Life Stage-Specific Hydropeaking Flow Rules
Abstract
1. Introduction
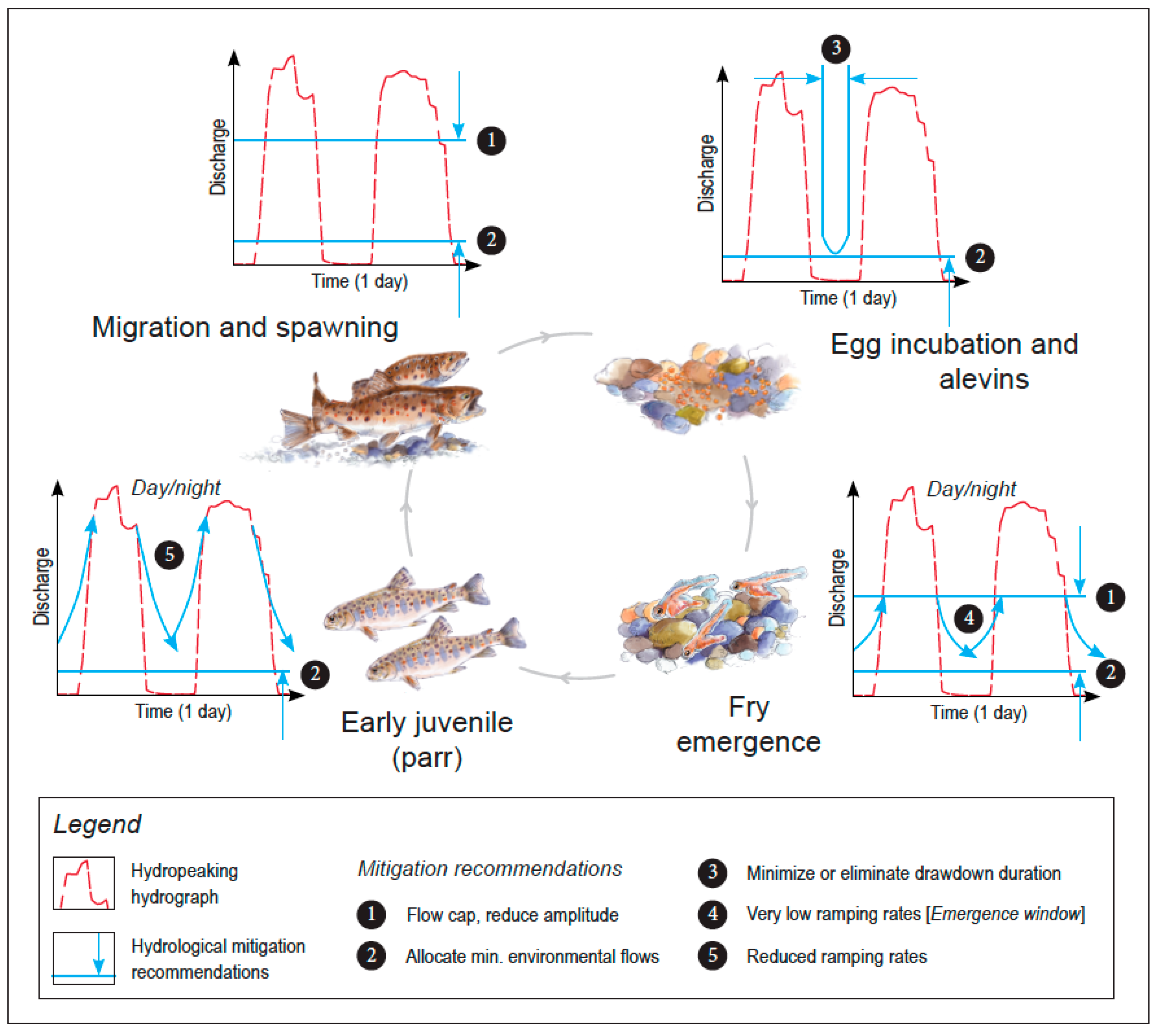
2. Life Cycle Stage-Specific Mitigation Approach
2.1. Migration and Spawning
2.2. Intragravel Life Cycle Stages: Egg Incubation and Alevins
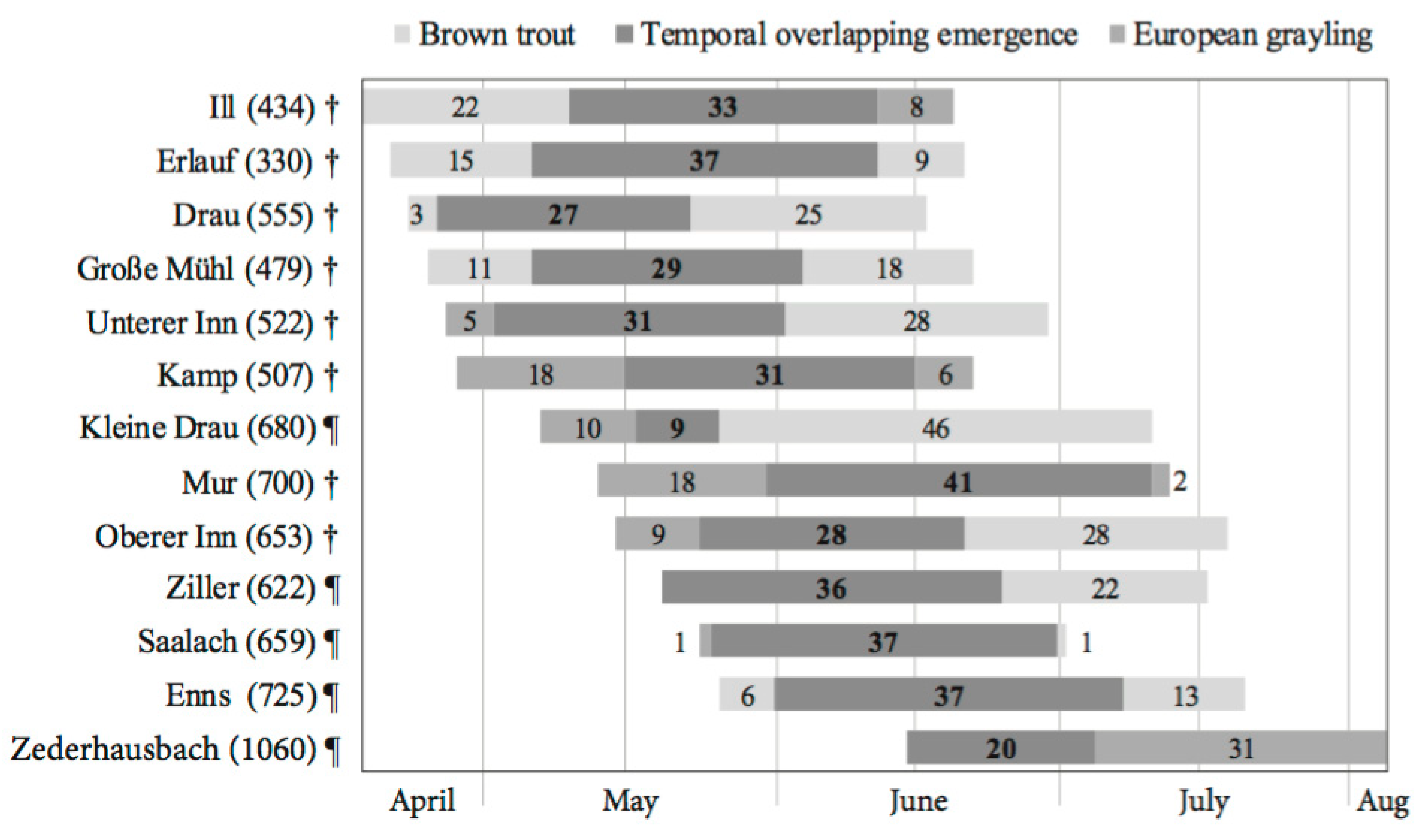
2.3. Fry Emergence and Early Juvenile Development
2.3.1. Thresholds for Impact Mitigation
2.3.2. Emergence Window Establishment
2.4. Parr to Adults
3. Discussion
3.1. The Effects of River Hydromorphology
3.2. The Effects of Sub-Daily Temperature Changes
3.3. Other Hydropeaking-Related Impacts
3.4. Achieving Hydrological Mitigation Measures and Their Economic Implications
3.5. Limitations of This Study and Research Needs
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ashraf, F.B.; Haghighi, A.T.; Riml, J.; Alfredsen, K.; Koskela, J.J.; Kløve, B.; Marttila, H. Changes in short term river flow regulation and hydropeaking in Nordic rivers. Sci. Rep. 2018, 8, 17232. [Google Scholar] [CrossRef] [PubMed]
- Greimel, F.; Neubarth, J.; Zeiringer, B.; Hayes, D.S.; Haslauer, M.; Führer, S.; Auer, S.; Höller, N.; Hauer, C.; Holzapfel, P.; et al. Sustainable River Management in Austria. In Proceedings of the 12th International Symposium on Ecohydraulics 2018, Tokyo, Japan, 19–24 August 2018; p. 4. [Google Scholar]
- Hauer, C.; Holzapfel, P.; Leitner, P.; Graf, W. Longitudinal assessment of hydropeaking impacts on various scales for an improved process understanding and the design of mitigation measures. Sci. Total Environ. 2017, 575, 1503–1514. [Google Scholar] [CrossRef] [PubMed]
- Schmutz, S.; Bakken, T.H.; Friedrich, T.; Greimel, F.; Harby, A.; Jungwirth, M.; Melcher, A.; Unfer, G.; Zeiringer, B. Response of Fish Communities to Hydrological and Morphological Alterations in Hydropeaking Rivers of Austria. River Res. Appl. 2015, 31, 919–930. [Google Scholar] [CrossRef]
- Auer, S.; Zeiringer, B.; Führer, S.; Tonolla, D.; Schmutz, S. Effects of river bank heterogeneity and time of day on drift and stranding of juvenile European grayling (Thymallus thymallus L.) caused by hydropeaking. Sci. Total Environ. 2017, 575, 1515–1521. [Google Scholar] [CrossRef] [PubMed]
- Saltveit, S.J.; Halleraker, J.H.; Arnekleiv, J.V.; Harby, A. Field experiments on stranding in juvenile Atlantic salmon (Salmo salar) and brown trout (Salmo trutta) during rapid flow decreases caused by hydropeaking. Regul. Rivers Res. Manag. 2001, 17, 609–622. [Google Scholar] [CrossRef]
- Casas-Mulet, R.; Alfredsen, K.; Brabrand, A.; Saltveit, S.J. Hydropower operations in groundwater-influenced rivers: Implications for Atlantic salmon, Salmo salar, early life stage development and survival. Fish. Manag. Ecol. 2016, 23, 144–151. [Google Scholar] [CrossRef][Green Version]
- Greimel, F.; Neubarth, J.; Fuhrmann, M.; Führer, S.; Habersack, H.; Haslauer, M.; Hauer, C.; Holzapfel, P.; Auer, S.; Pfleger, M.; et al. SuREmMa, Sustainable River Management—Energiewirtschaftliche und umweltrelevante Bewertung möglicher schwalldämpfender Maßnahmen; Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2017. [Google Scholar]
- IHA. Hydropower Status Report 2017; International Hydropower Association: London, UK, 2017. [Google Scholar]
