The Role of Hydrogen in the Ecological Benefits of Ultra Low Sulphur Diesel Production and Use: An LCA Benchmark
Abstract
:1. Introduction
1.1. SOx Pollution
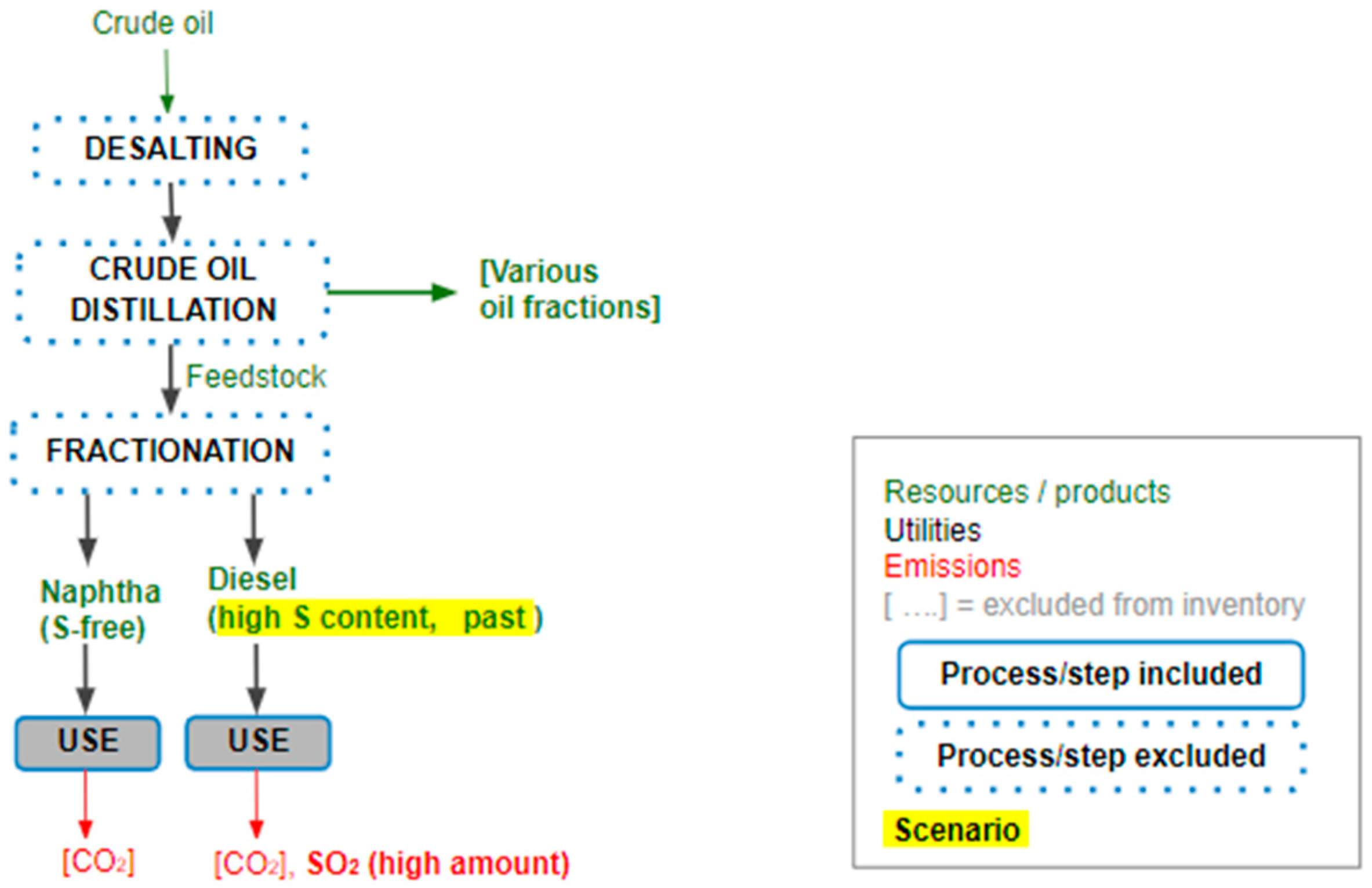
1.2. Desulphurization of Crude Oil
1.3. Knowledge Gap, Research Questions, and the Structure of This Paper
- RQ 1.
- What is the overall net ecological benefit of the current desulphurization practice (ULSD via H2 from SMR)?
- RQ 2.
- At which S ppm level in fuel does the additional ecological burden of desulphurization become higher than the additional ecological benefit of less SOx pollution from combustion?
- RQ 3.
- To what extent can cleaner hydrogen processes improve the overall net ecological benefit of diesel desulphurization?
2. Methods
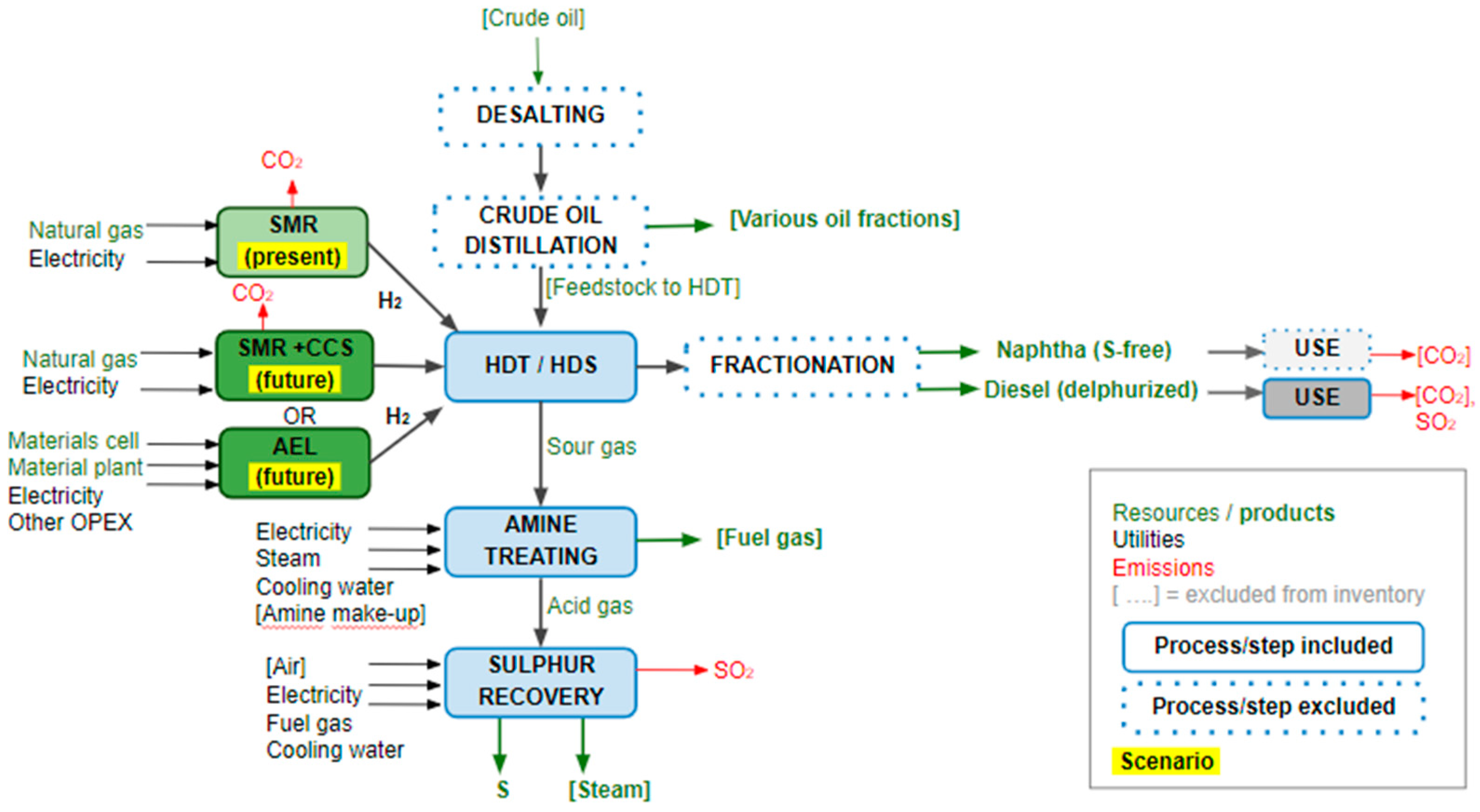
2.1. The System under Study
- Base case: SMR (9.0 ton/h H2) without CCS. The plant is equipped with feedstock pretreatment, pre-reformer, primary reformer, high temperature shift, and pressure swing adsorber (PSA) for H2-purification. Excess steam is converted into electricity, which is used to satisfy the plant needs. The excess power is exported to the grid.
