Chicago’s Urban Cemeteries as Habitat for Cavity-Nesting Birds
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Local-Scale Habitat Assessment
2.3. Landscape-Scale Habitat Assessment
2.4. Point Counts
2.5. Analysis
2.5.1. Cavities
2.5.2. Habitat Selection Models for Individual Cavity-Nesting Bird Species
2.5.3. Richness of Cavity-Nesting Bird Species
3. Results
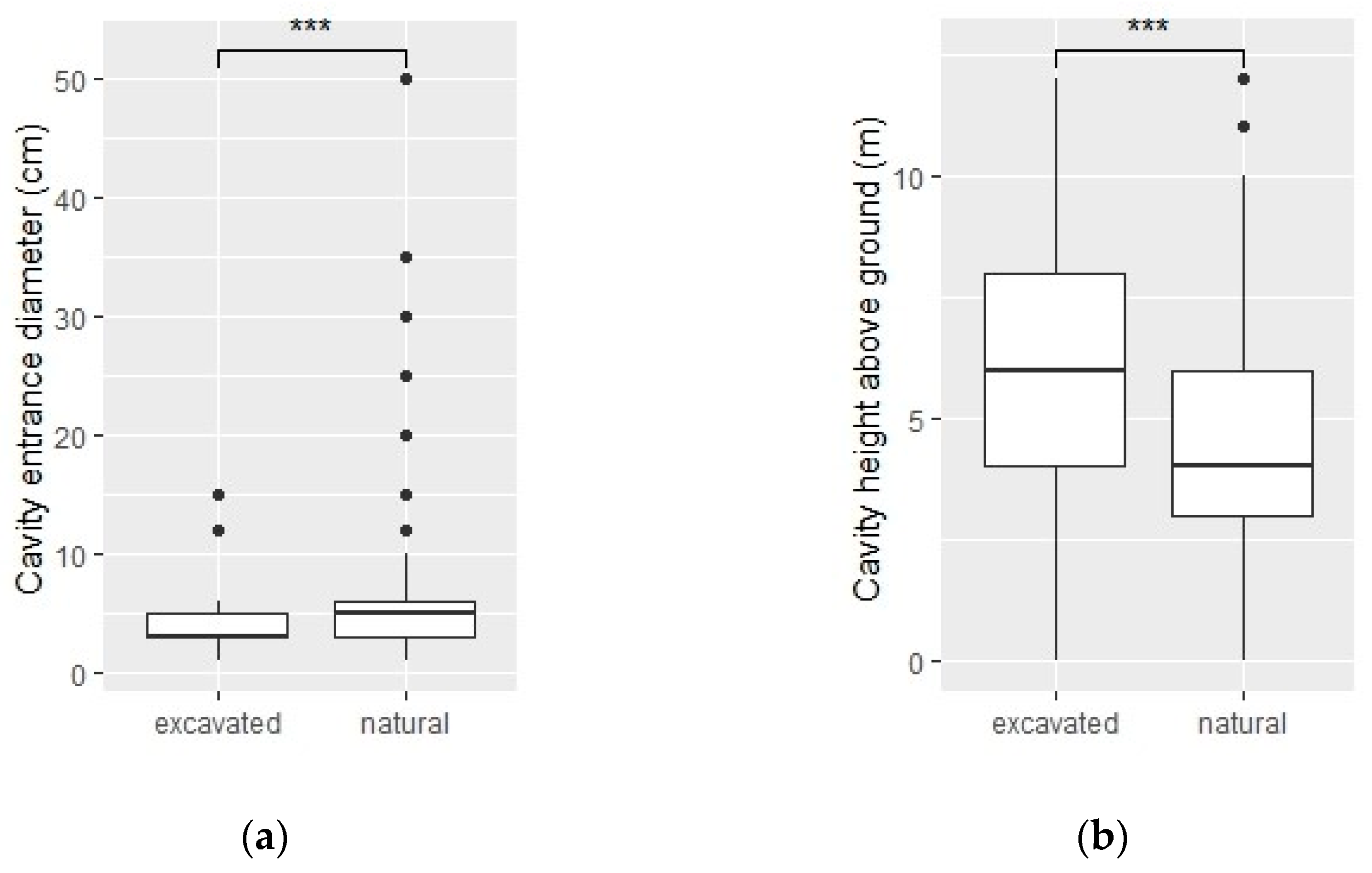
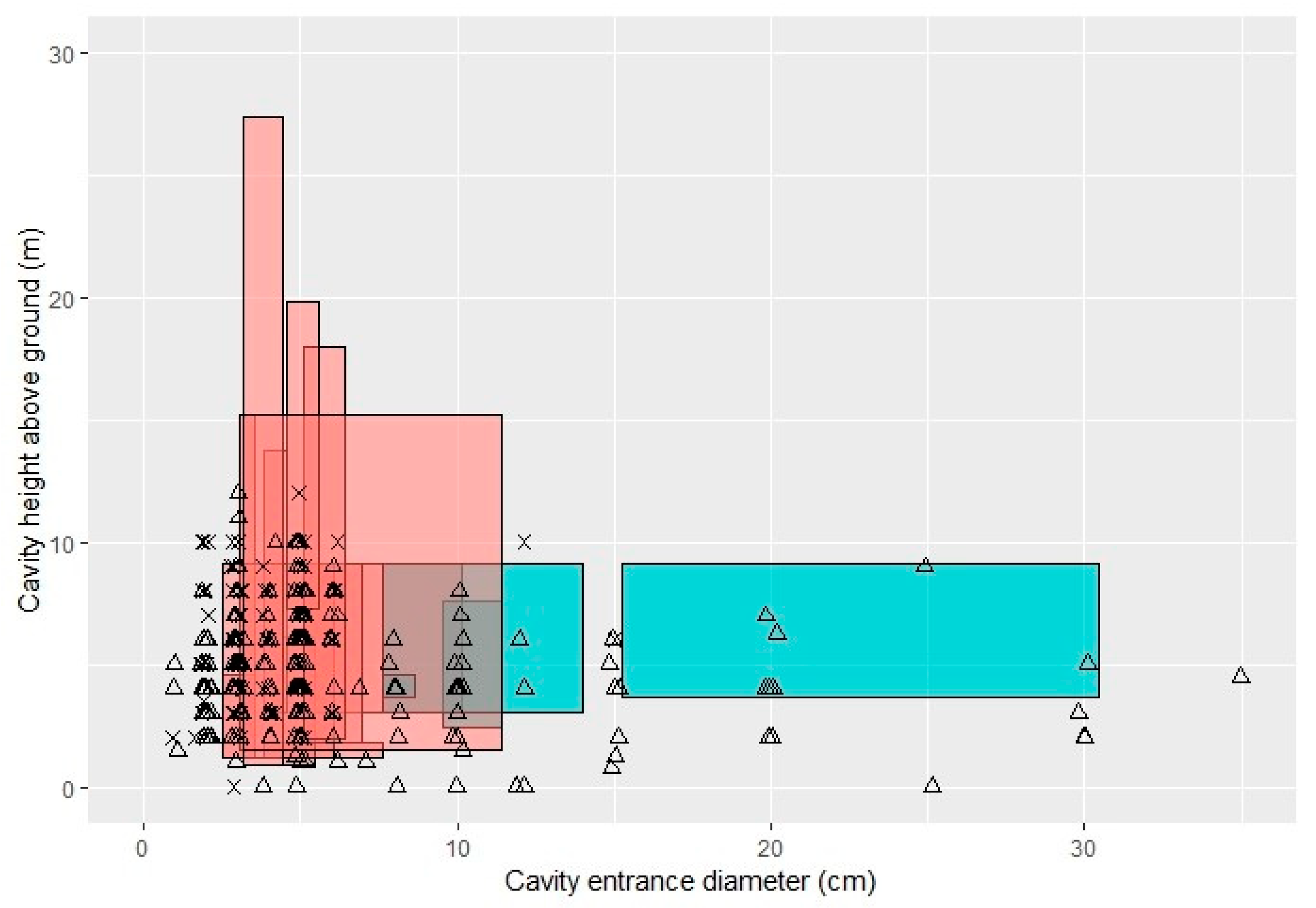
3.1. Cavities
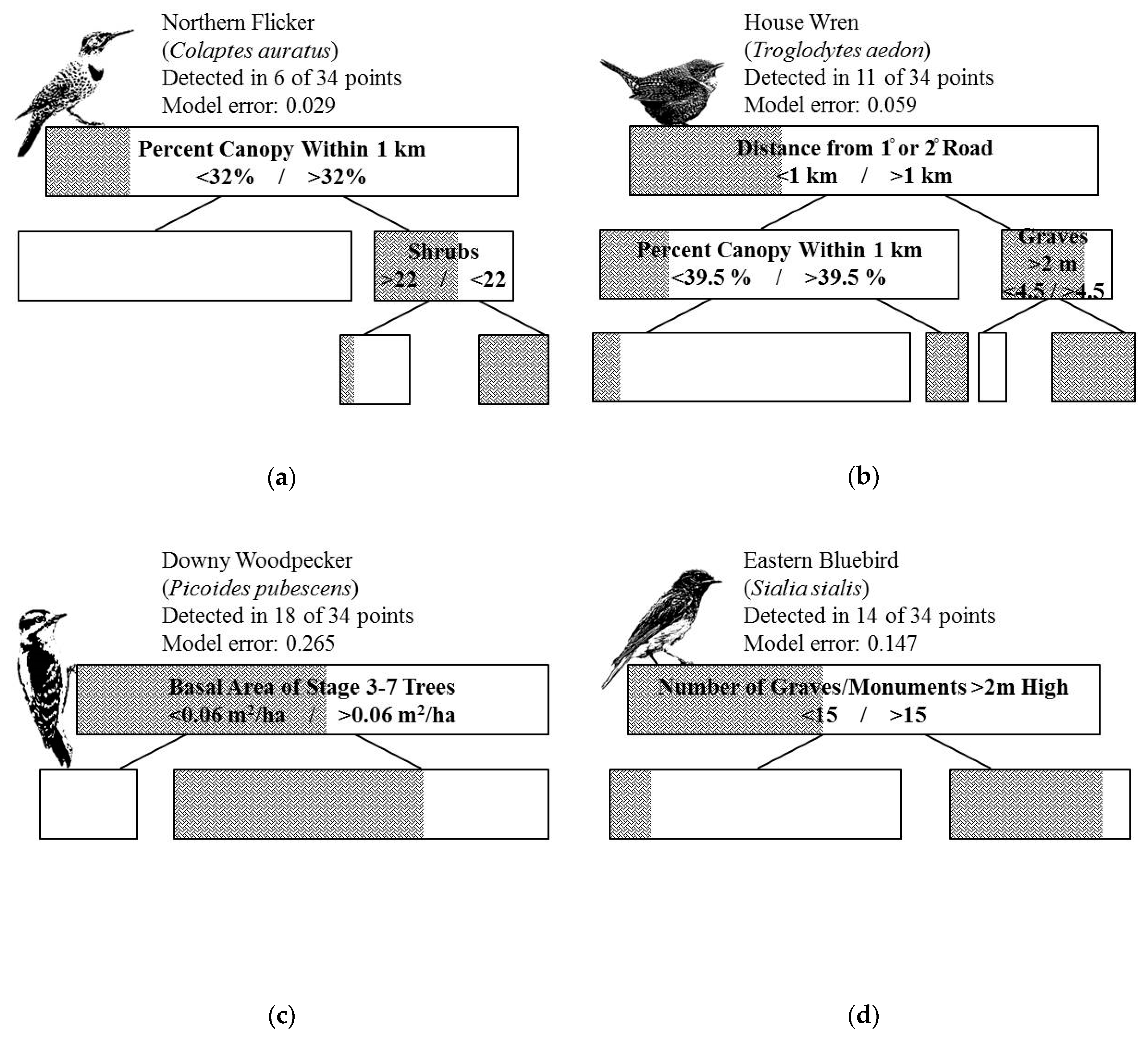
3.2. Habitat Selection Models
3.3. Richness of Cavity-Nesting Birds
4. Discussion
4.1. Site Selection for Establishment of New Cemeteries
4.2. Snag Provisioning
4.3. Headstones and Monuments
4.4. Landscaping Decisions
4.5. Cemeteries as Urban Habitat
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Species | Minimum Diameter (cm) | Maximum Diameter (cm) | Minimum Height (m) | Maximum Height (m) | Source(s) |
|---|---|---|---|---|---|
| American Kestrel (Falco sparverius) | 5.08 | 10.16 | 3.05 | 9.14 | [34,35] |
| Barn Owl (Tyto alba) | 9.53 | 11.43 | 2.44 | 7.62 | [35] |
