On HCap: Moraxellaceae (18%), Rhodobacteraceae (15.4%), Flavobacteriaceae (8.5%), Comamonadaceae (5.6%) | fermented food waste (30 v/v%) and sewage sludge (70 v/v%); VFA up to 29.5 g/L | HRT = 1 d | - 3HB/3HV: 94–97/3–6 wt% | [81] |
| (a) Paracoccus 26%, Lactococcus 28%, Enterococcus 15%; (b) Azospirillum 90% | synthtetic hemicellulose hydrolysates: (a) xylose 79.7%, Hac 8.9%; (b) xylose 42%, HAc 50% | HRT = 1 d SRT = 1 d | (a) 4% gPHA/gVSS (b) 18% gPHA/gVSS - | [82] |
| Paracoccus, Comamonas, Azoarcus, Thauera | acidified cooked mussel wastewater (62% gVFA/gCODSOL) | HRT = 1 d SRT = 1 d OLR = 1–2 gCOD/(L d) | - 3HB/3HV: 70–83/17–30 wt% | [83] |
| β-Proteobacteria up to 54% | synthetic VFA: 4.8 g COD/L; HAc/Hpr/Hbu = 16/1.5/8 (COD based) | HRT = 2 d SRT = 10 d OLR = 1.2 gCOD/(L d) | 71.4% gPHA/gVSS - | [84] |
| (a) fermented molasses: Thauera (33.3%), Azoarcus (64.6%), Paracoccus (15.9%); (b) cheese whey: Paenibacillus (26.5%), Lysinibacillus (13.2%) | (a) fermented molasses: HAc 28%, HPr 35%, HBut 20%, HVal 13%; (b) fermented cheese whey: HAc 60%, HPr 9%, HBut 14%, HVal 6%. | HRT = 1 d SRT = 4 d OLR = 2 gCOD/(L d) | - - | [85] |
| Lampropedia, Thauera, Azoarcus, Paracoccus | fermented cheese whey: HAc 41%, HBut 49%, HVal 6%, HPr 4%. | HRT = 1 d SRT = 4 d OLR = 40 Cmmol/(L d) | - 3HB/3HV: 89–92/8–11 wt% | [86] |
| Paracoccus (52.2%), Azoarcus (26%) and Thauera (8%) | synthetic VFA (HAc, HPr, HBut, HVal) | HRT = 1 d SRT = 4 d OLR = 50 Cmmol/(L d) | - 3HB/3HV: 33/67 wt% | [87] |
| Hydrogenophaga | fermented food waste and sewage sludge | HRT = 1 d SRT = 1 d OLR = 4 gCOD/(L d) | 45–50% gPHA/gVSS 3HB/3HV: 90/10 wt% | [18] |
| Proteobacteria (up to 88.1% HAc-fed), Vibrio up to (94.6% Starch-fed) | landfill leachate 600 mgCOD/L | HRT = 1 d SRT = 5 d | - 3HB/3HV: 93/7 wt% | [88] |
2.2. Marine Strains PHA-Producers
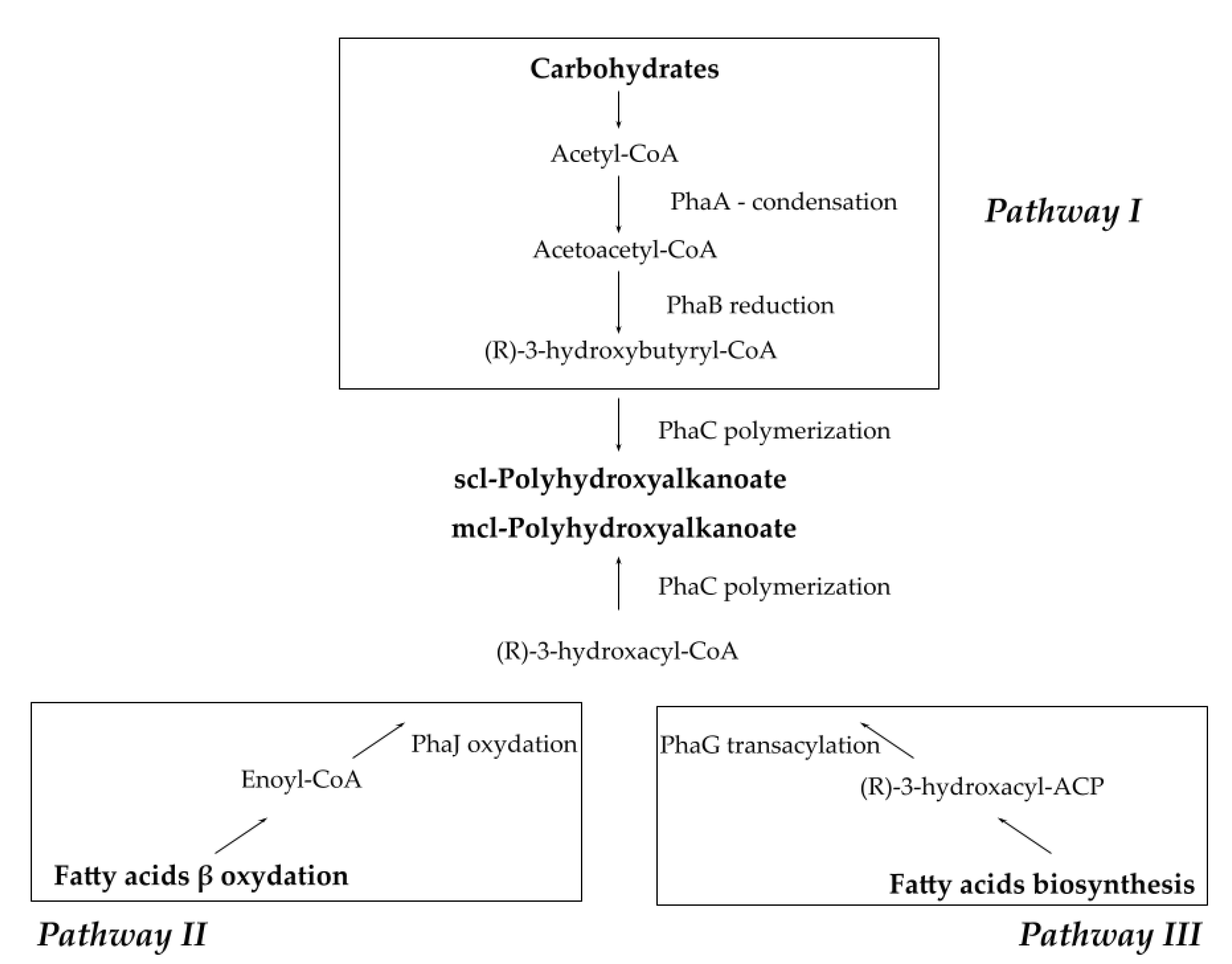
2.3. Known Metabolic Pathways for PHA Production from Organic Substrates
3. Bioconversion of Hydrocarbons to PHA by Terrestrial and Marine Bacteria
| Microorganism | PHA Produced | Carbon Source Used | Isolation Source | Reference |
|---|---|---|---|---|
| Alcanivorax borkumensis | PHA | Octadecane | Seawater | [144,154] |
| Pseudomonas fulva TY16 | mcl-PHAs * | Benzene, toluene, and ethylbenzene | Soil | [141] |
| Pseudomonas putida, Pseudomonas sp. and Ralstonia eutropha | mcl-PHA | Phenanthrene, pyrene and fluoranthene | PAH-contaminated site in Ao Tap Lamu, Phang-nga (Thailand) | [155] |
| P. putida CA-3 | mcl-PHA | Styrene and phenylacetic acid | Industrial bioreactor | [156] |
| Ochrobactrum intermedium | P(3HB) | Oily bilge water | Oily bilge waste contaminated seawater | [145] |
| Methylosinus trichosporium OB3b | P(3HB) | Trichloroethylene, methane | Terrestrial and aquatic environment | [146,147] |
| P. oleovorans ATCC 29347 | PHA | C8-C12 alkanes | Terrestrial and aquatic environment | [156] |
| Ralstonia eutropha H16 | a blend of P(3HB) and P(3HB-co-3HV) | Plant oils and 3-hydroxyvalerate | Sludge | [151] |
| Pseudomonas aeruginosa 47T2 | PHA | Waste frying oil | Waste frying oil | [157] |
| P. putida F1 | mcl-PHA | Toluene, benzene, or ethylbenzene | Terrestrial and aquatic environment | [140] |
| P. putida mt-2 | mcl-PHA | Toluene or p-xylene | Terrestrial and aquatic environment | [140] |
| Mixed culture of P. putida F1, mt-2, and CA-3 | mcl-PHA | Benzene, toluene, ethylbenzene, p-xylene, and styrene | Terrestrial and aquatic environment | [140] |
| P. saccharophila NRRL B-628 | mcl-PHA | Coconut oil, tallow | Terrestrial and aquatic environment | [158] |
Known Pathways for PHA Production from Hydrocarbons
4. Biodegradation of PHA by Marine Bacteria
5. Market Projections for Bio-Plastics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Andrady, A.L.; Neal, M.A. Applications and societal benefits of plastics. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 1977–1984. [Google Scholar] [CrossRef]
- Thushari, G.G.N.; Senevirathna, J.D.M. Plastic pollution in the marine environment. Heliyon 2020, 6, 8. [Google Scholar] [CrossRef]
- Hammer, J.; Kraak, M.H.S.; Parsons, J.R. Plastics in the Marine Environment: The Dark Side of a Modern Gift. In Reviews of Environmental Contamination and Toxicology (Continuation of Residue Reviews); Whitacre, D., Ed.; Springer: New York, NY, USA, 2012; Volume 220. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Lebreton, L.; van der Zwet, J.; Damsteeg, J.W.; Slat, B.; Andrady, A.; Reisser, J. River plastic emissions to the world’s oceans. Nat. Commun. 2017, 8, 15611. [Google Scholar] [CrossRef]
- Chowdhury, H.; Chowdhury, T.; Sait, S.M. Estimating marine plastic pollution from COVID-19 face masks in coastal regions. Mar. Pollut. Bull. 2021, 168, 112419. [Google Scholar] [CrossRef]
- Suaria, G.; Avio, C.; Mineo, A.; Lattin, G.L.; Magaldi, M.G.; Belmonte, G.; Moore, C.J.; Regoli, F.; Aliani, S. The Mediterranean Plastic Soup: Synthetic polymers in Mediterranean surface waters. Sci. Rep. 2016, 6, 37551. [Google Scholar] [CrossRef] [Green Version]
- Ford, H.V.; Jones, N.H.; Davies, A.J.; Godley, B.J.; Jambeck, J.R.; Napper, I.E.; Suckling, C.C.; Williams, G.J.; Woodall, L.C.; Koldewey, H.J. The fundamental links between climate change and marine plastic pollution. Sci. Total Environ. 2022, 806, 150392. [Google Scholar] [CrossRef]
- Bowley, J.; Baker-Austin, C.; Porter, A.; Hartnell, R.; Lewis, C. Oceanic hitchhikers–assessing pathogen risks from marine microplastic. Trends Microbiol. 2020, 29, 107–116. [Google Scholar] [CrossRef]
- Webb, H.; Arnott, J.; Crawford, R.; Ivanova, E. Plastic degradation and its environmental implications with special reference to poly(ethylene terephthalate). Polymers 2013, 5, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Muneer, F.; Rasul, I.; Azeem, F.; Siddique, M.H.; Zubair, M.; Nadeem, H. Microbial Polyhydroxyalkanoates (PHAs): Efficient Replacement of Synthetic Polymers. J. Polym. Environ. 2020, 28, 2301–2323. [Google Scholar] [CrossRef]
- Li, Z.; Yang, J.; Loh, X. Polyhydroxyalkanoates: Opening doors for a sustainable future. NPG Asia Mater. 2016, 8, e265. [Google Scholar] [CrossRef]
- Reddy, C.; Ghai, R.; Kalia, V.C. Polyhydroxyalkanoates: An overview. Bioresour. Technol. 2003, 87, 137–146. [Google Scholar] [CrossRef]
- Raza, Z.A.; Abid, S.; Banat, I.M. Polyhydroxyalkanoates: Characteristics, production, recent developments and applications. Int. Biodeterior. Biodegrad. 2018, 126, 45–56. [Google Scholar] [CrossRef]
- Możejko-Ciesielska, J.; Kiewisz, R. Bacterial Polyhydroxyalkanoates: Still Fabulous? Microbiol. Res. 2016, 192, 271–282. [Google Scholar] [CrossRef]
- Andler, R.; Vivod, R.; Steinbüchel, A. Synthesis of polyhydroxyalkanoates through the biodegradation of poly (cis-1, 4-isoprene) rubber. J. Biosci. Bioeng. 2019, 127, 360–365. [Google Scholar] [CrossRef]
- Choi, J.; Lee, S.Y. Process analysis and economic evaluation for poly(3-hydroxybutrate) production by fermentation. Bioprocess Eng. 1997, 17, 335–342. [Google Scholar] [CrossRef]
