Repeated-Batch Fermentation of Cheese Whey for Semi-Continuous Lactic Acid Production Using Mixed Cultures at Uncontrolled pH
Abstract
:1. Introduction
2. Materials and Methods
2.1. Substrate and Inoculum
2.2. Experimental Setup and Operational Conditions
2.2.1. Dark Fermentation Bioreactor
2.2.2. Experimental Conditions
2.3. Analytical Methods
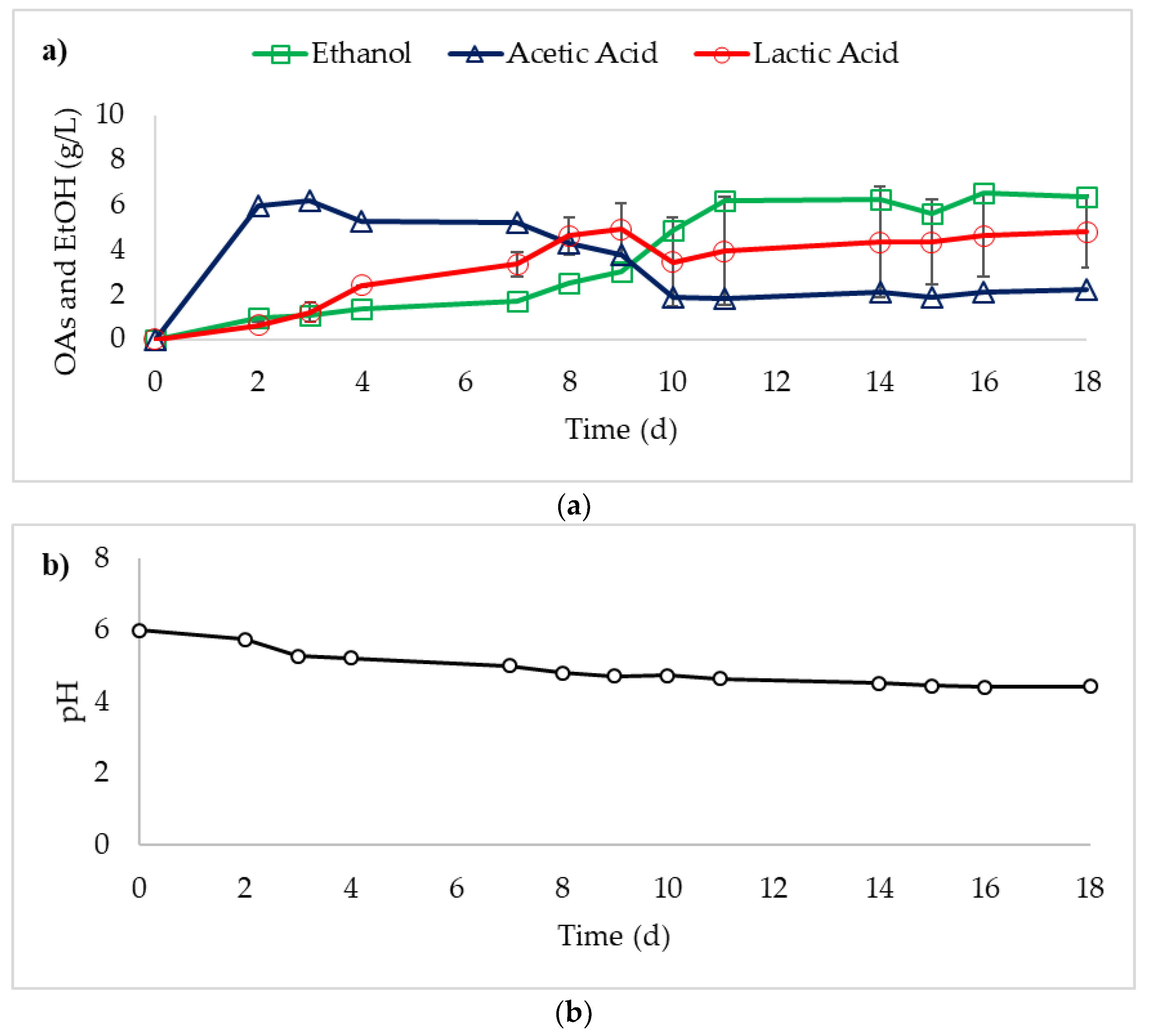
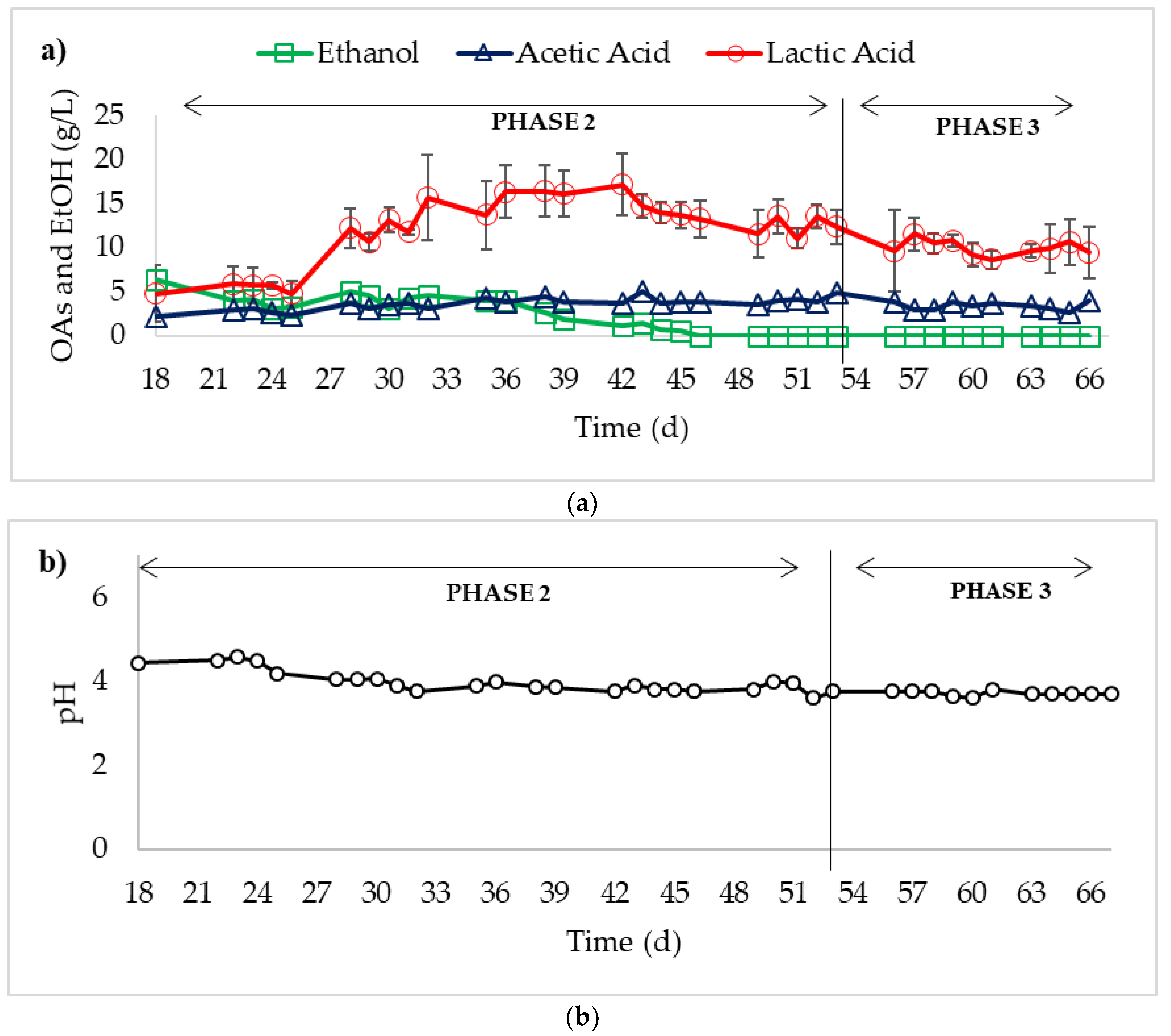
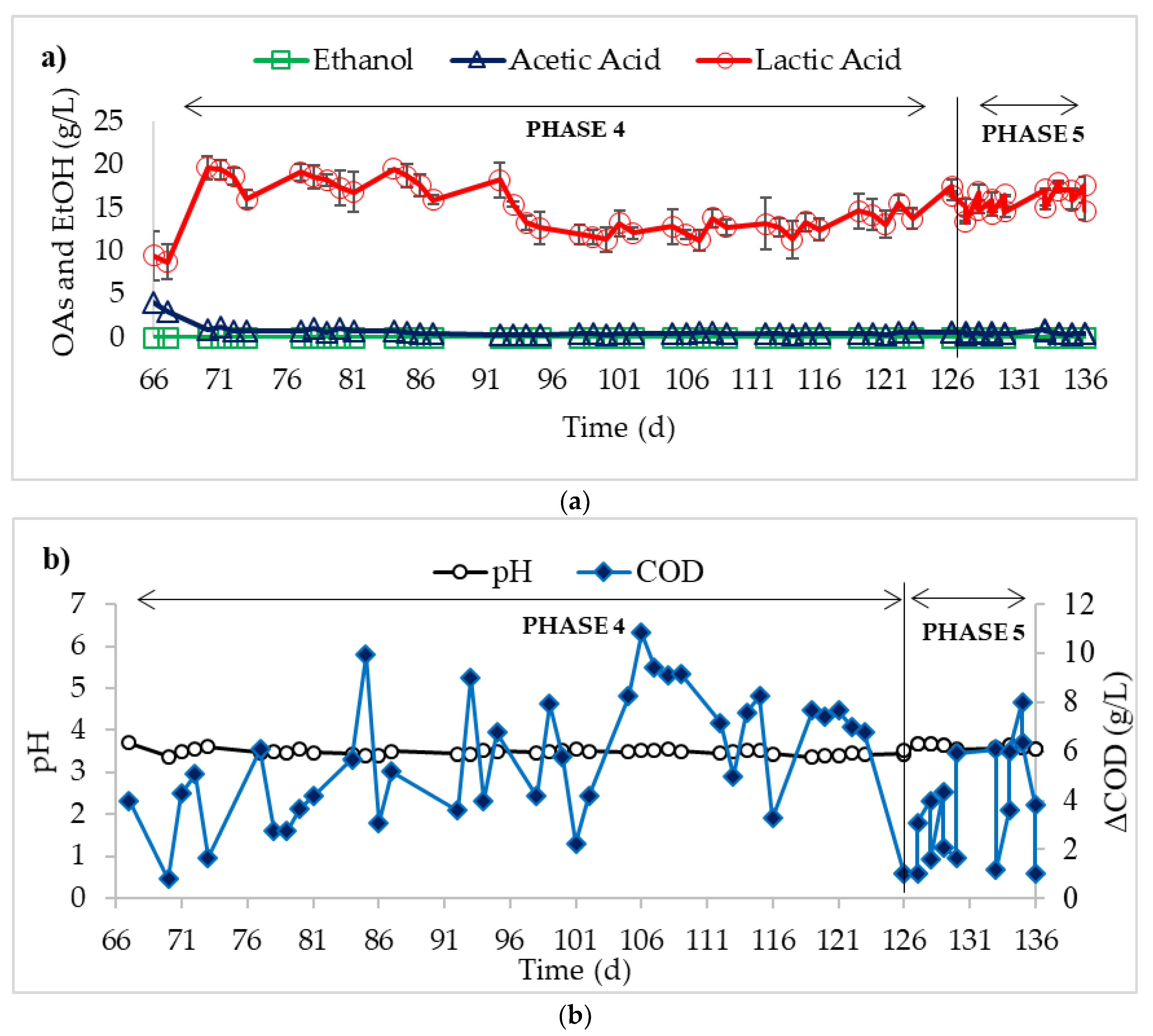
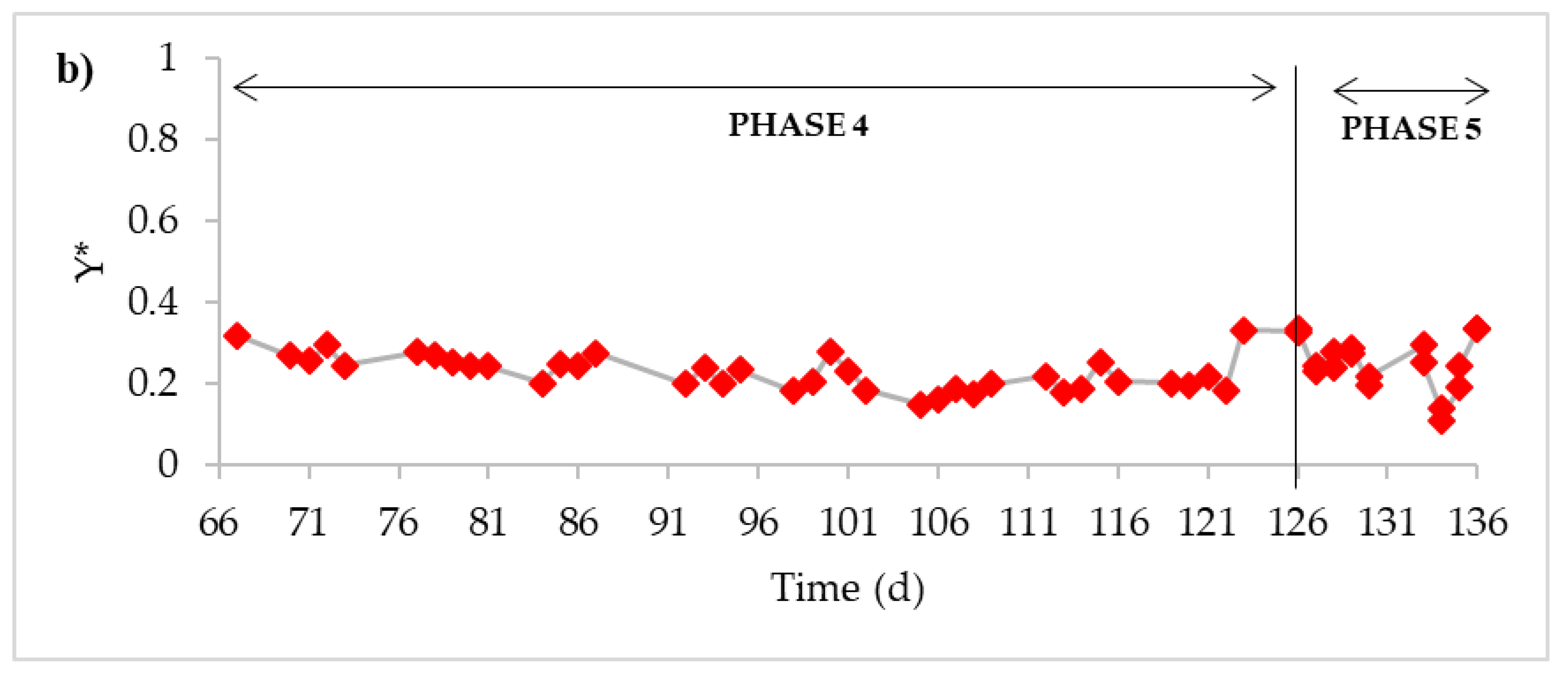
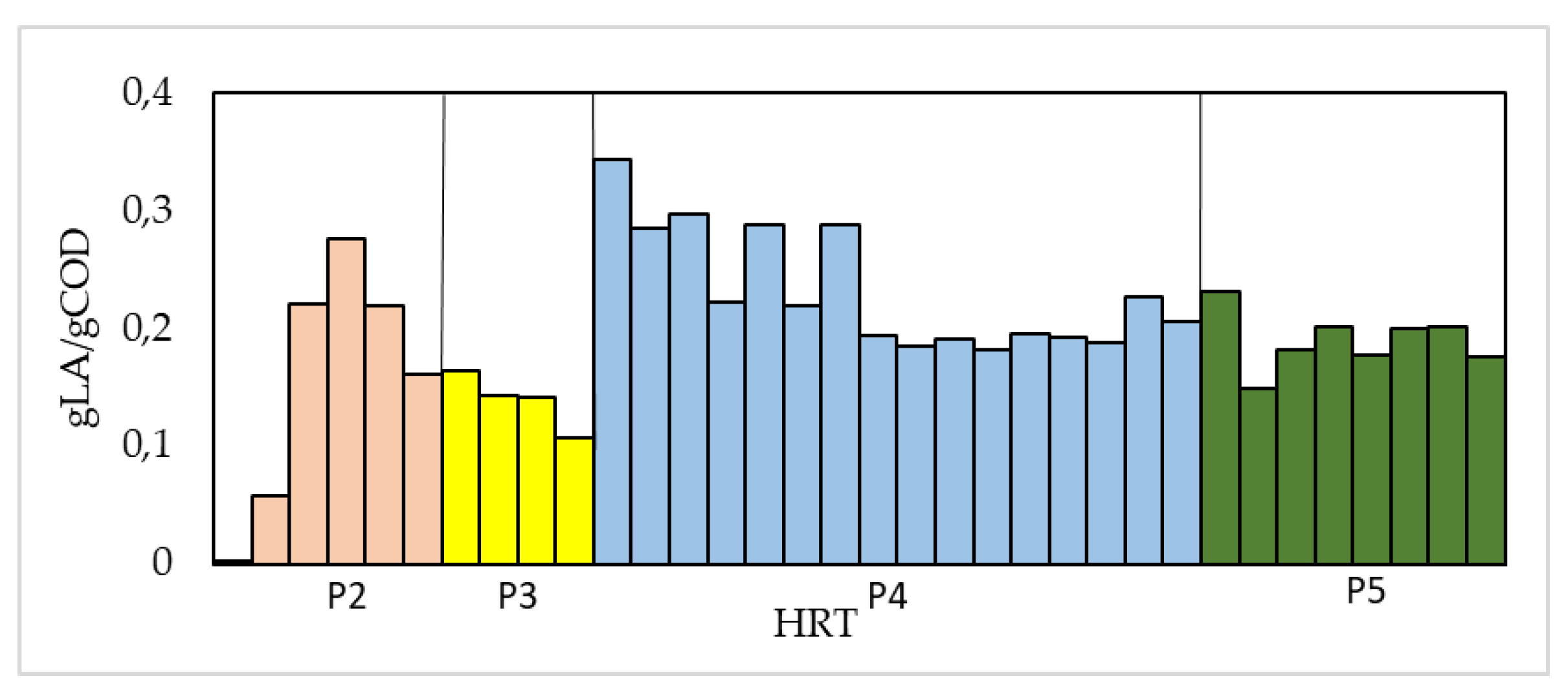
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ghimire, A.; Frunzo, L.; Salzano, E.; Panico, A.; Lens, P.N.L.; Pirozzi, F. Biomass Enrichment and Scale-up Implications for Dark Fermentation Hydrogen Production with Mixed Cultures. Chem. Eng. Trans. 2015, 43, 391–396. [Google Scholar]
- Carvalho, F.; Prazeres, A.R.; Rivas, J. Cheese whey wastewater: Characterization and treatment. Sci. Total Environ. 2013, 445–446, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Prazeres, A.R.; Carvalho, F.; Rivas, J. Cheese whey management: A review. J. Environ. Manag. 2012, 110, 48–68. [Google Scholar] [CrossRef] [PubMed]
- Ibarruri, J.; Hernández, I. Valorization of cheese whey and orange molasses for fungal biomass production by submerged fermentation with Rhizopus sp. Bioprocess Biosyst. Eng. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Sharma, P.; Jaimni, S.; Kehinde, B.A.; Kaur, S. Recent developments in purification techniques and industrial applications for whey valorization: A review. Chem. Eng. Commun. 2019, 1–16. [Google Scholar] [CrossRef]
