Abstract
Microalgae have received widespread interest owing to their potential in biofuel production. However, economical microalgal biomass production is conditioned by enhancing the lipid accumulation without decreasing growth rate or by increasing both simultaneously. While extensive investigation has been performed on promoting the economic feasibility of microalgal-based biofuel production that aims to increase the productivity of microalgae species, only a handful of them deal with increasing lipid productivity (based on lipid contents and growth rate) in the feedstock production process. The purpose of this review is to provide an overview of the recent advances and novel approaches in promoting lipid productivity (depends on biomass and lipid contents) in feedstock production from strain selection to after-harvesting stages. The current study comprises two parts. In the first part, bilateral improving biomass/lipid production will be investigated in upstream measures, including strain selection, genetic engineering, and cultivation stages. In the second part, the enhancement of lipid productivity will be discussed in the downstream measure included in the harvesting and after-harvesting stages. An integrated approach involving the strategies for increasing lipid productivity in up- and down-stream measures can be a breakthrough approach that would promote the commercialization of market-driven microalgae-derived biofuel production.
1. Introduction
The age of inexpensive fossil fuels is ending. The rapid increase in the world’s population and the rising demand for energy are global challenges that have presented themselves over the past couple of centuries [1,2,3]. However, fossil fuels are not renewable, and their resources are depleting day by day [4,5,6]. Among the potential energy sources, microalgae-derived biofuels are considered a comparable alternative for fossil fuels [5,7]. Microalgae is a potential feedstock for a number of applications such as the production of animal feed, value-added products, and biofuels [8,9]. However, the possibility of an economically sustainable microalgae feedstock production process is technology-driven, not commercially driven [10,11,12].
The main economic drawback cited in the literature is that algae species display two conflicting features: high biomass production with low lipid accumulation, or high lipid accumulation with low biomass production [13,14,15]. Under favorable growth conditions, the oil content of microalgae species typically is between ~10 and 30% dry weights. Meanwhile, there are algae species that produce higher lipid content (56% in Nannochloris sp., 80% in Schizochytrium sp.). However, the growth rate of such oleaginous species is often slow [16,17]. Alternatively, algae species such as Scenedesmus sp. and Chlorella sp. with a higher growth rate relatively possess lower lipid content [11,18]. It is commonly known that, for obtaining the best economic scenario, an optimal balance between lipid content and cell growth is required; because culturing either many cells with low lipid content or few cells with high lipid content will not lead to economically sustainable microalgae-derived biofuel production [13,17,19].
To overcome this obstacle, wide-ranging efforts are being made to improve biomass and lipid production in upstream and downstream stages. The upstream measure involves screening appropriate microalgae strain and further improvement of those ‘platform algal species’ by genetic manipulation to develop new organisms with higher lipid productivity. Furthermore, it is well established that the implementation of an efficient cultivation system that can deal with the contrast between lipid and biomass production is important in enhancing lipid productivity. Establishing the strategies that provide the best performance in the down-stream stage such as harvesting and after-harvesting stages can also provide an additional approach for promoting the lipid productivity of algae strains [20].
Screening for local oleaginous microalgae “bio-prospecting” is the first step in the optimization of the feedstock production process. One of the most important criteria for screening algae strains is lipid productivity (based on lipid contents and growth rate) [21]. It is commonly known that selecting the fast-growing oleaginous algal species would translate directly to an overall feedstock production process [21]. In this regard, researchers have focused their efforts on the screening of microalgae strains with higher biomass and lipid productivities. For instance, in a study, four oleaginous microalgae were investigated for biofuel production. Two of them, Monoraphidium dybowskii Y2 and Chlorella sp. L1 were found to produce the highest lipid content (32.45, 35.06 mg L−1 day−1) and biomass yield (106.61, 137.13 mg L−1 day−1) when cultured in photobioreactor (PBR) [22]. In another screening program, Přibyl et al. [23] evaluated the potential of 10 strains of Parachlorella and Chlorella for lipid and biomass production; among them, the strains C. vulgaris CCALA 256, with a biomass density of 5.7 g L−l, and overall lipid content of 30% dry weight, was the most promising strain for biofuel production when cultured in a PBR. Bioprospecting requires high throughput isolating procedures to screen local microalgae strain for biofuel production. However, conventional approaches utilized for strain selection mostly rely on complicated procedures that are labor intensive and time consuming [21,24]. In this regard, Kim et al. [25] studied the novel advances in the development of microfluidic systems for microalgae biotechnology. Similarly, Challagulla et al. [26] reviewed the application of the nuclear magnetic resonance spectroscopy, fluorescent lipid-soluble dyes, Raman spectroscopy, near-infrared spectroscopy, and Fourier transform techniques for analyzing lipid contents in microalgae. It was stated that some technologies such as spectroscopy, flow cytometry and microfluidic can be used alone or in combination with other strategies for the screening of strains with a high growth rate and lipid contents [26,27,28,29].
Genetic engineering has previously presented a promising approach for research in different scientific areas [30,31,32,33]. As is the case in other research fields, genetic engineering provides an alternative approach to bypass the controversial relationships between lipid accumulation and cell growth [34]. A number of the molecular studies focus on lipid metabolism engineering by over-expression of the enzymes involved in lipid synthesis or suppressing the competitive pathways for enhancing lipid content without comprising the growth rate. For instance, in an investigation, after inactivation of the specific multiuse enzymes acyltransferase/lipase/phospholipase, a mutant strain of Thalassiosira pseudonana showed 3.5 times more lipid content without decreasing growth rate [35]. In another study, Nguyen et al. [36] reported that the suppression of the gene involved in fatty acid (FA) degradation of Chlamydomonas mutant, Cre01.g 000300, could increase lipid content without impacting the growth rate. Up to date, a number of review articles have been published on genetic engineering techniques for promoting the lipid productivity of algae species. For instance, Park at al. [37] surveyed the strain improving strategies such as genetic engineering, random mutagenesis, and metabolic engineering pathways to promote lipid productivity of microalgae; and stated the combination of the appropriate tools and right targets can improve algal species that would promote the commercialization of market- microalgae-derived biofuel production. In similar investigations, Chen et al. [14] proposed that the lipid production efficiency in microalgae can be increased by integrating stress tolerance manipulation strategies with genetic engineering approaches.
It is commonly known that microalgae cultivation is the most important stage in microalgae biofuel production because the quantity and quality of the produced feedstock will strongly depend on this stage [19]. An ideal cultivation strategy would enable algae strains to grow rapidly with a synchronized increase in lipid content. However, cultivation conditions for cell growth typically differ from that required for lipid production. Generally, the microalgae species cultured under favorable conditions produce large amounts of biomass but with lower lipid yield [9]. Thus, for increasing lipid content in algae, additional approaches, such as applying different stresses during the biomass production process have been proposed. But such stressful conditions often have a negative impact on the microalgae growth rate, leading to a decrease in the desired product yield. Thus, obtaining the best economic scenario will require an optimal balance of lipid content and cell growth [13,19]. In this sense, the two-phase system has been proposed as a win-win strategy to overcome the trade-off effect between biomass and lipid yield. In the two-step cultivation system, a nutrient-rich growth medium is used in the first step to obtaining maximal biomass production. After an adequate concentration of algal biomass is produced, the medium condition changes into a stress induction condition in the second step. In an investigation, Xia et al. [38] developed a salinity-based two-phase cultivation for Scenedesmus obtusus XJ-15. In the first phase, the biomass productivity was increased from 139.4 to 212.1 mg L−1 day−1. In the second phase, lipid content was increased from 26.1% to 47.7%. In another investigation, Yun et al. [39] proposed a two-phase system for N. oleoabundans HK-129. The process resulted in a 40 and 60% increase in algal biomass and lipid content, respectively. Additionally, a number of review articles have been done as part of an effort to study the efficiency of various cultivation strategies in microalgae. One such investigation by Ho et al. [19] studied the efficiency of various cultivation systems for increasing lipid productivity in microalgae and stated two-stage and semi-continuous strategies can increase lipid content without impacting the growth rate. Nagappan et al. [40] compared lipid and carbohydrate productivities of two-stage strategies with single-stage systems. Aziz et al. [41] highlighted the potential of a two-stage culture strategy for simultaneous lipid and biomass production and modified the pre-harvesting stage to promote the economic viability of this strategy. In one of such studies, Bhatia et al. [42] discussed different types of wastewater and summarized the recent approaches in algal cultivation and harvesting technologies from wastewater. Table 1 shows a list including studies about increases in lipid and/or growth rate in microalgae, brief findings of which are given presently.

Table 1.
Summary of literature about the enhancing lipid and/or biomass production in microalgae.
Among the considered approaches, establishing the strategies that provide the best performance in the harvesting stage without having qualitative damages to microalgae biomass and/or lipid content is also important in the successful implementation of the feedstock production process [51]. For instance, Liu et al. [52] applied magnetite nanoparticles (nano-Fe3O4 coated) for the harvesting of C. pyrenoidosa and S. obliquus strains, and reported that this harvesting method did not reduce the lipid content in these species. Very few studies have been performed on harvesting and after-harvesting stages, although these stages should be taken into consideration for improving lipid productivity in the feedstock production process [50,51]. In one of such studies, Menegazzo et al. [49] reviewed the culture conditions for various algae species and their influence on the separation of microalgae biomass, lipid content, including biomass thickening methods, and methods of biomass depletion, the methods of cellular disruption and lipid extraction.
The above literature review shows that most of the studies provide only a partial picture of the bilateral increase in lipid productivity throughout the cultivation or genetic engineering stages. Meanwhile, there are review articles that focused on more aspects of the biomass production process. Studies such as that by Chu [45] present an overview of strain selection, genetic engineering, and cultivation without depth information about different cultivation strategies. Chen et al. [14] focused on recent advancements in the lipid enhancement process in the cultivation and genetic engineering stages. However, these articles are not containing all relevant information about increasing lipid productivity throughout the feedstock production process, and only scattered information is accessible (Table 1). To the best of the authors’ knowledge, no current review paper contains the findings of promoting lipid productivity in strain selection, genetic manipulation, cultivation, harvesting, and after harvesting stages. The purpose of this review is to provide an overview of the recent advances and novel approaches in promoting lipid productivity in the biofuel production process from strain selection to after-harvesting stages.
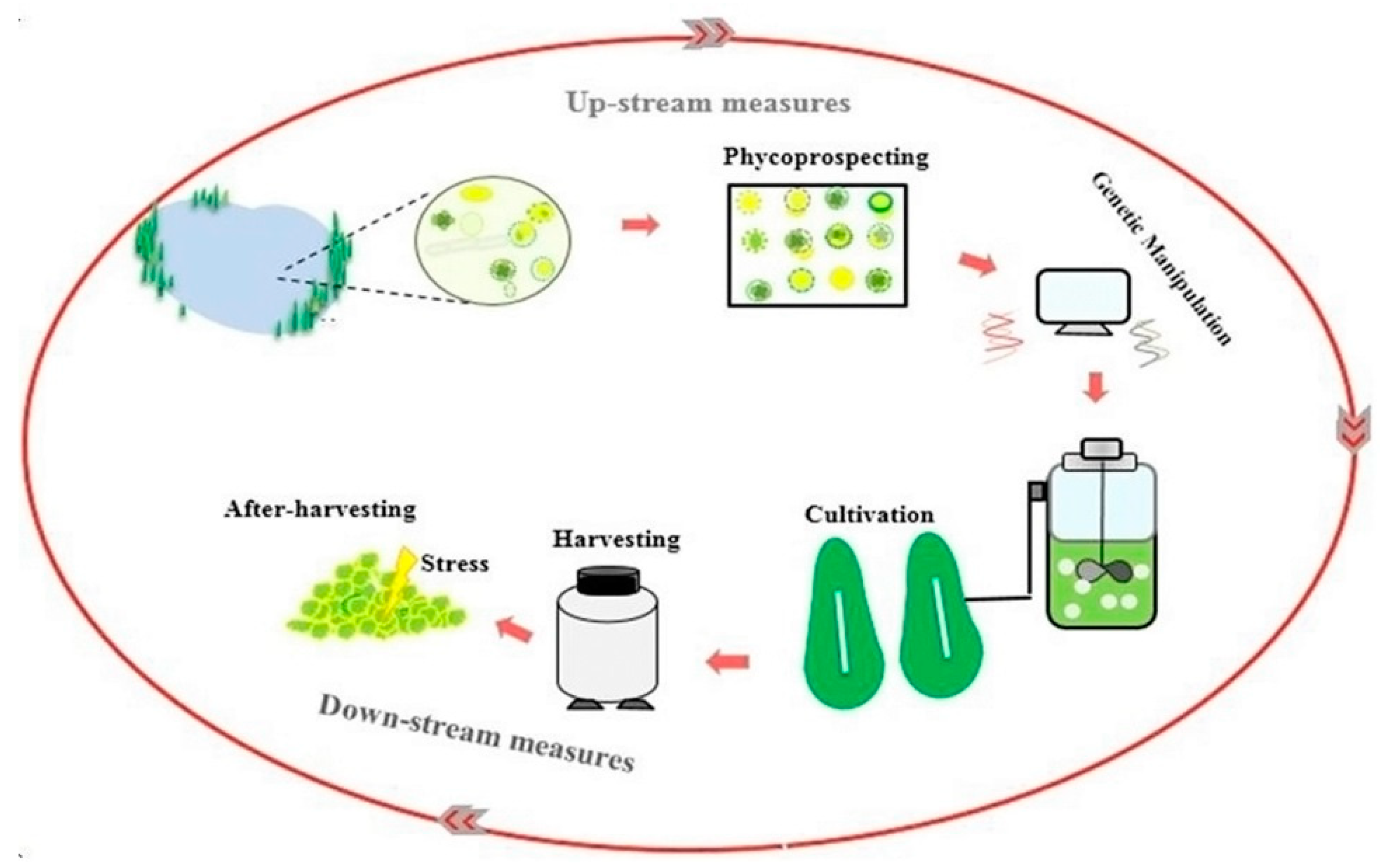
The first section of the article offers a discussion about the importance of bilateral biomass and lipid production in biofuel production and its potential in increasing microalgae economic feasibility of microalgae-derived biofuel production. In the following section, a bilateral increase in biomass/lipid production is investigated in upstream measures, including strain selection and genetic engineering stages. Finally, promoting lipid productivity will be discussed in the downstream measure included in the harvesting, and after-harvesting stages (Figure 1).
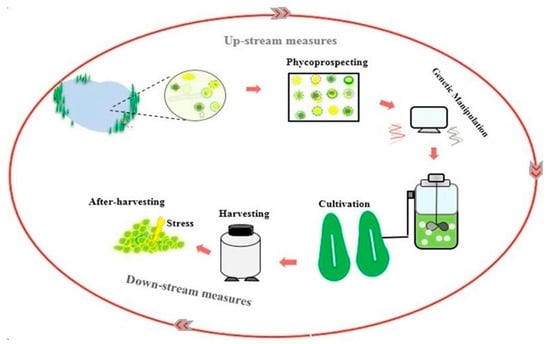
Figure 1.