- Valentino, F.; Moretto, G.; Lorini, L.; Bolzonella, D.; Pavan, P.; Majone, M. Pilot-Scale Polyhydroxyalkanoate Production from Combined Treatment of Organic Fraction of Municipal Solid Waste and Sewage Sludge. Ind. Eng. Chem. Res. 2019, 58, 12149–12158. [Google Scholar] [CrossRef]
- Izaguirre, J.K.; Barañano, L.; Castañón, S.; Santos, J.A.L.; Cesário, M.T.; da Fonseca, M.M.R.; Alkorta, I.; Garbisu, C. Economic and environmental assessment of bacterial poly(3-hydroxybutyrate) production from the organic fraction of municipal solid waste. Bioresour. Bioprocess. 2021, 8, 39. [Google Scholar] [CrossRef]
- Pagliano, G.; Gugliucci, W.; Torrieri, E.; Piccolo, A.; Cangemi, S.; Di Giuseppe, F.A.; Robertiello, A.; Faraco, V.; Pepe, O.; Ventorino, V. Polyhydroxyalkanoates (PHAs) from dairy wastewater effluent: Bacterial accumulation, structural characterization and physical properties. Chem. Biol. Technol. Agric. 2020, 7, 29. [Google Scholar] [CrossRef]
- Nielsen, C.; Rahman, A.; Rehman, A.U.; Walsh, M.K.; Miller, C.D. Food waste conversion to microbial polyhydroxyalkanoates. Microb. Biotechnol. 2017, 10, 1338–1352. [Google Scholar] [CrossRef]
- Estévez-Alonso, Á.; Pei, R.; van Loosdrecht, M.C.M.; Kleerebezem, R.; Werker, A. Scaling-up microbial community-based polyhydroxyalkanoate production: Status and challenges. Bioresour. Technol. 2021, 327, 124790. [Google Scholar] [CrossRef]
- Crutchik, D.; Franchi, O.; Caminos, L.; Jeison, D.; Belmonte, M.; Pedrouso, A.; Val del Rio, A.; Mosquera-Corral, A.; Campos, J.L. Polyhydroxyalkanoates (PHAs) Production: A Feasible Economic Option for the Treatment of Sewage Sludge in Municipal Wastewater Treatment Plants? Water 2020, 12, 1118. [Google Scholar] [CrossRef]
- Dietrich, K.; Dumont, M.-J.; Del Rio, L.F.; Orsat, V. Producing PHAs in the bioeconomy—Towards a sustainable bioplastic. Sustain. Prod. Consumpt. 2017, 9, 58–70. [Google Scholar] [CrossRef]
- Majone, M.; Dircks, K.; Beun, J.J. Aerobic storage under dynamic conditions in activated sludge processes. The state of the art. Wat. Sci. Technol. 1999, 39, 61–73. [Google Scholar] [CrossRef]
- Mitra, R.; Xu, T.; Xiang, H.; Han, J. Current developments on polyhydroxyalkanoates synthesis by using halophiles as a promising cell factory. Microb. Cell Fact. 2020, 19, 86. [Google Scholar] [CrossRef]
- Guzik, M.W.; Kenny, S.T.; Duane, G.F.; Casey, E.; Woods, T.; Babu, R.P.; Nikodinovic-Runic, J.; Murray, M.; O’Connor, K.E. Conversion of post consumer polyethylene to the biodegradable polymer polyhydroxyalkanoate. Appl. Microbiol. Biotechnol. 2014, 98, 4223–4232. [Google Scholar] [CrossRef]
- Khatami, K.; Perez-Zabaleta, M.; Owusu-Agyeman, I.; Cetecioglu, Z. Waste to bioplastics: How close are we to sustainable polyhydroxyalkanoates production? Waste Manag. 2021, 119, 374–388. [Google Scholar] [CrossRef]
- Ru, J.; Huo, Y.; Yang, Y. Microbial Degradation and Valorization of Plastic Wastes. Front. Microbiol. 2020, 11, 442. [Google Scholar] [CrossRef] [Green Version]
- Birolli, W.G.; Lima, R.N.; Porto, A.L.M. Applications of marine-derived microorganisms and their enzymes in biocatalysis and biotransformation, the underexplored potentials. Front. Microbiol. 2019, 10, 1453. [Google Scholar] [CrossRef]
- Smith, D.; Buddie, A.G.; Goss, R.J.M.; Overmann, J.; Lepleux, C.; Brönstrup, M.; Kloareg, B.; Meiners, T.; Brennecke, P.; Ianora, A.; et al. Discovery pipelines for marine resources: An ocean of opportunity for biotechnology. World J. Microbiol. Biotechnol. 2019, 35, 107. [Google Scholar] [CrossRef] [Green Version]
- Yakimov, M.M.; Timmis, K.N.; Golyshin, P.N. Obligate oil-degrading marine bacteria. Curr. Opin. Biotechnol. 2007, 18, 257–266. [Google Scholar] [CrossRef]
- Yakimov, M.M.; Bargiela, R.; Golyshin, P.N. Calm and Frenzy: Marine obligate hydrocarbonoclastic bacteria sustain ocean wellness. Curr. Opin. Biotechnol. 2022, 73, 337–345. [Google Scholar] [CrossRef]
- Valentino, F.; Morgan-Sagastume, F.; Campanari, S.; Villano, M.; Werker, A.; Majone, M. Carbon recovery from wastewater through bioconversion into biodegradable polymers. New Biotechnol. 2017, 37, 9–23. [Google Scholar] [CrossRef] [Green Version]
- Battista, F.; Frison, N.; Bolzonella, D. Can bioplastics be treated in conventional anaerobic digesters for food waste treatment? Environ. Technol. Innov. 2021, 22, 101393. [Google Scholar] [CrossRef]
- Lorini, L.; Martinelli, A.; Capuani, G.; Frison, N.; Reis, M.; Sommer Ferreira, B.; Villano, M.; Majone, M.; Valentino, F. Characterization of polyhydroxyalkanoates produced at pilot scale from different organic wastes. Front. Bioeng. Biotechnol. 2021, 9, 628719. [Google Scholar] [CrossRef]
- Gholami, A.; Mohkam, M.; Rasoul-Amini, S.; Ghasemi, Y. Industrial production of polyhydroxyalkanoates by bacteria: Opportunities and challenges. Minerva Biotecnol. 2016, 28, 59–74. [Google Scholar]
- Pagliano, G.; Galletti, P.; Samorì, C.; Zaghini, A.; Torri, C. Recovery of polyhydroxyalkanoates from single and mixed microbial cultures: A review. Front. Bioeng. Biotechnol. 2021, 9, 624021. [Google Scholar] [CrossRef]
- Rodriguez-Perez, S.; Serrano, A.; Pantio, A.A.; Alonso-Farinas, B. Challenges of scaling-up PHA production from waste streams. A review. J. Environ. Manag. 2018, 205, 215–230. [Google Scholar] [CrossRef] [Green Version]
- Ntaikou, I.; Koumelis, I.; Tsitsilianis, C.; Parthenios, J.; Lyberatos, G. Comparison of yields and properties of microbial polyhydroxyalkanoates generated from waste glycerol based substrates. Int. J. Biol. Macromol. 2018, 112, 273–283. [Google Scholar] [CrossRef]
- Catalán, A.I.; Malan, A.K.; Ferreira, F.; Gill, P.R.; Batista, S. Propionic acid metabolism and poly-3-hydroxybutyrate-co-3-hydroxyvalerate production by a prpC mutant of Herbaspirillum seropedicae Z69. J. Biotechnol. 2018, 286, 36–44. [Google Scholar] [CrossRef]
- Silva, F.; Matos, M.; Pereira, B.; Ralo, C.; Pequito, D.; Marques, N.; Carvalho, G.; Reis, M.A.M. An integrated process for mixed culture production of 3-hydroxyhexanoate-rich polyhydroxyalkanoates from fruit waste. Chem. Eng. J. 2022, 427, 131908. [Google Scholar] [CrossRef]
- Alfano, S.; Lorini, L.; Majone, M.; Sciubba, F.; Valentino, F.; Martinelli, A. Ethylic esters as green solvents for the extraction of intracellular polyhydroxyalkanoates produced by mixed microbial culture. Polymers 2021, 13, 2789. [Google Scholar] [CrossRef]
- Lorini, L.; Martinelli, A.; Pavan, P.; Majone, M.; Valentino, F. Downstream processing and characterization of polyhydroxyalkanoates (PHAs) produced by mixed microbial culture (MMC) and organic urban waste as substrate. Biomass Convers. Biorefinery 2021, 11, 693–703. [Google Scholar] [CrossRef]
- Morgan-Sagastume, F. Characterisation of open, mixed microbial cultures for polyhydroxyalkanoate (PHA) production. Rev. Environ. Sci. Biotechnol. 2016, 15, 593–625. [Google Scholar] [CrossRef]
