Aquaponics (s.l.) Production of Spearmint (Mentha spicata) with African Catfish (Clarias gariepinus) in Northern Germany
Abstract
1. Introduction
2. Materials and Methods
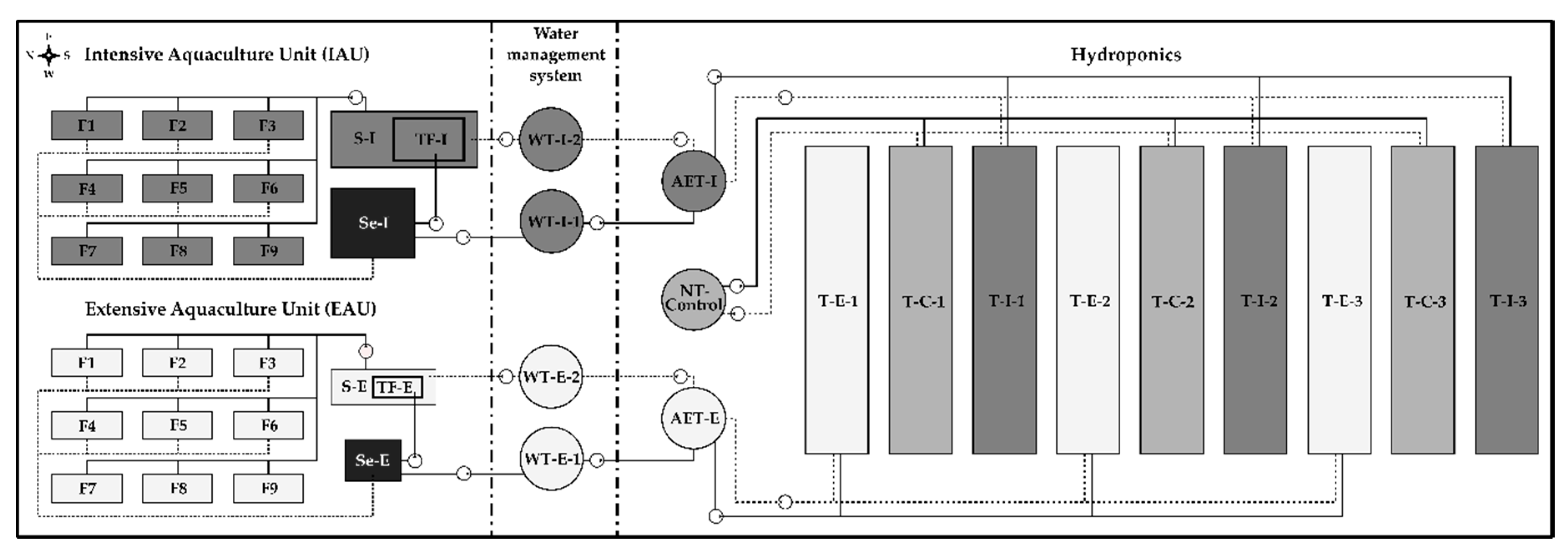
2.1. Experimental Facility and System Design
2.2. Fish Production and Feeding
2.3. Plant Production
2.4. Physical and Chemical Water Parameters
2.5. Mathematical and Statistical Analysis
3. Results
3.1. Fish and Plant Production
3.2. Physical and Chemical Water Parameters
3.3. Nutrient Ratios and Biomass Relations
4. Discussion
4.1. Fish Production
4.2. Mint Production
4.3. Nutrient Ratios and Biomass Relations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Somerville, C.; Cohen, M.; Pantanella, E.; Stankus, A.; Lovatelli, A. Small-Scale Aquaponic Food Production. Integrated Fish and Plant Farming; FAO Fisheries and Aquaculture Technical Paper No. 589; FAO: Rome, Italy, 2014; p. 262. [Google Scholar]
- Rakocy, J.E.; Masser, M.P.; Losordo, T.M. Recirculating Aquaculture Tank Production Systems: Aquaponics—Integrating Fish and Plant Culture. SRAC Publication No. 454, Southern Regional Aquaculture Center, USA 2006. Available online: http://aquaculture.ca.uky.edu/publication/recirculating-aquaculture-tank-production-systems-aquaponics-integrating-fish-and-plant (accessed on 6 October 2020).