- Case 2B: SMR with CO2 capture from the PSA tail gas using cryogenic and membrane separation technology (53% CO2 capture). Nowadays, there are several IG companies who have developed the cryogenic separation technology. In particular it should be noted that the Port-Jérôme project is the first large scale pilot demonstration of the CRYOCAP™ technology to capture CO2 from the hydrogen plants.
- Case 3: SMR with CO2 capture from the SMR’s flue gas using MEA (monoethanolamine) for chemical absorption (90% CO2 capture).
2.2. The Choice of the LCA Indicators
3. Results
3.1. A Comparison of the Ecological Burden and the Ecological Benefit of the Present Production System
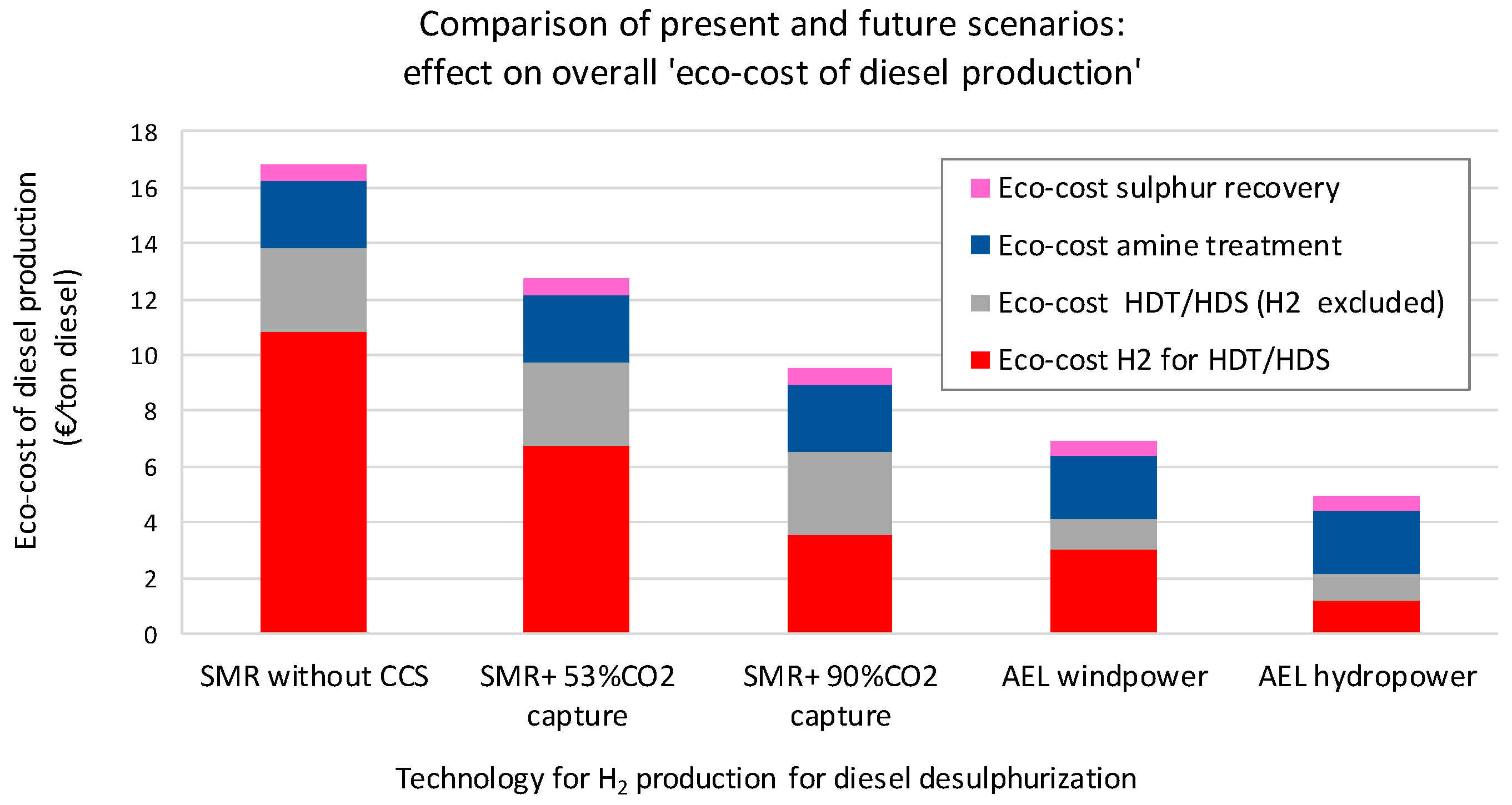
3.2. A Comparison of Present and Future Production Systems
4. Discussion
4.1. The Issue of Local Damage
4.2. The Issue of the Source of Electricity
4.3. The Issue of the Economic Feasibility
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
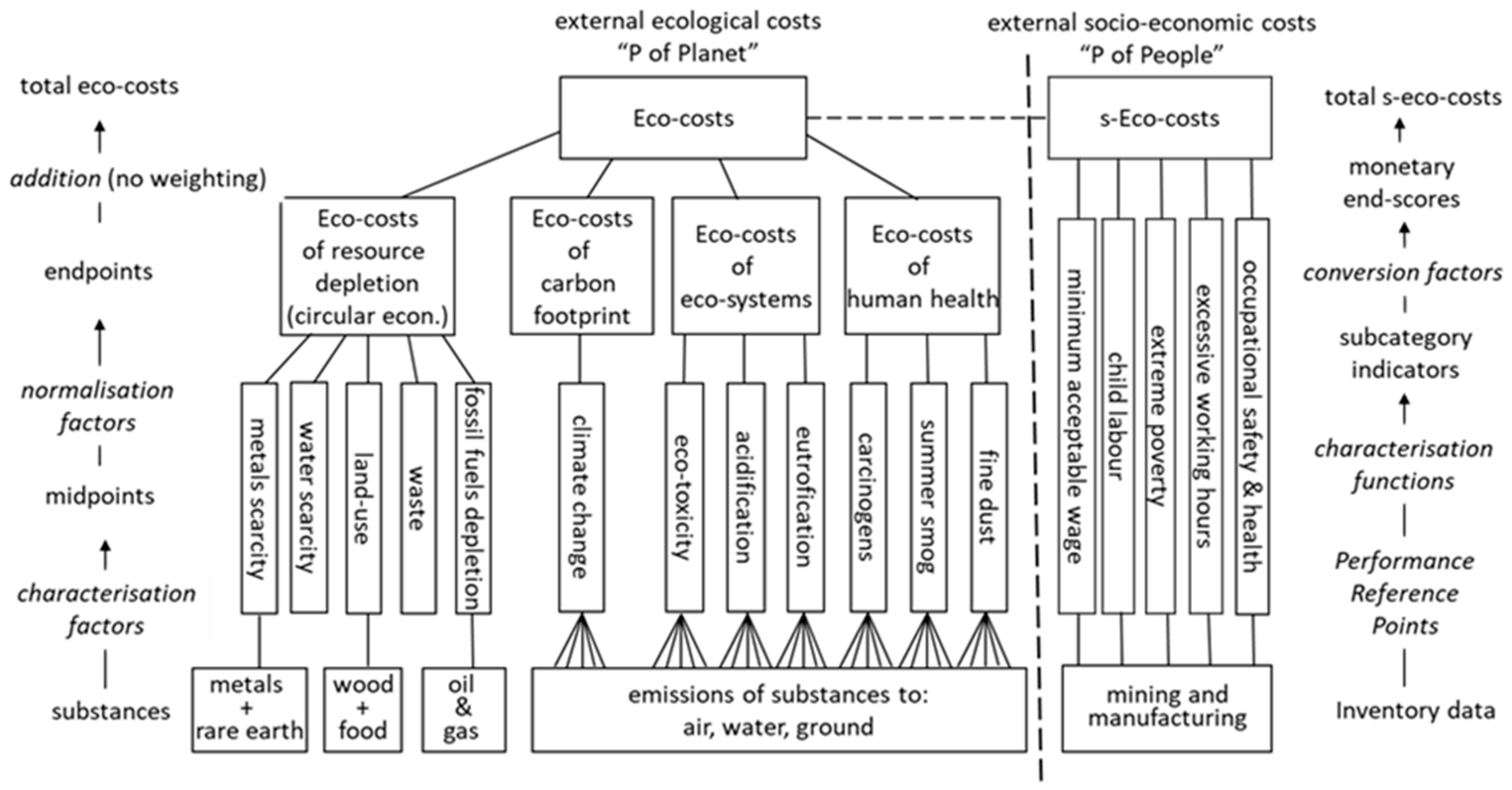
Appendix A. Short Description of the Eco-Costs Indicator System in LCA
| Eco-Costs of | Equivalent |
|---|---|
| acidification | 8.75 €/kg SOx equivalent |
| eutrophication | 4.17 €/kg phosphate equivalent |
| ecotoxicity | 55.0 €/kg Cu equivalent |
| human toxicity | 3754 €/kg Benzo(a)pyrene equivalent |
