Life Cycle Assessment (LCA) Proves that Manila Clam Farming (Ruditapes Philippinarum) is a Fully Sustainable Aquaculture Practice and a Carbon Sink
Abstract
1. Introduction
2. Materials and Methods
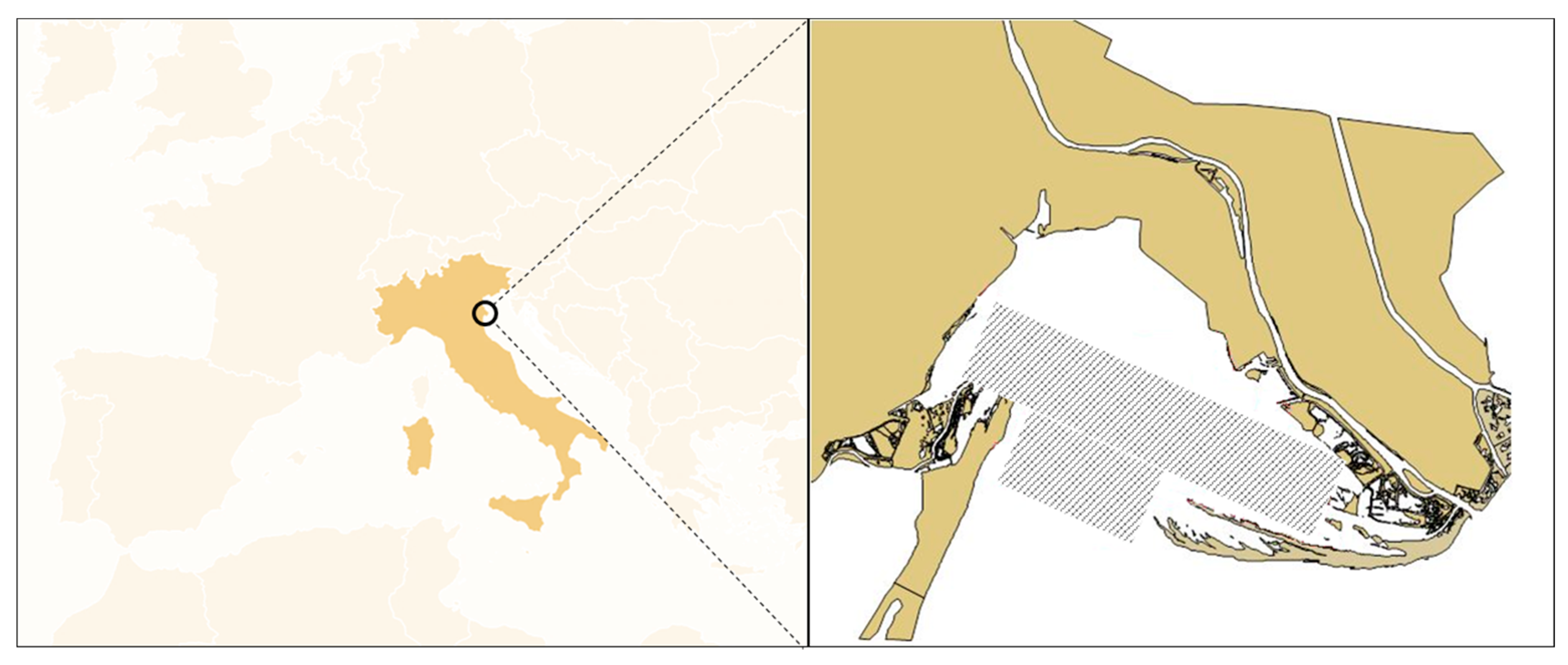
2.1. Study Area
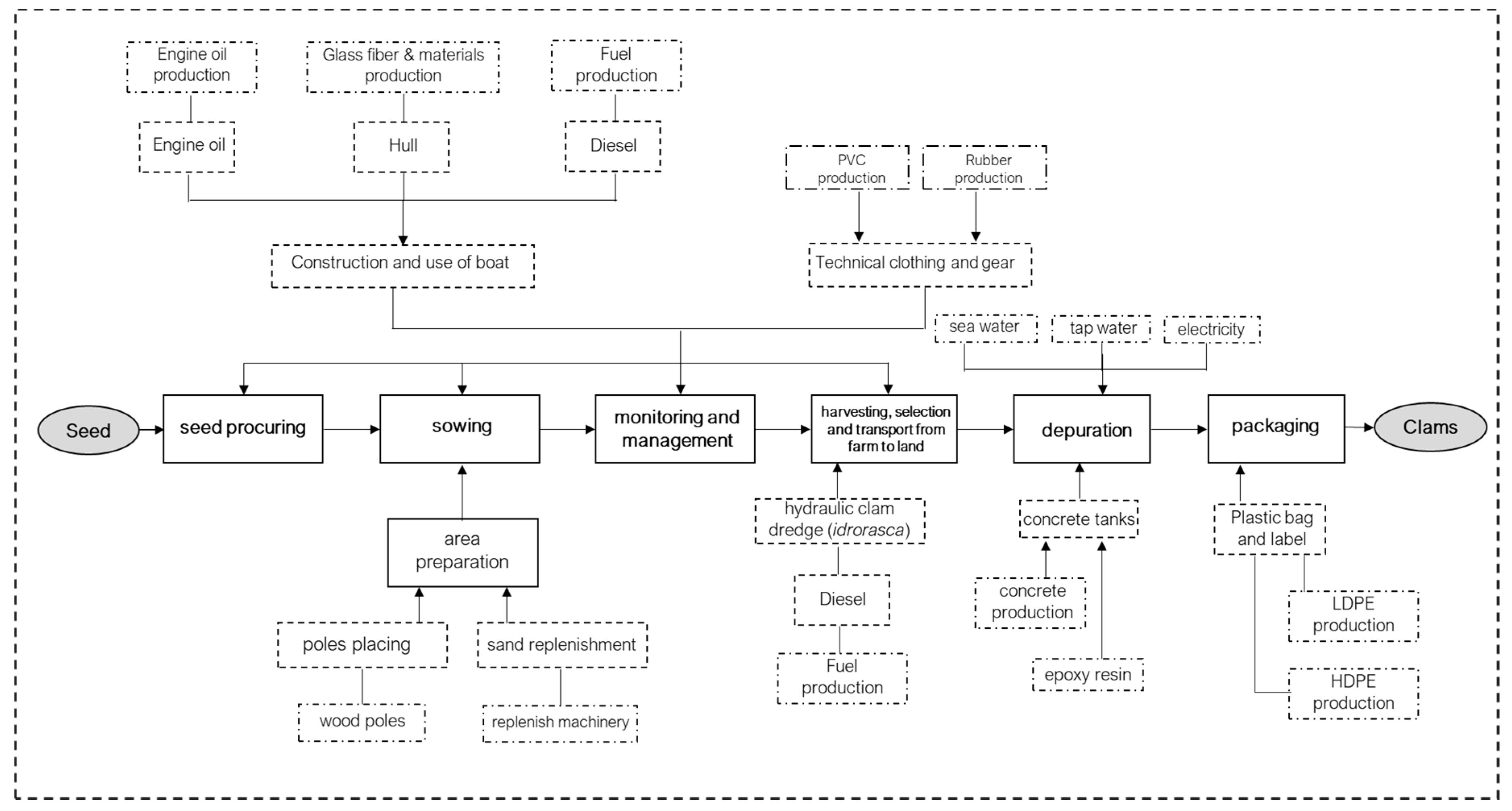
2.2. LCA Goal and Scope, System Boundaries and Functional Unit
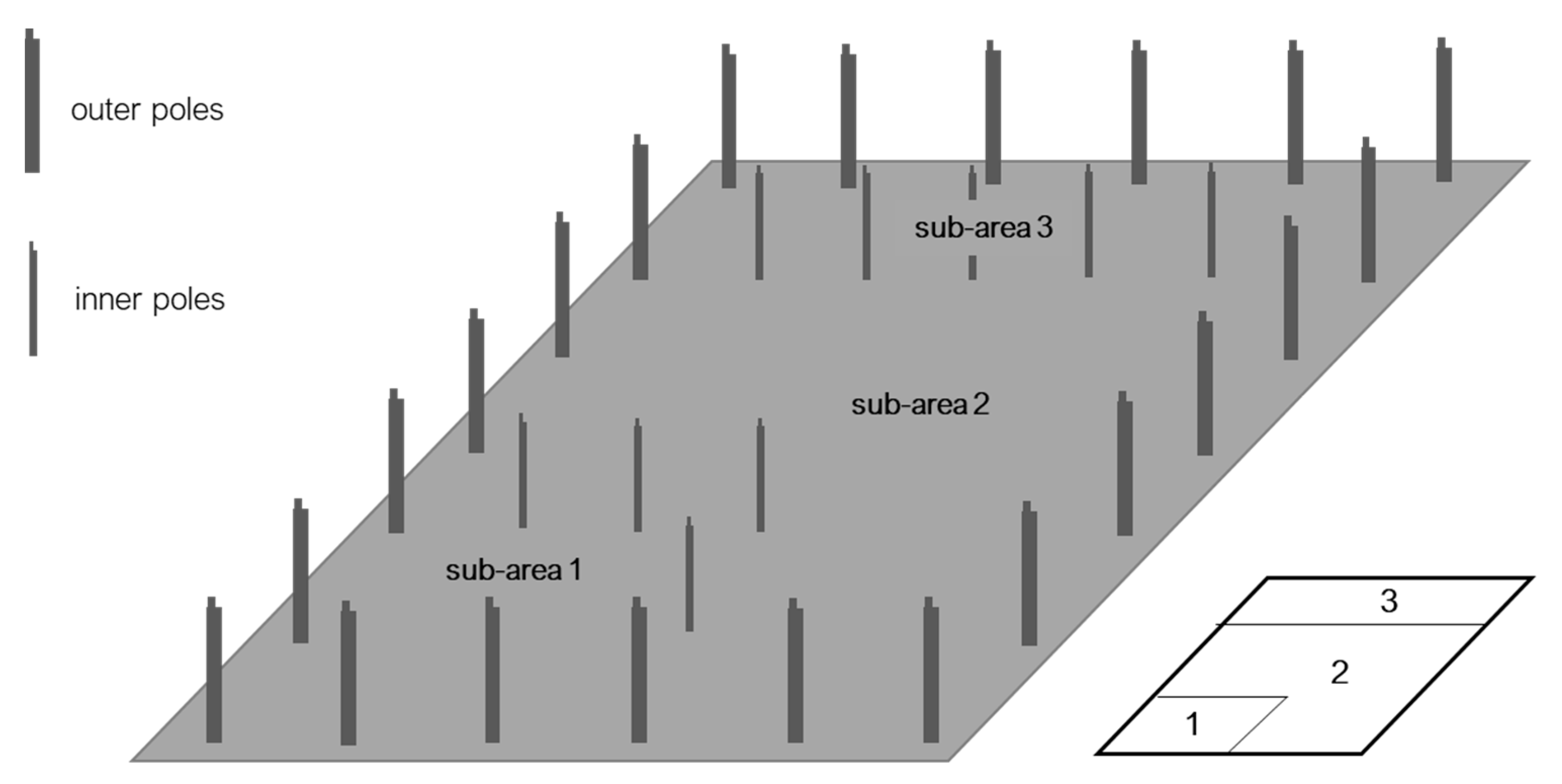
2.3. Life Cycle Inventory (LCI)
2.4. Elemental Balance (Carbon, Nitrogen and Phosphorous)
2.5. Life Cycle Impact Assessment (LCIA)
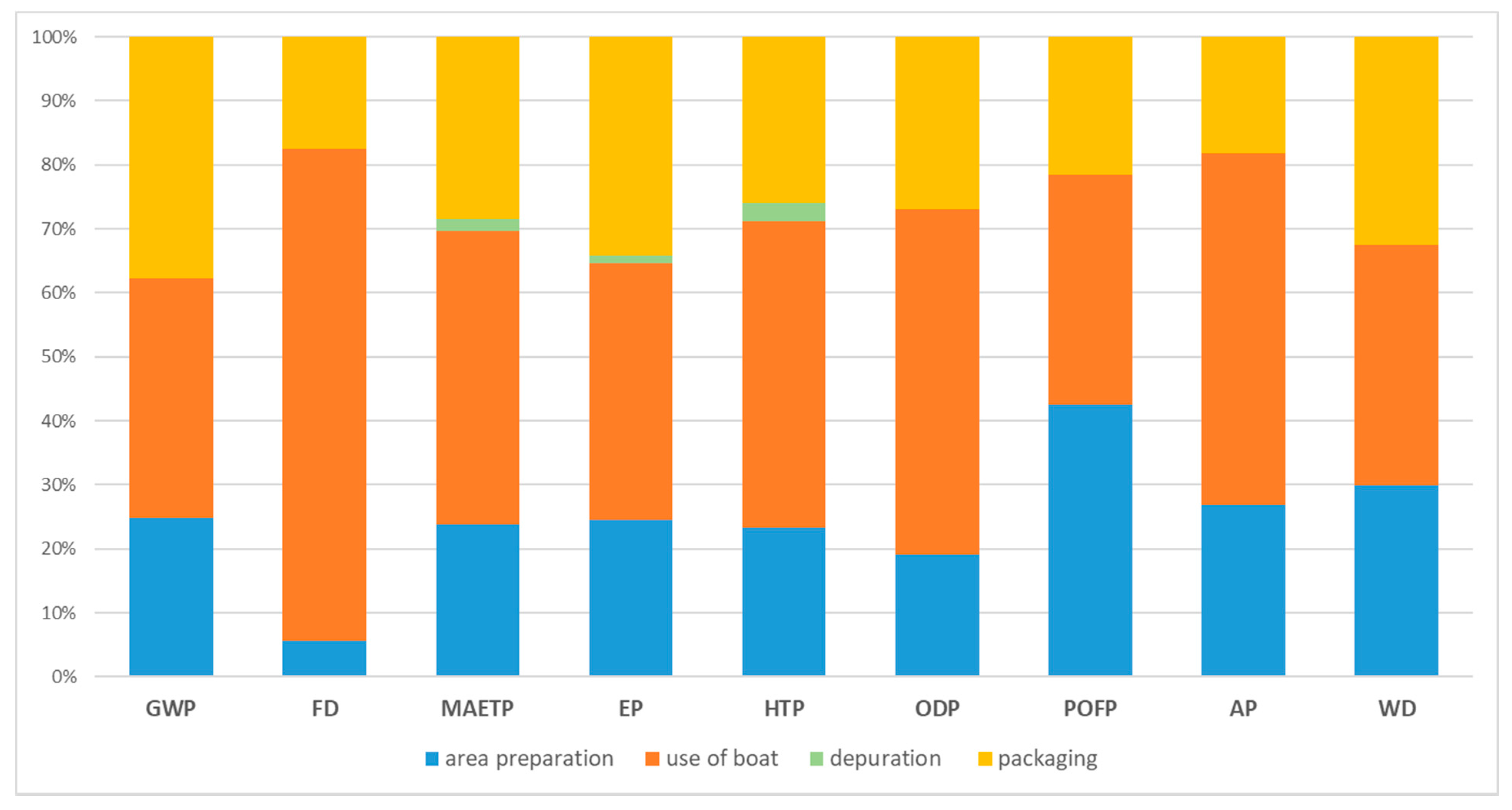
3. Results and Discussion
Clam Aquaculture as Net Carbon Sink
Author Contributions
Funding
Conflicts of Interest
References
- Shumway, S.E.; Davis, C.; Downey, R.; Karney, R.; Kraeuter, J.; Parsons, J.; Wikfors, G. Shellfish aquaculture–in praise of sustainable economies and environments. World Aquac. 2003, 34, 8–10. [Google Scholar]
- Gibbs, M.T. Sustainability performance indicators for suspended bivalve aquaculture activities. Ecol. Ind. 2007, 7, 94–107. [Google Scholar] [CrossRef]
- Froehlich, H.E.; Gentry, R.R.; Halpern, B.S. Global change in marine aquaculture production potential under climate change. Nat. Ecol. Evol. 2018, 2, 1745–1750. [Google Scholar] [CrossRef] [PubMed]
- Murray, G.; D’Anna, L. Seeing shellfish from the seashore: The importance of values and place in perceptions of aquaculture and marine social–ecological system interactions. Mar. Policy 2015, 62, 125–133. [Google Scholar] [CrossRef]