- Tamis, J.; Luzkova, K.; Jiang, Y.; van Loosdrecht, M.C.M.; Kleerebezem, R. Enrichment of Plasticicumulans acidivorans at pilot-scale for PHA production on industrial wastewater. J. Biotechnol. 2014, 192, 161–169. [Google Scholar] [CrossRef]
- Tamis, J.; Mulders, M.; Dijkman, H.; Rozendal, R.; van Loosdrecht, M.C.M.; Kleerebezem, R. Pilot-Scale Polyhydroxyalkanoate Production from Paper Mill Wastewater: Process Characteristics and Identification of Bottlenecks for Full-Scale Implementation. J. Environ. Eng. 2018, 144, 1–9. [Google Scholar] [CrossRef]
- Valentino, F.; Gottardo, M.; Micolucci, F.; Pavan, P.; Bolzonella, D.; Rossetti, S.; Majone, M. Organic fraction of municipal solid waste recovery by conversion into added-value polyhydroxyalkanoates and biogas. ACS Sustain. Chem. Eng. 2018, 6, 16375–16385. [Google Scholar] [CrossRef]
- Moretto, G.; Lorini, L.; Pavan, P.; Crognale, S.; Tonanzi, B.; Rossetti, S.; Majone, M.; Valentino, F. Biopolymers from urban organic waste: Influence of the solid retention time to cycle length ratio in the enrichment of a mixed microbial culture (MMC). ACS Sustain. Chem. Eng. 2020, 8, 14531–14539. [Google Scholar] [CrossRef]
- Moretto, G.; Russo, I.; Bolzonella, D.; Pavan, P.; Majone, M.; Valentino, F. An urban biorefinery for food waste and biological sludge conversion into polyhydroxyalkanoates and biogas. Water Res. 2020, 170, 115371. [Google Scholar] [CrossRef]
- Conca, V.; da Ros, C.; Valentino, F.; Eusebi, A.L.; Frison, N.; Fatone, F. Long-term validation of polyhydroxyalkanoates production potential from the sidestream of municipal wastewater treatment plant at pilot scale. Chem. Eng. J. 2020, 390, 124627. [Google Scholar] [CrossRef]
- Matos, M.; Cruz, R.A.; Cardoso, P.; Silva, F.; Freitas, E.B.; Carvalho, G.; Reis, M.A.M. Sludge retention time impacts on polyhydroxyalkanoate productivity in uncoupled storage/growth processes. Sci. Total Environ. 2021, 799, 149363. [Google Scholar] [CrossRef] [PubMed]
- Morgan-Sagastume, F.; Hjort, M.; Cirne, D.; Gerardin, F.; Lacroix, S.; Gaval, G.; Karabegovic, L.; Alexandersson, T.; Johansson, P.; Karlsson, A.; et al. Integrated production of polyhydroxyalkanoates (PHAs) with municipal wastewater and sludge treatment at pilot scale. Bioresour. Technol. 2015, 181, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, S.; Karlsson, A.; Alexandersson, T.; Quadri, L.; Hjort, M.; Johansson, P.; Morgan-Sagastume, F.; Anterrieu, S.; Arcos-Hernandez, M.; Karabegovic, L.; et al. A process for polyhydroxyalkanoate (PHA) production from municipal wastewater treatment with biological carbon and nitrogen removal demonstrated at pilot-scale. New Biotechnol. 2017, 35, 42–53. [Google Scholar] [CrossRef]
- Van Loosdrecht, M.C.M.; Pot, M.A.; Heijnen, J.J. Importance of bacterial storage polymers in bioprocesses. Water Sci. Technol. 1997, 35, 41–47. [Google Scholar] [CrossRef]
- Villano, M.; Lampis, S.; Valentino, F.; Vallini, G.; Majone, M.; Beccari, M. Effect of hydraulic and organic loads in Sequencing Batch Reactor on microbial ecology of activated sludge and storage of polyhydroxyalkanoates. Chem. Eng. Trans. 2010, 20, 187–192. [Google Scholar] [CrossRef]
- Matos, M.; Cruz, R.A.P.; Cardoso, P.; Silva, F.; Freitas, E.B.; Carvalho, G.; Reis, M.A.M. Combined strategies to boost polyhydroxyalkanoate production from fruit waste in a three-stage pilot plant. ACS Sustain. Chem. Eng. 2021, 9, 8270–8279. [Google Scholar] [CrossRef]
- Colombo, B.; Favini, F.; Scaglia, B.; Sciarria, T.P.; D’Imporzano, G.; Pognani, M.; Alekseeva, A.; Eisele, G.; Cosentino, C.; Adani, F. Enhanced polyhydroxyalkanoate (PHA) production from the organic fraction of municipal solid waste by using mixed microbial culture. Biotechnol. Biofuels 2017, 10, 201. [Google Scholar] [CrossRef]
- Jiang, Y.; Marang, L.; Kleerebezem, R.; Muyzer, G.; van Loosdrecht, M.C.M. Effect of temperature and cycle length on microbial competition in PHB-producing sequencing batch reactor. ISME J. 2011, 5, 896–907. [Google Scholar] [CrossRef]
- De Grazia, G.; Quadri, L.; Majone, M.; Morgan-Sagastume, F.; Werker, A. Influence of temperature on mixed microbial culture polyhydroxyalkanoate production while treating a starch industry wastewater. J. Environ. Chem. Eng. 2017, 5, 5067–5075. [Google Scholar] [CrossRef] [Green Version]
- Morgan-Sagastume, F.; Bengtsson, S.; De Grazia, G.; Alexandersson, T.; Quadri, L.; Johansson, P.; Magnusson, P.; Werker, A. Mixed-culture polyhydroxyalkanoate (PHA) production integrated into a food-industry effluent biological treatment: A pilot-scale evaluation. J. Environ. Chem. Eng. 2020, 8, 104469. [Google Scholar] [CrossRef]
- Patterson, T.; Massanet-Nicolau, J.; Jones, R.; Boldrin, A.; Valentino, F.; Dinsdale, R.; Guwy, A. Utilizing grass for the biological production of polyhydroxyalkanoates (PHAs) via green biorefining: Material and energy flows. J. Ind. Ecol. 2021, 25, 802–815. [Google Scholar] [CrossRef]
- Campanari, S.; Augelletti, F.; Rossetti, S.; Sciubba, F.; Villano, M.; Majone, M. Enhancing a multi-stage process for olive oil mill wastewater valorization towards polyhydroxyalkanoates and biogas production. Chem. Eng. J. 2017, 317, 280–289. [Google Scholar] [CrossRef]
- Bengtsson, S.; Werker, A.; Welander, T. Production of polyhydroxyalkanoates by glycogen accumulating organisms treating a paper mill wastewater. Water Sci. Technol. 2008, 58, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, M.G.E.; Torres, C.A.V.; Reis, M.A.M. Polyhydroxyalkanoate (PHA) production by a mixed microbial culture using sugar molasses: Effect of the influent substrate concentration on culture selection. Water Res. 2010, 44, 3419–3433. [Google Scholar] [CrossRef]
- Campanari, S.; Silva, A.F.; Bertin, L.; Villano, M.; Majone, M. Effect of the organic loading rate on the production of polyhydroxyalkanoates in a multi-stage process aimed at the valorization of olive oil mill wastewater. Int. J. Biol. Macromol. 2014, 71, 34–41. [Google Scholar] [CrossRef]
- Oliveira, C.S.; Silva, C.E.; Carvalho, G.; Reis, M.A.M. Strategies for efficiently selecting PHA producing mixed microbial cultures using complex feedstocks: Feast and famine regime and uncoupled carbon and nitrogen availabilities. New Biotechnol. 2017, 37, 69–79. [Google Scholar] [CrossRef]
- Silva, F.; Campanari, S.; Matteo, S.; Valentino, F.; Majone, M.; Villano, M. Impact of nitrogen feeding regulation on polyhydroxyalkanoates production by mixed microbial cultures. New Biotechnol. 2017, 37, 90–98. [Google Scholar] [CrossRef]
- Lorini, L.; di Re, F.; Majone, M.; Valentino, F. High rate selection of PHA accumulating mixed cultures in sequencing batch reactors with uncoupled carbon and nitrogen feeding. New Biotechnol. 2020, 56, 140–148. [Google Scholar] [CrossRef]
- Sruamsiri, D.; Thayanukul, P.; Suwannasilp, B.B. In situ identification of polyhydroxyalkanoate (PHA)-accumulating microorganisms in mixed microbial cultures under feast/famine conditions. Sci. Rep. 2020, 10, 3752. [Google Scholar] [CrossRef] [Green Version]