- Cherubini, F. The biorefinery concept: Using biomass instead of oil for producing energy and chemicals. Energy Convers. Manag. 2010, 51, 1412–1421. [Google Scholar] [CrossRef]
- Eş, I.; Mousavi Khaneghah, A.; Barba, F.J.; Saraiva, J.A.; Sant’Ana, A.S.; Hashemi, S.M.B. Recent advancements in lactic acid production-a review. Food Res. Int. 2018, 107, 763–770. [Google Scholar] [CrossRef]
- Dorgan, J.R.; Lehermeier, H.J.; Palade, L.-I.; Cicero, J. Polylactides: Properties and Prospects of an Environmentally Benigh Plastic from Renewable Resources. Macromol. Symp. 2001, 175, 55–66. [Google Scholar] [CrossRef]
- Bogaert, J.-C.; Coszach, P. Poly(lactic acids)- a potential solution to plastic waste dilemma. Macromol. Symp. 2000, 153, 287–303. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Recent advances in lactic acid production by microbial fermentation processes. Biotechnol. Adv. 2013, 31, 877–902. [Google Scholar] [CrossRef]
- Tan, J.; Mohamed Ali Abdel-Rahman, A.; Sonomoto, K. Biorefinery-Based Lactic Acid Fermentation: Microbial Production of Pure Monomer Product. Adv. Polym. Sci. 2018, 279, 27–66. [Google Scholar]
- Shahbazi, A.; Mims, M.R.; Li, Y.; Shirley, V.; Ibrahim, S.A.; Morris, A. Lactic Acid Production from Cheese Whey by Immobilized Bacteria. Appl. Biochem. Biotechnol. 2005, 122, 0529–0540. [Google Scholar] [CrossRef]
- Soriano-Perez, S.; Flores-Velez, L.; Alonso-Davila, P.; Cervantes-Cruz, G.; Arriaga, S. Production of lactic acid from cheese whey by batch cultures of Lactobacillus helveticus. Ann. Microbiol. 2012, 62, 313–317. [Google Scholar] [CrossRef]