- Rakocy, J.E. Chapter 14: Aquaponics—Integrating fish and plant culture. In Aquaculture Production Systems; Tidwell, J.H., Ed.; Wiley-Blackwell: Oxford, UK, 2012; pp. 344–386. [Google Scholar]
- Chauhan, R.S.; Kaul, M.K.; Shahi, A.K.; Kumar, A.; Ram, G.; Tawa, A. Chemical composition of essential oils in Mentha spicata L. accession [IIIM (J) 26] from North-West Himalayan region, India. Ind. Crops Prod. 2009, 29, 654–656. [Google Scholar] [CrossRef]
- Franzios, G.; Mirotsou, M.; Hatziapostolou, E.; Kral, J.; Scouras, Z.G.; Mavragani-Tsipidou, P. Insecticidal and genotoxic activities of mint essential oils. J. Agric. Food Chem. 1997, 45, 2690–2694. [Google Scholar] [CrossRef]
- Sivropoulou, A.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antimicrobial activity of mint essential oils. J. Agric. Food Chem. 1995, 43, 2384–2388. [Google Scholar] [CrossRef]
- Lawrence, B.M. Mint: The Genus Mentha; FL. 33487-2742; CRC Press: Boca Raton, FL, USA, 2006; p. 598. [Google Scholar]
- Tridge Top Producing Countries of Mint. 2020. Available online: https://www.tridge.com/intelligences/mint/production (accessed on 6 October 2020).
- Ross, J. Agricultural Statistics 2018. Table 3-54—Mint Oil: Production and Value, by State and United States, 2015–2017; United States Government Printing Office, National Agricultural Statistics Service, United States Department of Agriculture: Washington, DC, USA, 2018; p. III-26.
- CBI Exporting Fresh Herbs to Europe. CBI, the Centre for the Promotion of Imports from Developing Countries, The Hague the Netherlands. 2020. Available online: https://www.cbi.eu/market-information/fresh-fruit-vegetables/fresh-herbs/europe (accessed on 6 October 2020).
- Gershenzon, J.; Maffei, M.; Croteau, R. Biochemical and histochemical localization of monoterpene biosynthesis in the glandular trichomes of spearmint (Mentha spicata). Plant Physiol. 1989, 89, 1351–1357. [Google Scholar] [CrossRef]
- Tiwari, S. Plants: A rich source of herbal medicine. J. Nat. Prod. 2008, 1, 27–35. [Google Scholar]
- Knaus, U.; Appelbaum, S.; Palm, H.W. Significant factors affecting the economic sustainability of closed backyard aquaponics systems. Part IV: Autumn herbs and polyponics. AACL Bioflux 2018, 11, 1760–1775. [Google Scholar]
- Roosta, H.R.; Ghorbani, F. Investigation of the growth and development, essential oil and minerals content in two species of mint in hydroponics and aquaponics. J. Sci. Technol. Greenh. Cult. 2011, 2, 19–28. [Google Scholar]
- Wahap, N.; Estim, A.; Kian, A.Y.S.; Senoo, S.; Mustafa, S. Producing organic fish and mint in an aquaponic system. Aquaponics J. 2010, 58, 28–32. [Google Scholar]