- Gallardi, D. Effects of bivalve aquaculture on the environment and their possible mitigation: A review. Fish. Aquac. J. 2014, 105, 1–8. [Google Scholar] [CrossRef]
- Smaal, A.C.; Ferreira, J.G.; Grant, J.; Petersen, J.K.; Strand, Ø. (Eds.) Goods and Services of Marine Bivalves; Springer Nature Switzerland AG: Cham, Swizerland, 2019. [Google Scholar]
- Van der Schatte Olivier, A.; Jones, L.; Vay, L.L.; Christie, M.; Wilson, J.; Malham, S.K. A global review of the ecosystem services provided by bivalve aquaculture. Rev. Aquacul. 2020, 12, 3–25. [Google Scholar] [CrossRef]
- Ahmed, N.; Thompson, S. The blue dimensions of aquaculture: A global synthesis. Sci. Total Environ. 2019, 652, 851–861. [Google Scholar] [CrossRef]
- Little, D.C.; Young, J.A.; Zhang, W.; Newton, R.W.; Al Mamun, A.; Murray, F.J. Sustainable intensification of aquaculture value chains between Asia and Europe: A framework for understanding impacts and challenges. Aquaculture 2018, 493, 338–354. [Google Scholar] [CrossRef]
- Jennings, S.; Stentiford, G.D.; Leocadio, A.M.; Jeffery, K.R.; Metcalfe, J.D.; Katsiadaki, I.; Peeler, E.J. Aquatic food security: Insights into challenges and solutions from an analysis of interactions between fisheries, aquaculture, food safety, human health, fish and human welfare, economy and environment. Fish Fish. 2018, 17, 89–938. [Google Scholar] [CrossRef]