| Barred Owl (Strix varia) | 15.24 | 30.48 | 3.66 | 9.14 | [34,35] |
| Black-capped Chickadee (Poecile atricapillus) | 2.54 | 3.54 | 1.22 | 4.57 | [34,35] |
| Downy Woodpecker (Picoides pubescens) | 3.05 | 3.56 | 1.52 | 15.24 | [2,34] |
| Eastern Bluebird (Sialia sialis) | 3.81 | 7.62 | 1.22 | 1.83 | [34,35] |
| Eastern Screech Owl (Megascops asio) | 7.62 | 13.97 | 3.05 | 9.14 | [34,35] |
| Great Crested Flycatcher (Myiarchus crinitus) | 4.45 | 5.45 | 0.91 | 6.10 | [35] |
| Hairy Woodpecker (Picoides villosus) | 3.81 | 4.57 | 3.05 | 13.72 | [2,34] |
| House Wren (Troglodytes aedon) | 2.54 | 6.08 | 1.22 | 9.14 | [34,35] |
| Northern Flicker (Colaptes auratus) | 5.08 | 6.99 | 1.83 | 9.14 | [2,34,35] |
| Northern Saw-whet owl (Aegolius acadicus) | 7.62 | 8.62 | 3.66 | 4.57 | [35] |
| Red-bellied Woodpecker (Melanerpes carolinus) | 5.10 | 6.40 | 2.00 | 18.00 | [33] |
| Red-headed Woodpecker (Melanerpes erythrocephalus) | 4.57 | 5.57 | 7.32 | 19.81 | [2] |
| Tufted Titmouse (Baelophus bicolor) | 3.18 | 4.45 | 0.91 | 27.43 | [34,35] |
| White-breasted Nuthatch (Sitta carolinensis) | 3.18 | 11.43 | 1.52 | 15.24 | [34,35] |