- Itoh, Y.; Tada, K.; Kanno, T.; Horiuchi, J.I. Selective production of lactic acid in continuous anaerobic acidogenesis by extremely low pH operation. J. Biosci. Bioeng. 2012, 114, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, N.; Dipasquale, L.; D’Ippolito, G.; Panico, A.; Lens, P.N.L.; Esposito, G.; Fontana, A. Hydrogen production by the thermophilic bacterium Thermotoga neapolitana. Int. J. Mol. Sci. 2015, 16, 12578–12600. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; McDonald, A.G.; Coats, E.R. Lactic acid production from potato peel waste by anaerobic sequencing batch fermentation using undefined mixed culture. Waste Manag. 2015, 45, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Bonk, F.; Bastidas-Oyanedel, J.R.; Yousef, A.F.; Schmidt, J.E.; Bonk, F. Exploring the selective lactic acid production from food waste in uncontrolled pH mixed culture fermentations using different reactor configurations. Bioresour. Technol. 2017, 238, 416–424. [Google Scholar] [CrossRef]
- Tejayadi, S.; Cheryan, M. Lactic acid from cheese whey permeate. Productivity and economics of a continuous membrane bioreactor. Appl. Microbiol. Biotechnol. 1995, 43, 242–248. [Google Scholar] [CrossRef]
- Motte, J.C.; Sambusiti, C.; Dumas, C.; Barakat, A. Combination of dry dark fermentation and mechanical pretreatment for lignocellulosic deconstruction: An innovative strategy for biofuels and volatile fatty acids recovery. Appl. Energy 2015, 147, 67–73. [Google Scholar] [CrossRef]
- Bastidas-Oyanedel, J.-R.; Bonk, F.; Thomsen, M.H.; Schmidt, J.E. Dark fermentation biorefinery in the present and future (bio) chemical industry. Rev. Environ. Sci. Biol. Technol. 2015, 14, 473–498. [Google Scholar] [CrossRef]