| summer smog (respiratory diseases) | 10.38 €/kg C2H4 equivalent |
| fine dust | 34.0 €/kg fine dust PM2.5 |
| global warming (GWP 100) | 0.116 €/kg CO2 equivalent |
- IPPC 2013, 100 years, for greenhouse gasses
- USETOX 2, for human toxicity (carcinogens) and ecotoxicity
- ILCD, for acidification, eutrophication, and photochemical oxidant formation (summer smog)
- UNEP/SETAC 2016, for fine dust PM2.5 (for PM10 the default factors of the ILCD Midpoint+ are used)
- Eco-costs of abiotic scarcity (metals, including rare earth, and fossil fuels)
- Eco-costs of land-use change (based on loss of biodiversity, of vascular plants and mammals, used for eco-costs of tropical hardwood)
- Eco-costs of water scarcity (based on the midpoint Water Stress Indicator (WSI) of countries’ eco-costs of landfill)
- Eco-costs of human health = the sum of carcinogens, summer smog, and fine dust
- Eco-costs of ecosystems = the sum of acidification, eutrophication, and ecotoxicity
- Eco-costs of resource scarcity = the sum of abiotic depletion, land-use, water, and landfill
- Costs of global warming = the sum of CO2 and other greenhouse gases (the GWP 100 table)
- Total eco-costs = the sum of human health, ecosystems, resource scarcity, and global warming.