Appendix B
| Tree Taxon | Number of Points Where Encountered | Number of Trees | Total Basal Area (m2) |
|---|---|---|---|
| Acer platanoides* | 17 | 52 | 15.60 |
| Acer rubrum | 4 | 4 | 0.35 |
| Acer saccharinum | 23 | 65 | 35.29 |
| Acer saccharum | 11 | 19 | 4.47 |
| Aesculus glabra | 4 | 11 | 4.37 |
| Aesculus hippocastanum | 2 | 4 | 1.57 |
| Ailanthus altissima* | 4 | 9 | 5.52 |
| Betula alleghaniensis | 1 | 1 | 0.13 |
| Betula nigra | 1 | 1 | 0.11 |
| Betula papyrifera | 1 | 1 | 0.22 |
| Catalpa speciosa | 6 | 40 | 14.21 |
| Celtis occidentalis | 11 | 15 | 6.49 |
| Crataegus crus-galli | 2 | 5 | 0.28 |
| Fagus sylvatica | 1 | 1 | 0.01 |
| Fraxinus americana | 9 | 18 | 4.51 |
| Fraxinus pennsylvanica | 1 | 2 | 0.06 |
| Ginkgo biloba | 1 | 1 | 0.17 |
| Gleditsia triacanthos | 14 | 28 | 11.68 |
| Gymnocladus dioicus | 1 | 1 | 0.35 |
| Juglans nigra | 1 | 1 | 0.10 |
| Juniperus virginiana | 12 | 28 | 2.85 |
| Liliodendron tulipifera | 1 | 2 | 2.13 |
| Liquidambar styraciflua | 1 | 1 | 0.23 |
| Maclura pomifera* | 1 | 1 | 0.30 |
| Magnolia liliiflora | 2 | 2 | 0.24 |