- Shete, A.P.; Verma, A.K.; Chadha, N.K.; Prakash, C.; Peter, R.M.; Ahmad, I.; Nuwansi, K.K.T. Optimization of hydraulic loading rate in aquaponic system with Common carp (Cyprinus carpio) and Mint (Mentha arvensis). Aquac. Eng. 2016, 72, 53–57. [Google Scholar] [CrossRef]
- Shete, A.P.; Verma, A.K.; Chadha, N.K.; Prakash, C.; Chandrakant, M.H.; Nuwansi, K.K.T. Evaluation of different hydroponic media for mint (Mentha arvensis) with common carp (Cyprinus carpio) juveniles in an aquaponic system. Aquac. Int. 2017, 25, 1291–1301. [Google Scholar] [CrossRef]
- Kotzen, B.; Appelbaum, S. An investigation of aquaponics using brackish water resources in the Negev Desert. J. Appl. Aquac. 2010, 22, 297–320. [Google Scholar] [CrossRef]
- Espinosa Moya, E.A.; Angel Sahagún, C.A.; Mendoza Carrillo, J.M.; Albertos Alpuche, P.J.; Álvarez-González, C.A.; Martínez-Yáñez, R. Herbaceous plants as part of biological filter for aquaponics system. Aquac. Res. 2016, 47, 1716–1726. [Google Scholar] [CrossRef]
- Palm, H.W.; Bissa, K.; Knaus, U. Significant factors affecting the economic sustainability of closed aquaponic systems. Part II: Fish and plant growth. AACL Bioflux 2014, 7, 162–175. [Google Scholar]
- Palm, H.W.; Knaus, U.; Wasenitz, B.; Bischoff, A.A.; Strauch, S.M. Proportional up scaling of African catfish (Clarias gariepinus Burchell, 1822) commercial recirculating aquaculture systems disproportionally affects nutrient dynamics. Aquaculture 2018, 491, 155–168. [Google Scholar] [CrossRef]
- Knaus, U.; Palm, H.W. Effects of fish biology on ebb and flow aquaponical cultured herbs in northern Germany (Mecklenburg Western Pomerania). Aquaculture 2017, 466, 51–63. [Google Scholar] [CrossRef]
- Baßmann, B.; Brenner, M.; Palm, H.W. Stress and welfare of African catfish (Clarias gariepinus Burchell, 1822) in a coupled aquaponic system. Water 2017, 9, 504. [Google Scholar] [CrossRef]
- Pouomogne, V. FAO 2010–2020 Cultured Aquatic Species Information Programme Clarias gariepinus (Burchell, 1822). In FAO Fisheries Division [Online]; FAO: Rome, Italy, 2010; Available online: http://www.fao.org/fishery/culturedspecies/Clarias_gariepinus/en (accessed on 13 October 2020).
- Endut, A.; Jusoh, A.; Ali, N.; Nik, W.W.; Hassan, A. A study on the optimal hydraulic loading rate and plant ratios in recirculation aquaponic system. Bioresour. Technol. 2010, 101, 1511–1517. [Google Scholar] [CrossRef]
- Pantanella, E. Nutrition and Quality of Aquaponic Systems. Ph.D. Thesis, Università degli Studi della Tuscia, Viterbo, Italy, 2012; p. 124. [Google Scholar]
- Palm, H.W.; Knaus, U.; Appelbaum, S.; Goddek, S.; Strauch, S.M.; Vermeulen, T.; Jijakli, M.H.; Kotzen, B. Towards commercial aquaponics: A review of systems, designs, scales and nomenclature. Aquac. Int. 2018, 26, 813–842. [Google Scholar] [CrossRef]
