Chelating Agents in Assisting Phytoremediation of Uranium-Contaminated Soils: A Review
Abstract
1. Introduction
2. Interactions between Chelating Agents and U Ions
2.1. Mechanisms of Chelation
2.2. Mobilization of Soil U by Chelating Agents
3. Chelating Agent-Assisted Phytoremediation of U-Contaminated Soils
3.1. Mechanisms of Phytoextraction of U from Soils
3.2. Mechanisms of Assisted Phytoextraction of U from Soils
3.3. Environmental Risks
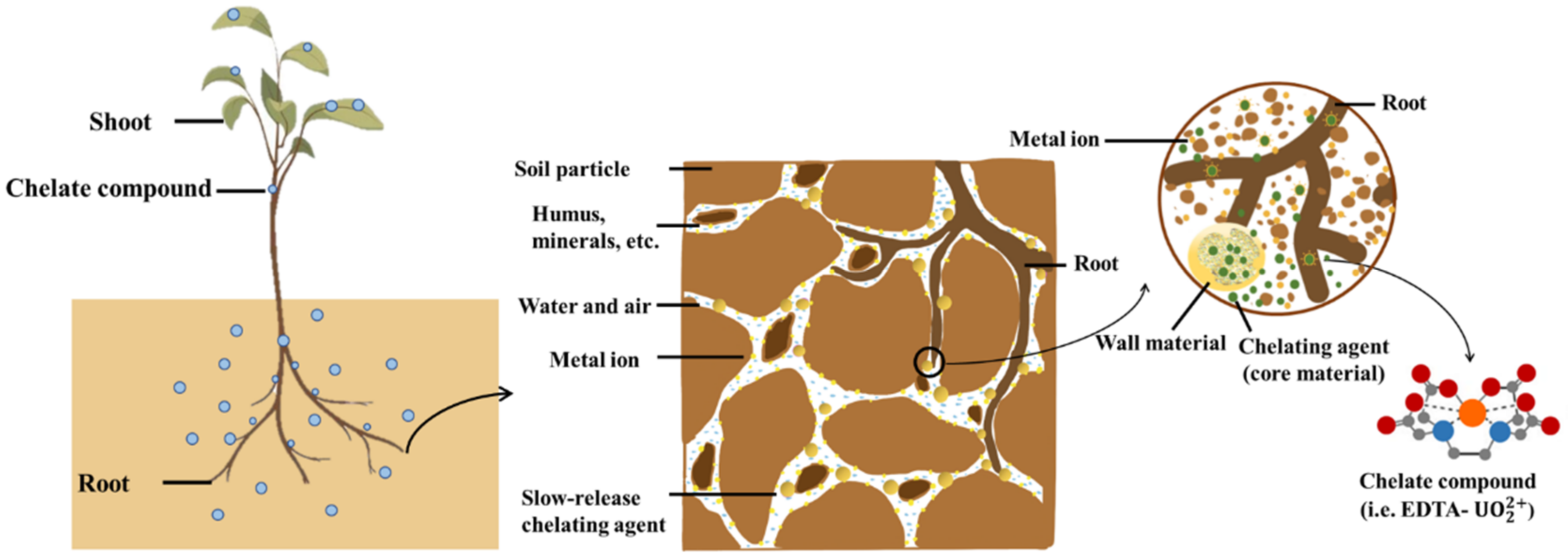
4. Slow-Release Chelating Agents
4.1. Slow-Release Technology
4.2. Slow-Release Chelating Agent-Assisted Phytoremediation
4.3. Evaluation of Slow-Release Performance
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Nuclear Association. World Nuclear Performance Report 2021—COP26 Edition; World Nuclear Performance Report; World Nuclear Association: London, UK, 2021; pp. 3–13. [Google Scholar]
- World Nuclear Association. The Nuclear Fuel Report: Global Scenarios for Demand and Supply Availability 2021–2040; World Nuclear Association: London, UK, 2021. [Google Scholar]
- Chen, L.; Liu, J.; Zhang, W.; Zhou, J.; Luo, D.; Li, Z. Uranium (U) Source, Speciation, Uptake, Toxicity and Bioremediation Strategies in Soil-Plant System: A Review. J. Hazard. Mater. 2021, 413, 125319. [Google Scholar] [CrossRef] [PubMed]
- Winde, F. Uranium Pollution of the Wonderfonteinspruit, 1997–2008 Part 1: Uranium Toxicity, Regional Background and Mining-Related Sources of Uranium Pollution. Water SA 2010, 36, 239–256. [Google Scholar]
- Yan, X.; Luo, X. Radionuclides Distribution, Properties, and Microbial Diversity of Soils in Uranium Mill Tailings from Southeastern China. J. Environ. Radioact. 2015, 139, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Liu, Z. Pollution Characteristics and Risk Assessment of Uranium and Heavy Metals of Agricultural Soil around the Uranium Tailing Reservoir in Southern China. J. Radioanal. Nucl. Chem. 2018, 318, 923–933. [Google Scholar] [CrossRef]
- Sabol, J. Uranium in the Beginning of the Nuclear Age: Reflections on the Historical Role of Jáchymov and an Overview of Early and Present Epidemiological Studies. In Uranium in Plants and the Environment; Gupta, D.K., Walther, C., Eds.; Springer International Publishing: Cham, Switzerland, 2020; ISBN 978-3-030-14961-1. [Google Scholar]
- Chen, L.; Wang, J.; Beiyuan, J.; Guo, X.; Wu, H.; Fang, L. Environmental and Health Risk Assessment of Potentially Toxic Trace Elements in Soils near Uranium (U) Mines: A Global Meta-Analysis. Sci. Total Environ. 2021, 816, 151556. [Google Scholar] [CrossRef]
- Gongalsky, K.B. Impact of Pollution Caused by Uranium Production on Soil Macrofauna. Environ. Monit. Assess. 2003, 89, 197–219. [Google Scholar] [CrossRef]
- Ma, W.; Gao, B.; Guo, Y.; Sun, Z.; Zhang, Y.; Chen, G.; Zhu, X.; Zhang, C. Occurrence and Distribution of Uranium in a Hydrological Cycle around a Uranium Mill Tailings Pond, Southern China. Int. J. Environ. Res. Public Health 2020, 17, 773. [Google Scholar] [CrossRef]
- Bourdon, B.; Turner, S.; Henderson, G.M.; Lundstrom, C.C. Introduction to U-Series Geochemistry. Rev. Mineral. Geochem. 2003, 52, 1–21. [Google Scholar] [CrossRef]
- Brugge, D.; de Lemos, J.L.; Oldmixon, B. Exposure Pathways and Health Effects Associated with Chemical and Radiological Toxicity of Natural Uranium: A Review. Rev. Environ. Health 2005, 20, 177–194. [Google Scholar] [CrossRef]
- Zhu, G.; Xiang, X.; Chen, X.; Wang, L.; Hu, H.; Weng, S. Renal Dysfunction Induced by Long-Term Exposure to Depleted Uranium in Rats. Arch. Toxicol. 2009, 83, 37–46. [Google Scholar] [CrossRef]
- Brugge, D.; Buchner, V. Health Effects of Uranium: New Research Findings. Doug Brugge Va Buchner 2011, 26, 231–249. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Lawrence Edwards, R.; Shen, C.-C.; Polyak, V.J.; Asmerom, Y.; Woodhead, J.; Hellstrom, J.; Wang, Y.; Kong, X.; Spötl, C.; et al. Improvements in 230Th Dating, 230Th and 234U Half-Life Values, and U–Th Isotopic Measurements by Multi-Collector Inductively Coupled Plasma Mass Spectrometry. Earth Planet. Sci. Lett. 2013, 371–372, 82–91. [Google Scholar] [CrossRef]
- Dewar, D. Uranium Mining: Environmental and Human Health Effects. In Nuclear Non-Proliferation in International Law—Volume IV: Human Perspectives on the Development and Use of Nuclear Energy; Black-Branch, J.L., Fleck, D., Eds.; T.M.C. Asser Press: Hague, The Netherlands, 2019; ISBN 978-94-6265-267-5. [Google Scholar]
- Ma, M.; Wang, R.; Xu, L.; Xu, M.; Liu, S. Emerging Health Risks and Underlying Toxicological Mechanisms of Uranium Contamination: Lessons from the Past Two Decades. Environ. Int. 2020, 145, 106107. [Google Scholar] [CrossRef] [PubMed]
- Meinrath, A.; Schneider, P.; Meinrath, G. Uranium Ores and Depleted Uranium in the Environment, with a Reference to Uranium in the Biosphere from the Erzgebirge/Sachsen, Germany. J. Environ. Radioact. 2003, 64, 175–193. [Google Scholar] [CrossRef]
- Mészáros, G.; Bognár, G.; Köteles, G.J. Long-Term Persistence of Chromosome Aberrations in Uranium Miners. J. Occup. Health 2004, 46, 310–315. [Google Scholar] [CrossRef]
- Wolf, G.; Arndt, D.; Kotschy-Lang, N.; Obe, G. Chromosomal Aberrations in Uranium and Coal Miners. Int. J. Radiat. Biol. 2009, 80, 147–153. [Google Scholar] [CrossRef]
- Duquène, L.; Vandenhove, H.; Tack, F.; Meers, E.; Baeten, J.; Wannijn, J. Enhanced Phytoextraction of Uranium and Selected Heavy Metals by Indian Mustard and Ryegrass Using Biodegradable Soil Amendments. Sci. Total Environ. 2009, 407, 1496–1505. [Google Scholar] [CrossRef]
- Mihalík, J.; Tlustoš, P.; Szaková, J. Comparison of Willow and Sunflower for Uranium Phytoextraction Induced by Citric Acid. J. Radioanal. Nucl. Chem. 2010, 285, 279–285. [Google Scholar] [CrossRef]
- Stojanović, M.D.; Stevanović, D.R.; Milojković, J.V.; Grubišić, M.S.; Ileš, D.A. Phytotoxic Effect of the Uranium on the Growing Up and Development the Plant of Corn. Water Air Soil Pollut. 2010, 209, 401–410. [Google Scholar] [CrossRef]
- Hou, J.; Wang, C.; Zhou, Y.; Li, S.; Hayat, T.; Alsaedi, A.; Wang, X. Effects of Uranium Stress on Physiological and Biochemical Characteristics in Seedlings of Six Common Edible Vegetables. J. Radioanal. Nucl. Chem. 2018, 316, 1001–1010. [Google Scholar] [CrossRef]
- Lai, J.; Liu, Z.; Luo, X. A Metabolomic, Transcriptomic Profiling, and Mineral Nutrient Metabolism Study of the Phytotoxicity Mechanism of Uranium. J. Hazard. Mater. 2020, 386, 121437. [Google Scholar] [CrossRef] [PubMed]