| Malus spp. | 1 | 1 | 0.16 |
| Metasequoia glyptostroboides | 1 | 1 | 0.24 |
| Morus alba* | 10 | 26 | 7.98 |
| Picea abies | 20 | 77 | 10.00 |
| Pinus resinosa | 4 | 10 | 2.49 |
| Pinus sylvestris | 1 | 1 | 0.24 |
| Planera aquatica | 1 | 1 | 0.16 |
| Platanus occidentalis | 7 | 8 | 4.09 |
| Populus deltoides | 3 | 5 | 4.21 |
| Prunus spp. | 4 | 5 | 1.46 |
| Quercus spp. | 13 | 88 | 40.49 |
| Robinia pseudoacacia* | 2 | 26 | 10.15 |
| Thuja occidentalis | 3 | 15 | 2.06 |
| Tilia americana | 13 | 28 | 11.78 |
| Tilia cordata | 1 | 1 | 0.52 |
| Ulmus americana | 4 | 5 | 3.73 |
| Ulmus pumila* | 5 | 6 | 2.67 |
| Ulmus rubra | 5 | 5 | 1.93 |
| Ulmus thomasii | 1 | 1 | 0.53 |
References
- Van der Hoek, Y.; Gaona, G.V.; Martin, K. The diversity, distribution and conservation status of the tree-cavity-nesting birds of the world. Divers. Distrib. 2017, 23, 1120–1131. [Google Scholar] [CrossRef]
- Scott, V.E.; Evans, K.E.; Patton, D.R.; Stone, C.P. Cavity-Nesting Birds of North American Forests; Agricultural Handbook; U.S. Department of Agriculture: Washington, DC, USA, 1977; Volume 511, pp. 1–97.