- Bruder, A.; Tonolla, D.; Schweizer, S.P.; Vollenweider, S.; Langhans, S.D.; Wüest, A. A conceptual framework for hydropeaking mitigation. Sci. Total Environ. 2016, 568, 1204–1212. [Google Scholar] [CrossRef]
- Greimel, F.; Schülting, L.; Wolfram, G.; Bondar-Kunze, E.; Auer, S.; Zeiringer, B.; Hauer, C. Hydropeaking Impacts and Mitigation. In Riverine Ecosystem Management; Schmutz, S., Sendzimir, J., Eds.; Springer: Berlin, Germany, 2018; pp. 91–110. ISBN 978-3-319-73250-3. [Google Scholar]
- Person, E.; Bieri, M.; Peter, A.; Schleiss, A.J. Mitigation measures for fish habitat improvement in Alpine rivers affected by hydropower operations. Ecohydrology 2014, 7, 580–599. [Google Scholar] [CrossRef]
- Freeman, M.C.; Bowen, Z.H.; Bovee, K.D.; Irwin, E.R. Flow and habitat effects on juvenile fish abundance in natural and altered flow regimes. Ecol. Appl. 2001, 11, 179–190. [Google Scholar] [CrossRef]
- Hayes, D.S.; Brändle, J.M.; Seliger, C.; Zeiringer, B.; Ferreira, T.; Schmutz, S. Advancing towards functional environmental flows for temperate floodplain rivers. Sci. Total Environ. 2018, 633, 1089–1104. [Google Scholar] [CrossRef]
- Moreira, M.; Hayes, D.S.; Boavida, I.; Schletterer, M.; Schmutz, S.; Pinheiro, A. Ecologically-based criteria for hydropeaking mitigation: A review. Sci. Total Environ. 2019, 657, 1508–1522. [Google Scholar] [CrossRef]
- Hunter, M.A. Hydropower Flow Fluctuations and Salmonids: A Review of the Biological Effects, Mechanical Causes and Options for Mitigation; Department of Fish and Wildlife: Olympia, WA, USA, 1992.
- Melcher, A.H.; Bakken, T.H.; Friedrich, T.; Greimel, F.; Humer, N.; Schmutz, S.; Zeiringer, B.; Webb, J.A. Drawing together multiple lines of evidence from assessment studies of hydropeaking pressures in impacted rivers. Freshw. Sci. 2017, 36, 220–230. [Google Scholar] [CrossRef]
- Nagrodski, A.; Raby, G.D.; Hasler, C.T.; Taylor, M.K.; Cooke, S.J. Fish stranding in freshwater systems: Sources, consequences, and mitigation. J. Environ. Manag. 2012, 103, 133–141. [Google Scholar] [CrossRef]
- Crisp, D.T. Trout and Salmon: Ecology, Conservation and Rehabilitation; Fishing News Books, Blackwell Science: Oxford, UK, 2000. [Google Scholar]
- Quinn, T.P. The Behavior and Ecology of Pacific Salmon and Trout; University of Washington Press: Seattle, WA, USA, 2005. [Google Scholar]
- Malcolm, I.A.; Gibbins, C.N.; Soulsby, C.; Tetzlaff, D.; Moir, H.J. The influence of hydrology and hydraulics on salmonids between spawning and emergence: Implications for the management of flows in regulated rivers. Fish. Manag. Ecol. 2012, 19, 464–474. [Google Scholar] [CrossRef]
- Mendez, R. Laichwanderung der Seeforelle im Alpenrhein; Swiss Federal Institute of Aquatic Science and Technology: Dübendorf, Switzerland, 2007. [Google Scholar]
- Jones, N.E.; Petreman, I.C. Environmental Influences on Fish Migration in a Hydropeaking River. River Res. Appl. 2015, 31, 1109–1118. [Google Scholar] [CrossRef]
- Haas, C.; Zinke, P.; Vollset, K.W.; Sauterleute, J.; Skoglund, H. Behaviour of spawning Atlantic salmon and brown trout during ramping events. J. Appl. Water Eng. Res. 2016, 4, 25–30. [Google Scholar] [CrossRef]