- Tufvesson, L.M.; Tufvesson, P.; Woodley, J.M.; Börjesson, P. Life cycle assessment in green chemistry: Overview of key parameters and methodological concerns. Int. J. Life Cycle Assess. 2013, 18, 431–444. [Google Scholar]
- Notarnicola, B.; Sala, S.; Anton, A.; McLaren, S.J.; Saouter, E.; Sonesson, U. The role of life cycle assessment in supporting sustainable agri-food systems: A review of the challenges. J. Clean. Prod. 2017, 140, 399–409. [Google Scholar] [CrossRef]
- Tamburini, E.; Pedrini, P.; Marchetti, M.G.; Fano, E.A.; Castaldelli, G. Life cycle based evaluation of environmental and economic impacts of agricultural productions in the Mediterranean area. Sustainability 2015, 7, 2915–2935. [Google Scholar] [CrossRef]
- Avadí, A.; Fréon, P. Life cycle assessment of fisheries: A review for fisheries scientists and managers. Fish. Res. 2013, 143, 21–38. [Google Scholar] [CrossRef]
- Iribarren, D.; Moreira, M.T.; Feijoo, G. Revisiting the life cycle assessment of mussels from a sectorial perspective. J. Clean. Prod. 2010, 18, 101–111. [Google Scholar] [CrossRef]
- Iribarren, D.; Moreira, M.T.; Feijoo, G. Life Cycle Assessment of fresh and canned mussel processing and consumption in Galicia (NW Spain). Resour. Conserv. Recy. 2010, 55, 106–117. [Google Scholar] [CrossRef]
- De Alvarenga, R.A.F.; Galindro, B.M.; de Fátima Helpa, C.; Soares, S.R. The recycling of oyster shells: An environmental analysis using Life Cycle Assessment. J. Environ. Manag. 2012, 106, 102–109. [Google Scholar] [CrossRef]