References
- World Health Organization. Chapter 7.4 “Sulfur dioxide”. In Air Quality Guidelines for Europe, 2nd ed.; WHO Regional Publications, European Series, No. 91; WHO: Geneva, Switzerland, 2000. [Google Scholar]
- World Health Organization. Air Quality Guidelines Global Update 2005. Particulate Matter, Ozone, Nitrogen Dioxide and Sulfur Dioxide; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- Fantke, P.; Evans, J.; Hodas, N.; Apte, J.; Jantunen, M.; Jolliet, O.; McKone, T.E. Health impacts of fine particulate matter. In Global Guidance for Life Cycle Impact Assessment Indicators; Frischknecht, R., Jolliet, O., Eds.; UNEP DTIE Sustainable Lifestyles, Cities and Industry Branch: Paris, France, 2016; Volume 1. [Google Scholar]
- Apte, J.S.; Marshall, J.D.; Cohen, A.J.; Brauer, M. Addressing Global Mortality from Ambient PM2.5. Environ. Sci. Technol. 2015, 49, 8057–8066. [Google Scholar] [CrossRef]
- Di, Q.; Dai, L.; Wang, Y.; Zanobetti, A.; Choirat, C.; Schwartz, J.D.; Dominici, F. Association of Short-term Exposure to Air Pollution with Mortality in Older Adults. JAMA 2017, 318, 2446–2456. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, C.T.; Lawrence, G.B.; Bulger, A.J.; Butler, T.J.; Cronan, C.S.; Eagar, C.; Lambert, K.F.; Likens, G.E.; Stoddard, J.L.; Weathers, K.C. Acidic Deposition in the Northeastern United States: Sources and Inputs, Ecosystem Effects, and Management Strategies: The effects of acidic deposition in the northeastern United States include the acidification of soil and water, which stresses terrestrial and aquatic biota. BioScience 2001, 51, 180–198. [Google Scholar]
- Harriman, R.; Wells, D.E. Causes and effects of surface water acidification. Water Pollut. Control 1985, 84, 215–224. [Google Scholar]
- Potting, J.; Schöpp, W.; Blok, K.; Hauschild, M. Site-Dependent Life-Cycle Impact Assessment of Acidification. J. Ecol. 1998, 2, 63–87. [Google Scholar] [CrossRef]
- Seppälä, J.; Posch, M.; Johansson, M.; Hettelingh, J.-P. Country-dependent Characterisation Factors for Acidification and Terrestrial Eutrophication Based on Accumulated Exceedance as an Impact Category Indicator. Int. J. Life Cycle Assess. 2006, 11, 403–416. [Google Scholar] [CrossRef]
- Posch, M.; Seppälä, J.; Hettelingh, J.-P.; Johansson, M.; Margni, M.; Jolliet, O. The role of atmospheric dispersion models and ecosystem sensitivity in the determination of characterisation factors for acidifying and eutrophying emissions in LCIA. Int. J. Life Cycle Assess. 2008, 13, 477–486. [Google Scholar] [CrossRef]
- National Research Council. Ocean Acidification: A National Strategy to Meet the Challenges of a Changing Ocean; National Academies Press: Washington, DC, USA, 2010. [Google Scholar] [CrossRef]
- Barthe, P.; Chaugny, M.; Roudier, S.; Delgado Sancho, L. Best Available Technique (BAT) Reference Document for the Refining and Mineral Oil and Gas; JRC Science and Policy Report EUR 27140EN; Publications Office of the European Union: Luxembourg City, Luxembourg, 2015. [Google Scholar]
- De Crisci, A.G.; Moniri, A.; Xu, Y. Hydrogen from hydrogen sulfide: Towards a more sustainable hydrogen economy. Int. J. Hydrog. Energy 2019, 44, 1299–1327. [Google Scholar] [CrossRef]
- De León, J.N.D.; Kumar, C.R.; Antúnez-García, J.; Fuentes-Moyado, S. Recent Insights in Transition Metal Sulfide Hydrodesulfurization Catalysts for the Production of Ultra Low Sulfur Diesel: A Short Review. Catalysts 2019, 9, 87. [Google Scholar] [CrossRef]
- IEA Energy Technology ETE 05 2017, Hydrogen Production & Distribution. Available online: https://webstore.iea.org/iea-energy-technology-essentials-hydrogen-production-distribution (accessed on 19 February 2019).
- IEAGHG Technical Report 2017-02. Techno-Economic Evaluation of SMR Based Standalone (Merchant) Hydrogen Plant with CCS; IEAGHG: Cheltenham, UK, 2007; Available online: https://ieaghg.org/exco_docs/2017-02.pdf (accessed on 19 February 2019).
- Figueroa, J.D.; Fout, T.; Plasynski, S.; McIlvried, H.; Srivastava, R.D. Advances in CO2 capture technology—The U.S. Department of Energy’s Carbon Sequestration Program. Int. J. Greenh. Gas Control 2008, 2, 9–20. [Google Scholar] [CrossRef]
- Kelly, N.A.A. Hydrogen production by water electrolysis (Chapter 6). In Advances in Hydrogen Production, Storage and Distribution; Basie, A., Iulianelli, A., Eds.; Elsevier Ltd.: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Burgess, A.A.; Brennan, D.J. Desulfurisation of gas oil A case study in environmental and economic assessment. J. Clean. Prod. 2001, 9, 465–472. [Google Scholar] [CrossRef]
- Wu, L.; Liu, Y. Environmental impacts of hydrotreating processes for the production of clean fuels based on life cycle assessment. Fuel 2016, 164, 352–360. [Google Scholar] [CrossRef]
- Wu, L.; Wang, Y.; Zheng, L.; Han, X.; Hong, F. Multi-objective Operational Optimization of a Hydrotreating Process Based on Hydrogenation Reaction Kinetics. Ind. Eng. Chem. Res. 2018, 57, 15785–15793. [Google Scholar] [CrossRef]
- Valente, A.; Irribaren, D.; Dofour, J. Harmonized Life-Cycle global warming impact of renewable hydrogen. J. Clean. Prod. 2017, 149, 762–772. [Google Scholar] [CrossRef]
- Suleman, F.; Dincer, I.; Agelin-Chaab, M. Comparative impact assessment study of various hydrogen production methods in terms of emissions. Int. J. Hydrog. Energy 2016, 41, 8364–8375. [Google Scholar] [CrossRef]