- Duquène, L.; Tack, F.; Meers, E.; Baeten, J.; Wannijn, J.; Vandenhove, H. Effect of Biodegradable Amendments on Uranium Solubility in Contaminated Soils. Sci. Total Environ. 2008, 391, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Lang, T.; Ding, D.; Hu, J.; Li, C.; Zhang, H.; Li, G. Enhancement of Repeated Applications of Chelates on Phytoremediation of Uranium Contaminated Soil by Macleaya cordata. J. Environ. Radioact. 2019, 199–200, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, J.; Wang, D. Phytoremediation of Uranium and Cadmium Contaminated Soils by Sunflower (Helianthus Annuus L.) Enhanced with Biodegradable Chelating Agents. J. Clean Prod. 2020, 263, 121491. [Google Scholar] [CrossRef]
- Balaram, V.; Rani, A.; Rathore, D.P.S. Uranium in Groundwater in Parts of India and World: A Comprehensive Review of Sources, Impact to the Environment and Human Health, Analytical Techniques, and Mitigation Technologies. Geosyst. Geoenviron. 2022, 1, 100043. [Google Scholar] [CrossRef]
- Kurttio, P.; Harmoinen, A.; Saha, H.; Salonen, L.; Karpas, Z.; Komulainen, H.; Auvinen, A. Kidney Toxicity of Ingested Uranium From Drinking Water. Am. J. Kidney Dis. 2006, 47, 972–982. [Google Scholar] [CrossRef]
- Bjørklund, G.; Semenova, Y.; Pivina, L.; Dadar, M.; Rahman, M.M.; Aaseth, J.; Chirumbolo, S. Uranium in Drinking Water: A Public Health Threat. Arch. Toxicol. 2020, 94, 1551–1560. [Google Scholar] [CrossRef]
- Arzuaga, X.; Gehlhaus, M.; Strong, J. Modes of Action Associated with Uranium Induced Adverse Effects in Bone Function and Development. Toxicol. Lett. 2015, 236, 123–130. [Google Scholar] [CrossRef]
- Guéguen, Y.; Souidi, M.; Baudelin, C.; Dudoignon, N.; Grison, S.; Dublineau, I.; Marquette, C.; Voisin, P.; Gourmelon, P.; Aigueperse, J. Short-Term Hepatic Effects of Depleted Uranium on Xenobiotic and Bile Acid Metabolizing Cytochrome P450 Enzymes in the Rat. Arch. Toxicol. 2006, 80, 187–195. [Google Scholar] [CrossRef]
- Wang, S.; Ran, Y.; Lu, B.; Li, J.; Kuang, H.; Gong, L.; Hao, Y. A Review of Uranium-Induced Reproductive Toxicity. Biol. Trace Elem. Res. 2020, 196, 204–213. [Google Scholar] [CrossRef]
- Angenard, G.; Muczynski, V.; Coffigny, H.; Duquenne, C.; Frydman, R.; Habert, R.; Livera, G.; Rouiller-Fabre, V. In Vitro Effects of Uranium on Human Fetal Germ Cells. Reprod. Toxicol. 2011, 31, 470–476. [Google Scholar] [CrossRef]
- Saint-Marc, B.; Elie, C.; Manens, L.; Tack, K.; Benderitter, M.; Gueguen, Y.; Ibanez, C. Chronic Uranium Contamination Alters Spinal Motor Neuron Integrity via Modulation of SMN1 Expression and Microglia Recruitment. Toxicol. Lett. 2016, 254, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Aide, M.T. Chemical Thermodynamics of Uranium in the Soil Environment. In Uranium—Safety, Resources, Separation and Thermodynamic Calculation; IntechOpen: London, UK, 2017; ISBN 978-1-78923-119-9. [Google Scholar]
- Wang, X.; Shi, Z.; Kinniburgh, D.G.; Zhao, L.; Ni, S.; Wang, R.; Hou, Y.; Cheng, K.; Zhu, B. Effect of Thermodynamic Database Selection on the Estimated Aqueous Uranium Speciation. J. Geochem. Explor. 2019, 204, 33–42. [Google Scholar] [CrossRef]
- Sarkar, D.; Andra, S.S.; Saminathan, S.K.M.; Datta, R. Chelant-Aided Enhancement of Lead Mobilization in Residential Soils. Environ. Pollut. 2008, 156, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Bucheli-Witschel, M.; Egli, T. Environmental Fate and Microbial Degradation of Aminopolycarboxylic Acids. Fems Microbiol. Rev. 2001, 25, 69–106. [Google Scholar] [CrossRef]
- Egli, T. Biodegradation of Metal-Complexing Aminopolycarboxylic Acids. J. Biosci. Bioeng. 2001, 92, 89–97. [Google Scholar] [CrossRef]
- Hu, Y.-H.; Wei, S.-H.; Zhou, Q.-X.; Zhan, J.; Ma, L.-H.; Niu, R.-C.; Li, Y.-M.; Wang, S.-S. Application of Chelator in Phytoremediation of Heavy Metals Contaminated Soils: A Review. J. Agro-Environ. Sci. 2010, 29, 2055–2063. [Google Scholar]
- Bian, X.; Cui, J.; Tang, B.; Yang, L. Chelant-Induced Phytoextraction of Heavy Metals from Contaminated Soils: A Review. Pol. J. Environ. Stud. 2018, 27, 2417–2424. [Google Scholar] [CrossRef]
- Pinto, I.S.S.; Neto, I.F.F.; Soares, H.M.V.M. Biodegradable Chelating Agents for Industrial, Domestic, and Agricultural Applications—a Review. Environ. Sci. Pollut. Res. 2014, 21, 11893–11906. [Google Scholar] [CrossRef]
- Huang, G.; You, J.; Zhou, X.; Ren, C.; Islam, M.S.; Hu, H. Effects of Low Molecular Weight Organic Acids on Cu Accumulation by Castor Bean and Soil Enzyme Activities. Ecotox. Environ. Safe 2020, 203, 110983. [Google Scholar] [CrossRef]
- Lai, H.-Y. Negative Effects of Chelants on Soil Qualities of Five Soil Series. Int. J. Phytoremediat. 2014, 17, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Meers, E.; Tack, F.M.G.; Verloo, M.G. Degradability of Ethylenediaminedisuccinic Acid (EDDS) in Metal Contaminated Soils: Implications for Its Use Soil Remediation. Chemosphere 2008, 70, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Baek, N.H.; Clesceri, N.L. NTA Biodegradation and Removal in Subsurface Sandy Soil. Water Res. 1986, 20, 345–349. [Google Scholar] [CrossRef]
- Means, J.L.; Kucak, T.; Crerar, D.A. Relative Degradation Rates of NTA, EDTA and DTPA and Environmental Implications. Environ. Pollut. 1980, 1, 45–60. [Google Scholar] [CrossRef]
- Wen, J.; Stacey, S.P.; McLaughlin, M.J.; Kirby, J.K. Biodegradation of Rhamnolipid, EDTA and Citric Acid in Cadmium and Zinc Contaminated Soils. Soil Biol. Biochem. 2009, 41, 2214–2221. [Google Scholar] [CrossRef]
- Gunina, A.; Smith, A.R.; Kuzyakov, Y.; Jones, D.L. Microbial Uptake and Utilization of Low Molecular Weight Organic Substrates in Soil Depend on Carbon Oxidation State. Biogeochemistry 2017, 133, 89–100. [Google Scholar] [CrossRef]
- Smith, R.M.; Martell, A.E. Critical Stability Constants Volume 6: Second Supplement; Springer: New York, NY, USA, 1989; Volume 6, ISBN 978-1-4615-6766-0. [Google Scholar]
- Leydier, A.; Lin, Y.; Arrachart, G.; Turgis, R.; Lecerclé, D.; Favre-Reguillon, A.; Taran, F.; Lemaire, M.; Pellet-Rostaing, S. EDTA and DTPA Modified Ligands as Sequestering Agents for Uranyl Decorporation. Tetrahedron 2012, 68, 1163–1170. [Google Scholar] [CrossRef]