- Palm, H.W.; Strauch, S.M.; Knaus, U.; Wasenitz, B. Das FischGlasHaus—Eine Innovationsinitiative zur energie und nährstoffeffizienten Produktion unterschiedlicher Fisch- und Pflanzenarten in Mecklenburg-Vorpommern (“Aquaponik in MV”). Fischerei Fischmarkt M-V 2016, 1, 38–47. (In German) [Google Scholar]
- Cho, Y.Y.; Oh, S.; Oh, M.M.; Son, J.E. Estimation of individual leaf area, fresh weight, and dry weight of hydroponically grown cucumbers (Cucumis sativus L.) using leaf length, width, and SPAD value. Sci. Hortic. 2007, 111, 330–334. [Google Scholar] [CrossRef]
- Robbins, N.S.; Pharr, D.M. Leaf area prediction models for cucumber from linear measurements. Hortscience 1987, 22, 1264–1266. [Google Scholar]
- Vazquez-Cruz, M.A.; Luna-Rubio, R.; Contreras-Medina, L.M.; Torres-Pacheco, I.; Guevara-Gonzalez, R.G. Estimating the response of tomato (Solanum lycopersicum) leaf area to changes in climate and salicylic acid applications by means of artificial neural networks. Biosyst. Eng. 2012, 112, 319–327. [Google Scholar] [CrossRef]
- Easlon, H.M.; Bloom, A.J. Easy Leaf Area: Automated digital image analysis for rapid and accurate measurement of leaf area. Appl. Plant Sci. 2014, 2, 1400033. [Google Scholar] [CrossRef] [PubMed]
- van de Nieuwegiessen, P.G.; Olwo, J.; Khong, S.; Verreth, J.A.; Schrama, J.W. Effects of age and stocking density on the welfare of African catfish, Clarias gariepinus Burchell. Aquaculture 2009, 288, 69–75. [Google Scholar] [CrossRef]
- Akinwole, A.O.; Faturoti, E.O. Biological performance of African Catfish (Clarias gariepinus) cultured in recirculating system in Ibadan. Aquac. Eng. 2007, 36, 18–23. [Google Scholar] [CrossRef]
- Leenhouwers, J.I.; ter Veld, M.; Verreth, J.A.; Schrama, J.W. Digesta characteristiscs and performance of African catfish (Clarias gariepinus) fed cereal grains that differ in viscosity. Aquaculture 2007, 264, 330–341. [Google Scholar] [CrossRef]
- Mota, V.C.; Limbu, P.; Martins, C.I.; Eding, E.H.; Verreth, J.A. The effect of nearly closed RAS on the feed intake and growth of Nile tilapia (Oreochromis niloticus), African catfish (Clarias gariepinus) and European eel (Anguilla anguilla). Aquac. Eng. 2015, 68, 1–5. [Google Scholar] [CrossRef]
- Hogendoorn, H.; Jansen, J.A.J.; Koops, W.J.; Machiels, M.A.M.; Van Ewijk, P.H.; Van Hees, J.P. Growth and production of the African catfish, Clarias lazera (C. & V.): II. Effects of body weight, temperature and feeding level in intensive tank culture. Aquaculture 1983, 34, 265–285. [Google Scholar] [CrossRef]