- Mehmeti, A.; Angelis-Dimakis, A.; Arampatzis, G.; McPhail, S.J.; Ulgiati, S. Life Cycle Assessment and Water Footprint of Hydrogen Production Methods: From Conventional to Emerging Technologies. Environments 2018, 5, 24. [Google Scholar] [CrossRef]
- Chui, F.; Elkamel, A.; Fowler, M. An integrated Decision Support Framework for the Assessment and Analysis of Hydrogen Production Pathways. Energy Fuels 2006, 20, 346–352. [Google Scholar] [CrossRef]
- Lee, D.-Y.; Elgowainy, A.; Dai, Q. Life cycle greenhouse gas emissions of hydrogen fuel production from chlor-alkali processes in the United States. Appl. Energy 2018, 217, 467–479. [Google Scholar] [CrossRef]
- Lee, D.-Y.; Elgowainy, A. By-product hydrogen from steam cracking of natural gas liquids (NGLs): Potential for large-scale hydrogen fuel production, life-cycle air emissions reduction, and economic benefit. Int. J. Hydrog. Energy 2018, 43, 20143–20160. [Google Scholar] [CrossRef]
- Koj, J.C.; Wulf, C.; Schreiber, A.; Zapp, P. Site-Dependent Environmental Impacts of Industrial Hydrogen Production by Alkaline Water Electrolysis. Energies 2017, 10, 860. [Google Scholar] [CrossRef]
- Koj, J.C.; Schreiber, A.; Zapp, P.; Marcuello, P. Life Cycle Assessment of improved high pressure alkaline electrolysis. Energy Procedia 2015, 75, 2871–2877. [Google Scholar] [CrossRef]
- Huijbregts, M.A.J.; Steinmann, Z.J.N.; Elshout, P.M.F.; Stam, G.; Verones, F.; Vieira, M.; Zijp, M.; Hollander, A.; Van Zelm, R. ReCiPe2016: A harmonised life cycle impact assessment method at midpoint and endpoint level. Int. J. Life Cycle Assess. 2017, 22, 138–147. [Google Scholar] [CrossRef]
- Rosenbaum, R.K.; Bachmann, T.M.; Gold, L.S.; Huijbregts, M.A.J.; Jolliet, O.; Juraske, R.; Koehler, A.; Larsen, H.F.; MacLeod, M.; Margni, M.; et al. USEtox—The UNEP-SETAC toxicity model: Recommended characterisation factors for human toxicity and freshwater ecotoxicity in life cycle impact assessment. Int. J. Life Cycle Assess. 2008, 13, 532–546. [Google Scholar] [CrossRef]
- Vogtlander, J.G.; Bijma, A. The ‘Virtual Pollution Prevention Costs ‘99’. J. Life Cycle Assess. 2000, 5, 113–124. [Google Scholar] [CrossRef]
- Vogtlander, J.G.; Brezet, H.C.; Hendriks, C.F. The virtual eco-costs ‘99. A Single LCA-Based Indicator for Sustainability and the Eco-Costs-Value Ratio (EVR) model for Economic Allocation. Int. J. Life Cycle Assess. 2001, 6, 157–166. [Google Scholar] [CrossRef]
- Vogtlander, J.G. A Practical Guide to LCA for Students, Designers and Business Managers, 5th ed.; Academic Press: Delft, The Netherlands, 2017. [Google Scholar]
- Mano, T.B.; Guillén-Gosálbez, G.; Jiménez, L.; Ravagnani, M.A.S.S. Synthesis of heat exchanger networks with economic and environmental assessment using fuzzy-Analytic Hierarchy Process. Chem. Eng. Sci. 2019, 195, 185–200. [Google Scholar] [CrossRef]
- Aghbashlo, M.; Rosen, M.A. Exergoeconoenvironmental analysis as a new concept for developing thermodynamically, economically, and environmentally sound energy conversion systems. J. Clean. Prod. 2018, 187, 190–204. [Google Scholar] [CrossRef]
- Dong, Y.; Hauschild, M.; Sørup, H.; Rousselet, R.; Fantke, P. Evaluating the costs of greenhouse gases emissions in the life cycle impact assessment. J. Clean. Prod. 2019, 209, 538–549. [Google Scholar] [CrossRef]
- Weidema, B.P. Using the budget constraint to monetarise impact assessment results. Ecol. Econ. 2009, 68, 1591–1598. [Google Scholar] [CrossRef]
- Goedkoop, M.; Spriensma, R. The Eco-indicator 99. A Damage Oriented Method for Life Cycle Impact Assessment. Methodology Report, 3rd ed.; Pré Consultants: Amersfoort, The Netherlands, 2001. [Google Scholar]
- Cédric, P. Renewable Energy Division, Updated 1st October 2017, IEA, Producing Ammonia and Fertilizers: New Opportunities from Renewables. Available online: https://www.iea.org/media/news/2017/Fertilizer_manufacturing_Renewables_01102017.pdf (accessed on 19 February 2019).
- Midpoint Characterisation Table for Acidification in of the ILCD Midpoint+ System. Available online: http://eplca.jrc.ec.europa.eu/?page_id=140 (accessed on 15 December 2018).
- Apte, J.S.; Bombrun, E.; Marshall, J.D.; Nazaroff, W.W. Global Intraurban Intake Fractions for Primary Air Pollutants from Vehicles and Other Distributed Sources. Environ. Sci. Technol. 2012, 46, 3415–3423. [Google Scholar] [CrossRef]
- Gupta, M.; He, J.; Nguyen, T.; Petzold, F.; Fonseca, D.; Jasinski, J.B.; Sunkara, M.K. Nanowire catalysts for ultra-deep hydro-desulfurization and aromatic hydrogenation. Appl. Catal. B Environ. 2016, 180, 246–254. [Google Scholar] [CrossRef]
- Salkuyeh, Y.K.; Saville, B.A.; MacLean, H.L. Techno-economic analysis and life cycle assessment of hydrogen production from natural gas using current and emerging technologies. Int. J. Hydrog. Energy 2017, 42, 18894–18909. [Google Scholar] [CrossRef]
- Ferdinand, F. EU ETS in 2030: A Long-Term Price Forecast. CEPS Task Force: EU ETS Market Stability Reserve, Brussels. 2014. Available online: https://www.ceps.eu/sites/default/files/Ferdinand.pdf (accessed on 19 February 2019).
- Lewis, M. EU Carbon Prices Could Double by 2012 and Quadruple by 2030. Carbon Tracker, 2018. Available online: https://www.carbontracker.org/eu-carbon-prices-could-double-by-2021-and-quadruple-by-2030/ (accessed on 19 February 2019).