Enhancing lipid productivity in the feedstock production process.
2. Bilateral Improvement in Biomass and Lipid Productivities
Microalgae are potential biofuel feedstock, owing to its rapid growth rate and ability to produce value-added lipids [16]. The lipid content of microalgae can be increased when the microalgae cells are subjected to stressful conditions such as culturing under a nutrient limitation or environmental stress [17]. However, such stresses often negatively affect algal growth, leading to decreased lipid productivity [47,53,54]. In an investigation, the effect of stressful conditions on biomass and lipid production of Chaetoceros muelleri was studied [55]. Under nitrogen-deficient conditions, the lipid content was elevated from 23% to 46%. However, on the other side, biomass productivity was decreased from 0.19 to 0.12 g L−1 day−1 [44]. Similarly, in another study, the impact of nutrient limitation on lipid and biomass yield of Scenedesmus destricola and Chorococcum spp. was investigated [56]. Under nitrogen-limited conditions, the lipid yield was increased from 48% to 54% for Scenedesmus destricola and 31.6% to 40.7% for Chlorococcum nivale. The biomass productivity of Scenedesmus destricola and Chlorococcum nivale, however, was decreased from 0.48 to 0.38 g L−1 day−1 and from 0.40 to 0.38 g L−1 day−1, respectively [44].
It is well established that increasing lipid content, without impacting the growth rate or increasing both, holds the key to obtaining economic viability of algae-based biofuel production [38]. The mathematical analyses of Yu et al. [57] showed how both biomass productivity and lipid contents are vital in determining the biofuel production cost. In their first analysis of Chlorella vulgaris CCAP 211/11B and Chlorella vulgaris F&M-M49 of similar lipid content and cell size, Chlorella vulgaris F&M-M49 incurred lower production costs because of its higher biomass yield. In other analyses of Nannochloropsis sp. and Nannochloropsis sp. F&M-M27 with similar biomass productivity and cell size, Nannochloropsis sp. F&M-M27 reduced the cost by 10–20%, owing to its 25% higher lipid content [47].
Conventional stress induction approaches seem to be useful for increasing algal lipid content but often fail to increase lipid productivity as the enhancing lipid content happens at the cost of biomass. Lipid productivity as expressed in Equation (1) usually reported in the unit of g lipid m−2 day−1 or g lipid L−1 day−1.
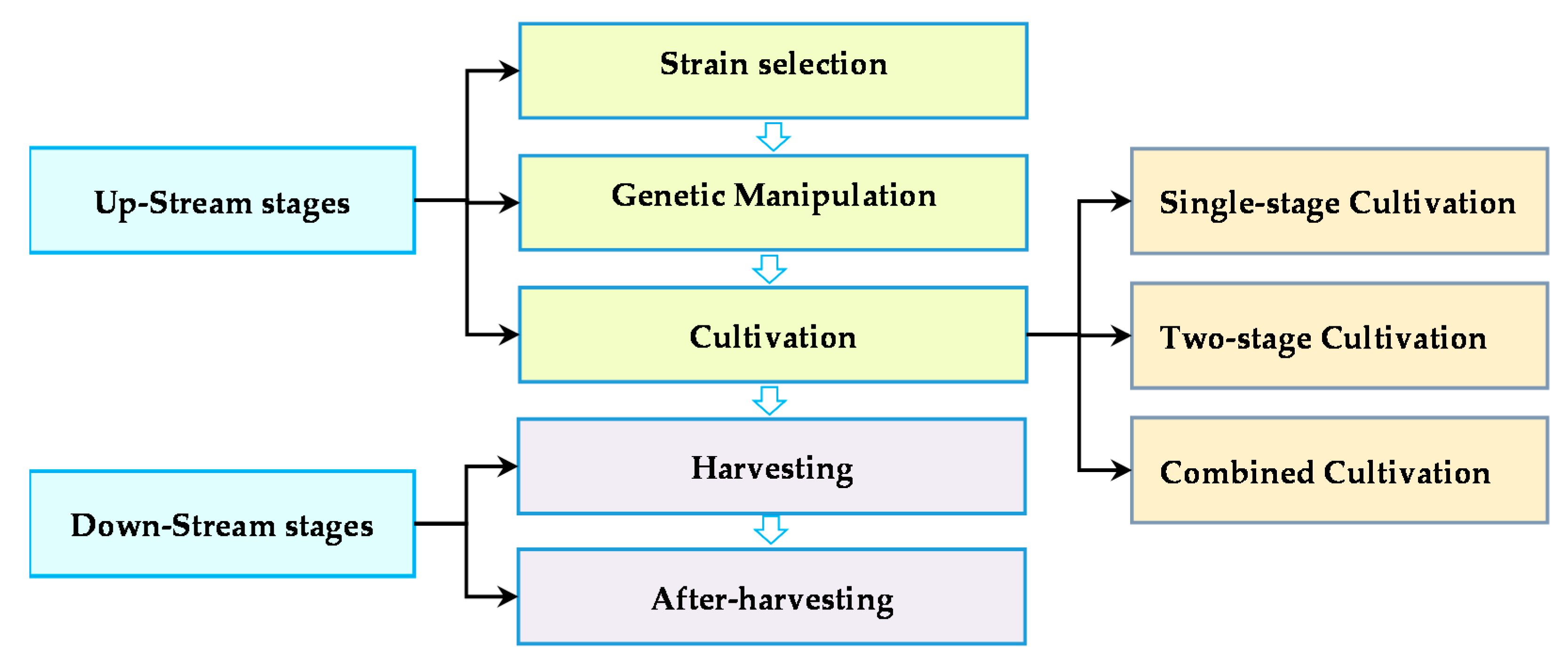
where Q is the amount of the microalgal yield per unit area of culture (m2) or volume (L) and is the specific growth rate (day−1) [37,43]. Therefore, microalgae-based biofuel production success is strongly influenced by two mutually exclusive factors, i.e., lipid content and growth rate. High lipid content is essential to decrease the processing costs per unit of biomass products, and a high growth rate is required to increase yield per unit culture area. Generally, lipid accumulation in algae typically is not concurrent with a fast growth rate, which is the potential conflict in the production process [58]. To achieve the economically feasible cost at a necessary production scale, it is reasonable to develop an optimal balance among upstream and downstream technologies to reduce overall process costs (Figure 2).
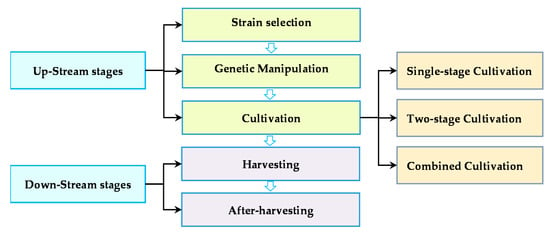
Figure 2.
Diagram of the microalgae feedstock production procedure.
Two trends have been proposed in biofuel-related algae studies for achieving high lipid productivity. The first trend focuses on enhancing lipid content without decreasing biomass yield, which is often achieved by genetic manipulation approaches. For instance, the co-expression of five acyltransferases from yeast in Chlorella minutissima, elevated lipid yield by two times without decreasing the growth rate [59]. The other trend has the aim of increasing biomass and lipid yields simultaneously that generally can be attained by applying a suitable cultivation strategy. For instance, Moussa et al. [60] designed a hybrid cultivation system for Picochlorum sp., in which maximal biomass productivity (0.427 g L−1 day−1) was obtained by the continuous culture at a dilution rate of 0.6 day−1, and high lipid contents, ranging from 499 to 698 g kg−1 dry weight (DW), were obtained under different nitrogen sources in the second step. In another study, San Pedro et al. [61] developed a two-stage cultivation method for Scenedesmus sp. In this cultivation method, microalgal cells were maximal biomass productivity of 0.49 g L−1 day−1 was attained with a nitrate concentration of 8.0 mM at a dilution rate of 0.42 1/day. A high increase in lipids content, 73.1% of overall lipids, was obtained in the second phase under nitrogen-depletion condition.
3. Upstream Measure
Oleaginous microalgae are potential cell factories for viable biodiesel production. It possesses the inherent ability to accumulate value-added products such as lipids and exhibits a high growth rate [20]. Commercially sustainable microalgae-based biofuel production requires screening local oleaginous species, which can be further promoted by strain engineering strategies [37,62]. Therefore, the upstream technologies mainly include three aspects. Firstly, screening appropriate algal species that are characterized by rich in lipid and fast-growing rate. Secondly, advanced molecular approaches can be manipulated so that produce the strains with high biomass and lipid productivity. Finally, selecting and establishing an efficient cultivation system that can deal with the contrast between lipid and biomass production.
3.1. Phycoprospecting
As estimated, there are 1 to 10,000,000 alga species on Earth; however, most of the on-going studies have been focused on a limited number of microalgae strains. Microalgae are characterized based on high lipid content and fast growth rate. But all of the algae species are not regarded as the best lipid producers [20,25,63]. Although documented in different studies about increasing the microalgal lipid productivity via various approaches, the potential limitation cannot be overcome if the screened algal species are not apt for biodiesel production [17,47]. Identifying best performing microalgae strains through ‘phycoprospecting’ can be a promising approach to obtain strains with higher lipid productivity for achieving commercially sustainable biofuel production [47,64].
In this respect, the fundamental requirement of microalgae-based biodiesel production is selecting appropriate strains with a high level of lipid accumulation and high biomass productivity [65]. Additionally, the optimal microalgae strains for biofuel production should have a number of important features: an optimal composition of lipid profiles; high cell density during cultivation; high carbon dioxide absorption rate; lack of need for expensive nutrients during cultivation process; resistance to temperature fluctuations; co-production of valuable by-products and short production cycle [17].
Besides, local algal species are more preferred for feedstock production purposes as they have better adaptability to environmental situations prevailing in a specific geographical location [15]. Several programs have focused on the isolation of the appropriate indigenous algal strains to enhance lipid and biomass productivities. Screening of indigenous microalgae strains for desirable characteristics or ‘phycoprospecting’ is important in determining potential strains for biodiesel production [65,66]. For example, an isolation project under the National Alliance for Advanced Biofuels and Bioproducts (NAABB) has successfully screened novel potential ‘platform strains’ with fast growth rates and high lipid content [66].
Bio-prospecting requires high throughput and rapid isolating procedures to screen new strains that are adapted to the production location [65]. Several techniques have been described, including single-cell isolation using serial dilutions, micromanipulation, atomized cell spray, and gravimetric separation [67,68]. However, such traditional microalgae isolation methods are time consuming and require further screening of axenic cultures to determine lipid and growth productivities [27,69,70]. Technologies such as automated processes including robotics, flowcytometry, and new strategies such as microfluidics and deep sequencing are being developed to facilitate the isolation and characterization of microalgae strains [70]. A combination of modern and conventional techniques may screen the suitable algal strain for biofuel production. In a study, flowcytometry technique coupled to cell sorting strategies has led to a rapid selection of strains with high lipid contents and fast growth rates [69,70]. In a study, Huang et al. [65] innovated a novel direct sampling technique that an enrichment strategy was coupled with a capillary aided sampling procedure. This approach sped up the isolation of desirable strains for both rapid growth and high lipid productivities. Kim et al. [71] innovated a droplet microfluidics analysis platform that is possible to study the differences in lipid and biomass productivities of Chlamydomonas reinhardtii under different nitrogen conditions. Table 2 highlights the techniques that have been applied for screening microalgae with lipid content/growth rate.

Table 2.
Some techniques have been applied for screening microalgae with lipid content/growth rate.
3.2. Molecular Approaches
Microalgae combine biotechnological properties of microbial cells (the ability to accumulate metabolites and fast growth) with typical characteristics pertaining to higher plants (simplicity of nutritional requirements and efficient oxygenic photosynthesis). This specific combination establishes the basis of biotechnological approaches for increasing lipid productivity in microalgae [72]. Oleaginous microalgae have attracted significant interest in the production of biofuel owing to its capacity in producing large amounts of lipid and fast growth rate [73]. However, lipid accumulation in microalgae is not conducive to a high growth rate, which is the fundamental limitation in the feedstock production process. To overcome such a drawback, genetic engineering provides an alternative approach to bypass the controversial relationships between lipid accumulation and growth rate [74]. It was reported that genetic engineering strategies that increase lipid accumulation without compromising growth rate could reduce production cost and fortify the economic sustainability of algae-based biofuels production [75].
So far, most of the molecular investigations focus on lipid metabolism engineering by over-expression of the enzymes involved in lipid synthesis or suppressing the competitive pathways in lipid or biomass production [14]. Meanwhile, genetic engineering of the genes involved in stress tolerance mechanisms exhibits a significant potential in improving lipid productivity [76]. The integration of these strategies may provide a potential approach for sustainable microalgae-based biofuel production at a competitive cost.
3.2.1. Lipid Biosynthesis Pathway
Triacylglycerol and FA synthesis included a series of biochemical reactions medicated by different enzymes. These enzymes’ over-expression would lead to enhancing the enzyme activity and thereby effectively triggers lipid accumulation [77]. Acetyl-CoA carboxylase (ACCase) is one of the most exploited enzymes for enhancing lipid accumulation in microalgae. As described by previous reports, the overexpression of ACCase had less effect on lipid production [45]. However, the synchronized overexpression of malic enzyme (ME) with a subunit of ACCase (accD) was successful in increasing the lipid productivity of Dunaliella salina [78]. Similarly, the overexpression of malic enzyme is reported to enhance the lipid production of Phaeodactylum tricornutum by 2.5 folds without a negative impact on the growth rate [79].
One of the advances in this area was obtained by the co-expression of five acyltransferases from yeast in Chlorella minutissima, which elevated lipid production by 2 times without compromising the growth rate [59]. In addition, the overexpression of several other enzymes, such as acetyl-CoA synthase, phosphoenolpyruvate carboxylase, pyruvate dehydrogenase, glycerol kinase, and NAD (H) kinase has been reported to increase lipid accumulation without compromising the growth rate. A summary of the molecular studies employed for increasing lipid content without sacrificing biomass production is listed in Table 3.

Table 3.
Overviews of molecular approaches employed for overexpression/suppression lipid content without comprising growth rate.
Among the considered strategies, suppressing competitive pathways such as lipid and carbohydrate catabolism is another approach for improving lipid productivity [85]. Carbohydrate metabolism is the most important pathway in microalgae for carbon storage and starch production. Knocking down the starch metabolism pathway may result in the carbon flow towards lipid synthesis [86]. However, the inhibition of the genes involved in starch biosynthesis may result in a reduced growth rate, consequently decreasing lipid productivity [87,88].