- Fagbenro, O.A. Comparative evaluation of heat-processed winged bean (Psophocarpus tetragonolobus) meals as partial replacement for fish meal in diets for the African catfish (Clarias gariepinus). Aquaculture 1999, 170, 297–305. [Google Scholar] [CrossRef]
- Oellermann, L.K. A Comparison of the Aquaculture Potential of Clarias gariepinus (Burchell, 1822) and Its Hybrid with Heterobranchus longifilis Valenciennes, 1840 in Southern Africa. Ph.D. Thesis, Rhodes University, Grahamstown, South Africa, 1995; p. 152. [Google Scholar]
- Britz, P.J. Environmental Requirements for the Hatchery Rearing of African Catfish Clarias gariepinus (Pisces: Clariidae) Larvae and Juveniles. Master’s Thesis, Rhodes University, Grahamstown, South Africa, 1988; p. 123. [Google Scholar]
- Schram, E.; Roques, J.A.C.; Abbink, W.; Spanings, T.; de Vries, P.; Bierman, S.; van de Vis, H.; Flik, G. The impact of elevated water ammonia concentration on physiology, growth and feed intake of African catfish (Clarias gariepinus). Aquaculture 2010, 306, 108–115. [Google Scholar] [CrossRef]
- Roques, J.A.C.; Schram, E.; Spanings, T.; van Schaik, T.; Abbink, W.; Boerrigter, J.; de Vries, P.; van de Vis, H.; Flik, G. The impact of elevated water nitrite concentration on physiology, growth and feed intake of African catfish Clarias gariepinus (Burchell 1822). Aquac. Res. 2015, 46, 1384–1395. [Google Scholar] [CrossRef]
- Schram, E.; Roques, J.A.C.; Abbink, W.; Yokohama, Y.; Spanings, T.; de Vries, P.; Bierman, S.; van de Vis, H.; Flik, G. The impact of elevated water nitrate concentration on physiology, growth and feed intake of African catfish Clarias gariepinus (Burchell 1822). Aquac. Res. 2014, 45, 1499–1511. [Google Scholar] [CrossRef]
- Huisman, E.A.; Richter, C.J.J. Reproduction, growth, health control and aquacultural potential of the African catfish, Clarias gariepinus (Burchell 1822). Aquaculture 1987, 63, 1–14. [Google Scholar] [CrossRef]
- Patel, A.; Patra, D.D. Effect of tannery sludge amended soil on glutathione activity of four aromatic crops: Tagetes minuta, Pelargonium graveolens, Ocimum basilicum and Mentha spicata. Ecol. Eng. 2015, 81, 348–352. [Google Scholar] [CrossRef]
- Savidov, N. Comparative study of aquaponically and hydroponically grown plants in model system. In Evaluation and Development of Aquaponics Production and Product Market Capabilities in Alberta; Chapter 3.2., Phase II; Crop Diversification Centre: South Brooks, NL, Canada, 2005; pp. 21–31. [Google Scholar]
- Lehmonen, R.; Sireeni, J. Comparison of Plant Growth and Quality in Hydroponic and Aquaponic Systems. Bachelor’s Thesis, University of Jyväskylä, Jyväskylä, Finland, 2017; p. 27. [Google Scholar]