- Moir, H.J.; Gibbins, C.N.; Soulsby, C.; Webb, J.H. Discharge and hydraulic interactions in contrasting channel morphologies and their influence on site utilization by spawning Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 2006, 63, 2567–2585. [Google Scholar] [CrossRef]
- Vollset, K.W.; Skoglund, H.; Wiers, T.; Barlaup, B.T. Effects of hydropeaking on the spawning behaviour of Atlantic salmon Salmo salar and brown trout Salmo trutta. J. Fish Biol. 2016, 88, 2236–2250. [Google Scholar] [CrossRef]
- Esteve, M. Observations of spawning behaviour in Salmoninae: Salmo, Oncorhynchus and Salvelinus. Rev. Fish Biol. Fish. 2005, 15, 1–21. [Google Scholar] [CrossRef]
- Chapman, D.W.; Weitkamp, D.E.; Welsh, T.L.; Dell, M.B.; Schadt, T.H. Effects of river flow on the distribution of Chinook salmon redds. Trans. Am. Fish. Soc. 1986, 115, 537–547. [Google Scholar] [CrossRef]
- IRKA. Alpenrhein: Quantitative Analyse von Schwall/Sunk-Ganglinien für unterschiedliche Anforderungsprofile; IRKA: Vaduz, Liechtenstein, 2012. [Google Scholar]
- Moir, H.J.; Gibbins, C.N.; Soulsby, C.; Webb, J. Linking channel geomorphic characteristics to spatial patterns of spawning activity and discharge use by Atlantic salmon (Salmo salar L.). Geomorphology 2004, 60, 21–35. [Google Scholar] [CrossRef]
- Becker, C.D.; Neitzel, D.A. Assessment of intergravel conditions influencing egg and alevin survival during salmonid redd dewatering. Environ. Biol. Fishes 1985, 12, 33–46. [Google Scholar] [CrossRef]
- McMichael, G.A.; Rakowski, C.L.; James, B.B.; Lukas, J.A. Estimated Fall Chinook Salmon Survival to Emergence in Dewatered Redds in a Shallow Side Channel of the Columbia River. N. Am. J. Fish. Manag. 2005, 25, 876–884. [Google Scholar] [CrossRef]
- Alfredsen, K.; Harby, A.; Linnansaari, T.; Ugedal, O. Development of an inflow-controlled environmental flow regime for a Norwegian river. River Res. Appl. 2012, 28, 731–739. [Google Scholar] [CrossRef]
- Hauer, C.; Holzapfel, P.; Tonolla, D.; Habersack, H.; Zolezzi, G. In situ measurements of fine sediment infiltration (FSI) in gravel-bed rivers with a hydropeaking flow regime. Earth Surf. Process. Landf. 2018. [Google Scholar] [CrossRef]
- Eberstaller, J.; Pinka, P. Trübung und Schwall Alpenrhein. Einfluss auf Substrat, Benthos, Fische. Teilbericht Fischökologie; BOKU: Wien, Austria, 2001. [Google Scholar]
- Meile, T.; Fette, M.; Baumann, P. Synthesebericht Schwall/Sunk; Rhone-Thur Project, Swiss Federal Institute of Aquatic Science and Technology (EAWAG): Dübendorf, Switzerland, 2005. [Google Scholar]
- Casas-Mulet, R.; Alfredsen, K.; Hamududu, B.; Timalsina, N.P. The effects of hydropeaking on hyporheic interactions based on field experiments. Hydrol. Process. 2015, 29, 1370–1384. [Google Scholar] [CrossRef]
- Sternecker, K.; Geist, J. The effects of stream substratum composition on the emergence of salmonid fry. Ecol. Freshw. Fish 2010, 19, 537–544. [Google Scholar] [CrossRef]
- O’Connor, W.C.K.; Andrew, T.E. The effects of siltation on Atlantic salmon, Salmo salar L., embryos in the River Bush. Fish. Manag. Ecol. 1998, 5, 393–401. [Google Scholar] [CrossRef]
- Schälchli, U.; Abegg, J.; Hunzinger, L. Kolmation: Methoden zur Erkennung und Bewertung; Swiss Federal Institute of Aquatic Science and Technology (EAWAG): Dübendorf, Switzerland, 2002. [Google Scholar]