- Smith, R.M.; Martell, A.E. Critical Stability Constants, Enthalpies and Entropies for the Formation of Metal Complexes of Aminopolycarboxylic Acids and Carboxylic Acids. Sci. Total Environ. 1987, 64, 125–147. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Chen, C.-Y. Stability Constants of Polymer-Bound Iminodiacetate-Type Chelating Agents with Some Transition-Metal Ions. J. Appl. Polym. Sci. 2002, 86, 1986–1994. [Google Scholar] [CrossRef]
- Vukovic, S.; Hay, B.P.; Bryantsev, V.S. Predicting Stability Constants for Uranyl Complexes Using Density Functional Theory. Inorg. Chem. 2015, 54, 3995–4001. [Google Scholar] [CrossRef]
- Singh, J.; Srivastav, A.N.; Singh, N.; Singh, A. Stability Constants of Metal Complexes in Solution. In Stability and Applications of Coordination Compounds; IntechOpen: London, UK, 2019; ISBN 978-1-83880-058-1. [Google Scholar]
- Tandy, S.; Bossart, K.; Mueller, R.; Ritschel, J.; Hauser, L.; Schulin, R.; Nowack, B. Extraction of Heavy Metals from Soils Using Biodegradable Chelating Agents. Environ. Sci. Technol. 2004, 38, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.J.; Luo, Y.M.; Zhao, Q.G.; Baker, A.J.M.; Christie, P.; Wong, M.H. Soil Cd Availability to Indian Mustard and Environmental Risk Following EDTA Addition to Cd-Contaminated Soil. Chemosphere 2003, 50, 813–818. [Google Scholar] [CrossRef]
- Wu, L.H.; Luo, Y.M.; Xing, X.R.; Christie, P. EDTA-Enhanced Phytoremediation of Heavy Metal Contaminated Soil with Indian Mustard and Associated Potential Leaching Risk. Agric. Ecosyst. Environ. 2004, 102, 307–318. [Google Scholar] [CrossRef]
- Liphadzi, M.S.; Kirkham, M.B. Availability and Plant Uptake of Heavy Metals in EDTA-Assisted Phytoremediation of Soil and Composted Biosolids. S. Afr. J. Bot. 2006, 72, 391–397. [Google Scholar] [CrossRef]
- Na, Z.; Yanshan, C.; Yu, F. Leaching of Cd and Pb from a Contaminated Soil with EDTA and EDDS: Effectiveness and Health Risk Assessment. Environ. Chem. 2011, 30, 958–963. [Google Scholar]
- Udovic, M.; Lestan, D. EDTA and HCl Leaching of Calcareous and Acidic Soils Polluted with Potentially Toxic Metals: Remediation Efficiency and Soil Impact. Chemosphere 2012, 88, 718–724. [Google Scholar] [CrossRef]
- Luo, C.; Shen, Z.; Li, X. Enhanced Phytoextraction of Cu, Pb, Zn and Cd with EDTA and EDDS. Chemosphere 2005, 59, 1–11. [Google Scholar] [CrossRef]
- Beiyuan, J.; Tsang, D.C.W.; Valix, M.; Baek, K.; Ok, Y.S.; Zhang, W.; Bolan, N.S.; Rinklebe, J.; Li, X.-D. Combined Application of EDDS and EDTA for Removal of Potentially Toxic Elements under Multiple Soil Washing Schemes. Chemosphere 2018, 205, 178–187. [Google Scholar] [CrossRef]
- Tao, Y.; Brigante, M.; Zhang, H.; Mailhot, G. Phenanthrene Degradation Using Fe(III)-EDDS Photoactivation under Simulated Solar Light: A Model for Soil Washing Effluent Treatment. Chemosphere 2019, 236, 124366. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, H.; Li, B.; Lai, Y.; Zang, L.; Tang, X. Process Design and Validation of a New Mixed Eluent for Leaching Cd, Cr, Pb, Cu, Ni, and Zn from Heavy Metal-Polluted Soil. Anal. Methods 2021, 13, 1269–1277. [Google Scholar] [CrossRef]
- Leibold, E.; Deckardt, K.; Mellert, W.; Potthoff-Karl, B.; Grundler, O.; Jäckh, R. NTA and Fe(III)NTA: Differential Patterns of Renal Toxicity in Subchronic Studies. Hum. Exp. Toxicol. 2002, 21, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Toyokuni, S. Reactive Oxygen Species-Induced Molecular Damage and Its Application in Pathology. Pathol. Int. 1999, 49, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.; Okazaki, Y.; Okada, S. Curcumin Attenuates Oxidative Damage in Animals Treated with a Renal Carcinogen, Ferric Nitrilotriacetate (Fe-NTA): Implications for Cancer Prevention. Mol. Cell. Biochem. 2009, 324, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T. Development of Chemical Tools for Imaging of Fe(II) Ions in Living Cells: A Review. Acta Histochem. Cytochem. 2018, 51, 137–143. [Google Scholar] [CrossRef]
- Huang, J.W.; Blaylock, M.J.; Kapulnik, Y.; Ensley, B.D. Phytoremediation of Uranium-Contaminated Soils: Role of Organic Acids in Triggering Uranium Hyperaccumulation in Plants. Environ. Sci. Technol. 1998, 32, 2004–2008. [Google Scholar] [CrossRef]
- Gavrilescu, M.; Pavel, L.V.; Cretescu, I. Characterization and Remediation of Soils Contaminated with Uranium. J. Hazard. Mater. 2009, 163, 475–510. [Google Scholar] [CrossRef]
- Prieto, C.; Lozano, J.C.; Rodríguez, P.B.; Tomé, F.V. Enhancing Radium Solubilization in Soils by Citrate, EDTA, and EDDS Chelating Amendments. J. Hazard. Mater. 2013, 250–251, 439–446. [Google Scholar] [CrossRef]
- Orr, R.; Hocking, R.K.; Pattison, A.; Nelson, P.N. Extraction of Metals from Mildly Acidic Tropical Soils: Interactions between Chelating Ligand, pH and Soil Type. Chemosphere 2020, 248, 126060. [Google Scholar] [CrossRef]
- Pepper, I.L.; Brusseau, M.L. Chapter 2—Physical-Chemical Characteristics of Soils and the Subsurface. In Environmental and Pollution Science, 3rd ed.; Brusseau, M.L., Pepper, I.L., Gerba, C.P., Eds.; Academic Press: Cambridge, MA, USA, 2019; ISBN 978-0-12-814719-1. [Google Scholar]
- Rakhsh, F.; Golchin, A.; Beheshti Al Agha, A.; Nelson, P.N. Mineralization of Organic Carbon and Formation of Microbial Biomass in Soil: Effects of Clay Content and Composition and the Mechanisms Involved. Soil Biol. Biochem. 2020, 151, 108036. [Google Scholar] [CrossRef]
- Crançon, P.; van der Lee, J. Speciation and Mobility of Uranium(VI) in Humic-Containing Soils. Radiochim. Acta 2003, 91, 673–679. [Google Scholar] [CrossRef]
- Kantar, C. Heterogeneous Processes Affecting Metal Ion Transport in the Presence of Organic Ligands: Reactive Transport Modeling. Earth-Sci. Rev. 2007, 81, 175–198. [Google Scholar] [CrossRef]
- Ratnikov, A.N.; Sviridenko, D.G.; Popova, G.I.; Sanzharova, N.I.; Mikailova, R.A. The Behaviour of Uranium in Soils and the Mechanisms of Its Accumulation by Agricultural Plants. In Uranium in Plants and the Environment; Gupta, D.K., Walther, C., Eds.; Springer International Publishing: Cham, Switzerland, 2020; ISBN 978-3-030-14961-1. [Google Scholar]
- Jiang, X.; Li, X.; Zhang, J.; Lv, J.; Chang, J.; Cui, D. Study of Citric Acid on Extraction and Washing from Different Soil Polluted by Cr. Chin. Agric. Sci. Bull. 2012, 28, 278–281. [Google Scholar]
- Wang, K.; Liu, Y.; Song, Z.; Khan, Z.H.; Qiu, W. Effects of Biodegradable Chelator Combination on Potentially Toxic Metals Leaching Efficiency in Agricultural Soils. Ecotox. Environ. Safe 2019, 182, 109399. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Luo, C.; Zhang, G.; Li, X.; Shen, Z. Extraction of Heavy Metals from E-Waste Contaminated Soils Using EDDS. J. Environ. Sci. 2012, 24, 1985–1994. [Google Scholar] [CrossRef]