The suppression of lipid catabolism, particularly the enzymes that catalyze the FA release, is another promising approach for increasing algal lipid productivity [35,88,89]. Lipid catabolism promotes membrane reconstruction by providing acyl groups, which is necessary for membrane reorganization of the photosynthetic system in microalgae [36,90]. However, some studies have indicated that, unlike the suppression of carbohydrate catabolism pathways, the inhibition of genes involved in lipid catabolism may have less influence on growth rate [35,89]. Therefore, enzymes involved in algal lipid catabolism have been considered as a potential alternative for a simultaneous increase in biomass and lipid yield. In an investigation, a mutant strain of Thalassiosira pseudonana showed 3.5 times more lipid synthesis after inactivation of the specific multiuse enzymes acyltransferase/lipase/phospholipase [35]. In another study, Nguyen et al. [36] reported that the suppression of the gene involved in FA degradation of Chlamydomonas mutant, Cre01.g 000300, could increase lipid content without impacting the growth rate.
3.2.2. Molecular Approaches for Modulation the Stress-Related Mechanisms
Lipid biosynthesis pathway in algae is a multi-step reaction, catalyzed by an enzyme complex [63]. Under optimal conditions, algae cell growth is endorsed by increased transcription and translation processes, which led to high biomass productivity [44]. However, under nutrient starvation conditions the microalgae growth is constrained, as most of the anabolic machinery is retarded. Therefore, identification of the behavior and mechanism of these enzymes under various environmental conditions is important in improving the lipid productivity of microalgae strains [14]. Wan et al. [88] studied the impacts of iron concentrations on the lipid yield of Chlorella sorokiniana. The expression of acc1, accD and rbcL genes are up-regulated at higher concentrations of iron, leading to greater lipid yield, without negatively affecting the growth rate. Similarly, Fan et al. [82] showed that the overexpression of the AtNADK3 of Chlorella pyrenoidosa considerably increased the lipid content with no adverse impact on the growth rate.
Additionally, manipulation of stress-responsive promoters is considered a potential approach to increase lipid production without negative impacts on growth rate [14]. For example, overexpression of the diacylglycerol acyltransferases gene controlled by the phosphorus limitation-inducible promoter has promoted lipid production by 2.5 folds in an engineered strain of Chlamydomonas reinhardtii, when compared to the control group [91]. Further heterologous expression of this construct in Nannochloropsis sp. NIES-2145 under phosphorus limitation also resulted in higher lipid yield (1.7 times more than the wild strain) [92].
Key transcription factors (TFs), as well as the enzymes and promoters, can be targeted for genetic engineering to achieve high lipid productivity [14]. Overexpression of the TFs involved in the regulation of lipid biosynthesis pathways can divert the metabolic flux toward lipids accumulation. Therefore, identification of TF-encoding genes and their subsequent manipulation in their hosts would be an efficient genetic approach for developing the robust microalgae strains [14]. Successful reports have emerged to verify the important roles of different TFs in enhancing the lipid yield without decreasing the growth rate (Table 4). For instance, TF GmDof4 expression from Glycine max in C. ellipsoidea contributed to enhancing the lipid accumulation from 46% to 53% with no negative impacts on growth rate [93]. In other investigations, the manipulation of CHT7 [94] and PSR1 [95] elevated lipid production in C. reinhardtii without comprising the growth rate. Similarly, Ajjawi et al. [96] studied the transcriptional profiling of N. gaditana under nutrient limitation conditions. They identified 20 efficient TFs for lipid accumulation in N. gaditana, and performed insertional knockout on 18 of these 20 TFs by CRISPR/Cas9 reverse-genetics pipeline.

Table 4.
Some genetic studies are employed for transcription factors (TFs) in different microalgae species.
3.3. Cultivation Stage
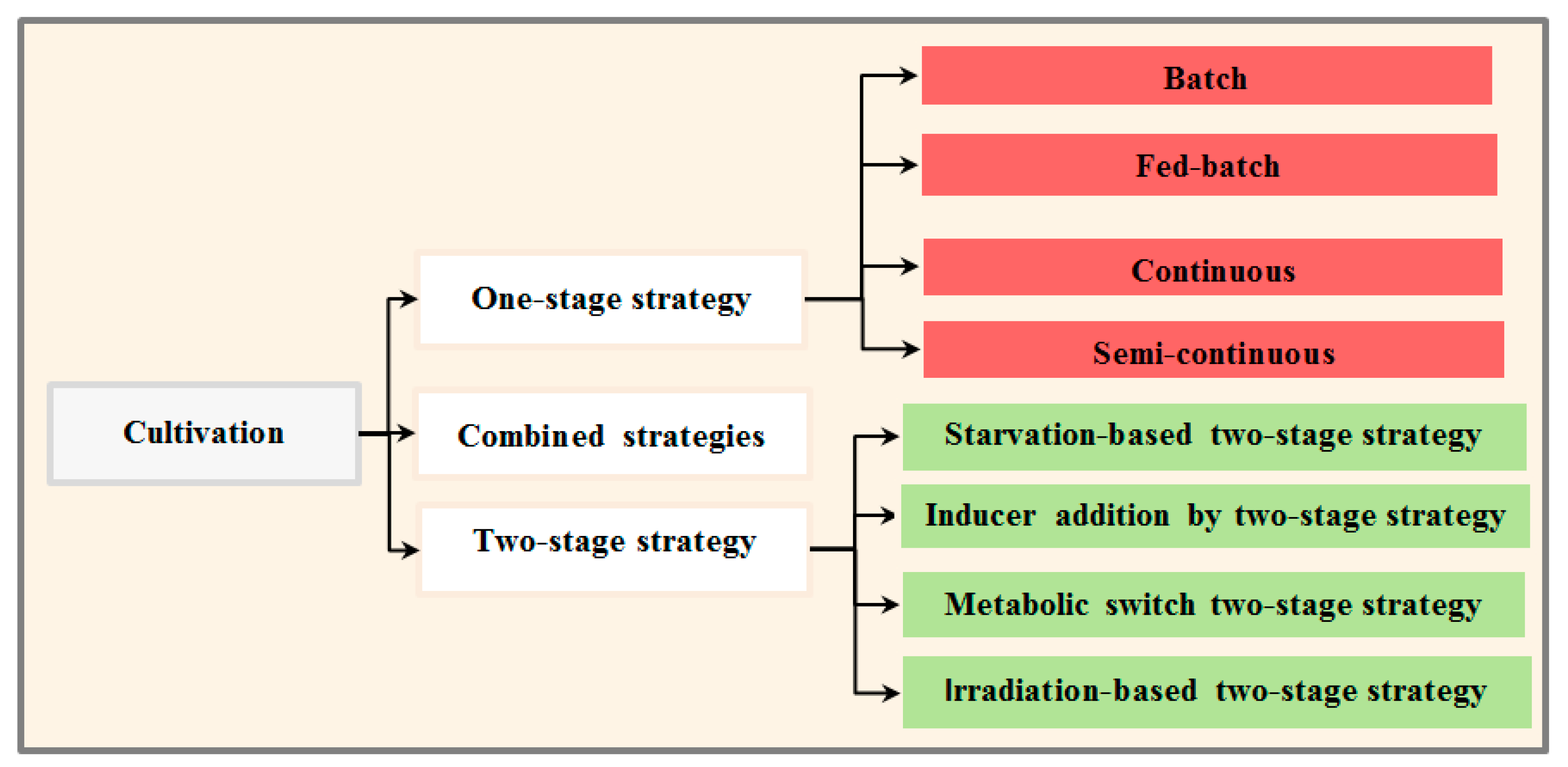
Although biomass/lipid production in microalgae is species-specific, the type of selected cultivation system is important in determining whether a feedstock production system will be economically feasible for biofuel production [44]. An ideal cultivation strategy would enable algae strains to grow rapidly with a synchronized increase in lipid content [19]. Cultivation strategies are categorized to single-stage strategies (e.g., semi-continuous, fed-batch, and continuous), integrated strategies and two-stage cultivation systems (Figure 3). [17,19,102]. These cultivation strategies, with emphasis on enhancing simultaneous biomass and lipid production, are discussed in the next sections.
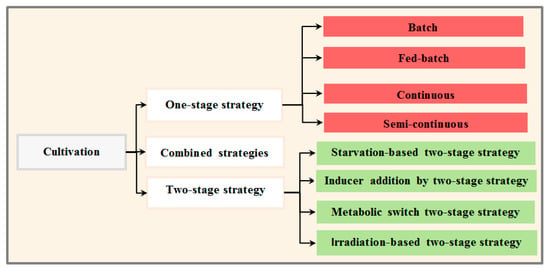
Figure 3.
Diagram of the different cultivation systems.
3.3.1. One-Stage Cultivation Strategy
Single-stage cultivation systems are categorized into five main groups: batch, continuous, fed-batch, and semi-continuous. As mentioned above, cultivation systems strongly influence the optimization of algal biomass and lipid production [102]. A comparison of the characteristics of various single-stage strategies indicates that the high lipid productivity can be achieved readily using semi-continuous strategy due to the strong stressful conditions associated with this cultivation system [19].
In contrast to the semi-continuous strategy, fed-batch and continuous cultivation systems are applied when the required nutrient level is available from the beginning of the cultivation process; in such cases, the medium gradually reaches a point of nutrient deprivation, which lead to lower lipid yield [79]. Generally, continuous systems tend to provide negligible lipid content, leading to their dismissal as sustainable platforms for microalgae-based biodiesel production [103]. However, some investigations have shown the possibility of using these cultivation strategies to improve microalgae biomass and lipid productivities through precision nutrient limitation. For instance, simultaneous lipid and biomass production was obtained in continuous chemostat culture when continuous feeding of BG11 media was supplemented with lipid inducers such as sodium chloride and sodium acetate [103]. Accordingly, Wen et al. [104] achieved high biomass and lipid productivity in continuous chemostat mode when the specific nitrate input rate was in the range of 0.78–4.56 mmol g−1 day−1. In other investigations, Del Rio et al. [105] cultivated Pseudokirchneriella subcapitata in continuous chemostat culture. The highest FA productivity was reported in nitrate concentration ranging from 3 to 5 mM. Some examples of such studies are outlined in Table 5.

Table 5.
Lipid/biomass productivities in different algae strain cultivated under a single-stage cultivation strategy.
3.3.2. Two-Stage Strategy
In microalgae, the cultivation conditions for lipid accumulation are different from those needed for biomass production. Thus, how to optimize cultivation conditions to achieve high lipid and biomass and production, is a big challenge in feedstock production [41,109]. In this sense, the two-phase system has been proposed as a win-win strategy to overcome the trade-off effect between biomass and lipid yield [20]. In the two-step cultivation system, a nutrient-rich growth medium is used in the first step to obtaining maximal biomass production. After an adequate concentration of algal biomass is produced, the medium condition changes into a stress induction condition in the second step [19,110]. It was reported, in the two-step system, that the average increase in biomass production is higher than in non-hybrid strategies, 12.5% more than photobioreactors, and 46–74% more than open ponds [41].
Based on various stimuli, two-stage cultivation strategies are classified into five groups: inducer addition by two-stage strategy, starvation-based two-stage strategy, metabolic switch by two-stage strategy, and irradiation-based two-stage strategy [40,41]. The next section of this article discusses various kinds of two-stage cultivation systems based on different stresses are described.
Nutrient Starvation
Nutrient starvation is demonstrated as an efficient approach to enhance lipid accumulation [111,112]. Of many various nutrient starvation strategies, nitrogen deprivation is one of the reliable methods to enhance lipid production in microalgae [16,113]. However, under nutrient depletion conditions, the growth rate is considerably decreased, resulting in lower lipid productivity. Accordingly, the two-stage strategy is proposed to address this issue, in which lipid and biomass production are split into separate steps [114,115]. In nutrient limitation strategy, the duration ratio between nitrogen-replete and deplete phases as well as the initial cell density in the second stage are crucial in optimization algal lipid and biomass production [11,116].
In terms of diatoms, silica deprivation is a preferred stress strategy for enhancing lipid accumulation [109]. The silica-limitation effect is more severe and rapid than nitrogen deficiency in this taxonomy. It was reported that silica deprivation can provide a controllable approach to enhance lipid biosynthesis in the two-step strategy. As an example, silica deprivation enhances the lipid content in diatoms of Cyclotella cryptica, Navicula saprophila and Chaetoceros muelleri up to almost 104%, 110%, and 89%, respectively [16,117].
Due to the efficiency of both nutrient limitation methods, a novel two-step model with nitrogen (N)-silicon(Si)-starvation has been suggested. In this strategy, the co-limitation of silicon and nitrogen not only improved lipid accumulation but also increased biodiesel quality of Skeletonema costatum [118].
Other than nitrogen and silicon, phosphorus is also vital for microalgal cell growth. In a number of cases, phosphate deprivation was reported to be more efficient than nitrogen starvation, such as the case in the two-step strategy of Ankistrodesmus falcatus where lipid productivity was higher than that in the nitrogen limitation method [119,120,121]. Additionally, reports on the effects of phosphate starvation on algal lipid productivity, based on the experimental setup, seem to be strain dependent. For instance, the lipid accumulation of Phaeodactylum tricornotum, Pavlova lutheri, Chaetoceros, and Dunaliella salina enhanced under phosphorus starvation, but, conversely, under such a condition, lipid content in Tetraselmis, Chlorella, and Nannochloris atomis was decreased. It is indicated that not all algae strains enhance their lipid accumulation during phosphate starvation conditions [55,120,122].
Inducer Addition
Although different inducers such as high salinity, low salinity, halo-alkalinity, and phytohormones could have a positive influence on algal lipid production [123,124], the changes in the culture medium are often associated with decreased cell growth in exchange for lipid production [125]. Therefore, integrating different inducers with a two-stage strategy can be an efficient strategy in optimization algal biomass and lipid productivities. For instance, applying salinity stress in the second phase of the two-step strategy increased the lipid content of I. galbana from 24% to 47% [126]. Similarly, inducing salinity stress in the two-step system was found to increase the lipid yield of Monoraphidium dybowskii LB50 [127]. It was reported that the salinity-based two-step cultivation strategy not only increases lipid productivity but also promotes the biodiesel quality obtained from the microalgae [40]. For instance, the biodiesel properties of saponification value, cetane number, long-chain saturation factor, and iodine value are considerably promoted in the salinity-based two-step strategy of Scenedesmus obtusus XJ-15 compared to one-step strategies [40,128].
A combination of high alkaline salt and pH (halo-alkalinity) can be readily adopted in this cultivation strategy. It was reported that the addition of hydrochloric acid and sodium bicarbonate can control pH in the culture medium in certain types of microalgae [40]. Wensel et al. [129] designed a halo-alkalinity-based two-step strategy in Chlorella pyrenoidosa and achieved a high lipid and biomass yield as well as high autoflocculation harvesting efficiency of 64.1%.
As mentioned above, microalgae lipid productivity can be improved by the induction of stressful conditions. However, such stresses often negatively affect the photosynthetic activity and, consequently, decrease the production of desired products [16]. The major reason for the decreased photosynthesis activity is the production of reactive oxygen species (ROS), which impairs the photosynthetic systems [130]. To solve this problem, a number of approaches have been suggested to reduce the oxidative stress caused by stressful conditions and hence increase lipid and biomass production [131,132].