- LaMontagne, J.M.; Kilgour, R.J.; Anderson, E.C.; Magle, S. Tree cavity availability across forest, park, and residential habitats in a highly urban area. Urban Ecosyst. 2015, 18, 151–167. [Google Scholar] [CrossRef]
- Blewett, C.M.; Marzluff, J.M. Effects of urban sprawl on snags and the abundance and productivity of cavity-nesting birds. Condor 2005, 107, 678–693. [Google Scholar] [CrossRef]
- Le Roux, D.S.; Ikin, K.; Lindenmayer, D.B.; Manning, A.D.; Gibbons, P. The future of large old trees in urban landscapes. PLoS ONE 2014, 9, e99403. [Google Scholar] [CrossRef] [PubMed]
- Beissinger, S.R.; Osborne, D.R. Effects of urbanization on avian community organization. Condor 1982, 84, 75–83. [Google Scholar] [CrossRef]
- Strausberger, B.M.; Ashley, M.V. Community-wide patterns of parasitism of a host “generalist” brood-parasitic cowbird. Oecologia 1997, 112, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, J.J.; Martel, M.; Markland, H.M.; Niklison, A.M.; Decker, K.L.; Martin, T.E. Testing ecological and behavioral correlates of nest predation. Oikos 2007, 116, 1887–1894. [Google Scholar] [CrossRef]
- Crooks, K.R.; Soulé, M.E. Mesopredator release and avifaunal extinctions in a fragmented system. Nature 1999, 400, 563–566. [Google Scholar] [CrossRef]
- Croci, S.; Butet, A.; Clergeau, P. Does urbanization filter birds on the basis of their biological traits? Condor 2008, 110, 223–240. [Google Scholar] [CrossRef]
- Lancaster, R.K.; Rees, W.E. Bird communities and the structure of urban habitats. Can. J. Zool. 1979, 57, 2358–2368. [Google Scholar] [CrossRef]
- Lim, H.C.; Sodhi, N.S. Responses of avian guilds to urbanisation in a tropical city. Landsc. Urban Plan. 2004, 66, 199–215. [Google Scholar] [CrossRef]
- Lussenhop, J. Urban cemeteries as bird refuges. Condor 1977, 79, 456–461. [Google Scholar] [CrossRef]
- Bovyn, R.A.; Lordon, M.C.; Grecco, A.E.; Leeper, A.C.; LaMontagne, J.M. Tree cavity availability in urban cemeteries and city parks. J. Urban Ecol. 2019, 5, juy030. [Google Scholar] [CrossRef]
- James, F.C.; Wamer, N.O. Relationships between temperate forest bird communities and vegetation structure. Ecology 1982, 63, 159–171. [Google Scholar] [CrossRef]
- Wiens, J.A. Habitat fragmentation: Island v landscape perspectives on bird conservation. Ibis 2008, 137, S97–S104. [Google Scholar] [CrossRef]
- Warren, T.L.; Betts, M.G.; Diamond, A.W.; Forbes, G.J. The influence of local habitat and landscape composition on cavity-nesting birds in a forested mosaic. Ecol. Manag. 2005, 214, 331–343. [Google Scholar] [CrossRef]
- Brown, J.S.; Kotler, B.P. Hazardous duty pay and the foraging cost of predation: Foraging cost of predation. Ecol. Lett. 2004, 7, 999–1014. [Google Scholar] [CrossRef]
- Burghardt, K.T.; Tallamy, D.W. Plant origin asymmetrically impacts feeding guilds and life stages driving community structure of herbivorous arthropods. Divers. Distrib. 2013, 19, 1553–1565. [Google Scholar] [CrossRef]
- Greenberg, J. A Natural History of the Chicago Region; University of Chicago Press: Chicago, IL, USA, 2002; Volume 219, pp. 1–15. [Google Scholar]
- United States Census Bureau. Annual Estimates of the Resident Population for Incorporated Places of 50,000 or More, Ranked by July 1, 2017 Population: April 1, 2010 to July 1, 2017; United States Census Bureau: Hutland Sutherland, MD, USA. Available online: https://factfinder.census.gov/faces/tableservices/jsf/pages/productview.xhtml?src=bkmk. (accessed on 12 November 2018).