- Buxton, T.H.; Buffington, J.M.; Yager, E.M.; Hassan, M.A.; Fremier, A.K. The relative stability of salmon redds and unspawned streambeds. Water Resour. Res. 2015, 51, 6074–6092. [Google Scholar] [CrossRef]
- Unfer, G.; Hauer, C.; Lautsch, E. The influence of hydrology on the recruitment of brown trout in an Alpine river, the Ybbs River, Austria. Ecol. Freshw. Fish 2011, 20, 438–448. [Google Scholar] [CrossRef]
- Casas-Mulet, R.; Saltveit, S.J.; Alfredsen, K. The Survival of Atlantic Salmon (Salmo salar) Eggs During Dewatering in a River Subjected to Hydropeaking. River Res. Appl. 2015, 31, 433–446. [Google Scholar] [CrossRef]
- Becker, C.D.; Neitzel, D.A.; Fickeisen, D.H. Effects of dewatering on Chinook salmon redds: Tolerance of four developmental phases to daily dewaterings. Trans. Am. Fish. Soc. 1982, 111, 624–637. [Google Scholar] [CrossRef]
- Becker, C.D.; Neitzel, D.A.; Abernethy, C.S. Effects of Dewatering on Chinook Salmon Redds: Tolerance of Four Development Phases to One-Time Dewatering. N. Am. J. Fish. Manag. 1983, 3, 373–382. [Google Scholar] [CrossRef]
- Becker, C.D.; Neitzel, D.A.; Carlile, D.W. Survival data for dewatered Rainbow Trout (Salmo gairdneri Rich.) eggs and alevins. J. Appl. Ichthyol. 1986, 3, 102–110. [Google Scholar] [CrossRef]
- Saltveit, S.J.; Brabrand, Å. Incubation, hatching and survival of eggs of Atlantic salmon (Salmo salar) in spawning redds influenced by groundwater. Limnologica 2013, 43, 325–331. [Google Scholar] [CrossRef]
- Connor, E.J.; Pflug, D.E. Changes in the Distribution and Density of Pink, Chum, and Chinook Salmon Spawning in the Upper Skagit River in Response to Flow Management Measures. N. Am. J. Fish. Manag. 2004, 24, 835–852. [Google Scholar] [CrossRef]
- Harnish, R.A.; Sharma, R.; McMichael, G.A.; Langshaw, R.B.; Pearsons, T.N. Effect of hydroelectric dam operations on the freshwater productivity of a Columbia River fall Chinook salmon population. Can. J. Fish. Aquat. Sci. 2014, 71, 602–615. [Google Scholar] [CrossRef]
- Irvine, R.L.; Thorley, J.L.; Westcott, R.; Schmidt, D.; Derosa, D. Why do fish strand? An analysis of ten years of flow reduction monitoring data from the Columbia and Kootenay rivers, Canada. River Res. Appl. 2015, 31, 1242–1250. [Google Scholar] [CrossRef]
- Young, P.S.; Cech, J.J.; Thompson, L.C. Hydropower-related pulsed-flow impacts on stream fishes: A brief review, conceptual model, knowledge gaps, and research needs. Rev. Fish Biol. Fish. 2011, 21, 713–731. [Google Scholar] [CrossRef]
- Schmutz, S.; Fohler, N.; Friedrich, T.; Fuhrmann, M.; Graf, W.; Greimel, F.; Höller, N.; Jungwirth, M.; Leitner, P.; Moog, O.; et al. Schwallproblematik an Österreichs Fließgewässern—Ökologische Folgen und Sanierungsmöglichkeiten; Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2013. [Google Scholar]
- Korman, J.; Walters, C.; Martell, S.J.D.; Pine Ill, W.E.; Dutterer, A. Effects of flow fluctuations on habitat use and survival of age-0 rainbow trout (Oncorhynchus mykiss) in a large, regulated river. Can. J. Fish. Aquat. Sci. 2011, 68, 1097–1109. [Google Scholar] [CrossRef]
- Jensen, A.J.; Johnsen, B.O. The functional relationship between peak spring floods and survival and growth of juvenile Atlantic Salmon (Salmo salar) and Brown Trout (Salmo trutta). Funct. Ecol. 1999, 13, 778–785. [Google Scholar] [CrossRef]