Recent studies on the bicarbonate application in the cultivation process show that not only is it considered as a carbon source, but also act as an oxidative stress mitigator [86]. Under nutrient depletion conditions, the addition of sodium bicarbonate considerably decreased the oxidative stress caused by ROS and promoted the activities of antioxidant enzymes of Dunaliella salina, resulting in increased lipid and biomass production [130]. In an investigation, manipulating the iron, nitrate, and carbonate was reported to increase lipid and biomass production of three microalgae Chlorella sp., Scenedesmus sp., and Chlamydomonas sp. [133]
Apart from bicarbonate addition, phytohormones also play an important role in metabolism regulation and growth of microalgae [86,131]. Phytohormone could increase the antioxidant system in the microalgae, which keeps a redox balance state under stressful conditions. The combination of plant hormones with a two-stage culture system may be efficient in increasing the higher biomass and lipid production in microalgae [131,134]. For instance, the integration of a two-stage strategy (heterotrophic-photoautotrophic) with fulvic acid results in an increase of 54–65% in the lipid content of Monoraphidium sp. [135]. Table 6 shows some examples of inducer-based two-stage cultivation.

Table 6.
Lipid/biomass concentration of growth mediums in different strains of microalgae supplemented with various types of inducers.
Metabolic Switch
Microalgae can adopt different trophic modes, including photoheterotrophic, heterotrophic, and mixotrophic, based on energy and the available carbon source [40]. Algal biomass is generally produced through a photoautotrophic culture where microalgae can convert water and carbon dioxide into feedstock through the photosynthesis process [141]. Among all the required cultivation conditions, adequate light is recognized as a crucial factor for photoautotrophic conditions. Generally, insufficient light penetration, resulting from mutual shading, is the main limiting factor for microalgae cultivation under an autotrophic mode [142]. Thus, transferring the microalgae growth culture into the second phase under mixotrophic or heterotrophic conditions can reduce the culture’s need for sunlight [17,143].
Microalgae cells are typically grown in a photoautotrophic mode in the first phase, and then transfer the cultured algal biomass into a heterotrophic reactor where algal cells use organic carbon to synthesize oil [144]. Additionally, integration of a two-step strategy with wastewaters, as a nutrient source, can considerably decrease feedstock production costs. Wastewater containing organic compounds generally can be applied as the nutritional sources for heterotrophic or mixotrophic culture media, while wastewaters without organic carbon can be used for phototrophic cultivation mode [145,146,147].
Up to date, various cultivation modes have been integrated with the treatment of different wastewater types for the production of the microalgae feedstock [142]. Pure simple strategies in phototrophic and heterotrophic, mixotrophic modes, and combinations of phototrophic, heterotrophic, and mixotrophic cultivations on microalgae growth have been studied in several studies (Table 7). These achievements demonstrated the notion of a two-stage strategy as a desirable strategy for obtaining high lipid and biomass concentrations. Xiong et al. [148] compared the efficiency of single-stage heterotrophic mode with photosynthesis–fermentation model on C. protothecoides. Photosynthesis–fermentation strategy presented a better performance in terms of lipid productivity (69% higher lipid content) than the one-step heterotrophic mode. Zhou et al. [149] combined a two-step mix-photoautotrophic cultivation strategy with wastewater treatment to produce animal feed and biofuel production. In a similar investigation, a hetero-photoautotrophic microalgal growth mode was investigated for improving wastewater treatment and the production of low-cost algal biofuel feedstock from Auxenochlorella protothecoides [150].

Table 7.
Comparison of the achieved lipid/biomass productivities of different wastewaters as growth media for cultivating various algal species.
Similarly, Liu et al. [151] studied the effects of cultivation strategies including heterotrophic + mixotrophic strategy, heterotrophic strategy, autotrophic cultivation, and heterotrophic + autotrophic strategy on the lipid /biomass production of Chlorella sp. HQ. The results showed that the heterotrophic + mixotrophic two-phase culture system was the best strategy for improving the microalgal biomass and heterotrophic cultivation was the best strategy for microalgae lipid accumulation of Chlorella sp. HQ. Although several studies are focusing on these aspects, an effective microalgae-based system that does not compromise the growth or lipid content is yet to be found [152]. Some of the obtained lipid/biomass productivity levels of various wastewaters as growth media are presented in Table 7.
Irradiation
Manipulation of irradiation-based stimuli, in terms of intensity and wavelength, has been recognized as a potential approach for enhancing lipid content in various kinds of cultivation strategies [61,126]. He et al. [22] found that fluctuating the light intensities (990 to 1486 μ mol photons m−2 s−1) increased the lipids yield of six microalgae strains. In another study, increasing the light intensity from 300 to 500 μ mol m−2 s−1 considerably increased the lipid productivity of Nannochloropsis oculata. In this study, the lipid productivity value obtained in the two-step strategy was nearly three times more than the one-step strategy [137].
Apart from light intensity, light frequency is also important in photosynthesis; it can influence algal biomass productivity [45]. Microalgae have a light-harvesting antenna, which primarily absorbs light wavelengths in the visible spectrum [158]. It has been shown that the coupling of red light-emitting diode (LED) and 10–30% blue LED provides the proper light frequency for feedstock production. In contrast, green wavelengths cannot be absorbed by microalgae cells but may have a positive impact on increasing lipid accumulation in microalgae [126,159,160]. Therefore, integrating different wavelengths of lights, with a two-stage strategy can be an efficient strategy in optimization algal biomass and lipid productivities. So that, in the first stage, the microalgae were cultivated under red and/or blue LEDs to achieve maximal biomass productivity. Then, in the second stage, a green LED (520 nm) stress was stimulated to increase lipids accumulation [126]. In an investigation, LEDs were applied to improved cell growth and lipid production of Picochlorum atomus by a two-step strategy. The results indicate that biomass productivity under red LED light was higher than that produced by yellow, blue, and purple LEDs in the first phase. The highest lipid production was achieved (50.3%) with green LED light in the second stage [161].
3.3.3. Combined Cultivation Strategies
Although microalgae can produce higher lipids/biomass productivity compared with terrestrial plants, commercialization of the microalgae-derived biofuels is hampered by the production of the high cell density culture with high lipid content [162]. Generally, each of the cultivation strategies has its own merits in terms of the biomass/lipid yield. It might be favorable to integrate two or more cultivation systems to obtain higher lipid/biomass productivity. Notably, whether the combined or single cultivation systems are applied; it is vital to ensure that microalgae-based-biofuel production with the proposed strategies is more feasible than the conventional culture strategy. For instance, the combination of a two-stage system with a fed-batch strategy led to higher lipid productivity in comparison with a fed-batch system [163]. Similarly, the integration of a semi-continuous system with a two-stage strategy could also offer better efficiency in terms of increasing lipid productivity in Neochloris oleoabundans [164].
In addition, the analysis of the biological characteristic of various algae species and optimization of cultivation strategies are of paramount importance in improving lipid and biomass productivity. For instance, Nayak et al. [165] optimized a continuous two-step strategy of Chlorella sp. HS2, with supplementation of additional phosphorus at the start of nitrogen starvation in the second phase. In other investigations, Ghidossi et al. [166] proposed an efficient two-stage fed-batch cultivation strategy based on the carbon to nitrogen mass ratio (C/N) the culture medium. In the first phase of this strategy, high cell concentrations were used under carbon starvation (lower C/N ratios). In the second phase, high lipid content was obtained under nitrogen depletion conditions (higher C/N ratios). In this study, lipid productivity attained 2- to 5-fold increase compared to other studies [166,167,168].
4. Downstream Measure
It is documented that determining the strategies that provide the best performance in harvesting, and after-harvesting stages are of paramount importance in achieving the commercially feasible feedstock production process [20]. The downstream technologies generally include the best harvesting and after-harvesting methods without having qualitative damages to algal biomass and/or lipid content [20,50,51].
4.1. Harvesting Stage
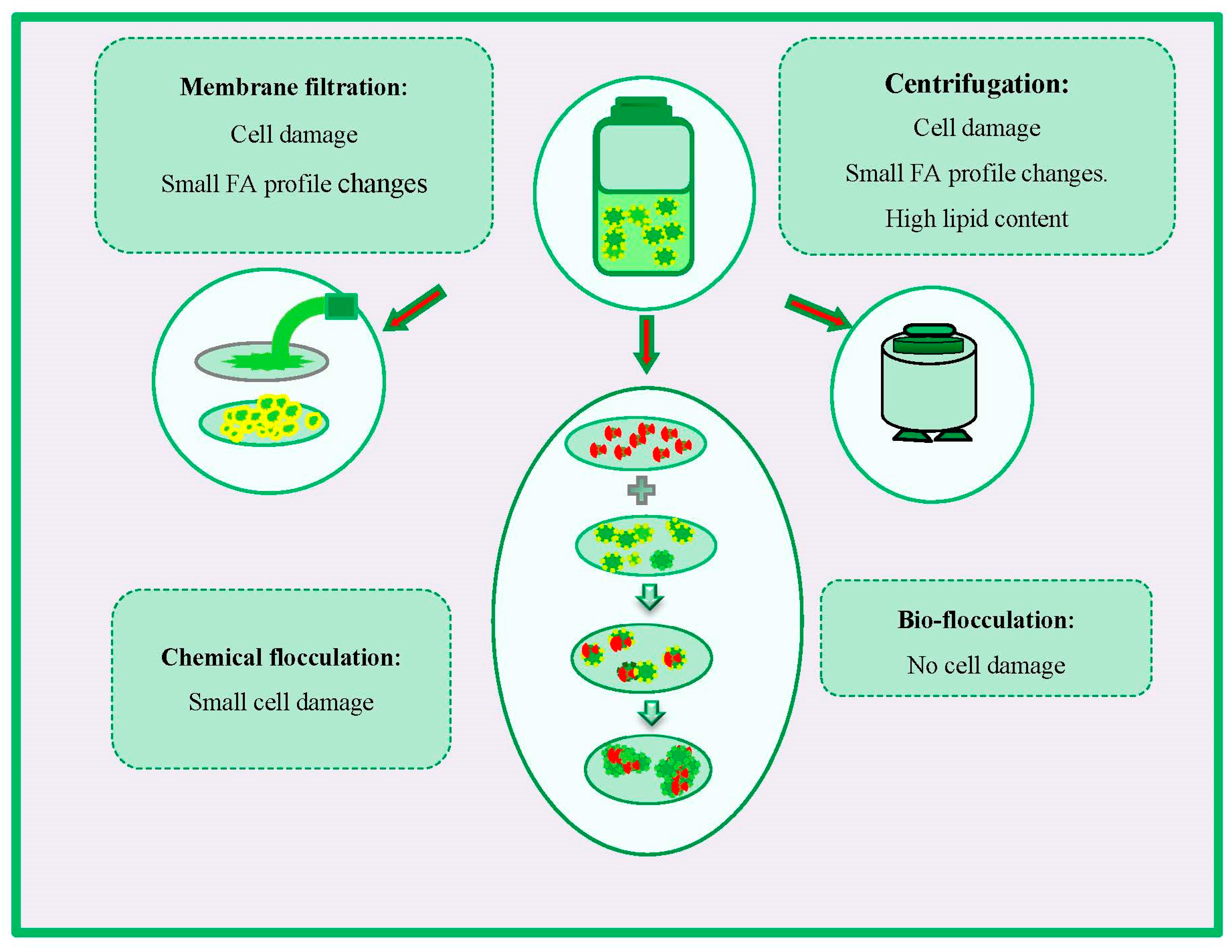
Harvesting is an important stage in the feedstock production process that requires to be investigated carefully throughout an integrated approach [49,51]. Accordingly, establishing the strategies that provide the best performance without qualitative damages to FA profiles, biomass, and lipid contents is crucial in implementing successful feedstock production [51]. Some strategies have been developed for algal biomass recovering, which the major ones include membrane filtration, centrifugation, and coagulation [124]. The effects of various harvesting methods on FA change and cell damage are presented in Figure 4.
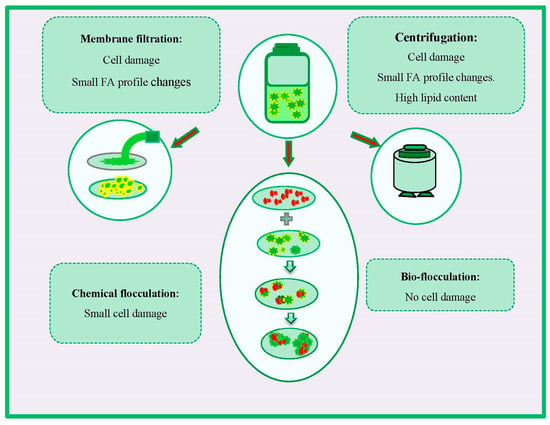
Figure 4.
Effects of different harvesting methods on fatty acid (FA) changes and cell damage.
Flocculation is a convenient harvesting strategy for recovering large volumes of algal biomass [169]. It can be performed by conventional harvesting methods, e.g., bioflocculation, chemical flocculation, or novel strategies such as magnetic nanoparticles [170]. It was reported that alum and alkaline flocculation did not severely affect total lipid content. In an investigation, Chatsungnoen et al. [171] found that metal salts such as ferric chloride and aluminum sulfate irreversibly bind to the biomass of Neochloris, Nannochloropsis, and Chlorella, sp. However, these metal coagulants did not significantly impact the biomass and total lipid content. In another study, Vandamme et al. [51] obtained slightly lower lipid content by alkaline or alum flocculation. The reduced lipid content might be explainable by the algal extracellular organic matter (EOM) interaction with the used coagulants [51,172]. Bioflocculants can reduce the demand for chemical flocculants. However, the competition between the bacteria and algae is a drawback for co-cultivation of bioflocculant-producing bacteria and microalgae, which subsequently can affect algal lipid content and cell density. In the recent past, some harvesting strategies have been proposed as promising approaches for algal biomass recovering. Among them, electro-coagulation–floatation (ECF) is considered a good substitute to conventional harvesting methods such as the chemical flocculation approach, due to the low energy requirement and no direct use of coagulant [173]. Fayad et al. [174] applied ECF, using iron and aluminum electrodes, for the harvesting of Chlorella vulgaris biomass and reported this harvesting method to have no impact on the amount of algal lipid production.
Additionally, magnetic particles have been described as an interesting flocculation option for microalgal harvesting, in which suspended algae cells were adsorbed or tagged to nano-sized or micron-sized magnetic particles. The tagged composites were separated using external magnet force because of intrinsic paramagnetic movement [175]. Liu et al. [52] applied magnetite nanoparticles (nano-Fe3O4 covered with polyethyleneimine) to recover microalgae C. pyrenoidosa and S. obliquus, and reported the process did not reduce the lipid content of microalgae. In another study, C. pyrenoidosa cells were harvested by the method of flocculation using Fe3O4-silica nanoparticles for improving microalgae lipid production [176].