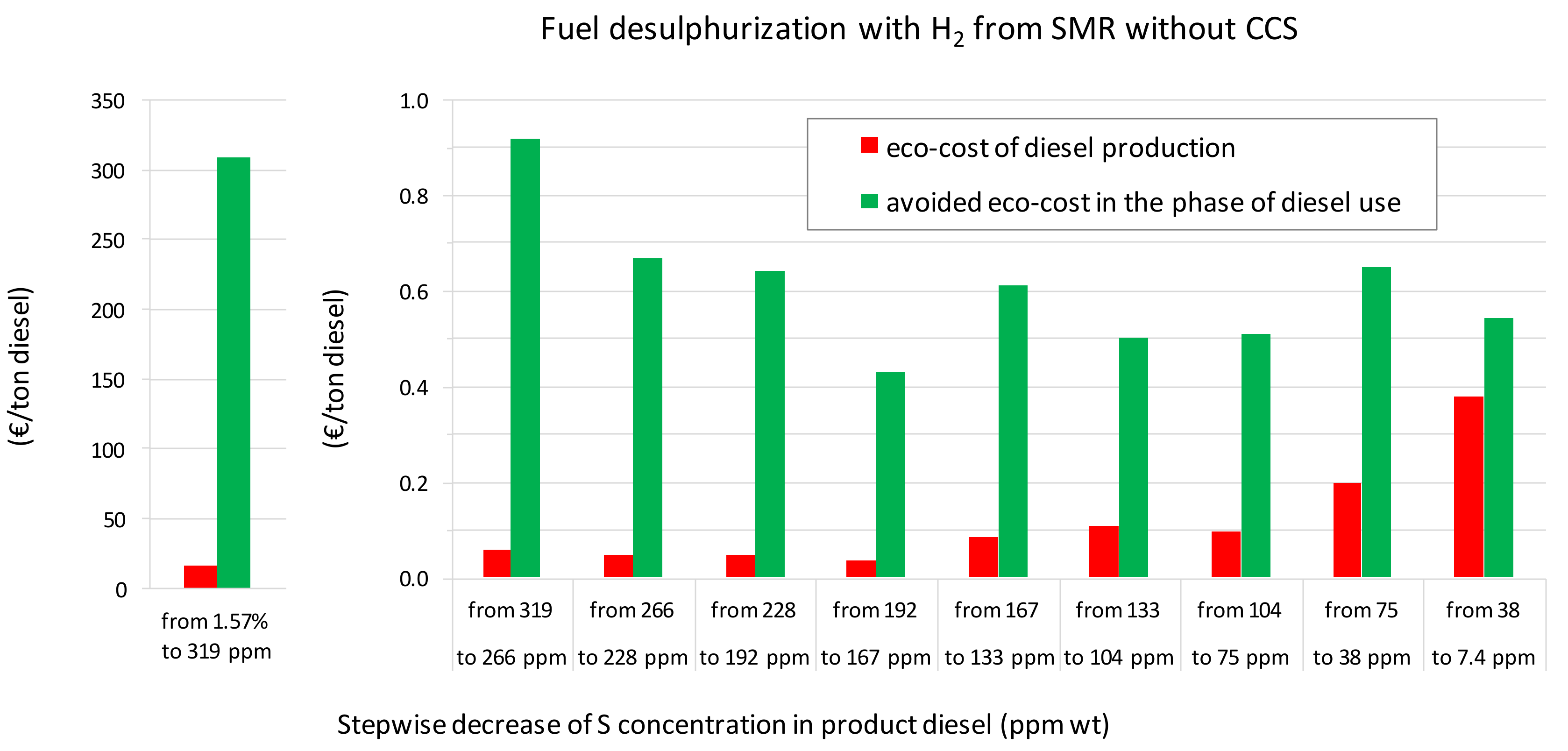
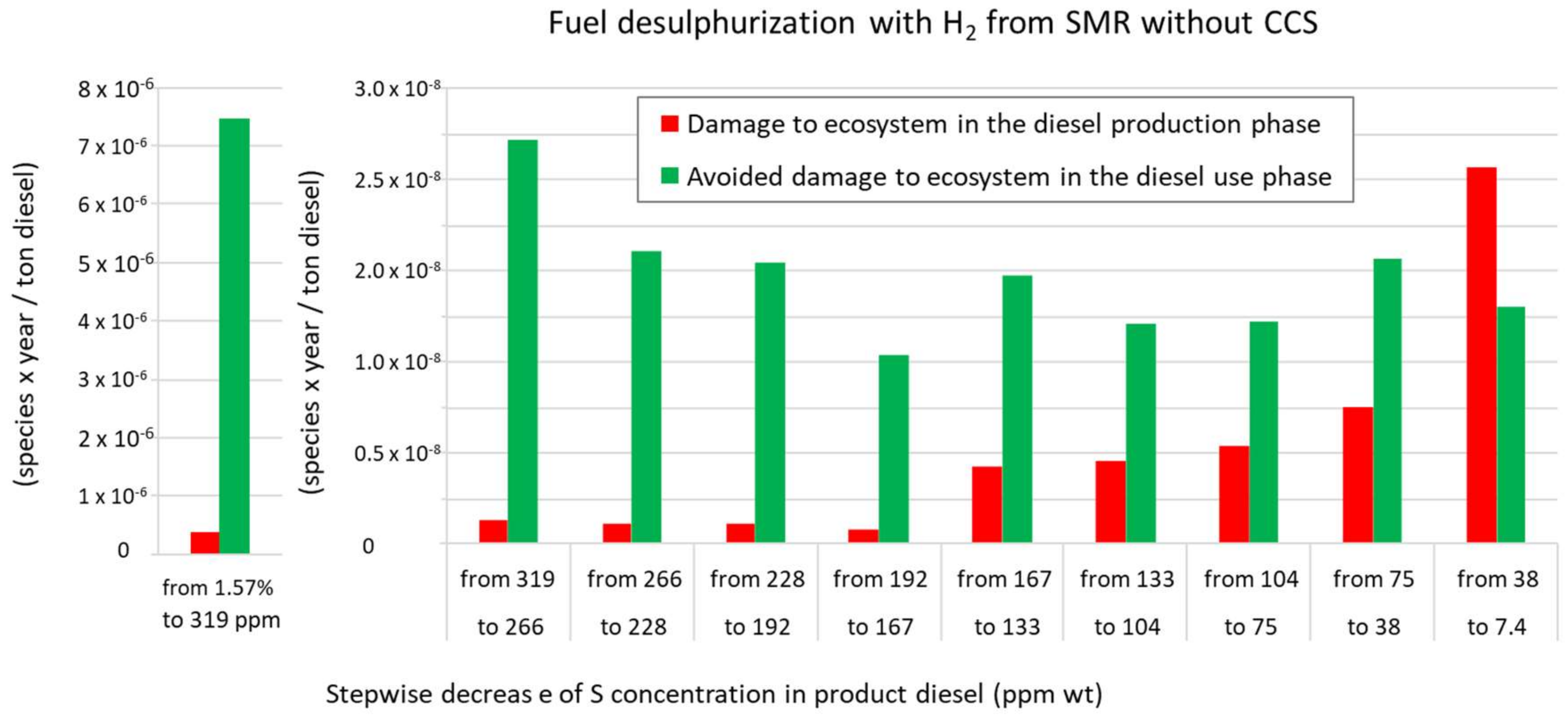
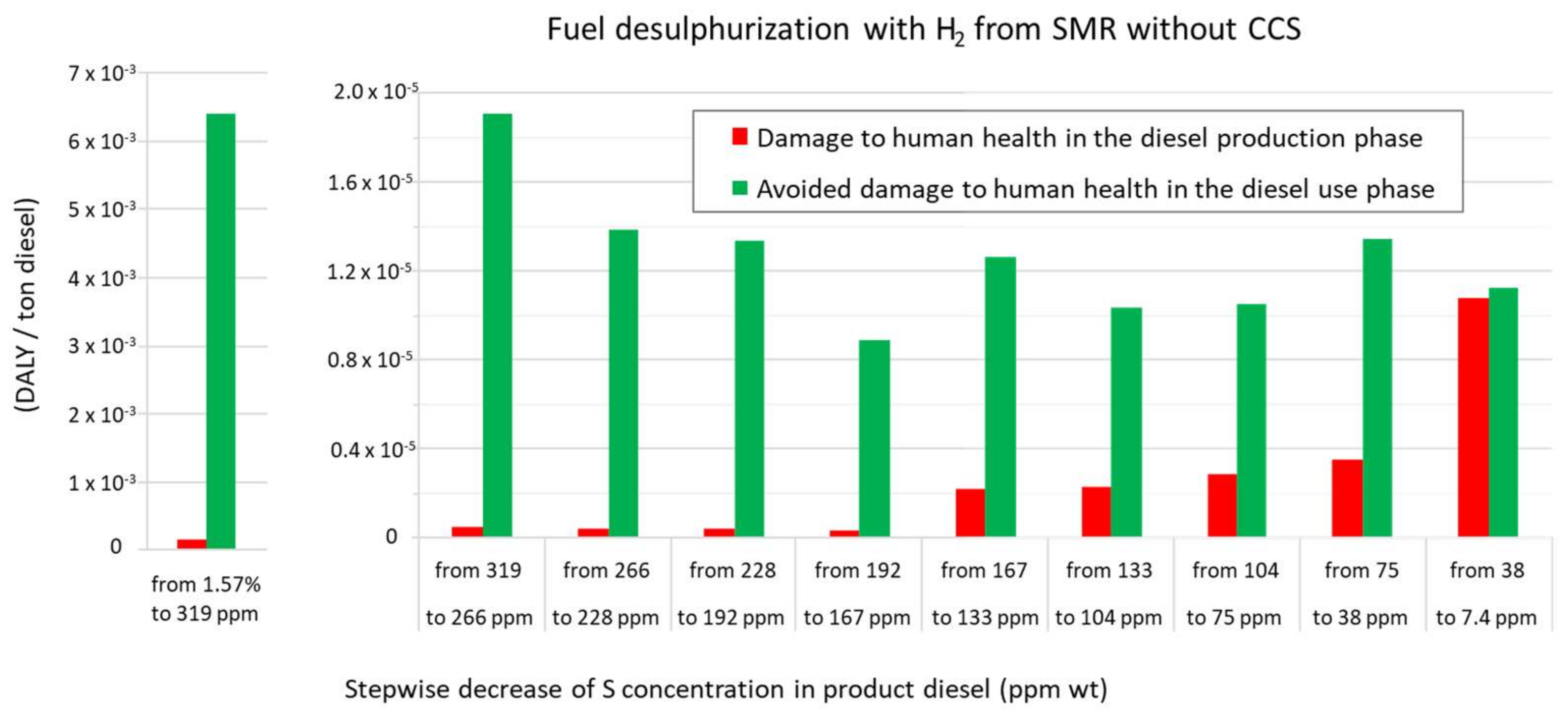
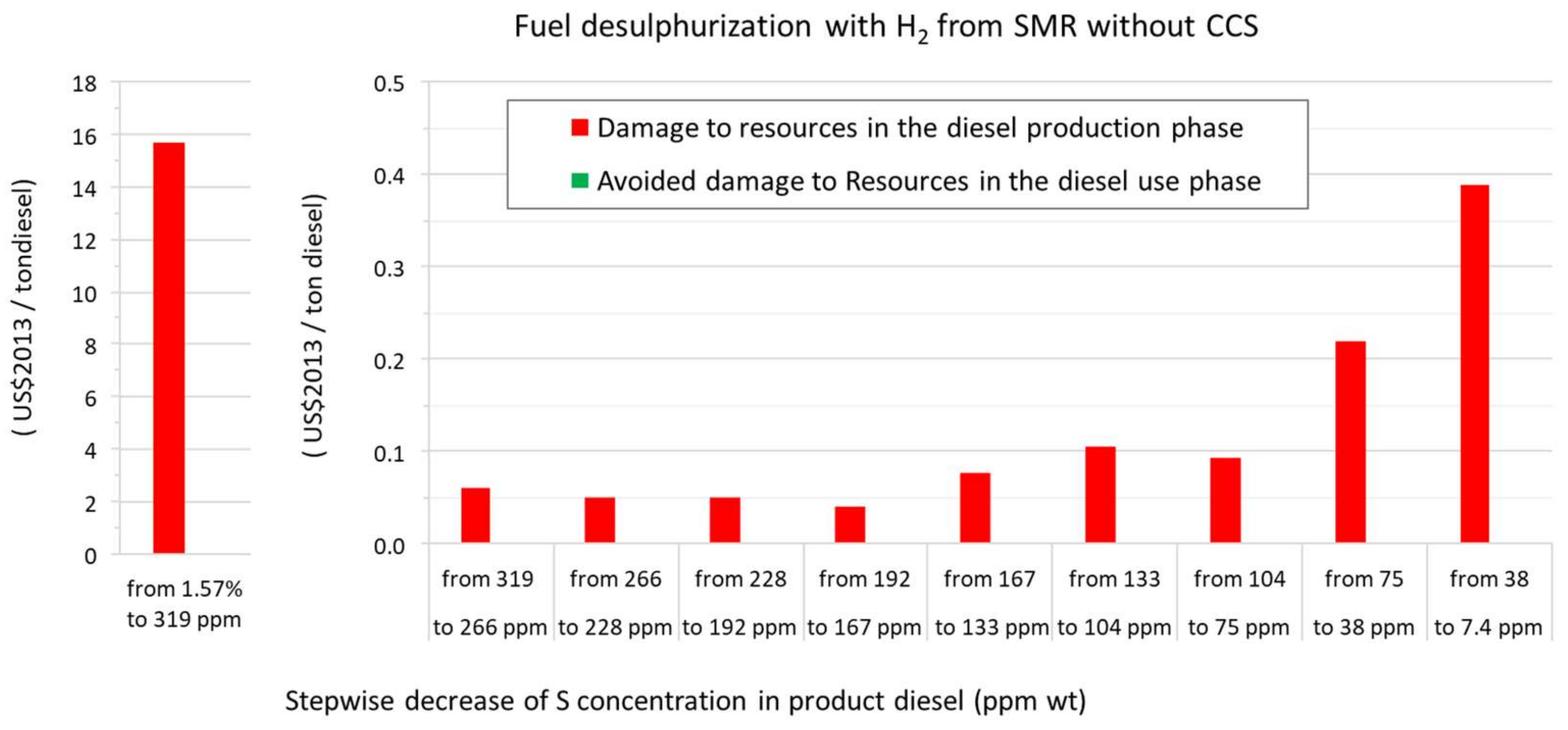
| SCENARIO (1.57% S in Feed to 7.4 ppm S in Diesel) | Present: H2 from SMR without CCS | Future: H2 from SMR with CCS | Future: H2 from Water Alkaline Electrolysis | |||
|---|---|---|---|---|---|---|
| 53% CO2 Capture | 90% CO2 Capture | |||||
| Electricity | EU Grid * | Windpower ** | Hydropower *** | |||
| Eco-costs H2 for HDT/HDS | (€/ton diesel) | 10.9 (64%) | 6.7 (53%) | 3.6 (37%) | 3.0 (44%) | 1.17 (24%) |
| Eco-costs HDT/HDS (H2 excluded) | (€/ton diesel) | 3.0 (18%) | 3.0 (23%) | 3.0 (31%) | 1.1 (16%) | 1.0 (20%) |
| Eco-costs amine treatment | (€/ton diesel) | 2.4 (14%) | 2.4 (19%) | 2.4 (25%) | 2.3 (33%) | 2.3 (46%) |
| Eco-costs sulphur recovery | (€/ton diesel) | 0.6 (4%) | 0.6 (5%) | 0.6 (6%) | 0.5 (7%) | 0.5 (10%) |
| Total eco-costs production | (€/ton diesel) | 16.9 (100%) | 12.7 (100%) | 9.5 (100%) | 6.9 (100%) | 4.9 (100%) |
| Avoided eco-costs (avoided SO2 emissions) | (€/ton diesel) | 313.4 | 313.4 | 313.4 | 313.4 | 313.4 |
| ‘Avoided eco-costs’/’Total eco-costs production’ | (€/€) | 18 | 25 | 33 | 46 | 64 |
| SCENARIO | Present: H2 from SMR without CCS | ||||||
|---|---|---|---|---|---|---|---|
| (Variable S Concentration in Feed to 38 ppmS Diesel) | |||||||
| S content in feed | (wt%) | 1.30 | 1.40 | 1.50 | 1.57 | 1.70 | 1.80 |