- Chrysargyris, A.; Xylia, P.; Botsaris, G.; Tzortzakis, N. Antioxidant and antibacterial activities, mineral and essential oil composition of spearmint (Mentha spicata L.) affected by the potassium levels. Ind. Crops Prod. 2017, 103, 202–212. [Google Scholar] [CrossRef]
- Vocke, G. Das Große Buch der Heilpflanzen: Die 110 Wichtigsten Heilpflanzen von A bis Z, mit Tipps für Anbau, Pflege und Verwendung; Compact-Verlag: Munich, Germany, 2002; p. 144. (In German) [Google Scholar]
- Tabatabaie, S.J.; Nazari, J.; Nazemiyeh, H.; Zehtab, S.; Azarmi, F. Influence of various electrical conductivity levels on the growth and essential oil content of peppermint (Menta piperita L.) grown in hydroponic. Acta Hortic. 2007, 747, 197–201. [Google Scholar] [CrossRef]
- Bittsanszky, A.; Uzinger, N.; Gyulai, G.; Mathis, A.; Junge, R.; Villarroel, M.; Kotzen, B.; Komives, T. Nutrient supply of plants in aquaponic systems. Ecocycles 2016, 2, 17–20. [Google Scholar] [CrossRef]
- Knaus, U.; Palm, H.W. Effects of the fish species choice on vegetables in aquaponics under spring-summer conditions in northern Germany (Mecklenburg Western Pomerania). Aquaculture 2017, 473, 62–73. [Google Scholar] [CrossRef]
- Nozzi, V.; Graber, A.; Schmautz, Z.; Mathis, A.; Junge, R. Nutrient management in aquaponics: Comparison of three approaches for cultivating lettuce, mint and mushroom herb. Agronomy 2018, 8, 27. [Google Scholar] [CrossRef]







| Parameters | Extensive (EAU) | Intensive (IAU) | p | ||||
|---|---|---|---|---|---|---|---|
| Initial Parameters | |||||||
| Tanks | 1, 4, 7 | 2, 5, 8 | 3, 6, 9 | 1, 4, 7 | 2, 5, 8 | 3, 6, 9 | - |
| Date of Stocking | 28 September 2015 | 17 August 2015 | 17 July 2015 | 28 September 2015 | 17 August 2015 | 17 July 2015 | - |
| Day of Grow Out (d) 1 | 74 | 32 | 1 | 74 | 32 | 1 | - |
| Individual Weight (g) | 275.0 | 275.0 | 275.0 | 275.0 | 275.0 | 275.0 | - |
| Fish Number (no) | 105 | 105 | 105 | 420 | 420 | 420 | - |
| Biomass Weight (kg) | 28.9 | 28.9 | 28.9 | 115.5 | 115.5 | 115.5 | - |
| Stocking Density (kg/m3) | 7.6 | 7.6 | 7.6 | 30.6 | 30.6 | 30.6 | - |
| Final Parameters | |||||||
| Date of Harvest | 29 March 2016 | 26 January 2016 | 24 November 2015 | 29 March 2016 | 26 January 2016 | 24 November 2015 | - |
| Day of Grow Out (d) 2 | 257 | 194 | 131 | 257 | 194 | 131 | - |
| Duration Grow Out (d) | 156.3 ± 23.0 a | 156.3 ± 23.0 a | 1.000 | ||||
| Individual Weight (g) | 1526.6 ± 284.0 a | 1458.8 ± 222.5 a | 0.436 | ||||
| Mean Fish Number (no) | 33.7 ± 2.2 b | 127.2 ± 8.8 a | 0.001 | ||||
| Survival (%) | 96.2 ± 6.4 a | 89.8 ± 5.3 b | 0.024 | ||||
| Stocking Density (kg/m3) | 40.6 ± 6.6 b | 146.8 ± 20.6 a | 0.001 | ||||
| Total Weight (kg/tank) | 41.5 ± 8.3 b | 146.4 ± 25.9 a | 0.001 | ||||
| Feed Input (kg/tank) | 38.6 ± 6.9 b | 141.2 ± 27.1 a | 0.001 | ||||