- Wijeyekoon, S.; Carere, C.R.; West, M.; Nath, S.; Gapes, D. Mixed culture polyhydroxyalkanoate (PHA) synthesis from nutrient rich wet oxidation liquors. Water Res. 2018, 140, 1–11. [Google Scholar] [CrossRef]
- Queirós, D.; Fonseca, A.; Rossetti, S.; Serafim, L.S.; Lemos, P.C. Highly complex substrates lead to dynamic bacterial community for polyhydroxyalkanoates production. J. Ind. Microbiol. Biotechnol. 2017, 44, 1215–1224. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.; Queirós, D.; Lemos, P.C.; Rossetti, S.; Serafim, L.S. Enrichment of a mixed microbial culture of PHA-storing microorganisms by using fermented hardwood spent sulfite liquor. New Biotechnol. 2020, 56, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Crognale, S.; Tonanzi, B.; Valentino, F.; Majone, M.; Rossetti, S. Microbiome dynamics and phaC synthase genes selected in a pilot plant producing polyhydroxyalkanoate from the organic fraction of urban waste. Sci. Total Environ. 2019, 689, 765–773. [Google Scholar] [CrossRef]
- Guerra-Blanco, P.; Cortes, O.; Poznyak, T.; Chairez, I.; García-Peña, E.I. Polyhydroxyalkanoates (PHA) production by photoheterotrophic microbial consortia: Effect of culture conditions over microbial population and biopolymer yield and composition. Eur. Polym. J. 2018, 98, 94–104. [Google Scholar] [CrossRef]
- Amulya, K.; Reddy, M.V.; Rohit, M.V.; Mohan, S.V. Wastewater as renewable feedstock for bioplastics production: Understanding the role of reactor microenvironment and system pH. J. Clean. Prod. 2016, 112, 4618–4627. [Google Scholar] [CrossRef]
- Burniol-Figols, A.; Varrone, C.; Le, S.B.; Daugaard, A.E.; Skiadas, I.V.; Gavala, H.N. Combined polyhydroxyalkanoates (PHA) and 1, 3-propanediol production from crude glycerol: Selective conversion of volatile fatty acids into PHA by mixed microbial consortia. Water Res. 2018, 136, 180–191. [Google Scholar] [CrossRef] [Green Version]
- Inoue, D.; Fukuyama, A.; Ren, Y.; Ike, M. Optimization of aerobic dynamic discharge process for very rapid enrichment of polyhydroxyalkanoates-accumulating bacteria from activated sludge. Bioresour. Technol. 2021, 336, 125314. [Google Scholar] [CrossRef]
- Mulders, M.; Tamis, J.; Abbas, B.; Sousa, J.; Dijkman, H.; Rozendal, R.; Kleerebezem, R. Pilot-scale polyhydroxyalkanoate production from organic waste: Process characteristics at high pH and high ammonium concentration. J. Environ. Chem. Eng. 2020, 146, 04020049. [Google Scholar] [CrossRef]
- Farghaly, A.; Enitan, A.M.; Kumari, S.; Bux, F.; Tawfik, A. Polyhydroxyalkanoates production from fermented paperboard mill wastewater using acetate-enriched bacteria. Clean Technol. Environ. Policy 2017, 19, 935–947. [Google Scholar] [CrossRef]
- Perez-Zabaleta, M.; Atasoy, M.; Khatami, K.; Eriksson, E.; Cetecioglu, Z. Bio-based conversion of volatile fatty acids from waste streams to polyhydroxyalkanoates using mixed microbial cultures. Bioresour. Technol. 2021, 323, 124604. [Google Scholar] [CrossRef]
- Pinto-Ibieta, F.; Cea, M.; Cabrera, F.; Abanto, M.; Felissia, F.E.; Area, M.C.; Ciudad, G. Strategy for biological co-production of levulinic acid and polyhydroxyalkanoates by using mixed microbial cultures fed with synthetic hemicellulose hydrolysate. Bioresour. Technol. 2020, 309, 123323. [Google Scholar] [CrossRef] [PubMed]
- Argiz, L.; Fra-Vázquez, A.; Del Río, Á.V.; Mosquera-Corral, A. Optimization of an enriched mixed culture to increase PHA accumulation using industrial saline complex wastewater as a substrate. Chemosphere 2020, 247, 125873. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, Z.; Wen, Q.; Lee, D.J. Enhanced polyhydroxyalkanoate production by mixed microbial culture with extended cultivation strategy. Bioresour. Technol. 2017, 241, 802–811. [Google Scholar] [CrossRef]
- Carvalho, G.; Pedras, I.; Karst, S.M.; Oliveira, C.S.; Duque, A.F.; Nielsen, P.H.; Reis, M.A.M. Functional redundancy ensures performance robustness in 3-stage PHA-producing mixed cultures under variable feed operation. New Biotechnol. 2018, 40, 207–217. [Google Scholar] [CrossRef]
- Oliveira, C.S.; Silva, M.O.; Silva, C.E.; Carvalho, G.; Reis, M.A.M. Assessment of protein-rich cheese whey waste stream as a nutrients source for low-cost mixed microbial PHA production. Appl. Sci. 2018, 8, 1817. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Oehmen, A.; Carvalho, G.; Reis, M.A.M. Community profile governs substrate competition in polyhydroxyalkanoate (PHA)-producing mixed cultures. New Biotechnol. 2020, 58, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Cui, Y.W.; Zhang, H.Y.; Gao, Z.L. Carbon source applied in enrichment stage of mixed microbial cultures limits the substrate adaptability for PHA fermentation using the renewable carbon. Appl. Biochem. Biotechnol. 2021, 193, 3253–3270. [Google Scholar] [CrossRef] [PubMed]
- Cram, J.; Chow, C.E.; Sachdeva, R.; Needham, D.M.; E Parada, A.; A Steele, J.; Fuhrman, J. Seasonal and interannual variability of the marine bacterioplankton community throughout the water column over ten years. ISME J. 2015, 9, 563–580. [Google Scholar] [CrossRef] [Green Version]
- Obruca, S.; Sedlacek, P.; Koller, M.; Kucera, D.; Pernicova, I. Involvement of polyhydroxyalkanoates in stress resistance of microbial cells: Biotechnological consequences and applications. Biotechnol. Adv. 2018, 36, 856–870. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Obruca, S.; Braunegg, G. Linking Salinity to Microbial Biopolyesters Biosynthesis. In The Handbook of Polyhydroxyalkanoates; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Jendrossek, D.; Pfeiffer, D. New insights in the formation of polyhydroxyalkanoate granules (carbonosomes) and novel functions of poly(3-hydroxybutyrate). Environ. Microbiol. 2014, 16, 2357–2373. [Google Scholar] [CrossRef]
- Zadjelovic, V.; Chhun, A.; Quareshy, M.; Silvano, E.; Hernandez-Fernaud, J.R.; Aguilo-Ferretjans, M.M.; Bosch, R.; Dorador, C.; Gibson, M.I.; Christie-Oleza, J.A. Beyond oil degradation: Enzymatic potential of Alcanivorax to degrade natural and synthetic polyesters. Environ. Microbiol. 2020, 22, 1356–1369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, M.; Tachibana, Y.; Kasuya, K. Biodegradability of poly(3-hydroxyalkanoate) and poly(ε-caprolactone) via biological carbon cycles in marine environments. Polym. J. 2021, 53, 47–66. [Google Scholar] [CrossRef]
- Eronen-Rasimus, E.; Hultman, J.; Hai, T.; Pessi, I.S.; Collins, E.; Wright, S.; Laine, P.; Viitamäki, S.; Lyra, C.; Thomas, D.N.; et al. Sea-Ice Bacteria Halomonas sp. Strain 363 and Paracoccus sp. Strain 392 Produce Multiple Types of Poly-3-Hydroxyalkaonoic Acid (PHA) Storage Polymers at Low Temperature. Appl. Environ. Microbiol. 2021, 87, AEM0092921. [Google Scholar] [CrossRef] [PubMed]
- Higuchi-Takeuchi, M.; Morisaki, K.; Numata, K. A Screening method for the isolation of polyhydroxyalkanoate-producing purple non-sulfur photosynthetic bacteria from natural seawater. Front. Microbiol. 2016, 7, 1509. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Tomasch, J.; Jarek, M.; Wagner-Dobler, I. A dual-species co-cultivation system to study the interactions between Roseobacters and dinoflagellates. Front. Microbiol. 2014, 5, 311. [Google Scholar] [CrossRef]