As for the FA profile, contradicting results have been reported in different harvesting methods. Some studies showed significant differences in obtained FA profiles in various recovering strategies, but in other investigations, the differences were not significant (Table 8). For instance, in a study, three harvesting methods of centrifugation, microfiltration membrane, and coagulation were analyzed in Chlorella sp. biomass recovering. Coagulation was found to exhibit the most poorly results in terms of obtained FA profiles compared to centrifugation and microfiltration membranes [177,178].

Table 8.
Lipid recovery under various harvesting strategies.
Membrane filtration is another efficient harvesting strategy for the aggregation of microalgal cells. Operating under a low trans-membrane pressure makes the approach less energy-intensive than centrifugation, and the long membrane lifespan makes the harvesting process more cost-effective in the long term [179]. Membrane-based separation processes also pose several challenges such as membrane clogging, and electrostatic repulsion from the negative surface charges of microalgae cells and membrane surface.
Centrifugation represents an alternative strategy that can be used in pilot-scale production. The harvesting by centrifugation generally presents better results in terms of lipid content when compared to filtration or flocculation [178,179]. However, it should be noted that algal cells exposed to high gravitational forces during centrifugation can result in structural cell damage and small FA profile changes. The effects of harvesting strategy on biomass quality are important when biochemical components must meet quality standards for further processing of obtained algal biomass, e.g., lipids for biodiesel production (Figure 3) [49].
4.2. Post-Harvesting Stage
Lipid enhancement approaches involving alteration of environmental conditions and nutrient limitation regimes such as temperature, light, and nutrient limitation (e.g., phosphorus and nitrogen) are conventional strategies applied to increase lipid production in microalgae [16]. Generally, conventional stress-inducing methods are applied during the cultivation stage [19]. However, recent investigations have shown that the application of different strategies in other stages of the feedstock production process also can improve lipid and/or biomass production in microalgae [51,123,181]. It was reported that the stress induction in the post-harvesting stage resulted in a positive effect in the lipid accumulation of Chlorella vulgaris [50]. In this investigation, the high lipid content in the after-harvesting stage was achieved in one day of nutrients starvation under dark conditions. The lipid production under these conditions was considerably increased compared to the control group. In addition, it was demonstrated that the main produced FAs of C. vulgaris were oleic acid, palmitic acid, and linoleic acid, sharing similar FA profiles to those of soybean, sunflower, and corn oil [50].
Additionally, microalgae-based biofuels are produced from algal feedstock by thermochemical, biochemical, and chemical methods. Among the thermochemical techniques, pyrolysis is considered as a potential approach involving high pressure and high temperature to produce bio-oil and biochar from the microalgal feedstock. Therefore, selecting a suitable pyrolysis method can influence obtaining a desirable quality and quantity of bio-oil from algae [182].
5. Conclusions
Microalgae have received widespread interest owing to their unique properties in producing large amounts of lipids and fast growth rates. However, algal strains exhibit conflicting features in terms of the conditions required for maximal lipid and biomass production. These contradictory features can be mitigated by applying appropriate strategies throughout the biomass production process. The purpose of this article is to review the technologies and advancements available for enhancing lipid productivity in microalgae species. The first step in the feedstock production process is screening the right alga with relevant properties and further improvement of those platform species by genetic manipulation. Approaches like genetic modification at the metabolic and genomic levels can be beneficial in improving biomass/lipid production. Additionally, it has been reported that, throughout cultivation, threshold nutrients give lower lipid content but high biomass yield and vice versa. These conditions can be mitigated by optimizing an appropriate two-phase cultivation strategy. Apart from the two-stage cultivation strategy, alteration growth parameters in some other cultivation strategies such as continuous chemostat can also produce higher lipid/biomass yield. Establishing the strategies that provide the best performance in the harvesting stage without qualitative damages to microalgae biomass and/or lipid content is important in the successful implementation of the feedstock production process. Magnetic and Bio-based flocculants are very promising for algal biomass recovering. Thus, choosing an appropriate screening, cultivation, harvesting, and after-harvesting strategies can influence obtaining desirable quantity and quality of bio-oil from algae. It is our hope that this review article could inspire ongoing efforts in developing sustainable microalgae-derived biofuel production with improved biomass/lipid yield at an economically feasible cost.
Author Contributions
Conceptualization, Z.S., and H.S.; software, Z.S., H.S., O.H.C., S.S.R.K., and M.P.; validation, Z.S.; formal analysis, Z.S., and H.S.; investigation, Z.S.; resources, Z.S., H.S., O.H.C., W.J.L., S.S.R.K., M.P., and A.F.I.; data curation, Z.S.; writing—original draft preparation, Z.S.; writing—review and editing, Z.S., H.S., O.H.C., W.J.L., and A.F.I.; visualization, Z.S., H.S., O.H.C., W.J.L., S.S.R.K., M.P., and A.F.I.; project administration, Z.S., H.S., O.H.C., W.J.L., S.S.R.K., M.P., and A.F.I.; funding acquisition, Z.S., H.S., O.H.C., W.J.L., S.S.R.K., M.P., and A.F.I. All authors have read and agreed to the published version of the manuscript.
Funding
Ministry of Education, Youth, and Sports of the Czech Republic and the European Union (European Structural and Investment Funds Operational Program Research, Development, and Education) in the framework of the project “Modular platform for autonomous chassis of specialized electric vehicles for freight and equipment transportation”, Reg. No. CZ.02.1.01/0.0/0.0/16_025/0007293, as well as the financial support from internal grants in the Institute for Nanomaterials, Advanced Technologies and Innovations (CXI), Technical University of Liberec (TUL).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Singh, D.; Sharma, D.; Soni, S.; Sharma, S.; Sharma, P.K.; Jhalani, A. A review on feedstocks, production processes, and yield for different generations of biodiesel. Fuel 2020, 262, 116553. [Google Scholar] [CrossRef]
- Günay, M.E.; Türker, L.; Tapan, N.A. Significant parameters and technological advancements in biodiesel production systems. Fuel 2019, 250, 27–41. [Google Scholar] [CrossRef]
- Milano, J.; Ong, H.C.; Masjuki, H.; Chong, W.; Lam, M.K.; Loh, P.K.; Vellayan, V. Microalgae biofuels as an alternative to fossil fuel for power generation. Renew. Sustain. Energy Rev. 2016, 58, 180–197. [Google Scholar] [CrossRef]
- Ullah, K.; Ahmad, M.; Sharma, V.K.; Lu, P.; Harvey, A.; Zafar, M.; Sultana, S. Assessing the potential of algal biomass opportunities for bioenergy industry: A review. Fuel 2015, 143, 414–423. [Google Scholar] [CrossRef]
- Shanmugam, S.; Hari, A.; Pandey, A.; Mathimani, T.; Felix, L.; Pugazhendhi, A. Comprehensive review on the application of inorganic and organic nanoparticles for enhancing biohydrogen production. Fuel 2020, 270, 117453. [Google Scholar] [CrossRef]
- Sharma, Y.; Singh, B.; Upadhyay, S. Advancements in development and characterization of biodiesel: A review. Fuel 2008, 87, 2355–2373. [Google Scholar] [CrossRef]
- Stephen, J.L.; Periyasamy, B. Innovative developments in biofuels production from organic waste materials: A review. Fuel 2018, 214, 623–633. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Vassileva, C.G. Composition, properties and challenges of algae biomass for biofuel application: An overview. Fuel 2016, 181, 1–33. [Google Scholar] [CrossRef]
- Khalid, A.A.H.; Yaakob, Z.; Abdullah, S.R.S.; Takriff, M.S. Growth improvement and metabolic profiling of native and commercial Chlorella sorokiniana strains acclimatized in recycled agricultural wastewater. Bioresour. Technol. 2018, 247, 930–939. [Google Scholar] [CrossRef]
- Deng, X.; Li, Y.; Fei, X. Microalgae: A promising feedstock for biodiesel. Afr. J. Microbiol. Res. 2009, 3, 1008–1014. [Google Scholar]
- Nayak, M.; Suh, W.I.; Chang, Y.K.; Lee, B. Exploration of two-stage cultivation strategies using nitrogen starvation to maximize the lipid productivity in Chlorella sp. HS2. Bioresour. Technol. 2019, 276, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Shokravi, H.; Shokravi, Z.; Heidarrezaei, M.; Lau, W.J.; Koloor, S.S.R.; Petrů, M.; Chyuan, O.H.; Ismail, A.F. Fourth Generation Biofuel: A Review on Challenges and Future Directions. Fuel 2020. under review. [Google Scholar]
- Adams, C.; Godfrey, V.; Wahlen, B.; Seefeldt, L.; Bugbee, B. Understanding precision nitrogen stress to optimize the growth and lipid content tradeoff in oleaginous green microalgae. Bioresour. Technol. 2013, 131, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Wan, C.; Mehmood, M.A.; Chang, J.-S.; Bai, F.; Zhao, X. Manipulating environmental stresses and stress tolerance of microalgae for enhanced production of lipids and value-added products—A review. Bioresour. Technol. 2017, 244, 1198–1206. [Google Scholar] [CrossRef]
- Ghosh, A.; Khanra, S.; Mondal, M.; Halder, G.; Tiwari, O.; Saini, S.; Bhowmick, T.K.; Gayen, K. Progress toward isolation of strains and genetically engineered strains of microalgae for production of biofuel and other value added chemicals: A review. Energy Convers. Manag. 2016, 113, 104–118. [Google Scholar] [CrossRef]
- Sajjadi, B.; Chen, W.-Y.; Raman, A.A.A.; Ibrahim, S. Microalgae lipid and biomass for biofuel production: A comprehensive review on lipid enhancement strategies and their effects on fatty acid composition. Renew. Sustain. Energy Rev. 2018, 97, 200–232. [Google Scholar] [CrossRef]
- Shokravi, H.; Shokravi, Z.; Aziz, M.A.; Shokravi, H. 11 Algal Biofuel: A Promising. In Fossil Free Fuels: Trends in Renewable Energy; Taylor & Francis Group: Oxford, UK, 2019; pp. 187–211. [Google Scholar]
- Shokravi, H.; Shokravi, Z.; Aziz, M.A.; Shokravi, H. 12 The Fourth-Generation. In Fossil Free Fuels: Trends in Renewable Energy; Taylor & Francis Group: Oxford, UK, 2019; pp. 213–251. [Google Scholar]
- Ho, S.-H.; Ye, X.; Hasunuma, T.; Chang, J.-S.; Kondo, A. Perspectives on engineering strategies for improving biofuel production from microalgae—A critical review. Biotechnol. Adv. 2014, 32, 1448–1459. [Google Scholar] [CrossRef]
- Peng, L.; Fu, D.; Chu, H.; Wang, Z.; Qi, H. Biofuel production from microalgae: A review. Environ. Chem. Lett. 2019, 18, 285–297. [Google Scholar] [CrossRef]
- Lim, D.K.Y.; Schenk, P.M. Microalgae selection and improvement as oil crops: GM vs non-GM strain engineering. AIMS Environ. Sci. 2017, 4, 151–161. [Google Scholar] [CrossRef]
- He, Q.; Yang, H.; Xu, L.; Xia, L.; Hu, C. Sufficient utilization of natural fluctuating light intensity is an effective approach of promoting lipid productivity in oleaginous microalgal cultivation outdoors. Bioresour. Technol. 2015, 180, 79–87. [Google Scholar] [CrossRef]
- Přibyl, P.; Cepák, V.; Zachleder, V. Production of lipids in 10 strains of Chlorella and Parachlorella, and enhanced lipid productivity in Chlorella vulgaris. Appl. Microbiol. Biotechnol. 2012, 94, 549–561. [Google Scholar] [CrossRef]
- Kwak, H.S.; Kim, J.Y.H.; Woo, H.M.; Jin, E.; Min, B.K.; Sim, S.J. Synergistic effect of multiple stress conditions for improving microalgal lipid production. Algal Res. 2016, 19, 215–224. [Google Scholar] [CrossRef]
- Kim, H.S.; Hsu, S.-C.; Han, S.-I.; Thapa, H.R.; Guzman, A.R.; Browne, D.R.; Tatli, M.; Devarenne, T.P.; Stern, D.B.; Han, A. High-throughput droplet microfluidics screening platform for selecting fast-growing and high lipid-producing microalgae from a mutant library. Plant Direct 2017, 1, e00011. [Google Scholar] [CrossRef]
- Challagulla, V.; Nayar, S.; Walsh, K.; Fabbro, L. Advances in techniques for assessment of microalgal lipids. Crit. Rev. Biotechnol. 2016, 37, 566–578. [Google Scholar] [CrossRef]
- Cabanelas, I.T.D.; Van Der Zwart, M.; Kleinegris, D.M.M.; Barbosa, M.J.; Wijffels, R.H. Rapid method to screen and sort lipid accumulating microalgae. Bioresour. Technol. 2015, 184, 47–52. [Google Scholar] [CrossRef]
- Erickson, R.A.; Jimenez, R. Microfluidic cytometer for high-throughput measurement of photosynthetic characteristics and lipid accumulation in individual algal cells. Lab Chip 2013, 13, 2893–2901. [Google Scholar] [CrossRef]
- Liu, J.; Mukherjee, J.; Hawkes, J.J.; Wilkinson, S.J. Optimization of lipid production for algal biodiesel in nitrogen stressed cells of Dunaliella salina using FTIR analysis. J. Chem. Technol. Biotechnol. 2013, 88, 1807–1814. [Google Scholar] [CrossRef]
- Shokravi, Z.; Mehrad, L.; Ramazani, A. Detecting the frequency of aminoglycoside modifying enzyme encoding genes among clinical isolates of methicillin-resistant Staphylococcus aureus. BioImpacts 2015, 5, 87–91. [Google Scholar] [CrossRef][Green Version]
- Shokravi, Z.; Haseli, M.; Mehrad, L.; Ramazani, A. Distribution of Staphylococcal cassette chromosome mecA (SCCmec) types among coagulase-negative Staphylococci isolates from healthcare workers in the North-West of Iran. Iran. J. Basic Med. Sci. 2020, 23, 1489–1493. [Google Scholar] [CrossRef]
- Ramazani, A.; Shokravi, Z.; Mehrad, L.; Sorouri, R.; Amirmoghaddami, H. Study the Genes Encoding Aminoglycoside-Modifying Enzymes among Clinical Isolates of Methicillin Resistance Staphylococcus Aureus. 2013. Available online: https://www.sid.ir/en/Seminar/ViewPaper.aspx?ID=32537 (accessed on 29 October 2020).