- Kantar, C.; Honeyman, B.D. Citric Acid Enhanced Remediation of Soils Contaminated with Uranium by Soil Flushing and Soil Washing. J. Environ. Eng. 2006, 132, 247–255. [Google Scholar] [CrossRef]
- Ebbs, S.D.; Norvell, W.A.; Kochian, L.V. The Effect of Acidification and Chelating Agents on the Solubilization of Uranium from Contaminated Soil. J. Environ. Qual. 1998, 27, 1486–1494. [Google Scholar] [CrossRef]
- Lozano, J.C.; Blanco Rodríguez, P.; Vera Tomé, F.; Calvo, C.P. Enhancing Uranium Solubilization in Soils by Citrate, EDTA, and EDDS Chelating Amendments. J. Hazard. Mater. 2011, 198, 224–231. [Google Scholar] [CrossRef]
- Sarthou, M.C.M.; Devime, F.; Baggio, C.; Figuet, S.; Alban, C.; Bourguignon, J.; Ravanel, S. Calcium-Permeable Cation Channels Are Involved in Uranium Uptake in Arabidopsis thaliana. J. Hazard. Mater. 2022, 424, 127436. [Google Scholar] [CrossRef]
- Croteau, M.-N.; Fuller, C.C.; Cain, D.J.; Campbell, K.M.; Aiken, G. Biogeochemical Controls of Uranium Bioavailability from the Dissolved Phase in Natural Freshwaters. Available online: https://pubs.acs.org/doi/pdf/10.1021/acs.est.6b02406 (accessed on 1 April 2022).
- Shahandeh, H.; Hossner, L.R. Enhancement of Uranium Phytoaccumulation from Contaminated Soils. Soil Sci. 2002, 167, 269–280. [Google Scholar] [CrossRef]
- Shahandeh, H.; Hossner, L. Role of Soil Properties in Phytoaccumulation of Uranium. Water Air Soil Pollut. 2002, 141, 165–180. [Google Scholar] [CrossRef]
- Laurette, J.; Larue, C.; Llorens, I.; Jaillard, D.; Jouneau, P.-H.; Bourguignon, J.; Carrière, M. Speciation of Uranium in Plants upon Root Accumulation and Root-to-Shoot Translocation: A XAS and TEM Study. Environ. Exp. Bot. 2012, 77, 87–95. [Google Scholar] [CrossRef]
- Shahid, M.; Austruy, A.; Echevarria, G.; Arshad, M.; Sanaullah, M.; Aslam, M.; Nadeem, M.; Nasim, W.; Dumat, C. EDTA-Enhanced Phytoremediation of Heavy Metals: A Review. Soil. Sediment. Contam. 2013, 23, 389–416. [Google Scholar] [CrossRef]
- Zhou, P.; Gu, B. Extraction of Oxidized and Reduced Forms of Uranium from Contaminated Soils: Effects of Carbonate Concentration and pH. Environ. Sci. Technol. 2005, 39, 4435–4440. [Google Scholar] [CrossRef] [PubMed]
- Doustaly, F.; Combes, F.; Fiévet, J.B.; Berthet, S.; Hugouvieux, V.; Bastien, O.; Aranjuelo, I.; Leonhardt, N.; Rivasseau, C.; Carrière, M.; et al. Uranium Perturbs Signaling and Iron Uptake Response in Arabidopsis thaliana Roots. Metallomics 2014, 6, 809–821. [Google Scholar] [CrossRef] [PubMed]
- Berthet, S.; Villiers, F.; Alban, C.; Serre, N.B.C.; Martin-Laffon, J.; Figuet, S.; Boisson, A.-M.; Bligny, R.; Kuntz, M.; Finazzi, G.; et al. Arabidopsis thaliana Plants Challenged with Uranium Reveal New Insights into Iron and Phosphate Homeostasis. New Phytol. 2018, 217, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Malaviya, P.; Singh, A. Phytoremediation Strategies for Remediation of Uranium-Contaminated Environments: A Review. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2575–2647. [Google Scholar] [CrossRef]
- Chang, P.; Kim, K.-W.; Yoshida, S.; Kim, S.-Y. Uranium Accumulation of Crop Plants Enhanced by Citric Acid. Environ. Geochem. Health 2005, 27, 529–538. [Google Scholar] [CrossRef]
- Rong, L.; Zhang, S.; Wang, J.; Li, S.; Xie, S.; Wang, G. Phytoremediation of Uranium-Contaminated Soil by Perennial Ryegrass (Lolium perenne L.) Enhanced with Citric Acid Application. Environ. Sci. Pollut. Res. 2022, 29, 33002–33012. [Google Scholar] [CrossRef]
- Li, C.; Dan, W.; Chan, L.; Cui, Z. Effect of Three Kinds of Chelating Agents on Phytoremediation in U and Cd Contaminated Soil by Zebrina pendula. Atomic Energy Sci. Technol. 2018, 52, 1359–1366. [Google Scholar] [CrossRef]
- Manara, A.; Fasani, E.; Furini, A.; DalCorso, G. Evolution of the Metal Hyperaccumulation and Hypertolerance Traits. Plant Cell Environ. 2020, 43, 2969–2986. [Google Scholar] [CrossRef]
- Sevostianova, E.; Lindemann, W.C.; Ulery, A.L.; Remmenga, M.D. Plant Uptake of Depleted Uranium from Manure-Amended and Citrate Treated Soil. Int. J. Phytoremediat. 2010, 12, 550–561. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Pan, N.; Jin, Y.; Xia, C. Study on remediation for uranium contaminated soils enhanced by chelator using brassica mustard. In Progress Report on Nuclear Science and Technology in China; Atomic Energy Press: Beijing, China, 2012; Volume 2, ISBN 978-7-5022-5602-9. [Google Scholar]
- Qi, F.; Zha, Z.; Du, L.; Feng, X.; Wang, D.; Zhang, D.; Fang, Z.; Ma, L.; Jin, Y.; Xia, C. Impact of Mixed Low-Molecular-Weight Organic Acids on Uranium Accumulation and Distribution in a Variant of Mustard (Brassica juncea VarTumida). J. Radioanal. Nucl. Chem. 2014, 302, 149–159. [Google Scholar] [CrossRef]
- Nowack, B.; Schulin, R.; Robinson, B.H. Critical Assessment of Chelant-Enhanced Metal Phytoextraction. Environ. Sci. Technol. 2006, 40, 5225–5232. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, Q.; Cui, Y.; Dong, Y.; Christie, P. Slow Release Chelate Enhancement of Lead Phytoextraction by Corn (Zea mays L.) from Contaminated Soil—A Preliminary Study. Sci. Total Environ. 2005, 339, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.K. Heavy Metals Toxicity in Plants: An Overview on the Role of Glutathione and Phytochelatins in Heavy Metal Stress Tolerance of Plants. S. Afr. J. Bot. 2010, 76, 167–179. [Google Scholar] [CrossRef]
- Yan, A.; Wang, Y.; Tan, S.N.; Mohd Yusof, M.L.; Ghosh, S.; Chen, Z. Phytoremediation: A Promising Approach for Revegetation of Heavy Metal-Polluted Land. Front. Plant Sci. 2020, 11, 359. [Google Scholar] [CrossRef] [PubMed]
- Pietrini, I.; Grifoni, M.; Franchi, E.; Cardaci, A.; Pedron, F.; Barbafieri, M.; Petruzzelli, G.; Vocciante, M. Enhanced Lead Phytoextraction by Endophytes from Indigenous Plants. Soil Syst. 2021, 5, 55. [Google Scholar] [CrossRef]
- Römkens, P.; Bouwman, L.; Japenga, J.; Draaisma, C. Potentials and Drawbacks of Chelate-Enhanced Phytoremediation of Soils. Environ. Pollut. 2002, 116, 109–121. [Google Scholar] [CrossRef]
- Epelde, L.; Ma Becerril, J.; Alkorta, I.; Garbisu, C. Heavy Metal Phytoremediation: Microbial Indicators of Soil Health for the Assessment of Remediation Efficiency. In Advances in Applied Bioremediation; Singh, A., Kuhad, R.C., Ward, O.P., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-540-89621-0. [Google Scholar]