- Pattison, W.D. The cemeteries of Chicago: A phase of land utilization. Ann. Assoc. Am. Geogr. 1955, 45, 245–257. [Google Scholar] [CrossRef]
- French, S. The cemetery as cultural institution: The establishment of Mount Auburn and the “rural cemetery” movement. Am. Q. 1974, 26, 37–59. [Google Scholar] [CrossRef]
- Ralph, C.J.; Droege, S.; Sauer, J.R. Managing and monitoring birds using point counts: Standards and applications. In Monitoring Bird Populations by Point Counts; USDA Forest Service General Technical Report; US Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 1995; pp. 161–168. [Google Scholar]
- Imbeau, L.; Desrochers, A. Foraging ecology and use of drumming trees by three-toed woodpeckers. J. Wildl. Manag. 2002, 66, 222–231. [Google Scholar] [CrossRef]
- ESRI. ArcGIS Desktop: 10.5; Environmental Systems Research Institute: Redlands, CA, USA, 2017. [Google Scholar]
- United States Census Bureau. TIGER/Line® Shapefiles; United States Census Bureau: Hutland Sutherland, MD, USA. Available online: https://www.census.gov/geo/maps-data/data/tiger-line.html (accessed on 15 October 2017).
- United States Geological Survey. USGS National Hydrography Dataset (NHD); United States Geological Survey: Reston, VA, USA. Available online: https://www.usgs.gov/core-science-systems/ngp/national-hydrography (accessed on 15 October 2017).
- O’Neil-Dunne, J. Cook County Land Cover. Available online: http://www.uvm.edu/~joneildu/downloads/CookCounty/ (accessed on 22 November 2017).
- Nur, N.; Jones, S.L.; Geupel, G.R. Statistical Guide to Data Analysis of Avian Monitoring Programs; U.S. Department of the Interior, Fish and Wildlife Service: Washington, DC, USA, 1999; pp. 1–46.
- Hollander, M.; Wolfe, D. Nonparametric Statistical Methods; Wiley: New York, NY, USA, 1973; pp. 68–75. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Jackson, J.A. A comparison of some aspects of the breeding ecology of red-headed and red-bellied woodpeckers in Kansas. Condor 1976, 78, 67–76. [Google Scholar] [CrossRef]
- Stokes, D.; Stokes, L. Stokes Nature Guides: A Guide to Bird Behavior; Little, Brown and Company: Boston, MA, USA, 1979; Volume I–III. [Google Scholar]
- Cornell Lab of Ornithology. All about Birdhouses; Cornell Lab of Ornithology: Ithaca, NY, USA; Available online: https://nestwatch.org/learn/all-about-birdhouses/ (accessed on 22 March 2019).
- De’ath, G.; Fabricius, K.A. Classification and regression trees: A powerful yet simple technique for ecological data analysis. Ecology 2000, 81, 3178–3192. [Google Scholar] [CrossRef]
- Therneau, T.; Atkinson, B. Rpart: Recursive Partitioning and Regression Trees, R Package Version 4.1-13. 2018.