- Hurel, G. Impacts du fonctionnement par éclusées d’une usine hydroélectrique sur une population de truites communes (Salmo trutta L.) dans les Pyrénées ariégeoises (09); L’Ecole Nationale Supérieure Agronomique de Toulouse: Toulouse, France, 2010. [Google Scholar]
- Auer, S.; Fohler, N.; Zeiringer, B.; Führer, S. Experimentelle Untersuchungen zur Schwallproblematik. Drift und Stranden von Äschen und Bachforellen während der ersten Lebensstadien; Institute of Hydrobiology and Aquatic Ecosystem Management, University of Natural Resources and Life Sciences, Vienna: Vienna, Austria, 2014. [Google Scholar]
- Bauersfeld, K. Stranding of juvenile salmon by flow reductions at Mayfield Dam on the Cowlitz River, 1976; Washington Department of Fisheries: Tacoma, WA, USA, 1978. [Google Scholar]
- Unfer, G.; Leitner, P.; Graf, W.; Auer, S. Der Einfluss von Schwallbetrieb auf den Fischbestand der Oberen Drau; VERBUND—Austrian Hydro Power AG and Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2011. [Google Scholar]
- Halleraker, J.H.; Sundt, H.; Alfredsen, K.T.; Dangelmaier, G. Application of multiscale environmental flow methodologies as tools for optimized management of a Norwegian regulated national salmon watercourse. River Res. Appl. 2007, 23, 493–510. [Google Scholar] [CrossRef]
- Halleraker, J.H.; Saltveit, S.J.; Harby, A.; Arnekleiv, J.V.; Fjeldstad, H.-P.; Kohler, B. Factors influencing stranding of wild juvenile brown trout (Salmo trutta) during rapid and frequent flow decreases in an artificial stream. River Res. Appl. 2003, 19, 589–603. [Google Scholar] [CrossRef]
- Liebig, H.; Cereghino, R.; Lim, P.; Belaud, A.; Lek, S. Impact of hydropeaking on the abundance of juvenile brown trout in a Pyrenean stream. Arch. für Hydrobiol. 1999, 4, 439–454. [Google Scholar] [CrossRef]
- Jungwirth, M.; Winkler, H. The temperature dependence of embryonic development of grayling (Thymallus thymallus), Danube salmon (Hucho hucho), Arctic char (Salvelinus alpinus) and brown trout (Salmo trutta fario). Aquaculture 1984, 38, 315–327. [Google Scholar] [CrossRef]
- Bradford, M.J. An experimental study of stranding of juvenile salmonids on gravel bars and in sidechannels during rapid flow decreases. Regul. Rivers Res. Manag. 1997, 13, 395–401. [Google Scholar] [CrossRef]
- Bradford, M.J.; Taylor, G.C.; Allan, J.A.; Higgins, P.S. An experimental study of the stranding of juvenile coho salmon and rainbow trout during rapid flow decreases under winter conditions. N. Am. J. Fish. Manag. 1995, 15, 473–479. [Google Scholar] [CrossRef]
- Heggenes, J.; Saltveit, S.J. Summer stream habitat partitioning by sympatric Arctic charr, Atlantic salmon and brown trout in two sub-arctic rivers. J. Fish Biol. 2007, 71, 1069–1081. [Google Scholar] [CrossRef]
- Heggenes, J.; Krog, O.M.W.; Lindås, O.R.; Dokk, J.G.; Bremnes, T. Homeostatic behavioural responses in a changing environment: Brown trout (Salmo trutta) become nocturnal during winter. J. Anim. Ecol. 1993, 62, 295–308. [Google Scholar] [CrossRef]
- Robertson, M.J.; Pennell, C.J.; Scruton, D.A.; Robertson, G.J.; Brown, J.A. Effect of increased flow on the behaviour of Atlantic salmon parr in winter. J. Fish Biol. 2004, 65, 1070–1079. [Google Scholar] [CrossRef]