- Lee, J.; Sung, K. Effects of Chelates on Soil Microbial Properties, Plant Growth and Heavy Metal Accumulation in Plants. Ecol. Eng. 2014, 73, 386–394. [Google Scholar] [CrossRef]
- Cao, A.; Carucci, A.; Lai, T.; La Colla, P.; Tamburini, E. Effect of Biodegradable Chelating Agents on Heavy Metals Phytoextraction with Mirabilis jalapa and on Its Associated Bacteria. Eur. J. Soil Biol. 2007, 43, 200–206. [Google Scholar] [CrossRef]
- Barona, A.; Aranguiz, I.; Elías, A. Metal Associations in Soils before and after EDTA Extractive Decontamination: Implications for the Effectiveness of Further Clean-up Procedures. Environ. Pollut. 2001, 113, 79–85. [Google Scholar] [CrossRef]
- Udovic, M.; Lestan, D. Pb, Zn and Cd Mobility, Availability and Fractionation in Aged Soil Remediated by EDTA Leaching. Chemosphere 2009, 74, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Luo, D.; Chen, X.; Wei, L.; Liu, Y.; Wu, Q.; Xiao, T.; Mai, X.; Liu, G.; Liu, L. Insights into Heavy Metals Leakage in Chelator-Induced Phytoextraction of Pb- and Tl-Contaminated Soil. Int. J. Environ. Res. Public Health 2019, 16, 1328. [Google Scholar] [CrossRef] [PubMed]
- Thayalakumaran, T.; Robinson, B.; Vogeler, I.; Scotter, D.; Clothier, B.; Percival, H. Plant Uptake and Leaching of Copper during EDTA-Enhanced Phytoremediation of Repacked and Undisturbed Soil. Plant Soil 2003, 254, 415–423. [Google Scholar] [CrossRef]
- Mastromatteo, M.; Mastromatteo, M.; Conte, A.; del Nobile, M.A. Advances in Controlled Release Devices for Food Packaging Applications. Trends Food Sci. Technol. 2010, 21, 591–598. [Google Scholar] [CrossRef]
- Yusoff, S.N.M.; Kamari, A.; Aljafree, N.F.A. A Review of Materials Used as Carrier Agents in Pesticide Formulations. Int. J. Environ. Sci. Technol. 2016, 13, 2977–2994. [Google Scholar] [CrossRef]
- Al-Rawajfeh, A.E.; Alrbaihat, M.R.; AlShamaileh, E.M. Chapter 4—Characteristics and Types of Slow- and Controlled-Release Fertilizers. In Controlled Release Fertilizers for Sustainable Agriculture; Lewu, F.B., Volova, T., Thomas, S., Rakhimol, K.R., Eds.; Academic Press: Cambridge, MA, USA, 2021; ISBN 978-0-12-819555-0. [Google Scholar]
- Lubkowski, K.; Grzmil, B. Controlled Release Fertilizers. Pol. J. Chem. Technol. 2007, 9, 83–84. [Google Scholar] [CrossRef]
- Liu, G.; Zotarelli, L.; Li, Y.; Dinkins, D.; Wang, Q.; Ozores-Hampton, M. Controlled-Release and Slow-Release Fertilizers as Nutrient Management Tools. Available online: https://edis.ifas.ufl.edu/publication/HS1255 (accessed on 21 September 2021).
- Almasi, H.; Oskouie, M.J.; Saleh, A. A Review on Techniques Utilized for Design of Controlled Release Food Active Packaging. Crit. Rev. Food Sci. Nutr. 2020, 61, 2601–2621. [Google Scholar] [CrossRef]
- Kuai, L.; Liu, F.; Chiou, B.-S.; Avena-Bustillos, R.J.; McHugh, T.H.; Zhong, F. Controlled Release of Antioxidants from Active Food Packaging: A Review. Food Hydrocoll. 2021, 120, 106992. [Google Scholar] [CrossRef]
- Vasile, C.; Baican, M. Progresses in Food Packaging, Food Quality, and Safety—Controlled-Release Antioxidant and/or Antimicrobial Packaging. Molecules 2021, 26, 1263. [Google Scholar] [CrossRef]
- Heirlings, L.; Siró, I.; Devlieghere, F.; Bavel, E.V.; Cool, P.; Meulenaer, B.D.; Vansant, E.F.; Debevere, J. Influence of Polymer Matrix and Adsorption onto Silica Materials on the Migration of α-Tocopherol into 95% Ethanol from Active Packaging. Food Addit. Contam. 2007, 21, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Cerisuelo, J.P.; Bermúdez, J.M.; Aucejo, S.; Catalá, R.; Gavara, R.; Hernández-Muñoz, P. Describing and Modeling the Release of an Antimicrobial Agent from an Active PP/EVOH/PP Package for Salmon. J. Food Eng. 2013, 116, 352–361. [Google Scholar] [CrossRef]
- LaCoste, A.; Schaich, K.M.; Zumbrunnen, D.; Yam, K.L. Advancing Controlled Release Packaging through Smart Blending. Packag. Technol. Sci. 2005, 18, 77–87. [Google Scholar] [CrossRef]
- Han, J.H.; Floros, J.D. Simulating Diffusion Model and Determining Diffusivity of Potassium Sorbate Through Plastics to Develop Antimicrobial Packaging Films. J. Food Process Preserv. 1998, 22, 107–122. [Google Scholar] [CrossRef]
- Whelehan, M.; Marison, I.W. Microencapsulation Using Vibrating Technology. J. Microencapsul. 2011, 28, 669–688. [Google Scholar] [CrossRef] [PubMed]
- Rathore, S.; Desai, P.M.; Liew, C.V.; Chan, L.W.; Heng, P.W.S. Microencapsulation of Microbial Cells. J. Food Eng. 2013, 116, 369–381. [Google Scholar] [CrossRef]
- Xie, Z.; Wu, L.; Chen, N.; Liu, C.; Zheng, Y.; Xu, S.; Li, F.; Xu, Y. Phytoextraction of Pb and Cu Contaminated Soil With Maize and Microencapsulated EDTA. Int. J. Phytoremediat. 2012, 14, 727–740. [Google Scholar] [CrossRef][Green Version]
- Ding, C.; Li, Y. A Slow-Release Chelating Agent for Heavy Metal Soil Remediation. Patent Registration Number 201910960626, 14 January 2020. [Google Scholar]
- Ocak, B. Complex Coacervation of Collagen Hydrolysate Extracted from Leather Solid Wastes and Chitosan for Controlled Release of Lavender Oil. J. Environ. Manag. 2012, 100, 22–28. [Google Scholar] [CrossRef]
- O’Connor, D.; Hou, D.; Ok, Y.S.; Song, Y.; Sarmah, A.K.; Li, X.; Tack, F.M.G. Sustainable in Situ Remediation of Recalcitrant Organic Pollutants in Groundwater with Controlled Release Materials: A Review. J. Control Release 2018, 283, 200–213. [Google Scholar] [CrossRef]
- Christenson, M.; Kambhu, A.; Reece, J.; Comfort, S.; Brunner, L. A Five-Year Performance Review of Field-Scale, Slow-Release Permanganate Candles with Recommendations for Second-Generation Improvements. Chemosphere 2016, 150, 239–247. [Google Scholar] [CrossRef]
- Tang, X.; Yu, C.; Lei, Y.; Wang, Z.; Wang, C.; Wang, J. A Novel Chitosan-Urea Encapsulated Material for Persulfate Slow-Release to Degrade Organic Pollutants. J. Hazard. Mater. 2022, 426, 128083. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.T.; Liu, J.K.; Chang, Y.M.; Chen, K.F.; Kao, C.M. Application of Polycolloid-Releasing Substrate to Remediate Trichloroethylene-Contaminated Groundwater: A Pilot-Scale Study. J. Hazard. Mater. 2014, 268, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Yeum, Y.; Han, K.; Kang, J.-H.; Kim, D.-W.; Park, C.-W.; Kwon, S.; Kim, Y. Production, Characterization, and Evaluation of Two Types of Slow-Releasing Carbon Source Tablets for in-Situ Heterotrophic Nitrate Denitrification in Aquifers. Chemosphere 2020, 260, 127478. [Google Scholar] [CrossRef] [PubMed]