- Xiao, N.; Jiao, N.Z. Formation of polyhydroxyalkanoate in aerobic anoxygenic phototrophic bacteria and its relationship to carbon source and light availability. Appl. Environ. Microbiol. 2011, 77, 7445–7450. [Google Scholar] [CrossRef] [Green Version]
- Doronina, N.V.; Trotsenko, Y.A.; Tourova, T.P. Methylarcula marina gen. nov., sp. nov. and Methylarcula terricola sp. nov.: Novel aerobic, moderately halophilic, facultatively methylotrophic bacteria from coastal saline environments. Int. J. Syst. Evol. Microbiol. 2000, 50, 1849–1859. [Google Scholar] [CrossRef]
- Zhang, B.; Matchinski, E.J.; Chen, B.; Ye, X.; Jing, L.; Lee, K. Marine oil spills—Oil pollution, sources and effects. In World Seas: An Environmental Evaluation; Issues Environ. Impacts; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; Volume III, pp. 391–406. [Google Scholar]
- Sharma, P.K.; Fu, J.; Mohanan, N.; Levin, D.B. Genomics of PHA Synthesizing Bacteria. In The Handbook of Polyhydroxyalkanoates; A Routledge Handbooks Online; Koller, M., Ed.; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Kumar, P.; Kim, B.S. Paracoccus sp. Strain LL1 as a Single Cell Factory for the Conversion of Waste Cooking Oil to Polyhydroxyalkanoates and Carotenoids. Appl. Food Biotechnol. 2019, 6, 53–60. [Google Scholar] [CrossRef]
- Yu, L.P.; Yan, X.; Zhang, X.; Chen, X.B.; Wu, Q.; Jiang, X.R.; Chen, G.Q. Biosynthesis of functional polyhydroxyalkanoates by engineered Halomonas bluephagenesis. Metab. Eng. 2020, 59, 119–130. [Google Scholar] [CrossRef]
- Stanley, A.; Murthy, P.S.K.; Vijayendra, S.V.N. Characterization of Polyhydroxyalkanoate Produced by Halomonas venusta KT832796. J. Polym. Environ. 2020, 28, 973–983. [Google Scholar] [CrossRef]
- Simon-Colin, C.; Raguénès, G.; Cozien, J.; Guezennec, J.G. Halomonas profundus sp. nov., a new PHA-producing bacterium isolated from a deep-sea hydrothermal vent shrimp. J. Appl. Microbiol. 2008, 104, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.; Mishra, S. Efficient Production of Polyhydroxyalkanoate Through Halophilic Bacteria Utilizing Algal Biodiesel Waste Residue. Front. Bioeng. Biotechnol. 2021, 9, 624859. [Google Scholar] [CrossRef]
- Shrivastav, A.; Mishra, S.K.; Shethia, B.; Pancha, I.; Jain, D.; Mishra, S. Isolation of promising bacterial strains from soil and marine environment for polyhydroxyalkanoates (PHAs) production utilizing Jatropha biodiesel byproduct. Int. J. Biol. Macromol. 2010, 47, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Satoh, Y.; Kuriki, Y.; Seino, T.; Fujita, S.; Suda, T.; Kobayashi, T.; Tajima, K. Polyhydroxyalkanoate production by a novel bacterium Massilia sp. UMI-21 isolated from seaweed, and molecular cloning of its polyhydroxyalkanoate synthase gene. J. Biosci. Bioeng. 2014, 118, 514–519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, D.; Wang, Y.; Tong, Y.; Chen, G.Q. Grand Challenges for Industrializing Polyhydroxyalkanoates (PHAs). Trends Biotechnol. 2021, 39, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Veizán, N.; Daga-Quisbert, J.; Perez-Zabaleta, M.; Guevara-Martínez, M.; Larsson, G.; Quillaguamán, J. Improvements of poly(3-hydroxybutyrate) production in an air-lift reactor using simple production media. Bioresour. Bioprocess. 2020, 7, 22. [Google Scholar] [CrossRef] [Green Version]
- Muhammadi, S.; Afzal, M.; Hameed, S. Bacterial polyhydroxyalkanoates-eco-friendly next generation plastic: Production, biocompatibility, biodegradation, physical properties and applications. Green Chem. Lett. Rev. 2015, 8, 56–77. [Google Scholar] [CrossRef] [Green Version]
- Mezzolla, V.; D’Urso, O.F.; Poltronieri, P. Role of PhaC Type I and Type II Enzymes during PHA Biosynthesis. Polymers 2018, 10, 910. [Google Scholar] [CrossRef] [Green Version]
- Thomas, T.; Elain, A.; Bazire, A.; Bruzaud, S. Complete genome sequence of the halophilic PHA-producing bacterium Halomonas sp. SF2003: Insights into its biotechnological potential. World J. Microbiol. Biotechnol. 2019, 35, 50. [Google Scholar] [CrossRef]
- Rehm, B.H. Polyester synthases: Natural catalysts for plastics. Biochem. J. 2003, 376 Pt 1, 15–33. [Google Scholar] [CrossRef] [Green Version]
- Madison, L.L.; Huisman, G.W. Metabolic engineering of poly (3-hydroalkanoates): From DNA to plastic. Microbiol. Mol. Biol. Rev. 1999, 63, 21–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sasidharan, R.S.; Bhat, S.G.; Chandrasekaran, M. Amplification and sequence analysis of phaC gene of polyhydroxybutyrate producing Vibrio azureus BTKB33 isolated from marine sediments. Ann. Microbiol. 2016, 66, 299–306. [Google Scholar] [CrossRef]
- Hai, T.; Lange, D.; Rabus, R.; Steinbüchel, A. Polyhydroxyalkanoate (PHA) accumulation in sulfate-reducing bacteria and identification of a class III PHA synthase (PhaEC) in Desulfococcus multivorans. Appl. Environ. Microbiol. 2004, 70, 4440–4448. [Google Scholar] [CrossRef] [Green Version]
- Higuchi-Takeuchi, M.; Numata, K. Marine Purple Photosynthetic Bacteria as Sustainable Microbial Production Hosts. Front. Bioeng. Biotechnol. 2019, 7, 258. [Google Scholar] [CrossRef]
- Hai, T.; Hein, S.; Steinbüchel, A. Multiple evidence for widespread and general occurrence of type-III PHA synthases in cyanobacteria and molecular characterization of the PHA synthases from two thermophilic cyanobacteria: Chlorogloeopsis fritschii PCC 6912 and Synechococcus sp. strain MA19. Microbiology 2001, 147, 3047–3060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pu, N.; Wang, M.R.; Li, Z.J. Characterization of polyhydroxyalkanoate synthases from the marine bacterium Neptunomonas concharum JCM17730. J. Biotechnol. 2020, 319, 69–73. [Google Scholar] [CrossRef]
- Tsuge, T.; Hyakutake, M.; Mizuno, K. Class IV polyhydroxyalkanoate (PHA) synthases and PHA-producing Bacillus. Appl. Microbiol. Biotechnol. 2015, 99, 6231–6240. [Google Scholar] [CrossRef] [PubMed]
- Vandi, L.J.; Chan, C.M.; Werker, A.; Richardson, D.; Laycock, B.; Pratt, S. Wood-PHA Composites: Mapping Opportunities. Polymers 2018, 10, 751. [Google Scholar] [CrossRef] [Green Version]
- Bugnicourt, E.; Cinelli, P.; Lazzeri, A.; Alvarez, V.A. Polyhydroxyalkanoate (PHA): Review of synthesis, characteristics, processing and potential applications in packaging. Express Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef] [Green Version]
- Kourmentza, C.; Costa, J.; Azevedo, Z.; Servin, C.; Grandfils, C.; de Freitas, V.; Reis, M. Burkholderia thailandensis as a microbial cell factory for the bioconversion of used cooking oil to polyhydroxyalkanoates and rhamnolipids. Bioresour. Technol. 2018, 247, 829–837. [Google Scholar] [CrossRef]
- Surendran, A.; Lakshmanan, M.; Chee, J.Y.; Sulaiman, A.M.; Thuoc, D.V.; Sudesh, K. Can Polyhydroxyalkanoates Be Produced Efficiently from Waste Plant and Animal Oils? Front. Bioeng. Biotechnol. 2020, 8, 169. [Google Scholar] [CrossRef] [PubMed]