- Ghimire, A.; Trably, E.; Frunzo, L.; Pirozzi, F.; Lens, P.N.L.; Esposito, G.; Cazier, E.A.; Escudié, R. Effect of total solids content on biohydrogen production and lactic acid accumulation during dark fermentation of organic waste biomass. Bioresour. Technol. 2018, 248, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, A.; Frunzo, L.; Pirozzi, F.; Trably, E.; Escudie, R.; Lens, P.N.L.; Esposito, G. A review on dark fermentative biohydrogen production from organic biomass: Process parameters and use of by-products. Appl. Energy 2015, 144, 73–95. [Google Scholar] [CrossRef]
- Maharaj, B.C.; Mattei, M.R.; Frunzo, L.; va Hullebusch, E.D.; Esposito, G. ADM1 based mathematical model of trace element complexation in anaerobic digestion processes. Bioresour. Technol. 2019, 276, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Luongo, V.; Ghimire, A.; Frunzo, L.; Fabbricino, M.; d’Antonio, G.; Pirozzi, F.; Esposito, G. Photofermentative production of hydrogen and poly-β-hydroxybutyrate from dark fermentation products. Bioresour. Technol. 2017, 228, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, A.; Luongo, V.; Frunzo, L.; Pirozzi, F.; Lens, P.N.L.; Esposito, G. Continuous biohydrogen production by thermophilic dark fermentation of cheese whey: Use of buffalo manure as buffering agent. Int. J. Hydrogen Energy 2017, 42, 4861–4869. [Google Scholar] [CrossRef]
- Ragsdale, S.W. Enzymology of the Wood-Ljungdahl pathway of acetogenesis. Ann. N. Y. Acad. Sci. 2008, 1125, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Djukić-Vuković, A.; Mladenović, D.; Ivanović, J.; Pejin, J.; Mojović, L. Towards sustainability of lactic acid and poly-lactic acid polymers production. Renew. Sustain. Energy Rev. 2019, 108, 238–252. [Google Scholar] [CrossRef]