- Aubin, J.; Fontaine, C.; Callier, M.; Roque d’Orbcastel, E. Blue mussel (Mytilus edulis) bouchot culture in Mont-St Michel Bay: Potential mitigation effects on climate change and eutrophication. Int. J. Life Cycle Assess. 2018, 23, 1030–1041. [Google Scholar] [CrossRef]
- Lourguioui, H.; Brigolin, D.; Boulahdid, M.; Pastres, R. A perspective for reducing environmental impacts of mussel culture in Algeria. Int. J. Life Cycle Assess. 2017, 22, 1266–1277. [Google Scholar] [CrossRef]
- Tamburini, E.; Fano, E.A.; Castaldelli, G.; Turolla, E. Life Cycle Assessment of Oyster Farming in the Po Delta, Northern Italy. Resources 2019, 8, 170. [Google Scholar] [CrossRef]
- Tamburini, E.; Turolla, E.; Fano, E.A.; Castaldelli, G. Sustainability of Mussel (Mytilus galloprovincialis) Farming in the Po River Delta, Northern Italy, Based on a Life Cycle Assessment Approach. Sustainability 2020, 12, 3814. [Google Scholar] [CrossRef]
- Çolakoğlu, S.; Palaz, M. Some population parameters of Ruditapes philippinarum (Bivalvia, Veneridae) on the southern coast of the Marmara Sea, Turkey. Helgol. Mar. Res. 2014, 68, 539–548. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals, CC BY-NC-SA 3.0 IGO; Licence: Rome, Italy, 2018. [Google Scholar]