- Goudarztalejerdi, A.; Tabatabaei, M.; Eskandari, M.H.; Mowla, D.; Iraji, A. Evaluation of bioremediation potential and biopolymer production of pseudomonads isolated from petroleum hydrocarbon-contaminated areas. Int. J. Environ. Sci. Technol. 2015, 12, 2801–2808. [Google Scholar] [CrossRef] [Green Version]
- Page, W.J. Waste Sources for Polyhydroxyalkanoate Production. In Proceedings of the 1996 International Symposium on Bacterial Polyhydroxyalkanoates, Davos, Switzerland, 18–23 August 1996; Eggink, G., Steinbuchel, A., Poirier, Y., Witholt, B., Eds.; National Research Council of Canada: Davos, Switzerland, 1996; pp. 56–65. [Google Scholar]
- Ayub, N.D.; Pettinari, M.J.; Ruiz, J.A.; Lopez, N.I. A polyhydroxybutyrate-producing Pseudomonas sp. isolated from Antarctic environments with high stress resistance. Curr. Microbiol. 2004, 49, 170–174. [Google Scholar] [CrossRef]
- Kadouri, D.; Jurkevitch, E.; Okon, Y.; Castro-Sowinski, S. Ecological and agricultural significance of bacterial polyhydroxyalkanoates. Crit. Rev. Microbiol. 2005, 31, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Kourmentza, C.; Plácido, J.; Venetsaneas, N.; Burniol-Figols, A.; Varrone, C.; Gavala, H.N.; Reis, M. Recent Advances and Challenges towards Sustainable Polyhydroxyalkanoate (PHA) Production. Bioengineering 2017, 4, 55. [Google Scholar] [CrossRef] [Green Version]
- Zobell, C.E. Action of Microorganisms on Hydrocarbons. Bacteriol. Rev. 1946, 10, 1–49. [Google Scholar] [CrossRef]
- Imam, A.; Kanaujia, P.K.; Ray, A.; Suman, S.K. Removal of Petroleum Contaminants Through Bioremediation with Integrated Concepts of Resource Recovery: A Review. Indian J. Microbiol. 2021, 61, 250–261. [Google Scholar] [CrossRef]
- Denaro, R.; Pippo, F.D.; Crisafi, F.; Rossetti, S. Biodegradation of Hydrocarbons in Marine Environment. In Water Pollution and Remediation: Organic Pollutants; Springer: Cham, Switzerland, 2021; pp. 195–228. [Google Scholar]
- Prince, R.C.; Amande, T.J.; McGenity, T.J. Prokaryotic hydrocarbon degraders. In Taxonomy, Genomics and Ecophysiology of Hydrocarbon-Degrading Microbes, Handbook of Hydrocarbon and Lipid Microbiology; McGenity, T.J., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 1–39. [Google Scholar] [CrossRef]
- McGenety, T.J.; Folwell, B.D.; McKew, B.A.; Sanni, G. Marine crude-oil biodegradation: A central role for interspecies interactions. Aquat. Biosyst. 2012, 8, 10–29. [Google Scholar] [CrossRef] [Green Version]
- Berry, D.; Gutierrez, T. Evaluating the detection of hydrocarbon-degrading bacteria in 16S rRNA gene sequencing surveys. Front. Microbiol. 2017, 8, 896. [Google Scholar] [CrossRef] [Green Version]
- Head, I.M.; Jones, D.M.; Röling, W.F.M. Marine microorganisms make a meal of oil. Nat. Rev. Microbiol. 2006, 4, 173–182. [Google Scholar] [CrossRef]
- de Smet, M.J.; Eggink, G.; Witholt, B.; Kingma, J.; Wynberg, H. Characterization of intracellular inclusions formed by Pseudomonas oleovorans during growth on octane. J. Bacteriol. 1983, 154, 870–878. [Google Scholar] [CrossRef] [Green Version]
- Tobin, K.M.; O’Connor, K.E. Polyhydroxyalkanoate accumulating diversity of Pseudomonas species utilising aromatic hydrocarbons. FEMS Microbiol. Lett. 2005, 253, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Nikodinovic, J.; Kenny, S.T.; Babu, R.P.; Woods, T.; Blau, W.J.; O’Connor, K.E. The conversion of BTEX compounds by single and defined mixed cultures to medium-chain-length polyhydroxy-alkanoate. Appl. Microbiol. Biotechnol. 2008, 80, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.Y.; Kim, D.Y.; Chung, M.G.; Lee, S.H.; Park, H.Y.; Rhee, Y.H. Biosynthesis of medium-chain-length poly(3-hydroxyalkanoates) by volatile aromatic hydrocarbons-degrading Pseudomonas fulva TY16. Bioresour. Technol. 2010, 101, 8485–8488. [Google Scholar] [CrossRef] [PubMed]
- Hori, K.; Kobayashi, A.; Ikeda, H.; Unno, H. Rhodococcus aetherivorans IAR1, a new bacterial strain synthesizing poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from toluene. J. Biosci. Bioeng. 2009, 107, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Yajima, T.; Nagatomo, M.; Wakabayashi, A.; Sato, M.; Taguchi, S.; Maeda, M. Bioconversion of biphenyl to a polyhydroxyalkanoate copolymer by Alcaligenes denitrificans A41. AMB Express 2020, 10, 155. [Google Scholar] [CrossRef]
- Sabirova, J.S.; Ferrer, M.; Lünsdorf, H.; Wray, V.; Kalscheuer, R.; Steinbüchel, A.; Timmis, K.N.; Golyshin, P.N. Mutation in a “tesB-like” hydroxyacyl-coenzyme A-specific thioesterase gene causes hyperproduction of extracellular polyhydroxyalkanoates by Alcanivorax borkumensis SK2. J. Bacteriol. 2006, 188, 8452–8459. [Google Scholar] [CrossRef] [Green Version]
- Mahendhran, K.; Arthanari, A.; Dheenadayalan, B.; Ramanathan, M. Bioconversion of oily bilge waste to polyhydroxybutyrate (PHB) by marine Ochrobactrum intermedium. Bioresour. Technol. Rep. 2018, 4, 66–73. [Google Scholar] [CrossRef]
- Fitch, M.W.; Speitel, G.E.; Georgiou, G. Degradation of Trichloroethylene by Methanol-Grown Cultures of Methylosinus trichosporium OB3b PP358. Appl. Environ. Microbiol. 1996, 62, 1124–1128. [Google Scholar] [CrossRef] [Green Version]
- Doronina, N.V.; Ezhov, V.A.; Trotsenko, Y.A. Growth of Methylosinus trichosporium OB3b on methane and poly-β-hydroxybutyrate biosynthesis. Appl. Biochem. Microbiol. 2008, 44, 182–185. [Google Scholar] [CrossRef]
- Goff, M.; Ward, P.G.; O’Connor, K.E. Improvement of the conversion of polystyrene to polyhydroxyalkanoate through the manipulation of the microbial aspect of the process: A nitrogen feeding strategy for bacterial cells in a stirred tank reactor. J. Biotechnol. 2007, 132, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Kenny, S.T.; Runic, J.N.; Kaminsky, W.; Woods, T.; Babu, R.P.; Keely, C.M.; Blau, W.; O’Connor, K.E. Up-cycling of PET (polyethylene terephthalate) to the biodegradable plastic PHA (polyhydroxyalkanoate). Environ. Sci. Technol. 2008, 42, 7696–7701. [Google Scholar] [CrossRef] [PubMed]
- Santolin, L.; Waldburger, S.; Neubauer, P.; Riedel, S.L. Substrate-Flexible Two-Stage Fed-Batch Cultivations for the Production of the PHA Copolymer P(HB-co-HHx) With Cupriavidus necator Re2058/pCB113. Front. Bioeng. Biotechnol. 2021, 9, 623890. [Google Scholar] [CrossRef]
- Lee, W.H.; Loo, C.Y.; Nomura, C.T.; Sudesh, K. Biosynthesis of polyhydroxyalkanoate copolymers from mixtures of plant oils and 3-hydroxyvalerate precursors. Bioresour. Technol. 2008, 99, 6844–6851. [Google Scholar] [CrossRef] [PubMed]
- Perez-Pantoja, D.; De la Iglesia, R.; Pieper, D.H.; Gonzalez, B. Metabolic reconstruction of aromatic compounds degradation from the genome of the amazing pollutant-degrading bacterium Cupriavidus necator JMP134. FEMS Microbiol. Rev. 2008, 25, 736–794. [Google Scholar] [CrossRef] [Green Version]