| FCR | 0.94 ± 0.04 a | 0.96 ± 0.04 a | 0.164 | ||||
| SGR (% d−1) | 1.09 ± 0.06 a | 1.07 ± 0.08 a | 0.545 | ||||
| Extensive (EAU) | Intensive (IAU) | |||||
|---|---|---|---|---|---|---|
| Tanks | 1, 4, 7 | 2, 5, 8 | 3, 6, 9 | 1, 4, 7 | 2, 5, 8 | 3, 6, 9 |
| Total Fish Biomass (kg) | ||||||
| Beginning of the Experiment | 0 | 37 | 43 | 0 | 146 | 174 |
| Day 74 before Stocking Tanks 1, 4, 7 | 0 | 68 | 75 | 0 | 242 | 270 |
| Day 74 after Stocking Ranks 1, 4, 7 | 29 | 68 | 75 | 116 | 242 | 270 |
| Parameters | Fertilizer (Control) | Extensive (EAU) | Intensive (IAU) | p-I 1 | p-II 1 | p-III 1 |
|---|---|---|---|---|---|---|
| Initial Parameters | ||||||
| Initial Biomass (g/plant) | 2.7 ± 1.0 a | 2.3 ± 0.6 a | 2.8 ± 0.8 a | 0.831 | 0.979 | 0.725 |
| Plant Height (cm) | 8.5 ± 1.7 a | 9.7 ± 2.0 a | 11.2 ± 2.4 a | 0.558 | 0.558 | 0.558 |
| Leaf Length (cm) | 3.4 ± 0.6 a | 4.0 ± 0.5 a | 3.7 ± 1.5 a | 0.773 | 0.956 | 0.912 |
| Leaf Width (cm) | 2.2 ± 0.3 a | 2.3 ± 0.3 a | 2.4 ± 0.7 a | 0.961 | 0.812 | 0.933 |
| Final Parameters | ||||||
| Plant Height (cm) | 38.4 ± 8.2 b | 45.2 ± 11.5 a | 51.0 ± 16.9 a | 0.009 | 0.001 | 0.109 |
| Shoot Number (no) | 1.6 ± 0.7 b | 1.6 ± 0.6 b | 2.3 ± 1.2 a | 0.848 | 0.004 | 0.002 |
| Leaf Area (cm2) | 5.7 ± 2.2 b | 5.6 ± 2.1 b | 10.9 ± 2.5 a | 0.977 | 0.001 | 0.001 |
| Leaf Length (cm) | 5.5 ± 1.4 b | 5.4 ± 1.4 b | 8.6 ± 1.6 a | 0.997 | 0.001 | 0.001 |
| Fresh Biomass (g/plant) | 120.6 ± 51.8 b | 165.5 ± 71.7 a | 190.7 ± 105.6 a | 0.034 | 0.003 | 0.621 |
| Total Fresh Biomass (kg) * | 50.7 | 69.5 | 80.1 | - | - | - |
| Fresh Cut Biomass (g/plant) ** | 11.2 ± 5.3 c | 17.4 ± 4.7 b | 31.8 ± 13.8 a | 0.006 | 0.001 | 0.005 |
| Total Fresh Cut Biomass (kg) * | 4.7 | 7.3 | 13.4 | - | - | - |
| Dry Biomass (g/plant) | 18.3 ± 7.4 a | 23.6 ± 12.4 a | 21.3 ± 14.2 a | 0.375 | 0.375 | 0.375 |
| Parameters | Extensive (EAU) | Intensive (IAU) | p-Value |
|---|---|---|---|
| O2 (mg/L) | 7.1 ± 0.6 a | 4.9 ± 1.7 b | 0.001 |
| O2 (%) | 88.6 ± 7.9 a | 60.1 ± 20.4 b | 0.001 |
| Temperature (°C) | 26.7 ± 0.2 a | 25.8 ± 0.8 b | 0.001 |
| pH | 7.2 ± 0.5 a | 6.6 ± 0.2 b | 0.001 |
| Salinity (‰) | 0.5 ± 0.1 b | 0.6 ± 0.1 a | 0.001 |
| Conductivity (µS/cm) | 952.3 ± 87.2 b | 1247.7 ± 128.9 a | 0.001 |
| Redox Potential (mV) | 122.6 ± 32.4 a | 131.0 ± 26.7 a | 0.241 |
| NH4+–N (mg/L) | 0.38 ± 0.23 b | 16.69 ± 5.11 a | 0.001 |
| NO2−–N (mg/L) | 0.29 ± 0.17 a | 0.48 ± 0.54 a | 0.842 |
| TON (mg/L) | 44.49 ± 8.73 b | 76.50 ± 12.53 a | 0.001 |
| NO3−–N (mg/L) | 44.20 ± 8.69 b | 76.02 ± 12.37 a | 0.001 |
| TDN (mg/L) | 44.87 ± 8.82 b | 93.19 ± 14.04 a | 0.001 |
| PO43−–P (mg/L) | 3.06 ± 1.20 b | 7.06 ± 1.11 a | 0.001 |