- Stickler, M.; Alfredsen, K.; Scruton, D.A.; Pennell, C.; Harby, A.; Økland, F. Mid-winter activity and movement of Atlantic salmon parr during ice formation events in a Norwegian regulated river. Hydrobiologia 2007, 582, 81–89. [Google Scholar] [CrossRef]
- Berland, G.; Nickelsen, T.; Heggenes, J.; Okland, F.; Thorstad, E.B.; Halleraker, J. Movements of wild Atlantic salmon parr in relation to peaking flows below a hydropower station. River Res. Appl. 2004, 20, 957–966. [Google Scholar] [CrossRef]
- Puffer, M.; Berg, O.K.; Huusko, A.; Vehanen, T.; Forseth, T.; Einum, S. Seasonal Effects of Hydropeaking on Growth, Energetics and Movement of Juvenile Atlantic Salmon (Salmo salar). River Res. Appl. 2015, 31, 1101–1108. [Google Scholar] [CrossRef]
- Boavida, I.; Harby, A.; Clarke, K.D.; Heggenes, J. Move or stay: Habitat use and movements by Atlantic salmon parr (Salmo salar) during induced rapid flow variations. Hydrobiologia 2017, 785, 261–275. [Google Scholar] [CrossRef]
- Holzapfel, P.; Leitner, P.; Habersack, H.; Graf, W.; Hauer, C. Evaluation of hydropeaking impacts on the food web in alpine streams based on modelling of fish- and macroinvertebrate habitats. Sci. Total Environ. 2017, 575, 1489–1502. [Google Scholar] [CrossRef] [PubMed]
- Sauterleute, J.F.; Hedger, R.D.; Hauer, C.; Pulg, U.; Skoglund, H.; Sundt-Hansen, L.E.; Bakken, T.H.; Ugedal, O. Modelling the effects of stranding on the Atlantic salmon population in the Dale River, Norway. Sci. Total Environ. 2016, 573, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Scruton, D.A.; Pennell, C.J.; Robertson, M.J.; Ollerhead, L.M.N.; Clarke, K.D.; Alfredsen, K.; Harby, A.; McKinsley, R.S. Seasonal Response of Juvenile Atlantic Salmon to Experimental Hydropeaking Power Generation in Newfoundland, Canada. N. Am. J. Fish. Manag. 2005, 25, 964–974. [Google Scholar] [CrossRef]
- Scruton, D.A.; Pennell, C.; Ollerhead, L.M.N.; Alfredsen, K.; Stickler, M.; Harby, A.; Robertson, M.; Clarke, K.D.; LeDrew, L.J. A synopsis of “hydropeaking” studies on the response of juvenile Atlantic salmon to experimental flow alteration. Hydrobiologia 2008, 609, 263–275. [Google Scholar] [CrossRef]
- Casas-Mulet, R.; Alfredsen, K.; Killingtveit, A. Modelling of environmental flow options for optimal Atlantic salmon, Salmo salar, embryo survival during hydropeaking. Fish. Manag. Ecol. 2014, 21, 480–490. [Google Scholar] [CrossRef]
- Yarnell, S.M.; Petts, G.E.; Schmidt, J.C.; Whipple, A.A.; Beller, E.E.; Dahm, C.N.; Goodwin, P.; Viers, J.H. Functional Flows in Modified Riverscapes: Hydrographs, Habitats and Opportunities. Bioscience 2015, 65, 963–972. [Google Scholar] [CrossRef]
- Naiman, R.J.; Latterell, J.J.; Pettit, N.E.; Olden, J.D. Flow variability and the biophysical vitality of river systems. C. R. Geosci. 2008, 340, 629–643. [Google Scholar] [CrossRef]
- Greimel, F.; Zeiringer, B.; Höller, N.; Grün, B.; Godina, R.; Schmutz, S. A method to detect and characterize sub-daily flow fluctuations. Hydrol. Process. 2016, 30, 2063–2078. [Google Scholar] [CrossRef]
- Hauer, C.; Unfer, G.; Holzapfel, P.; Haimann, M.; Habersack, H. Impact of channel bar form and grain size variability on estimated stranding risk of juvenile brown trout during hydropeaking. Earth Surf. Process. Landf. 2014, 39, 1622–1641. [Google Scholar] [CrossRef]