- Heidarrezaei, M.; Shokravi, H.; Huyop, F.; Koloor, S.R.; Petru, M. Isolation and Characterization of a Novel Bacterium from the Marine Environment for Trichloroacetic Acid Bioremediation. Appl. Sci. 2020, 10, 4593. [Google Scholar] [CrossRef]
- Sharma, P.K.; Saharia, M.; Srivstava, R.; Kumar, S.; Sahoo, L. Tailoring Microalgae for Efficient Biofuel Production. Front. Mar. Sci. 2018, 5, 382. [Google Scholar] [CrossRef]
- Trentacoste, E.M.; Shrestha, R.P.; Smith, S.R.; Glé, C.; Hartmann, A.C.; Hildebrand, M.; Gerwick, W.H. Metabolic engineering of lipid catabolism increases microalgal lipid accumulation without compromising growth. Proc. Natl. Acad. Sci. USA 2013, 110, 19748–19753. [Google Scholar] [CrossRef]
- Nguyen, T.H.T.; Park, S.; Jeong, J.; Shin, Y.S.; Sim, S.J.; Jin, E. Enhancing lipid productivity by modulating lipid catabolism using the CRISPR-Cas9 system in Chlamydomonas. Environ. Biol. Fishes 2020, 32, 2829–2840. [Google Scholar] [CrossRef]
- Park, S.; Nguyen, T.H.T.; Jin, E. Improving lipid production by strain development in microalgae: Strategies, challenges and perspectives. Bioresour. Technol. 2019, 292, 121953. [Google Scholar] [CrossRef]
- Xia, L.; Ge, H.; Zhou, X.; Zhang, D.; Hu, C. Photoautotrophic outdoor two-stage cultivation for oleaginous microalgae Scenedesmus obtusus XJ-15. Bioresour. Technol. 2013, 144, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.-H.; Cho, D.-H.; Lee, S.; Heo, J.; Tran, Q.-G.; Chang, Y.K.; Kim, H.-S. Hybrid operation of photobioreactor and wastewater-fed open raceway ponds enhances the dominance of target algal species and algal biomass production. Algal Res. 2018, 29, 319–329. [Google Scholar] [CrossRef]
- Nagappan, S.; Devendran, S.; Tsai, P.-C.; Dahms, H.-U.; Ponnusamy, V.K. Potential of two-stage cultivation in microalgae biofuel production. Fuel 2019, 252, 339–349. [Google Scholar] [CrossRef]
- Aziz, M.A.; Kassim, K.A.; Shokravi, Z.; Jakarni, F.M.; Liu, H.Y.; Zaini, N.; Tan, L.S.; Islam, A.S.; Shokravi, H.; Lieu, H.Y. Two-stage cultivation strategy for simultaneous increases in growth rate and lipid content of microalgae: A review. Renew. Sustain. Energy Rev. 2020, 119, 109621. [Google Scholar] [CrossRef]
- Bhatia, S.; Mehariya, S.; Bhatia, R.K.; Kumar, M.; Pugazhendhi, A.; Awasthi, M.K.; Atabani, A.; Kumar, G.; Kim, W.; Seo, S.-O.; et al. Wastewater based microalgal biorefinery for bioenergy production: Progress and challenges. Sci. Total. Environ. 2021, 751, 141599. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Species and strain selection. In Algae for Biofuels and Energy; Springer: Berlin, Germany, 2013; pp. 77–89. [Google Scholar]
- Singh, P.; Kumari, S.; Guldhe, A.; Misra, R.; Rawat, I.; Bux, F. Trends and novel strategies for enhancing lipid accumulation and quality in microalgae. Renew. Sustain. Energy Rev. 2016, 55, 1–16. [Google Scholar] [CrossRef]
- Chu, W.-L. Strategies to enhance production of microalgal biomass and lipids for biofuel feedstock. Eur. J. Phycol. 2017, 52, 419–437. [Google Scholar] [CrossRef]
- Chung, Y.-S.; Lee, J.-W.; Chung, C.-H. Molecular challenges in microalgae towards cost-effective production of quality biodiesel. Renew. Sustain. Energy Rev. 2017, 74, 139–144. [Google Scholar] [CrossRef]
- Shin, Y.S.; Choi, H.I.; Choi, J.W.; Lee, J.S.; Sung, Y.J.; Sim, S.J. Multilateral approach on enhancing economic viability of lipid production from microalgae: A review. Bioresour. Technol. 2018, 258, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Piligaev, A.V.; Sorokina, K.N.; Samoylova, Y.V.; Parmon, V.N. Production of Microalgal Biomass with High Lipid Content and Their Catalytic Processing Into Biodiesel: A Review. Catal. Ind. 2019, 11, 349–359. [Google Scholar] [CrossRef]
- Menegazzo, M.L.; Fonseca, G.G. Biomass recovery and lipid extraction processes for microalgae biofuels production: A review. Renew. Sustain. Energy Rev. 2019, 107, 87–107. [Google Scholar] [CrossRef]
- Poh, Z.L.; Kadir, W.N.A.; Lam, M.-K.; Uemura, Y.; Suparmaniam, U.; Lim, J.W.; Show, P.L.; Lee, K.T. The effect of stress environment towards lipid accumulation in microalgae after harvesting. Renew. Energy 2020, 154, 1083–1091. [Google Scholar] [CrossRef]
- Vandamme, D.; Gheysen, L.; Muylaert, K.; Foubert, I. Impact of harvesting method on total lipid content and extraction efficiency for Phaeodactylum tricornutum. Sep. Purif. Technol. 2018, 194, 362–367. [Google Scholar] [CrossRef]
- Liu, Y.; Jin, W.; Zhou, X.; Han, S.-F.; Tu, R.; Feng, X.; Jensen, P.D.; Wang, Q. Efficient harvesting of Chlorella pyrenoidosa and Scenedesmus obliquus cultivated in urban sewage by magnetic flocculation using nano-Fe3O4 coated with polyethyleneimine. Bioresour. Technol. 2019, 290, 121771. [Google Scholar] [CrossRef]
- Nwokoagbara, E.; Olaleye, A.K.; Wang, M. Biodiesel from microalgae: The use of multi-criteria decision analysis for strain selection. Fuel 2015, 159, 241–249. [Google Scholar] [CrossRef]
- Sibi, G.; Shetty, V.; Mokashi, K. Enhanced lipid productivity approaches in microalgae as an alternate for fossil fuels—A review. J. Energy Inst. 2016, 89, 330–334. [Google Scholar] [CrossRef]
- Gao, Y.; Yang, M.; Wang, C. Nutrient deprivation enhances lipid content in marine microalgae. Bioresour. Technol. 2013, 147, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wan, L.; Li, A.; Zhang, C. Responses in growth, lipid accumulation, and fatty acid composition of four oleaginous microalgae to different nitrogen sources and concentrations. Chin. J. Oceanol. Limnol. 2013, 31, 1306–1314. [Google Scholar] [CrossRef]
- Yu, N.; Dieu, L.T.J.; Harvey, S.; Lee, D.-Y. Optimization of process configuration and strain selection for microalgae-based biodiesel production. Bioresour. Technol. 2015, 193, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Ren, Y.; Mao, X.; Li, X.; Zhang, H.; Lao, Y.; Chen, F. Harnessing C/N balance of Chromochloris zofingiensis to overcome the potential conflict in microalgal production. Commun. Biol. 2020, 3, 1–13. [Google Scholar] [CrossRef]
- Hsieh, H.-J.; Su, C.-H.; Chien, L.-J. Accumulation of lipid production in Chlorella minutissima by triacylglycerol biosynthesis-related genes cloned from Saccharomyces cerevisiae and Yarrowia lipolytica. J. Microbiol. 2012, 50, 526–534. [Google Scholar] [CrossRef]
- Moussa, I.D.-B.; Chtourou, H.; Hassairi, I.; Sayadi, S.; Dhouib, A. The effect of switching environmental conditions on content and structure of lipid produced by a wild strain Picochlorum sp. Renew. Energy 2019, 134, 406–415. [Google Scholar] [CrossRef]
- Pedro, A.S.; González-López, C.; Acién, F.; Molina-Grima, E. Marine microalgae selection and culture conditions optimization for biodiesel production. Bioresour. Technol. 2013, 134, 353–361. [Google Scholar] [CrossRef]
- Pal, P.; Chew, K.W.; Yen, H.-W.; Lim, J.W.; Lam, M.K.; Show, P.L. Cultivation of oily microalgae for the production of third-generation biofuels. Sustainability 2019, 11, 5424. [Google Scholar] [CrossRef]
- De Bhowmick, G.; Koduru, L.; Sen, R. Metabolic pathway engineering towards enhancing microalgal lipid biosynthesis for biofuel application—A review. Renew. Sustain. Energy Rev. 2015, 50, 1239–1253. [Google Scholar] [CrossRef]
- El Arroussi, H.; Benhima, R.; El Mernissi, N.; Bouhfid, R.; Tilsaghani, C.; Bennis, I.; Wahby, I. Screening of marine microalgae strains from Moroccan coasts for biodiesel production. Renew. Energy 2017, 113, 1515–1522. [Google Scholar] [CrossRef]
- Huang, S.T.; Goh, J.L.; Ahmadzadeh, H.; Murry, M.A. A rapid sampling technique for isolating highly productive lipid-rich algae strains from environmental samples. Biofuel Res. J. 2019, 6, 920–926. [Google Scholar] [CrossRef]
- Neofotis, P.; Huang, A.; Sury, K.; Chang, W.; Joseph, F.; Gabr, A.; Twary, S.; Qiu, W.; Holguin, O.; Polle, J.E. Characterization and classification of highly productive microalgae strains discovered for biofuel and bioproduct generation. Algal Res. 2016, 15, 164–178. [Google Scholar] [CrossRef]
- Andersen, R.A.; Kawachi, M. Traditional Microalgae Isolation Techniques. In Algal Culturing Techniques; Elsevier Academic Press: Burlington, MA, USA, 2005; pp. 83–100. [Google Scholar]
- Mutanda, T.; Ramesh, D.; Karthikeyan, S.; Kumari, S.; Anandraj, A.; Bux, F. Bioprospecting for hyper-lipid producing microalgal strains for sustainable biofuel production. Bioresour. Technol. 2011, 102, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Cabanelas, I.T.D.; Van Der Zwart, M.; Kleinegris, D.M.M.; Wijffels, R.H.; Barbosa, M.J. Sorting cells of the microalga Chlorococcum littorale with increased triacylglycerol productivity. Biotechnol. Biofuels 2016, 9, 1–12. [Google Scholar] [CrossRef]
- Pereira, H.; Barreira, L.; Mozes, A.; Florindo, C.; Polo, C.; Duarte, C.V.; Custodio, L.; Varela, J. Microplate-based high throughput screening procedure for the isolation of lipid-rich marine microalgae. Biotechnol. Biofuels 2011, 4, 61. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Guzman, A.R.; Thapa, H.R.; Devarenne, T.P.; Han, A. A droplet microfluidics platform for rapid microalgal growth and oil production analysis. Biotechnol. Bioeng. 2016, 113, 1691–1701. [Google Scholar] [CrossRef]
- Del Río, E.; Acién, F.G.; Guerrero, M.G.; Sanchez, E.D.R. Photoautotrophic production of astaxanthin by the microalga Haematococcus pluvialis. In Sustainable Biotechnology; Springer: Dordrecht, The Netherlands, 2010; pp. 247–258. [Google Scholar]
- Sung, M.-G.; Lee, B.; Kim, C.W.; Nam, K.; Chang, Y.K. Enhancement of lipid productivity by adopting multi-stage continuous cultivation strategy in Nannochloropsis gaditana. Bioresour. Technol. 2017, 229, 20–25. [Google Scholar] [CrossRef]
- Abdullah, B.; Muhammad, S.A.F.S.; Shokravi, Z.; Ismail, S.; Kassim, K.A.; Mahmood, A.N.; Aziz, M.A. Fourth generation biofuel: A review on risks and mitigation strategies. Renew. Sustain. Energy Rev. 2019, 107, 37–50. [Google Scholar] [CrossRef]
- Davis, R.; Aden, A.; Pienkos, P.T. Techno-economic analysis of autotrophic microalgae for fuel production. Appl. Energy 2011, 88, 3524–3531. [Google Scholar] [CrossRef]
- Eguiheneuf, F.; Ekhan, A.; Tran, L.-S.P. Genetic Engineering: A Promising Tool to Engender Physiological, Biochemical, and Molecular Stress Resilience in Green Microalgae. Front. Plant Sci. 2016, 7, 400. [Google Scholar] [CrossRef]
- Tan, K.W.M.; Lee, Y.-K. The dilemma for lipid productivity in green microalgae: Importance of substrate provision in improving oil yield without sacrificing growth. Biotechnol. Biofuels 2016, 9, 255. [Google Scholar] [CrossRef] [PubMed]
- Talebi, A.F.; Tohidfar, M.; Bagheri, A.; Lyon, S.R.; Salehi-Ashtiani, K.; Tabatabaei, M. Manipulation of carbon flux into fatty acid biosynthesis pathway in Dunaliella salina using AccD and ME genes to enhance lipid content and to improve produced biodiesel quality. Biofuel Res. J. 2014, 1, 91–97. [Google Scholar] [CrossRef]
- Xue, J.; Niu, Y.-F.; Huang, T.; Yang, W.-D.; Liu, J.-S.; Li, H.-Y. Genetic improvement of the microalga Phaeodactylum tricornutum for boosting neutral lipid accumulation. Metab. Eng. 2015, 27, 1–9. [Google Scholar] [CrossRef]
- Ma, Y.-H.; Wang, X.; Niu, Y.-F.; Yang, Z.-K.; Zhang, M.-H.; Wang, Z.; Yang, W.-D.; Liu, J.-S.; Li, H.-Y. Antisense knockdown of pyruvate dehydrogenase kinase promotes the neutral lipid accumulation in the diatom Phaeodactylum tricornutum. Microb. Cell Factories 2014, 13, 100. [Google Scholar] [CrossRef]
- Muto, M.; Tanaka, M.; Liang, Y.; Yoshino, T.; Matsumoto, M.; Tanaka, T. Enhancement of glycerol metabolism in the oleaginous marine diatom Fistulifera solaris JPCC DA0580 to improve triacylglycerol productivity. Biotechnol. Biofuels 2015, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Ning, K.; Zeng, X.; Luo, Y.; Wang, D.; Hu, J.; Li, J.; Xu, H.; Huang, J.; Wan, M.; et al. Genomic foundation of starch-to-lipid switch in Oleaginous Chlorella spp. Plant Physiol. 2015, 169, 2444–2461. [Google Scholar]
- Deng, X.; Li, Y.; Fei, X. The mRNA abundance of pepc2 gene is negatively correlated with oil content in Chlamydomonas reinhardtii. Biomass Bioenergy 2011, 35, 1811–1817. [Google Scholar] [CrossRef]
- Muñoz, C.F.; Weusthuis, R.A.; D’Adamo, S.; Wijffels, R.H. Effect of Single and Combined Expression of Lysophosphatidic Acid Acyltransferase, Glycerol-3-Phosphate Acyltransferase, and Diacylglycerol Acyltransferase on Lipid Accumulation and Composition in Neochloris oleoabundans. Front. Plant Sci. 2019, 10, 1573. [Google Scholar] [CrossRef]