- Essawy, H.A.; Ghazy, M.B.M.; El-Hai, F.A.; Mohamed, M.F. Superabsorbent Hydrogels via Graft Polymerization of Acrylic Acid from Chitosan-Cellulose Hybrid and Their Potential in Controlled Release of Soil Nutrients. Int. J. Biol. Macromol. 2016, 89, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Pang, L.; Gao, Z.; Feng, H.; Wang, S.; Wang, Q. Cellulose Based Materials for Controlled Release Formulations of Agrochemicals: A Review of Modifications and Applications. J. Control Release 2019, 316, 105–115. [Google Scholar] [CrossRef]
- Wang, G.; Wang, B.; Fan, W.; Deng, N. Enhanced Phytoremediation of Uranium-Contaminated Soils by Indian Mustard (Brassica juncea L.) Using Slow Release Citric Acid. Environ. Sci. Pollut. Res. 2021, 28, 61061–61071. [Google Scholar] [CrossRef]
- Shibata, M.; Konno, T.; Akaike, R.; Xu, Y.; Shen, R.; Ma, J.F. Phytoremediation of Pb Contaminated Soil with Polymer-Coated EDTA. Plant Soil 2007, 290, 201–208. [Google Scholar] [CrossRef]
- Xie, Z.; Chen, N.; Liu, C.; Zhou, J.; Xu, S.; Zheng, Y.; Li, F.; Xu, Y. Synthesis and Characterization of Ethylenediamine Tetraacetic Acid Tetrasodium Salt Loaded in Microcapsules with Slow Release Properties. Chin. J. Chem. Eng. 2010, 18, 149–155. [Google Scholar] [CrossRef]
- Yoo, J.; Won, Y.-Y. Phenomenology of the Initial Burst Release of Drugs from PLGA Microparticles. ACS Biomater. Sci. Eng. 2020, 6, 6053–6062. [Google Scholar] [CrossRef]
- Chen, J.; Lü, S.; Zhang, Z.; Zhao, X.; Li, X.; Ning, P.; Liu, M. Environmentally Friendly Fertilizers: A Review of Materials Used and Their Effects on the Environment. Sci. Total Environ. 2018, 613–614, 829–839. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, G.; Lu, J.; Liang, H. Preparation and Characterization of Carboxymethyl Cellulose/Polyvinyl Alcohol Blend Film as a Potential Coating Material. Polym. Plast. Technol. Eng. 2013, 52, 163–167. [Google Scholar] [CrossRef]
- Han, X.; Chen, S.; Hu, X. Controlled-Release Fertilizer Encapsulated by Starch/Polyvinyl Alcohol Coating. Desalination 2009, 240, 21–26. [Google Scholar] [CrossRef]
- Anghel, N.; Marius, N.; Spiridon, I. Heavy Metal Adsorption Ability of a New Composite Material Based on Starch Strengthened with Chemically Modified Cellulose. Polym. Adv. Technol. 2019, 30, 1453–1460. [Google Scholar] [CrossRef]
- Wan, M.-W.; Petrisor, I.G.; Lai, H.-T.; Kim, D.; Yen, T.F. Copper Adsorption through Chitosan Immobilized on Sand to Demonstrate the Feasibility for in Situ Soil Decontamination. Carbohydr. Polym. 2004, 55, 249–254. [Google Scholar] [CrossRef]
- Zhou, Y.; Gao, B.; Zimmerman, A.R.; Fang, J.; Sun, Y.; Cao, X. Sorption of Heavy Metals on Chitosan-Modified Biochars and Its Biological Effects. Chem. Eng. J. 2013, 231, 512–518. [Google Scholar] [CrossRef]
- Li, B.; Li, M.; Zhang, P.; Pan, Y.; Huang, Z.; Xiao, H. Remediation of Cd (II) Ions in Aqueous and Soil Phases Using Novel Porous Cellulose/Chitosan Composite Spheres Loaded with Zero-Valent Iron Nanoparticles. React. Funct. Polym. 2022, 173, 105210. [Google Scholar] [CrossRef]
- O’Connell, D.W.; Birkinshaw, C.; O’Dwyer, T.F. Heavy Metal Adsorbents Prepared from the Modification of Cellulose: A Review. Bioresour. Technol. 2008, 99, 6709–6724. [Google Scholar] [CrossRef]
- Kos, B.; Le tan, D. Influence of a Biodegradable ([S,S]-EDDS) and Nondegradable (EDTA) Chelate and Hydrogel Modified Soil Water Sorption Capacity on Pb Phytoextraction and Leaching. Plant Soil 2003, 253, 403–411. [Google Scholar] [CrossRef]
- Noppakundilograt, S.; Pheatcharat, N.; Kiatkamjornwong, S. Multilayer-Coated NPK Compound Fertilizer Hydrogel with Controlled Nutrient Release and Water Absorbency. J. Appl. Polym. Sci. 2015, 132, 41249. [Google Scholar] [CrossRef]
- Ramli, R.A. Slow Release Fertilizer Hydrogels: A Review. Polym. Chem. 2019, 10, 6073–6090. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Lu, P.; Zhang, M. Preparation and Properties of Hydrogel Based on Sawdust Cellulose for Environmentally Friendly Slow Release Fertilizers. Green Process. Synth. 2020, 9, 139–152. [Google Scholar] [CrossRef]
- Kareem, S.A.; Dere, I.; Gungula, D.T.; Andrew, F.P.; Saddiq, A.M.; Adebayo, E.F.; Tame, V.T.; Kefas, H.M.; Joseph, J.; Patrick, D.O. Synthesis and Characterization of Slow-Release Fertilizer Hydrogel Based on Hydroxy Propyl Methyl Cellulose, Polyvinyl Alcohol, Glycerol and Blended Paper. Gels 2021, 7, 262. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Rubina Gilani, S.; Iqbal Durani, A.; Naseem, S. Materials Diversity of Hydrogel: Synthesis, Polymerization Process and Soil Conditioning Properties in Agricultural Field. J. Adv. Res. 2021, 33, 15–40. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, S.; Xu, L.; Li, Q. The Slow-Release Chelating Agent and Its Preparation Method for Phytoremediation of Soil. Patent Registration Number 201610567644.8, 7 December 2016. [Google Scholar]
- Chen, L.; Wang, D.; Long, C.; Cui, Z. Effect of Biodegradable Chelators on Induced Phytoextraction of Uranium- and Cadmium- Contaminated Soil by Zebrina Pendul. Schnizl. Sci. Rep. 2019, 9, 19817. [Google Scholar] [CrossRef]
- Arshad, M.; Naqvi, N.; Gul, I.; Yaqoob, K.; Bilal, M.; Kallerhoff, J. Lead Phytoextraction by Pelargonium hortorum: Comparative Assessment of EDTA and DIPA for Pb Mobility and Toxicity. Sci. Total Environ. 2020, 748, 141496. [Google Scholar] [CrossRef]
- Broschat, T.K.; Moore, K.K. Release Rates of Ammonium-Nitrogen, Nitrate-Nitrogen, Phosphorus, Potassium, Magnesium, Iron, and Manganese from Seven Controlled-Release Fertilizers. Commun. Soil Sci. Plant Anal. 2007, 38, 843–850. [Google Scholar] [CrossRef]
- Dai, J.; Fan, X.; Yu, J.; Liu, F.; Zhang, Q. Study on the Rapid Method to Predict Longevity of Controlled Release Fertilizer Coated by Water Soluble Resin. Agric. Sci. China 2008, 7, 1127–1132. [Google Scholar] [CrossRef]
- Medina, L.C.; Sartain, J.B.; Obreza, T.A.; Hall, W.L.; Thiex, N.J. Evaluation of a Soil Incubation Method to Characterize Nitrogen Release Patterns of Slow- and Controlled-Release Fertilizers. J. AOAC Int. 2014, 97, 643–660. [Google Scholar] [CrossRef]
- Jia, Y.; Hu, Z.; Mu, J.; Zhang, W.; Xie, Z.; Wang, G. Preparation of Biochar as a Coating Material for Biochar-Coated Urea. Sci. Total Environ. 2020, 731, 139063. [Google Scholar] [CrossRef]