- Cordero, D.; Delgado, M.; Liu, B.; Ruesink, J.; Saavedra, C. Population genetics of the Manila clam (Ruditapes philippinarum) introduced in North America and Europe. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Lin, Z. Development of Manila clam industry in China. Bull. Jpn. Fish. Res. Educ. Agen. 2016, 42, 29–34. [Google Scholar]
- Sladonja, B.; Bettoso, N.; Zentilin, A.; Tamberlich, F.; Acquavita, A. Manila Clam (Tapes Philippinarum Adams & Reeve, 1852) in the Lagoon of Marano and Grado (Northern Adriatic Sea, Italy): Socio Economic and Environmental Pathway of a Shell Farm. Aquaculture and the Environment—A Shared Destiny; Barbara, S., Ed.; InTech Publisher: London, UK, 2011; pp. 51–78. [Google Scholar]
- Chew, K.K. Recent advances in the cultivation of molluscs in the Pacific United States and Canada. Aquaculture 1984, 39, 69–81. [Google Scholar] [CrossRef]
- Humphreys, J.; Caldow, R.W.; McGrorty, S.; West, A.D.; Jensen, A.C. Population dynamics of naturalised Manila clams Ruditapes philippinarum in British coastal waters. Mar. Biol. 2007, 151, 2255–2270. [Google Scholar] [CrossRef]
- Bartoli, M.; Castaldelli, G.; Nizzoli, D.; Fano, E.A.; Viaroli, P. Manila clam introduction in the Sacca di Goro Lagoon (Northern Italy): Ecological implications. Bull. Jpn. Fish. Res. Educ. Agen. 2016, 42, 43–52. [Google Scholar]
- Solidoro, C.; Pastres, R.; Canu, D.M.; Pellizzato, M.; Rossi, R. Modelling the growth of Tapes philippinarum in Northern Adriatic lagoons. Mar. Ecol. Prog. Ser. 2000, 199, 137–148. [Google Scholar] [CrossRef]