- Hofvendahl, K.; Hahn–Hägerdal, B. Factors affecting the fermentative lactic acid production from renewable resources1. Enzyme Microb. Technol. 2000, 26, 87–107. [Google Scholar] [CrossRef]
- Vijayakumar, J.; Aravindan, R.; Viruthagiri, T. Recent Trends in the Production, Purification and Application of Lactic Acid. Chem. Biochem. Eng. 2008, 22, 245–264. [Google Scholar]
- Ghimire, A.; Sposito, F.; Frunzo, L.; Trably, E.; Escudié, R.; Pirozzi, F.; Lens, P.N.L.; Esposito, G. Effects of operational parameters on dark fermentative hydrogen production from biodegradable complex waste biomass. Waste Manag. 2016, 50, 55–64. [Google Scholar] [CrossRef]
- Maharaj, B.C.; Mattei, M.R.; Frunzo, L.; van Hullebusch, E.D.; Esposito, G. ADM1 based mathematical model of trace element precipitation/dissolution in anaerobic digestion processes. Bioresour. Technol. 2018, 267, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Frunzo, L.; Fermoso, F.G.; Luongo, V.; Mattei, M.R.; Esposito, G. ADM1-based mechanistic model for the role of trace elements in anaerobic digestion processes. J. Environ. Manag. 2019, 241, 587–602. [Google Scholar] [CrossRef] [PubMed]
- Colombo, B.; Sciarria, T.P.; Reis, M.; Scaglia, B.; Adani, F. Polyhydroxyalkanoates (PHAs) production from fermented cheese whey by using a mixed microbial culture. Bioresour. Technol. 2016, 218, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.K.; Chen, W.H.; Li, L.; Sung, S. Biological hydrogen production: Effects of pH and intermediate products. Int. J. Hydrogen Energy 2004, 29, 1123–1131. [Google Scholar] [CrossRef]