- Purnomo, C.W.; Saputra, H. Chapter 6—Manufacturing of Slow and Controlled Release Fertilizer. In Controlled Release Fertilizers for Sustainable Agriculture; Lewu, F.B., Volova, T., Thomas, S., Rakhimol, K.R., Eds.; Academic Press: Cambridge, MA, USA, 2021; ISBN 978-0-12-819555-0. [Google Scholar]
- Kottegoda, N.; Sandaruwan, C.; Priyadarshana, G.; Siriwardhana, A.; Rathnayake, U.A.; Berugoda Arachchige, D.M.; Kumarasinghe, A.R.; Dahanayake, D.; Karunaratne, V.; Amaratunga, G.A.J. Urea-Hydroxyapatite Nanohybrids for Slow Release of Nitrogen. ACS Nano 2017, 11, 1214–1221. [Google Scholar] [CrossRef]
- Ni, B.; Liu, M.; Lü, S. Multifunctional Slow-Release Urea Fertilizer from Ethylcellulose and Superabsorbent Coated Formulations. Chem. Eng. J. 2009, 155, 892–898. [Google Scholar] [CrossRef]
- Xie, Z.; Chen, N.; Xu, S.; Xu, Y. A Complexing Agent Microcapsule for Phytoremediation of Soil and Its Preparation Method. Patent Registration Number 200810220712.9, 2 September 2009. [Google Scholar]
- Cagnasso, C.E.; López, L.B.; Rodríguez, V.G.; Valencia, M.E. Development and Validation of a Method for the Determination of EDTA in Non-Alcoholic Drinks by HPLC. J. Food Compos. Anal. 2007, 20, 248–251. [Google Scholar] [CrossRef]
- Khan, M.S.; Zaidi, A.; Wani, P.A.; Oves, M. Role of Plant Growth Promoting Rhizobacteria in the Remediation of Metal Contaminated Soils. Environ. Chem. Lett. 2009, 7, 1–19. [Google Scholar] [CrossRef]
- Saleh, H.M. Water Hyacinth for Phytoremediation of Radioactive Waste Simulate Contaminated with Cesium and Cobalt Radionuclides. Nucl. Eng. Des. 2012, 242, 425–432. [Google Scholar] [CrossRef]
- Saleh, H.M.; Bayoumi, T.A.; Mahmoud, H.H.; Aglan, R.F. Uptake of Cesium and Cobalt Radionuclides from Simulated Radioactive Wastewater by Ludwigia stolonifera Aquatic Plant. Nucl. Eng. Des. 2017, 315, 194–199. [Google Scholar] [CrossRef]
- Saleh, H.M.; Aglan, R.F.; Mahmoud, H.H. Ludwigia stolonifera for Remediation of Toxic Metals from Simulated Wastewater. Chem. Ecol. 2018, 35, 164–178. [Google Scholar] [CrossRef]
- Saleh, H.M.; Mahmoud, H.H.; Aglan, R.F.; Bayoumi, T.A. Biological Treatment of Wastewater Contaminated with Cu (II), Fe (II) and Mn (II) Using Ludwigia stolonifera Aquatic Plant. Environ. Eng. Manag. J. 2019, 18, 1327–1336. [Google Scholar] [CrossRef]
- Saleh, H.M.; Moussa, H.R.; El-Saied, F.A.; Dawoud, M.; Nouh, E.S.A.; Abdel Wahed, R.S. Adsorption of Cesium and Cobalt onto Dried Myriophyllum spicatum L. from Radio-Contaminated Water: Experimental and Theoretical Study. Prog. Nucl. Energy 2020, 125, 103393. [Google Scholar] [CrossRef]
- Saleh, H.M.; Moussa, H.R.; Mahmoud, H.H.; El-Saied, F.A.; Dawoud, M.; Abdel Wahed, R.S. Potential of the Submerged Plant Myriophyllum spicatum for Treatment of Aquatic Environments Contaminated with Stable or Radioactive Cobalt and Cesium. Prog. Nucl. Energy 2020, 118, 103147. [Google Scholar] [CrossRef]
| Category | Chelator | Chelator Structure | Biodegradability | U Chelate Structure | logKU * |
|---|---|---|---|---|---|
| Aminopolycarboxylate chelating agents (APCAs) | Ethylenediaminetetraacetic acid (EDTA) | 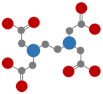 | Half-life > 40 d [46] |  | 7.4 (UO22+:EDTA = 1:1) 17.87 (UO22+:EDTA = 2:1) |
| Ethylenediaminedisuccinic acid (EDDS) | 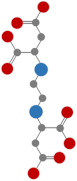 | Completely degraded after 54 d [47] | 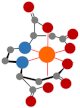 | 10.7 | |
| Nitrilotriacetic acid (NTA) | 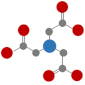 | 75% degraded after 21 d [48] | 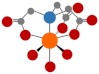 | 9.5 | |
| Diethylenetriaminepentaacetic acid (DTPA) | 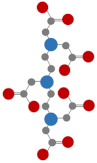 | Similar to EDTA [49] | 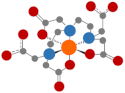 | 11 | |
| Low molecular weight organic acids (LMWOAs) | Citric acid (CA) | 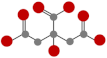 | 69% degraded after 20 d [50] |  | 8.96 |
| Oxalic acid (OA) | 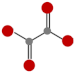 | Hard to degrade in nature due to the formation of Ca-oxalate [21] | 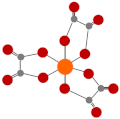 | 6.36 | |
| Succinic acid (SA) | 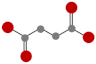 | Half-life < 2 min and 200 h in soil solution and for microbial mineralization, respectively [51] | 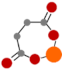 | 3.87 |
| Item | Reference | ||||||
|---|---|---|---|---|---|---|---|
| [85] | [72] | [86] | [26] | [27] | |||
| Soil characteristics | |||||||
| Texture | Silty loam | Loam | Sandy loam | Sand | Loamy sand | Heavy clay | Medium clay |
| OM (%) | 3.4 | 4.2 | 3.47 | 4.9 | 7.8 | 9.2 | 12.1 |
| pH | 6.8 | 7.3 | 5.58 | 6.9 | 7.2 | 6.7 | 6.67 |
| Total U (mg/kg) | 909 | 280 | 272 | 14 | 13 | 41 | 18 |
| Source of U contamination | Industrial | Industrial | U mine site | Industrial | Industrial | Uraniferous shale | UO2(NO3)3·6H2O |
| Experimental design | |||||||
| Chelator | CA and OA | CA, EDTA, and MA | CA, EDTA, and EDDS | CA, EDDS, OA, NTA, and NH4-citrate | CA and EDDS | ||
| Chelator dosage (mmol/kg) | 2, 10, and 20 | 0.5, 1, 5, 10, and 20 | 0.5, 2, 2.5, 5, 10, 15, 25, and 50 | 5 | 5, 5 + 5, 10, and 10 + 10 | ||
| Test conditions | Soil incubation | Soil incubation and pot experiment | Column leaching test | Soil incubation | Soil incubation | Soil incubation | Soil incubation and pot experiment |
| Result of most effective mobilization of soil U | |||||||
| Chelator and dosage (mmol/kg) | CA 20 | CA 20 | CA 50 | CA 5 | NH4-citrate 5 | NH4-citrate 5 | CA 10 + 10 |
| Days taken to reach the maximum U concentration in soil solution | 1 | 1 | 6 | 1 | 1 | 1 | 8 |
| Maximum U concentration in soil solution and increment compared to control | 775 mg/kg, increased by 140 times | 240 mg/L, increased by 200 times | 2000–2400 Bq/kg, increased by 356 times | 5019 µg/L, increased by 479 times | 1106 µg/L, increased by 368 times | 733 µg/L, increased by 366 times | 1463.6 µg/L, increased by 215 times |
| Chelator | Plant | Total U (mg/kg) | Source of U Contamination | Incubation Period | Dosage (mmol/kg) | Application Frequency | Result of Most Effective Mobilization of Soil U | Ref. |
|---|---|---|---|---|---|---|---|---|
| AA 1, CA, and MA | 10 species 6 | 280 and 750 | Industrial | 28 d of growth prior to chelator addition and harvested 7 d after addition | 20 | Single | CA was the most effective in enhancing U accumulation in plants. After 20 mmol/kg CA addition, shoot U concentrations in four plant species (B. juncea, B. chinensis, B. narinosa, and amaranth) increased by more than 1000 times compared to control within a few days. | [72] |