- Tuhtan, J.A.; Noack, M.; Wieprecht, S. Estimating stranding risk due to hydropeaking for juvenile European grayling considering river morphology. KSCE J. Civ. Eng. 2012, 16, 197–206. [Google Scholar] [CrossRef]
- Hauer, C.; Unfer, G.; Graf, W.; Holzapfel, P.; Leitner, P.; Habersack, H. Grundlagenuntersuchungen und Methodikentwicklung zur Bewertung des Wasserkraft-Schwalls bei unterschiedlichen Flusstypen. Österreichische Wasser- und Abfallwirtschaft 2013, 65, 324–338. [Google Scholar] [CrossRef]
- Harby, A.; Noack, M. Rapid Flow Fluctuations and Impacts on Fish and the Aquatic Ecosystem. In Ecohydraulics: An Integrated Approach; Maddock, I., Harby, A., Kemp, P., Wood, P., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; pp. 323–335. [Google Scholar]
- Zolezzi, G.; Siviglia, A.; Toffolon, M.; Maiolini, B. Thermopeaking in alpine streams: Event characterization and time scales. Ecohydrology 2011, 4, 564–576. [Google Scholar] [CrossRef]
- Olden, J.D.; Naiman, R.J. Incorporating thermal regimes into environmental flows assessments: Modifying dam operations to restore freshwater ecosystem integrity. Freshw. Biol. 2010, 55, 86–107. [Google Scholar] [CrossRef]
- Schülting, L.; Feld, C.K.; Graf, W. Effects of hydro- and thermopeaking on benthic macroinvertebrate drift. Sci. Total Environ. 2016, 573, 1472–1480. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.B.; Webster, J.R. The role of macroinvertebrates in stream ecosystem function. Annu. Rev. Entomol. 1996, 41, 115–139. [Google Scholar] [CrossRef] [PubMed]
- Pulg, U.; Vollset, K.W.; Velle, G.; Stranzl, S. First observations of saturopeaking: Characteristics and implications. Sci. Total Environ. 2016, 573, 1615–1621. [Google Scholar] [CrossRef] [PubMed]
- Lumsdon, A.E.; Artamonov, I.; Bruno, M.C.; Righetti, M.; Tockner, K.; Tonolla, D.; Zarfl, C. Soundpeaking—Hydropeaking induced changes in river soundscapes. River Res. Appl. 2018, 34, 3–12. [Google Scholar] [CrossRef]
- Tonolla, D.; Bruder, A.; Schweizer, S. Evaluation of mitigation measures to reduce hydropeaking impacts on river ecosystems—A case study from the Swiss Alps. Sci. Total Environ. 2017, 574, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Pragana, I.; Boavida, I.; Cortes, R.; Pinheiro, A. Hydropower Plant Operation Scenarios to Improve Brown Trout Habitat. River Res. Appl. 2017, 33, 364–376. [Google Scholar] [CrossRef]
- L’Abée-Lund, J.H.; Otero, J. Hydropeaking in small hydropower in Norway—Compliance with license conditions? River Res. Appl. 2018, 34, 372–381. [Google Scholar] [CrossRef]
- Costa, M.J.; Boavida, I.; Almeida, V.; Cooke, S.J.; Pinheiro, A.N. Do artificial velocity refuges mitigate the physiological and behavioural consequences of hydropeaking on a freshwater Iberian cyprinid? Ecohydrology 2018, 11, e1983. [Google Scholar] [CrossRef]
- Storli, P.-T. Novel methods of increasing the storage volume at Pumped Storage Power plants. Int. J. Fluid Mach. Syst. 2017, 10, 209–2017. [Google Scholar] [CrossRef]
- HydroFlex. Social Acceptance and Mitigation of Environmental Impact. Available online: https://h2020hydroflex.eu/work-packages/wp-5/ (accessed on 6 March 2019).