- Gottwald, M.; Gottschalk, G. The internal pH of Clostridium acetobutylicum and its effect on the shift from acid to solvent formation. Arch. Microbiol. 1985, 143, 42–46. [Google Scholar] [CrossRef]
- Hutkins, R.W.; Nannen, N.L. pH Homeostasis in Lactic Acid Bacteria. J. Dairy Sci. 1993, 76, 2354–2365. [Google Scholar] [CrossRef]
- Corcoran, B.M.; Stanton, C.; Fitzgerald, G.F.; Ross, R.P. Survival of probiotic lactobacilli in acidic environments is enhanced in the presence of metabolizable sugars. Appl. Environ. Microbiol. 2005, 71, 3060–3067. [Google Scholar] [CrossRef]
- Choi, G.; Kim, J.; Lee, C. Effect of low pH start-up on continuous mixed-culture lactic acid fermentation of dairy effluent. Appl. Microbiol. Biotechnol. 2016, 100, 10179–10191. [Google Scholar] [CrossRef]
- Akao, S.; Tsuno, H.; Horie, T.; Mori, S. Effects of pH and temperature on products and bacterial community in l-lactate batch fermentation of garbage under unsterile condition. Water Res. 2007, 41, 2636–2642. [Google Scholar] [CrossRef]
- Roy, D.; Goulet, J.; LeDuy, A. Batch fermentation of whey ultrafiltrate by Lactobacillus helveticus for lactic acid production. Appl. Microbiol. Biotechnol. 1986, 24, 206–213. [Google Scholar] [CrossRef]
- Wu, Y.; Ma, H.; Zheng, M.; Wang, K. Lactic acid production from acidogenic fermentation of fruit and vegetable wastes. Bioresour. Technol. 2015, 191, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Ghaly, A.E.; Tango, M.S.A.; Mahmoud, N.S.; Avery, A.C. Batch propagation of Lactobacillus helveticus for production of lactic acid from lactose concentrated cheese whey with microaeration and nutrient supplementation. World J. Microbiol. Biotechnol. 2004, 20, 65–75. [Google Scholar] [CrossRef]