| Eco-costs H2 for HDT/HDS | (€/ton diesel) | 9.78 (65.8%) | 10.18 (65.9%) | 10.58 (65.9%) | 10.86 (65.9%) | 11.38 (66.0%) | 11.78 (66.1%) |
| Eco-costs HDT/HDS (H2 excluded) | (€ ton diesel) | 2.59 | 2.58 | 2.60 | 2.60 | 2.59 | 2.60 |
| Eco-costs amine treatment | (€/ton diesel) | 1.98 | 2.13 | 2.29 | 2.40 | 2.59 | 2.75 |
| Eco-costs sulphur recovery | (€/ton diesel) | 0.51 | 0.55 | 0.59 | 0.61 | 0.67 | 0.70 |
| Total ‘eco-costs production’ | (€/ton diesel) | 14.87 (100%) | 15.44 (100%) | 16.05 (100%) | 16.47 (100%) | 17.23 (100%) | 17.83 (100%) |
| ‘Avoided eco-costs’ (avoided SO2 emissions) | (€/ton diesel) | 258.77 | 278.73 | 298.70 | 312.88 | 338.60 | 358.55 |
| ‘Avoided eco-costs’/Total ‘eco-costs production’ | (€/€) | 17.4 | 18.1 | 18.6 | 19.0 | 19.7 | 20.1 |
| Factor | Country | Factor | Country | Factor | Country |
|---|---|---|---|---|---|
| 0.024 | Albania | 1.741 | Germany | 3.453 | Norway |
| 0.543 | Austria | 0.906 | Great Britain | 1.330 | Poland |
| 0.525 | Belarus | 0.009 | Greece | 0.028 | Portugal |
| 1.205 | Belgium | 0.414 | Hungary | 0.153 | Romania |
| 0.077 | Bosnia and Herzegovina | 0.426 | Ireland | 0.028 | Russia |
| 0.031 | Bulgaria | 0.084 | Italy | 0.111 | Serbia and Montenegro |
| 0.143 | Croatia | 0.329 | Latvia | 0.181 | Slovenia |
| 1.618 | Czech Republic | 0.434 | Lithuania | 0.600 | Slovakia |
| 1.560 | Denmark | 0.961 | Luxemburg | 0.082 | Spain |
| 0.282 | Estonia | 0.027 | Macedonia | 1.521 | Sweden |
| 0.353 | Finland | 0.134 | Moldova | 0.377 | Switzerland |