- Ekere, A.I.; Johnston, B.; Zięba, M.; Chaber, P.; Adamus, G.; Tchuenbou-Magaia, F.; Barsi, D.; Amaro, L.P.; Chiellini, E.; Radecka, I.; et al. Environmental cleaning mission Bioconversion of oxidatively fragmented polyethylene plastic waste to value-added copolyesters. Chim. Oggi-Chem. Today 2019, 37, 36–40. [Google Scholar]
- Denaro, R.; Aulenta, F.; Crisafi, F.; Di Pippo, F.; Cruz Viggi, C.; Matturro, B.; Tomei, P.; Smedile, F.; Martinelli, A.; Di Lisio, V.; et al. Marine hydrocarbon-degrading bacteria breakdown poly(ethylene terephthalate) (PET). Sci. Total Environ. 2020, 749, 141608. [Google Scholar] [CrossRef] [PubMed]
- Sangkharak, K.; Choonut, A.; Rakkan, T.; Prasertsan, P. The Degradation of Phenanthrene, Pyrene, and Fluoranthene and Its Conversion into Medium-Chain-Length Polyhydroxyalkanoate by Novel Polycyclic Aromatic Hydrocarbon-Degrading Bacteria. Curr. Microbiol. 2020, 77, 897–909. [Google Scholar] [CrossRef]
- Lageveen, R.G.; Huisman, G.W.; Preusting, H.; Ketelaar, P.; Eggink, G.; Witholt, B. Formation of Polyesters by Pseudomonas oleovorans: Effect of Substrates on Formation and Composition of Poly-(R)-3-Hydroxyalkanoates and Poly-(R)-3-Hydroxyalkenoates. Appl. Environ. Microbiol. 1988, 54, 2924–2932. [Google Scholar] [CrossRef] [Green Version]
- Vidal-Mas, J.; Resina-Pelfort Haba, E.; Comas, J.; Manresa, A.; Vives-Rego, J. Rapid flow cytometry--Nile red assessment of PHA cellular content and heterogeneity in cultures of Pseudomonas aeruginosa 47T2 (NCIB 40044) grown in waste frying oil. Antonie Van Leeuwenhoek 2001, 80, 57–63. [Google Scholar] [CrossRef]
- Solaiman, D.K.; Ashby, R.D.; Foglia, T.A. Medium- chain-length poly(b-hydroxyalkanoate) synthesis from triacylglycerols by Pseudomonas saccharophila. Curr. Microbiol. 1999, 38, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Sabirova, J. Polyhydroxyalkanoates Produced by Hydrocarbon-Degrading Bacteria. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar] [CrossRef]
- Yoon, M.G.; Jeon, H.J.; Kim, M.N. Biodegradation of polyethylene by a soil bacterium and AlkB cloned recombinant cell. J. Bioremediat. Biodegrad. 2012, 3, 145. [Google Scholar] [CrossRef] [Green Version]
- Jeon, H.J.; Kim, M.N. Functional analysis of alkane hydroxylase system derived from Pseudomonas aeruginosa E7 for low molecular weight polyethylene biodegradation. Int. Biodeter. Biodegr. 2015, 103, 141–146. [Google Scholar] [CrossRef]
- Rojo, F. Degradation of alkanes by bacteria. Environ. Microbiol. 2009, 11, 2477–2490. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.G.; de Roo, G.; O’Connor, K.E. Accumulation of polyhydroxyalkanoate from styrene and phenyla- cetic acid by Pseudomonas putida CA-3. Appl. Envion. Microbiol. 2005, 71, 2046–2052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikodinovic-Runic, J.; Casey, E.; Duane, G.F.; Mitic, D.; Hume, A.R.; Kenny, S.T.; O’Connor, K.E. Process analysis of the conversion of styrene to biomass and medium chain length polyhydroxyalkanoate in a two- phase bioreactor. Biotechnol. Bioeng. 2011, 108, 2447–2455. [Google Scholar] [CrossRef]
- Anderson, A.J.; Dawes, E.A. Occurrence, metabolism, metabolic role, and industrial uses of bacterial polyhydroxyalkanoates. Microbiol. Mol. Biol. Res. 1990, 54, 450–472. [Google Scholar] [CrossRef]
- O’Leary, N.D.; O’Connor, K.E.; Ward, P.; Go, M.; Dobson, A.D. Genetic characterization of accumulation of polyhydroxyalkanoate from styrene in Pseudomonas putida CA-3. Appl. Environ. Microbiol. 2005, 71, 4380–4387. [Google Scholar] [CrossRef] [Green Version]
- Oelschlägel, M.; Zimmerling, J.; Tischler, D. A review: The styrene metabolizing cascade of side-chain oxygenation as biotechnological basis to gain various valuable compounds. Front. Microbiol. 2018, 9, 490. [Google Scholar] [CrossRef]
- Kasuya, K.; Ohura, T.; Masuda, K.; Doi, Y. Substrate and binding specificities of bacterial polyhydroxybutyrate depolymerases. Int. J. Biol. Macromol. 1999, 24, 329–336. [Google Scholar] [CrossRef]
- Kroeker, K.J.; Kordas, R.L.; Crim, R.; Hendriks, I.E.; Ramajo, L.; Singh, G.S.; Duarte, C.M.; Gattuso, J. Impacts of ocean acidification on marine organisms: Quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 2013, 19, 1884–1896. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uefuji, M.; Kasuya, K.; Doi, Y. Enzymatic degradation of poly (R)-3-hydroxybutyrate: Secretion and properties of PHB depolymerase from Pseudomonas stutzeri. Polym. Degrad. Stab. 1997, 58, 275–281. [Google Scholar] [CrossRef]
- Kasuya, K.; Mitomo, H.; Nakahara, M.; Akiba, A.; Kudo, T.; Doi, Y. Identification of a marine benthic P(3HB)-degrading bacterium isolate and characterization of its P(3HB) depolymerase. Biomacromolecules 2000, 1, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Laycock, B.; Halley, P.; Pratt, S.; Werker, A.; Lant, P. The chemomechanical properties of microbial polyhydroxyalkanoates. Prog. Polym. Sci. 2013, 38, 536–583. [Google Scholar] [CrossRef]
- Baric, M.; Pierro, L.; Pietrangeli, B.; Petrangeli Papini, M. Polyhydroxyalkanoate (PHB) as a slow-release electron donor for advanced in situ bioremediation of chlorinated solvent-contaminated aquifers. New Biotechnol. 2014, 31, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Melendez-Rodriguez, B.; Torres-Giner, S.; Lorini, L.; Valentino, F.; Sammon, C.; Cabedo, L.; Lagaron, J.M. Valorization of municipal biowaste into electrospun poly(3-hydroxybutyrate-co-3-hydroxyvalerate) biopapers for food packaging applications. ACS App. Bio Mater. 2020, 3, 6110–6123. [Google Scholar] [CrossRef]
- Cavaliere, C.; Capriotti, A.L.; Cerrato, A.; Lorini, L.; Montone, C.M.; Valentino, F.; Laganà, A.; Majone, M. Identification and quantification of polycyclic aromatic hydrocarbons in polyhydroxyalkanoates produced from mixed microbial cultures and municipal organic wastes at pilot scale. Molecules 2021, 26, 539. [Google Scholar] [CrossRef]
- Riccardi, C.; Buiarelli, F.; Castellani, F.; Di Filippo, P.; Lorini, L.; Majone, M.; Matos, M.; Pomata, D.; Simonetti, G.; Sommer-Ferreira, B.; et al. Polychlorinated biphenyl profile in polyhydroxyalkanoates synthetized from urban organic wastes. Polymers 2020, 2, 659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Astolfi, M.L.; Marconi, E.; Lorini, L.; Valentino, F.; Silva, F.; Sommer Ferreira, B.; Canepari, S.; Majone, M. Elemental concentration and migratability in bioplastics derived from organic waste. Chemosphere 2020, 259, 127472. [Google Scholar] [CrossRef] [PubMed]
- Samrot, A.V.; Samanvitha, S.K.; Shobana, N.; Renitta, E.R.; Senthilkumar, P.; Kumar, S.S.; Abirami, S.; Dhiva, S.; Bavanilatha, M.; Prakash, P.; et al. The Synthesis, characterization and applications of polyhydroxyalkanoates (PHAs) and PHA-based nanoparticles. Polymers 2021, 13, 3302. [Google Scholar] [CrossRef]
- European Commission. Directive 2018/851 of the European Parliament and of the Council of 30 May 2018 Amending Directive 2008/98/EC on Waste. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2018.150.01.0109.01.ENG (accessed on 1 May 2022).