- Juodeikiene, G.; Zadeike, D.; Bartkiene, E.; Klupsaite, D. Application of acid tolerant Pedioccocus strains for increasing the sustainability of lactic acid production from cheese whey. LWT Food Sci. Technol. 2016, 72, 399–406. [Google Scholar] [CrossRef]
- Tango, M.S.A.; Ghaly, A.E. Effect of nutrient addition on batch production of lactic acid from cheese whey using Lactobacillus helveticus under batch conditions. Biomass Bioenergy 1998, 16, 61–78. [Google Scholar] [CrossRef]
- Ferraro, A.; Fabbricino, M.; van Hullebusch, E.D.; Esposito, G. Investigation of different ethylenediamine-N,N′-disuccinic acid-enhanced washing configurations for remediation of a Cu-contaminated soil: Process kinetics and efficiency comparison between single-stage and multi-stage configurations. Environ. Sci. Pollut. Res. 2017, 24, 21960–21972. [Google Scholar] [CrossRef] [PubMed]
- Mattei, M.R.; Frunzo, L.; D’Acunto, B.; Pechaud, Y.; Pirozzi, F.; Esposito, G. Continuum and discrete approach in modeling biofilm development and structure: A review. J. Math. Biol. 2018, 76, 945–1003. [Google Scholar] [CrossRef] [PubMed]
| TS (g/L) | VS (g/L) | COD (gCOD/L) | pH (-) | Soluble Carbohydrates (g/L) | |
|---|---|---|---|---|---|
| CW | 47.1 ± 2.5 | 31 ± 2 | 41 ± 2 | 5.6 | 31 ± 2 |
| Inoculum | 51 ± 2 | 31 ± 2 | 41 ± 2 | 7.5 | - |
| Phase | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| OLR (kg VS/m3/day) | batch | 19.6 | 32.4 | 32.4 | 49 |