- Anderson, D.R. Model Based Inference in the Life Sciences: A Primer on Evidence; Springer Science and Business Media: New York, NY, USA, 2008; pp. 1–184. [Google Scholar]
- Mazerolle, M. AICcmodavg: Model Selection and Multimodel Inference Based on (Q)AIC(c); R Package Version 2.1-1. 2017; Available online: https://www.fwspubs.org/doi/suppl/10.3996/102016-JFWM-079/suppl_file/10.3996102016-jfwm-079.s6.pdf (accessed on 10 June 2019).
- Paradis, E.; Schliep, K. ape 5.0: An environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 2018, 35, 526–528. [Google Scholar] [CrossRef] [PubMed]
- Ahlmann-Eltze, C. Ggsignif: Significance Brackets for “ggplot2”; R Package Version 0.4.0. 2017; Available online: https://cran.r-project.org/web/packages/ggsignif/ggsignif.pdf (accessed on 10 June 2019).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Green Burial Council. Our Standards. Available online: https://www.greenburialcouncil.org/our_standards.html (accessed on 22 March 2019).
- Basmajian, C.; Coutts, C. Planning for the disposal of the dead. J. Am. Plan. Assoc. 2010, 76, 305–317. [Google Scholar] [CrossRef]
- Lomolino, M.V. Ecology’s most general, yet protean pattern: The species-area relationship. J. Biogeogr. 2000, 27, 17–26. [Google Scholar] [CrossRef]
- Jackson, J.A.; Ouellet, H.R. Downy woodpecker (Dryobates pubescens). Birds N. Am. Online 2018. [Google Scholar] [CrossRef]
- Cockle, K.L.; Martin, K.; Wesołowski, T. Woodpeckers, decay, and the future of cavity-nesting vertebrate communities worldwide. Front. Ecol. Environ 2011, 9, 377–382. [Google Scholar] [CrossRef]
- Bull, E.L.; Partridge, A.D. Methods of killing trees for use by cavity nesters. Wildl. Soc. Bull. 1986, 14, 142–146. [Google Scholar]
- Swearingen, J.; Bargeron, C. Trees: Invasive Plant Atlas of the United States. Available online: https://www.invasiveplantatlas.org/trees.cfm (accessed on 22 March 2019).
- Martin, P.H.; Marks, P.L. Intact forests provide only weak resistance to a shade-tolerant invasive Norway maple (Acer platanoides L.). J. Ecol. 2006, 94, 1070–1079. [Google Scholar] [CrossRef]
- Yosef, R.; Grubb, T.C. Resource dependence and territory size in loggerhead shrikes (Lanius ludovicianus). Auk 1994, 111, 465–469. [Google Scholar] [CrossRef]
- Wesołowski, T. “Lifespan” of woodpecker-made holes in a primeval temperate forest: A thirty year study. Ecol. Manag. 2011, 262, 1846–1852. [Google Scholar] [CrossRef]
- Frei, B.; Smith, K.G.; Withgott, J.H.; Rodewald, P.G.; Pyle, P.; Patten, M.A. Red-headed woodpecker (Melanerpes erythrocephalus). Birds N. Am. 2000, 518, 28. [Google Scholar] [CrossRef]
- King, R.S.; Brashear, K.E.; Reiman, M. Red-headed woodpecker nest-habitat thresholds in restored savannas. J. Wildl. Manag. 2007, 71, 30–35. [Google Scholar] [CrossRef]
- Wiebe, K.L.; Moore, W.S. Northern flicker (Colaptes auratus). Birds N. Am. 2008, 166a, 167–173. [Google Scholar] [CrossRef]
- Smith, R.J.; Hatch, M.I.; Cimprich, D.A.; Moore, F.R. Gray catbird (Dumetella carolinensis). Birds N. Am. 1995. [Google Scholar]