| Strain | Phylum | Isolation Source | PHA Produced | Carbon Source Used |
|---|---|---|---|---|
| Afifella marina | α-Proteobacteria | seawater | P(3HB) | nutrient rich medium |
| Alcanivorax borkumensis | γ-Proteobacteria | seawater sediments | PHA | sodium acetate |
| Alteromonas lipolytica | γ-Proteobacteria | seawater | P(3HB) | marine broth |
| Bacillus cereus MCCB 281 | Firmicutes | seawater | P(3HB-co-3HV) | glycerol |
| Bacillus licheniformis MSBN12 | Firmicutes | marine sponge callyspongia diffusa | P(3HB) | palm jaggery |
| Bacillus megaterium | Firmicutes | sediment | PHA | glucose |
| Bacillus sp. NQ-11/A2, | Firmicutes | sediment | P(3HB) | glucose |
| Bacillus thuringiensis | Firmicutes | seashore | P(3HB), P(3HB-co-3HV) | glucose |
| Brevibacterium casei MSI04 | Actinobacteria | marine sponge dendrilla nigra | P(3HB) | starch |
| Burkholderia sp. AIU M5M02 | β-Proteobacteria | shallow sea mud | P(3HB) | nitrogen-limiting mineral salt medium mannitol as a carbon source |
| Colwellia sp. JAMM-0421 | γ-Proteobacteria | deep sea | P(3HB), P(3HB-co-3HV) | glucose, fructose, sodium gluconate or soybean oil |
| Desulfobacterium autotrophicum | δ-Proteobacteria | sediment | P(3HB), P(3HB-co-3HV) | caproate |
| Desulfobotulus sapovorans | δ-Proteobacteria | sediment | P(3HB), P(3HB-co-3HV) | caproate |
| Desulfococcus multivorans | δ-Proteobacteria | sediment | P(3HB), P(3HB-co-3HV) | benzoate |
| Desulfonema magnum | δ-Proteobacteria | sediment | P(3HB), P(3HB-co-3HV) | benzoate |
| Desulfosarcina variabilis | δ-Proteobacteria | sediment | P(3HB), P(3HB-co-3HV) | benzoate |
| Dinoreseobacter shibae DFL 12T | α-Proteobacteria | prorocentrum lima | PHA | sodium acetate glucose |
| Erythrobacter longus DSMZ 6997 | α-Proteobacteria | enteromorpha linza | PHA | glucose |
| Halomonas boliviensis | γ-Proteobacteria | seawater | P(3HB) | different combinations of carbohydrates and hydrolysed polysaccharides |
| Halomonas campisalis | γ-Proteobacteria | seawater | P(3HB-co-3HV), 3.6 mol% 3HV | maltose and yeast extract |
| Halomonas halophila | γ-Proteobacteria | seawater | P(3HB) | hydrolysates of cheese whey, spent coffee grounds, sawdust and corn stover, lignocellulose |
| Halomonas hydrothermalis | γ-Proteobacteria | seawater | P(3HB) | waste frying oil |
| Halomonas marina | γ-Proteobacteria | seawater | P(3HB-co-3HV), 12.8 mol% 3HV | glucose yeast extract alkanoic acids (C3–C6) |
| Halomonas profundus | γ-Proteobacteria | deep sea hydrothermal vent shrimp | P(3HB), P(3HB-co-3HV) | acetate, pyruvate, propionate, valerate, octanoate, glucose and glycerol |
| Labrenzia alexandrii DFL 11T | α-Proteobacteria | alexandrium lusitanicum | PHA | ASW with 1 g/L peptone and 1 g/L yeast extract |
| Marinobacter guineae | γ-Proteobacteria | seawater | PHA | nutrient rich medium |
| Massilia sp. UMI-21 | β-Proteobacteria | seaweed | PHA | starch, maltotriose, or maltose as a sole carbon source |
| Methylarcula marina | α-Proteobacteria | coastal seawater | P(3HB) | starch hydrolysate |
| Methylarcula terricola | α-Proteobacteria | coastal sediment | P(3HB) | starch hydrolysate |
| Methylobacterium sp. | α-Proteobacteria | sediment | P(3HB) | valeric acid and methanol |
| Moritella sp. JCM21335 | γ-Proteobacteria | deep sea | P(3HB-co-3HV) | glucose, fructose, gluconate and plant oils |
| Neptunomonas antarctica | γ-Proteobacteria | sediment | P(3HB) | bacto tryptone, yeast extract and fructose |
| Oceanicola granulosus | α -Proteobacteria | seawater | P(3HB) | pentoses, hexoses, oligosaccharides, sugar alcohols, organic acids and amino acids. |
| Oceanimonas doudoroffii | γ-Proteobacteria | seawater | P(3HB) | lignin or several lignin derivatives |
| Paracoccus sp. LL1 | α-Proteobacteria | seawater | P(3HB) | waste cooking oil |
| Paracoccus seriniphilus | α-Proteobacteria | marine bryozoan | PHA | peptone–yeast marine medium |
| Photobacterium leiognathi 208 | γ-Proteobacteria | seawater | P(3HB) | peptone, glycerol and valeric acid |
| Photobacterium leiognathi 683 | γ-Proteobacteria | fish | P(3HB-co-3HV) | water fish extract followed by peptone, glycerol and valeric acid |
| Pseudoalteromonas sp. SM9913 | γ-Proteobacteria | deep sea sediment | P(3HD-co-3HDD) | glucose, decanoic acid, or olive oil |
| Pseudomonas guezennei | γ-Proteobacteria | marine microbial mat | P(3HO-co-3HD) ** | glucose |
| Rhodovulum euryhalinum | α-Proteobacteria | seawater | PHA | malate, pyruvate and acetate |
| Roseobacter denitrificans OCh 114 | α-Proteobacteria | enteromorpha linza | PHA | sodium acetate followed by glucose |
| Roseospira goensis | α-Proteobacteria | sediment | P(3HB-co-3HV) | sodium acetate |
| Saccharophagus degradans ATCC 43961 | γ-Proteobacteria | salt marsh grass | P(3HB) | glucose |
| Shewanella basaltis | γ-Proteobacteria | seawater | PHA | nutrient rich medium |
| Shewanella surugensis JAMM-0036 | γ-Proteobacteria | deep sea | Oligohydroxyalkanoate | glucose, fructose, sodium gluconate, or soybean oil |
| Sphingopyxis alaskensis | α-Proteobacteria | seawater | P(3HB) | waste vegetable oil |
| Thiohalocapsa marina | γ-Proteobacteria | seawater | P(3HB) | sodium acetate |
| Vibrio azureus BTKB33 | γ-Proteobacteria | sediment | P(3HB) | glucose |
| Vibrio harveyi MCCB 284 | γ-Proteobacteria | tunicate phallusia nigra | P(3HB) | glycerol |
| Vibrio proteolyticus | γ-Proteobacteria | seashore | P(3HB), P(3HB-co-3HV) | fructose, yeast extract |
| Vibrio sp. KN01 | γ-Proteobacteria | seawater | P(3HB), P(3HB-co-5HV-co-3HP) *** | glucose, fructose, gluconate (sodium gluconate), or soybean oil |
| Yangia sp. ND199 | α-Proteobacteria | mangrove samples | P(3HB-co-3HV) | glucose |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crisafi, F.; Valentino, F.; Micolucci, F.; Denaro, R. From Organic Wastes and Hydrocarbons Pollutants to Polyhydroxyalkanoates: Bioconversion by Terrestrial and Marine Bacteria. Sustainability 2022, 14, 8241. https://doi.org/10.3390/su14148241
Crisafi F, Valentino F, Micolucci F, Denaro R. From Organic Wastes and Hydrocarbons Pollutants to Polyhydroxyalkanoates: Bioconversion by Terrestrial and Marine Bacteria. Sustainability. 2022; 14(14):8241. https://doi.org/10.3390/su14148241
Chicago/Turabian StyleCrisafi, Francesca, Francesco Valentino, Federico Micolucci, and Renata Denaro. 2022. "From Organic Wastes and Hydrocarbons Pollutants to Polyhydroxyalkanoates: Bioconversion by Terrestrial and Marine Bacteria" Sustainability 14, no. 14: 8241. https://doi.org/10.3390/su14148241
APA StyleCrisafi, F., Valentino, F., Micolucci, F., & Denaro, R. (2022). From Organic Wastes and Hydrocarbons Pollutants to Polyhydroxyalkanoates: Bioconversion by Terrestrial and Marine Bacteria. Sustainability, 14(14), 8241. https://doi.org/10.3390/su14148241