| HRT (days) | batch | 4 | 2 | 2 | 1 |
| Time length (days) | 18 | 31 | 11 | 53 | 10 |
| Phase | Y (gCOD(LA)/gCOD(CW)) | P (gLA/L) | YMAX (gCOD(LA)/gCOD(CW)) | PMAX (gLA/L) | Y*MAX % |
|---|---|---|---|---|---|
| 2 | 0.191 ± 2.09 | 2.261 ± 2.017 | 0.33 | 3.75 | - |
| 3 | 0.141 ± 2.02 | 3.401 ± 2.287 | 0.2 | 4.55 | - |
| 4 | 0.241 ± 2.05 | 5.671 ± 2.626 | 0.37 | 8.55 | 0.33 |
| 5 | 0.201 ± 2.02 | 9.191 ± 2.289 | 0.23 | 10.6 | 0.33 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luongo, V.; Policastro, G.; Ghimire, A.; Pirozzi, F.; Fabbricino, M. Repeated-Batch Fermentation of Cheese Whey for Semi-Continuous Lactic Acid Production Using Mixed Cultures at Uncontrolled pH. Sustainability 2019, 11, 3330. https://doi.org/10.3390/su11123330
Luongo V, Policastro G, Ghimire A, Pirozzi F, Fabbricino M. Repeated-Batch Fermentation of Cheese Whey for Semi-Continuous Lactic Acid Production Using Mixed Cultures at Uncontrolled pH. Sustainability. 2019; 11(12):3330. https://doi.org/10.3390/su11123330
Chicago/Turabian StyleLuongo, Vincenzo, Grazia Policastro, Anish Ghimire, Francesco Pirozzi, and Massimiliano Fabbricino. 2019. "Repeated-Batch Fermentation of Cheese Whey for Semi-Continuous Lactic Acid Production Using Mixed Cultures at Uncontrolled pH" Sustainability 11, no. 12: 3330. https://doi.org/10.3390/su11123330
APA StyleLuongo, V., Policastro, G., Ghimire, A., Pirozzi, F., & Fabbricino, M. (2019). Repeated-Batch Fermentation of Cheese Whey for Semi-Continuous Lactic Acid Production Using Mixed Cultures at Uncontrolled pH. Sustainability, 11(12), 3330. https://doi.org/10.3390/su11123330