- Vincenzetti, S.; Felici, A.; Ciarrocchi, G.; Pucciarelli, S.; Ricciutelli, M.; Ariani, A.; Polidori, P. Comparative proteomic analysis of two clam species: Chamelea gallina and Tapes philippinarum. Food Chem. 2017, 219, 223–229. [Google Scholar] [CrossRef]
- Nizzoli, D.; Bartoli, M.; Viaroli, P. Nitrogen and phosphorous budgets during a farming cycle of the Manila clam Ruditapes philippinarum: An in situ experiment. Aquaculture 2006, 261, 98–108. [Google Scholar] [CrossRef]
- Malta, E.J.; Stigter, T.Y.; Pacheco, A.; Dill, A.C.; Tavares, D.; Santos, R. Effects of external nutrient sources and extreme weather events on the nutrient budget of a southern European coastal lagoon. Estuar. Coast 2017, 40, 419–436. [Google Scholar] [CrossRef]
- ISTAT (Italian National Institute of Statistics). Available online: https://www.istat.it/it/archivio/pesca (accessed on 31 May 2020).
- Marinov, D.; Galbiati, L.; Giordani, G.; Viaroli, P.; Norro, A.; Bencivelli, S.; Zaldívar, J.M. An integrated modelling approach for the management of clam farming in coastal lagoons. Aquaculture 2007, 269, 306–320. [Google Scholar] [CrossRef]
- Ecoinvent Database®. Available online: https://www.ecoinvent.org/database/database.html (accessed on 11 May 2020).
- GSE (Gestore dei Servizi Energetici S.p.A). Rapporto Statistico 2018. Available online: http://enerweb.casaccia.enea.it/enearegioni/UserFiles/GSE_Rapporto_Statistico_FER_2018.pdf (accessed on 15 March 2020).