| CA, CDTA 2, DTPA, EDTA, HEDTA 3, and OA | Sunflower (Helianthus annuus L.) and Indian mustard (Brassica juncea L.) | 300 | Additional UO2(NO3)3·6H2O | 28 d of growth prior to chelator addition and harvested 42–56 d after addition | 1–25 | Single | CA was the most effective in desorption of U ions and enhancing plant accumulation of U. 20 mmol/kg CA addition to loamy acid soil resulted in the highest U concentration in sunflower shoots, being 150 times greater than control. | [89] |
| AC 4, CA, EDDS, NTA, and OA | Indian mustard (Brassica juncea cv. Vitasso) and ryegrass (Lolium perenne cv. Melvina) | 14 | Industrial U-contaminated soil (sand, labelled as BK1) | 28 d of growth prior to chelator addition and harvested 4–14 d after addition | 5 | Single | The Indian mustard shoot U concentration was in the following decreasing order: EDDS > AC > CA > OA > NTA in BK1; and CA > AC > EDDS > OA = NTA in BI. The ryegrass shoot U concentration was in the following decreasing order: CA > AC > OA > EDDS > NTA in BK1; and CA = AC > OA = EDDS > NTA in BI. | [21] |
| 41 | Natural U-enriched soil (heavy clay, labelled as BI) | |||||||
| AC and CA | Kochia (Kochia scoparia L. Schrad.), sunflower (Helianthus annuus L.), and sweet corn (Zea mays L.) | 78.7 | Depleted U-contaminated soil | 45 d of growth prior to chelator addition and harvested 6 d after addition | 20 | Single | AC was as effective in enhancing plant uptake of U as CA. The kochia leaves U concentration was in the following decreasing order: CA > AC, whereas that in the stems: AC > CA. The sunflower leaves U concentration was in the following decreasing order: AC > CA, whereas that in the stems: AC = CA. Corn had the lowest U uptake capacity and was considered as a negative control. | [101] |
| CA and MA | Indian mustard (Brassica juncea L.) | 100 | Additional UO2(NO3)3·6H2O | 66 d of growth prior to chelator addition and harvested 7 d after addition | 5, 10, and 20 | Single | A total of two restorations were carried out. In the first round, the maximum plant accumulation of U was noted in 10 mmol/kg CA treatment after 45 d and in 20 mmol/kg CA treatment after 55 d. In the second round, the maximum plant accumulation of U was noted in 20 mmol/kg MA treatment after both 55 and 65 d. | [102] |
| CA and mixture of CA, MA, OA, and LA 5 (labelled as Mix) | Mustard (Brassica juncea var. tumida) | 47.75 | Additional UO2(NO3)3·6H2O | 60 d of growth prior to chelator addition and harvested 7 d after addition | 5 for CA and molar ratio of CA: MA: OA: LA in Mix = 2.5:2.31:1.15:0.044 | Single | Mix was more effective in enhancing U accumulation in mustard. | [103] |
| CA, EDDS, and OA | Red wetter grass (Zebrina pendula Schnizl) | 150 | Additional UO2(CH3CO2)2·2H2O | 90 d of growth prior to chelator addition and harvested 7 d after the last addition | 2.5, 5, and 7.5 | Three consecutive applications every three days | CA was the most effective in enhancing U accumulation in Zebrina pendula Schnizl. Addition of 5 mmol/kg CA resulted in the highest U concentration in the plant, being 5.7 times greater than control. | [99] |
| CA, EDDS, and OA | Macleaya cordata | 18 | Additional UO2(NO3)3·6H2O | Chelator addition at the beginning of flowering and harvested 14 d after the last addition | 5 and 10 | Except for 10 mmol/kg EDDS, all other chelators were applied both in a single application and two consecutive applications. | The enhanced U uptake was in the following decreasing order: CA > EDDS > OA. Two consecutive applications of 10 mmol/kg CA resulted in the most significantly promoted solubilization of soil U, being 215 times greater than control. | [27] |
| CA, EDDS, and OA | Sunflower (Helianthus annuus L.) | 15 | Additional UO2(CH3CO2)2·2H2O | 60 d of growth prior to chelator addition and harvested 7 d after the last addition | 2.5, 5, and 7.5 | Three consecutive applications every three days | 5 mmol/kg CA addition resulted in the highest U phytoextraction efficiency, being 1.78 times greater than control. | [28] |
| Wall/Carrier Material | Core Material | Synthesis Method | Particle Size | Slow-Release Period | Contaminant | Dosage (mmol/kg) | Test Conditions | Ref. |
|---|---|---|---|---|---|---|---|---|
| Silicate | EDTA-Na2 | Spray drying | 3–5 mm | Release of EDTA from slow-release EDTA was still significantly lower than from uncoated solid EDTA in soil after 18 d. | Pb and Zn | 13 | Pot experiment | [105] |
| Talc, polyethylene, ethylene-vinyl acetate copolymer, ethylene-octene-1 copolymer, and polyoxyethylene monomethyl ether | EDTA-Na2 | Coating | - | Release of 75% EDTA ranged from 3 to 210 d in water | Pb | 4 | Pot experiment | [142] |
| Diethylenetriamine and hexamethylene diisocyanate | EDTA-Na4 | Interfacial polymerization | 5.78 μm in average | Release of 85% EDTA in water after 5 d | - | - | Pot experiment | [143] |
| Polycaprolactone | EDTA | Solvent evaporation | 65 ± 15 μm | Release of 93% EDTA in water after 30 d | Cu and Pb | 3 and 6 | Pot experiment | [131] |
| Chitosan and its derivatives | EDTA-Na2 | Spray drying | 5–20 μm | Release of EDTA from slow-release EDTA was still significantly lower than from non-microencapsulated EDTA in water after 3 d. | Cd, Cu, and Pb | 4 and 8 | Pot experiment | [159] |
| Hydroxypropyl chitosan-graft-carboxymethyl-β-cyclodextrin | CA | Spray drying | - | Cumulative release of 80% CA in water after 2 d | U | 5 | Pot experiment | [141] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
You, Y.; Dou, J.; Xue, Y.; Jin, N.; Yang, K. Chelating Agents in Assisting Phytoremediation of Uranium-Contaminated Soils: A Review. Sustainability 2022, 14, 6379. https://doi.org/10.3390/su14106379
You Y, Dou J, Xue Y, Jin N, Yang K. Chelating Agents in Assisting Phytoremediation of Uranium-Contaminated Soils: A Review. Sustainability. 2022; 14(10):6379. https://doi.org/10.3390/su14106379
Chicago/Turabian StyleYou, Yue, Junfeng Dou, Yu Xue, Naifu Jin, and Kai Yang. 2022. "Chelating Agents in Assisting Phytoremediation of Uranium-Contaminated Soils: A Review" Sustainability 14, no. 10: 6379. https://doi.org/10.3390/su14106379
APA StyleYou, Y., Dou, J., Xue, Y., Jin, N., & Yang, K. (2022). Chelating Agents in Assisting Phytoremediation of Uranium-Contaminated Soils: A Review. Sustainability, 14(10), 6379. https://doi.org/10.3390/su14106379