| 0.546 | France | 1.186 | Netherlands | 0.117 | Ukraine |
| City | Factor | City | Factor | City | Factor | City | Factor |
|---|---|---|---|---|---|---|---|
| normal city | 1.78 | Den Haag | 0.73 | London | 2.79 | Munich | 1.05 |
| Amsterdam | 0.84 | Eindhoven | 0.30 | Lyon | 0.93 | Paris | 2.71 |
| Barcelona | 2.05 | Frankfurt | 0.57 | Madrid | 1.62 | Rome | 1.00 |
| Berlin | 1.57 | Hannover | 0.46 | Marseille | 0.69 | Rotterdam | 0.55 |
| Brussels | 1.19 | Koln | 0.66 | Milano | 1.26 | rural average | 0.22 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olindo, R.; Vogtländer, J.G. The Role of Hydrogen in the Ecological Benefits of Ultra Low Sulphur Diesel Production and Use: An LCA Benchmark. Sustainability 2019, 11, 2184. https://doi.org/10.3390/su11072184
Olindo R, Vogtländer JG. The Role of Hydrogen in the Ecological Benefits of Ultra Low Sulphur Diesel Production and Use: An LCA Benchmark. Sustainability. 2019; 11(7):2184. https://doi.org/10.3390/su11072184
Chicago/Turabian StyleOlindo, Roberta, and Joost G. Vogtländer. 2019. "The Role of Hydrogen in the Ecological Benefits of Ultra Low Sulphur Diesel Production and Use: An LCA Benchmark" Sustainability 11, no. 7: 2184. https://doi.org/10.3390/su11072184
APA StyleOlindo, R., & Vogtländer, J. G. (2019). The Role of Hydrogen in the Ecological Benefits of Ultra Low Sulphur Diesel Production and Use: An LCA Benchmark. Sustainability, 11(7), 2184. https://doi.org/10.3390/su11072184