- Vezzani, D.; Velázquez, S.M.; Soto, S.; Schweigmann, N.J. Environmental characteristics of the cemeteries of Buenos Aires City (Argentina) and infestation levels of Aedes aegypti (Diptera: Culicidae). Mem. Inst. Oswaldo Cruz 2001, 96, 467–471. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Variable | Minimum | Median | Maximum | Mean ± SD |
|---|---|---|---|---|
| Tree density (trees/ha) | 6.4 | 25.5 | 42.0 | 23.3 ± 9.0 |
| Snag basal area per hectare (m2/ha) | 0 | 0.6 | 2.6 | 0.8 ± 0.8 |
| Cemetery area (ha) | 0.9 | 35.7 | 141 | 48.9 ± 44.0 |
| Number of shrubs | 0 | 15.5 | 39 | 15.6 ± 11.9 |
| Number of headstones taller than 2 m | 0 | 11 | 77 | 17.1 ± 19.6 |
| Distance from a 1° or 2° road (km) | 0.1 | 0.7 | 3.4 | 0.9 ± 0.9 |
| Distance from a body of water (km) | 0.1 | 0.4 | 2.1 | 0.6 ± 0.5 |
| Percent canopy cover within 1 km (%) | 18 | 29 | 51 | 29.1 ± 8.0 |
| Common Name | Scientific Name | Number of Points |
|---|---|---|
| Black-capped Chickadee | Poecile atricapillus | 30 |
| White-breasted Nuthatch | Sitta carolinensis | 21 |
| Downy Woodpecker | Picoides pubescens | 18 |
| Eastern Bluebird | Sialia sialis | 14 |
| Red-bellied Woodpecker | Melanerpes carolinus | 12 |
| House Wren | Troglodytes aedon | 11 |
| Northern Flicker | Colaptes auratus | 6 |
| Great Crested Flycatcher | Myiarchus crinitus | 6 |
| Hairy Woodpecker | Picoides villosus | 6 |
| American Kestrel | Falco sparverius | 4 |
| Tufted Titmouse | Baeolophus bicolor | 1 |
| Turkey Vulture | Cathartes aura | 1 |
| Model | K | AICc | ΔAICc | AICc Weight | LL | Adjusted r2 | Coefficient of Variable in Single Variable Model | Coefficient of Variable in Combined Model |
|---|---|---|---|---|---|---|---|---|
| Combined model | 6 | 119.12 | 0.00 | 0.88 | −52.00 | 0.39 | N/A | N/A |
| Canopy within 1 km | 3 | 124.55 | 5.43 | 0.06 | −58.88 | 0.17 | 0.09 ± 0.03 | 0.04 ± 0.03 |
| Distance to a body of water | 3 | 125.27 | 6.15 | 0.04 | −59.23 | 0.15 | −1.24 ± 0.47 | −0.97 ± 0.41 |
| Tree density | 3 | 127.58 | 8.47 | 0.01 | −60.39 | 0.09 | 0.06 ± 0.03 | 0.06 ± 0.03 |
| Distance to a 1° or 2° road | 3 | 128.39 | 9.27 | 0.01 | −60.79 | 0.07 | 0.54 ± 0.29 | 0.60 ± 0.24 |
| Null | 2 | 129.51 | 10.40 | 0.00 | −62.56 | 0.00 | 3.82 ± 0.27 | N/A |
| Snag basal area | 3 | 131.33 | 12.22 | 0.00 | −62.27 | −0.01 | 0.26 ± 0.34 | N/A |
| Number of shrubs | 3 | 131.61 | 12.49 | 0.00 | −62.41 | −0.02 | −0.01 ± 0.02 | N/A |
| Cemetery area | 3 | 131.66 | 12.54 | 0.00 | −62.43 | −0.02 | 0.00 ± 0.01 | N/A |
| Number of headstones >2m | 3 | 131.88 | 12.77 | 0.00 | −62.54 | −0.03 | 0.00 ± 0.01 | N/A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, A.D.; Minor, E. Chicago’s Urban Cemeteries as Habitat for Cavity-Nesting Birds. Sustainability 2019, 11, 3258. https://doi.org/10.3390/su11123258
Smith AD, Minor E. Chicago’s Urban Cemeteries as Habitat for Cavity-Nesting Birds. Sustainability. 2019; 11(12):3258. https://doi.org/10.3390/su11123258
Chicago/Turabian StyleSmith, Alexis D., and Emily Minor. 2019. "Chicago’s Urban Cemeteries as Habitat for Cavity-Nesting Birds" Sustainability 11, no. 12: 3258. https://doi.org/10.3390/su11123258
APA StyleSmith, A. D., & Minor, E. (2019). Chicago’s Urban Cemeteries as Habitat for Cavity-Nesting Birds. Sustainability, 11(12), 3258. https://doi.org/10.3390/su11123258