- Northcote, T.G.; Lobón-Cerviá, J. Increasing experimental approaches in stream trout research—1987–2006. Ecol. Freshw. Fish 2008, 17, 349–361. [Google Scholar] [CrossRef]
- Northcote, T.G. Comparative biology and management of Arctic and European grayling (Salmonidae, Thymallus). Rev. Fish Biol. Fish. 1995, 5, 141–194. [Google Scholar] [CrossRef]
- Jonsson, B.; Jonsson, N. A review of the likely effects of climate change on anadromous Atlantic salmon Salmo salar and brown trout Salmo trutta, with particular reference to water temperature and flow. J. Fish Biol. 2009, 75, 2381–2447. [Google Scholar] [CrossRef] [PubMed]
- Bejarano, M.D.; Jansson, R.; Nilsson, C. The effects of hydropeaking on riverine plants: A review. Biol. Rev. 2018, 93, 658–673. [Google Scholar] [CrossRef]
- Lucas, M.C.; Baras, E. Migration of Freshwater Fishes; Blackwell Science Ltd.: Hoboken, NJ, USA, 2001. [Google Scholar]
- Crisp, D.T. Environmental requirements of common riverine European salmonid fish species in fresh water with particular reference to physical and chemical aspects. Hydrobiologia 1996, 323, 201–221. [Google Scholar] [CrossRef]
- Lobón-Cerviá, J.; Sanz, N. (Eds.) Brown Trout: Biology, Ecology and Management; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018. [Google Scholar]
- Fabricius, E.; Gustafson, K.-J. Observations on the spawning behaviour of the grayling, Thymallus thymallus (L.). Rep. Inst. Freshw. Res. Drottningholm 1955, 36, 75–103. [Google Scholar]
- Gibson, R.J. The Atlantic salmon in fresh water: Spawning, rearing and production. Rev. Fish Biol. Fish. 1993, 3, 39–73. [Google Scholar] [CrossRef]
- Jonsson, B.; Jonsson, N. Ecology of Atlantic Salmon and Brown Trout; Springer Science & Business Media: Berlin, Germany, 2011. [Google Scholar]
- Flodmark, L.E.W.; Vøllestad, L.A.; Forseth, T. Performance of juvenile brown trout exposed to fluctuating water level and temperature. J. Fish Biol. 2004, 65, 460–470. [Google Scholar] [CrossRef]
- Vehanen, T.; Huusko, A.; Yrjänä, T.; Lahti, M.; Mäki-Petäys, A. Habitat preference by grayling (Thymallus thymallus) in an artificially modified, hydropeaking riverbed: A contribution to understand the effectiveness of habitat enhancement measures. J. Appl. Ichthyol. 2003, 19, 15–20. [Google Scholar] [CrossRef]
| Life-History Stage (and Activity) | Ecological Research | Literature Example(s) 1 | Hydropeaking Research 2 | Literature Example(s) | Particularities on Hydropeaking Studies | |
|---|---|---|---|---|---|---|
| Spawning | Migration | + | [20,98,100,101,102] | – | [22,23] | |
| Behavior 3 | + | [27,102,103] | – | [24,25,26] | ||
| Intragravel life stages | Egg incubation | + | [19,20,101] | ± | [31,43,44,45,46] | Aside from studies on short- or long-term desiccation, information on repeated wetting and drying is largely missing for different species. |
| Alevin | + | [19,20,98] | – | [44] | ||
| Young-of-the-year (0+) | Fry | + | [97,98] | ± | [52,55,56,57] | Only studies regarding few selected species, topics mostly restricted to stranding and drift. |
| Parr | + | [19,20,104] | ± | [5,6,70,71] | Only studies regarding few selected species, topics mostly restricted to drift, stranding, movements, habitat use, and growth. | |
| Juvenile | 1+ fish (smolt) | + | [19,102,104,105] | – | [61,106] | |
| Adult | 2+ fish | + | [105] | – | [107] | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayes, D.S.; Moreira, M.; Boavida, I.; Haslauer, M.; Unfer, G.; Zeiringer, B.; Greimel, F.; Auer, S.; Ferreira, T.; Schmutz, S. Life Stage-Specific Hydropeaking Flow Rules. Sustainability 2019, 11, 1547. https://doi.org/10.3390/su11061547
Hayes DS, Moreira M, Boavida I, Haslauer M, Unfer G, Zeiringer B, Greimel F, Auer S, Ferreira T, Schmutz S. Life Stage-Specific Hydropeaking Flow Rules. Sustainability. 2019; 11(6):1547. https://doi.org/10.3390/su11061547
Chicago/Turabian StyleHayes, Daniel S., Miguel Moreira, Isabel Boavida, Melanie Haslauer, Günther Unfer, Bernhard Zeiringer, Franz Greimel, Stefan Auer, Teresa Ferreira, and Stefan Schmutz. 2019. "Life Stage-Specific Hydropeaking Flow Rules" Sustainability 11, no. 6: 1547. https://doi.org/10.3390/su11061547
APA StyleHayes, D. S., Moreira, M., Boavida, I., Haslauer, M., Unfer, G., Zeiringer, B., Greimel, F., Auer, S., Ferreira, T., & Schmutz, S. (2019). Life Stage-Specific Hydropeaking Flow Rules. Sustainability, 11(6), 1547. https://doi.org/10.3390/su11061547