- Shahid, A.; Rehman, A.U.; Usman, M.; Ashraf, M.U.F.; Javed, M.R.; Khan, A.Z.; Gill, S.S.; Mehmood, M.A. Engineering the metabolic pathways of lipid biosynthesis to develop robust microalgal strains for biodiesel production. Biotechnol. Appl. Biochem. 2020, 67, 41–51. [Google Scholar] [CrossRef]
- Ran, W.; Wang, H.; Liu, Y.; Qi, M.; Xiang, Q.; Yao, C.; Zhang, Y.; Lan, X. Storage of starch and lipids in microalgae: Biosynthesis and manipulation by nutrients. Bioresour. Technol. 2019, 291, 121894. [Google Scholar] [CrossRef]
- Zhu, B.-H.; Shi, H.-P.; Yang, G.; Lv, N.-N.; Yang, M.; Pan, K.-H. Silencing UDP-glucose pyrophosphorylase gene in Phaeodactylum tricornutum affects carbon allocation. New Biotechnol. 2016, 33, 237–244. [Google Scholar] [CrossRef]
- Wan, M.; Jin, X.; Xia, J.; Rosenberg, J.N.; Yu, G.; Nie, Z.; Oyler, G.A.; Betenbaugh, M. The effect of iron on growth, lipid accumulation, and gene expression profile of the freshwater microalga Chlorella sorokiniana. Appl. Microbiol. Biotechnol. 2014, 98, 9473–9481. [Google Scholar] [CrossRef]
- Chauton, M.S.; Winge, P.; Brembu, T.; Vadstein, O.; Bones, A.M. Gene Regulation of Carbon Fixation, Storage, and Utilization in the Diatom Phaeodactylum tricornutum Acclimated to Light/Dark Cycles. Plant Physiol. 2012, 161, 1034–1048. [Google Scholar] [CrossRef]
- Solovchenko, A. Physiological role of neutral lipid accumulation in eukaryotic microalgae under stresses. Russ. J. Plant Physiol. 2012, 59, 167–176. [Google Scholar] [CrossRef]
- Iwai, M.; Ikeda, K.; Shimojima, M.; Ohta, H. Enhancement of extraplastidic oil synthesis in C hlamydomonas reinhardtii using a type-2 diacylglycerol acyltransferase with a phosphorus starvation–inducible promoter. Plant Biotechnol. J. 2014, 12, 808–819. [Google Scholar] [CrossRef] [PubMed]
- Eiwai, M.; Ehori, K.; Esasaki-Sekimoto, Y.; Eshimojima, M.; Ohta, H. Manipulation of oil synthesis in Nannochloropsis strain NIES-2145 with a phosphorus starvation–inducible promoter from Chlamydomonas reinhardtii. Front. Microbiol. 2015, 6, 912. [Google Scholar] [CrossRef]
- Zhang, J.; Hao, Q.; Bai, L.; Xu, J.; Yin, W.-B.; Song, L.; Xu, L.; Guo, X.; Fan, C.; Chen, Y.; et al. Overexpression of the soybean transcription factor GmDof4 significantly enhances the lipid content of Chlorella ellipsoidea. Biotechnol. Biofuels 2014, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-H.; Warakanont, J.; Takeuchi, T.; Sears, B.B.; Moellering, E.R.; Benning, C. The protein Compromised Hydrolysis of Triacylglycerols 7 (CHT7) acts as a repressor of cellular quiescence in Chlamydomonas. Proc. Natl. Acad. Sci. USA 2014, 111, 15833–15838. [Google Scholar] [CrossRef]
- Ngan, C.Y.; Wong, C.-H.; Choi, C.; Yoshinaga, Y.; Louie, K.; Jia, J.; Chen, C.; Bowen, B.; Cheng, H.; Leonelli, L.; et al. Lineage-specific chromatin signatures reveal a regulator of lipid metabolism in microalgae. Nat. Plants 2015, 1, 15107. [Google Scholar] [CrossRef]
- Ajjawi, I.; Verruto, J.; Aqui, M.; Soriaga, L.B.; Coppersmith, J.; Kwok, K.; Peach, L.; Orchard, E.; Kalb, R.; Xu, W.; et al. Lipid production in Nannochloropsis gaditana is doubled by decreasing expression of a single transcriptional regulator. Nat. Biotechnol. 2017, 35, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.K.; Jeon, S.; Kwon, S.; Koh, H.G.; Shin, S.-E.; Lee, B.; Choi, G.-G.; Yang, J.-W.; Jeong, B.-R.; Chang, Y.K. Effects of overexpression of a bHLH transcription factor on biomass and lipid production in Nannochloropsis salina. Biotechnol. Biofuels 2015, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.; Kang, N.K.; Koh, H.G.; Shin, S.-E.; Lee, B.; Jeong, B.-R.; Chang, Y.K. Enhancement of biomass and lipid productivity by overexpression of a bZIP transcription factor in Nannochloropsis salina. Biotechnol. Bioeng. 2017, 115, 331–340. [Google Scholar] [CrossRef]
- Liang, M.-H.; Jiang, J.-G. Analysis of carotenogenic genes promoters and WRKY transcription factors in response to salt stress in Dunaliella bardawil. Sci. Rep. 2017, 7, 37025. [Google Scholar] [CrossRef]
- Matthijs, M.; Fabris, M.; Broos, S.; Vyverman, W.; Goossens, A. Profiling of the Early Nitrogen Stress Response in the Diatom Phaeodactylum tricornutum Reveals a Novel Family of RING-Domain Transcription Factors. Plant Physiol. 2015, 170, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Li, Y.; Wu, G.; Li, G.; Sun, H.; Deng, S.; Shen, Y.; Chen, G.; Zhang, R.; Meng, C.; et al. Transcriptome Analysis in Haematococcus pluvialis: Astaxanthin Induction by Salicylic Acid (SA) and Jasmonic Acid (JA). PLoS ONE 2015, 10, e0140609. [Google Scholar] [CrossRef]
- Tevatia, R.; Allen, J.; Blum, P.; Demirel, Y.; Black, P. Modeling of rhythmic behavior in neutral lipid production due to continuous supply of limited nitrogen: Mutual growth and lipid accumulation in microalgae. Bioresour. Technol. 2014, 170, 152–159. [Google Scholar] [CrossRef]
- Kumar, V.; Muthuraj, M.; Palabhanvi, B.; Das, D. Synchronized growth and neutral lipid accumulation in Chlorella sorokiniana FC6 IITG under continuous mode of operation. Bioresour. Technol. 2016, 200, 770–779. [Google Scholar] [CrossRef]
- Wen, X.; Geng, Y.; Li, Y. Enhanced lipid production in Chlorella pyrenoidosa by continuous culture. Bioresour. Technol. 2014, 161, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Del Río, E.; García-Gómez, E.; Moreno, J.; Guerrero, M.G.; Garcia-González, M.; Sanchez, E.D.R. Microalgae for oil. Assessment of fatty acid productivity in continuous culture by two high-yield strains, Chlorococcum oleofaciens and Pseudokirchneriella subcapitata. Algal Res. 2017, 23, 37–42. [Google Scholar] [CrossRef]
- Sobczuk, T.M.; Chisti, Y. Potential fuel oils from the microalga Choricystis minor. J. Chem. Technol. Biotechnol. 2010, 85, 100–108. [Google Scholar] [CrossRef]
- Tang, H.; Chen, M.; Ng, K.S.; Salley, S.O. Continuous microalgae cultivation in a photobioreactor. Biotechnol. Bioeng. 2012, 109, 2468–2474. [Google Scholar] [CrossRef] [PubMed]
- Klok, A.J.; Martens, D.E.; Wijffels, R.H.; Lamers, P.P. Simultaneous growth and neutral lipid accumulation in microalgae. Bioresour. Technol. 2013, 134, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.R.; Glé, C.; Abbriano, R.M.; Traller, J.C.; Davis, A.; Trentacoste, E.; Vernet, M.; Allen, A.E.; Hildebrand, M. Transcript level coordination of carbon pathways during silicon starvation-induced lipid accumulation in the diatom Thalassiosira pseudonana. New Phytol. 2016, 210, 890–904. [Google Scholar] [CrossRef] [PubMed]
- Zaparoli, M.; Ziemniczak, F.G.; Mantovani, L.; Costa, J.A.V.; Colla, L.M. Cellular Stress Conditions as a Strategy to Increase Carbohydrate Productivity in Spirulina platensis. Bioenergy Res. 2020, 1–14. [Google Scholar] [CrossRef]
- Jiang, X.-M.; Han, Q.; Gao, X.; Gao, G. Conditions optimising on the yield of biomass, total lipid, and valuable fatty acids in two strains of Skeletonema menzelii. Food Chem. 2016, 194, 723–732. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Rezania, S.; Oryani, B.; Cho, J.; Sabbagh, F.; Rupani, P.F.; Talaiekhozani, A.; Rahimi, N.; Ghahroud, M.L. Technical Aspects of Biofuel Production from Different Sources in Malaysia—A Review. Processes 2020, 8, 993. [Google Scholar] [CrossRef]
- Hu, X.; Liu, B.; Deng, Y.; Bao, X.; Yang, A.; Zhou, J. A novel two-stage culture strategy used to cultivate Chlorella vulgaris for increasing the lipid productivity. Sep. Purif. Technol. 2019, 211, 816–822. [Google Scholar] [CrossRef]
- Rezania, S.; Mohamad, S.E. Response Surface Methodology for Optimization of Ethanol Production from Cocoa Waste. J. Energy Environ. Pollut. 2020, 1, 7–12. [Google Scholar]
- Cheng, D.; Li, D.; Yuan, Y.; Zhou, L.; Li, X.; Wu, T.; Wang, L.; Zhao, Q.; Wei, W.; Sun, Y. Improving carbohydrate and starch accumulation in Chlorella sp. AE10 by a novel two-stage process with cell dilution. Biotechnol. Biofuels 2017, 10, 1–14. [Google Scholar] [CrossRef]
- Griffiths, M.J.; Harrison, S.T.L. Lipid productivity as a key characteristic for choosing algal species for biodiesel production. Environ. Biol. Fishes 2009, 21, 493–507. [Google Scholar] [CrossRef]
- Gao, G.; Wu, M.; Fu, Q.; Li, X.; Xu, J. A two-stage model with nitrogen and silicon limitation enhances lipid productivity and biodiesel features of the marine bloom-forming diatom Skeletonema costatum. Bioresour. Technol. 2019, 289, 121717. [Google Scholar] [CrossRef]
- Álvarez-Díaz, P.D.; Ruiz, J.; Arbib, Z.; Barragán, J.; Garrido-Pérez, C.; Perales, J.A. Lipid Production of Microalga Ankistrodesmus falcatus Increased by Nutrient and Light Starvation in a Two-Stage Cultivation Process. Appl. Biochem. Biotechnol. 2014, 174, 1471–1483. [Google Scholar] [CrossRef] [PubMed]
- Anne-Marie, K.; Yee, W.; Loh, S.H.; Aziz, A.; Cha, T.S. Effects of Excess and Limited Phosphate on Biomass, Lipid and Fatty Acid Contents and the Expression of Four Fatty Acid Desaturase Genes in the Tropical Selenastraceaen Messastrum gracile SE-MC4. Appl. Biochem. Biotechnol. 2019, 190, 1438–1456. [Google Scholar] [CrossRef] [PubMed]
- Shokravi, H.; Shaghaghi, T.M. Experimental Study on After-Mixing Temperature Control of Concrete in Batching Plant Production. IOP Mater. Sci. Eng. 2020, 713. [Google Scholar] [CrossRef]
- Ahmad, A.; Osman, S.M.; Cha, T.C.; Loh, S.H. Phosphate-induced changes in fatty acid biosynthesis in Chlorella sp. KS-MA2 Strain. Bio Technologia 2016, 97, 295–304. [Google Scholar] [CrossRef]
- Xia, L.; Rong, J.; Yang, H.; He, Q.; Zhang, D.; Hu, C. NaCl as an effective inducer for lipid accumulation in freshwater microalgae Desmodesmus abundans. Bioresour. Technol. 2014, 161, 402–409. [Google Scholar] [CrossRef]
- Goh, B.H.H.; Ong, H.C.; Cheah, M.Y.; Chen, W.-H.; Yu, K.L.; Mahlia, T.M.I. Sustainability of direct biodiesel synthesis from microalgae biomass: A critical review. Renew. Sustain. Energy Rev. 2019, 107, 59–74. [Google Scholar] [CrossRef]
- Park, H.; Jung, D.; Lee, J.; Kim, P.; Cho, Y.; Jung, I.; Kim, Z.-H.; Lim, S.-M.; Lee, C.-G. Improvement of biomass and fatty acid productivity in ocean cultivation of Tetraselmis sp. using hypersaline medium. Environ. Biol. Fishes 2018, 30, 2725–2735. [Google Scholar] [CrossRef]
- Ra, C.-H.; Kang, C.-H.; Jung, J.-H.; Jeong, G.-T.; Kim, S.-K. Effects of light-emitting diodes (LEDs) on the accumulation of lipid content using a two-phase culture process with three microalgae. Bioresour. Technol. 2016, 212, 254–261. [Google Scholar] [CrossRef]
- Yang, H.; He, Q.; Rong, J.; Xia, L.; Hu, C. Rapid neutral lipid accumulation of the alkali-resistant oleaginous Monoraphidium dybowskii LB50 by NaCl induction. Bioresour. Technol. 2014, 172, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Pancha, I.; Chokshi, K.; Maurya, R.; Trivedi, K.; Patidar, S.K.; Ghosh, A.; Mishra, S. Salinity induced oxidative stress enhanced biofuel production potential of microalgae Scenedesmus sp. CCNM 1077. Bioresour. Technol. 2015, 189, 341–348. [Google Scholar] [CrossRef]
- Wensel, P.; Helms, G.; Hiscox, B.; Davis, W.C.; Kirchhoff, H.; Bule, M.; Yu, L.; Chen, S. Isolation, characterization, and validation of oleaginous, multi-trophic, and haloalkaline-tolerant microalgae for two-stage cultivation. Algal Res. 2014, 4, 2–11. [Google Scholar] [CrossRef]
- Srinivasan, R.; Mageswari, A.; Subramanian, P.; Suganthi, C.; Chaitanyakumar, A.; Aswini, V.; Gothandam, K.M. Bicarbonate supplementation enhances growth and biochemical composition of Dunaliella salina V-101 by reducing oxidative stress induced during macronutrient deficit conditions. Sci. Rep. 2018, 8, 6972. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, H.-P.; Han, B.; Yu, X. Coupling of abiotic stresses and phytohormones for the production of lipids and high-value by-products by microalgae: A review. Bioresour. Technol. 2019, 274, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Guldhe, A.; Renuka, N.; Singh, P.; Bux, F. Effect of phytohormones from different classes on gene expression of Chlorella sorokiniana under nitrogen limitation for enhanced biomass and lipid production. Algal Res. 2019, 40, 101518. [Google Scholar] [CrossRef]