- Nizzoli, D.; Welsh, D.T.; Fano, E.A.; Viaroli, P. Impact of clam and mussel farming on benthic metabolism and nitrogen cycling, with emphasis on nitrate reduction pathways. Mar. Ecol. Prog. Ser. 2006, 315, 151–165. [Google Scholar] [CrossRef]
- Zan, X.; Xu, B.; Zhang, C.; Ren, Y. Annual variations of biogenic element contents of manila clam (Ruditapes philippinarum) bottom-cultivated in Jiaozhou Bay, China. J. Ocean Univ. China 2014, 13, 637–646. [Google Scholar] [CrossRef]
- Melzner, F.; Stange, P.; Trübenbach, K.; Thomsen, J.; Casties, I.; Panknin, U.; Gutowska, M.A. Food supply and seawater pCO2 impact calcification and internal shell dissolution in the blue mussel Mytilus edulis. PLoS ONE 2011, 6, e24223. [Google Scholar] [CrossRef]
- Frankignoulle, M.; Canon, C.; Gattuso, J.P. Marine calcification as a source of carbon dioxide: Positive feedback of increasing atmospheric CO2. Limnol. Oceanogr. Meth. 1994, 39, 458–462. [Google Scholar] [CrossRef]
- Ray, N.E.; O’Meara, T.; Wiliamson, T.; Izursa, J.L.; Kangas, P.C. Consideration of carbon dioxide release during shell production in LCA of bivalves. Int. J. Life Cycle Assess. 2018, 23, 1042–1048. [Google Scholar] [CrossRef]
- Humphreys, M.P.; Daniels, C.J.; Wolf-Gladrow, D.A.; Tyrrell, T.; Achterberg, E.P. On the influence of marine biogeochemical processes over CO2 exchange between the atmosphere and ocean. Mar. Chem. 2006, 199, 1–11. [Google Scholar] [CrossRef]
- Morris, J.P.; Humphreys, M.P. Modelling seawater carbonate chemistry in shellfish aquaculture regions: Insights into CO2 release associated with shell formation and growth. Aquaculture 2019, 501, 338–344. [Google Scholar] [CrossRef]
- ReCiPe®. Available online: https://www.pre-sustainability.com/recipe (accessed on 19 May 2020).
- Portier, C.J.; Tart, K.T.; Carter, S.R.; Dilworth, C.H.; Grambsch, A.E.; Gohlke, J.; Maslak, T. A human health perspective on climate change: A report outlining the research needs on the human health effects of climate change. J. Curr. Issues Glob. 2013, 6, 621. [Google Scholar] [CrossRef]
- Goudarzi, G.; Geravandi, S.; Foruozandeh, H.; Babaei, A.A.; Alavi, N.; Niri, M.V.; Khodayar, M.J.; Salmanzadeh, S.; Mohammadi, M.J. Cardiovascular and respiratory mortality attributed to ground-level ozone in Ahvaz, Iran. Environ. Monit. Assess. 2015, 187, 487. [Google Scholar] [CrossRef] [PubMed]
- Beckers, F.; Rinklebe, J. Cycling of mercury in the environment: Sources, fate, and human health implications: A review. Crit. Rev. Environ. Sci. Technol. 2017, 47, 693–794. [Google Scholar] [CrossRef]
- Forrest, B.M.; Keeley, N.B.; Hopkins, G.A.; Webb, S.C.; Clement, D.M. Bivalve aquaculture in estuaries: Review and synthesis of oyster cultivation effects. Aquaculture 2009, 298, 1–15. [Google Scholar] [CrossRef]
- Nizzoli, D.; Welsh, D.T.; Viaroli, P. Seasonal nitrogen and phosphorus dynamics during benthic clam and suspended mussel cultivation. Mar. Pollut. Bull. 2011, 62, 1276–1287. [Google Scholar] [CrossRef] [PubMed]
- Bartoli, M.; Nizzoli, D.; Viaroli, P.; Turolla, E.; Castaldelli, G.; Fano, E.A.; Rossi, R. Impact of Tapes philippinarum farming on nutrient dynamics and benthic respiration in the Sacca di Goro. Hydrobiologia 2001, 455, 203–212. [Google Scholar] [CrossRef]
- Lodeiros, C.J.; Rengel, J.J.; Guderley, H.E.; Nusetti, O.; Himmelman, J.H. Biochemical composition and energy allocation in the tropical scallop Lyropecten (Nodipecten) nodosus during the months leading up to and following the development of gonads. Aquaculture 2001, 199, 63–72. [Google Scholar] [CrossRef]
- Brandao, M.; Levasseur, A. Assessing Temporary Carbon Storage in Life Cycle Assessment and Carbon Footprinting. European Commission Joint Research Centre Institute for Environment and Sustainability Scientific and Technical Reports. 2010. Available online: http://www.avnir.org/documentation/e_book/Workshop-Report-final.pdf (accessed on 21 May 2020).