| Parameters | Fertilizer (Control) | Extensive (EAU) | Intensive (IAU) | p-I * | p-II * | p-III * |
|---|---|---|---|---|---|---|
| O2 (mg/L) | 8.3 ± 0.8 a | 5.1 ± 1.2 b | 2.8 ± 1.7 c | 0.001 | 0.001 | 0.001 |
| O2 (%) | 87.4 ± 12.1 a | 56.1 ± 13.0 b | 31.2 ± 18.6 c | 0.001 | 0.001 | 0.001 |
| Temperature (°C) | 18.9 ± 2.0 b | 20.9 ± 2.6 a | 20.8 ± 2.7 a | 0.001 | 0.001 | 0.900 |
| pH | 8.1 ± 0.2 a | 7.3 ± 0.3 b | 7.4 ± 0.3 b | 0.001 | 0.001 | 0.622 |
| Salinity (‰) | 0.4 ± 0.0 c | 0.5 ± 0.1 b | 0.6 ± 0.1 a | 0.001 | 0.001 | 0.001 |
| Conductivity (µS/cm) | 673.7 ± 120.1 c | 821.0 ± 149.3 b | 1095.2 ± 122.4 a | 0.001 | 0.001 | 0.001 |
| Redox Potential (mV) | 117.1 ± 102.4 a,b | 142.1 ± 136.9 a | 100.6 ± 46.1 b | 0.130 | 1.000 | 0.042 |
| NH4⁺–N (mg/L) | 0.24 ± 0.31 c | 2.11 ± 3.44 b | 20.46 ± 6.53 a | 0.001 | 0.001 | 0.001 |
| NO2−–N (mg/L) | 0.52 ± 1.11 b | 0.55 ± 0.30 a | 0.60 ± 0.49 a | 0.001 | 0.001 | 0.843 |
| TON (mg/L) | 7.55 ± 2.12 b | 40.59 ± 12.40 a | 43.99 ± 17.94 a | 0.001 | 0.001 | 0.469 |
| NO3−–N (mg/L) | 7.03 ± 1.95 b | 40.04 ± 12.38 a | 43.38 ± 17.85 a | 0.001 | 0.001 | 0.444 |
| TDN (mg/L) | 7.79 ± 2.14 c | 42.70 ± 13.92 b | 64.45 ± 20.55 a | 0.001 | 0.001 | 0.001 |
| PO43−–P (mg/L) | 3.44 ± 1.05 b | 3.30 ± 1.16 b | 7.98 ± 1.69 a | 0.392 | 0.001 | 0.001 |
| N1/P2 | 2.4 ± 0.7 c | 13.4 ± 3.1 a | 8.2 ± 2.3 b | 0.001 | 0.001 | 0.001 |
| Leaf Area/N 1 | 0.7 ± 0.3 a | 0.2 ± 0.1 b | 0.2 ± 0.1 b | 0.001 | 0.001 | 0.356 |
| Leaf Area/P 2 | 1.6 ± 0.8 a | 2.2 ± 1.5 a | 1.6 ± 0.7 a | 0.148 | 0.148 | 0.148 |
| Fresh Biomass/Leaf Area | 23.0 ± 10.5 a,b | 29.2 ± 19.8 a | 16.9 ± 11.0 b | 0.313 | 0.069 | 0.009 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knaus, U.; Wenzel, L.C.; Appelbaum, S.; Palm, H.W. Aquaponics (s.l.) Production of Spearmint (Mentha spicata) with African Catfish (Clarias gariepinus) in Northern Germany. Sustainability 2020, 12, 8717. https://doi.org/10.3390/su12208717
Knaus U, Wenzel LC, Appelbaum S, Palm HW. Aquaponics (s.l.) Production of Spearmint (Mentha spicata) with African Catfish (Clarias gariepinus) in Northern Germany. Sustainability. 2020; 12(20):8717. https://doi.org/10.3390/su12208717
Chicago/Turabian StyleKnaus, Ulrich, Lisa Carolina Wenzel, Samuel Appelbaum, and Harry Wilhelm Palm. 2020. "Aquaponics (s.l.) Production of Spearmint (Mentha spicata) with African Catfish (Clarias gariepinus) in Northern Germany" Sustainability 12, no. 20: 8717. https://doi.org/10.3390/su12208717
APA StyleKnaus, U., Wenzel, L. C., Appelbaum, S., & Palm, H. W. (2020). Aquaponics (s.l.) Production of Spearmint (Mentha spicata) with African Catfish (Clarias gariepinus) in Northern Germany. Sustainability, 12(20), 8717. https://doi.org/10.3390/su12208717