- Sivaramakrishnan, R.; Incharoensakdi, A. Enhancement of total lipid yield by nitrogen, carbon, and iron supplementation in isolated microalgae. J. Phycol. 2017, 53, 855–868. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, D.; Xu, J.-W.; Zhao, P.; Li, T.; Ma, H.; Yu, X. Melatonin enhances lipid production in Monoraphidium sp. QLY-1 under nitrogen deficiency conditions via a multi-level mechanism. Bioresour. Technol. 2018, 259, 46–53. [Google Scholar] [CrossRef]
- Che, R.; Ding, K.; Huang, L.; Zhao, P.; Xu, J.-W.; Li, T.; Ma, H.; Yu, X. Enhancing biomass and oil accumulation of Monoraphidium sp. FXY-10 by combined fulvic acid and two-step cultivation. J. Taiwan Inst. Chem. Eng. 2016, 67, 161–165. [Google Scholar] [CrossRef]
- Babu, A.G.; Wu, X.; Kabra, A.N.; Kim, D.-P. Cultivation of an indigenous Chlorella sorokiniana with phytohormones for biomass and lipid production under N-limitation. Algal Res. 2017, 23, 178–185. [Google Scholar] [CrossRef]
- Su, C.-H.; Chien, L.-J.; Gomes, J.; Lin, Y.-S.; Yu, Y.-K.; Liou, J.-S.; Syu, R.-J. Factors affecting lipid accumulation by Nannochloropsis oculata in a two-stage cultivation process. Environ. Biol. Fishes 2010, 23, 903–908. [Google Scholar] [CrossRef]
- Abomohra, A.E.-F.; El-Naggar, A.H.; Alaswad, S.O.; Elsayed, M.; Li, M.; Li, W. Enhancement of biodiesel yield from a halophilic green microalga isolated under extreme hypersaline conditions through stepwise salinity adaptation strategy. Bioresour. Technol. 2020, 310, 123462. [Google Scholar] [CrossRef]
- Hang, L.T.; Mori, K.; Tanaka, Y.; Morikawa, M.; Toyama, T. Enhanced lipid productivity of Chlamydomonas reinhardtii with combination of NaCl and CaCl2 stresses. Bioprocess Biosyst. Eng. 2020, 43, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Mirizadeh, S.; Nosrati, M.; Shojaosadati, S.A. Synergistic Effect of Nutrient and Salt Stress on Lipid Productivity of Chlorella vulgaris through Two-Stage Cultivation. BioEnergy Res. 2019, 13, 507–517. [Google Scholar] [CrossRef]
- Acién, F.; Molina, E.; Reis, A.; Torzillo, G.; Zittelli, G.; Sepúlveda, C.; Masojídek, J. Photobioreactors for the production of microalgae. In Microalgae-Based Biofuels and Bioproducts; Elsevier: Cambridge, UK, 2017; pp. 1–44. [Google Scholar]
- Yen, H.-W.; Chang, J.-T. A two-stage cultivation process for the growth enhancement of Chlorella vulgaris. Bioprocess Biosyst. Eng. 2013, 36, 1797–1801. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Show, P.-L.; Juan, J.C.; Chang, J.-S.; Ling, T.C. Enhancing biomass and lipid productions of microalgae in palm oil mill effluent using carbon and nutrient supplementation. Energy Convers. Manag. 2018, 164, 188–197. [Google Scholar] [CrossRef]
- Caprio, F.D.; Andrea, V.; Altimari, P.; Toro, L.; Masciocchi, B.; Iaquaniello, G.; Pagnanelli, F. Two stage process of microalgae cultivation for starch and carotenoid production. Chem. Eng. Trans. 2016, 49, 415–420. [Google Scholar]
- Ra, C.H.; Kang, C.-H.; Kim, N.K.; Lee, C.-G.; Kim, S.-K. Cultivation of four microalgae for biomass and oil production using a two-stage culture strategy with salt stress. Renew. Energy 2015, 80, 117–122. [Google Scholar] [CrossRef]
- Chen, G.; Zhao, L.; Qi, Y. Enhancing the productivity of microalgae cultivated in wastewater toward biofuel production: A critical review. Appl. Energy 2015, 137, 282–291. [Google Scholar] [CrossRef]
- Bora, A.P.; Gupta, D.P.; Durbha, K.S. Sewage sludge to bio-fuel: A review on the sustainable approach of transforming sewage waste to alternative fuel. Fuel 2020, 259, 116262. [Google Scholar] [CrossRef]
- Xiong, W.; Gao, C.; Yan, N.; Wu, C.; Wu, Q. Double CO2 fixation in photosynthesis–fermentation model enhances algal lipid synthesis for biodiesel production. Bioresour. Technol. 2010, 101, 2287–2293. [Google Scholar] [CrossRef]
- Zhou, W.; Hu, B.; Li, Y.; Min, M.; Mohr, M.; Du, Z.; Chen, P.; Ruan, R. Mass Cultivation of Microalgae on Animal Wastewater: A Sequential Two-Stage Cultivation Process for Energy Crop and Omega-3-Rich Animal Feed Production. Appl. Biochem. Biotechnol. 2012, 168, 348–363. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Min, M.; Li, Y.; Hu, B.; Ma, X.; Cheng, Y.; Liu, Y.; Chen, P.; Ruan, R. A hetero-photoautotrophic two-stage cultivation process to improve wastewater nutrient removal and enhance algal lipid accumulation. Bioresour. Technol. 2012, 110, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Hong, Y.; Liu, P.; Zhan, J.; Yan, R. Effects of cultivation strategies on the cultivation of Chlorella sp. HQ in photoreactors. Front. Environ. Sci. Eng. 2019, 13, 78. [Google Scholar] [CrossRef]
- Jagadevan, S.; Banerjee, C.; Banerjee, C.; Guria, C.; Tiwari, R.; Baweja, M.; Shukla, P. Recent developments in synthetic biology and metabolic engineering in microalgae towards biofuel production. Biotechnol. Biofuels 2018, 11, 1–21. [Google Scholar] [CrossRef]
- Wang, X.-W.; Huang, L.; Ji, P.-Y.; Chen, C.-P.; Li, X.-S.; Gao, Y.-H.; Liang, J.-R. Using a mixture of wastewater and seawater as the growth medium for wastewater treatment and lipid production by the marine diatom Phaeodactylum tricornutum. Bioresour. Technol. 2019, 289, 121681. [Google Scholar] [CrossRef]
- Farooq, W.; Lee, Y.-C.; Ryu, B.-G.; Kim, B.-H.; Kim, H.-S.; Choi, Y.-E.; Yang, J.-W. Two-stage cultivation of two Chlorella sp. strains by simultaneous treatment of brewery wastewater and maximizing lipid productivity. Bioresour. Technol. 2013, 132, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Hena, S.; Fatihah, N.; Tabassum, S.; Ismail, N. Three stage cultivation process of facultative strain of Chlorella sorokiniana for treating dairy farm effluent and lipid enhancement. Water Res. 2015, 80, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zheng, H.; Addy, M.; Anderson, E.; Liu, Y.; Chen, P.; Ruan, R. Cultivation of Chlorella vulgaris in wastewater with waste glycerol: Strategies for improving nutrients removal and enhancing lipid production. Bioresour. Technol. 2016, 207, 252–261. [Google Scholar] [CrossRef]
- Ge, S.; Qiu, S.; Tremblay, D.; Viner, K.; Champagne, P.; Jessop, P.G. Centrate wastewater treatment with Chlorella vulgaris: Simultaneous enhancement of nutrient removal, biomass and lipid production. Chem. Eng. J. 2018, 342, 310–320. [Google Scholar] [CrossRef]
- Johnson, T.J.; Katuwal, S.; Anderson, G.A.; Gu, L.; Zhou, R.; Gibbons, W.R. Photobioreactor cultivation strategies for microalgae and cyanobacteria. Biotechnol. Prog. 2018, 34, 811–827. [Google Scholar] [CrossRef] [PubMed]
- Sirisuk, P.; Ra, C.-H.; Jeong, G.-T.; Kim, S.-K. Effects of wavelength mixing ratio and photoperiod on microalgal biomass and lipid production in a two-phase culture system using LED illumination. Bioresour. Technol. 2018, 253, 175–181. [Google Scholar] [CrossRef]
- Ramanna, L.; Rawat, I.; Bux, F. Light enhancement strategies improve microalgal biomass productivity. Renew. Sustain. Energy Rev. 2017, 80, 765–773. [Google Scholar] [CrossRef]
- Ra, C.H.; Kang, C.-H.; Jung, J.-H.; Jeong, G.-T.; Kim, S.-K. Enhanced biomass production and lipid accumulation of Picochlorum atomus using light-emitting diodes (LEDs). Bioresour. Technol. 2016, 218, 1279–1283. [Google Scholar] [CrossRef]
- Huerlimann, R.; Denys, R.; Heimann, K. Growth, lipid content, productivity, and fatty acid composition of tropical microalgae for scale-up production. Biotechnol. Bioeng. 2010, 107, 245–257. [Google Scholar] [CrossRef]
- Wang, T.; Tian, X.; Liu, T.; Wang, Z.-J.; Guan, W.; Guo, M.; Chu, J.; Zhuang, Y. A two-stage fed-batch heterotrophic culture of Chlorella protothecoides that combined nitrogen depletion with hyperosmotic stress strategy enhanced lipid yield and productivity. Process. Biochem. 2017, 60, 74–83. [Google Scholar] [CrossRef]
- Yoon, S.Y.; Hong, M.E.; Chang, W.S.; Sim, S.J. Enhanced biodiesel production in Neochloris oleoabundans by a semi-continuous process in two stage photobioreactors. Bioprocess Biosyst. Eng. 2015, 38, 1415–1421. [Google Scholar] [CrossRef]
- Nayak, M.; Suh, W.I.; Cho, J.M.; Kim, H.S.; Lee, B.; Chang, Y.K. Strategic implementation of phosphorus repletion strategy in continuous two-stage cultivation of Chlorella sp. HS2: Evaluation for biofuel applications. J. Environ. Manag. 2020, 271, 111041. [Google Scholar] [CrossRef]
- Ghidossi, T.; Marison, I.; Devery, R.; Gaffney, D.; Forde, C. Characterization and Optimization of a Fermentation Process for the Production of High Cell Densities and Lipids Using Heterotrophic Cultivation of Chlorella protothecoides. Ind. Biotechnol. 2017, 13, 253–259. [Google Scholar] [CrossRef]
- Palabhanvi, B.; Muthuraj, M.; Mukherjee, M.; Kumar, V.; Das, D. Process engineering strategy for high cell density-lipid rich cultivation of Chlorella sp. FC2 IITG via model guided feeding recipe and substrate driven pH control. Algal Res. 2016, 16, 317–329. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, T.; Yu, X.; Bates, P.D.; Dong, T.; Chen, S. High-density fed-batch culture of a thermotolerant microalga Chlorella sorokiniana for biofuel production. Appl. Energy 2013, 108, 281–287. [Google Scholar] [CrossRef]
- Muylaert, K.; Bastiaens, L.; Vandamme, D.; Gouveia, L. Harvesting of microalgae: Overview of process options and their strengths and drawbacks. In Microalgae-Based Biofuels and Bioproducts; Elsevier: Cambridge, UK, 2017; pp. 113–132. [Google Scholar]
- Zhang, B.; Chen, S. Effect of different organic matters on flocculation of Chlorella sorokiniana and optimization of flocculation conditions in swine manure wastewater. Bioresour. Technol. 2015, 192, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Chatsungnoen, T.; Chisti, Y. Oil production by six microalgae: Impact of flocculants and drying on oil recovery from the biomass. Environ. Biol. Fishes 2016, 28, 2697–2705. [Google Scholar] [CrossRef]
- Vandamme, D.; Foubert, I.; Fraeye, I.; Muylaert, K. Influence of organic matter generated by Chlorella vulgaris on five different modes of flocculation. Bioresour. Technol. 2012, 124, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Shah, R.; Yadav, P.; Verma, R.; Srivastava, S. Harvesting of freshwater microalgae Scenedesmus sp. by electro–coagulation–flocculation for biofuel production: Effects on spent medium recycling and lipid extraction. Environ. Sci. Pollut. Res. 2019, 27, 3497–3507. [Google Scholar] [CrossRef]
- Fayad, N.; Yehya, T.; Audonnet, F.; Vial, C. Harvesting of microalgae Chlorella vulgaris using electro-coagulation-flocculation in the batch mode. Algal Res. 2017, 25, 1–11. [Google Scholar] [CrossRef]
- Mathimani, T.; Mallick, N. A comprehensive review on harvesting of microalgae for biodiesel—Key challenges and future directions. Renew. Sustain. Energy Rev. 2018, 91, 1103–1120. [Google Scholar] [CrossRef]
- Vashist, V.; Chauhan, D.; Bhattacharya, A.; Rai, M.P. Role of silica coated magnetic nanoparticle on cell flocculation, lipid extraction and linoleic acid production from Chlorella pyrenoidosa. Nat. Prod. Res. 2019, 34, 2852–2856. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Yasin, N.M.; Derek, C.; Lim, J. Comparison of harvesting methods for microalgae Chlorella sp. and its potential use as a biodiesel feedstock. Environ. Technol. 2014, 35, 2244–2253. [Google Scholar] [CrossRef]
- Borges, L.; Caldas, S.; D’Oca, M.G.M.; Abreu, P.C. Effect of harvesting processes on the lipid yield and fatty acid profile of the marine microalga Nannochloropsis oculata. Aquac. Rep. 2016, 4, 164–168. [Google Scholar] [CrossRef]
- Yasin, N.H.M.; Shafei, N.I.; Rushan, N.H.; Abu Sepian, N.R.; Said, F.M. The Effect of Microalgae Harvesting on Lipid for Biodiesel Production. Mater. Today Proc. 2019, 19, 1582–1590. [Google Scholar] [CrossRef]
- Coward, T.; Lee, J.; Caldwell, G.S. Harvesting microalgae by CTAB-aided foam flotation increases lipid recovery and improves fatty acid methyl ester characteristics. Biomass Bioenergy 2014, 67, 354–362. [Google Scholar] [CrossRef]
- Magnusson, M.; Mata, L.; Denys, R.; Paul, N. Biomass, Lipid and Fatty Acid Production in Large-Scale Cultures of the Marine Macroalga Derbesia tenuissima (Chlorophyta). Mar. Biotechnol. 2014, 16, 456–464. [Google Scholar] [CrossRef]
- Sekar, M.; Mathimani, T.; Alagumalai, A.; Chi, N.T.L.; Duc, P.A.; Bhatia, S.K.; Brindhadevi, K.; Pugazhendhi, A. A review on the pyrolysis of algal biomass for biochar and bio-oil-Bottlenecks and scope. Fuel 2021, 283, 119190. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).