- Filgueira, R.; Byron, C.J.; Comeau, L.A.; Costa-Pierce, B.; Cranford, P.J.; Ferreira, J.G.; Grant, J.; Guyondet, T.; Jansen, H.; Landry, T.; et al. An integrated ecosystem approach for assessing the potential role of cultivated bivalve shells as part of the carbon trading system. Mar. Ecol. Prog. Ser. 2015, 518, 281–287. [Google Scholar] [CrossRef]
| Input | |
|---|---|
| From the Technosphere | |
| Materials and Fuels | |
| Low-density polyethylene (LDPE) (g) | 6.0 |
| Polyvinyl chloride (PVC) (g) | 0.8 |
| Rubber (g) | <0.1 |
| Concrete (kg) | 22.5 |
| Chestnut wood (m3) | 0.3 |
| Gasoline (g) | 30 |
| Engine oil (l) | 0.2 |
| Electricity (kW) | 2.3 |
| Emissions to air | |
| Carbon dioxide (kg) | 8.28 |
| Nitrous oxide (kg) | 0.13 |
| Sulfur oxide (kg) | 0.22 |
| Methane (kg) | 0.21 |
| Non-methane volatile organic carbon (NMVOC) (kg) | 0.13 |
| Chemical oxygen demand (COD) (kg) | 0.10 |
| Dissolved organic carbon (DOC) (g) | 3.50 |
| Heat, waste (J) | 4.21 |
| PM < 2.5 µm (g) | 3.4 |
| PM > 2.5 µm and < 10 µm (g) | 27.0 |
| PM > 10 µm (g) | 38.0 |
| From the Environment | |
| Resources | |
| Seawater (m3) | 2.28 |
| Freshwater (m3) | 0.3 |
| Clams Harvested | |
|---|---|
| C fixed in shells (kg) | 88.00 ± 3.34 |
| N fixed in shells (kg) | 1.54 ± 0.14 |
| P fixed in shells (kg) | 0.31 ± 0.02 |
| CO2 released by calcification (kg) | 124.20 ± 6.21 |
| Impact Category | Value | CV% | Unit |
|---|---|---|---|
| Climate change—GWP100 * | 75.95 | 26% | kg CO2 eq. |
| Acidification potential (AP) | 0.56 | 34% | kg SO2 eq. |
| Eutrophication potential (EP) | 0.16 | 24% | kg PO4 eq. |
| Fossil depletion (FD) | 60.29 | 18% | kg oil eq. |
| Water depletion (WD) | 69.96 | 32% | m3 |
| Ozone layer depletion potential (ODP) | 1.81 × 10−5 | 33% | kg CFC-11 eq. |
| Photochemical oxidant formation potential (POFP) | 0.75 | 33% | kg NMVOC eq. |
| Human toxicity potential (HTP) | 8.63 | 60% | kg 1,4-DCB eq.** |
| Marine water aquatic ecotoxicity potential (MAETP) | 1.89 | 46% | kg 1,4-DCB eq. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turolla, E.; Castaldelli, G.; Fano, E.A.; Tamburini, E. Life Cycle Assessment (LCA) Proves that Manila Clam Farming (Ruditapes Philippinarum) is a Fully Sustainable Aquaculture Practice and a Carbon Sink. Sustainability 2020, 12, 5252. https://doi.org/10.3390/su12135252
Turolla E, Castaldelli G, Fano EA, Tamburini E. Life Cycle Assessment (LCA) Proves that Manila Clam Farming (Ruditapes Philippinarum) is a Fully Sustainable Aquaculture Practice and a Carbon Sink. Sustainability. 2020; 12(13):5252. https://doi.org/10.3390/su12135252
Chicago/Turabian StyleTurolla, Edoardo, Giuseppe Castaldelli, Elisa Anna Fano, and Elena Tamburini. 2020. "Life Cycle Assessment (LCA) Proves that Manila Clam Farming (Ruditapes Philippinarum) is a Fully Sustainable Aquaculture Practice and a Carbon Sink" Sustainability 12, no. 13: 5252. https://doi.org/10.3390/su12135252
APA StyleTurolla, E., Castaldelli, G., Fano, E. A., & Tamburini, E. (2020). Life Cycle Assessment (LCA) Proves that Manila Clam Farming (Ruditapes Philippinarum) is a Fully Sustainable Aquaculture Practice and a Carbon Sink. Sustainability, 12(13), 5252. https://doi.org/10.3390/su12135252








